Abstract
Non-isothermal crystallization kinetics of virgin polypropylene (PP) and its nanocomposites have been evaluated using differential scanning calorimetric technique. The nanocomposites were prepared using melt intercalation method. It is observed that the crystallization peak temperature of nanocomposite is marginally higher than virgin PP at various cooling rates. The non-isothermal crystallization melt data were analysed using Avrami, Ozawa and Mos models. The half-time for crystallization decreased with incorporation of clay nanoparticles. The values of crystallization rate constant and cooling rate at unit crystallization time showed that the crystallization rate increased with the increasing of cooling rates for virgin PP and nanocomposite, but the crystallization rate of nanocomposite was faster than that of PP at a given cooling rate. The activation energy for non-isothermal crystallization of virgin polymer and nanocomposites based on Kissinger method has been determined to be 186 and 196 kJ mol−1, respectively. The polarized micrographs showed that the number of effective nuclei increased in the PP-clay nanocomposite, where the clay acts as a heterogeneous nucleating agent during the crystallization of the nanocomposite.
Keywords
Introduction
In the recent years, clay-based polyolefin nanocomposites have gained considerable research interests ranging from packaging to automotive applications. Polypropylene (PP) is one of the most widely used polyolefin polymers. Incorporation of fillers has been proved to be an effective way of improving the mechanical properties of PP. It is a semi-crystalline thermoplastic polymeric material that finds wide application because of its excellent processibility, good mechanical properties and very good chemical resistance. 1 –4 Smectite clays, such as montmorillonite, saponite and surface-modified montmorillonite, are valuable minerals used in many industrial applications because of their high aspect ratio and plate-like morphology. Among all, montmorillonite is a naturally abundant and environment-friendly cheap clay material used in polymeric matrices such as PP to make material with superior final properties 5 . PP does not contain any polar groups in its backbone for which it has been difficult to attain homogeneous dispersion of clay particles inside the polymer matrix. According to chemical thermodynamic study, entropic and enthalpic balances determine the dispersion of the clay particles in the polymer matrix. As the polymer chains become constrained between silicate layers, the entropy decreases. In order to get a homogeneous dispersion, the interaction between the clay and the polymer matrix must be favourable. 6 –8 Hence, to improve the interaction between polymer matrix and clay, montmorillonite is usually modified with alkyl ammonium, which makes the hydrophilic clay surface organophilic 9 . Several methods have been adopted to prepare organoclay/polyolefin nanocomposites, including in situ polymerization of propylene in the presence of layered silicates, 10,11 , solvent blending 12 and melt compounding. 1,13 Among all, melt compounding has been the most convenient process for preparation of the nanocomposites.
Kawasumi et al. firstly prepared PP/clay nanocomposites by direct melt blending technique with the use of maleic anhydride-modified PP (PP-
The subject of polymer crystallization kinetics has been of great interest for several decades and still provides fruitful areas for research. Crystallization, like any phase transformation, obeys the fundamental laws of thermodynamics, which suggest whether, under specific conditions and circumstances, there is existence of crystal or not. In ideal conditions where external conditions are constant, the studies of crystallization are limited because of fewer problems connected to cooling rates and thermal gradients inside the polymer matrix. In reality, during various industrial processes, it is seen that external conditions change continuously for which the studies of non-isothermal crystallization are more complex in nature. Whatever it may be, the study of crystallization of polymers is of great significance in a continuously varying situation since many manufacturing and engineering processes in industry proceed under nonisothermal conditions. If we see from a scientific point of view, the study of crystallization in dynamic conditions may enlarge our knowledge of understanding regarding crystallization of polymers since many isothermal processes occur frequently under constrained to narrow temperature ranges.
The purpose of this work is to provide the current state of art for non-isothermal crystallization of polymer and its nanocomposites during cooling from the melt. This article also reviews the various theories of non-isothermal crystallization processes of virgin polymer and its nanocomposites and then its experimental observations in dynamic conditions. The aim of the present work is to arouse discussion pertaining to non-isothermal crystallization kinetics in order to get better prophecy of the reaction parameters of dynamic solidification under genuine processing conditions. This comprises an imperative industrial and scientific confront in the area of non-isothermal crystallization of polymer and its nanocomposites.
It is evident that the mechanical and physical properties of polymers are invariably dependent on morphology and degree of crystallinity during the time of processing. The surface of the filler particles acts as nucleation site for semi-crystalline polymer matrix and affects the crystallization behaviour. Therefore, it is very important to study the nonisothermal crystallization kinetics of polymer materials
20
. In this article, PP and organophilic montmorillonite were used as the base polymer and reinforcing agent, respectively, for the preparation of nanocomposites. Comprehensive study has been made on the nonisothermal crystallization kinetics of PP and clay nanocomposites. In the recent years, the nonisothermal crystallization kinetics of several polymers was investigated by several authors
21
–23
and different kinetic models were applied to deal with the experimental data derived using the differential scanning calorimetry (DSC) technique. The main objective in this work is to make a relative comparison of crystallization kinetics under nonisothermal condition of virgin PP and PP/PP-
Experimental
Materials
PP (H100EY, weight-average molecular weight = 180,000 g mol−1) was obtained from the Reliance Industries Ltd (Jamnagar, Gujarat, India). Virgin PP was in the form of pellets with a melt flow index (MFI) of 11 g/10 min at the rate of 230°C/2.16 kg and a density of 0.9 g cm−3. PP-
Preparation of PP/PP-g -MAH/C15A nanocomposite
Prior to extrusion, the clay powder was dried in a vacuum chamber oven at 80°C for 12 h to remove the absorbed moisture. Three PP master batches were prepared containing PP and C15A along with virgin PP master batch. The composition of each master batch containing C15A was in the range of 3, 5 and 7% by weight without using compatibilizer. Nanocomposites were prepared by melt mixing of the two components using torque rheometer (Haake Rheomix OS, Germany) with counterrotating roller rotors having a chamber size of 66 cm3. The screw speed was 80 r min−1 and the mixing time was 15 min for all the compositions. The barrel temperature profile was optimized from 175 to 190oC from feed to die zone. After compounding, the mixed material was extruded to produce cylindrical extrudes. The optimized composition of the master batch was further mixed with PP-
Instruments and conditions
Differential scanning calorimetry
Crystallization of PP/PP-
Polarized light microscopy
Crystalline morphology studies were carried out using a DM 4500 P, LED Leica; Germany PLM equipped with crossed polar in conjunction with a Linkam THMS 600 hot stage. Samples of specific thickness were prepared by cutting small pieces from films. These samples were heated to 200°C at 20°C min−1, kept for 10 min at 200°C, and then quickly cooled at the rate of 40°C min−1 to reach the desired crystallization temperature. The temperature was kept constant for 2 min at this stage and then further cooled at the rate of 2°C min−1 to 100°C. The growth of spherulite during that stage was monitored. Micrographs were taken at various time intervals for interpreting the spherulite size.
