Abstract
Novel hybrid blend with the composition of m-aramid and sodium ionomers with different weight ratio percentage were prepared by the solution blending technique. The prepared composite films were characterized by the Fourier-transform infrared (FTIR), X-ray diffraction (XRD), scanning electron microscopy (SEM), differential scanning calorimetry, thermogravimetric analysis (TGA), and dynamic mechanical analysis (DMA). The phase structure, compatibility, and morphologies of the composite film were investigated with SEM, DMA, XRD, and transmission electron microscopy (TEM) analysis. The TGA of the composite films shows improvement in the thermal stability. The temperature corresponding to 5% (T 5%) weight losses is in the range of 101–414°C. The storage modulus (in the range of 01.55–3.0 GPa) and glass transition temperature (in the range of 244–276°C) decreased with an increase in the content of sodium ionomers. The tensile strength of the composite films is in the range of 160–185 MPa. This effective approach shows a potential application in the field of m-aramid-based composites.
Introduction
The design flexibility afforded by many thermoplastic compositions and their relatively lightwbeights and corrosion resistances make them attractive materials for the replacement of metal components in many applications. However, thermoplastic compositions often possess an insufficient combination of stiffness, strength, toughness, and/or other physical properties to satisfy the requirements of many of these applications. 1 –4 The polyamides, in particular, would be desirable materials to use in many metal replacement applications as they can exhibit a roughly constant stiffness over a wide and useful temperature range for many applications, including automotive uses. The addition of fibrous reinforcing agents to polyamide compositions can produce materials having good stiffness, but this gain is often at the expense of the toughness of the resulting material. 5 –7 It would thus be desirable to produce polyamide structures having physical properties that rendered them suitable for use as replacements for metal components in various applications. There is a need for the blend composition having better thermal and mechanical properties.
Ionomers are ion-containing polymers typically hydrophobic backbone and a relatively small amount of ionic groups such as sulphonic, carboxylic, and phosphoric acids, where the acid groups are partially or fully neutralized with metal cations. 8 –11 The incorporation of ionomers into the polymer blends modifies greatly their characteristic properties such as stiffness, yield strength, and moisture absorption. The development of new multiphase methods with desired mechanical properties often involves the strategy of blending. 12,13 A majority of polymer blends are thermodynamically immiscible in nature due to the low entropy of mixing. 14 The advantage of the ionomer compared to conventional polymers is that the ionomer can be molded into complex shapes by conventional solution blending methods. This study relates to solvents for ethylene copolymers. In one aspect, it relates to solvent blends for copolymers of ethylene and ethylenically unsaturated carboxylic acid. In another aspect, it relates to solvent blends that will hold such copolymers in solution at a temperature of about 60°C.
Further, such copolymers are characterized, among other things, by their outstanding adhesion to a variety of metallic and nonmetallic substrates, outstanding toughness, and tensile strength. Because of these unique properties, such copolymers find commercial utility, among others, in packaging, building products, communications, and transportation end use of applications. 9,12,15 It has been found that the most important property of such copolymers is their adhesive characteristics. Thus, such copolymers are used widely as adhesive for laminating two or more substrates into a laminate.
Conversely, aramid can be considered to be a tough material at room temperature, and it also possesses good chemical resistance. Subsequently, the blending of sodium ionomers with aramid can be an expected approach for increasing the thermal and impact strengths of sodium ionomers. Sodium ionomer of poly(ethylene-co-methacrylic) acid was carefully chosen such that all other specifications, namely melt index, acid content, and degree of neutralization, are comparable to other ionomers. It is expected that the m-aramid/sodium ionomer fiber composites developed in this work will gain more prominent impact toughness, much lighter weight, better dielectric properties, and higher fatigue resistance compared to conventional inorganic fiber–reinforced thermoplastic ones. Such composites highlight their specific applications for injection molding of the structural components used for automobiles, high-speed trains and airplanes, electrical apparatus, military equipment, and so on. 16 –18
Therefore, the present work investigated the m-aramid in the sodium ionomer matrix and its thermophysical properties, the effect of molecular structure of relatively formed copolymer on compatibilization, and morphological properties of the blends. Furthermore, the miscibility of the blends with different weight ratio was scrutinized by thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), and dynamic mechanical analysis (DMA) measurements. The m-aramid/sodium ionomers with different weight ratio (3:0.25, 3:0.50, 3:0.75, and 3:1) were prepared.
Materials
Sodium ionomers (Surlyn 8150, purchased from DuPont, South Korea); m-aramid (30%) solution from Huvis R&D Centre, Seoul, South Korea; tetrahydrofuran (THF), N-methyl-2-pyrrolidinone (NMP), and dimethylacetamide (DMAc) purchased from Daejung chemicals from South Korea.
Characterization methods
The surface of the films was examined by Fourier-transform infrared (FTIR) (Spectrum 100, PerkinElmer, Waltham, Massachusetts, USA) spectroscopy in attenuated total reflectance (ATR) mode. Germanium (Ge) ATR crystals were used as the internal reflection elements. The film was held against one side of the Ge crystal. The background spectra were obtained with a Ge crystal and nitrogen gas in the absence of the film. After drying at 105°C for 2 h, the FTIR spectra of the films were scanned from 700 cm−1 to 4000 cm−1 for 64 scans at a 2 cm−1 resolution. X-ray diffraction (XRD) (D/Max, Rigaku, Tokyo, Japan) was performed to examine crystal formation under the molecular interaction between sodium ionomer and m-aramid. Cu Kα radiation (wavelength = 1.5418 Å) was used as an incident X-ray source (40 kV, 200 mA), and the films were scanned from 10° to 80° 2θ at a rate of 2° min–1. The thermal behavior of the films was examined by DSC (DSC 2920, TA Instruments, New Castle, Delaware, USA) in the temperature range from 25°C to 300°C under nitrogen purge gas and at a heating rate of 0°C min–1. DMA was performed on the samples of size 20 × 10 × 0.1 mm3 using a dynamic mechanical analyzer (N535-0001, PerkinElmer), which was used for the evaluation of dynamic moduli and mechanical damping (tan δ) under nitrogen purge gas in the temperature range of 10°C to 280°C at a frequency of 1 Hz and a heating rate of 2°C min–1. The tensile strength and elongation at break were assessed in a universal testing machine (Model 3345, Instron, Canton, Massachusetts, USA) at a crosshead speed of 300 mm min–1 according to the KS K 0521:2006 method. The gauge length (distance between jaws) was 200 mm. Ten samples were tested in each case and the average was taken. TGA was carried out using a TA Instruments, SDT Q600 model at a heating rate of 10°C min–1 up to 800°C under N2 atmosphere. Ultraviolet–Visible (UV-Vis) spectrophotometer used in this study is a double beam spectrophotometer (Shimadzu UV-1601, Japan). It has built-in tungsten and deuterium lamps, which provide measurement of optical density in the range of 200–900 nm (near UV and visible regions). The samples were dissolved in triple distilled water for recording UV-Vis spectra. The absorbance was measured with reference to triple distilled water as blank. The change in the absorption spectrum was determined spectrochemically by observing absorbance of chromophore at respective λ max using double beam UV-Vis spectrophotometer with reference solvent in Teflon-stoppered quartz cuvettes on the path length of 1 cm for all optical experiments. For the measurement of moisture uptakes, film specimens were immersed in deionized water at 25°C, and the weight difference was calculated after 1 week. The mean of five specimens per sample was taken as the final values for the water absorption. A transmission electron microscope (H-7600, Hitachi, Tokyo, Japan) was used to observe at the acceleration voltage of 120 kV with a resolution of 0.32 nm.
