Abstract
A series of binary composite blends of m-aramid/zinc ionomer (m-Ar/ZnI) with different weight ratio were prepared by the solution blend technique. These composite films were characterized in terms of the structure, crystallinity, morphology, thermal, and mechanical properties. The temperature corresponding to 5% (T5%) weight loss are in the range of 101–438°C. The composite films showed good storage modulus (in the range of 2.1–3.1 GPa) and high glass transition temperature (Tg) (in the range of 254–278°C) and exhibited good tensile strength but it decreased with an increase in the content of ZnI. The strong compatibilizing effect was observed between the m-Ar and ZnI components, where exceptionally self-assembled morphology was formed in the m-Ar/ZnI composite films. The most favorable mechanical and thermal data supported by the finest structure were observed with 3:0.50 weight ratio. This study evaluates the efficiency of ionomer in m-Ar/ZnI composite blends.
Introduction
The polymer blends are among the most investigated in the area of polymer science based on both theoretical and practical points of view because of the technical advantages of obtaining specific physical properties and cost-effective route to prepare without the need to synthesize new specialized polymer systems. 1 There are increasing usage of thermoplastic materials to replace thermosets for fabrication of laminates, because of the advantages such as high toughness, durability, low density, low cost, ease of processing, corrosion resistance, and design freedom. 2 The concept of physical mixing of two or more polymers to obtain new materials is an attractive and powerful route to design with new properties in user-tailored materials from available polymers. This approach has a number of advantages over the tedious synthesizing of various copolymers or block copolymers. There is no additional chemical synthesis required. 3 Through various physical interactions like hydrogen bonding, dipole–dipole forces, or charge transfer complexes for homo polymer mixtures in the blends are organized supramolecular structure by the blend components.
Aromatic polyamides have been useful as high performance materials because of their high thermal stability, specific strength, high degree of stiffness, and low densities. 4 –8 There is not much attention has been paid to the blending of the aromatic polyamides because upon heating they decomposed rather than melted. This discouraged their melt blending with other polymers. To overcome their problem, we have blended the m-aramid (m-Ar) with zinc ionomer (ZnI) using solution blending technique. 9 –13
Ionomers are ion-containing polymers typically hydrophobic backbones and a relatively small amount of ionic groups such as carboxylic, sulfonic, and phosphoric acid, where the acid groups are partially or fully neutralized with metal cations. Extensive research on the microstructure of ionomers has been done over the three decades and it has been shown that ionic groups tend to form microphase-separated ionic domains, which are crucial for the unique physiochemical properties of ionomers. Commercially available ionomers include Surlyn 8150 and Surlyn 9910 which are copolymers of ethylene and sodium and zinc acrylate. Ionomers can carry out specific interaction with polyamides. However, the polymer blends are most often immiscible because of the small combinatorial entropy of mixing for high molecular weight compounds; the miscibility of polyamides and ionomers can be achieved by this inter molecular exothermic interaction. These specific interactions between the polymers and ionomers which leads to effective strategies in the area of high performance composite blends. 14 –20 In this study, polymethacrylic ionomers are used as a copolymer of methyl methacrylate and zinc methacrlate.
In this study, we prepare the m-Ar/ZnI blends by solution blending method and investigate the influence of the polymethacrylic ionomers in the degree of the order of hydrogen bonding with m-Ar. The interaction between m-Ar/ZnI was studied with Fourier transform infrared (FTIR), differential scanning calorimetry (DSC), and dynamic mechanical analysis (DMA), respectively. The thermal stability and morphological properties were also studied by thermogravimetric analysis (TGA), X-ray diffraction (XRD), and scanning electron microscope (SEM) analysis.
Materials
ZnIs (Surlyn 9910 from Dupont, South Korea), m-Ar (30%) solution was purchased from Huvis R&D Centre, Seoul, South Korea. Tetrahydrofuran (THF), N-methyl-2-pyrrolidinone (NMP), dimethylacetamide (DMAc) were purchased from Daejung chemicals (South Korea).
Characterization methods
The FTIR spectra of the films were examined using FTIR (Spectrum 100, PerkinElmer, Waltham, Massachusetts, USA) spectroscopy in attenuated total reflectance (ATR) mode. Germanium (Ge) ATR crystals were used as the internal reflection elements. The film was held against one side of the Ge crystal. The X-ray diffraction patterns (D/Max, Rigaku, Tokyo, Japan) was performed to examine crystal formation under the molecular interaction between PAN and m-Ar. Cu Kα radiation (wavelength = 1.5418 Å) was used as an incident X-ray source (40 kV, 200 mA), and the films were scanned from 10° to 80° 2θ at a rate of 2° min−1. DSC was performed with TA Instruments, New Castle, Delaware, USA, in the temperature range from 25°C to 300°C under nitrogen (N2) purge gas and at a heating rate of 5°C min−1. DMA was performed on the samples of size 20 × 10 × 0.1 mm3 using a dynamic mechanical analyzer (N535−0001, PerkinElmer), which was used for the evaluation of dynamic moduli and mechanical damping (tan δ) under N2 purge gas in a temperature range of 10°C to 280°C at a frequency of 1 Hz and heating rate of 2°C min−1. The mechanical properties of the film were assessed in a universal testing machine (Model 3345, Instron, Canton, Massachusetts, USA) at a cross head speed of 300 mm min−1 according to the KS K 0521: 2006 method. The gauge length (distance between jaws) was 200 mm. The tensile property reported here represents an average value of at least five measurements of each sample. TGA was carried out using a TA Instruments, SDT Q600 model at a heating rate of 10°C min−1 up to 800°C under N2 atmosphere. Ultraviolet–visible (UV-vis) spectrophotometer used in this study is a double beam spectrophotometer (Shimadzu UV-1601, Japan). The samples were dissolved in triple distilled water for recording UV-vis spectra. The absorbance was measured with reference to triple distilled water as blank. The change in absorption spectrum was determined spectrochemically by observing absorbance of chromophore at respective λ max using double beam UV-vis spectrophotometer with reference solvent in Teflon stoppered quartz cuvettes on the path length of 1 cm for all optical experiments. For the measurement of moisture uptakes, film specimens were immersed in deionized water at 25°C, and the weight difference was calculated after 1 week. The mean of five specimens per sample was taken as the final value for the water absorption.
Preparation of m-Ar/sodium ionomer film
The m-Ar/ZnI film was prepared by the solution blending technique using THF as a solvent. The m-Ar/ZnI composite blends were prepared with a different solid weight ratio (3:0.25, 3:0.5, 3:0.75, 3:1). For the preparation of the m-Ar/ZnI composite films, first a 10-g solution of m-Ar was added in the 250 ml three-necked round-bottomed flask equipped with a refluxing condenser and a magnetic stirrer under continuous stirring at room temperature. Then the suitable amounts of ZnI were mixed at above 60°C and THF, NMP, and DMAc solvents are added slowly into the reaction mixture. The reaction mixture was stirred for 1 h at room temperature and then heated at 90°C for 1 h. Then the solution was cooled to 60°C and the resultant viscous solution poured into a flat glass surface and make the film using applicator. All the cast films were initially dried in a dry air-flowing oven at 60°C for 12 h to remove most of the solvent. Followed by curing the composite film was then heated to temperatures at 100°C and 160°C for 1 h. Finally, the films were cooled to room temperature and peeled from the glass plate to obtain the m-Ar/sodium ionomer composite film. A series of composite films with different m-Ar and ZnI contents (i.e. 3:0.25, 3:0.5, 3:0.75, and 3:1) was prepared using the above approach. The preparation of m-Ar/ZnI composite is presented in Figure 1.