Results and discussions
Crystallization behaviour
The effects of clay on the crystallization behaviour of PP were quantitatively analysed through nonisothermal DSC experiments. The crystallization thermograms of PP and PP/PP-
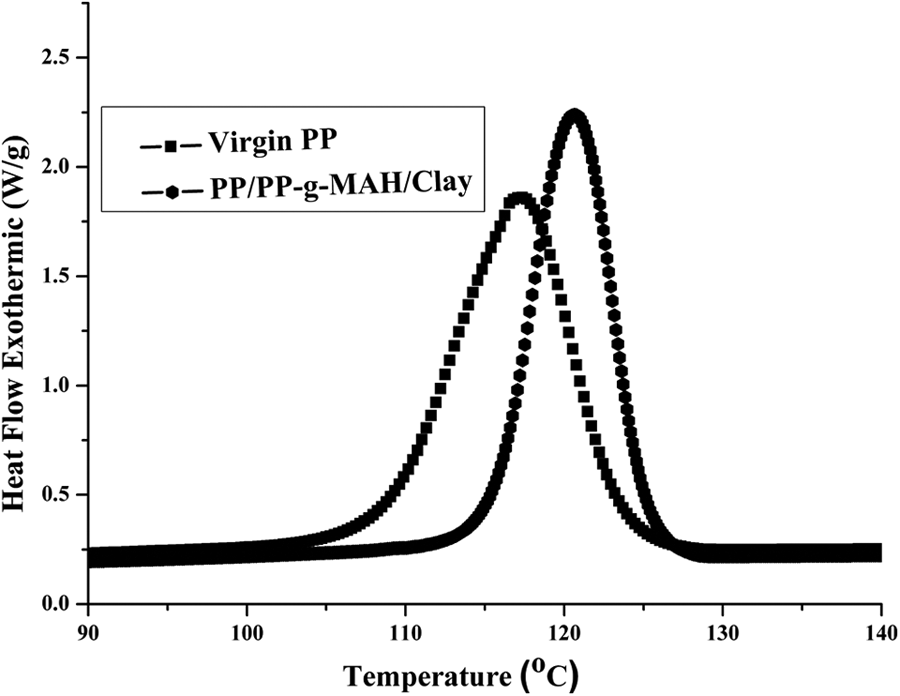
DSC thermograms virgin PP and PP/PP-
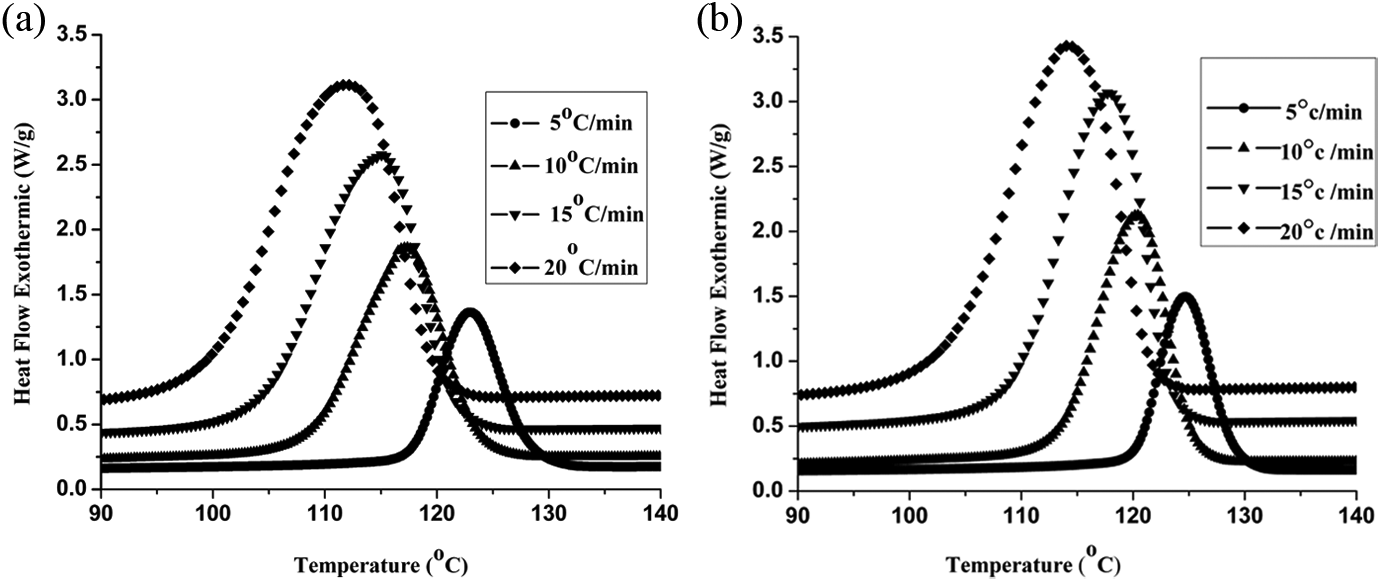
(a) DSC thermograms of virgin PP recorded during non-isothermal crystallization at different cooling rates. (b) DSC thermograms PP/PP-