Preparation of m-aramid/sodium ionomer film
The m-aramid/sodium ionomer film was prepared by the solution blending technique using THF as a solvent. The m-aramid/sodium ionomer composite blends were prepared with a different solid weight ratio (3:0.25, 3:0.5, 3:0.75, and 3:1). For the preparation of the polymer composite blend (m-aramid/sodium ionomer) film, first a 10 g solution of m-aramid was dissolved in a 250 ml three-necked round-bottomed flask equipped with a refluxing condenser and a magnetic stirrer under continuous stirring at room temperature. Then suitable amounts of sodium ionomer were mixed at above 60°C and added slowly THF and NMP into the reaction mixture. The reaction mixture was stirred for 1 h at room temperature and then heated at 90°C for 1 h. Then the viscous solution at 50°C was poured into a flat glass surface and made the film using applicator. All the cast films were initially dried in a dry air-flowing oven at 60°C for 12 h to remove most of the solvent. Followed by curing, the composite film was then heated to temperatures of 100°C and 160°C for 1 h. Finally, the films were cooled to room temperature and peeled from the glass plate to obtain the m-aramid/sodium ionomer composite film. A series of composite films with different m-aramid and sodium contents (i.e. 3:0.25, 3:0.5, 3:0.75, and 3:1) were prepared using the above approach. The preparation of m-aramid/sodium ionomer composite is shown in Figure 1.
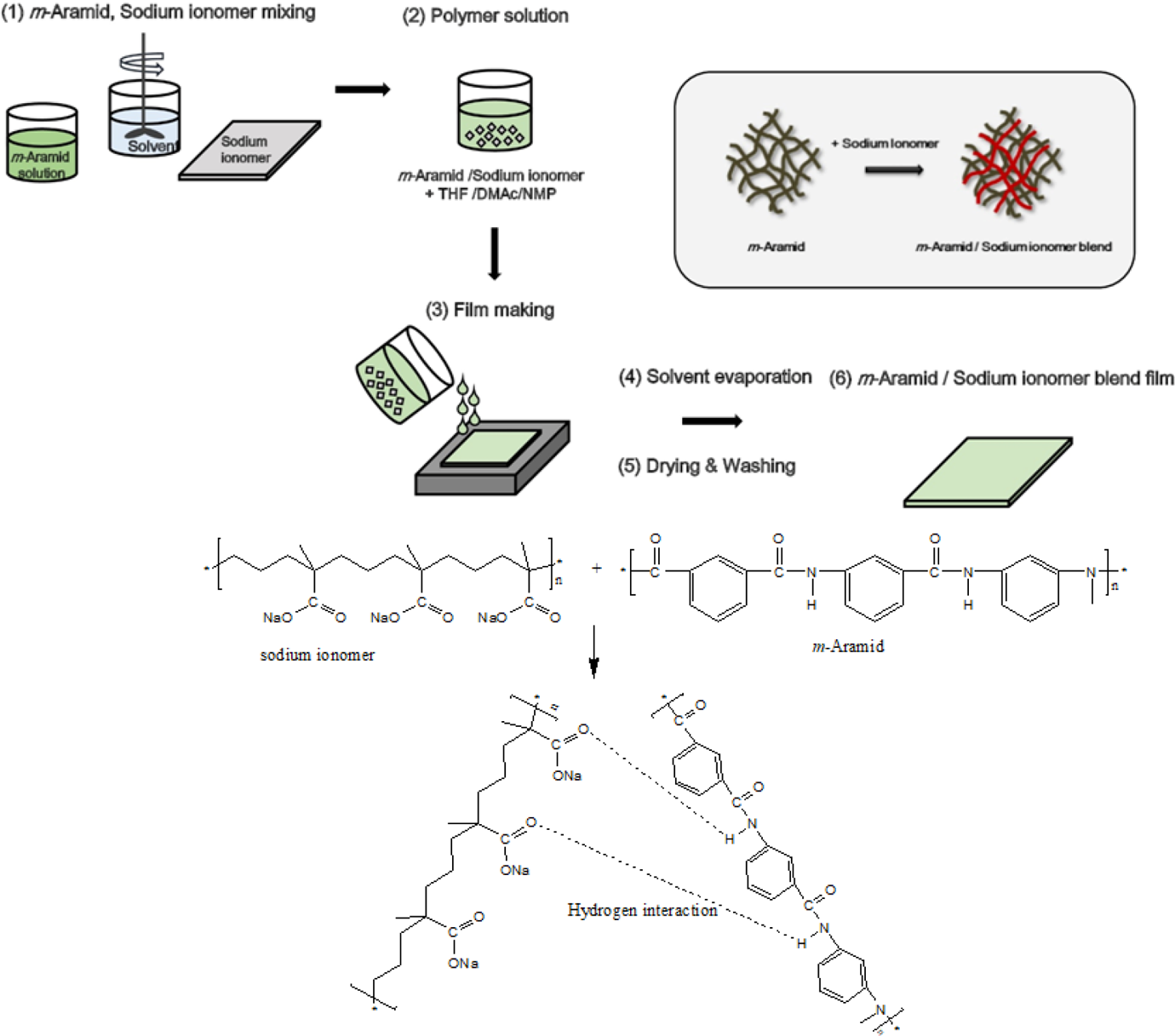
Preparation of m-aramid/sodium ionomer composite film and interaction between the m-aramid and sodium ionomer components.