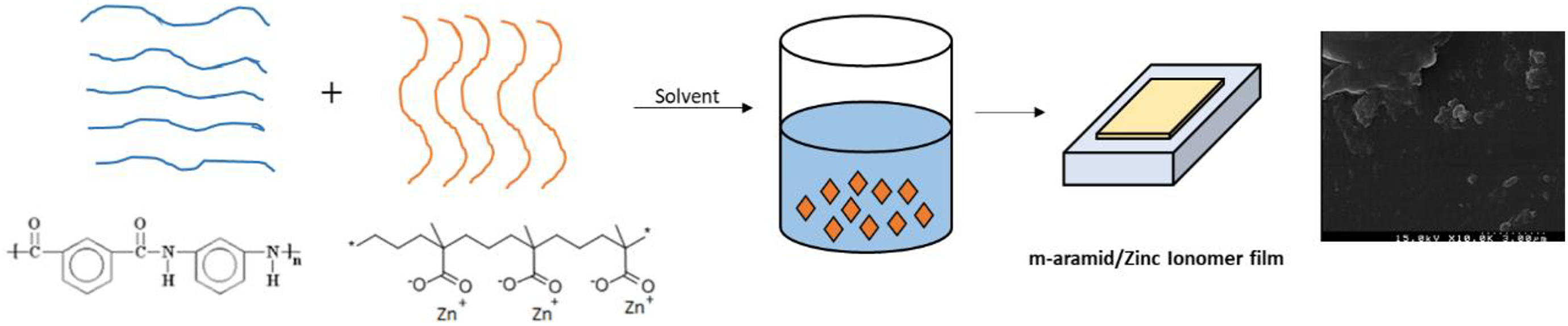
Preparation of m-Ar/ZnI composite film.
Result and discussion
The FTIR spectra of m-Ar, ZnIs, and m-Ar/ZnI composite films are shown in Figure 2. Figure 2(a) shows the FTIR spectra of ZnIs. Figure 2(b) shows the FTIR spectra of m-Ar. The strong and intense band at 1466 cm−1 corresponds to the –CH2– bending and a weak band at 1372 cm−1 which indicates –CH2– wagging present ZnI. The ZnI also exhibit a broad absorption peak around 1536 cm−1 in the asymmetric carboxylate stretching region. This broad band is associated with the coupled asymmetric carboxylate vibration of zinc carboxylate ions present in the neat ionomers. The C=O stretching vibration is typical of the ester/acid carbonyl groups at 1700 and region below 1600 cm−1 presents some difference in the profile pattern of the absorptions. The carbonyl absorption region presents the ester/acid absorptions at 1720 cm−1 and the asymmetric carboxylate stretching band near 1585 cm−1. Figure 2(b) shows the FTIR spectra of m-Ar. In Figure 2(b), the band at about 3310 and 1532 cm−1 indicates a hydrogen bonded N−H stretching and bending vibration. The stretching frequency of C=O for m-Ar appeared at 1650 cm−1. The FTIR spectra of the m-Ar/ZnIs are shown in Figure 2(c) to (f). The major absorption band observed at 3291, 1532, 2928, and 2855 for NH2 groups and –CH3– and –CH2– groups, respectively. Both the aliphatic and aromatic –CH– bands observed m-Ar/ZnI composite film, signifying the integration of m-Ar chains into the m-Ar/ZnI composite matrix. This physical interaction shows compatibility between the two components, which improves the thermal and mechanical properties. The FTIR spectra confirm the structure of m-Ar/ZnI film.
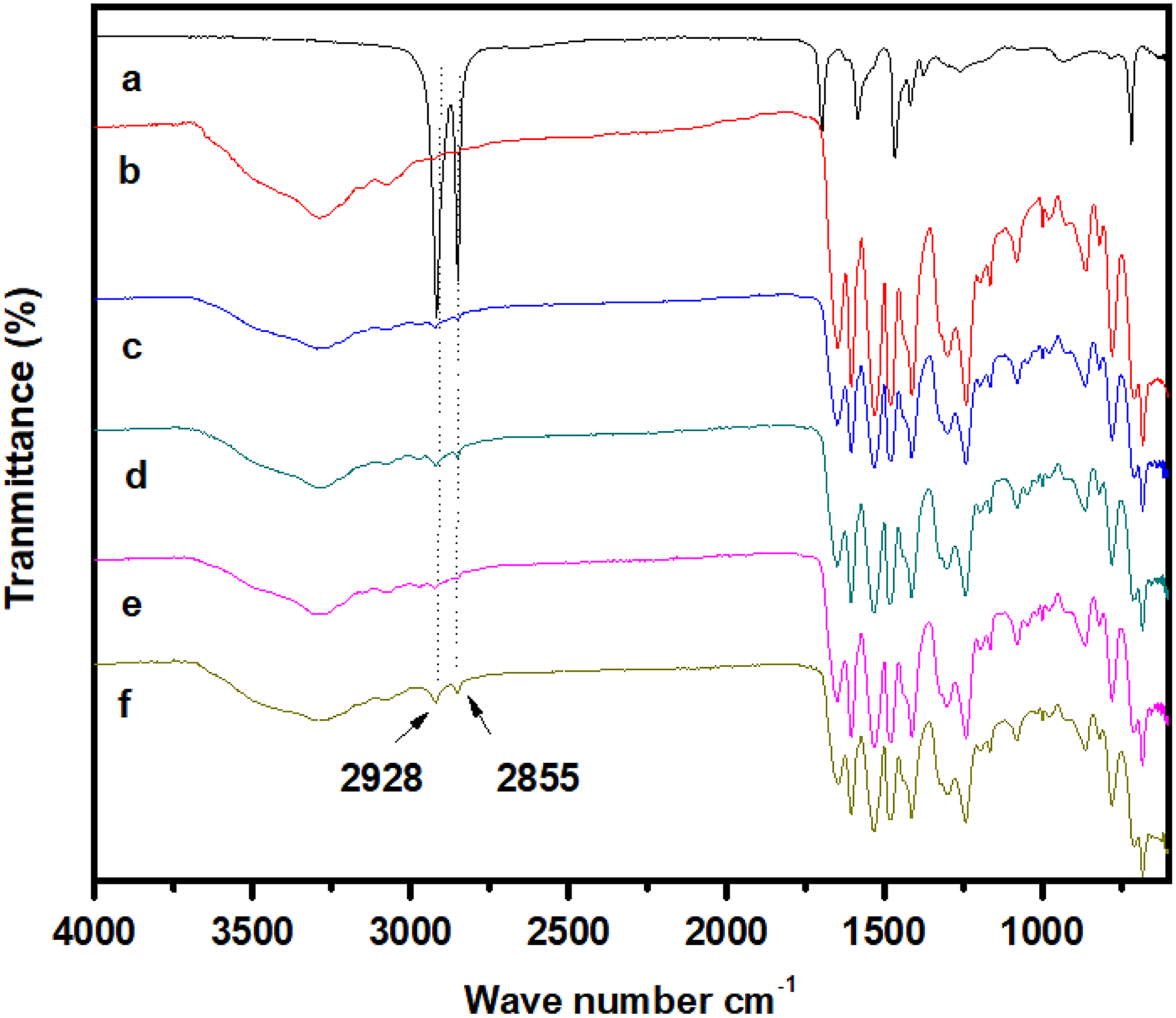
FTIR spectra of (a) ZnI, (b) m-Ar, (c) m-Ar/ZnI 3:0.25, (d) m-Ar/ZnI 3:0.5, (e) m-Ar/ZnI 3:0.75, (f) m-Ar/ZnI 3:1.
