Abstract
Polypropylene (PP) composites containing magnesium oxysulfate particle (MOSp), magnesium oxysulfate whisker (MOSw), or magnesium oxysulfate sector (MOSs) were prepared via melt blending method. Scanning electron microscopy results showed that three magnesium oxysulfate (MOS) fillers all dispersed homogeneously in PP matrix and displayed vague and fuzzy interfaces. Wide-angle X-ray diffraction (WXRD) patterns showed that MOSp induced the most amount of β-PP, which was supported by polarized light microscopy (PLM) photographs. Moreover, PLM photographs also showed that the presence of MOSp, MOSw, or MOSw decreased the PP spherulites, especially for MOSp. As such, mechanical tests showed that incorporation of MOSp into PP matrix greatly improved the impact strength and least lowered the nominal strain at break. The yield strength and Young’s modulus of composites were greatly enhanced with MOSw. Two possible reasons for this phenomenon are rigidity of MOSw and microstructure of composites. Rheological properties were measured via small amplitude oscillatory shear. The results showed that PP melts containing MOSw exhibited significant yield stress and “shear-thinning” behaviors, which indicated the formation of MOSw network and the transition from “liquid-like” PP matrix to “solid-like” composites. The rheological results greatly proved the enhancement in tensile properties of MOSw-incorporated composites.
Introduction
Magnesium oxysulfate (MOS), with three different types of morphologies, has been growing interest in several academic and industrial studies since its discovery. 1 –3 Initially, MOS with particle form (MOSp) was prepared as a substitute for magnesium oxychloride cement because of its excellent resistance efflorescence behavior, as reported in the study by Temple and Rocks. 4 Furthermore, MOSp decomposes endothermically in theory, accompanied by the release of water vapor, which completely meets the requirement of flame-retardant additives. Eills 5 reported MOSp acted as a composition of fire barrier coating on susceptible wood or plastic substrates, or other substances. The coating effectively prevented the ignition of flame and spread of fire on the coated substrate.
Actually, the presence of whisker-shaped MOS (MOSw) at the 1990s greatly promotes its application, especially in polymeric composites area, because of its inherent outstanding mechanical and flame-retardant properties. 6,7 So far, there are many reports about MOSw-reinforced or flame-retarded polymeric composites, such as polypropylene/MOSw (PP/MOSw), 8,9 polyethylene/MOSw (PE/MOSw), 10,11 silicone rubber/MOSw (SR/MOSw), 12 ethylene-vinyl acetate/MOSw (EVA/MOSw), 13 acryonitrile butadiene styrene copolymer/MOSw (ABS/MOSw), 14,15 polyvinyl chloride/MOSw (PVC/MOSw) 16,17 composites, and so on. Almost all these composites exhibited significant improvement in the flame-retardant properties but scarcely enhancing the mechanical behaviors. For example, Wu et al. 13 declared that EVA/MOSw composite exhibited significantly higher limit oxygen index (LOI) and lower heat release rate than EVA/magnesium hydroxide composite. The mechanical properties of composites were not enhanced as the authors expected. Except this, Fang et al. 12 found that both the tensile strength and the elongation at break of SR/MOSw composite decreased with the incorporation of MOSw, although significant improvement happened on the flame-retardant properties. Jiang and Li 17 observed a slight decrease in tensile strength of PVC/MOSw composite compared with neat PVC. In other study, Jiang et al. 8 also found that the tensile strength reduced obviously when MOSw was added into PP matrix. They ascribed it to no chemical bond formed between PP matrix and MOSw. In fact, the main reason of these phenomena can be attributed to the agglomeration of whiskers. Therefore, surface modification is the most common solution for improving polymer/MOSw mechanical properties. 18
Analogously, sector-shaped MOS (MOSs) is structured similar to the agglomerated MOSw by one head. The mechanical properties of polymer/MOSs composites are not as high as expected. Actually, MOSs is more easily prepared than MOSw via hydrothermal process, 19,20 but the large dimensions and complicated structures limited its application as a reinforcing filler. Only few attempt has been made to prepare polymer/MOSs composites. Gao et al. 21 found that MOSs performed much higher caloric receptivity and decomposition temperature than magnesium hydroxide and aluminum hydroxide, two kinds of flame retardant commonly used in plastic industry. When 20 wt% methyl methacrylate (MMA) modified MOSs was added, the LOI of PP composites increased to 22.4%, while the tensile strength greatly increased by 92%. However, there was no further investigation about the reinforcing mechanism of MOSs for PP matrix, neither for other polymer matrices.
Based on the above samples, we found that the morphology of MOS greatly affected the mechanical properties of composites, while almost no influence on the flame-retardant properties. In fact, the size or morphology of all fillers plays a vital role in modifying the mechanical properties of polymer matrices. 22 –26 Tang et al. 23 declared that both micro and nanoparticles of zinc oxide reduced spherulite size in isotactic polypropylene (iPP), but smaller spherulites were observed when the inorganic nanoparticles exhibited dimensions on the order of 40–150 nm, relative to micron-size particles. The mechanical properties changed with the size of spherulites. Besides, Khoshkava and Kamal 24 reported that different cellulose nanocrystals’ morphologies greatly affected the rheological and mechanical properties of its PP composites. With reference to MOS, the study of its morphology affected on dispersion and other properties of polymeric composites has not been reported, as far as we know. Therefore, in this article, three different MOS microstructures obtained by different synthesis processes and conditions were incorporated into PP via melt mixing. Maleic anhydride-grafted polypropylene (PP-g-MAH) was chosen as a compatibilizer according to our previous studies. 27 Field emission scanning electron microscope (FESEM) was used to observe the morphology of prepared PP/MOS composites. Wide-angle X-ray diffraction (WXRD) and polarized light microscope (PLM) were employed to investigate the crystallization behaviors of samples. The mechanical properties were characterized in terms of tensile and impact behavior aspects. Furthermore, rheological behaviors were also studied with frequency sweeps in small amplitude oscillatory shear (SAOS), as rheology is a powerful tool to examine the microstructure of composites.
Experimental
Materials
iPP (T30s) was purchased from Maoming Petrochemical Co., Ltd., Maoming, China. It has a melt flow rate of 3.7 g/10 min (230°C, 2.16 kg−1) and a density of 0.91 g cm−3. PP-g-MAH with a density of 0.90 g cm−3 was purchased from Fine-blend Compatibilizer Jiangsu Co., Ltd, China. The MAH content was 0.92% (±0.03%). Antioxidant 1010 (95.0%) was purchased from Tokyo Chemical Industry (TCI), Tokyo, Japan. Whisker-shaped MOS (MOSw) with a diameter of 0.5–1 μm and a length of 10–50 μm was obtained from a procedure described as follows: Magnesium sulfate and sodium hydroxide were mixed in aqueous solution by the molar ratio of 1/0.8, and then heated and vigorous stirring for several hours. Impurities were removed by the step of centrifuging and washing with plenty of water. The product was obtained after drying at 60°C. Sector-shaped MOSw (MOSs) was obtained from almost the same procedure as MOSw, just changed the molar ratio of magnesium sulfate and sodium hydroxide to 1/1.2. 19 Particle-shaped MOS (MOSp) was obtained from MOSw after 2 h ball-milling process. 22 The morphology and the XRD analysis of MOS are presented in Online Supplementary Figures S1 and S2.