Result and discussion
FTIR analysis
The FTIR spectra of m-aramid, sodium ionomer, and m-aramid/sodium ionomer composite films are shown in Figure 2. The FT-IR spectra shows,(Figure 2(b)), the characteristic absorption bands at 3290 and 1532 cm–1 were attributed to the stretching and bending vibrations of N–H group, whereas bands at 1651 cm–1 were attributed to the stretching vibrations of C=O bond. The FTIR spectrum of sodium ionomer (Figure 2(a)) shows that the sharp band at 718 cm–1 is due to the methylene rocking in the pendent backbone. The absorbance band at 1254 cm–1 was attributed to the C–O stretching vibration of COOH group in the hydrogen-bonded dimers. The peak with a wavelength of 1700 cm–1 is due to the C=O stretching vibration.
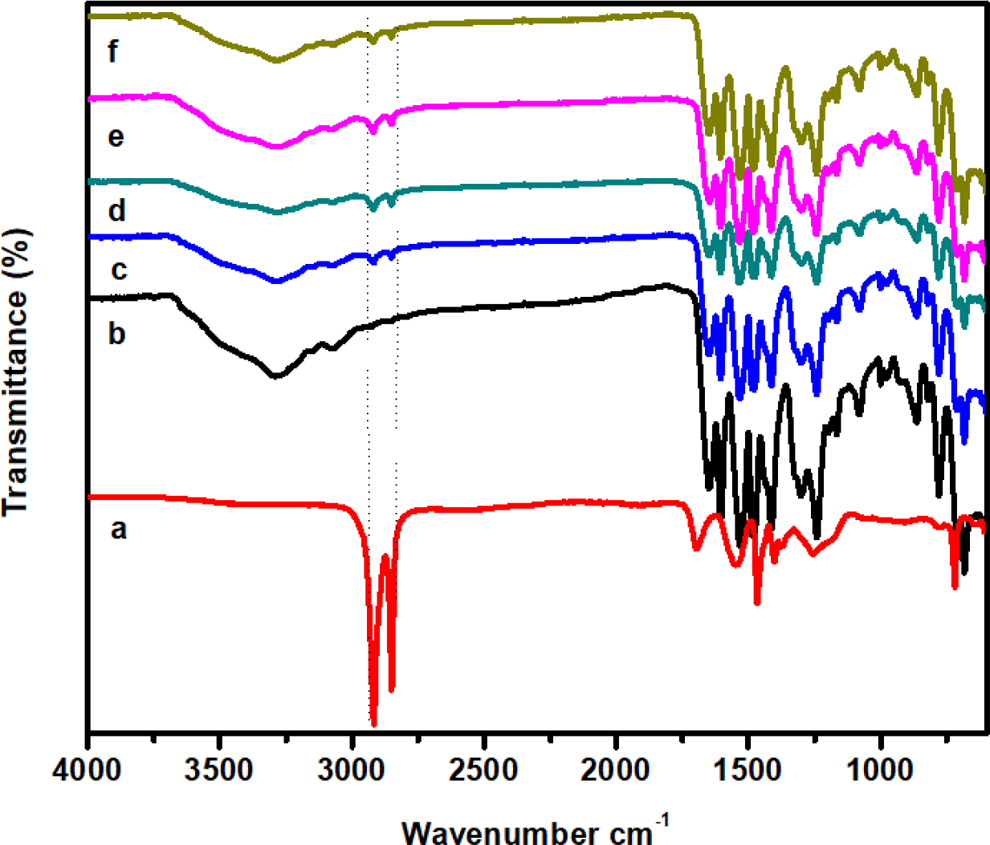
FTIR spectra of (a) sodium ionomer, (b) m-aramid, (c) m-aramid/sodium ionomer 3:0.25, (d) m-aramid/sodium ionomer 3:0.5, (e) m-aramid/sodium ionomer 3:0.75, and (f) m-aramid/sodium ionomer 3:1. FTIR: Fourier-transform infrared.
The absorbance probably results from the combinations involving the C–H stretching bands and the C–H deformation bands. The stretching absorption bands for –CH3 and –CH2 groups at 2917 and 2849 cm–1 respectively. The deformation bands of these groups were observed at 1466 and 1403 cm–1. In the FTIR spectrum of m-aramid/sodium ionomer blend, the major peaks were observed at 2920, 2850, 3210, and 1532 cm–1 for –CH3 groups, –CH2 groups, and N–H groups, respectively. The FTIR spectrum for the m-aramid showed no absorbance band for aliphatic C–H stretching vibration, but m-aramid/sodium ionomer blend spectrum showed both aromatic and aliphatic C–H bands were present, signifying the integration of aramid chains into the composite matrix. This physical interaction shows compatibility among the blend components, which improved mechanical and thermal properties. 2
X-ray diffraction
The XRD patterns of m-aramid, sodium ionomer, and m-aramid/sodium ionomer composite film are shown in Figure 3. For the sodium ionomer, the characteristic sharp diffraction peaks were found at diffraction angle (2θ) of 20.11 (correspond to interlayer spacing of about 4.4 Å) and the diffraction peaks of the m-aramid located at 24.03 (interlayer spacing of about 3.7 Å). The composite film, the m-aramid/sodium ionomer had a small and weak diffraction peak at 2θ = 21.97, as shown in Figure 3. The disappearance of the characteristic sodium ionomer peaks indicates that the intercalation of sodium interfaces m-aramid interlayers during the fabrication. The pattern of m-aramid and its composite film reinforced by sodium ionomer are similar to that of the unfilled aramid film, but the intensity of peaks becomes stronger than that of the unfilled one. The addition of sodium ionomer improves the crystallization of aramid film, resulting in a high degree of crystallinity. 19
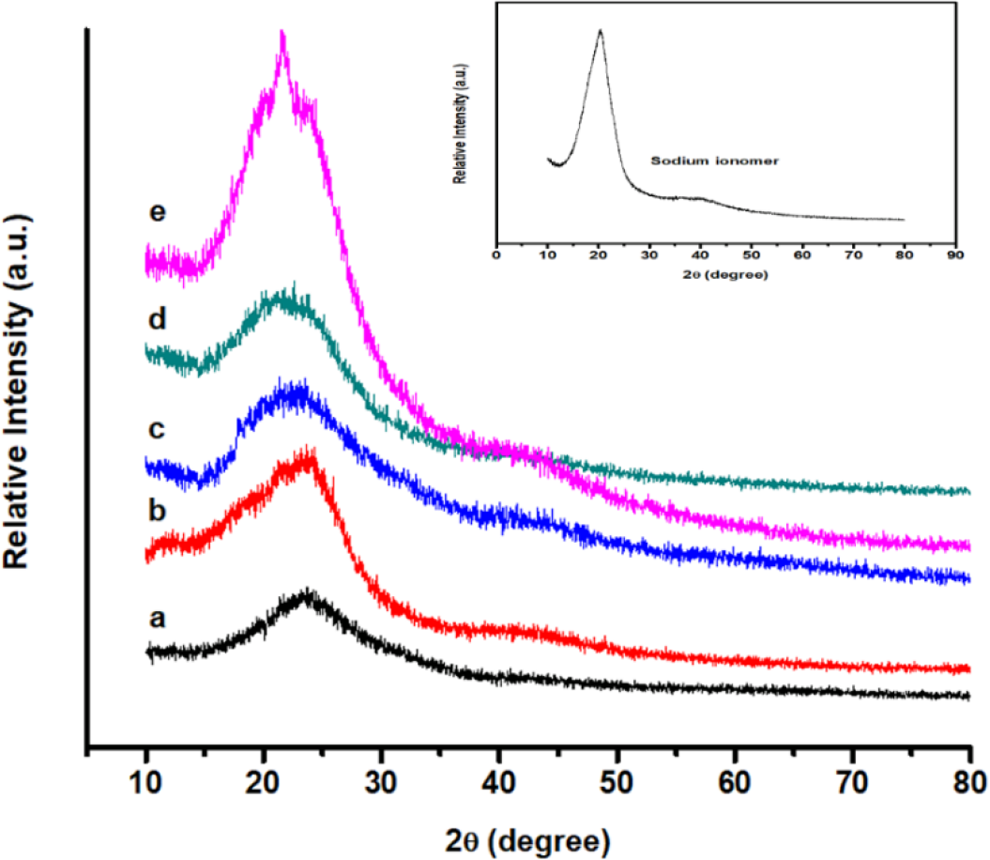
X-ray powder diffractogram of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1.
