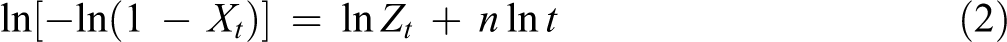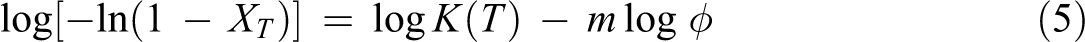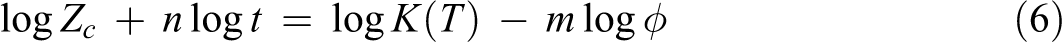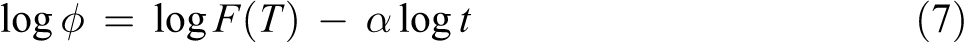Abstract
The aim of this study is to investigate the kinetics of non-isothermal crystallization of polypropylene/high-density polyethylene/halloysite nanotube (PP/HDPE/HNT) nanocomposites using three methods, that is, Avrami equation, combined Ozawa–Avrami method (hereafter called Mo model), and Kissinger equation. The Avrami exponent (n) is in the range of 1–2 for all the PP/HDPE/HNT nanocomposites indicating instantaneous nucleation while the crystallization rate constant (Zt ) values of PP/HDPE increased with the addition of HNT. This proved that addition of HNT increases the crystallization rate. The reduction of half crystallization time (t 1/2) for PP/HDPE as the increasing HNT loading indicates faster crystallization rate. In the Mo model, the cooling rate chosen at unit crystallization time F(T) values for PP/HDPE decreases with the addition of HNT. Kissinger equation showed that the activation energy (E a) of crystallization for the PP/HDPE decreases with the addition of HNT. All the results demonstrated that HNT can accelerate the crystallization rate for the PP/polyethylene blends.
Introduction
Polymer blends have been intensively studied due to their industrial importance. Polypropylene (PP) and polyethylene (PE) are versatile commercial thermoplastics with low density, easy processing, and low cost. PE offers good impact strength while PP could improve environmental stress cracking resistance for PE/PP blends. 1 The properties of the blends that have two crystallizable components depend strongly on their crystallinity, crystalline morphology, and degree of dispersion. 2 Also, adding filler into a polymer blend could further influence their crystallization behavior. The incorporation of organoclay into PP/PE blend significantly enhance their properties and contribute to a more complex phase morphology as well as crystallization kinetics for both of the crystalline components. 3 A beneficial result from the incorporation of effective nucleating agent is reduced cycle time in polymer processing (e.g. extrusion and molding). 4
Halloysite nanotube (HNT) is widely used to reinforce polymer nanocomposites due to their high aspect ratio, high dispersibility, low external hydroxyl groups, biocompatibility, and high temperature resistance. 5 –8 The incorporation of HNT has proved to enhance the mechanical strength, modulus, stiffness, and impact resistance of polymers. 9 In terms of crystallization behavior, HNT can affect the crystallization behavior of polymers by acting as heterogeneous nucleation points. According to Ning et al., 10 HNT can act as a nucleation agent, resulting in an enhancement of the overall crystallization rate and the non-isothermal crystallization temperature of PP. Another study done by Du et al. 11 reported that the PP nanocomposites show faster crystallization process due to the nucleating effect of HNT. Liu et al. 12 reported that HNT has dual nucleating ability for α-isotactic PP and β-isotactic PP under non-isothermal and isothermal crystallization.
High-density polyethylene (HDPE) is chosen to blend with PP attributed to its ease of processing and good impact resistance. However, the crystallization of PP often influenced by the addition of HDPE, due to the fact that nucleation in HDPE is more difficult to be controlled than in PP. Further, incorporation of HNT into PP/HDPE blends is worth to be studied, especially the effects of HNT on the kinetics of crystallization for the PP/HDPE blends. The production of thermoplastic composites in actual industrial processes usually occurs under non-isothermal conditions including injection and extrusion molding. Thus, the investigation of the crystallization behavior can enable optimization of processing technology of PP blend nanocomposites. 13 The analysis of crystallization kinetics can provide knowledge to control processing parameters such as cycle time, blend ratios, and processing temperature in injection molding. 14
The Avrami equation is widely used to quantify crystallization kinetics and give an indication of the nature of the crystallization process, including nucleation and growth. 15 –17 Xu et al. 18 and Mosavian et al. 19 used Avrami equation to study the non-isothermal crystallization of PP nanocomposites and HDPE nanocomposites, respectively. Xu et al. 18 reported that the difference of Avrami exponent between PP and PP/montmorillonite nanocomposite was approximately 1 at the same cooling rate indicating that the montmorillonite nanoclay can act as nucleating agent in the PP matrix. According to Mosavian et al., 19 the Avrami exponent increased with an increase in cooling rate and alumina content in HDPE matrix.
The other reliable approach adopted for describing non-isothermal crystallization is the Mo model. 20 By combining the Avrami and Ozawa equations, Mo 21 proposed a new method for non-isothermal crystallization, which is considered as having more applicability than the Ozawa method. This method is useful to dealing with the kinetics of non-isothermal crystallization of various polymer systems such as polyamides, poly(ethylene oxide), polyolefines, poly (3-alkylthiophenes), poly(beta-hydroxybutyrate), and poly(aryl ether ketone). 22 The Mo model has been used to study the non-isothermal crystallization of PP nanocomposites and HDPE nanocomposites. 23,24 According to Yuan et al., 23 the value of F(T) (note: F(T) refers to the value of the cooling rate chosen at unit crystallization time, when the system has a defined degree of crystallinity) decreases with increasing the content of clay, indicating that the nanocomposites can achieve the same degree of crystallinity faster than neat PP. This implies faster kinetics of crystallization which proved that nanocomposites can act as a nucleating agent to accelerate the crystallization of PP. In the research done by Chen et al., 24 the F(T) values of the HDPE/barium sulfate (BaSO4) nanocomposites are lower than that of neat HDPE; and they demonstrated that the Mo method is successful in describing the non-isothermal process of the neat HDPE and the HDPE/BaSO4 nanocomposites.