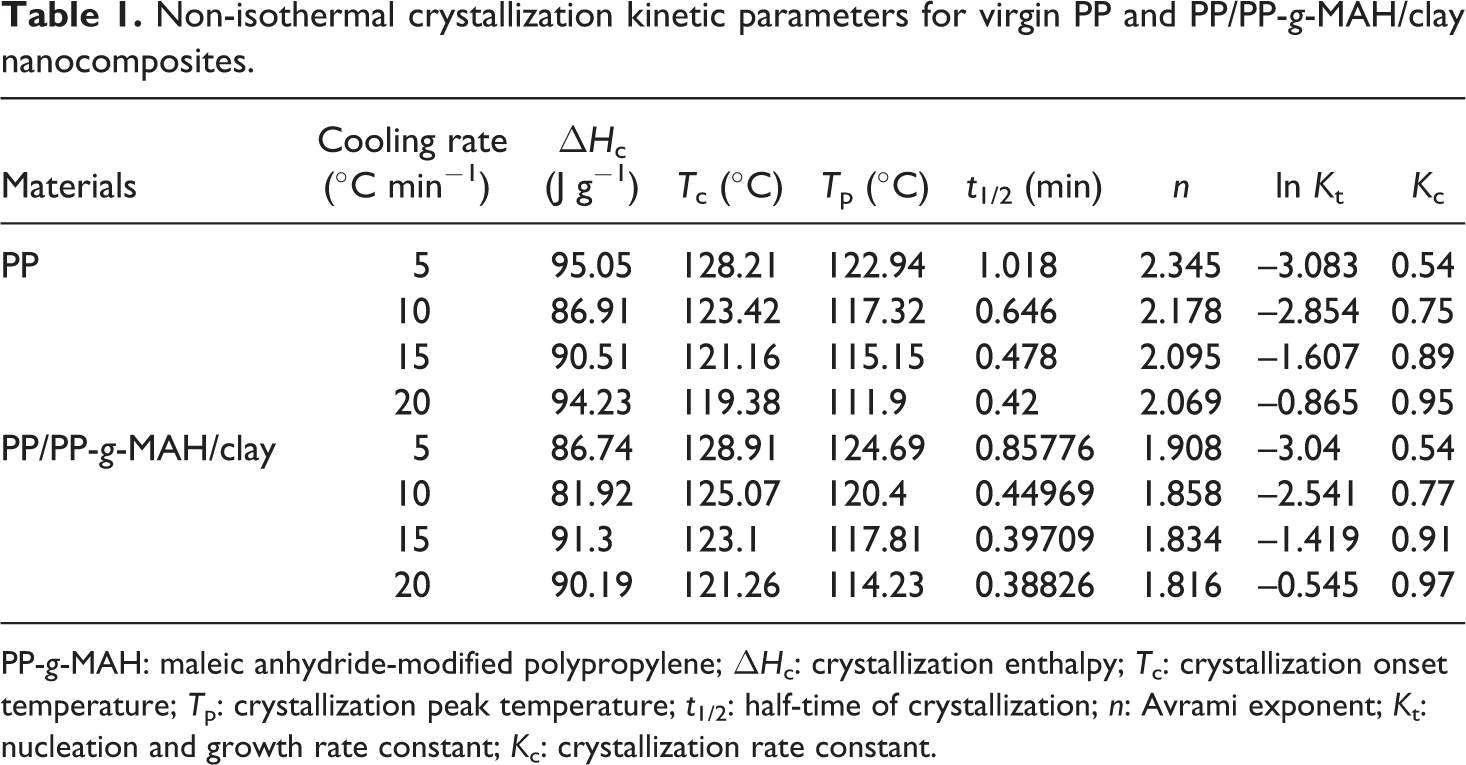
Non-isothermal crystallization kinetic parameters for virgin PP and PP/PP-
PP-
Figure 3 shows the relationship between crystallization
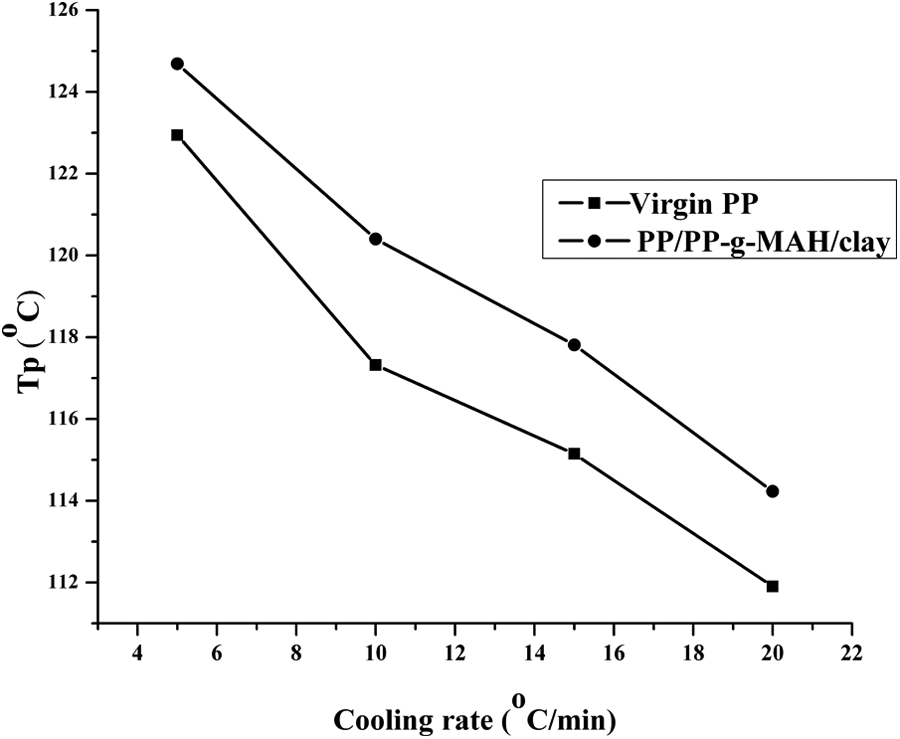
Crystallization peak temperature (
Under the same cooling rate, the
Figure 4(a) and (b) shows the relationship between the relative degree of crystallinity ( (a) Relative crystallinity (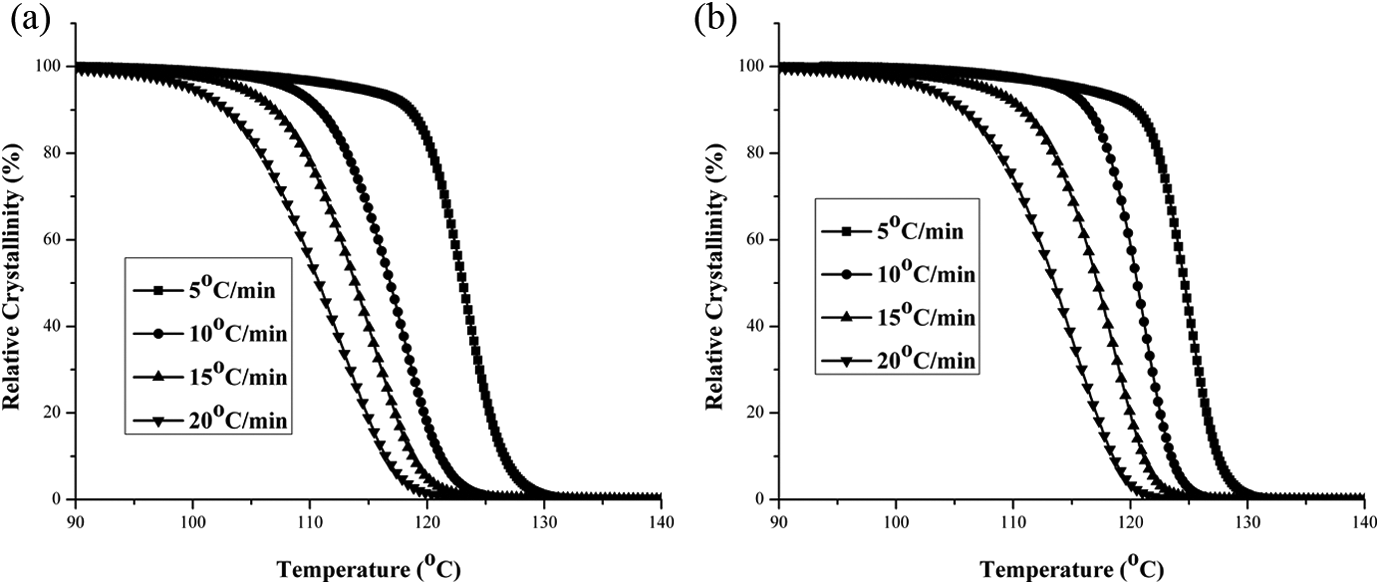

where
