Abstract
This article highlights the potential of halloysite nanotube (HNT) as a multifunctional filler for polypropylene/polyamide 66 (PP/PA66) blends, for their roles as the reinforcing agent, compatibilizer, nucleating agent, and thermal stabilizer. The flexural modulus and strength of PP/PA66 were improved by the incorporation of HNT due to the reinforcing ability and compatibilization effects of HNT. 4 parts per hundred resin (phr) of HNT improved the thermal stability of PP/PA66 blends due to its heat barrier effects. Adding HNT increased the degree of crystallinity for PP and PA66, where the effects of HNT were more prominent in the PA66 phase, attributed to additional hydrogen bonding formation between the PA66 and HNT.
Introduction
Polypropylene (PP) is a commodity polymer that is commonly used in many applications, such as home appliances and automotive parts. However, the usage of PP often limited due to its low impact strength, particularly at low temperatures. Blending PP with different polymers is an economic and effective way to improve these drawbacks. 1 PP and polyamide (PA) blending has been attempted to achieve improvement in mechanical properties, paintability, and barrier properties. In general, PA contributes to mechanical and thermal properties, while PP ensures good processing ability and high moisture barrier properties. 2,3 Polyamide 66 (PA66) is an engineering thermoplastic that is widely used in numerous engineering applications. The reason behind this is that PA66 possesses outstanding properties such as high strength, toughness, and corrosion resistance. 4 Besides that, PA66 can be easily processed using injection molding attributed to its rapid crystallization and melt strength. 5
The mechanical and thermal properties of PP/PA can be further tailored by adding of nanofillers, for example, titanium dioxide (TiO2) nanoparticles, 6 organo-montmorillonite nanoclay, 7 and carbon nanotube. 8,9 The presence of functionalized TiO2 nanoparticles in PP/PA6 blend increases the crystallization rate due to the induction of heterogeneous nucleation of PP and PA6 chains. 6 The storage modulus of PP/PA is improved by the incorporation of nanoclay. 7 The crystallization temperature and thermal stability of the PP/PA blends increased in the presence of 5 wt% of CNT. 8
Halloysite nanotube with the chemical formula of Al2Si2O5(OH)4.nH2O is a two-layered natural aluminosilicate clay minerals in nanotubular form. 10 The advantages of HNT are attributing to their natural availability, nanosize structure, and suitability for polymer nanocomposite application. 11,12 HNT is more easily dispersed in polymer matrix compared to other silicates (e.g. montmorillonite) using commercial processing methods. 13,14
Studies on HNT-filled single polymer nanocomposites, for example, PP/HNT nanocomposites 15 -17 and PA/HNT nanocomposites, 18,19 have been reported. The thermomechanical properties of the PP and PA are enhanced by the addition of HNT. HNT-filled binary polymer blends nanocomposites also being developed, for example, PP/acrylonitrile–styrene copolymer/HNT, 20 poly(lactic acid)/poly(methyl methacrylate)/HNT, 21 and poly(ether ether ketone)/liquid crystal polymer/HNT. 22 To the best of the author’s knowledge, there is no study on PP/PA/HNT binary nanocomposites.
Note that, the blending of PP (major matrix) and PA66 (minor phase) that required to be melt-compounded at temperature 260°C (Tm of PA66) may induce thermal degradation of PP. The selection of PA66 was based on its high melting temperature (higher than PA6 = 230°C) so that the workable temperature of the HNT (as both reinforcing agent and thermal stabilizer) can be determined. It was believed that if the HNT can perform well for the PP/PA66, then it should be feasible for other PP/PA blends (e.g. PP/PA6). It was known that adding antioxidant is one of the possible approaches to stabilize the thermal properties of polymer blends. However, some of the issues need to be concerned, for example, selection of a suitable antioxidant and antioxidant effects on mechanical properties of the polymer blend. Thus, in this study, the “multi-functionality” of HNT is explored in terms of reinforcing ability, thermal stabilization, and crystallization for PP/PA66 blends.