The activation energy for the transport of the polymer segments to the growing crystal surface can be determined using the Kissinger approach. Kissinger equation has been used to study the activation energy, E a, of PP nanocomposites and HDPE nanocomposites. 18,25 The research carried out by Xu et al. 18 showed that the E a of PP/montmorillonite is lower than that of PP which indicated that the nanoclay facilitates the acceleration of the overall non-isothermal crystallization process of the PP. According to Kim et al., 25 the lower activation energy of the HDPE nanocomposites implies faster crystallization due to the large density of nuclei in the sample.
In this study, Avrami equation, Mo model, and Kissinger equation are used to study the non-isothermal crystallization of PP/HDPE/HNT nanocomposites. These data are useful to demonstrate the potential of HNT as nanofiller in controlling the crystallization behavior of the PP/HDPE blends.
Materials and methods
PP (TITANPRO PM–255) was purchased from Titan PP Polymers (M) Sdn. Bhd, Malaysia. The HDPE (TITANEX HF0961) was obtained from Lotte Chemical Titan Titanex, Malaysia. HNT (Aldrich 685445) was supplied by Sigma-Aldrich, Malaysia.
Sample preparation
PP/HDPE (blend ratio = 80/20) was prepared by mixing the PP, HDPE, and HNT using an internal mixer (Haake PolyDrive R600, Germany) at 160–166°C with a rotor speed of 50 r min−1. The HNT loading was fixed at 2, 5, and 8 phr. All the materials were dehumidified in a vacuum oven at 100°C for 24 h before compounding. The samples were compression molded with a hot press machine (Go Tech, Taiwan) at 180°C into sheets of 1 mm thickness. The preheating, compression molding, and cooling times was set at 6, 2, and 3 min, respectively.
Kinetics of crystallization
The thermal properties of PP/HDPE/HNT nanocomposites were characterized using differential scanning calorimeter (DSC) (Mettler Toledo, DSC 200, Columbus, Ohio, USA) under nitrogen atmosphere. Using DSC, the PP/HDPE/HNT nanocomposites were scanned from 30°C to 200°C at a heating rate of 10°C min−1 and a different cooling rate of 2, 5, 10, and 30°C min−1.
Avrami model
The Avrami equation used to describe the non-isothermal crystallization kinetics can be expressed in the following equations 26
where Xt is the relative crystallinity at crystallization time t, n is the Avrami exponent associated with the crystallization mechanism, and Zt is the crystallization rate constant related to both nucleation and growth rate parameters. The values of n and Zt can be obtained from the slope and intercept of the plots of ln [−ln(1 − Xt )] versus ln t, respectively.
The non-isothermal crystallization rate can be characterized by means of halftime of crystallization (t 1/2), it represents the time for the system to reach 50% of relative degree of crystallinity, and it is used to evaluate the rate of crystallization of different systems. The t 1/2 can be calculated using the following equation
Mo model
Ozawa extended Avrami equation by considering the influence of the cooling rate, and the relative degree of crystallization at temperature T, XT , can be calculated using the following equation 27
where K(T) is the cooling function which relates to the overall crystallization rate and indicates how fast the crystallization proceeds, XT is the relative crystallinity as a function of temperature, m is the Ozawa exponent depending on the dimension of crystal growth, n is the Avrami exponent, and ϕ is the cooling rate. In order to describe the non-isothermal crystallization process exactly, a new kinetics model is proposed by combining the models of Avrami and Ozawa
where F(T) = [K(T)/Zc ]1/m is the cooling rate chosen at unit crystallization time. When log ϕ is plotted against log t, the intercept is log F(T), and the slope is −α. A higher F(T) value means that a higher cooling rate is needed to reach a certain XT in a unit of crystallization time, indicating a slow crystallization rate. α = n/m is the ratio of Avrami exponent to Ozawa exponent, n is Avrami exponent, m is Ozawa exponent, and Zc is the modified crystallization rate constant.
Kissinger equation
The Kissinger equation provides a simple and direct model-free way to estimate the activation energy, E a, for the non-isothermal cold crystallization of polymers. 28 The crystallization activation energy during non-isothermal processes for polymer systems can be evaluated using the Kissinger equation
where E
a is the crystallization activation energy, ϕ is the cooling rate, T
p is the crystallization peak temperature, and R is the gas constant. The activation energy E
a can be obtained from the slope of the plot of ln (ϕ/
Results and discussions
Avrami model
The plots of relative crystallinity (Xt ) with t at different cooling rates are illustrated in Figure 1. A series of S-shaped curves is obtained because of the spherulitic impingement in the later crystallization stage. The values of t (the proceeding time of crystallization) at the various cooling rates can be obtained at a random relative crystallinity. It is clearly observed that the higher the cooling rate, the shorter the time of crystallization completion.