The relation between crystallization time and temperature during the nonisothermal crystallization process is given by equation (2):
where
Figure 5(a) and (b) shows the plots of the
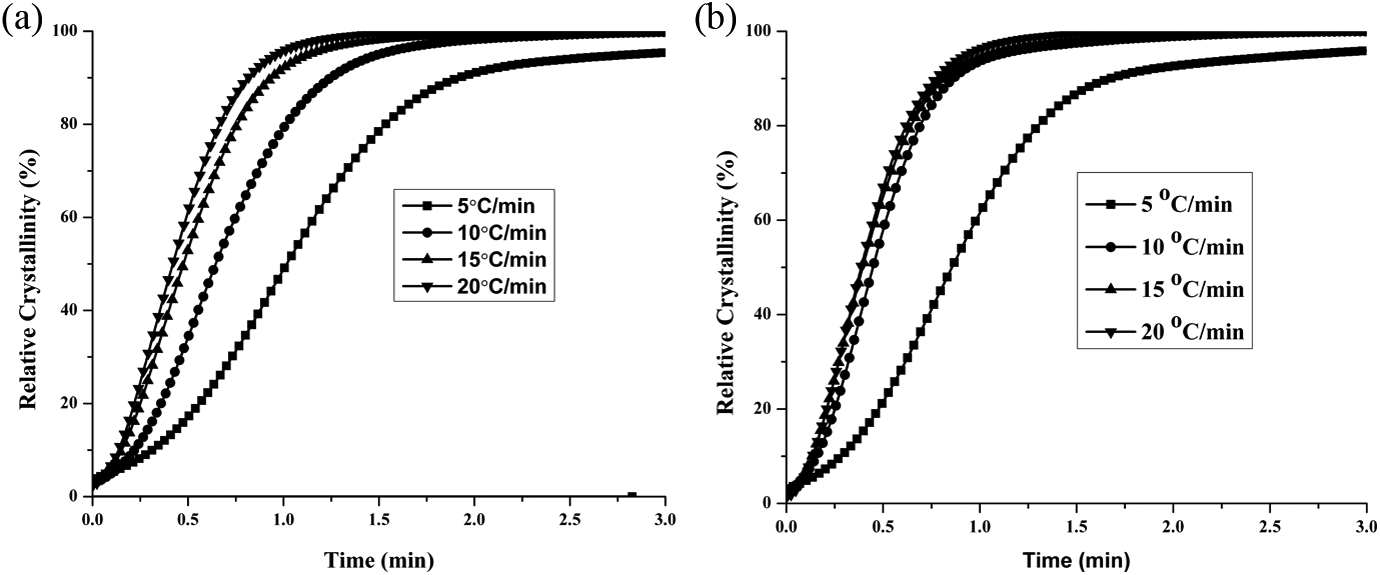
(a) Variation of relative crystallinity (
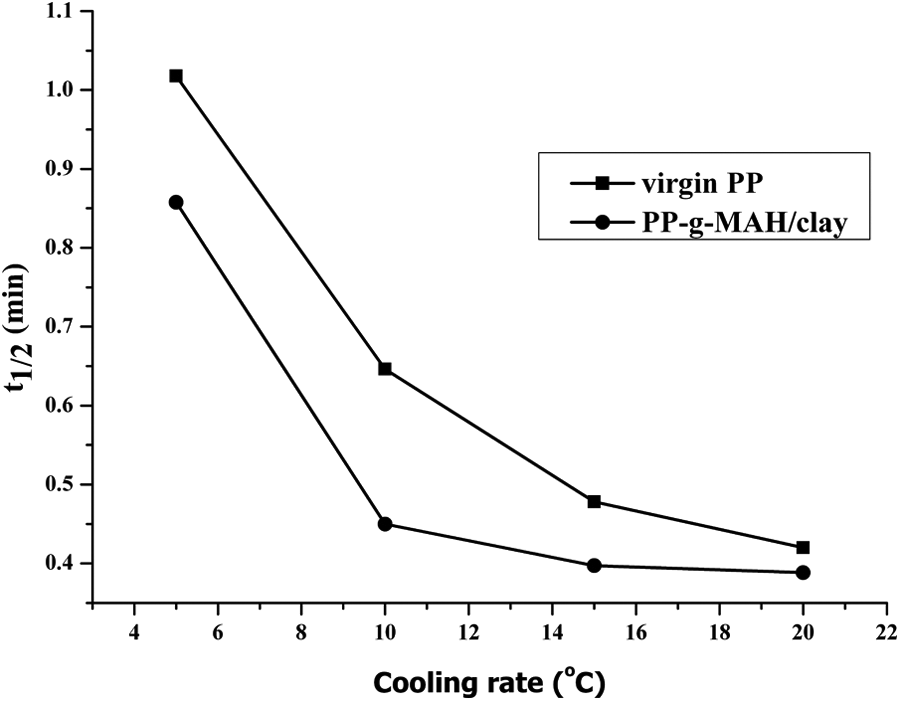
Non-isothermal crystallization half-time (
Nonisothermal crystallization kinetics
The primary stage of nonisothermal crystallization kinetics could be described by the Avrami equation. The equation is represented as follows:
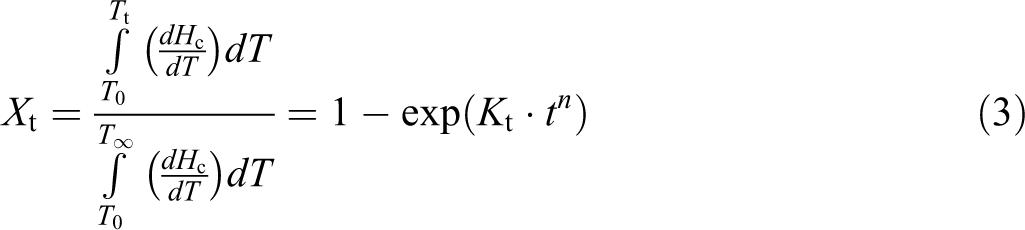