Sample preparation
Before blending, all materials were dried at 70°C for 8 h to eliminate the effect of moisture, and then the PP pellets, MOS, and antioxidant 1010 were mixed based on a certain percentage with an RM-200C torque rheometer (HAPRO, Harbin, China) at 190°C with a rotor speed of 60 r min−1 for 15 min. The weight fractions of MOSp, MOSw, and MOSs were 5 wt%, 10 wt%, and 15 wt%, respectively. The content of PP-g-MAH and antioxidant 1010 was maintained 15 wt% and 0.2 wt% of PP composites separately.
Table 1 summarizes the nomenclature used to identify different samples. Film samples with different thicknesses were obtained by compression molding with an XH-406B press vulcanizer (Xihua, Dongguan, China) at 190°C for 8 min without pressure and for 7 min under a pressure of 15 MPa, subsequently. Then, the film samples were cooled to room temperature at the same pressure for 5 min. All the samples were stored in a dry and room temperature (23 ± 2°C).
Nomenclature used for samples in this article.

PP-g-MAH: maleic anhydride-grafted polypropylene MOSp: magnesium oxysulfate particle; MOSw: magnesium oxysulfate whisker; MOSs: magnesium oxysulfate sector.
Characterization
The cryo-fractured and impact-fractured surface of specimens were both observed after sputtering coated with gold with a JSM-6701F FESEM from JEOL, Japan, using an acceleration voltage of 5.0 kV. WXRD patterns of the samples were acquired using a Philips X’Pert X-ray spectrometer, Netherlands, with Cu Kα radiation (λ = 0.154 Å). The spectrum scanned ranges from 10° to 30° for 2θ value by 2° min−1 speed. An Olympus BX51 polarized light microscopy (PLM) equipped with a Linkman, UK, THMS600 hot-stage thermal controller was used to study the spherulitic morphology of neat PP and PP composites in the isothermal crystallization process. The samples were placed between two glass slides on the hot-stage and melted at 200°C for 3 min to erase any thermal history and then cooled down to the 130°C at a rate of 100°C min−1. The crystallization procedure and the morphology were observed and recorded by taking photographs. The rheological properties of PP and these composites were measured using a stress-controlled TA, US AR2000ex rheometer. The experiments were conducted under nitrogen atmosphere to avoid oxidation of the samples. A parallel plate geometry was used with a gap of 1 mm and a plate diameter of 25 mm. Frequency sweeps in SAOS on the samples at 190°C.
Tensile strength and modulus were measured at room temperature with CMT 6000 Electronic Universal Testing Machine according to ISO 527-2:1993, using dumbbell-shaped specimens (1BB) with a length of 30 mm and a thickness of 2 mm. The test speed was 10 mm min−1. Impact strength was measured at room temperature with XJJD-50 Charpy Impact Tester, China, using a 7.5J hammer according to ISO 179-1:2000. The specimens were cut with the dimensions of 80 × 10 × 4 mm3, and V-shaped gap of 2 mm in the middle of samples. The results for each measurement were the average value of six samples test at least.
Results and discussion
SEM analysis
Figure 1 shows the scanning electron microscopy (SEM) results of neat PP, PP/15PP-g-MAH, PP/15PP-g-MAH/5MOSp, PP/15PP-g-MAH/5MOSw, and PP/15PP-g-MAH/5MOSs composites. The smooth surface of PP/15PP-g-MAH as neat PP and no apparent new phase indicates that PP-g-MAH is well compatible with the PP matrix. As such, the interfaces between MOS fillers and the PP matrix are vague and fuzzy. For PP/15PP-g-MAH/5MOSp composite, the small voids in Figure 1(c) and 1(d) are possibly MOSp that were pulled out during the fracture process. 28

SEM micrographs of neat PP (a), PP/15PP-g-MAH (b), PP/15PP-g-MAH/5MOSp (c) and (d), PP/15PP-g-MAH/5MOSw (e) and (f), and PP/15PP-g-MAH/5MOSs (g) and (h). SEM: scanning electron microscopy; PP: polypropylene; PP-g-MAH: maleic anhydride-grafted polypropylene; MOSp: magnesium oxysulfate particle; MOSw: magnesium oxysulfate whisker; MOSs: magnesium oxysulfate sector.
For PP/15PP-g-MAH/5MOSw composite, it is more obvious that polymer melts wrapped on the surface of MOSw and the interfaces between MOSw and the PP matrix are well-knit, as shown in Figure 1(f). Unlike MOSp and MOSw, the structures of MOSs are much more complicated so that almost polymer melt infiltration took place within the sector structures, as shown in Figure 1(h). In Figure 1(c), (e), and (g), we also observe how the compatibilization lead to a dispersive state of MOS fillers. However, it should be noticed that MOSs possibly acts as agglomerations in the PP matrix because of its immense size and complicated structures. These analogous agglomerations usually lead to the stress concentration and microcrack initiation, which negatively affect the mechanical properties of composites.