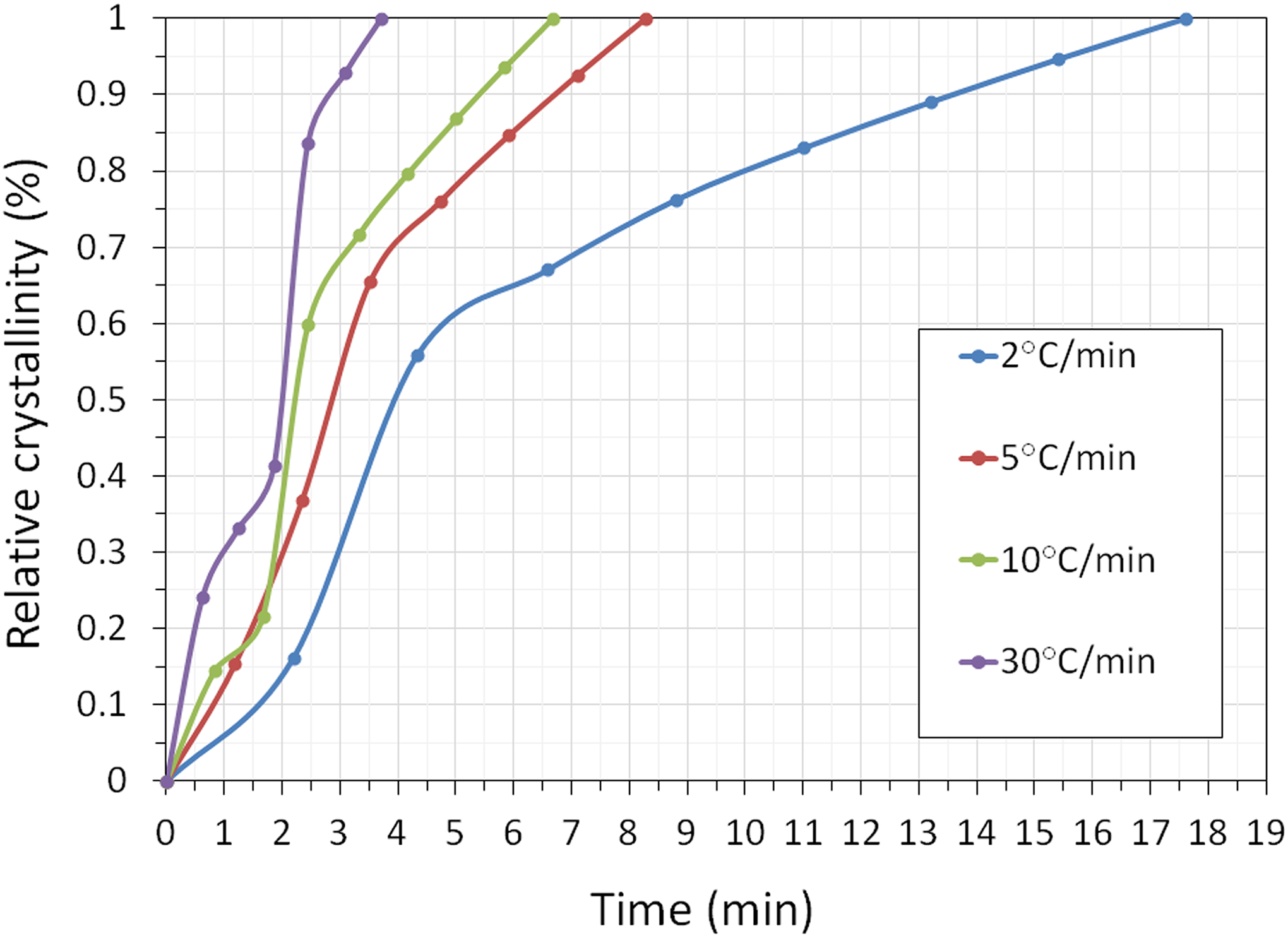
Relative crystallinity versus time at various cooling rates of PP/HDPE/HNT5 nanocomposites.
The crystallization halftime, t 1/2, is defined as the time at which the relative crystallization degree is completed 50%. The halftime t 1/2 presents the overall crystallization rate. The shorter the halftime, the faster is the overall crystallization rate. 29 From Table 1, it can be seen that the t 1/2 of PP/HDPE and PP/HDPE/HNT composites decreased gradually with increasing cooling rate, indicating a progressively faster crystallization rate as the cooling rate increased.
The half crystallization time (t 1/2) of PP/HDPE/HNT nanocomposites.
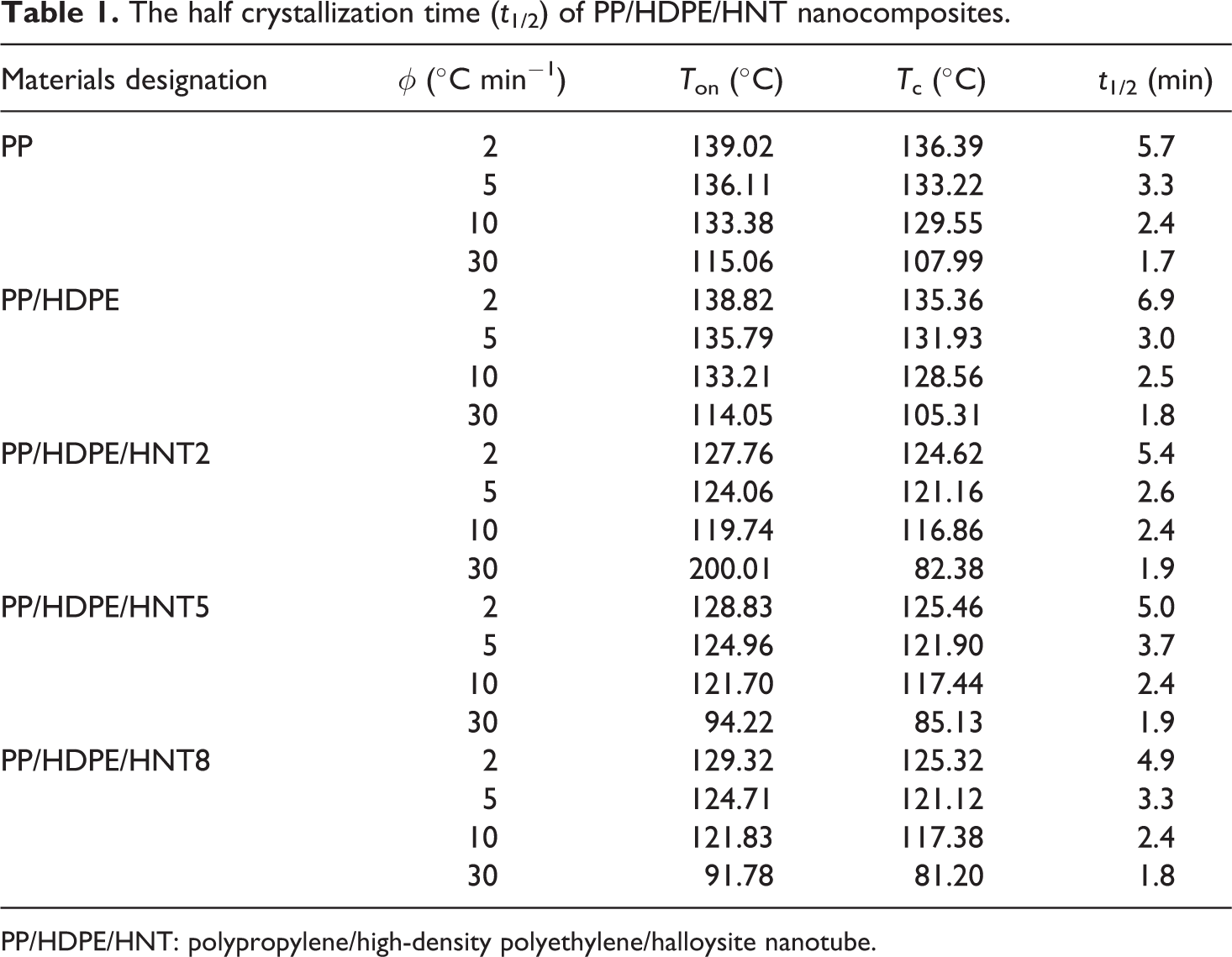
PP/HDPE/HNT: polypropylene/high-density polyethylene/halloysite nanotube.
Figure 2 shows the plots of ln (−ln(1 − Xt )) versus ln t of PP/HDPE/HNT5 nanocomposites. The Avrami exponent n and rate constant Zt can be determined from the slope and intercept of the linear segment. From the data presented in Table 2, it is evident that the Avrami exponent n values of PP/HDPE are the lowest at high cooling