Materials and methods
Sample preparation
PP (TitanPro SM-128) was supplied by Lotte Chemical Titan Holding Sdn. Bhd. (Malaysia), while PA66 (Amilan CM3001-N) was obtained from Toray Nylon Resin Amilan (Malaysia). HNT was purchased from Sigma-Aldrich (Malaysia). PP, PA66, and HNT were pre-dried in an oven at 100°C for 24 h. PP/PA66 (blending ratio = 80/20) and HNT (loading = 2 to 8 phr) were compounded using a single screw extruder (Kompaktextruder KE19, Brabender, Germany). The temperatures of the feeding zone, compression zone, metering zone, and die zone were 200°C, 240°C, 260°C, and 255°C, respectively. The rotor speed was set at 40 r min−1. The extrudate obtained was granulated using a granulator (Brabender, Germany). The granules were pre-dried at 100°C for 24 h prior to injection molding. The temperatures of the injection molding machine (BOY 22M, Exton, PA, USA) at the feeding-, compression-, metering-, and nozzle-zone were set at 210°C, 260°C, 260°C, and 250°C, respectively. The mold temperature was set at 40°C, while the cooling time was 20 s. Samples obtained from the injection molding (125 mm length × 13 mm width × 3 mm thickness) were used for the flexural test.
Characterization
Flexural tests
The flexural properties of the nanocomposites were determined by three-point bending test using a universal testing machine (Instron 3366, Norwood, MA, USA). The length of the support span was set at 50 mm, while the crosshead speed was set at 1.50 mm min−1. The flexural modulus and strength of PP/PA66 nanocomposites were determined.
Morphology assessment
The morphology of the cryogenic-fractured surface of PP/PA66 and PP/PA66/HNT nanocomposites was investigated using field emission scanning electron microscope (FESEM; Zeiss Supra, Germany). The samples were immersed in liquid nitrogen for 10 s before cryogenic fractured. The fractured samples were sputter coated with a layer of gold to prevent electrostatic charging during FESEM inspection.
Thermogravimetric analysis
Thermal decomposition of the PP/PA66/HNT nanocomposites was analyzed using the thermogravimetric analyzer (Pyris 6, Perkin Elmer, Boston, MA, USA) under N2 atmosphere. The sample weight about 10 mg was scanned at a heating rate of 10°C min−1 from 30°C to 600°C. The weight change was recorded as a function of the heating temperature. From the TGA and derivative thermogravimetry (DTG) curves obtained, the onset of thermal decomposition (Tonset) refers to the starting of the degradation temperature, the temperature of maximum decomposition (Td) refers to the temperature at the end of decomposition, and the peak temperature (Tmax) refers to the temperature where the rate of degradation was maximum (detected from DTG curves).
Differential scanning calorimetry
The thermal properties of the PP/PA66/HNT nanocomposites were characterized using differential scanning calorimeter (DSC 200, Mettler Toledo, Columbus, OH, USA). For the first heating, the samples were scanned from 0°C to 300°C at a heating rate of 10°C min−1, held for 1 min, and cooled back to 0°C at the same rate to remove the thermal history. For the second heating, the same process was repeated where the samples were scanned from 0°C to 300°C at a heating rate of 10°C min−1, held for 1 min, and cooled back to 0°C at the same rate (10°C min−1). The melting temperature for PP (Tm(pp)), PA66 (Tm(PA66)) and crystallization temperature for PP (Tc(pp)), PA66 (Tc(PA66)) were determined. The degree of crystallinity, χc for PP and PA66 was calculated based on equation (1).
where ΔHm refers to the enthalpy of fusion of the blend component, while ΔHfº refers to the blend component at 100% degree of crystallinity, 207.1 J g−1 for PP and 255.8 J g−1 for PA66. 23 w denotes the net weight percentage of the blend component.