Crystallization behaviors
WXRD analysis
WXRD characterization is used to identify the crystallization structure of neat PP and its composites. As shown in Figure 2, the WXRD pattern of neat PP shows peaks at 2θ = 14.2°, 17.0°, 18.6°, 21.3°, and 21.9°, respectively, corresponding to (110), (040), (130), (131), and (111) reflections, indicating that only α-crystal form exists in neat PP. 29,30 With respect to PP/15PP-g-MAH composites, a weak extra peak arise at 2θ = 16.0° corresponding to (300) reflection of β-crystal form. 30 For PP/15PP-g-MAH/15MOSp composite, a stronger β-crystal peak is observed, indicating that MOSp and PP-g-MAH act synergistically to induce β-crystal PP. Nevertheless, when MOSw or MOSs is added, the intensity of β-crystal peak decreases notably, indicating that MOSw and MOSs are detrimental to inducing β-crystal PP. These different effects between MOSp and MOSw (or MOSs) are probably due to the smaller dimension of MOSp. Moreover, the peaks at 2θ = 13.0°, 17.3°, and 22.9° correspond to (201), (202), and (203) reflections of MOS, respectively. 18
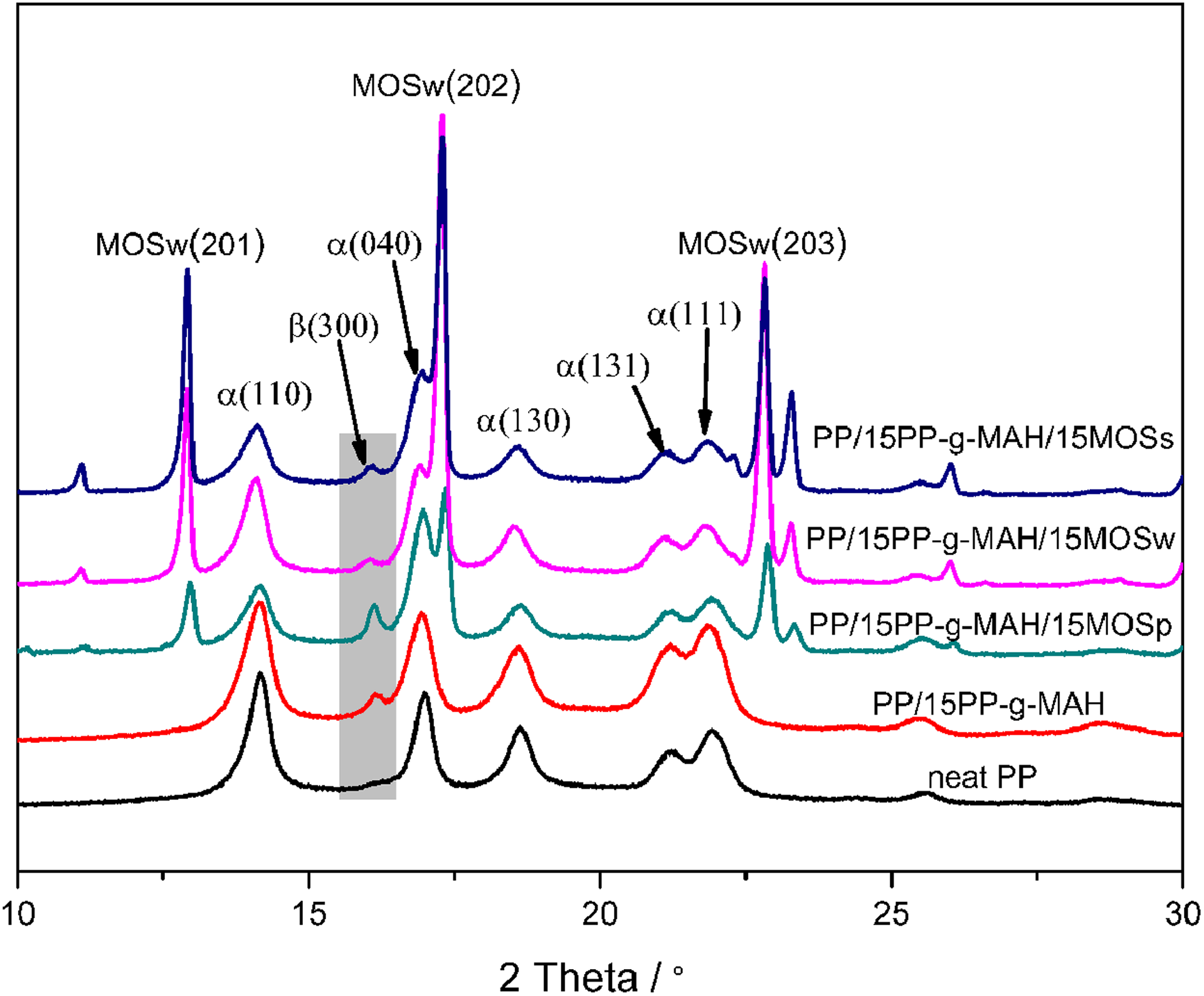
WXRD patterns of neat PP, PP/15PP-g-MAH, and PP composites with different MOS. WXRD: wide-angle X-ray diffraction; PP: polypropylene; PP-g-MAH: maleic anhydride-grafted polypropylene MOS: magnesium oxysulfate.
PLM results
The influences of different appearances of MOS on the crystallization procedure and the morphology of PP spherulites are directly observed by PLM characterization. Figure 3 shows the photographs of neat PP, PP/15PP-g-MAH, PP/15PP-g-MAH/15MOSp, PP/15PP-g-MAH/15MOSw, and PP/15PP-g-MAH/15MOSs composites crystallized at 130°C. Neat PP presents extremely huge spherulites with diameters more than 130 μm. The interfaces between the PP spherulites are crowded but still clear. The PP matrix containing this kind of spherulites is easily fractured. The introduction of PP-g-MAH remarkably decreased the size of PP spherulites to about 80 μm, as shown in Figure 3(b). The boundaries between spherulites become vague and fuzzy, but no other phase is observed. This is the indication of high compatibility of PP-g-MAH within the PP matrix. Further, when MOSp is added into PP/15PP-g-MAH composite, the size of spherulites decreases quite small and their boundaries are almost indistinct because MOSp promotes the heterogeneous nucleation of PP. The formation of smaller spherulites is conductive to toughening the PP matrix. 31 Besides, it is also observed a few different phases in PP/15PP-g-MAH/15MOSp composite, as shown in the squares of Figure 3(c). These spherulites are more bright and bigger size than others. We also observed their melting process at about 140°C. Hence, it is concluded that MOSp induces a few amount of β-PP. For PP/15PP-g-MAH/15MOSw, the size of spherulites is not as small as PP/15PP-g-MAH/15MOSp composite probably due to the larger size of MOSw. As such, the largest size of MOSs induces the biggest PP spherulites with diameter about 20 μm, as shown in Figure 3(e). On the one hand, MOSs induces fewer nucleus at the beginning of crystallization. On the other hand, the complicated three-dimensional structure of MOSs also hindered the migration of PP chains, so that the biggest spherulites are formed in PP/15PP-g-MAH/15MOSs composite.

PLM photographs of crystallized neat PP (a), PP/15PP-g-MAH (b), PP/15PP-g-MAH/15MOSp (c), PP/15PP-g-MAH/15MOSw (d), and PP/15PP-g-MAH/15MOSs (e). PLM: polarized light microscopy; PP-g-MAH: maleic anhydride-grafted polypropylene; MOSp: magnesium oxysulfate particle; MOSw: magnesium oxysulfate whisker; MOSs: magnesium oxysulfate sector.