rates, which indicates a simpler nucleation mechanism and geometry as the cooling rate increases. The change in n could indicate differences in crystal growth geometry and the type of nucleation because it is a function of the number of dimensions in which growth takes place; it reflects the details of crystal nucleation and growth mechanisms. According to Meng et al., 15 n = 1 represents rod-like growth from instantaneous nuclei; n = 2 indicates high nucleation rate and plate-like growth, where growth is mainly along two dimensions; n = 3 represents spherulitic growth from instantaneous nuclei, whereas an n = 4 represents heterogeneous nucleation and spherulitic growth from sporadic nuclei. Since the n values in all of the PP/HDPE/HNT nanocomposites are in the range of 1–2, the growth mechanism is mainly instantaneous nucleation. In general, low values of n and high values of Zt indicate an increased rate of crystallization and a more instantaneous nucleation process.
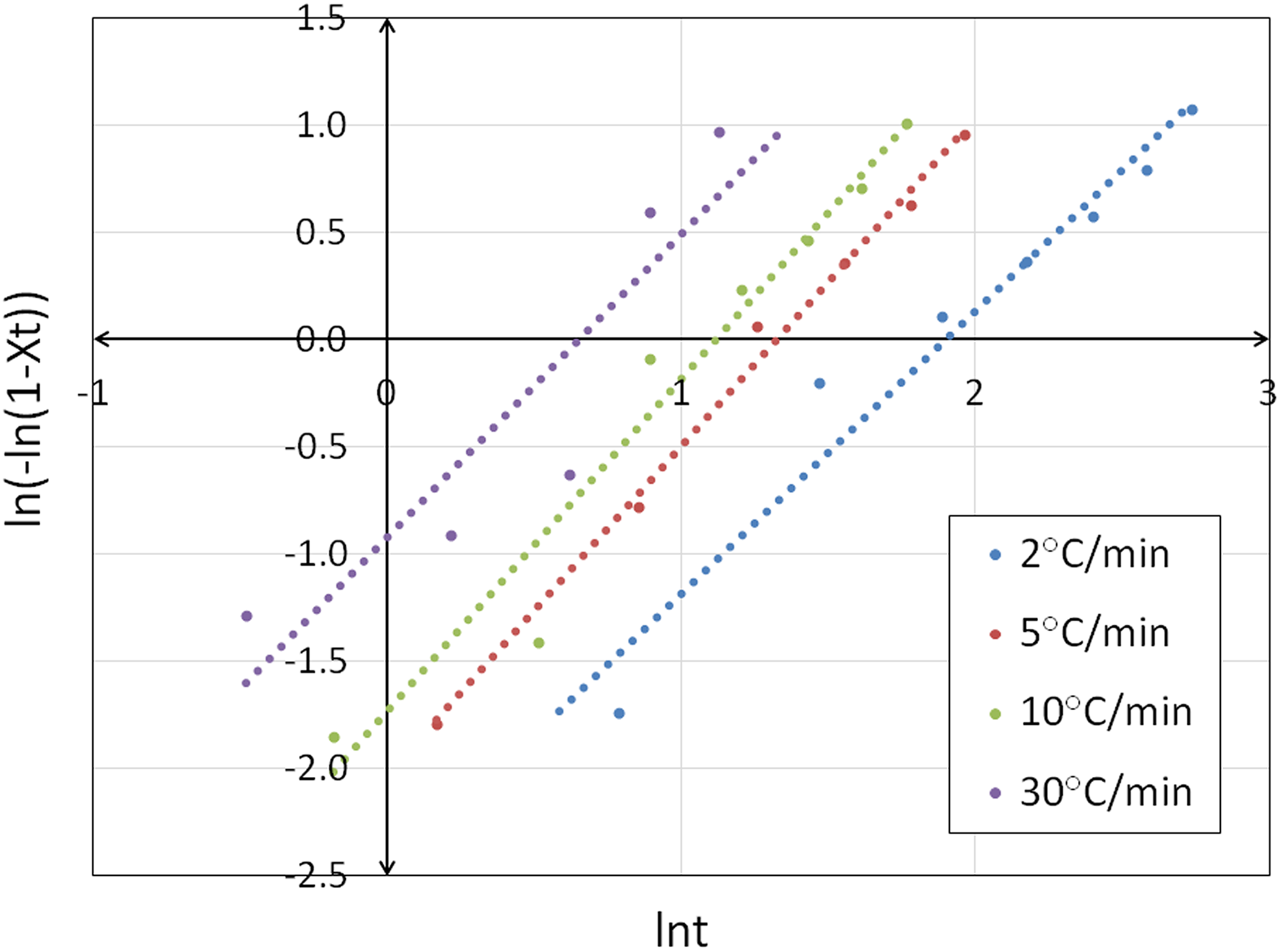
Plots of ln (−ln(1 − Xt )) versus ln t at various cooling rates of PP/HDPE/HNT5 nanocomposites.