Results and discussions
Flexural properties
Figures 1 and 2 show the flexural modulus and flexural strength of PP/PA66 blends with different HNT loadings. The flexural modulus of PP/PA66 increased with increasing loading of HNT, attributed to the stiffness and reinforcing effects of HNT. Similar findings had been reported by Du et al. 24 and Handge et al. 25 on the PP/HNT nanocomposites and the PA6/HNT nanocomposites. The enhancement in mechanical properties of polymer nanocomposites is associated with the loading of HNT. Note that the increment of modulus also can be attributed to good dispersion of the HNT in the polymer matrix. 26
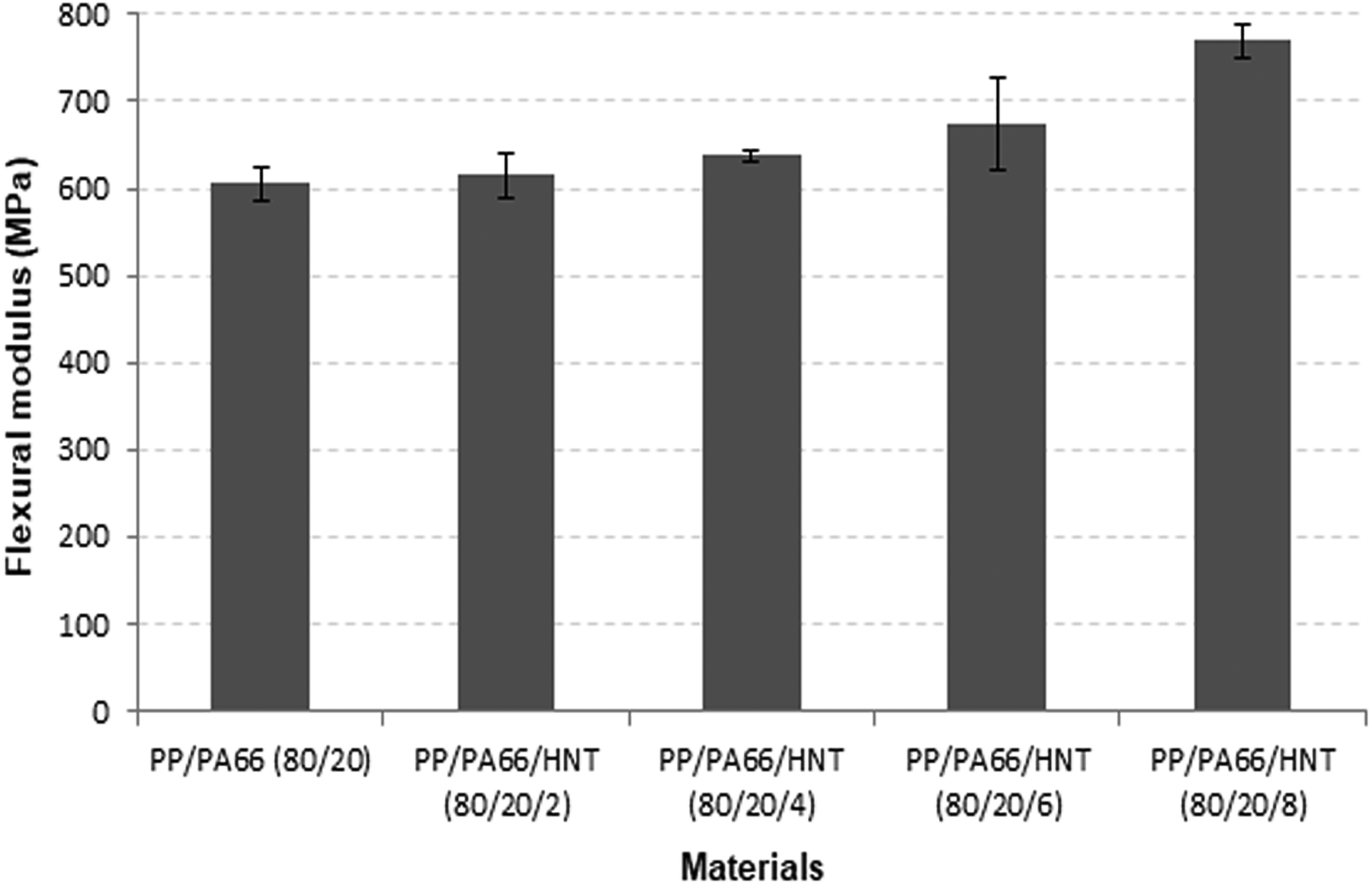
Effects of HNT on the flexural modulus of PP/PA66 blends.

Effects of HNT on the flexural strength of PP/PA66 blends.