Mechanical properties
Tensile behaviors
The tensile strength, Young’s modulus, and nominal strain at break of the PP/15PP-g-MAH/iMOSp, PP/15PP-g-MAH/iMOSw, and PP/15PP-g-MAH/iMOSs composites are compared to those of PP/15PP-g-MAH in Figure 4. As shown in Figure 4(a), the yield strength of PP/15PP-g-MAH decreases by approximately 5.6% in case of adding 5 wt% of MOSp. With the increase of MOSp content, the yield strength of PP/15PP-g-MAH/iMOSp composites slightly increases. As for PP/15PP-g-MAH/15MOSp composite, the yield strength even increases by approximately 3.9% than that of PP/15PP-g-MAH. The most prominent enhancement is observed in the PP/15PP-g-MAH/iMOSw composites as the yield strength increased by approximately 11.2% when only 5 wt% of MOSw was added. This outstanding reinforcement is mainly attributed to the high mechanical strength and modulus and aspect ratios of MOSw own, as reported by other authors. 22,28 MOSs also improves the yield strength of the matrix inferior to MOSw because of the large dimensions and complicated structures of MOSs that are detrimental for the stress transfer from the matrix to the filler.
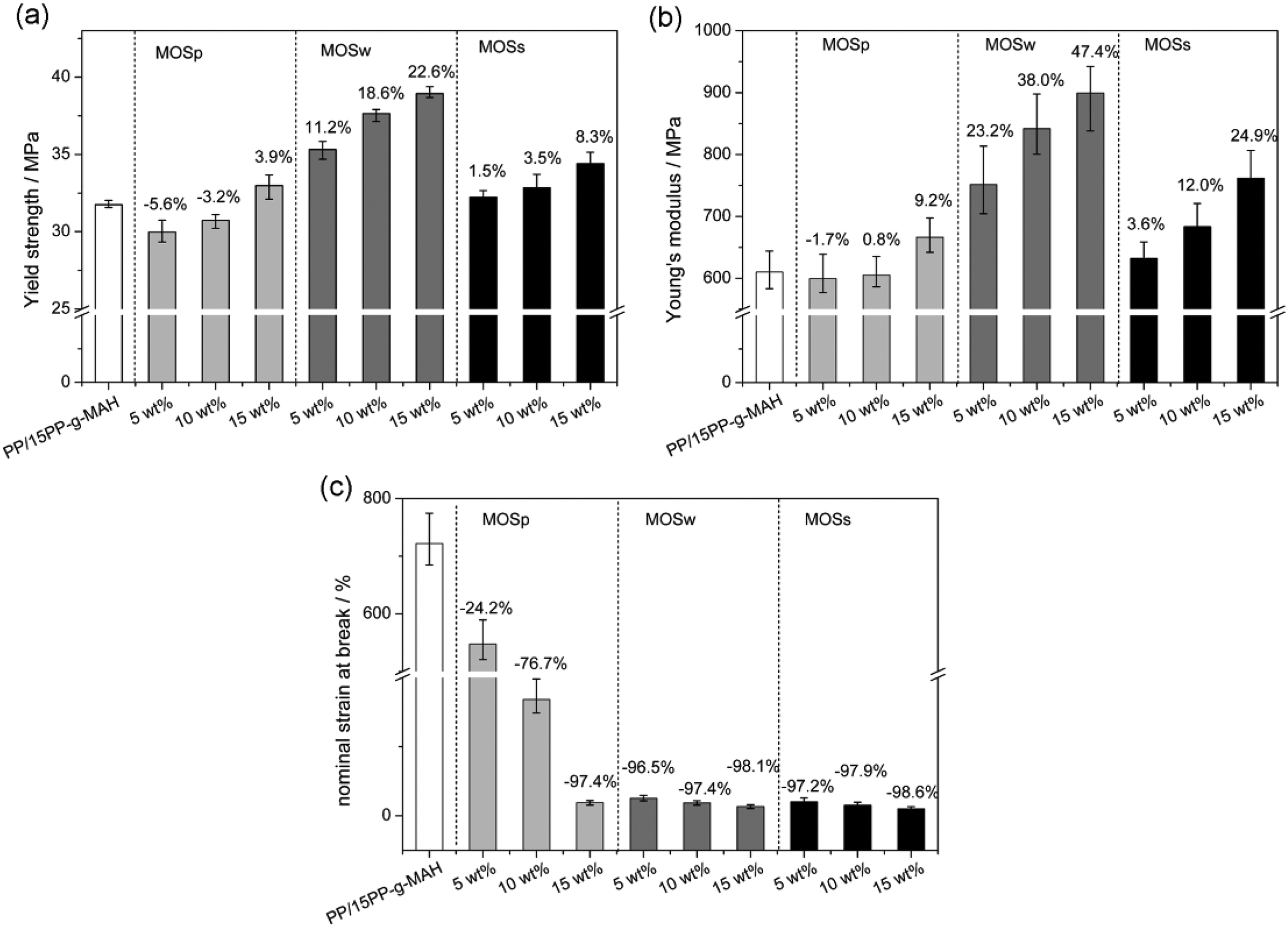
Comparison of the yield strength (a), Young’s modulus (b), and nominal strain at break (c) of composites filled with different MOS. The numbers above the bars represent the changes in percentage with respect to the PP/15PP-g-MAH. PP-g-MAH: maleic anhydride-grafted polypropylene; MOS: magnesium oxysulfate.
In Figure 4(b), Young’s modulus variations of these composites which are very similar to the yield strength, and Young’s modulus are more sensitive to these fillers than yield strength. For example, Young’s modulus of PP/15PP-g-MAH/15MOSp, PP/15PP-g-MAH/15MOSw, and PP/15PP-g-MAH/15MOSs composites increases by approximately 9.2%, 47.4%, and 24.9%, respectively. The significant increase in Young’s modulus is mainly ascribed to the rigidity of MOS fillers. The enhancement of interfacial adhesion between MOS fillers and the PP matrix by PP-g-MAH is another non-negligible reason.
Finally, as shown in Figure 4(c), the nominal strain at break decreases dramatically when MOSw or MOSs is added, which is probably due to the retardation of chain mobility by the fillers. 32 However, MOSp-filled composites show rather less lowering nominal strain at break, especially for the low levels of MOSp. This is mainly due to the small size of MOSp that hardly reduces the mobility of PP chains. In more detail, the nominal strain at break decreases by approximately 24.2%, 96.5%, and 97.2% in the presence of 5 wt% of MOSp, MOSw, and MOSs, respectively.
