Abstract
Layered double hydroxides (LDHs) were synthesized by a coprecipitation method. The synergistic flame retardant effect of nano-silicon dioxide (nano-SiO2) on ethylene vinyl acetate (EVA)/LDHs composites was studied using limiting oxygen index (LOI), cone calorimeter test (CCT), scanning electron microscopy (SEM), and thermogravimetric analysis (TGA). Results showed that the LOI values of the EVA/LDHs/nano-SiO2 composites were basically higher than that of the EVA/LDHs composite, and the EVA composite with 48% LDHs and 2% nano-SiO2 reached an LOI value of up to 31.2%. The CCT results indicate that the addition of nano-SiO2 greatly reduced the heat release rate, total heat release, mass loss, smoke production rate, total smoke release, and smoke factor. The morphology and structures of residues investigated by SEM gave positive evidence that char layers formed from the EVA/LDHs/nano-SiO2 composites were improved. The TGA data showed that the EVA/LDHs/nano-SiO2 composites show a higher thermal stability than the EVA/LDHs composites.
Keywords
Introduction
Layered double hydroxides (LDHs) are a relatively new class of inorganic layered crystalline materials attracting considerable attention as extraction sorbents, stabilizers, drug delivery agents, flame retardants, and catalysts due to their special structure and unique properties. 1 –5 The general formula of LDHs was often written as [MII 1−x MIII 1−x (OH)2] x +(A n −)x/n ·m H2O, where MII is a divalent metal cation, MIII is a trivalent metal cation, and A stands for n valence inorganic or organic acid anions. 6 –8 There are many methods to prepare LDHs, such as coprecipitation, 9,10 calcination–rehydration, 11,12 the exchanges of the ion, 13,14 and hydrothermal synthesis method. 15 The LDHs as a kind of halogen-free flame retardant reagent have been extensively used in the flame retardation of polymers due to their advantages such as low smoke, no toxicity, and no corrosive gas generation. 16 –19 However, its fatal disadvantages are very large usage amount, which lead the mechanical properties of flame retardant materials to drop down sharply due to the poor compatibility between LDHs and polymer resin. 16,17 In order to minimize this effect, many works show that the synergistic effects of some environment friendly flame retardant agents (such as ammonium polyphosphate, 8,20,21 red phosphorus, 17,22 and titanium dioxide 23 ) with LDHs can enhance flame retardancy and reduce the high loading level of LDHs.
Silicon dioxide (SiO2), with high toughness, high strength, and abrasion, is one of reinforced filling materials that are extensively used. 24 It usually used as an enhancing agent in thermoplastic polymers to increase the mechanical properties. 25 Kashiwagi et al. 26 studied the flame retardant mechanism of SiO2 in polypropylene composites. Fu and Qu 27 investigated the synergistic flame retardant mechanism of fumed silica in ethylene vinyl acetate (EVA)/magnesium hydroxide composites. Pan et al. 28 reported the synergistic effect of nano-SiO2 and ammonium polyphosphate on flame retardancy of wood fiber–polyethylene composites. However, as far as we know, few research reports have focused on the synergistic effect of nano-SiO2 and LDHs on flame retardancy of EVA composites.
In this work, LDHs were synthesized and characterized by X-ray diffraction (XRD). Moreover, the synergistic effects of nano-SiO2 and LDHs on the flame retardant and smoke suppression properties of the EVA/LDHs/nano-SiO2 composites have been studied using cone calorimeter test (CCT), limiting oxygen index (LOI), scanning electron microscopy (SEM), and thermogravimetric analysis (TGA).
Experimental
Materials
EVA containing 18 wt% vinyl acetate was bought from Beijing Eastern Petrochemical Company Ltd (China). Nano-SiO2 (30 nm) was purchased from Aladdin industrial corporation (China). Other reagents were the analytical reagent and were used for the synthesis of LDHs. All of these materials were used as received without any further purification.
Synthesis of LDHs
LDHs were synthesized by a coprecipitation method. Firstly, salt solution (with Mg/(Al+Fe) molar ratio of 3:1 and total metal ion concentration of 1.0 mol·L−1) was prepared by dissolving the required amount of MgCl2·6H2O, FeCl3·6H2O, and AlCl3·6H2O in 100 mL of deionized water at room temperature. Meanwhile, alkali solution was also prepared by dissolving the required amount of NaOH and Na2CO3 in 100 mL of deionized water at room temperature. Then, the above solutions were separately added dropwise into a beaker containing suitable deionized water with electric stirring at 80°C. During the coprecipitation reaction, the pH in the reaction mixture was maintained at approximately 8–9. Finally, the suspension was filtered and washed repeatedly with deionized water until the filtrate liquor was neutral. The filter products were dried at 80°C for 24 h to obtain LDHs. Then, the LDHs were grinded into 200-mesh pass particles (with a particle size of about 74 μm) as flame retardant additives. It contains O (62.75 wt%), C (9.35 wt%), Na (0.13 wt%), Mg (18.13 wt%), Al (3.89 wt%), Cl (0.17 wt%), and Fe (5.58 wt%) by an Oxford MAX-80 energy dispersive X-ray spectrometer.
Preparation of samples
All compositions were melt-compounded with a mixer at 130°C for 10 min. And the mixing was carried out at 30 r min−1. Then, the obtained composites were hot-pressed at 130°C for 10 min under a pressure of 10 Mpa into sheets of suitable thickness (3 mm). Finally, the compressed sheets were cut into suitable size for fire testing according to the standards mentioned in the following part. The formulations of the EVA composites are listed in Table 1.
Formulations of all samples.