The improvement of mechanical properties such as modulus and strength of nanocomposites is mainly because of the high aspect ratio of nanosized filler and strong polymer–filler interactions that impart effective load transfer between the filler and the matrix. 27 HNT can easily disperse in a nonpolar polymer matrix such as PP due to secondary interactions among the nanotubes. Furthermore, hydrogen bonding can occur between the polymer matrix and the filler due to the polarity of polyamide. 28 HNT contains inner and outer hydroxyl groups. Most of the hydroxyl groups are inner groups, with relatively small amount are located on the surface due to the multilayer structure of HNT. 29 The reactive hydroxyl site of Al–OH groups is present at the edges and the surfaces of HNT, leading to a certain level of interfacial chemical interaction such as hydrogen bonding with PA6. 30
Morphological properties
The morphology of cryogenic-fractured PP/PA66 blend and its nanocomposite was analyzed using FESEM. Figure 3 shows the FESEM image of PP/PA66 (80/20), while Figure 4 shows the FESEM image of PP/PA66/HNT (80/20/4). From Figure 3, the minor phase (PA66 in the form of droplets) is dispersed well in the major phase (PP matrix). The PA66 minor phase appears in both circular and elongated (ellipse) shape. This is common for a polymer blend with sea-and-island microstructure. Note that, the size of PA66 varied from the ranges of 2.2 µm to 10 µm. Furthermore, the droplet indicated by the arrow clearly shows that PA66 was deformed and sheared. This is due to the shearing effect generated from extrusion and injection molding process. From Figure 4, it can be seen that the size of the PA66 droplet was in the range of 0.5 µm to 2.5 µm. The reduction of droplet size of the PA66 dispersed phase can be due to compatibilization effects induced by HNT.

FESEM image of PP/PA66 blend.

FESEM image of PP/PA66/HNT (80/20/4).
The addition of mineral filler into a blend may affect the compatibility between the two polymers. Nanoclay can act as a compatibilizer in an immiscible blend and reduces the size of the dispersed phase. 31 According to Pal et al., 32 the compatibility of polyoxymethylene/PP blends was enhanced by the addition of HNT. They found that after incorporation of the HNT, the size of the dispersed domain decreased. HNT restricts the small droplets to agglomerate themselves hence droplets remain in a smaller size. This indicates the bridging effect of HNT, and thus it increases the compatibility of the blends. Besides that, according to Kubade and Tambe, 33 the addition of HNT reduces the acrylonitrile butadiene styrene (ABS) dispersed phase size in the PP/ABS nanocomposites, which attributes to the compatibilization effects of HNT.
Thermogravimetric analysis
Figures 5 and 6 show the TGA curve and DTG of PP/PA66 and PP/PA66/HNT nanocomposites. Table 1 lists the corresponding temperatures of Tonset, Td, and Tmax obtained from the TGA curve and DTG curve. Based on Table 1, one may see that only the Tonset of PP/PA66/HNT(80/20/4) is higher than that of PP/PA66. On the other hands, the Tonset of the PP/PA66/HNT(80/20/2), PP/PA66/HNT(80/20/6), and PP/PA66/HNT(80/20/8) nanocomposites is lower than that of the PP/PA66 blends. Three factors should be considered to explain this phenomenon: (1) the appropriate amount of HNT, (2) the barrier properties and thermal resistance of HNT, and (3) the catalytic effects of HNT due to the presence of hydroxyl groups as Bronsted acid sites. In this study, the low loading of HNT in PP/PA66/HNT(80/20/2) is not sufficient to capture the volatile products effectively at the initial degradation stage, and thus records low Tonset. A significant improvement for the Tonset of the PP/PA66/HNT(80/20/4) nanocomposite is associated with the barrier properties and thermal resistance of the HNT. The tubular structure of HNT entraps the volatile products and reduces the mass transfer to slow down the degradation process. 34 The barrier effects of HNT are more dominant than the catalytic effects in the PP/PA66/HNT(80/20/4) nanocomposites. However, it should be mentioned that further loading of HNT (more than 4 phr) reduced the Tonset of PP/PA66 blend. This is due to the catalytic effect of HNT, which increases the thermal degradation of PP/PA66. According to Wang and Huang, 35 HNT can cause a catalytic effect on the thermal degradation of PP as a result of the presence of hydroxyl groups which played a role as Bronsted acid sites. A similar finding also reported by Luyt and Kelnar 36 where the HNT initiates an early degradation by catalyzing the formation of the free radicals. Note that the Td and Tmax for PP/PA66/HNT nanocomposite are comparable with the PP/PA66 blend.