Non-isothermal crystallization kinetics parameters of PP/HDPE/HNT nanocomposites determined from Avrami plot.
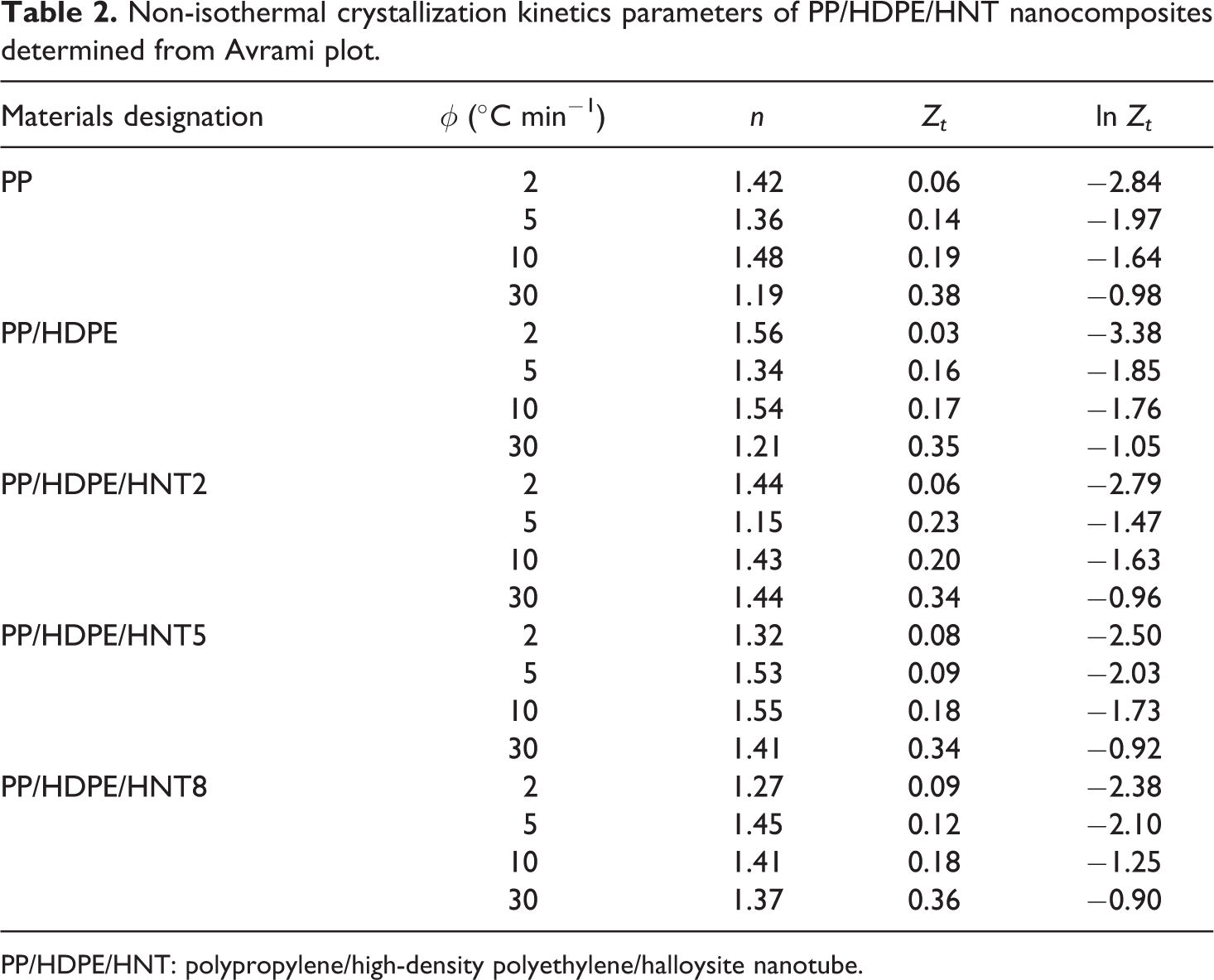
PP/HDPE/HNT: polypropylene/high-density polyethylene/halloysite nanotube.
In addition, the crystallization rate constant Zt values of all the PP/HDPE/HNT nanocomposites increase with increasing cooling rate. This is because at higher cooling rate, melt crystallization occurs at lower temperature, resulting in higher crystallization rate due to higher undercooling. 13 Furthermore, at a certain cooling rate, it can be clearly determined that the Zt values of PP/HDPE/HNT composites are higher than the PP/HDPE, indicating faster overall crystallization rate of the HNT-filled polymer blend composites.