The double logarithmic form of equation (3) is given as: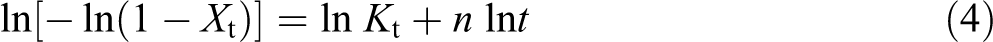
Where
Figure 7(a) and (b) shows the plots of ln[–ln(1–
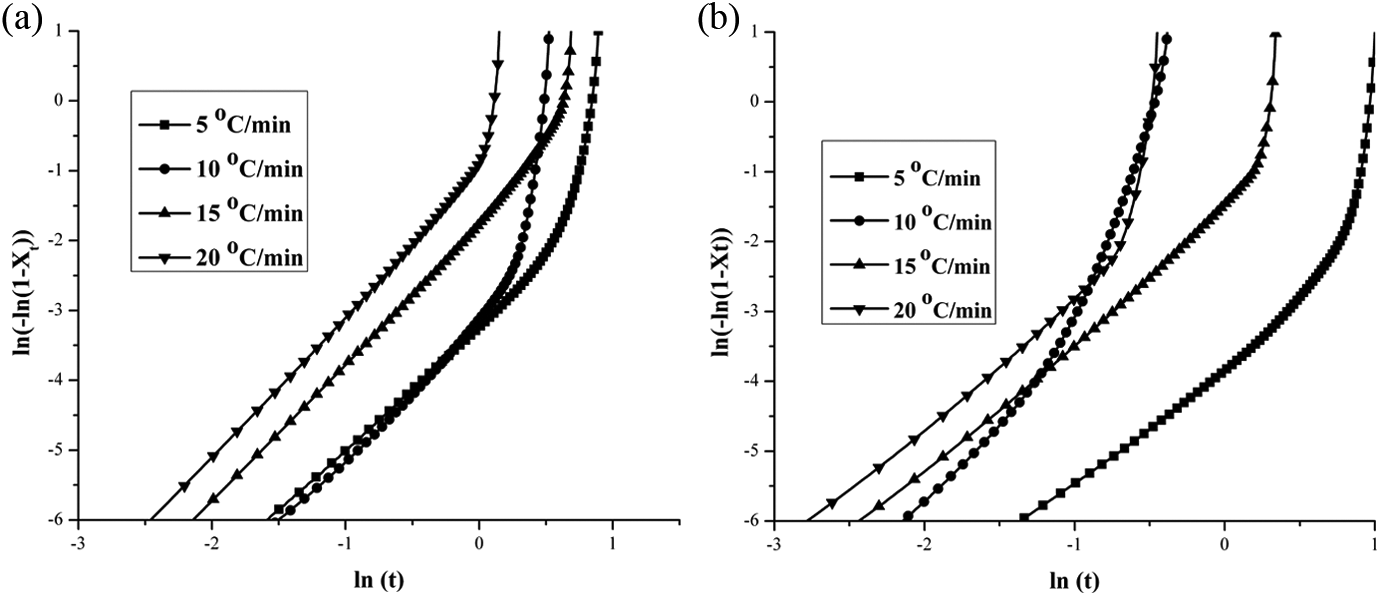
(a) Plots of ln(–ln(1–
The value of simultaneous occurrence of two- and three-dimensional spherulite growths with heterogeneous nucleation; simultaneous occurrence of two-dimensional spherulite growth with heterogeneous and homogeneous nucleation; and combined effect of the above two mechanisms.