Impact behaviors
The variations of impact strength for the PP/MOS composites with different MOS fillers are shown in Figure 5. Obviously, the incorporation of MOSp dramatically increases the impact strength of PP/15PP-g-MAH and displays a maximum by approximately 193.2% higher at 15 wt% of MOSp loading. The similar behavior has been observed for many polymer composites containing nanoparticles such as calcium carbonate, silica, zinc oxide, and so on. As Van Der Wal et al. 33 reported that the rigid filler particles must meet certain requirements to act as toughening agents: (i) the size of particles should be less than 5 μm to avoid the initiate fracture, (ii) the aspect ratio must be close to avoid high stress concentrations, and (iii) the particles must be dispersed homogeneously in the matrix polymer. Therefore, it is considered that MOSp acts as a toughening agent for PP matrix. Besides, WXRD and PLM results indicate that β-crystal PP is mostly induced by MOSp, while the β-crystal PP with looser structure is also conducive to improve the impact strength. 22 For MOSw, the impact strength of composites increases with the increasing content of MOSw and reaches to maximum by approximately 120.8% higher at 10 wt% content. Further addition of MOSw leads to a slight decrease in the impact strength, which possibly results from the aggregations formed in composites at higher MOSw content. In general, the key mechanism of improving toughness of whisker-reinforced polymer is the pullout effect. 28 When the composite samples are fractured in the impact process, the pullout and breakage of MOSw can absorb a part of impact energy since friction sliding so that improving the impact strength of composites. For MOSs, the maximum value for impact strength is approximately 36.0% higher than that of PP/15pp-g-MAH when 5 wt% of MOSs is added. With an increase of MOSs, the impact strength of composites decreases significantly due to the complicated structures of MOSs, which act as agglomerations in the matrix, as mentioned previously. Besides, the immense size of MOSs is far more than 5 μm which is liable to cause initiate fractures. Hence, the incorporation of MOSs slightly improves the impact strength of composites.
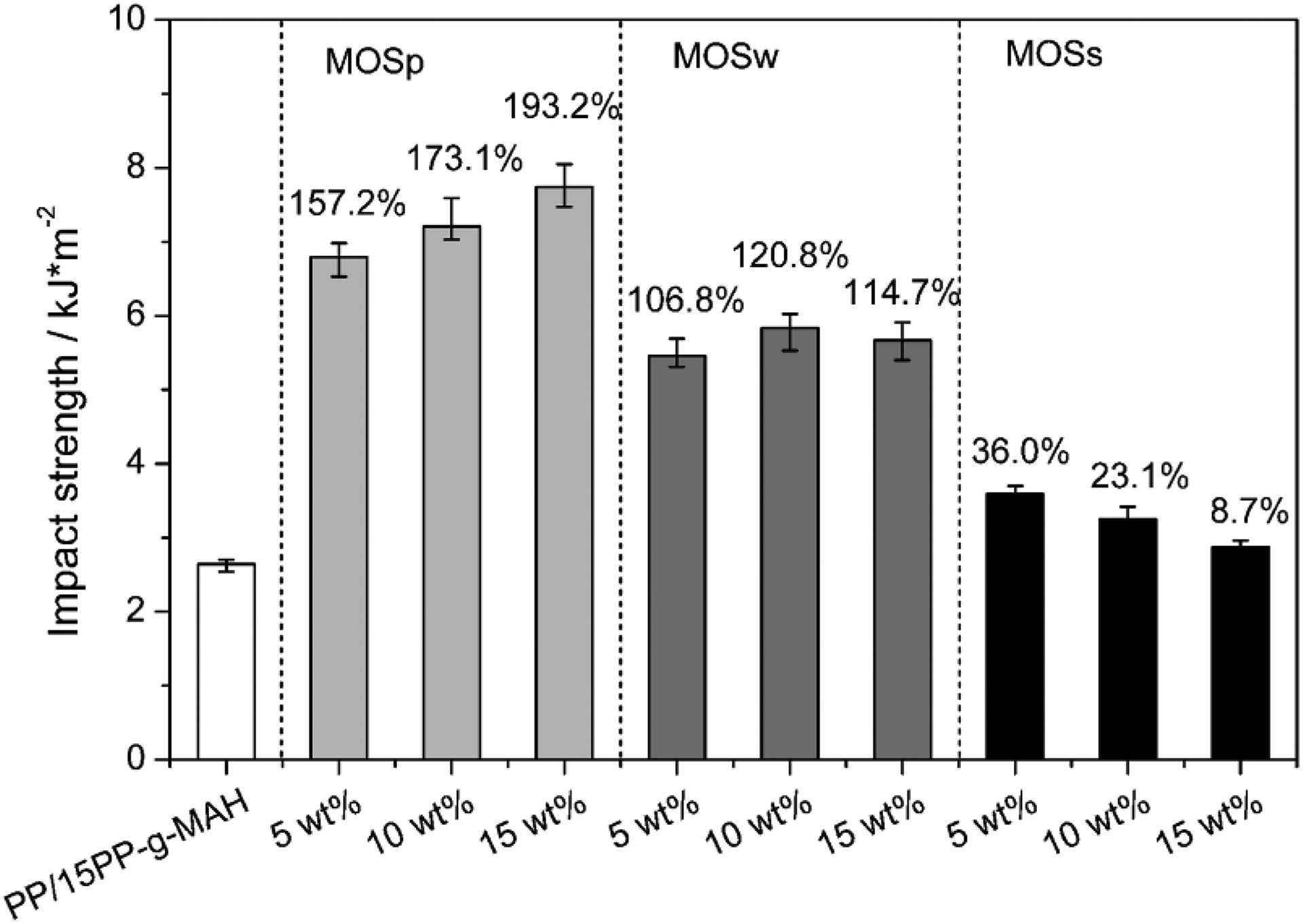
Variation of the impact strength of composites filled with different MOS. The numbers above the bars represent the changes in percentage with respect to the PP/15PP-g-MAH. PP-g-MAH: maleic anhydride-grafted polypropylene; MOS: magnesium oxysulfate.
Rheology analysis
Rheology analysis is a promising method to investigate the microstructure of composites because the viscoelastic response is not only related to the short-range structure of fillers but also reflected the long-range interaction between fillers and matrix. 34,35 Figure 6 shows the variation of complex viscosity (η*) for neat PP and its composites in the frequency range of 0.01–100 rad s−1. At low frequencies, η* of MOSp- or MOSs-containing composites present obvious “plateau region,” which is not much different from neat PP and PP/15PP-g-MAH. This typical phenomenon of molten polymers suggests that the addition of MOSp or MOSs does not alter the intrinsic quality of PP matrix. 36 However, MOSw-reinforced composites show weakening “plateau region,” especially for the PP/15PP-g-MAH/15MOSw composite, whose “plateau region” completely disappears at low frequencies. This apparent yield stress can be attributed to the particle–particle contact effect of MOSw, and the specific quantify of apparent yield stress (σ y) can be determined by applying the modified four-parameter Cross model 24 to the experimental data

where σ y is the yield stress and b (Pa·s), c (s), and d (dimensionless) are model parameters. The curve fitting results are plotted as solid lines in Figure 6, and the relevant model parameters are listed in Table 2. It is worth noting that composites containing MOSw show higher yield stress values than composites containing MOSp or MOSs. For example, the yield stress value of PP/15PP-g-MAH/15MOSw composite is up to 1600.49 Pa. Hence, it is considered that MOSw formed a strong network within the PP matrix as a result of suitable dimension. At high frequencies, PP/15PP-g-MAH/15MOSw composite shows more pronounced “shear-thinning” behavior, which could be ascribed to the destruction of the MOSw network at high frequencies shearing.