Mo model
Figure 3 shows the plots of log ϕ versus log t of PP/HDPE/HNT5 nanocomposites. Table 3 summarizes the non-isothermal crystallization kinetics parameters of PP/HDPE/HNT nsnocomposites determined using the Mo model. From Figure 3, a good linear relationship is obtained. This indicates that the Mo method is a good approach in describing the non-isothermal crystallization kinetics of the PP/HDPE and its nanocomposites. The cooling rate chosen at unit crystallization time (F(T)) and the Mo exponent (α) related to a given relative crystallinity (Xt ) can be obtained from the intercept and slope of each straight line, respectively. 30 From Table 3, the values of F(T) and α increase gradually with the increase of relative crystallinity, indicating that the crystallization becomes more difficult as the crystallization progresses. 31 Overall, at the same Xt , the F(T) values of PP/HDPE blends decrease as the amount of HNT increases. Since F(T) reflects the difficulty of crystallization process, thus the lower the F(T) value, the faster the crystallization rate is. This indicates that HNT can act as nucleating agent for PP/PE blends.
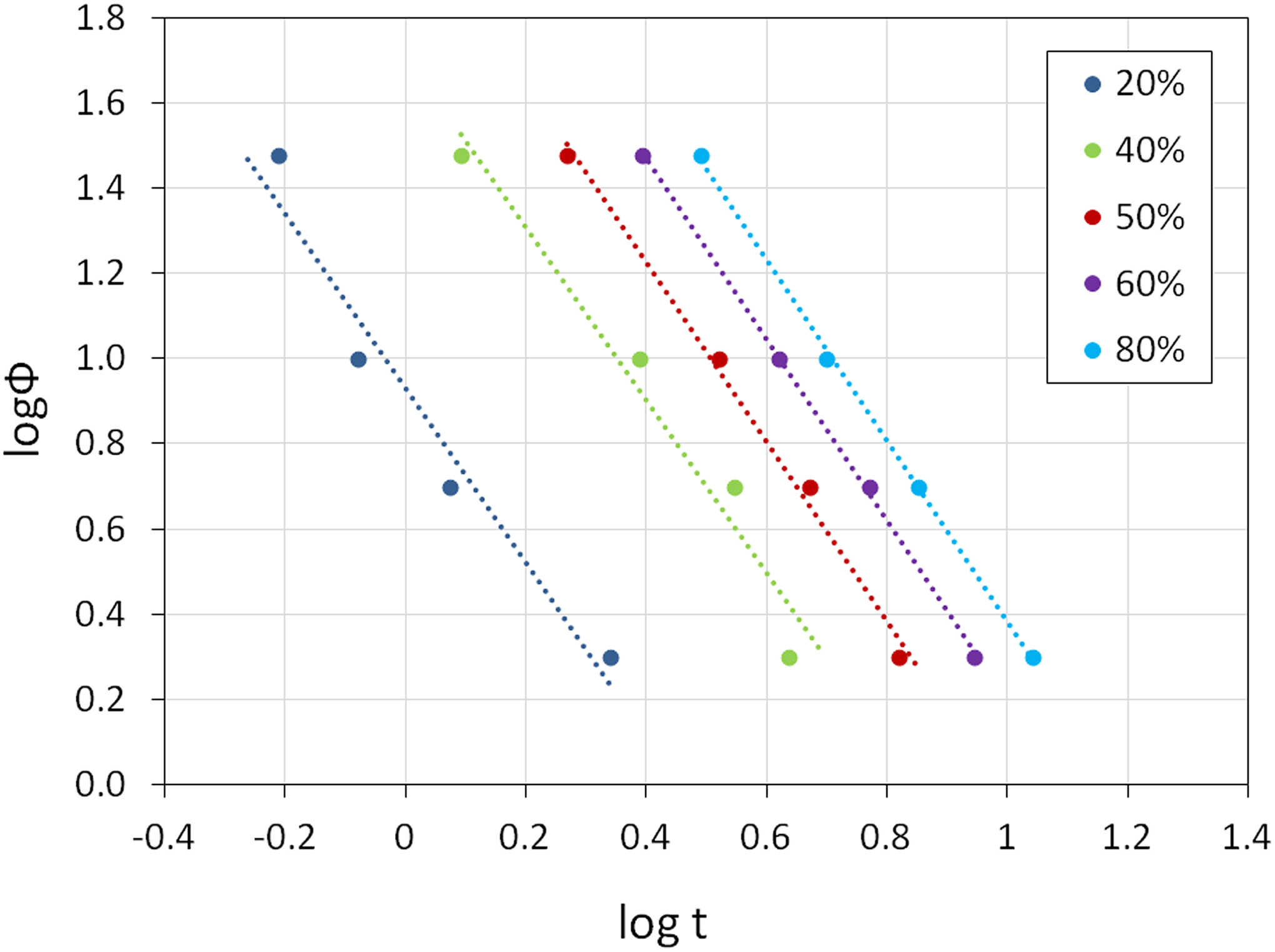
Plots of log ϕ versus log t of PP/HDPE/HNT5 nanocomposites.
Non-isothermal crystallization kinetics parameters of PP/HDPE/HNT nanocomposites determined using Mo model.