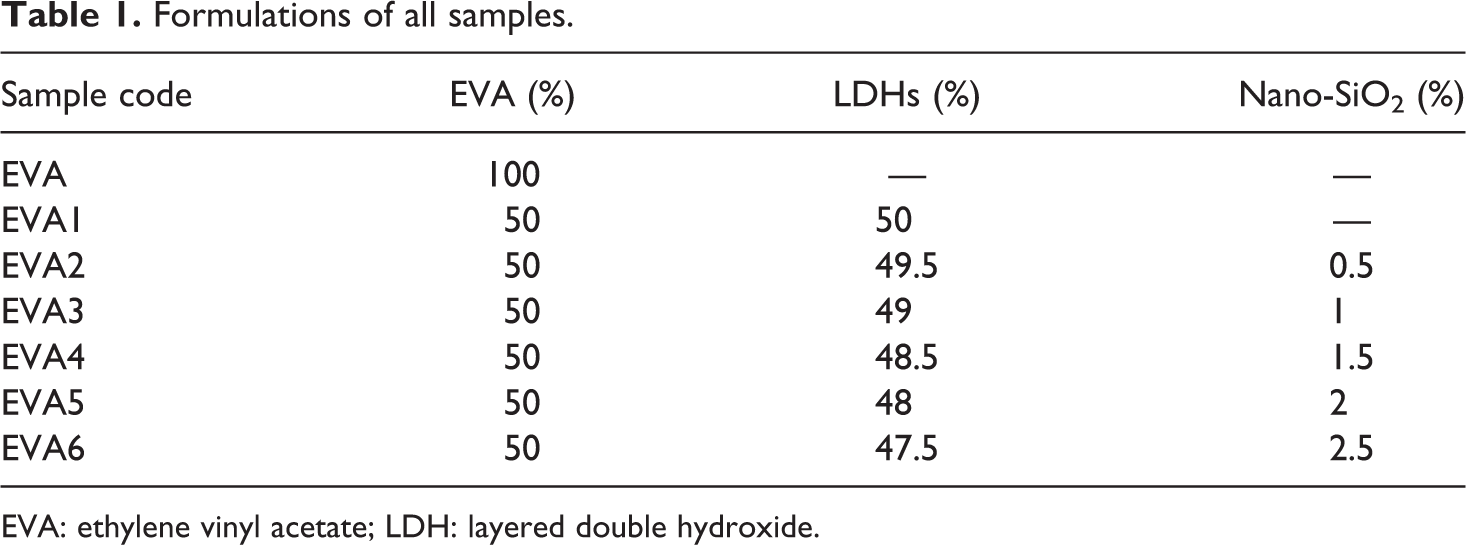
EVA: ethylene vinyl acetate; LDH: layered double hydroxide.
Characterization of LDHs
XRD analysis
The XRD pattern of LDHs was recorded using a Philips X’Pert Pro Super apparatus (Nicolet Instrument Co., Madison, Wisconsin, USA) with copper Kα radiation (λ = 1.5418 Å). The test was carried out under the tube voltage of 40 kV and current of 150 mA at room temperature, and the 2θ diffractive angle was scanned from 5° to 65° with the step size of 0.02° steps and scanning rate was 8° min−1.
Measurements of samples
Cone calorimetry test
The CCT (Stanton Redcroft, UK) was performed under a heat flux of 50 kW m−2 according to ISO 5660 standard procedures. Each specimen of dimensions 100 × 100 × 3 mm3 was wrapped in aluminum foil.
LOI analysis
LOI measurements were carried out using an HC-2 oxygen index meter (Jiangning Analysis Instrument Company, China) according to ASTM D 2863.
SEM analysis
The surface morphology of char residues of samples after CCT was investigated by SEM (Hitachi, Japan) using a Hitachi X650 SEM.
Thermogravimetric analysis
TGA was carried out under nitrogen atmosphere on a TG209 F1 apparatus (Netzsch Company, Germany) at a heating rate of 10°C min−1. About 5 mg of sample, placed in a crucible sample holder, was heated from 30°C to 800°C.
Results and discussion
XRD characterization of LDHs
Figure 1 presents the XRD pattern of LDHs. As shown in Figure 1, the (003), (006), (009), (015), (018), (110), and (113) diffraction peaks of LDHs are consistent with hydrotalcite lattice structure at around 12°, 24°, 36°, 39°, 46°, 61°, and 62°, respectively. 29,30 And the diffraction peaks of the sample are sharp and the baselines are low, being indicative of relatively well-formed crystalline layered structures. 31 In addition, 2θ values of (003), (006), and (009) crystal planes have good multiple relationship, showing the LDHs synthesized have well-formed crystalline layered structures; around 61°, diffraction peaks of (110) and (113) crystal planes are obviously separated, indicating that negative ions among layers have high degree of regularity, and the LDHs synthesized have good symmetry. 19 It can be concluded that LDHs can be synthesized successfully by a coprecipitation method.
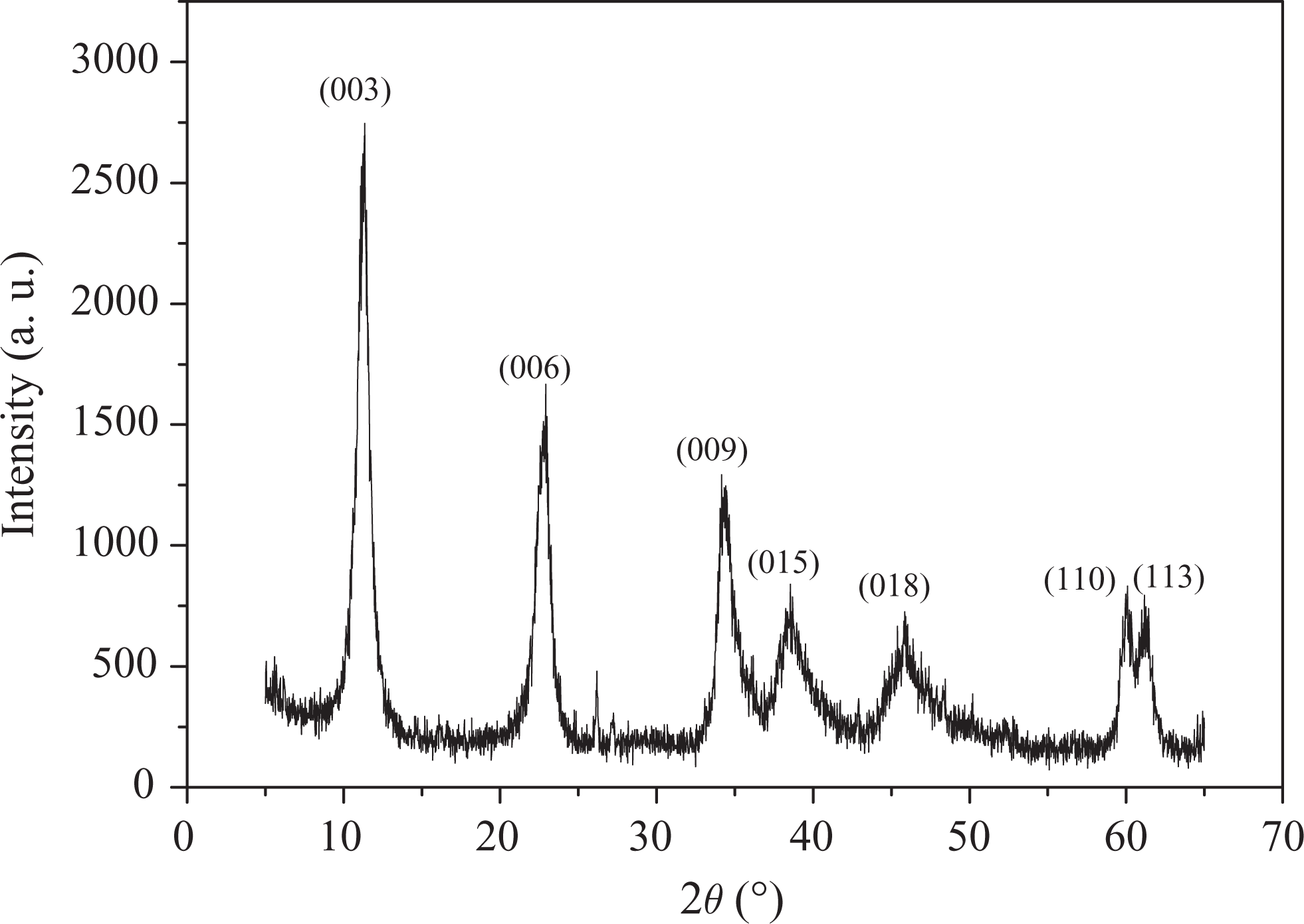
The XRD pattern of LDHs. XRD: X-ray diffraction; LDH: layered double hydroxide.