X-ray diffraction
Figure 3 shows the X-ray scattering patterns of the ZnI, m-Ar, and m-Ar/ZnI composite films. All the samples show a single broad peak except the ZnI component. The characteristic sharp diffraction peaks for the ZnI found at 20.3 (corresponding to interlayer spacing of about 4.37 Ǻ) and the diffraction peak of m-Ar appeared at 24.04 (d-spacing 3.69 Ǻ). The m-Ar was characterized by intense narrow diffraction reflections, indicates high longitudinal ordering of their structure. 21 The m-Ar/Zn composite film had a small and weak diffraction peak appeared at 22.03 (d-spacing 4.03 Ǻ). The disappearance of the characteristic peak of ZnI and m-Ar/Zn broad peak is slightly moved compared with m-Ar composite film, indicating the interaction of ZnI interface into the m-Ar interlayer during the fabrication. The peak pattern of m-Ar and composite films is similar but the peak intensity is stronger than that of the unfilled film.
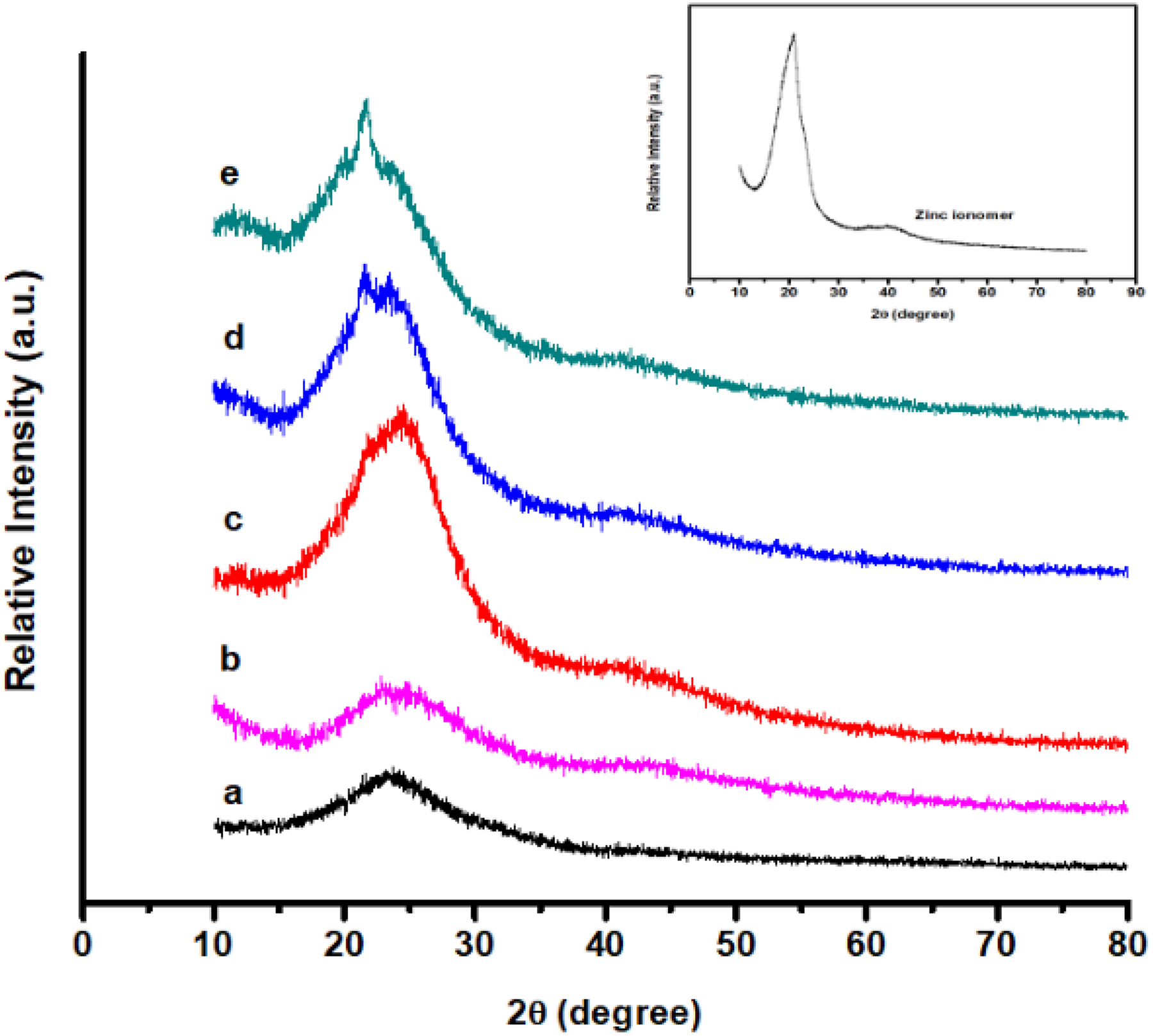
X-ray powder diffractogram of (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, (e) m-Ar/ZnI 3:1.
Thermal properties
The thermal properties of the m-Ar and m-Ar/ZnI composite films are summarized in Table 1, whereas the glass transition temperature (Tg) of the films were determined from the DSC heating scans. The Tg of the m-Ar and m-Ar/ZnI composites are in the range of 250°C to 274°C. The Tg of the composite film decreased with increase in the percentage of the ZnI content. In this composite systems, a single Tg observed corresponding to the segmental motion of the molecular chains (Figure 4). This indicates the miscibility of the components in the composite films, relevant changes in the crystallization, and melting points of composite film. The Tg of m-Ar/ZnI composites shifted toward the lower temperature, and this confirms the better compatibility between the blend components.
Thermal analysis and DMA of m-Ar/ZnI composites.