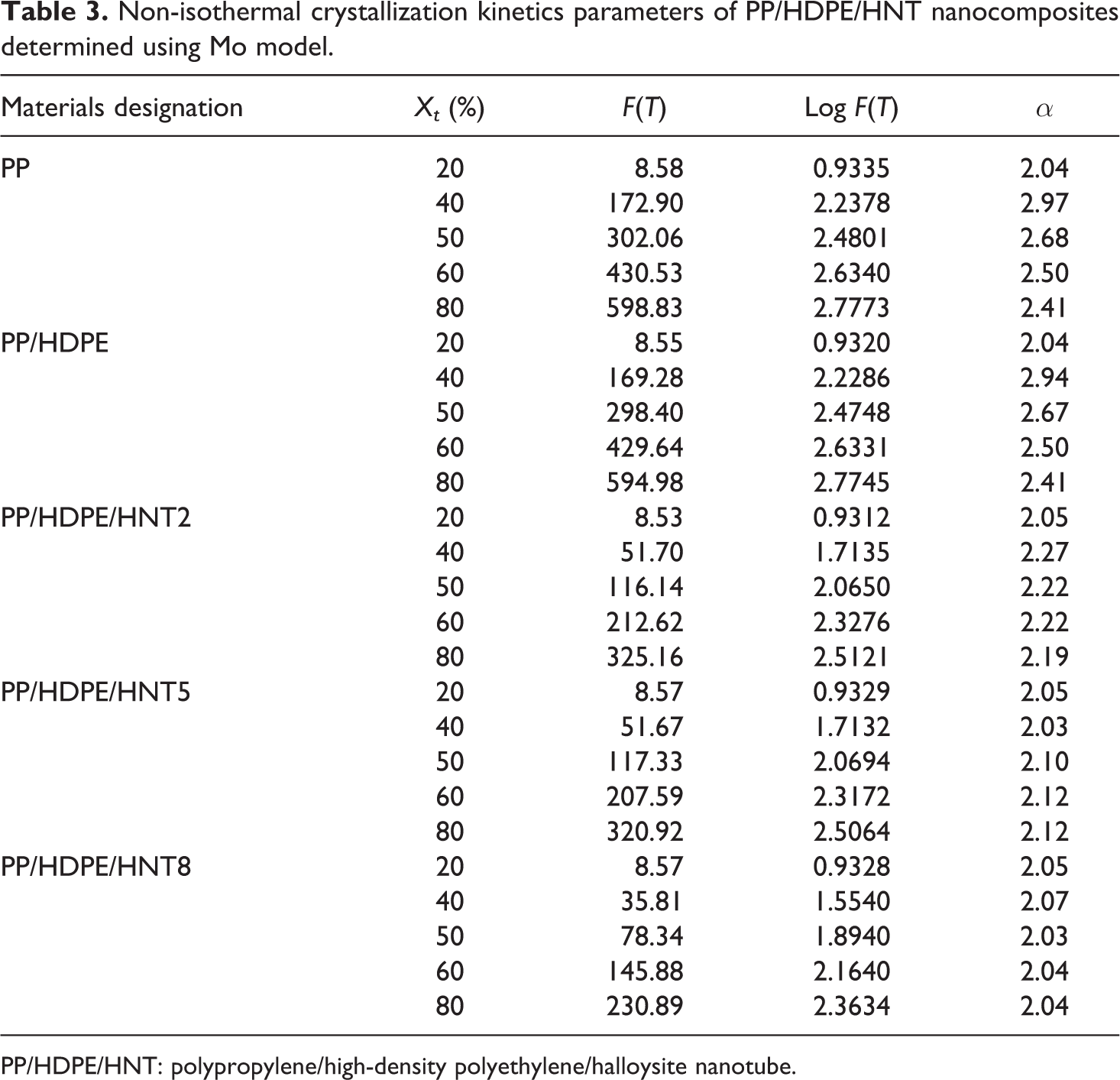
PP/HDPE/HNT: polypropylene/high-density polyethylene/halloysite nanotube.
Kissinger equation
For non-isothermal crystallization, the crystallization activation energy (E
a) can be estimated from the variation of crystallization peak temperature (T
c) with cooling rates (ϕ) using the Kissinger equation. It enables us to determine the difficulty for the transport of the macromolecular segments to the growing crystal surface. Plots of ln (ϕ/
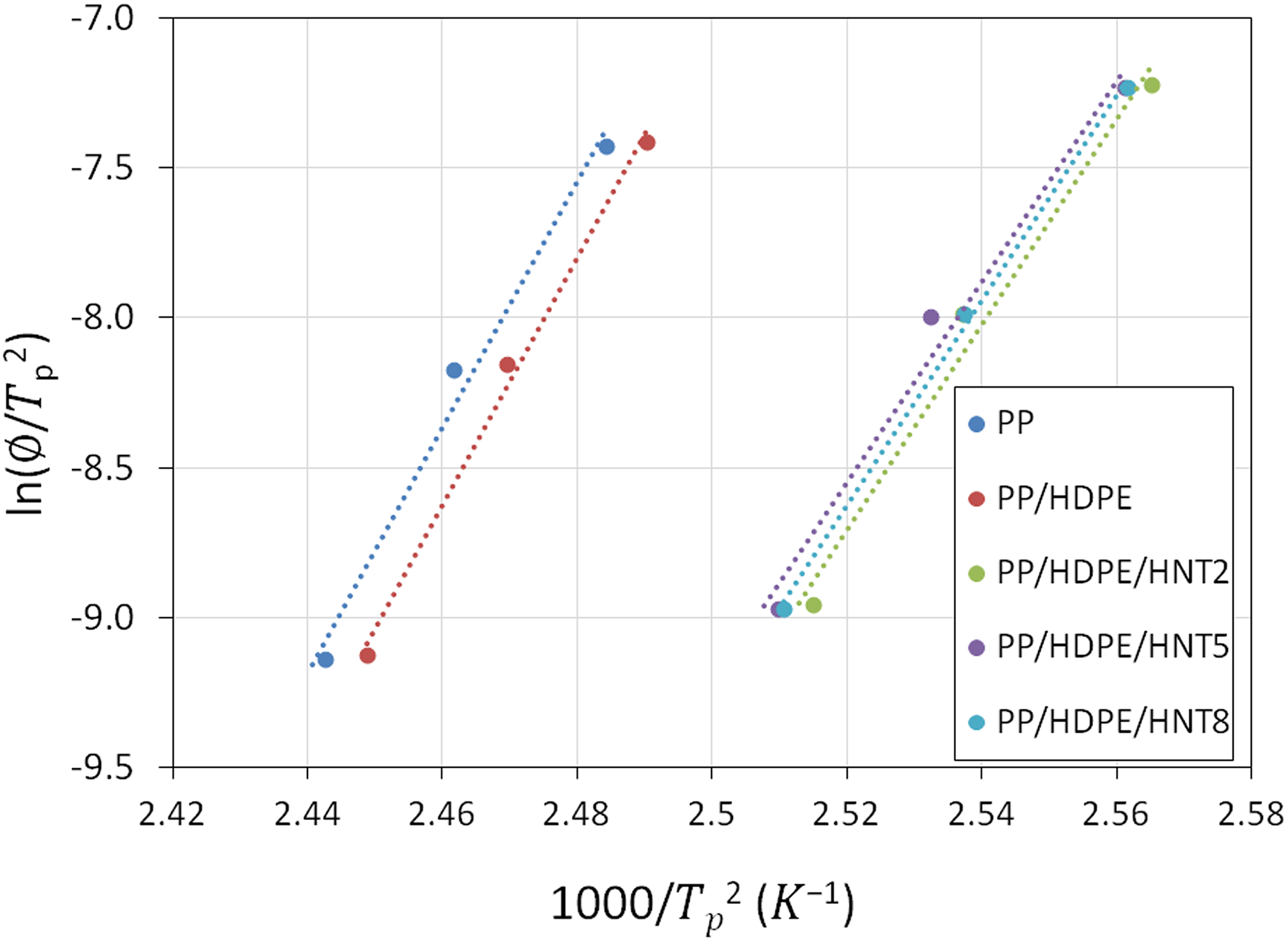
Kissinger plot for PP/HDPE/HNT nanocomposites.
E a values of PP/HDPE/HNT nanocomposites determined using Kissinger equation.
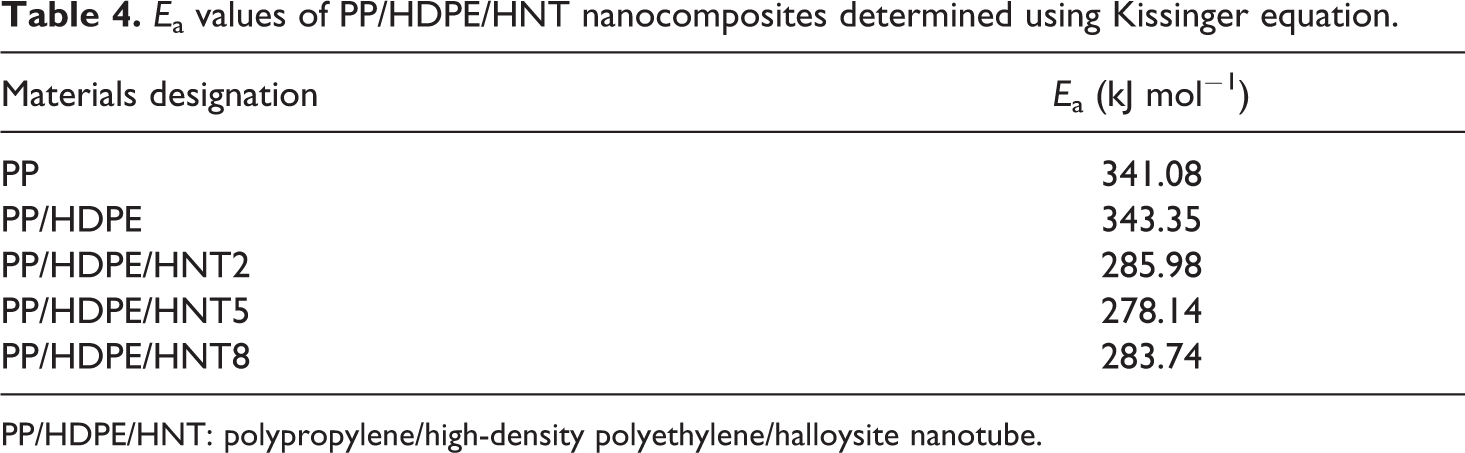
PP/HDPE/HNT: polypropylene/high-density polyethylene/halloysite nanotube.
Conclusions
Based on this study devoted to determine the kinetics of non-isothermal crystallization of PP/HDPE/HNT nanocomposites using three methods, that is, Avrami equation, Mo model, and Kissinger equation, the following conclusions can be drawn. The Avrami exponent (n) is ranged from 1 to 2 for all PP/HDPE/HNT nanocomposites indicating instantaneous nucleation. The Zt values of PP/HDPE blend (0.03) increased to 0.06, 0.08, and 0.09 by the addition of 2, 5, and 8 phr of HNT, respectively. The increased Zt
values indicate increased rate of crystallization with increased loading of HNT of PP/HDPE/HNT composites. The half crystallization time (t
1/2) for PP/HDPE (6 min) decreased to 4 min for PP/HDPE/HNT composites regardless of HNT loading, where lower t
1/2 values indicate faster crystallization rate. In the Mo model, the cooling rate chosen at unit crystallization time (F(T)) values for PP/HDPE, which is 594.98, decreases to 325.16, 320.92, and 230.89 for 2, 5, and 8 phr of HNT, respectively. The increasing loading of HNT decreases the values of F(T), where the lower values of F(T) indicate higher crystallization rate. Kissinger equation showed that the activation energy E
a of the PP/HDPE (343.35 kJ mol−1) decreases to approximately 278.14 kJ mol−1 by the addition of 5 wt% HNT, indicating that HNT accelerates the crystallization rate.
Footnotes
Acknowledgement
The authors would like to thank Universiti Sains Malaysia for providing Bridging Fund.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universiti Sains Malaysia by providing Bridging Fund (304.PBAHAN.6316090).