The values of
Hao et al.
39
reported the modified Jeziorny equation for the non-isothermal crystallization process and represented as follows:
where
Ozawa analysis
Ozawa modified the Avrami equation by incorporating the cooling rate factor 
where 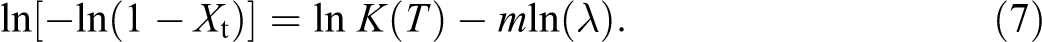
Several authors have claimed that Ozawa model is not valid for the nonisothermal crystallization process.
20,22
But we have used this model for the nanocomposite. Drawing the plot between ln[–ln(1–
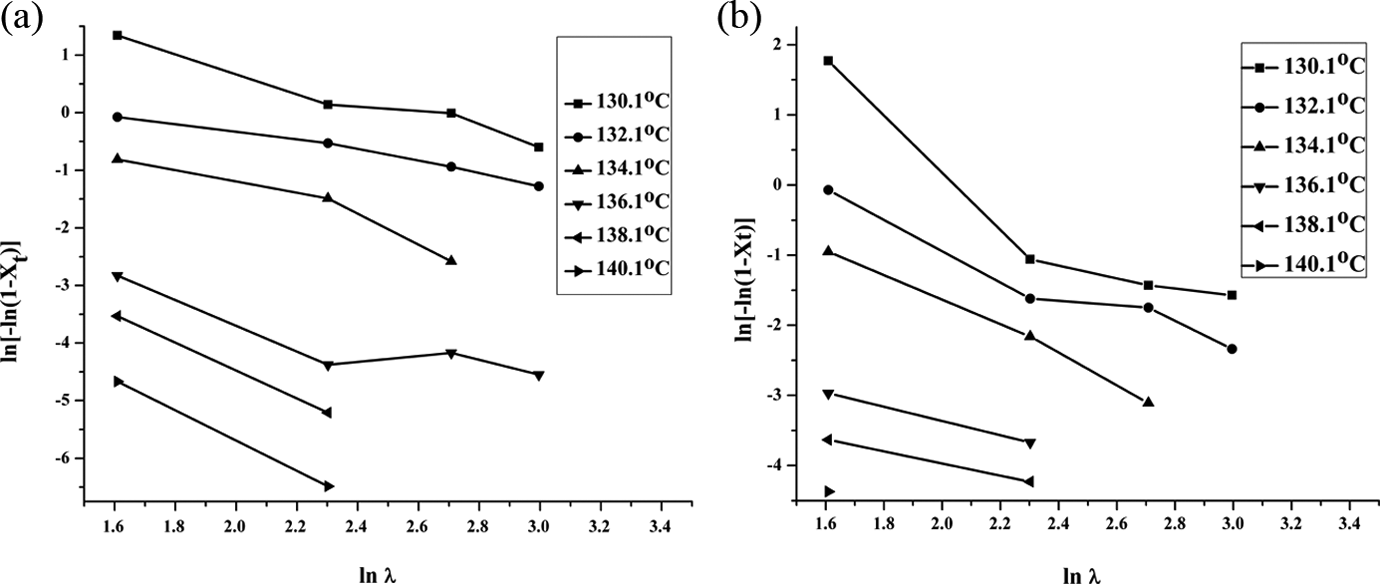
(a) Ozawa plots of ln[–ln(1–
It is clearly seen from the Ozawa plots that the curves do not exhibit linearity in the upper parts. This might be due to retardation in crystal growth at higher cooling rate, as it takes place in a much constrained environment. It is explained that during cooling, secondary crystallization could not take place due to continuous decrease in temperature.
Mo’s analysis
In order to explain properly the kinetics of non-isothermal crystallization Mo et al.