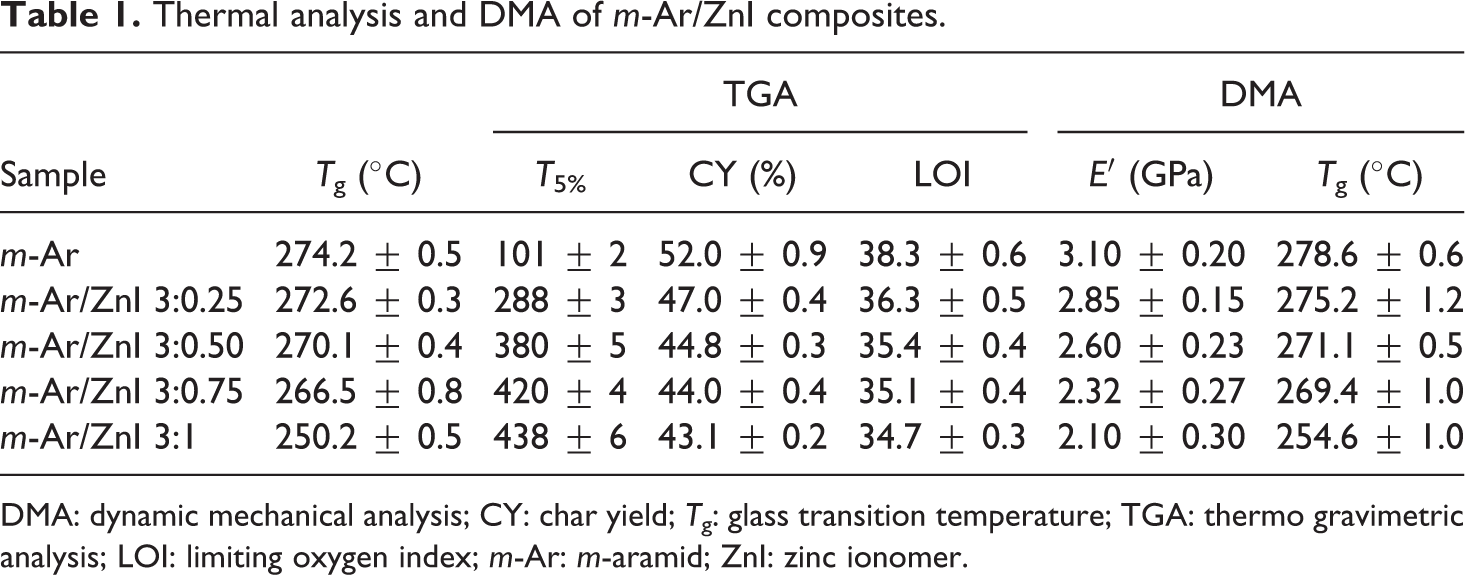
DMA: dynamic mechanical analysis; CY: char yield; Tg: glass transition temperature; TGA: thermo gravimetric analysis; LOI: limiting oxygen index; m-Ar: m-aramid; ZnI: zinc ionomer.
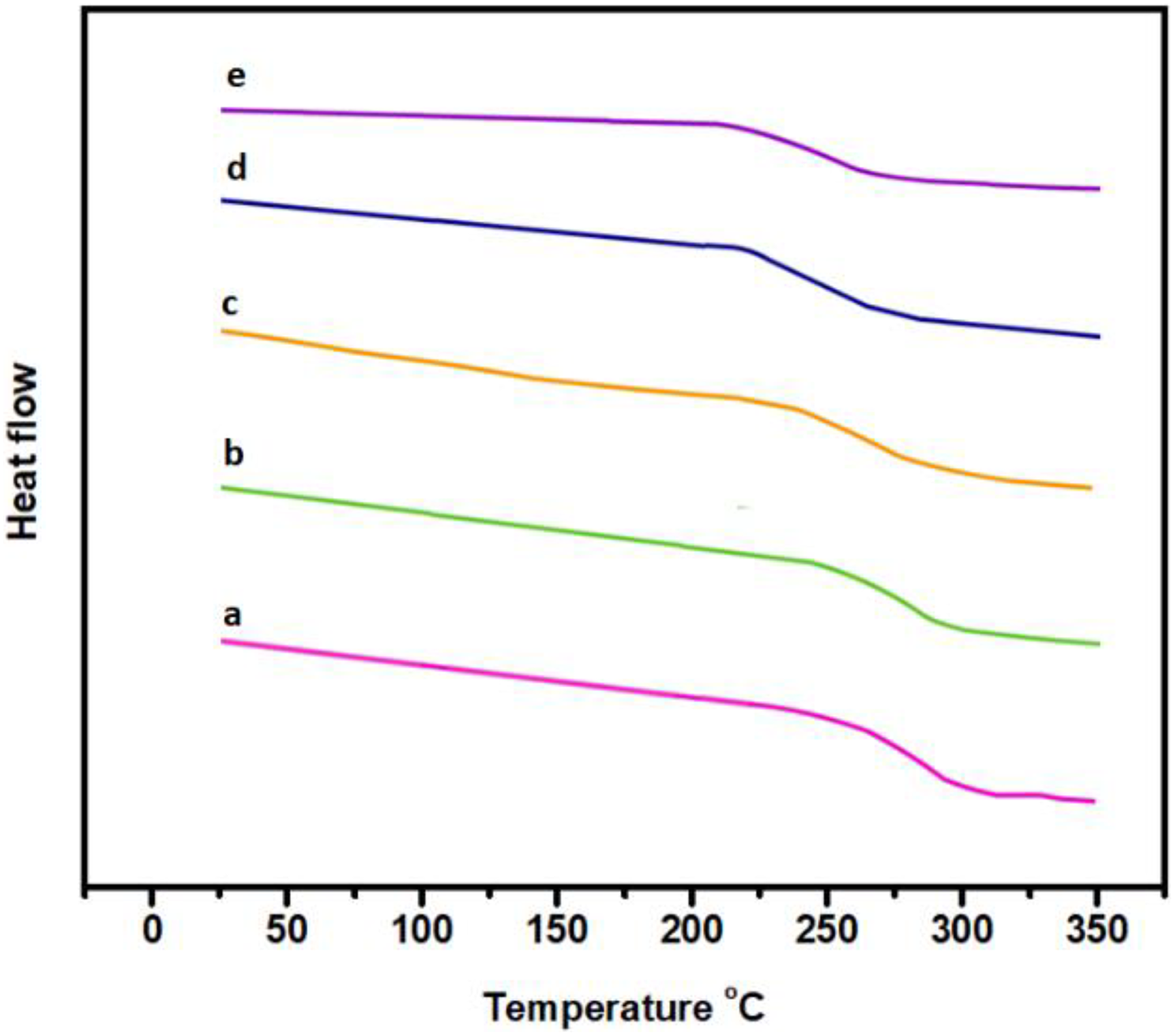
DSC thermogram of (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, and (e) m-Ar/ZnI 3:1.
The thermal degradation temperature and compatibility were investigated from the TGA curves of the m-Ar and m-Ar/ZnI blends presented in Figure 5. The TGA revealed that the pure m-Ar film had good thermal stability than the composite film, indicating that the additional amide linkage in m-Ar/ZnI blend was effective in disturbing the interchain interaction of Ar. The ZnI improves the initial thermal degradation temperature of m-Ar/ZnI composite film. The 5% gravimetric loss temperatures are in the range of 101–438°C. There is a constant weight loss which indicates that the miscibility and compatibility between the m-Ar and ZnI components. The char yields (CYs) are in the range of 43.11–52%. The CY of the m-Ar/ZnI composite films decreases with increase the percentage of ZnI in the blend. The limiting oxygen index (LOI) values which can be taken as an indicator to evaluate the polymer’s flame retardancy were measured and are shown in Table 1. The CY of a material can be used to estimate LOI according to Van Krevelen and Hoftyzer equation. 22 The CY of the composite films were found to be high in the range of 34.7–38.3% indicating high flame retardancy, which is expressed in terms of their LOI value. The LOI values of the polymers should be above the threshold value of 26 to render them self-extinguishing and for their qualification for many applications requiring good flame resistance. 23 The prepared m-Ar/ZnI composites show LOI value greater than 26 confirming their good flame-retardant properties.