TGA curves of PP/PA66/HNT nanocomposites.
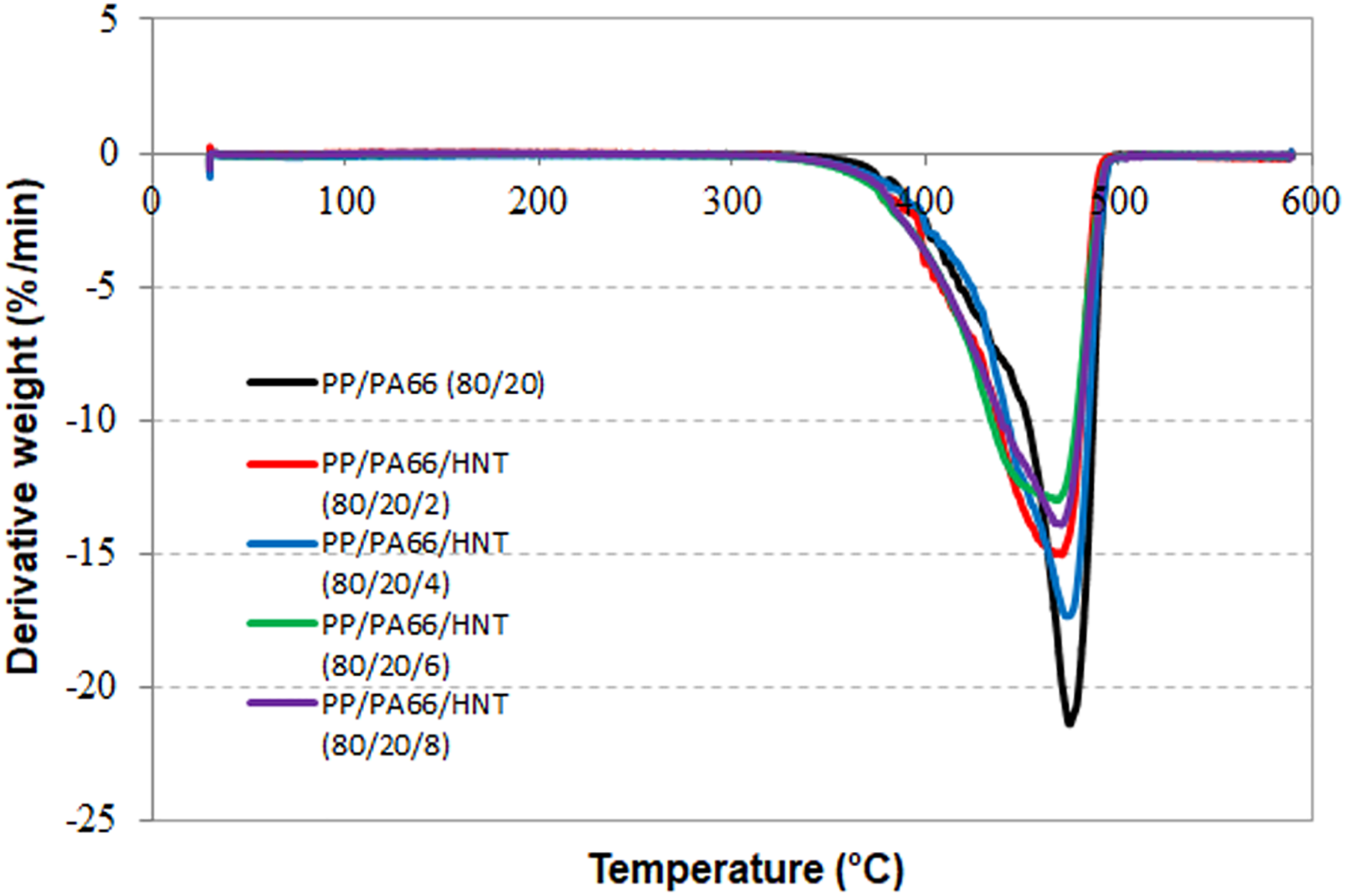
DTG curves of PP/PA66/HNT nanocomposites.