Thermal properties
The typical DSC thermogram of the blend composite and the neat polymer and the peak temperature along with associated with glass transition temperature (T g) values are summarized in Figure 4 and Table 1. The characterization of T g is widely used to study the polymer blend miscibility. In this blend system, a single T g was exhibited corresponding to the cooperative segmental motion of the molecular chains. The T g for the composite films shown in figure 4 that with single glass transition is termed as the miscible or single phase system. This is attributed to the strong intermolecular between N–H moiety (m-aramid) and carbonyl group (sodium ionomers). This physical interaction makes the miscible composite blends of m-aramid/sodium ionomers. The T g of the m-aramid and the m-aramid/sodium ionomer blends was in the range of 273–241°C. The portion of the sodium ionomer increased in the composite film, and T g decreased or shifted to lower temperature. This is due to the internal friction of m-aramid molecule reduced. The lower T g of m-aramid/sodium ionomers film has greater low-temperature flexibility and it improves the processability. The polymer blends containing a crystallizable material have attracted applied research areas. The majority of commercially available thermoplastic blends and alloys contain at least one crystallizable material. In amorphous/crystalline polymer blends, the crystallization behavior is often strongly influenced by the amorphous component. The crystallization rate of the polymer is reduced by the amorphous polymers. This increases in the crystallization rate may be caused partially by a lowering of the T g in the system. This may be attributed to dissociation of the aramid–aramid interactions such as strong hydrogen bonds.
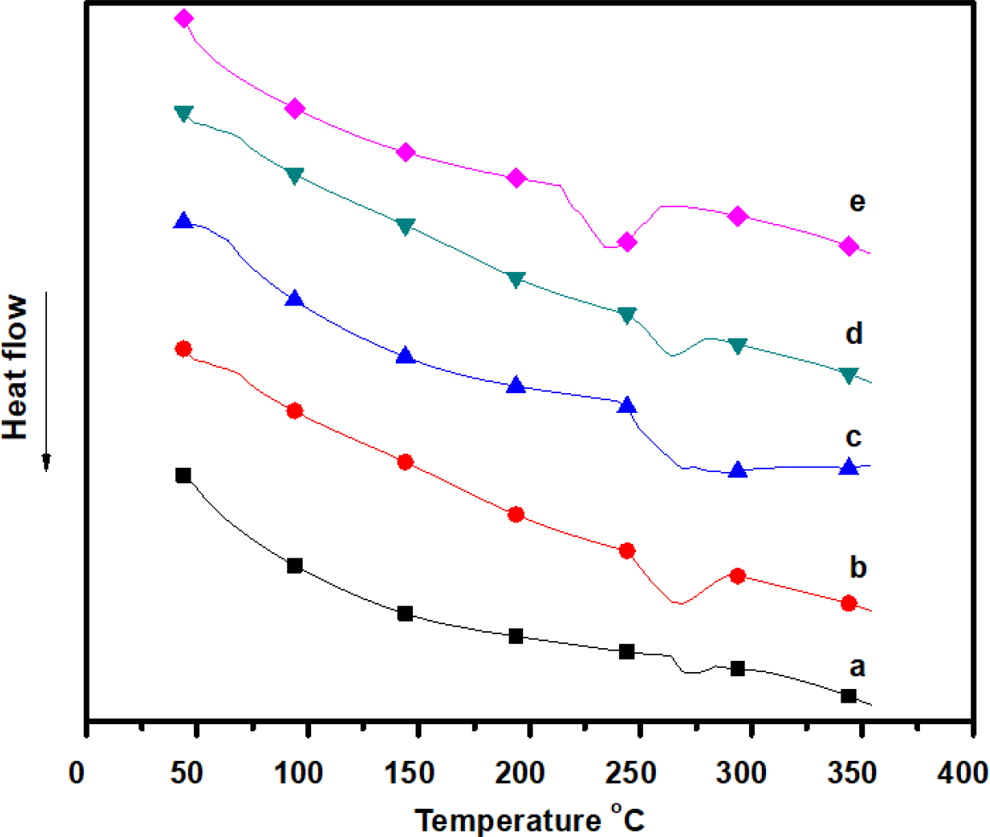
DSC thermogram of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1. DSC: differential scanning calorimetry.
Thermal, water absorption, and mechanical properties of m-aramid/sodium ionomer composites.