CCT of the EVA/LDHs/nano-SiO2 composites
The CCT based on the oxygen consumption principle has been widely used to evaluate the flammability properties of materials because its results correlate well with those obtained from large-scale fire tests and can be used to predict the fire performance of materials in real fires. 32 –34 It provides comprehensive insights into fire risk via parameters such as the peak of heat release rate (pHRR), the heat release rate (HRR), the total heat release (THR), the time to pHRR, the time to ignition (TTI), the mass loss, and the time to flame out. 35
Heat release rate
Flame retardant properties of the samples were tested with cone calorimeter. The HRR, especially pHRR, measured by CCT is a very important parameter as it expresses the intensity of a fire, which in turn determines other parameters. 27,36
The HRR of pure EVA and EVA composites as a function of burning time is shown in Figure 2, and the correlated data are given in Table 2. As shown in Figure 2 and Table 2, the HRR curve of pure EVA is very narrow and sharp with a pHRR of 1510.1 kW m−2, which indicates that pure EVA is easily flammable polymeric material. In contrast, the HRR curves of all the flame retardant EVA composites are obviously declined, and the combustion processes are all prolonged markedly in comparison with that of pure EVA resin. Additionally, the combustion tended to be mild after the introduction of flame retardants. For EVA1, its maximal pHRR has an about 82% reduction in comparison with that of pure EVA; the TTI and burning time are increased to 41 and 677 s, respectively. This may be due to the decomposition of LDHs and the formation of char layers. LDHs can decompose into H2O, CO2, and mixed metal oxides. On the one hand, H2O and CO2 can decline the temperature and affect the diffusion of oxygen and volatile products; on the other hand, the presence of alkaline metal oxides can help acid-catalyzed dehydration of polymers and consequently promote a charring on the surface of the composite. The protective char residue can reduce the heat transfer between the EVA matrix and the heat source by absorbing the primary heat from the source and disturbing the effective heat convection at the surface of the substrate. 18,19,37 For EVA2-EVA6, with the increase of nano-SiO2, HRR obviously decreases; the HRR of EVA6 (with a maximal pHRR of 180.8 kW m−2) is the lowest among all samples. It should also be found that the TTI and flame out are almost prolonged for EVA2-EVA6. EVA2 takes more time to be ignited than the other composites; the time to flame out for EVA6 is prolonged by 75 s in comparison with that of EVA1. These phenomena indicate that an appropriate amount of nano-SiO2 is needed for the improvement of the flame retardancy due to the synergism effect between LDHs and nano-SiO2. Nano-SiO2 can catalyze the dehydration and cross-linking reactions between polymer and LDHs, thus promoting the formation of char residues and improving char structure, which leads to a significant improvement on the flame retardancy of EVA composites. 28
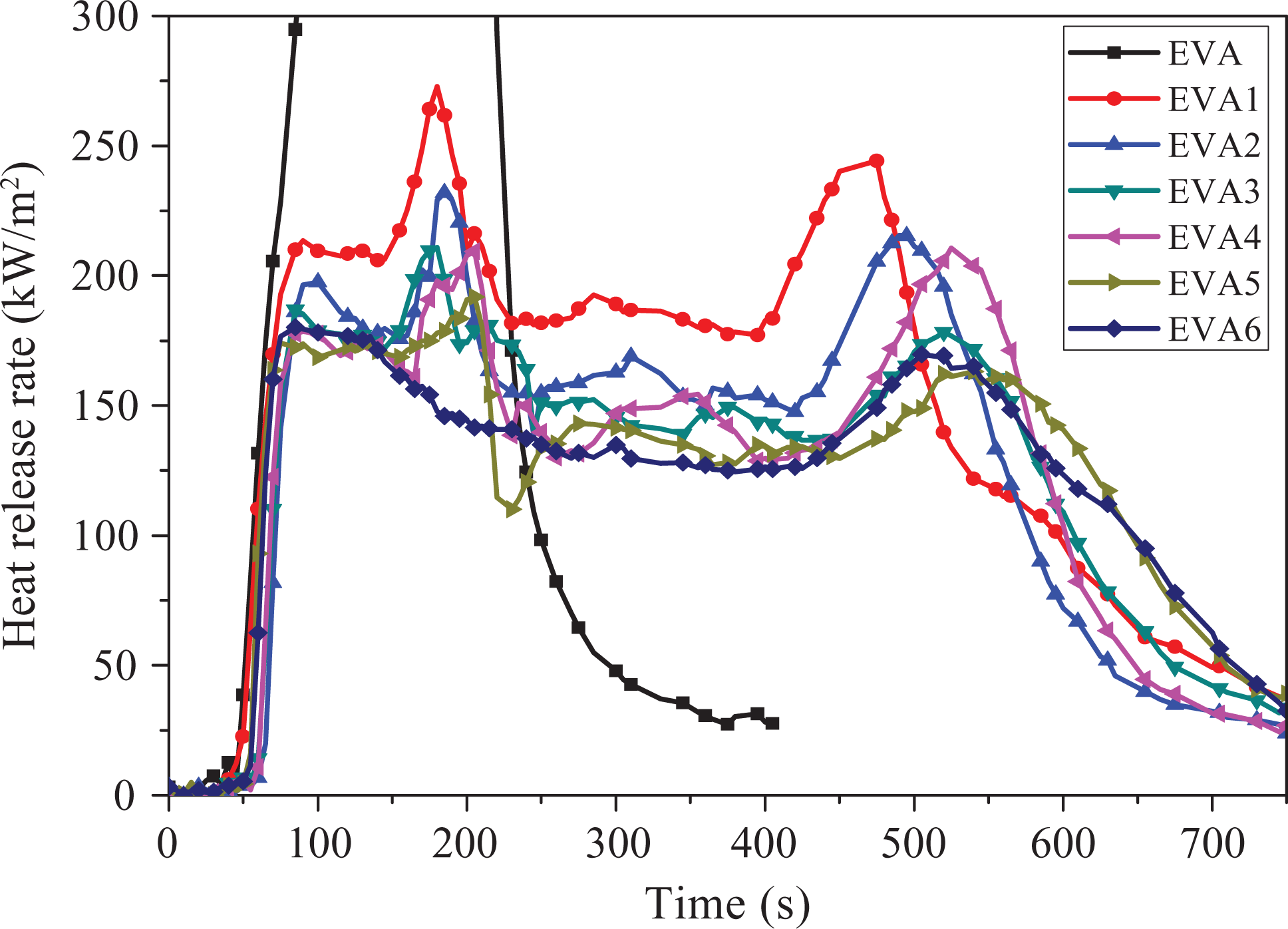
Heat release rate curves of samples.