40
derived a relation between the cooling rate (
where
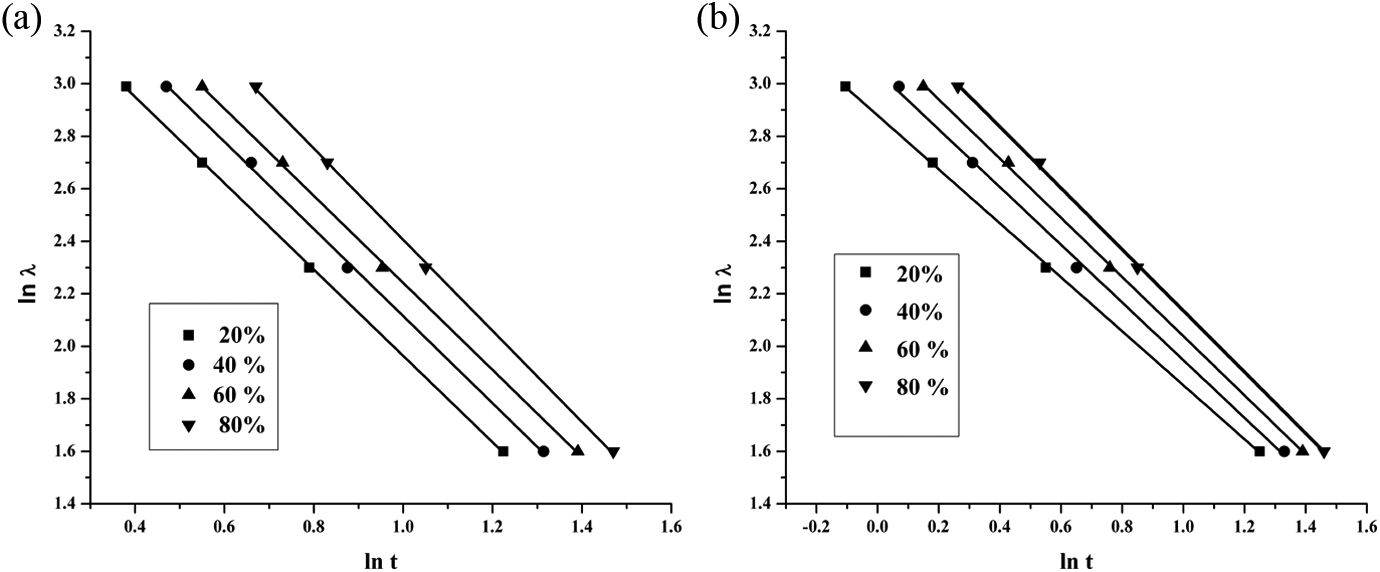
(a) Mo plots of ln
At a given degree of crystallization, a linear relationship has been seen. Using linearity fitting method, we can obtain the slope (–α) and the intercept (ln
Non-isothermal crystallization kinetics parameters for the PP and PP/PP-g-MAH/clay nanocomposite at different relative degrees of crystallinity by combination of Avrami–Ozawa equation.
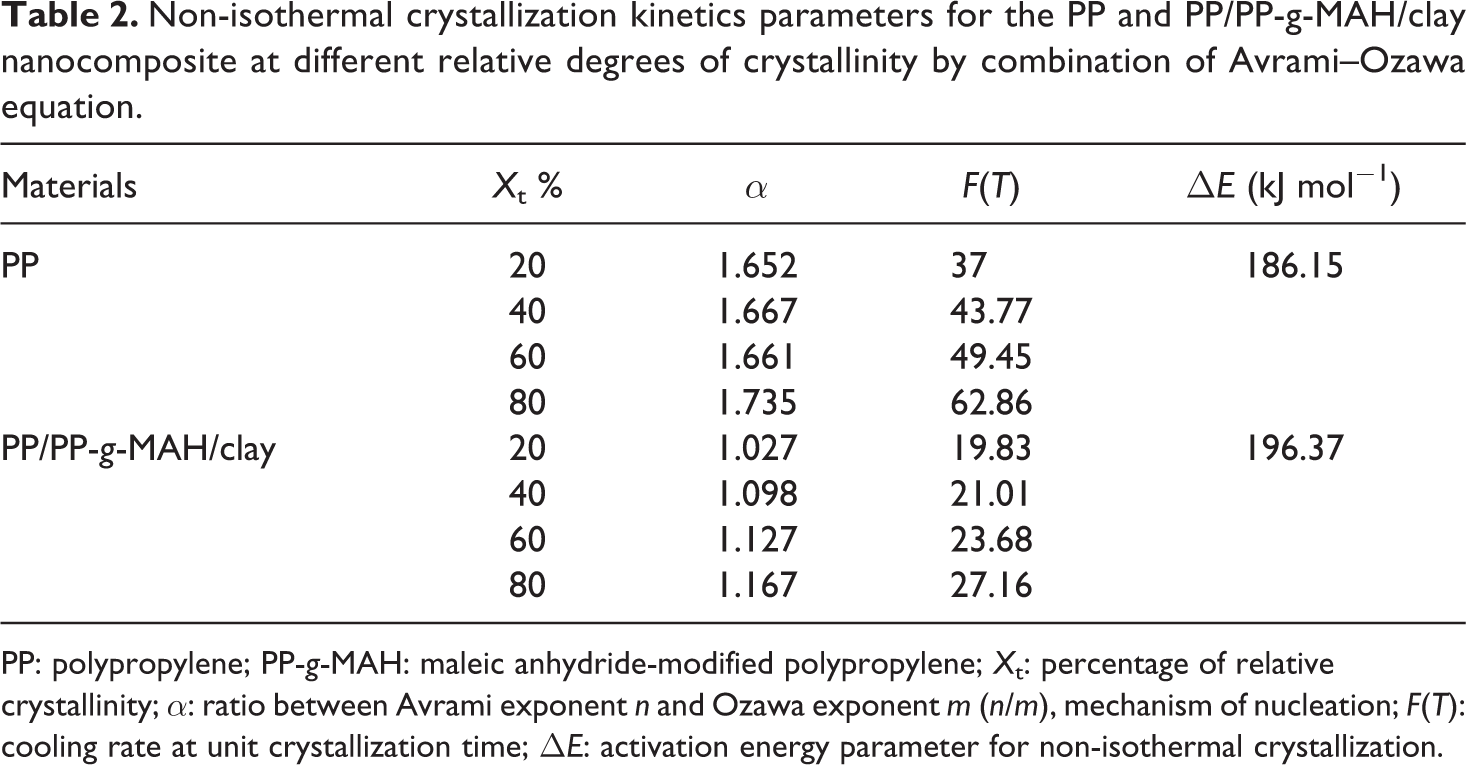
PP: polypropylene; PP-
Crystallization activation energy
Apart from all the mentioned crystallization kinetic study, it is also necessary to calculate the activation energy for a non-isothermal crystallization process. Kissinger approach is widely used in estimating the overall effective energy required for the transport of the polymeric chain segments to the growing crystal surface. The Kissinger equation is 41 :