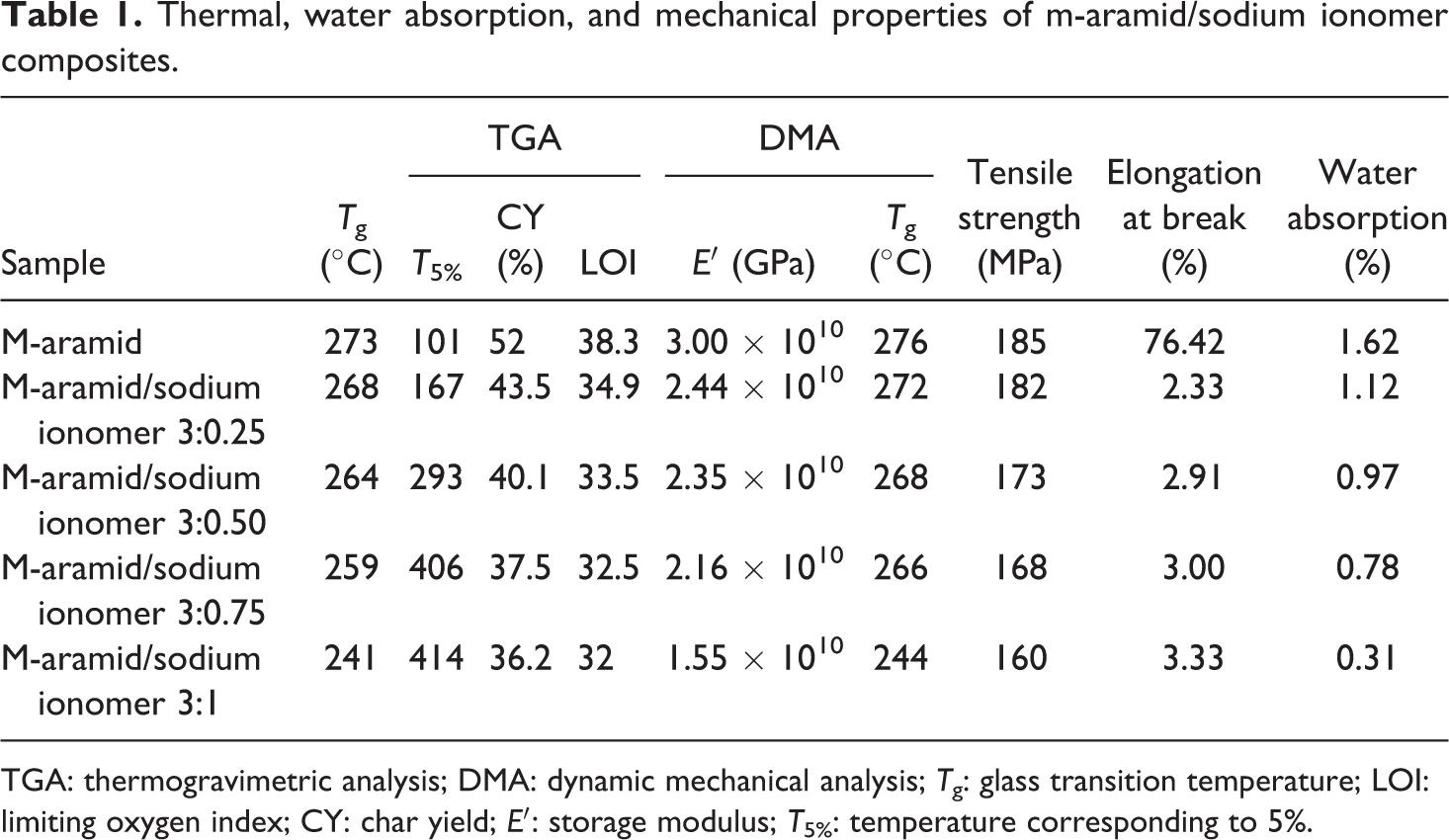
TGA: thermogravimetric analysis; DMA: dynamic mechanical analysis; T g: glass transition temperature; LOI: limiting oxygen index; CY: char yield; E′: storage modulus; T 5%: temperature corresponding to 5%.
TGA was employed to investigate the thermal stability of the m-aramid/sodium ionomer composite films. Figure 5 shows the TGA curve of m-aramid/sodium ionomer under N2 atmosphere. As described in Table 1, the 5% and 10% weight loss temperatures (T 5% and T 10%) are in the range of 101–414°C and 406–445°C, respectively. The thermal stability of the composite films was effectively improved by the combination of m-aramid/sodium ionomer. The char yield of the composite film is in the range of 36.2–52%. The combination of m-aramid and sodium ionomers greatly stabilized the polymer intermolecular interaction.
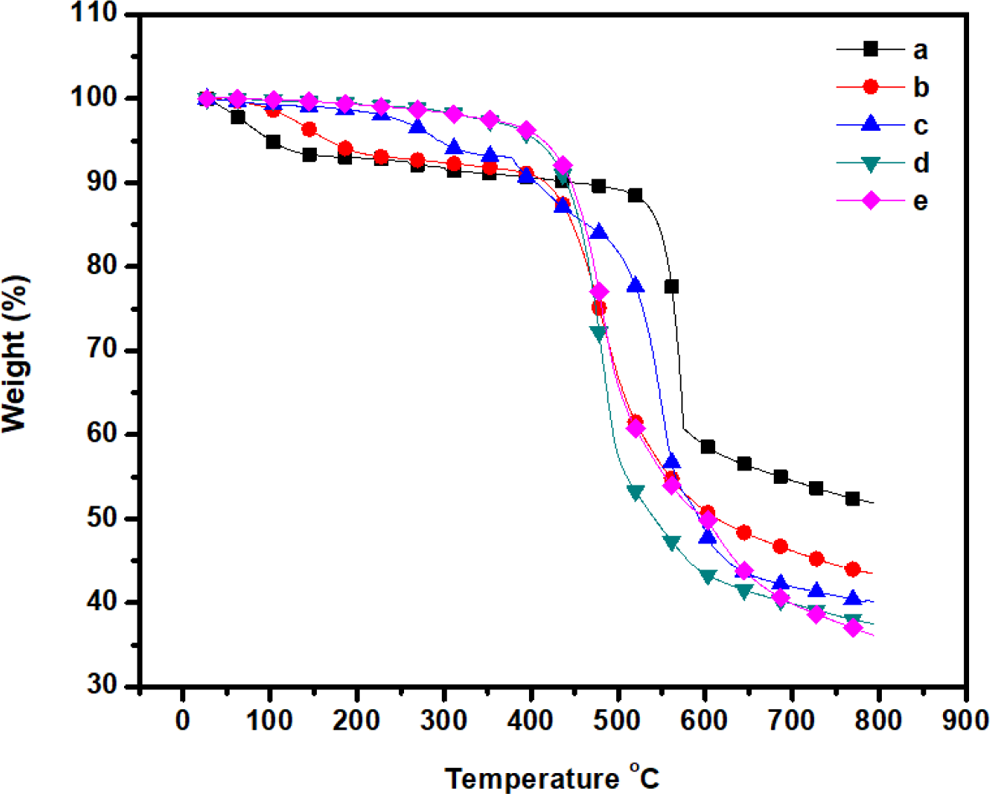
TGA thermogram of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1. TGA: thermogravimetric analysis.