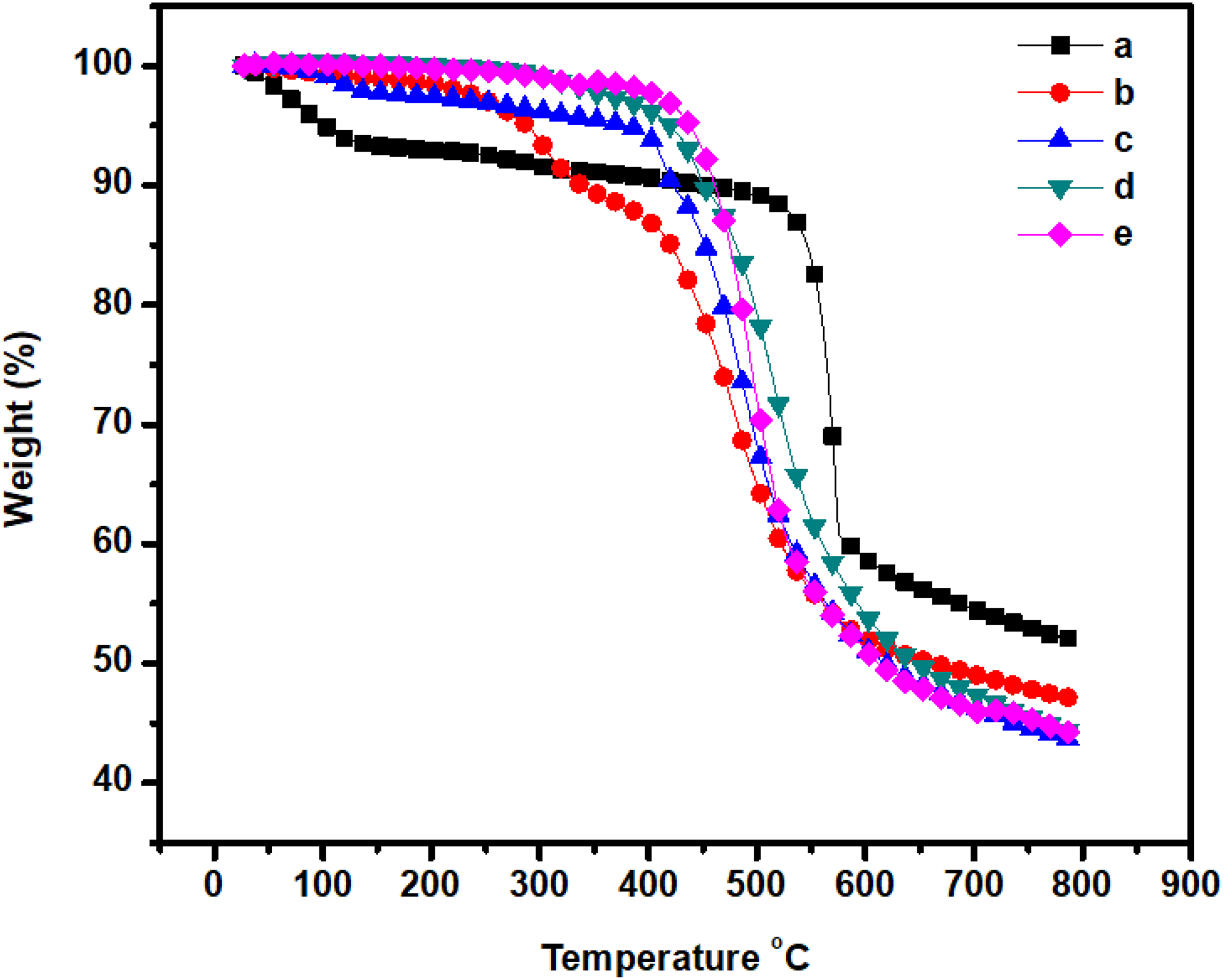
TGA thermogram of (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, and (e) m-Ar/ZnI 3:1.
Optical properties
The optical properties of m-Ar/ZnI composites films are shown in Figure 6. The cutoff wavelengths of the composite films were in the range of 360–380 nm, respectively. The spectral shapes and thickness of m-Ar/ZnI films were similar to each other and the transmittance of the films measured at 600 nm are in the range of 78.45–64.22 nm. The transparency of these composite film depends on the ZnI dispersion in the polymer matrix. The UV results indicate that the m-Ar/ZnI composite films were not transparent because the average size of the particle is bigger than that of the wavelength of the light, and the distribution of particles is relatively uniform. The tendency for the agglomeration of small particles into larger size decreases the homogeneity of the system and the particle size becomes larger, the transmittance values decreased.