The data from CCT.
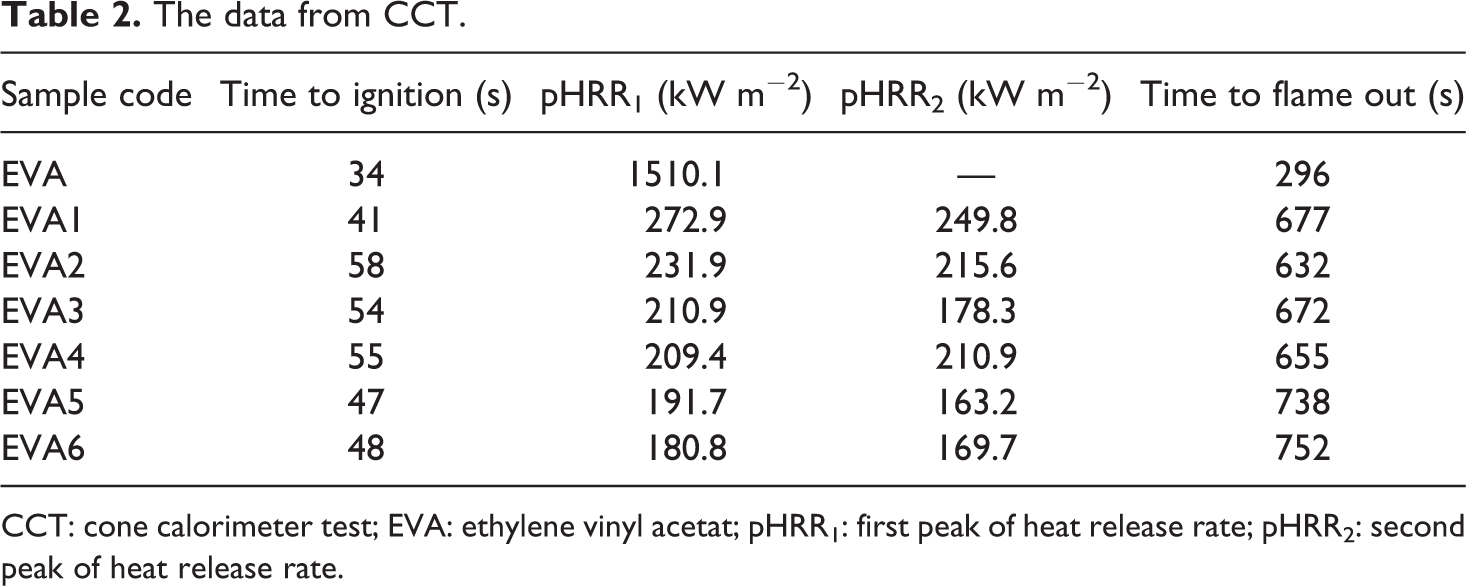
CCT: cone calorimeter test; EVA: ethylene vinyl acetat; pHRR1: first peak of heat release rate; pHRR2: second peak of heat release rate.
At the same time, it is interesting to find from Figure 2 that there are two peaks in the HRR curves for EVA1-EVA6. It can be easily understood that the first peak of HRR curves (pHRR1) is due to the development of the protective char. After the first peak, the HRR curve forms a plateau in which the increase in HRR is suppressed due to the presence of efficient protective char. The second peak of HRR curves (pHRR2) may be caused by cracking char or increase in the effective pyrolysis. 38
Total heat release
Figure 3 shows the THR of the flame retardant EVA composites. The slope of THR curve can be assumed as representative of fire spread rate. 16,17 It can be found from Figure 3 that the flame spread rate of samples (EVA1 to EVA6) decreases apparently compared to EVA. Moreover, with the addition of nano-SiO2, the THR energy of samples (EVA2 to EVA6) further decreases than that of EVA1. Among all the samples, the THR of EVA6 is the lowest. This phenomenon could also be explained by the synergistic effect of flame retardance between LDHs and nano-SiO2.