The limiting oxygen index (LOI) value indicates the flame retardant property of the composite film (Table 1). The flame retardancy of material seems to be closely concerned with the maximum decomposition rate of polymer matrix but less relative to the thermal stability of the materials. 20 The char yield of the materials can be used to calculate the LOI values from the TGA analysis using the Van Krevelen and Hoftyzer equation 21 as shown below
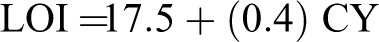
where LOI is the limiting oxygen index and CY is the char yield.
The LOI values of the films are in the range of 32–38.3%, indicating high flame retardancy. The LOI values of the polymers should be above the threshold value of 26% to render them self-extinguishing and for their qualification for many applications requiring good flame retardancy. The prepared composite films show LOI leading to good flame retardancy.
Dynamic mechanical analysis
The dynamic mechanical properties of the m-aramid/sodium ionomers composite film with different weight percentage are shown in Figures 6 and 7 where the storage modulus and tan δ were plotted versus temperature dependencies and the data are summarized in Table 1. The storage modulus (E′) values are in the range of 1.55 × 1010 to 3.0 × 1010 GPa. The modulus observed in the binary blends with sodium ionomers was reduced in all weight ratios. The incorporation of the sodium ionomers into the composite film increased the storage at the expense of severe decrease in the ductility of the blends. The blends of sodium ionomers show strong interaction with m-aramid which results in the miscibility of both materials at certain composition ranges. The interaction strength decreases when the weight percentage of sodium ionomers content increases. However, theses blends exhibited brittle fracture. In the higher weight percentage 3:1 m-aramid/sodium ionomer composite films, the mechanical behavior changed with the ionomer in the blends. The brittle fracture in the binary blend was converted into more ductile fracture. The ionomer incorporation was not as effective as in the blend, even though the mechanical behavior corresponded to a ductile material. With addition, the modulus of the films showed slight variation in the 3:0.25, 3:0.50, and 3:0.75 m-aramid/sodium ionomer blends. But in the 3:1 ratio blend, the storage modulus decreased from 3.0 × 1010 to 1.55 × 1010 GPa compared to neat m-aramid. This reduction was caused by the low value of the ionomer modulus and the concentration of the ionomer.
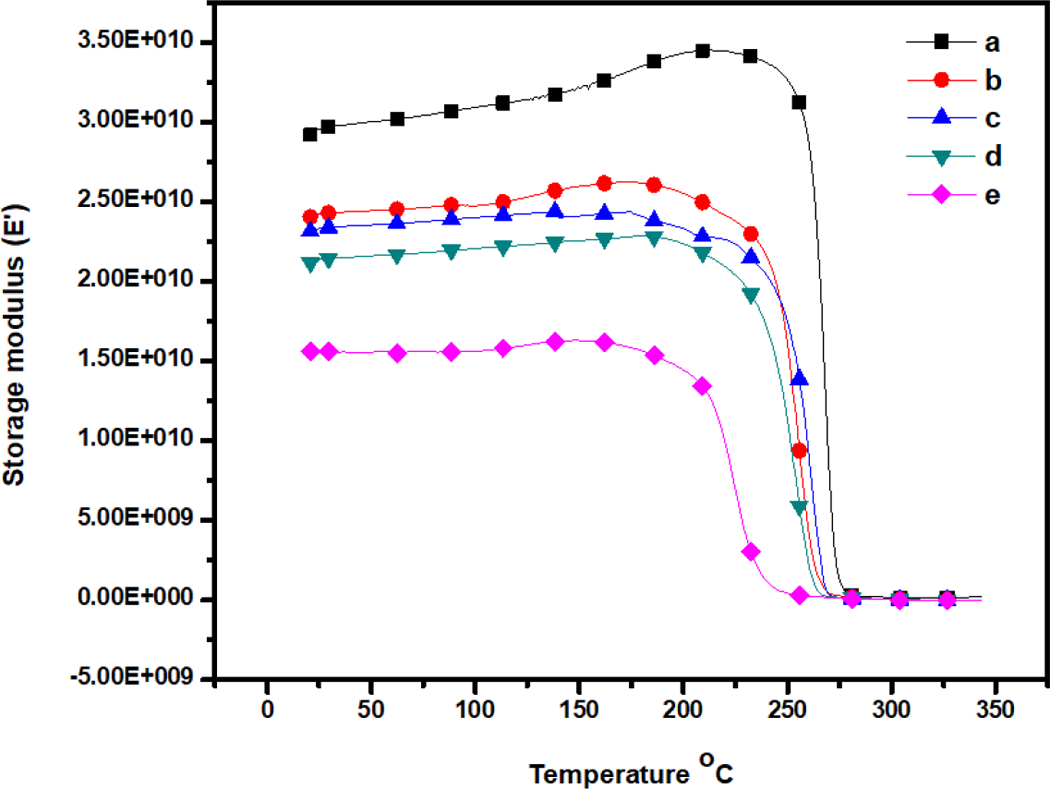
DMA curves show the plot of storage modulus (E′) versus temperature of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1. DMA: dynamic mechanical analysis.