TGA results of PP/PA66/HNT nanocomposites.

TGA: thermogravimetric analysis; PP/PA66: polypropylene/polyamide 66; HNT: halloysite nanotube.
Differential scanning calorimetry
Figures 7 and 8 show the DSC heating and cooling curves of PP/PA66 blend with different HNT loadings. Table 2 summarizes the thermal characteristics of PP/PA66 and PP/PA66/HNT nanocomposites. Based on Table 2, the melting temperature (Tm) and crystallization temperature (Tc) for both PP and PA66 do not change much. However, it can be seen that the crystallinity of PP increased from 36.5% to 48.2% as the HNT loading increased from 2 phr to 8 phr. At the same time, the crystallinity of PA66 also increased from 13.6% to 27.9% as the HNT loading increased from 2 phr to 8 phr. This is due to the fact that the HNT can act as heterogeneous nuclei and induce crystallization for both PP and PA66. The increase of these nuclei promoted the site for the nucleation of the polymer and thus enhanced the crystallinity of both PP and PA66. A similar finding was reported by Prashantha et al. 37 and Prashantha et al. 38 for the PP/HNT nanocomposites and PA6/HNT nanocomposites, respectively. According to Li et al., 39 incorporation of the HNT enhances the crystallization process and leads to perfect crystal formation in PA6/melamine cyanurate composites. Liu et al. 40 reported that the HNT significantly increased numbers of nucleation site by acting as heterogeneous nuclei for PP. The HNT in the form of tubes with small diameter enables the polymer chains to adsorb and align on the surface.

DSC heating curves of PP/PA66/HNT nanocomposites.

DSC cooling curves of PP/PA66/HNT nanocomposites.
DSC characteristics of PP/PA66/HNT nanocomposites.

DSC: differential scanning calorimetry; PP/PA66: polypropylene/polyamide 66; HNT: halloysite nanotube.
From Table 2, it is worth to mention that the χc increment for PA66 is relatively higher than that of PP upon the addition of HNT. It is known that one of the steps involved in heterogeneous nucleation is the adsorption of the polymer on the surface of the nuclei. The effectiveness of this adsorption is associated with the formation of bonding, for example, hydrogen bonds. Since the outer surface of HNT can form hydrogen bonding with PA66, the nucleation for PA66 was relatively higher as the polyamide chains could be easily adsorbed and induced crystal growth as compared to PP. The HNT not only works as a nucleating agent but also caused an enthalpic interaction with amide groups of PA66. This phenomenon causes the formation of the secondary bond that packs the molecular chain tightly, promotes the arrangement orderly, and thus increases the crystallinity of the polymer. 41
Conclusions
Based on the study to investigate the effects of HNT on the flexural and thermal properties of PP/PA66 blends, the following conclusions can be drawn:
The flexural modulus and strength of the PP/PA66 were improved by the incorporation of HNT due to the reinforcing ability and compatibilization effects of HNT.
TGA results suggested that the thermal decomposition of PP/PA66 blends was influenced by the loading of HNT. 4 phr of HNT can improve the thermal stability of PP/PA66 blends due to its heat barrier effects.
DSC results indicated that adding of HNT increased the degree of crystallinity of PP and PA66, where the effects of HNT are more prominent in the PA66 phase due to the additional hydrogen bonding formation between PA66 and HNT. The HNT is a good nucleating agent for the PP/PA66 blends.
The multifunctionality of HNT in PP/PA66 blends was clearly shown by its outstanding performance in flexural properties reinforcement, thermal stability, and crystallization.
Footnotes
Acknowledgement
The authors thankfully acknowledge the financial support provided by the Bridging Fund from Universiti Sains Malaysia (USM), Malaysia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Bridging Fund (304.PBAHAN.6316090) from Universiti Sains Malaysia (USM), Malaysia.