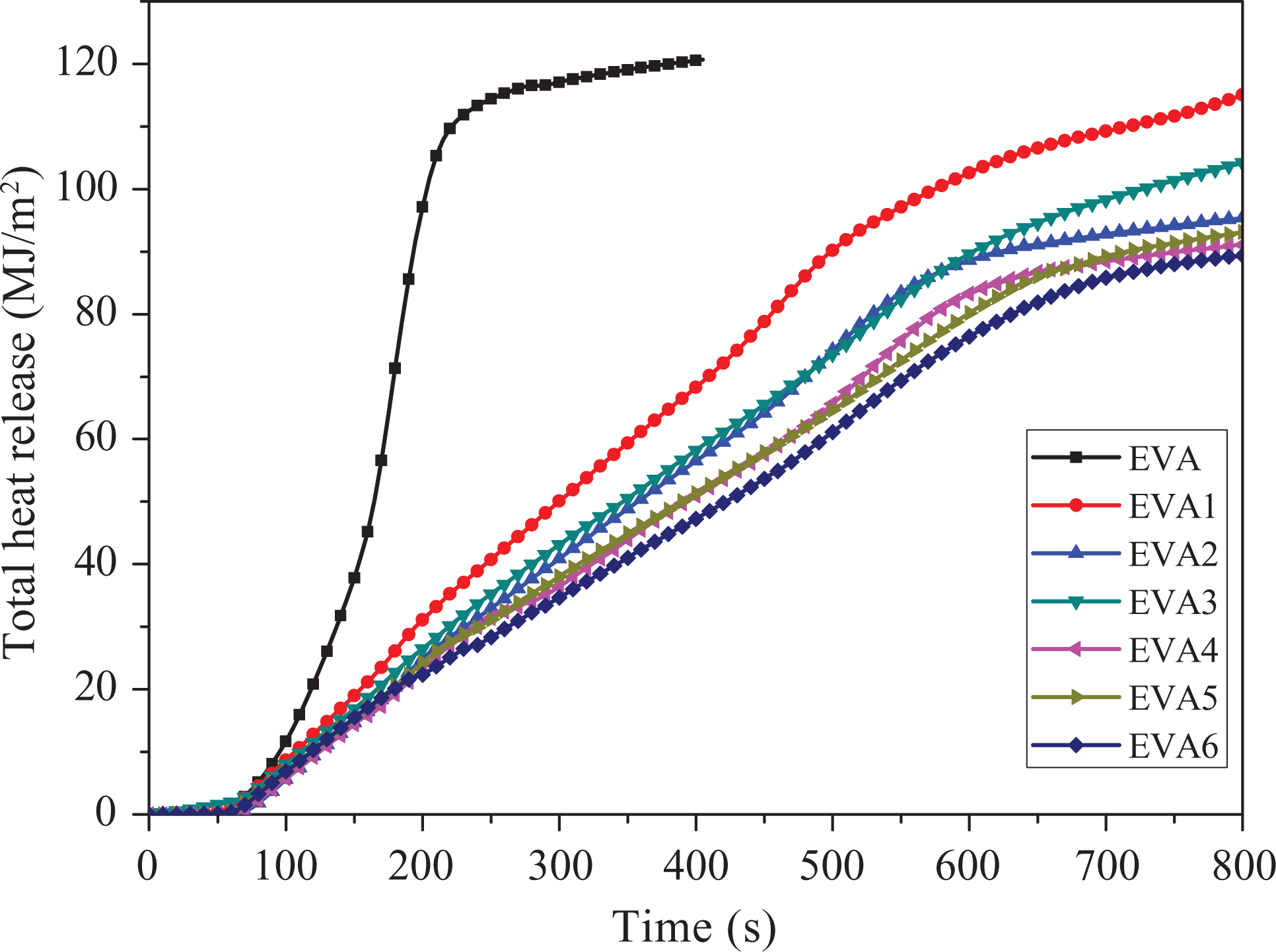
Total heat release curves of samples.
Mass
Figure 4 shows the mass of the char residues. It can be noticed in Figure 4 that all composites containing flame retardant have a higher weight of char residues than that of pure EVA. Moreover, with the increase of nano-SiO2, the weight of the char residues gradually increases, and the mass loss curve of EVA6 is the highest due to the synergistic effect of LDHs and nano-SiO2 on flame retardant properties, which is in accordance with the HRR results. During combustion, a compact char may occur on the surface of material, creating a physical protective barrier. At the time, nano-SiO2 effectively improve char structure, thus enhance the strength of the char layer, which leads to improved flame retardancy of samples.
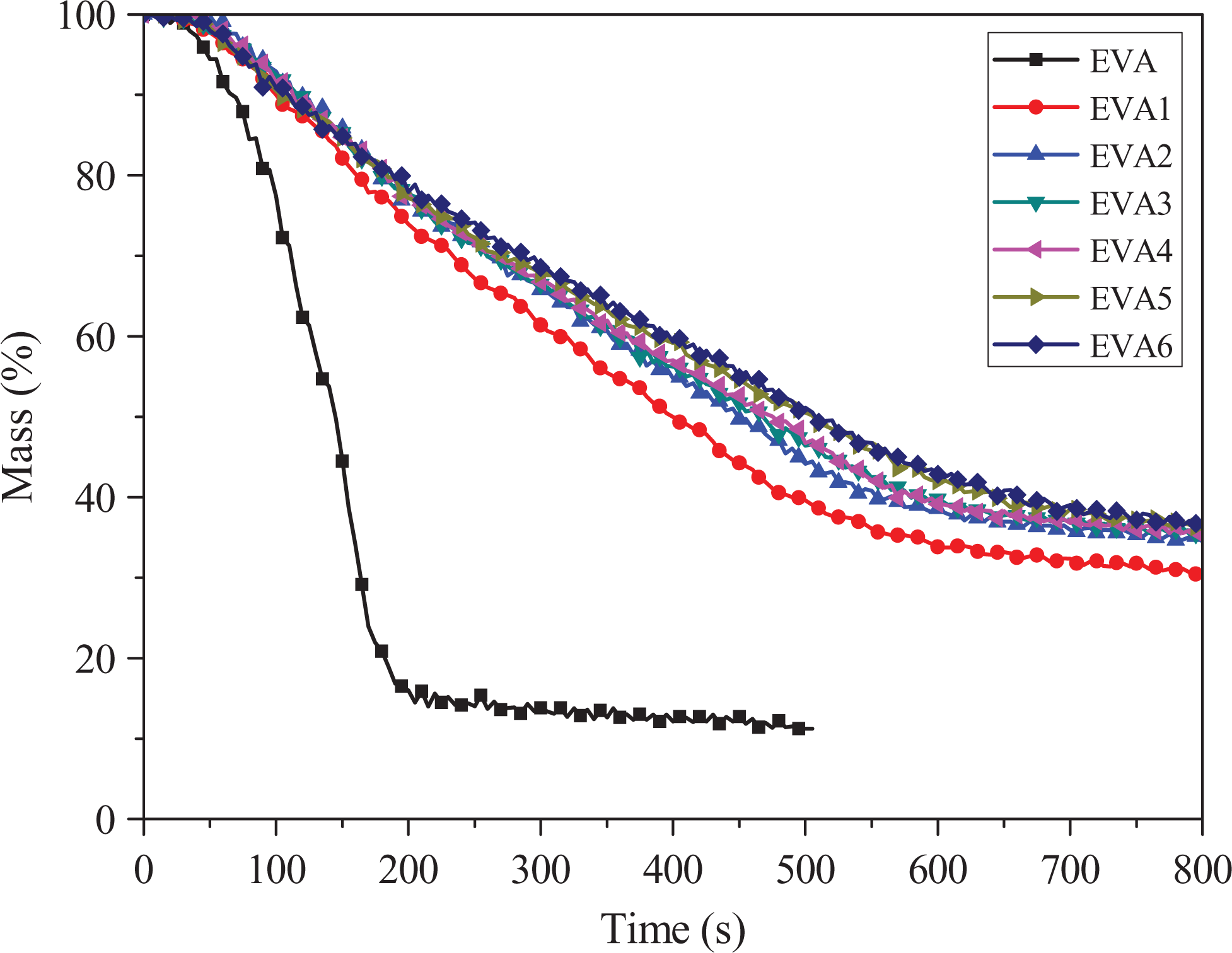
Mass loss curves of samples.