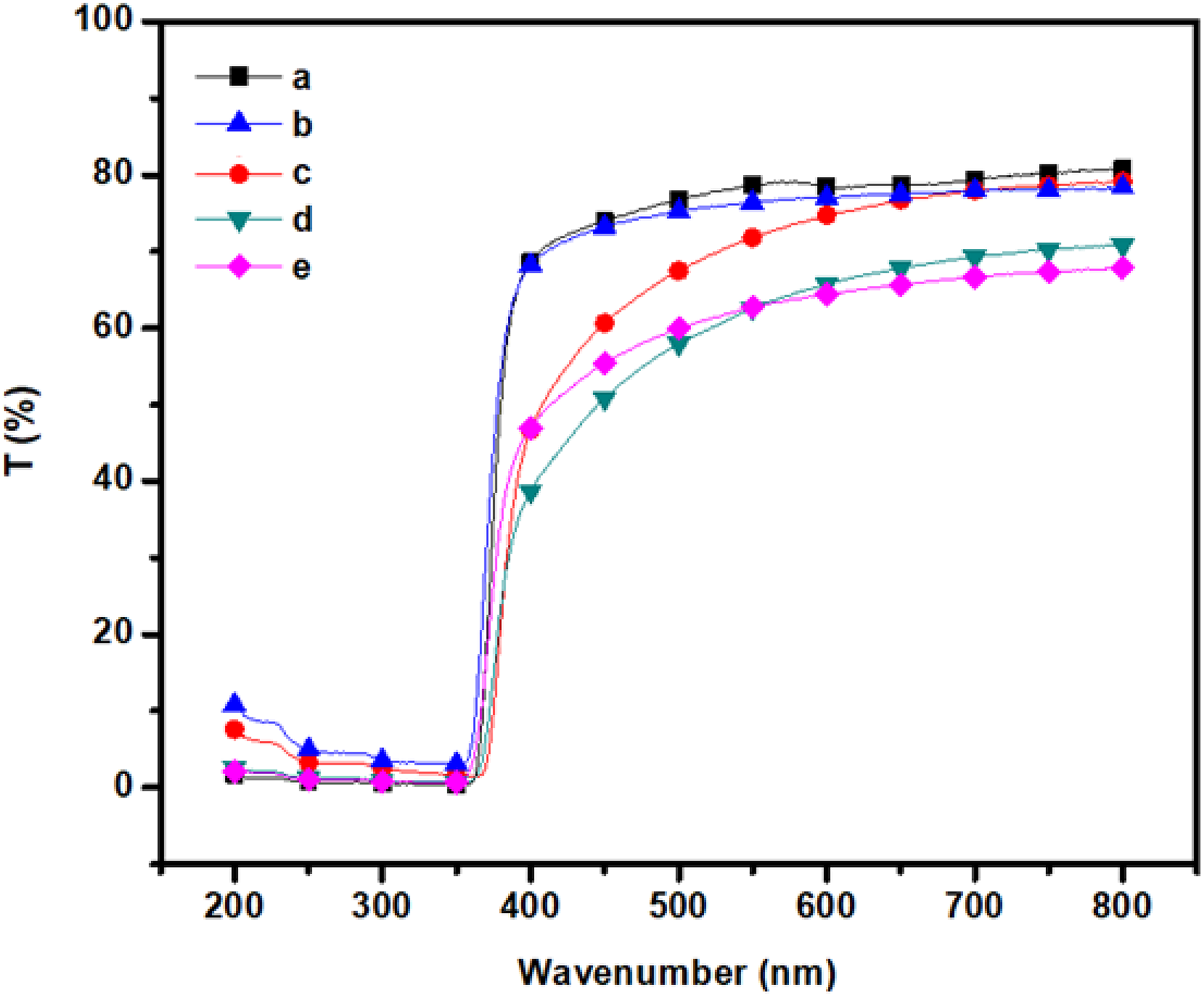
UV transmittance spectra of (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, (e) m-Ar/ZnI 3:1.
Dynamic mechanical analysis
The DMA of the composites blend is shown in Figure 7 and data are summarized in Table 1. From Figure 7, over a temperature range of 30°C to 350°C, single transition regions as indicated by damping maxima are recorded for each specimen. The small strain rheological behaviors for the system at the highest m-Ar content seem to change with the ZnI content. The storage modulus decreased as corresponding to the melting transition of film shifted to a higher temperature when ZnI content increased in the composite films. 1,24 The storage modulus values are in the range of 2.10–3.10 GPa (at 50°C). The percentage of ZnI increased in the composite film as the storage modulus decreased. This is due to the low storage modulus value of the ZnI. The incorporation of ZnI produces significant trend in displacement of the α-relaxation peak for the films. The composite film with 3:1 weight ratio shows the lowest Tg (Figure 8) compared to the other composition. It is suggested that the notching affect to the composite film. They may introduce a micro void in the m-Ar/ZnI composites. This results indicated an improvement in interfacial bonding in the composites because of the higher the damping at the interface, the poorer the interface adhesion. The Tg shift to the lower temperature when incorporation of ZnI into the matrix. This behavior is derived from the increase of the molecular mobility. The results confirm that some compatibility exist between the m-Ar and ZnI components. This is between the amide group and methyl group of ZnI resulting in hetero chain association. Thus, the extent of the microphase separation between the segments increased with an increase in the interaction, Tg gradually decreased with an increase in the interaction.
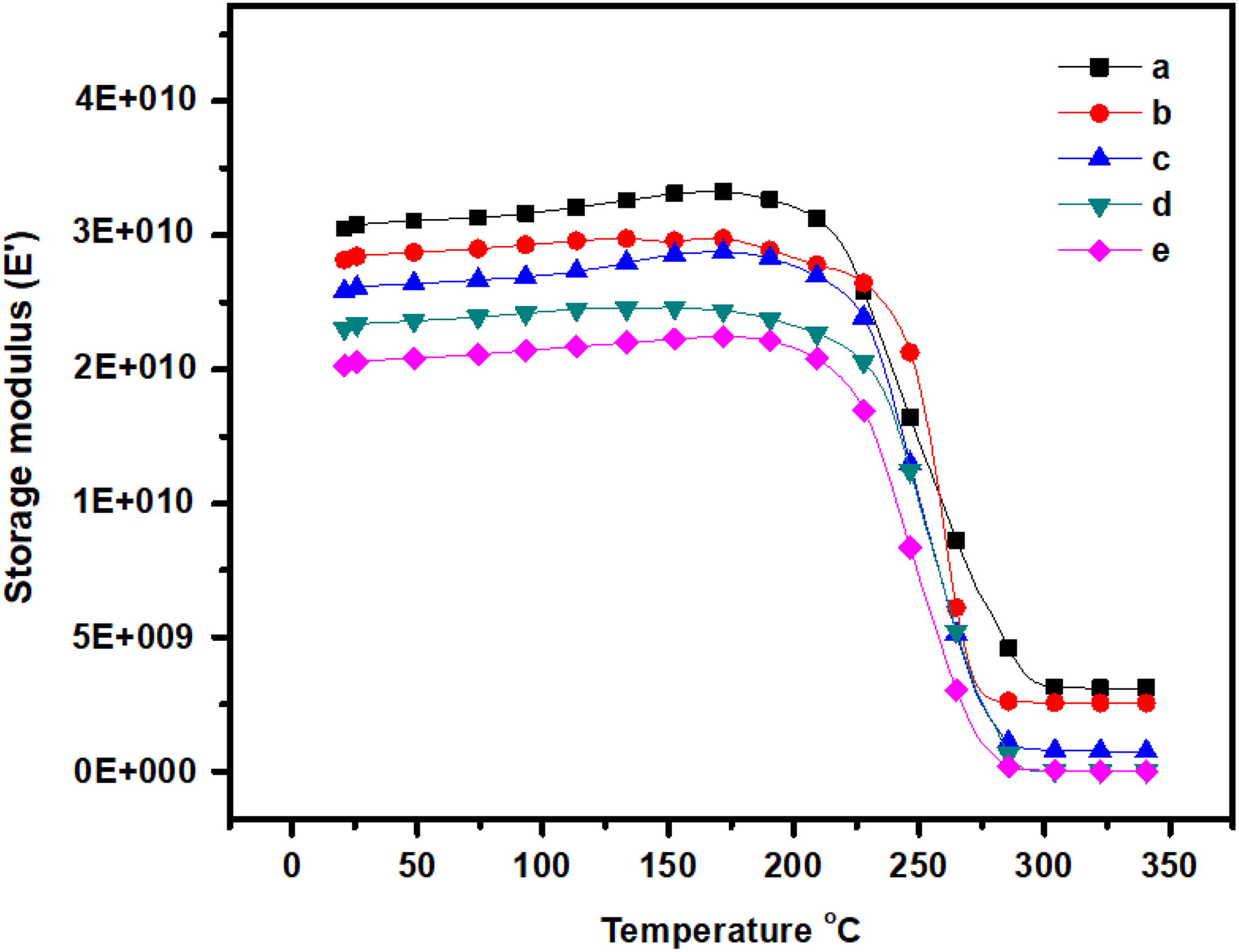
DMA curves show the plot of storage modulus (E′) versus temperature of the (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, and (e) m-Ar/ZnI 3:1.