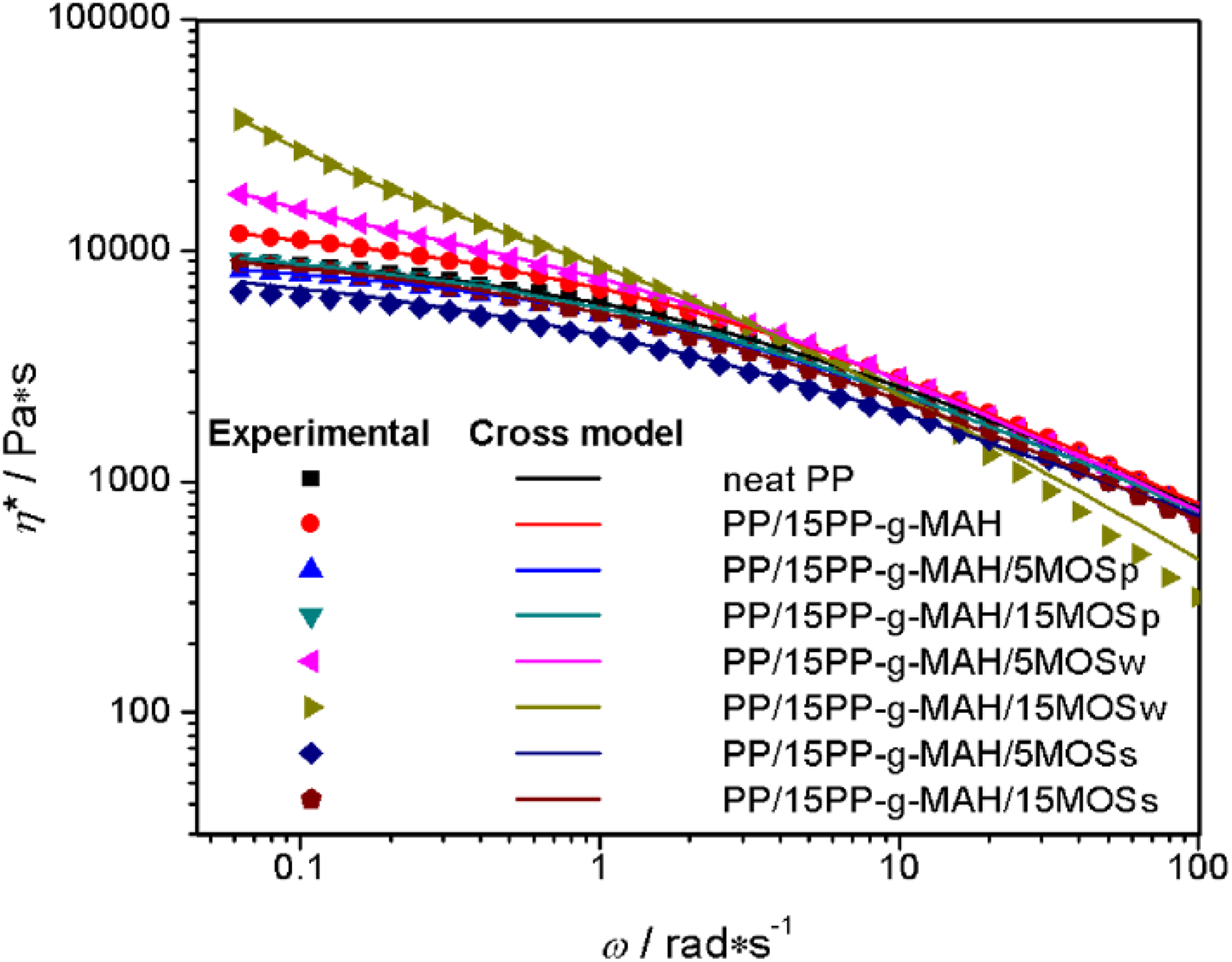
Plots of complex viscosity versus angular frequency for neat PP and its composites (symbols) and the curve fitting in the Cross model (solid lines). PP: polypropylene.
Values of the parameters in the Cross model used for curve fitting of the experimental complex viscosity.
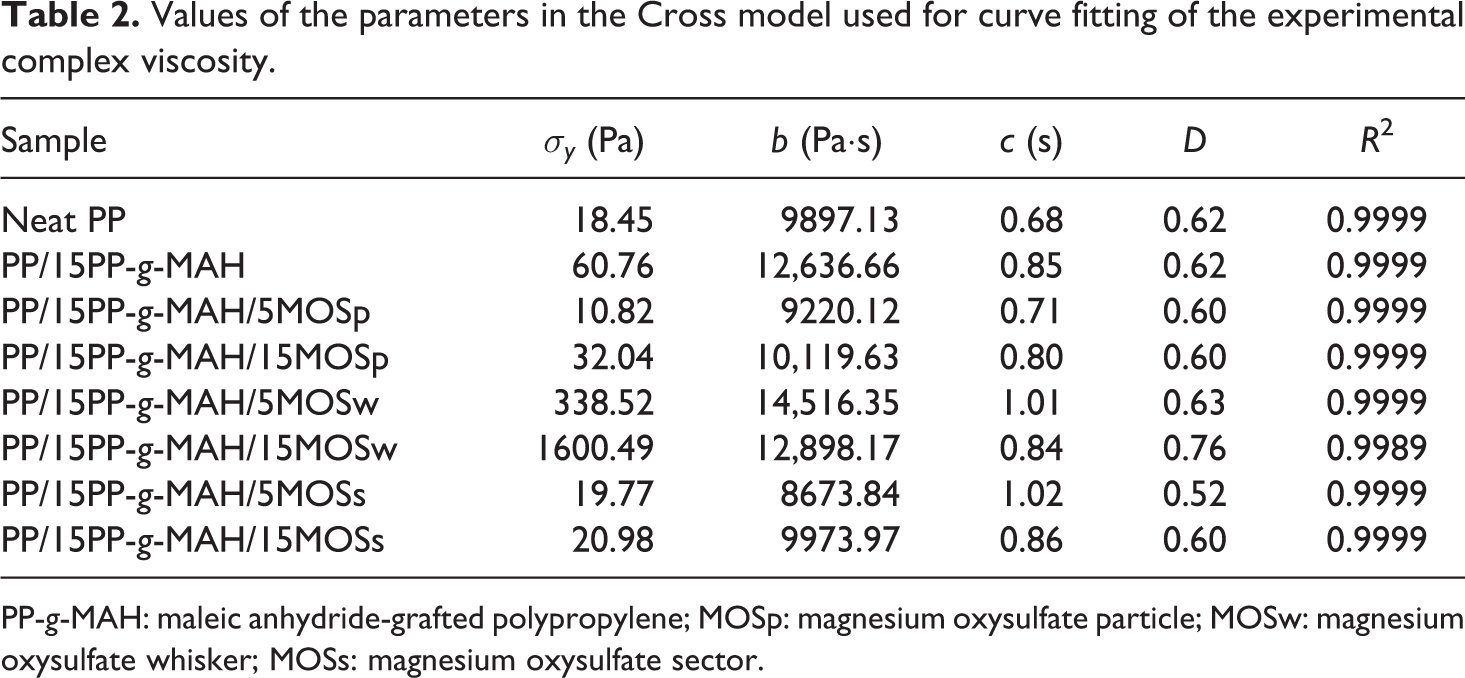
PP-g-MAH: maleic anhydride-grafted polypropylene; MOSp: magnesium oxysulfate particle; MOSw: magnesium oxysulfate whisker; MOSs: magnesium oxysulfate sector.