Smoke production rate
Evaluation of the fire performance often involves quantifying not only heat generation but also smoke generation as the smoke production rate (SPR). 38 It can be seen from Figure 5 that the peak SPR value of EVA is 0.063 m 2 s−1 at 155 s in the combustion process. Comparing with pure EVA, the peak SPR value of EVA1 is 0.024 m 2 s−1 at 455 s, which is much lower than that of EVA. It can be concluded that LDHs can obviously decrease the SPR of EVA composites. In addition, the SPR values of the EVA/LDHs/nano-SiO2 composites are all lower than that of EVA1 in the combustion process. For example, the peak SPR value of EVA5 is 0.015 m 2 s−1 at 520 s, which is the lowest one among all the samples. The main reason may be that nano-SiO2 could change the structure of char residue layer. The SPR may decrease in the formation process of carbon layer. Comparing EVA1 with the samples containing nano-SiO2, nano-SiO2 is an effective additive to suppress smoke production in the combustion process of EVA composites.
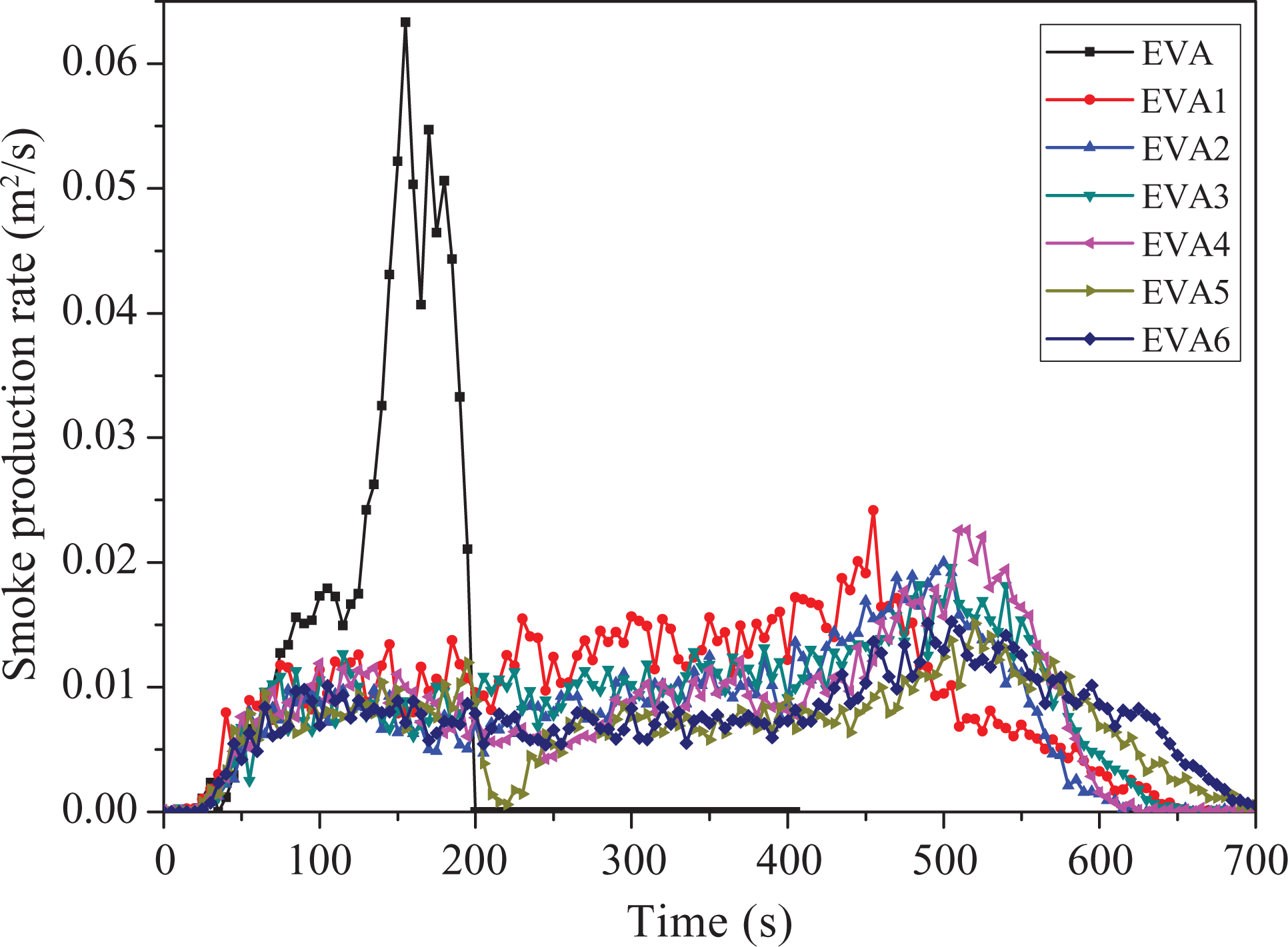
Smoke production rate curves of samples.
Total smoke release
The major factor leading to death is smoke and toxic substances released in most cases of real fire hazards. 39 Figure 6 shows the total smoke release (TSR) curves of EVA composites at a flux of 50 kW m−2 in the CCT. As is presented in Figure 6, the TSR value of pure EVA and EVA1 is 454.3 and 743.7 m 2 m−2 at the end of the experiments, respectively. The reason may be that the solid particles formed from the depolymerization of the polymers are mainly burned out to form gases but do not migrate directly into the gas phase to increase the amount of smoke. Comparing with EVA1, the samples containing nano-SiO2 have lower TSR value at the end of test, which is in accordance with the SPR results. And, the TSR value of EVA5 is only 550.8 m 2 m-2. It can be concluded that nano-SiO2 could help smoke suppression on the EVA/LDHs system, which can illustrated that nano-SiO2 can change the melt viscosity of EVA composites and the structure of char residue. So, the volatile products decreased, including the smoke particulates.