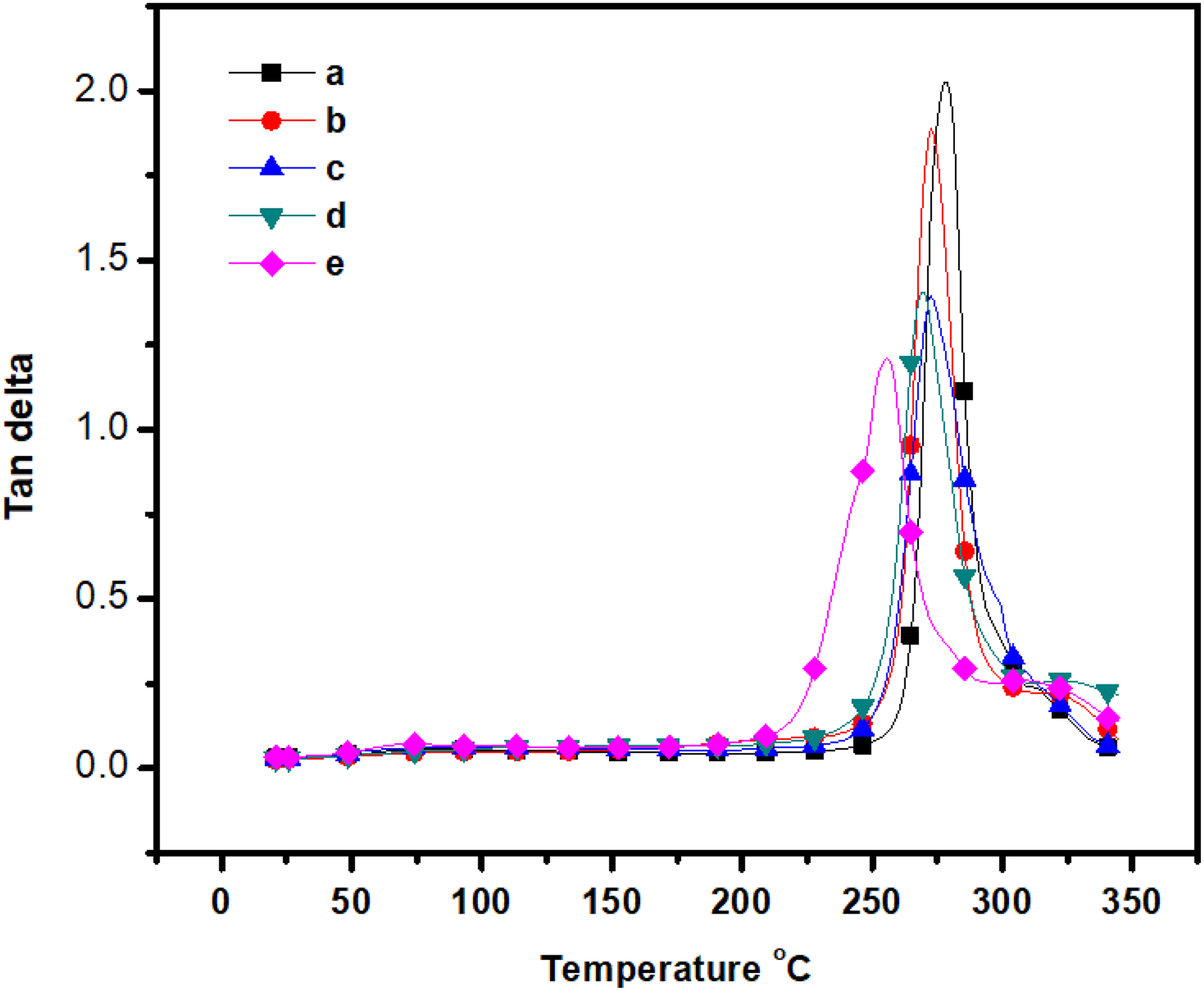
DMA curves shows the plot of loss modulus versus temperature of the (a) m- Aramid, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, and (e) m-Ar/ZnI 3:1.
Mechanical properties
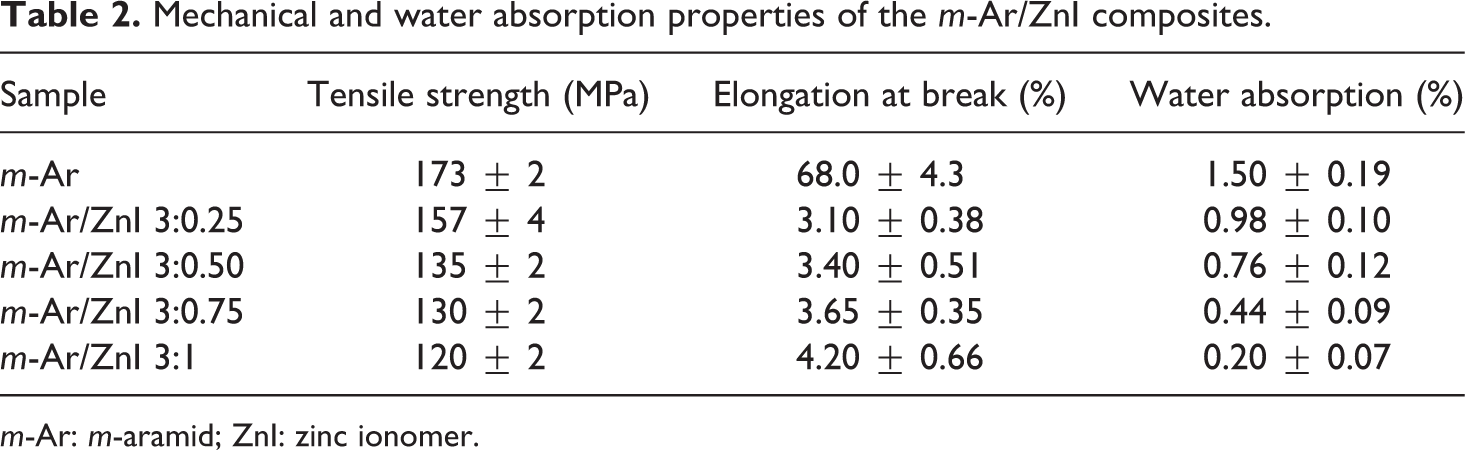
The mechanical studies of the m-Ar and m-Ar/ZnI composite films were prepared with different weight ratio. All the results are listed in Table 2 and Figure 9, it shows that the tensile strength of neat m-Ar is higher than those of composite films. The tensile strength of the composite system increased with high m-Ar content. The results show that m-Ar/ZnI composite films exhibit a lower strength compared to the pure m-Ar. This was also observed that the application of tensile load leads to strain hardening effects. This was explained by the fact that the m-Ar fiber as a chain of crystallites was slightly disoriented with respect to the fiber axis and the application of tensile load results in the alignment of crystallites in the loading direction. 9 On the other hand, the elongations at break for specimens are slightly changed. At high particle contents, ZnIs tend to agglomerate due to strong interaction between the particles. This agglomeration of particles causes premature failure of matrix when an external force is imposed on the composite. The addition of ZnIs to the Ar matrix resulted in the deterioration of mechanical properties. 3,25 –29 The overall mechanical properties of this system show no clear trend or pattern with variation in data. 13
Mechanical and water absorption properties of the m-Ar/ZnI composites.
m-Ar: m-aramid; ZnI: zinc ionomer.