In theory, storage modulus (G′) and loss modulus (G″), respectively, reflect the elastic property of microstructures and the intermolecular interaction and molecular chains migration of molten polymers. Figure 7 shows, for neat PP and its composites, the dependence of G′ and G″ variation on frequency. At low frequencies, both G′ and G″ increase with the amount of MOSw but not for MOSp- or MOSs-contained samples. There is a “plateau region” appeared in G′ for PP/15PP-g-MAH/15MOSw sample. These phenomena imply that adding MOSw significantly improves the elasticity of PP matrix but also hinders the migration of PP molecular chains. 24,36 That is to say PP matrix converts from “liquid-like” behavior to “solid-like” behavior, probably due to the formation of MOSw network.
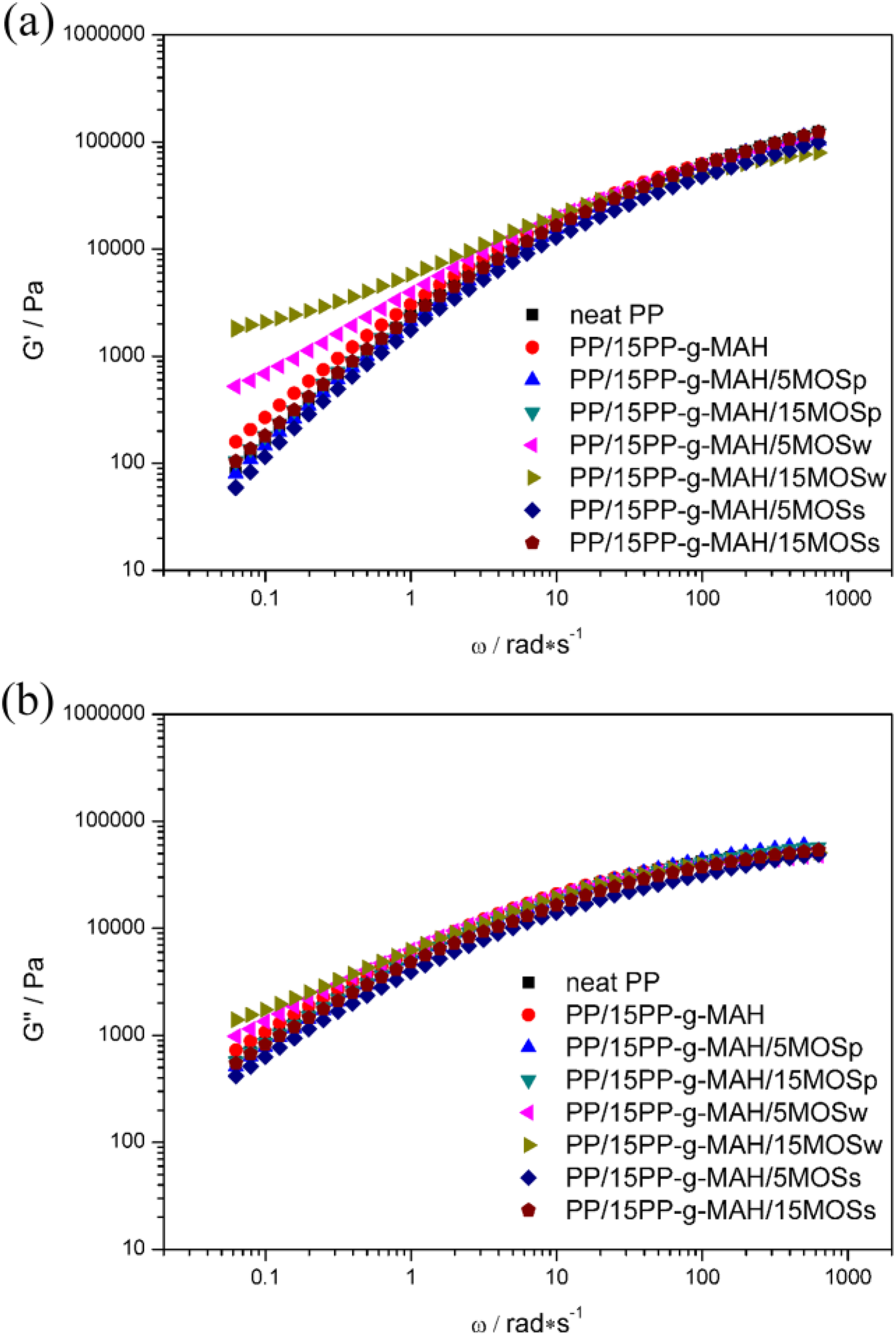
Storage modulus (a) and loss modulus (b) versus frequency of neat PP and its composites at 190°C. PP: polypropylene.