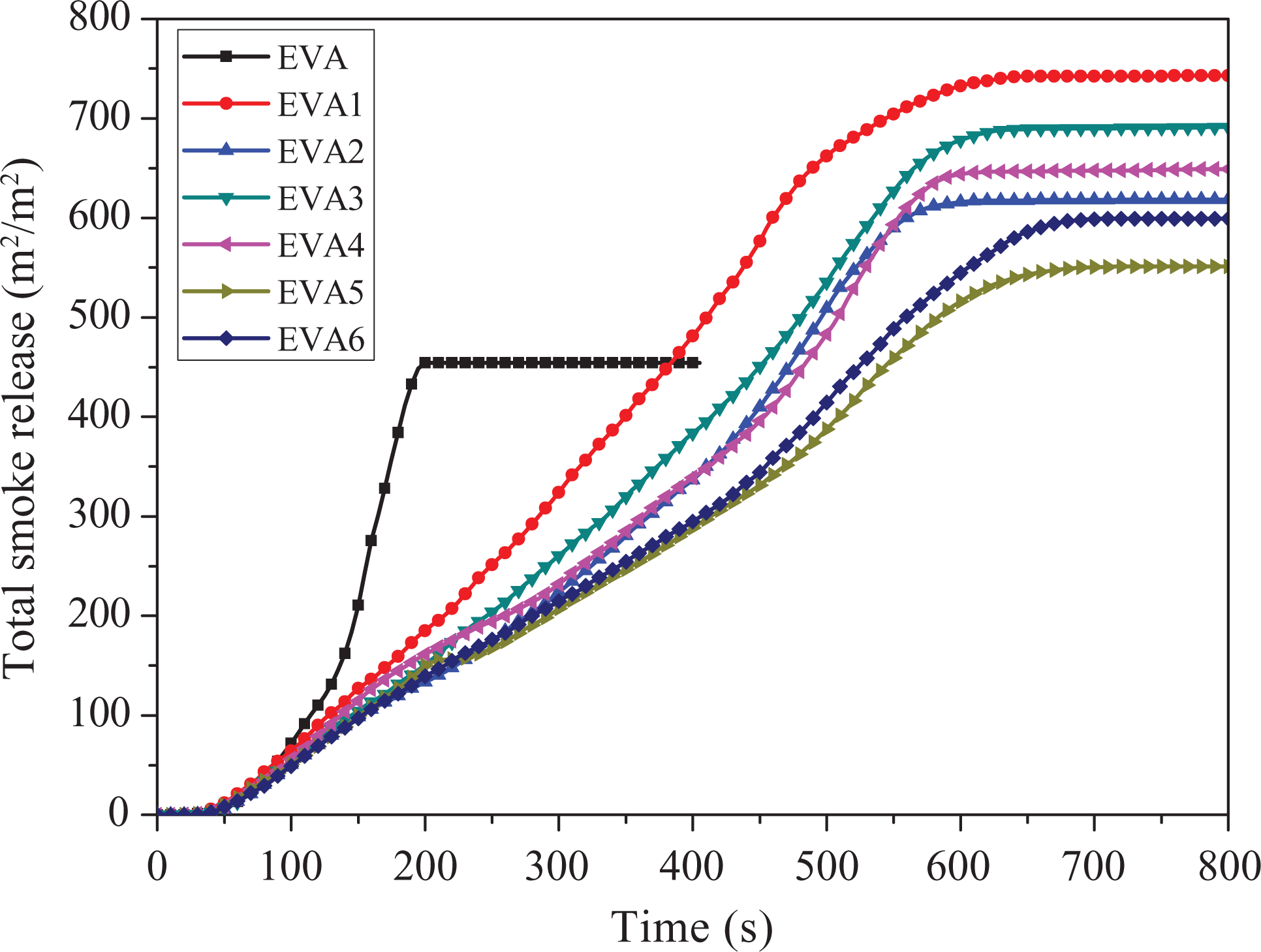
Total smoke release curves of samples.
Smoke factor
Figure 7 gives the smoke factor (SF) as a function of time for all samples. The value of SF is the product of pHRR and TSR. 40 At the end of the experiment, the SF value of EVA is up to 686.1 MW m−2. However, the SF value of EVA1 containing only LDHs (202.8 MW m−2) is much lower than that of EVA. Additionally, the addition of nano-SiO2 can significantly reduce the SF values of EVA composites, especially for EVA5 composites which had a lowest SF value (105.6 MW m−2), approximately 47.9% decrease relative to EVA1. That indicates that an appropriate amount of nano-SiO2 has positive effect on smoke suppression of EVA composites, which is in good agreement with the above results of SPR and TSR.
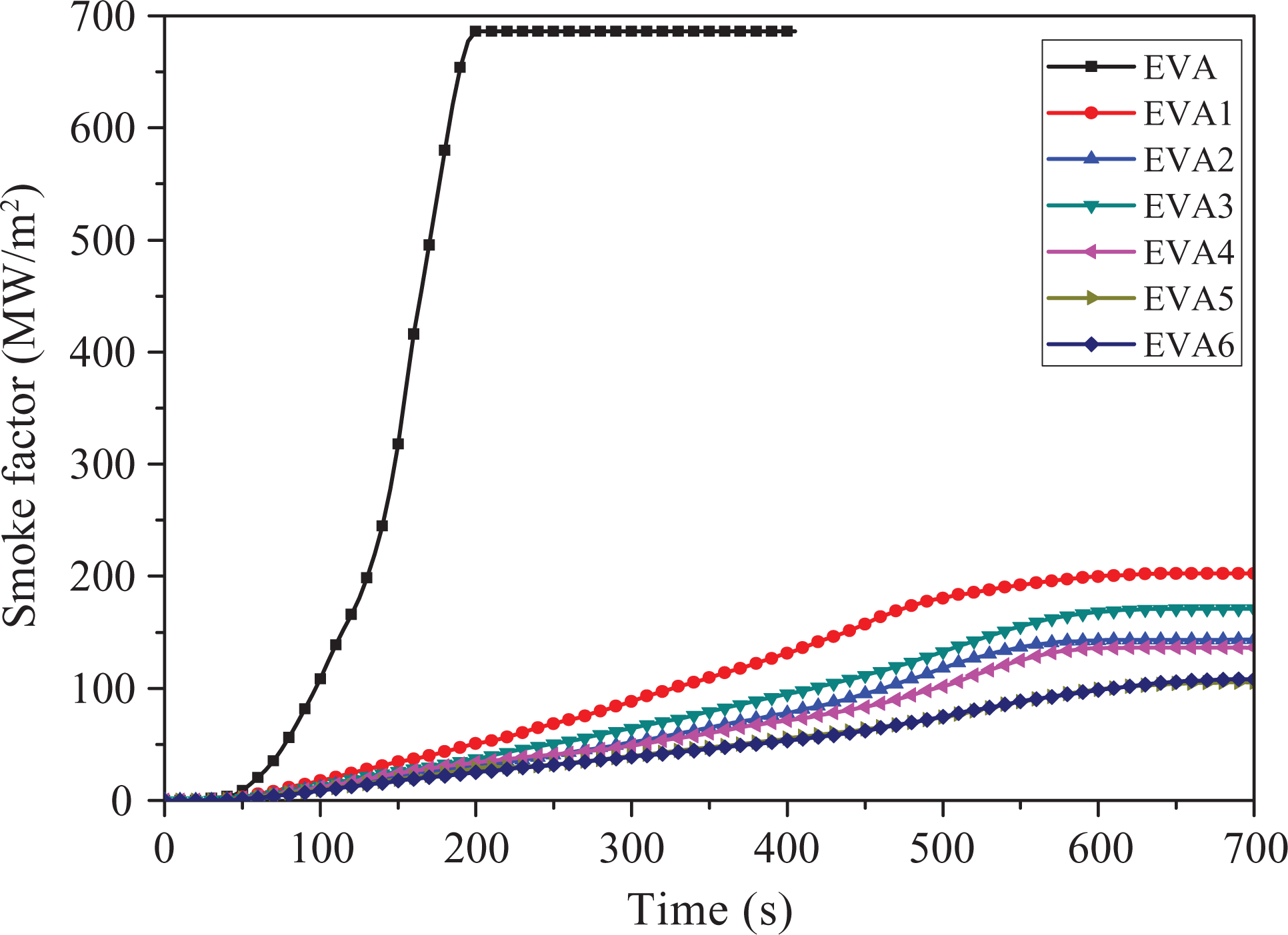
Smoke factor of samples.