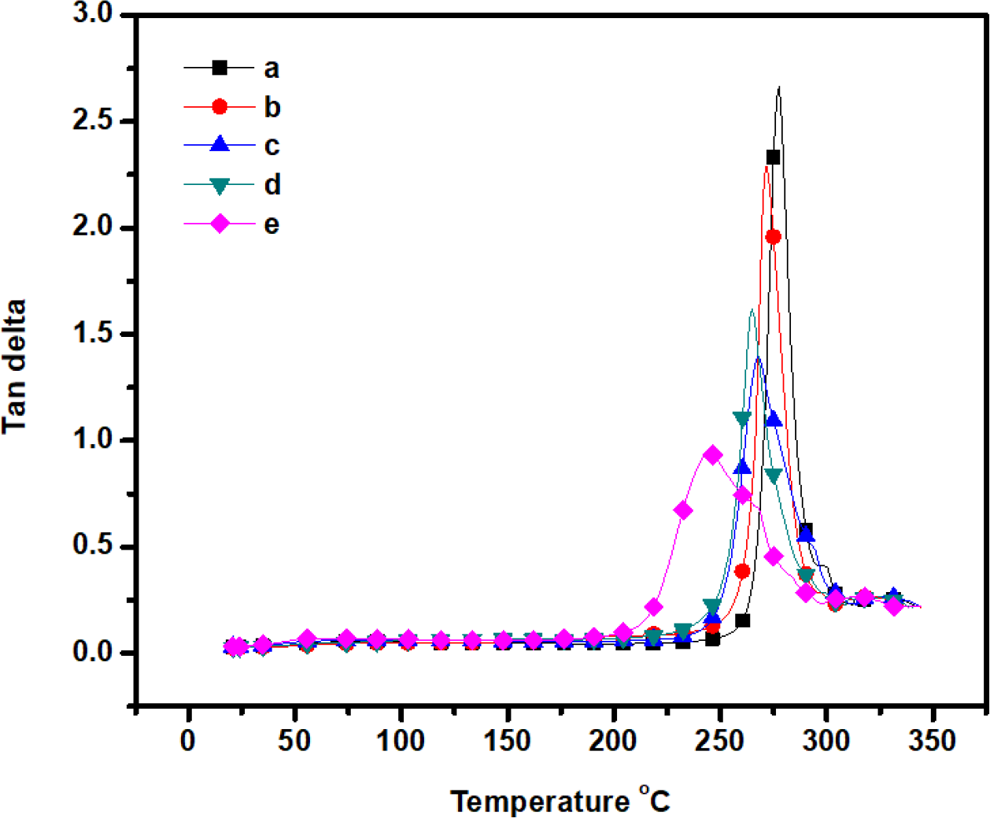
DMA curves show the plot of loss modulus versus temperature of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1. DMA: dynamic mechanical analysis.
Tan δ (loss factor/damping ration) is defined as the ratio of the loss modulus to the storage modulus. It is the ratio of the energy dissipated to the energy stored during a dynamic loading cycle. The contribution to damping in a composite is due to the nature of the matrix or fiber materials. 22 The T g of the m-aramid and composites films was in the range of 244–276°C. The T g decreases with an increase in the content of sodium ionomers in the composite. The sodium ionomers reduce the cohesiveness of the matrix and stiffness of the composite. This indicates that the composites underwent non-covalent interaction between m-aramid and sodium ionomer phases resulting in good mechanical properties. 23 A decline in the tan δ values with the addition of sodium ionomer indicates an improvement in interfacial bonding in composites because the higher the damping at the interface, the poorer the interface adhesion.
Water absorption
In this study to observe the effects of differing moisture content on the properties of the m-aramid/sodium ionomer composites. The difference in moisture content has been achieved by the wet conditioning method. The rate of water absorption depends on the internal material states, nature of fiber constituents, fiber–matrix interface, as well as environment factors like temperature and applied stress. 24 The percentage of weight change for the specimen was determined using the following equation
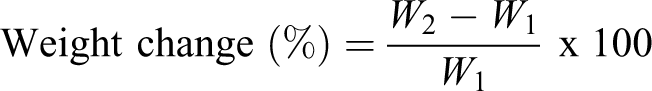
where W 1 and W 2 are the weight (in grams) of the specimen before and after immersion in water. The aramid contains the polar –CO-NH groups, which have a tendency to absorb water through hydrogen bonding. The results show that the maximum water absorption for the pure aramid (1.62%) and m-aramid/sodium ionomer composite decreased (1.12% to 0.31%). The diffusion of water molecule into the matrix can take places by capillary action causing the fiber to swell and create more voids. The hydrophobic nature of the sodium ionomer present in the composite, the extent of water absorption at saturation point considerably decreased. The enhanced barrier characteristic, chemical resistance, reduced solvent uptake and flame retardancy of m-aramid/sodium ionomer composites benefit from the hindered diffusion pathway through the composite films.
Optical properties
The optical transmittance parameter of the composite films is a crucial factor for their use in optical applications. It depends on their structures and external factors that affect the colors of the composite films, including purity of the polymers’ residual solvent and film-forming conditions. 25 Figure 8 shows that the optical properties of the m-aramid and the m-aramid/sodium ionomer composite films were investigated by UV-Vis spectroscopy (0.05 mm thickness). The results are summarized in Table1. The composite films have a cutoff wavelength of 900 nm. The pure m-aramid film exhibits good transparency over 800 nm with the transmittance higher than 80%. The transmittance of the 3:0.25 ratio film at 600 nm is 77%, while that of 3:0.50, 3:0.75, and 3:1 ratio is 66%, 60%, and 55%, respectively. The maximum transmittance was found for the hybrid film containing 3:0.25 weight ratio of m-aramid/sodium ionomer content in the matrix, beyond which it decreased gradually. The transparency of these composite films depends upon the size and spatial distribution of ionomers in the polymer matrix. Ultimately, the tendency for the larger once may increase, which decreases the homogeneity of the system. As particle size becomes larger, the transmittance value decreases. 26
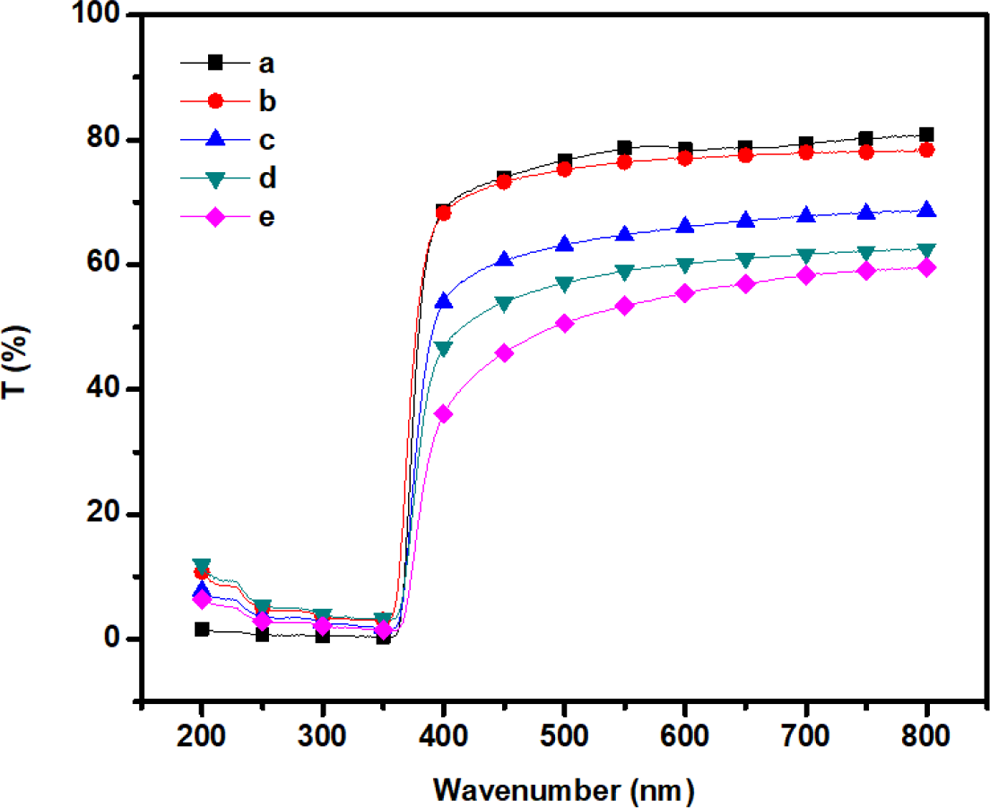
UV transmittance spectra of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, and (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1. UV: ultraviolet.