The relationship between G′ and G″ of neat PP and its composites is also investigated, as shown in Figure 8(a). Obviously, the addition of MOSp or MOSs to the PP melt does not change the G′–G″ curve, which is almost superimposed with the neat PP curve. However, the curves of MOSw containing composites gradually deviate from those of neat PP with the increase of MOSw content. Specific to PP/15PP-g-MAH/15MOSw composite, a significant deviation is observed. This deviation phenomenon again confirms the transition from “liquid-like” to “solid-like” when MOSw is added. 36,37
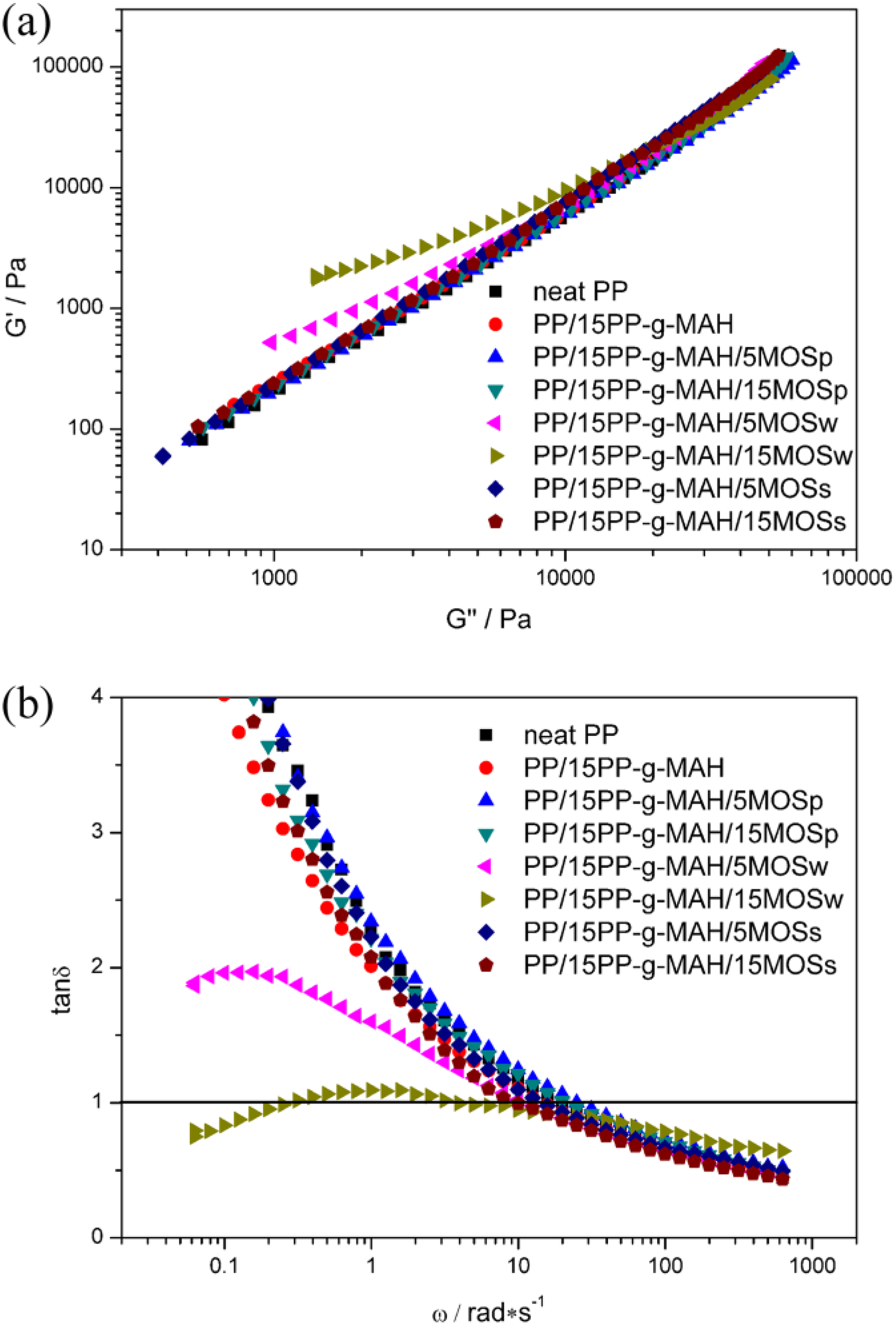
Plots of G′ versus G″ (a) and tan δ versus frequency (b) for neat PP and its composites at 190°C. PP: polypropylene.
The plots of damping factor (tan δ) versus frequency (ω) of neat PP and its composites are presented in Figure 8(b). It is reported that the formation of filler network in polymer matrix occurs at the two crossover frequencies (tan δ = 1). 24 As for neat PP, the crossover frequency is about 19.9 rad s−1 and changes little after introducing MOSp or MOSs. PP/15PP-g-MAH/5MOSw composite still shows only one crossover frequency; however, the value decreases to 9.9 rad s−1. The decrease of crossover frequency implies the retardation of PP chain relaxation. As for PP/15PP-g-MAH/15MOSw composite, two crossover frequencies are observed at 3.2 and 0.32 rad s−1, respectively, suggesting that an MOSw network has been formed in the PP/15PP-g-MAH/15MOSw composite. In one word, the formation of continuous network greatly enhances the mechanical strength of PP matrix.
Conclusion
In this work, the effects of three kinds of MOS fillers (MOSp, MOSw, and MOSs) on the crystallization, mechanical, and rheological properties of polypropylene were investigated. SEM micrographs show that all the MOS fillers disperse homogeneously in PP matrix and display vague and fuzzy interfaces. However, MOSs, analogous agglomerations, usually leads to the stress concentration and microcrack initiation, which negatively affects the mechanical properties of composites. WXRD and PLM observation both indicates the formation of β-PP in MOSp added composites, which greatly improves the impact strength of PP composites. Besides, PLM photographs also show that the diameters of PP spherulites decrease in the presence of MOSp, MOSw, and MOSs, especially for MOSp. For these reasons, the incorporation of MOSp into PP matrix greatly improves the impact strength and least lowers the nominal strain at break. The tensile test shows that incorporation of MOSw greatly improves the yield strength and Young’s modulus of the PP matrix. Except for the rigidity of MOSw, the microstructure of composites is another important reason for enhancing tensile properties. Rheological property measurements suggest that incorporation of 15 wt% of MOSw generates a continuous network in the PP matrix. The formation of MOSw network results in a transition from “liquid-like” PP matrix to “solid-like” composites, which greatly impacts the tensile properties of PP composites.
Supplemental material
Supplemental Material, Supporting_Information - Effect of magnesium oxysulfate (MOS) morphology on the crystallization, mechanical, and rheological properties of polypropylene/MOS composites
Supplemental Material, Supporting_Information for Effect of magnesium oxysulfate (MOS) morphology on the crystallization, mechanical, and rheological properties of polypropylene/MOS composites by Li Dang, Xueying Nai, Xin Liu, Zhihui Lv and Wu Li in Journal of Thermoplastic Composite Materials
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the “135” Foundation of the Qinghai Institute of Salt Lake of the Chinese Academy of Sciences (No. Y460321111) and the Developing Foundation of the School of Chemical Engineering, Qinghai University (No. QHHY-2017-01).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