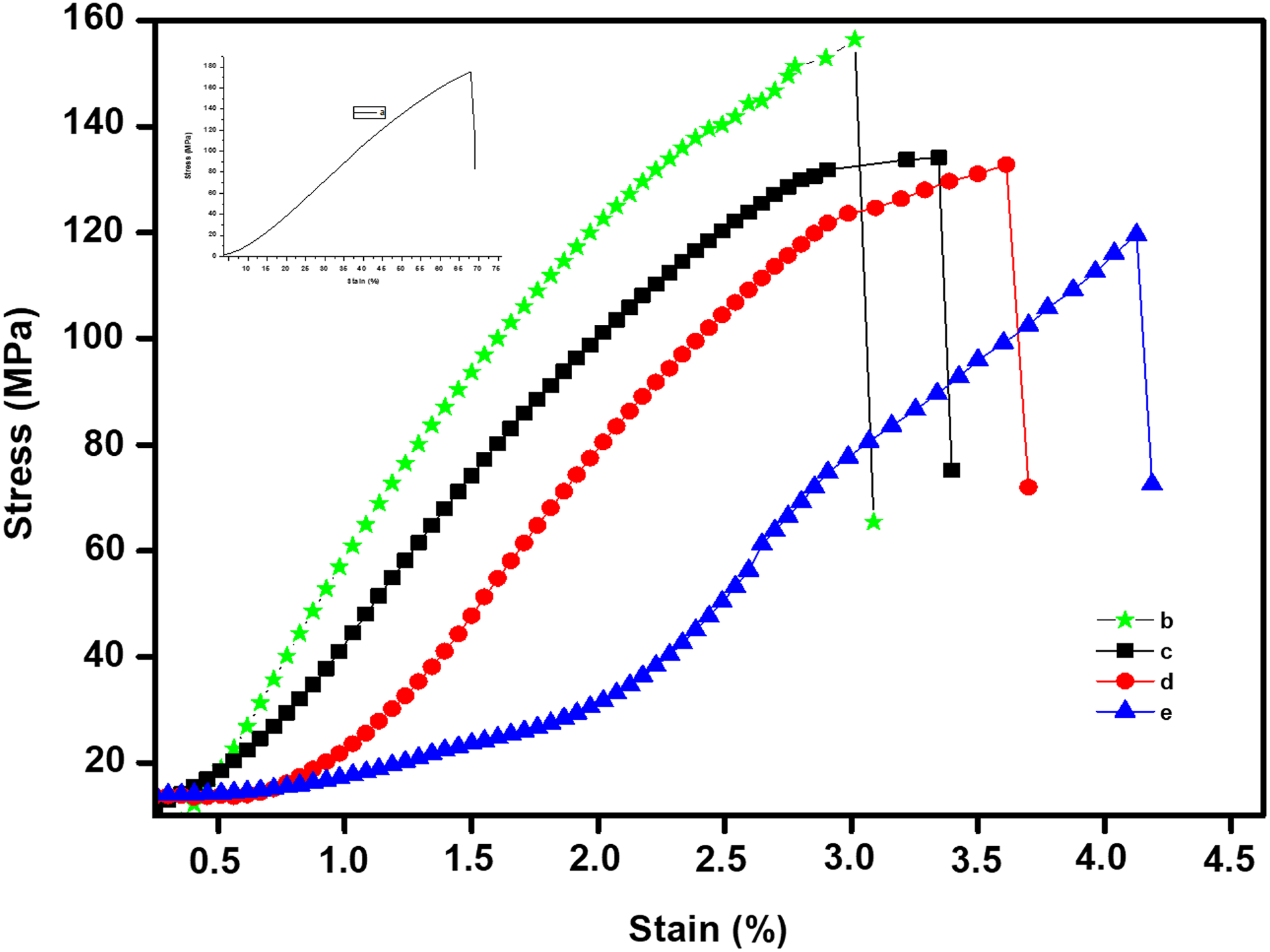
Typical stress–stain curves of the (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, and (e) m-Ar/ZnI 3:1.
Morphological study
The morphology of the composite film has been studied by SEM. The influence of the ZnI content on the matrix is shown in Figure 10. All the composite blends show compatibility or homogeneity of the blend components, which is believed to be due to the interfacial ionic interactions as discussed earlier. The morphology of this composite films exhibits self-assembled surface, uniform, no aggregated particles, and homogenously dispersed. The SEM micrographs are consistent with the DMA results. The results indicated that the addition of an ionomeric compatibilizer dramatically changed the morphology of these m-Ar/ZnI composite films.

SEM images of the (a) m-Ar, (b) m-Ar/ZnI 3:0.25, (c) m-Ar/ZnI 3:0.5, (d) m-Ar/ZnI 3:0.75, and (e) m-Ar/ZnI 3:1.
Water absorption
The water absorption of the neat m-Ar and m-Ar/ZnI composite films were studied, and the water uptake by the neat m-Ar under saturation conditions (7 days) was found to be 1.50%. The m-Ar contains polar –CO–NH groups which have a tendency to absorb water through hydrogen bonding. This is obviously important since it can adversely affect the mechanical and dielectric properties of the composite film. The higher moisture absorption for the pure m-Ar is caused by the higher hydrophilic nature and the greater interfacial area (capillary effect). 30 The moisture absorption m-Ar/ZnI composite films are in the range of 0.98–0.20%. The different matrix systems have different interface characteristics. The moisture character do not only depend on the fiber but also could be caused by numerous effects, for example, void content, matrix interfaces, and binding of the water molecules to the molecule structure of polymer matrix. The addition of ZnI into the m-Ar matrix improved the moisture absorption properties.
Conclusion
The m-Ar and m-Ar/ZnI composite films were prepared by solution blending method. The ionomers used in this study were copolymers of zinc-neutralized ethylene/methacrylic acid. The ZnI had a strong interaction with m-Ar molecular chains. The entire study established influence of this interaction on the degree of hydrogen bonding was confirmed by FTIR. The better compatibility of these two components can be explained by the results of TGA and DMA. The Tg of the pure m-Ar system shifted to the lower temperature and the thickness of the amorphous phase of the system increases. This confirms the existence of compatibility between m-Ar and ZnI.
Footnotes
Acknowledgement
The authors would like to thank Prof. Taehwan Oh for his support in the tensile analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Technology Innovation Program (or industrial strategic technology development program (10076409, establishment of solution for optical/light design/dynamic concept hybrid 2-D/3-D thermoplastic prepreg for design performance) funded by the Ministry of Trade, Industry & Energy (MOTIE Korea). This research was also partially supported by Yeungnam University in the form of research grant in 2017.