Morphology
In order to investigate the morphology, micro-structure of the m-aramid/sodium ionomer composite films was studied using SEM and TEM. The SEM micrograph images of the m-aramid/sodium ionomer composite film with different weight percentage are shown in Figure 9. The blend system exhibited self-assembled nano-phase morphologies ranging from finely dispersed granular matrix to well-patterned knitted nano-fibrillar network. There are many factors to affect the structure and morphology such as glass transition temperature, crystallinity as well as specific interaction between the blend materials. But the morphology of this composite film appears uniform, self-assembled surface, no aggregated particles, and homogenously dispersed. In particulary the m-aramid/sodium ionomer weight ratio in 3:0.50, the two phases merged completely forming a fine continuous pattern. The fine dispersion of polymers with different weight ratio can be clearly understood from the SEM images. The strength of intermolecular interaction to some extent depends on the composition. Figure 10 shows the TEM images of the m-aramid/sodium ionomers composite films with different weight ratio. Both components are well dispersed in the matrix, and slight agglomerates are found. The TEM images confirmed the good interaction between the matrices and showed consistent results with those given by XRD and SEM.

SEM images of (a) m-aramid, (b) m-aramid/sodium ionomer 3:0.25, (c) m-aramid/sodium ionomer 3:0.5, (d) m-aramid/sodium ionomer 3:0.75, and (e) m-aramid/sodium ionomer 3:1. SEM: scanning electron microscopy.
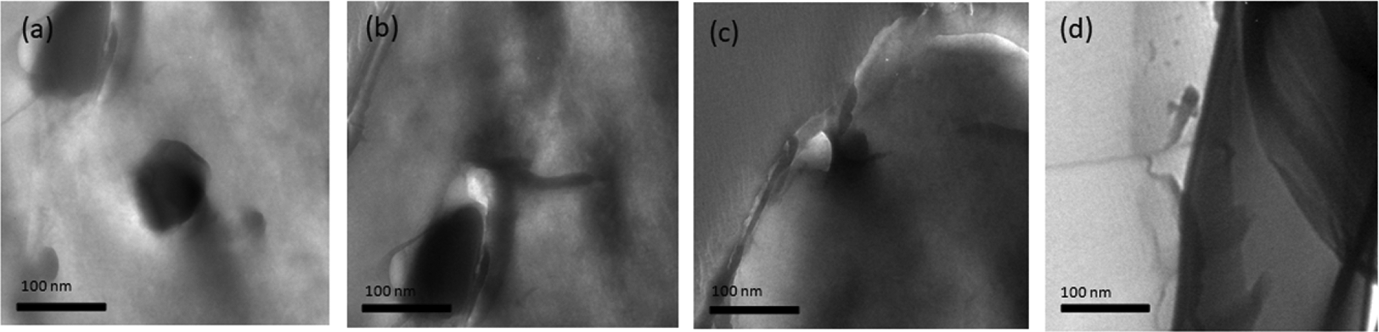
TEM images of (a) m-aramid/sodium ionomer 3:0.25, (b) m-aramid/sodium ionomer 3:0.5, (c) m-aramid/sodium ionomer 3:0.75, and (d) m-aramid/sodium ionomer 3:1. TEM: transmission electron microscopy.
Mechanical properties
The tensile test results of the m-aramid and composite films with different ratio are summarized in Table 1. The tensile strength and elongation at break of neat m-aramid film is higher than the m-aramid/sodium ionomer composite films. With increasing weight percentage of sodium ionomers in the composite film, the tensile strength and elongation at break slightly decreased. The ultimate stress at break for composite films increased with increasing sodium ionomer content. The elongation at break of the composite films increased from 2.33% to 3.33% with weight of 3:0.25 to 3:1 of m-aramid/sodium ionomer. The maximum strength of aramid was 185 MPa which decreased up to 160 MPa with 3:1 weight ratio of m-aramid/sodium ionomer. The addition of sodium ionomer to the aramid matrix resulted in the deterioration of mechanical properties. The property of the fiber-reinforced films relies on the balance of processing ability and end properties.
Conclusion
In this study, several polymer blends with different weight ratios of m-aramid/sodium ionomer composite film were prepared by the solution blending technique. The physical interaction of composites was confirmed by FTIR analysis. The entire investigation established that physical interaction contributed to the enhanced compatibility of m-aramid/sodium ionomer binary blends, thus improving the morphologies and thermal properties. The composites exhibit high thermal stability (T 5%) 101–414°C and further suggest that the composite films can be used for flame retardant application as shown by their LOI values. SEM and TEM images confirmed that the two matrices are well dispersed and compatible with each other. The better compatibility of film can be explained by the DMA analysis, compared with that of pure m-aramid, the Tg of the composite film decreases to lower temperature. This study indicated the feasibility of producing advanced thermoplastic laminates with enhanced thermoplastic laminates with enhanced thermal stability, mechanical properties and clearly shows the potential of conventional functional polymers to obtain multifunctional properties for an expected the material useful in the field composite polymers. Thermoformability characteristics of the films and barrier properties as the function of relative humidity are under study to assess whether these blends can become an advantage in the film-packaging applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Technology Innovation Program (or industrial strategic technology development program (10076409, establishment of solution for optical/light design/dynamic concept hybrid 2D/3D thermoplastic prepreg for design performance) funded by the Ministry of Trade, Industry & Energy (MOTIE Korea). This research was also partially supported by Yeungnam University in the form of research grant in 2017 and the authors thank Prof. Taehwan Oh for supporting the tensile analysis.