where
Taking logarithm of equation (9), we get:
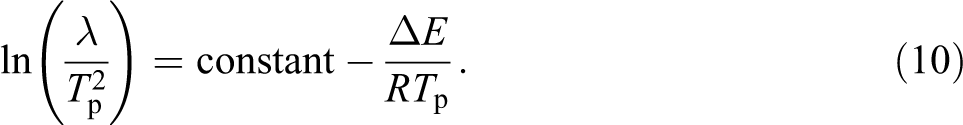
Figure 10 shows the plots based on the Kissinger method and the slopes of the lines equal to –Δ
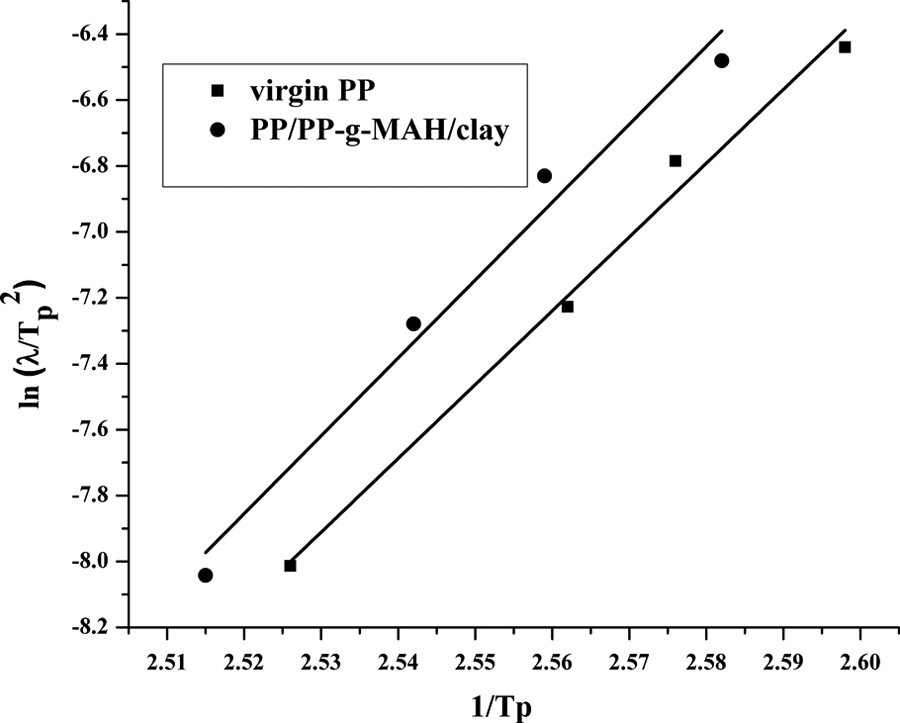
Plots of Kissinger treatment for evaluating non-isothermal crystallization activation energy of (a) virgin PP and (b) PP/PP-
Morphological observations
Morphological observations were studied with the help of PLM. Figure 11(a) and (b) shows the micrographs of virgin PP and PP/PP-
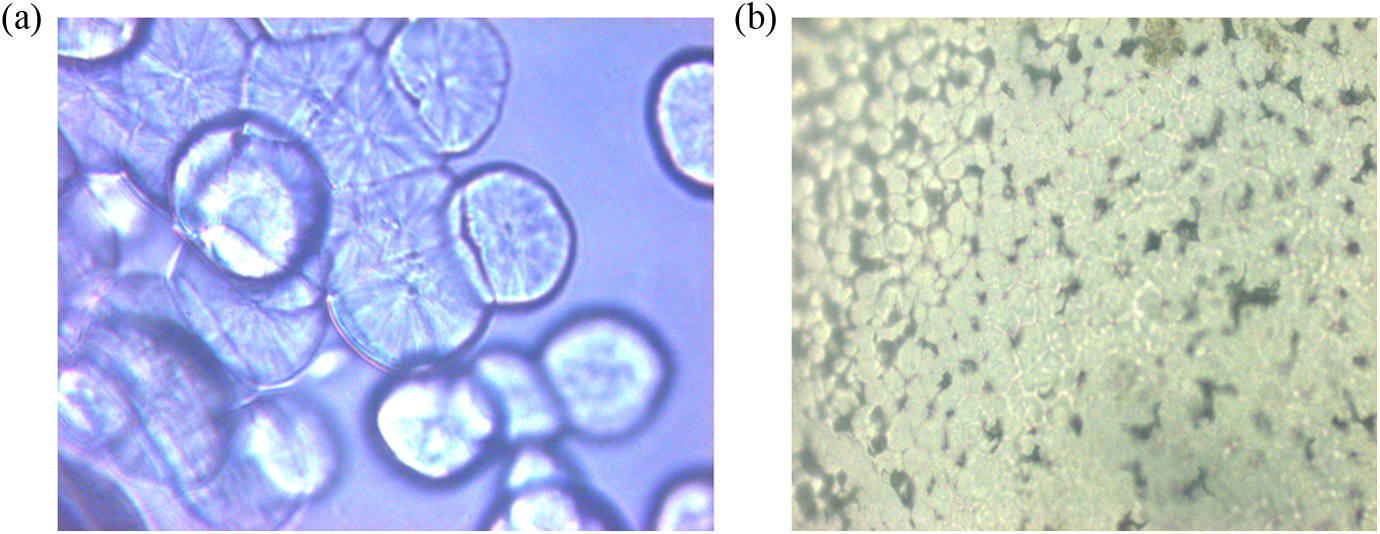
(a) The polarized light micrographs showing the morphology of crystals of virgin PP at respective crystallization temperature (magnification ×500). (b) The polarized light micrographs showing the morphology of crystals of PP/PP-
Conclusion
The non-isothermal crystallization behaviour of virgin PP and PP/PP-
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