SEM of char residues after CCT
In order to investigate the relationship between microstructure of char layer and flame retardancy performance, the morphologies of char residues left after cone calorimetry test were characterized by SEM, and the results are provided in Figure 8. It can be found from Figure 8 that many loose and separated particles are dispersed on the surface of the residues of EVA1, which can’t segregate oxygen and combustible gases effectively. However, the char residue of EVA6 is much compacter and denser than that of EVA1, which forms protective shields on the surface of the residues, thus inhibiting the transmission of heat and heat diffusion when exposed to flame or heat source. What’s more, the compact carbon layer can also prevent smoke release. The main reason is the addition of nano-SiO2 changing the structure of char residue. As a result, we can draw the conclusion that nano-SiO2 can promote the formation of char residue in the combustion process of EVA composites.

The SEM images of the charred residues: (a) EVA1 and (b) EVA6. SEM: scanning electron microscopy; EVA: ethylene vinyl acetate.
LOI values of the EVA/LDHs/nano-SiO2
The LOI is widely used to evaluate flame retardant properties of materials. Figure 9 presents the LOI values of the flame retardant EVA/LDHs/nano-SiO2 composites with different loading of nano-SiO2. From Figure 9, it can be seen that the LOI value of EVA1 increases rapidly from 19.2% for EVA to 28.3% for EVA1. This may be due to the decomposition of LDHs and the formation of char layers, which is helpful to LOI test. In addition, compared to EVA1 (with a LOI value of 28.3%), the LOI value of the EVA/LDHs/nano-SiO2 composites (EVA2 to EVA6) increases obviously to 28.8, 29.3, 30.4, 31.2, and 29.6%, respectively. These phenomena indicate that nano-SiO2 is very beneficial to LOI test due to the synergistic effects between nano-SiO2 and LDHs.
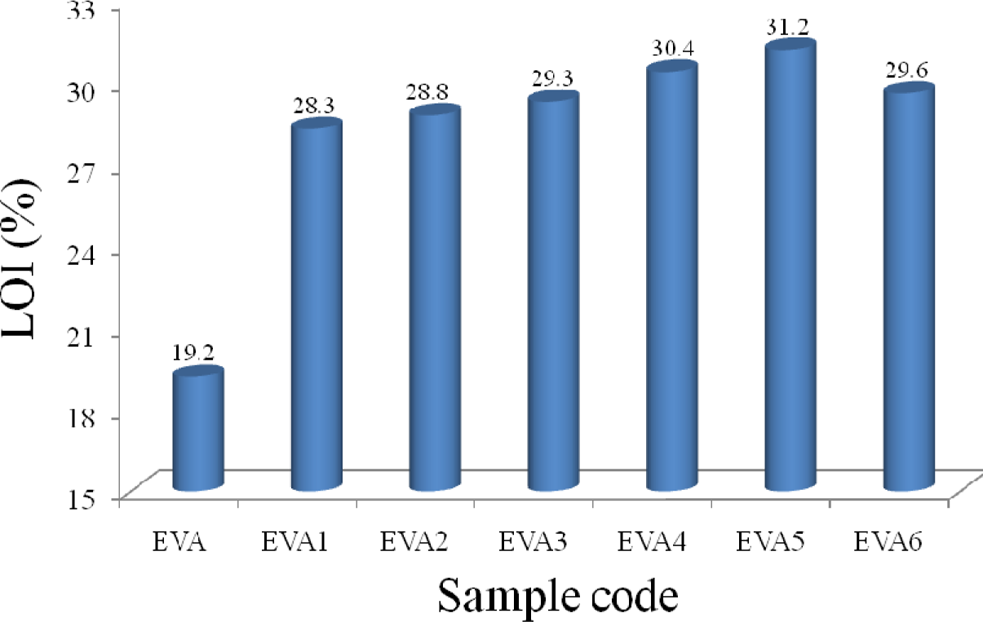
The LOI values of samples. LOI: limiting oxygen index.
Thermogravimetric analysis
The thermal stability of a polymeric material is extremely important when it comes to a flame retardant. 8 Figure 10 shows the thermogravimetry and differential thermogravimetry curves of samples EVA, EVA1, and EVA6 under nitrogen atmosphere. It can be found that EVA undergoes two degradation steps as shown in Figure 10. The first decomposition step is due to the loss of carboxylic acid; the second involves random chain scission of the remaining material, forming unsaturated vapor species, such as butene and ethylene. 41 EVA1 and EVA6 show three mass loss steps. The first step of the composites is attributed to the loss of loosely bound water in the interlayer space of LDHs in the composites. The second and the third steps belong to the simultaneous dehydroxylation and decarbonization of the lattice of LDHs, which overlap with the decomposition of the acetate groups in EVA side chains and the scission of the main chains of EVA. 16,17,41
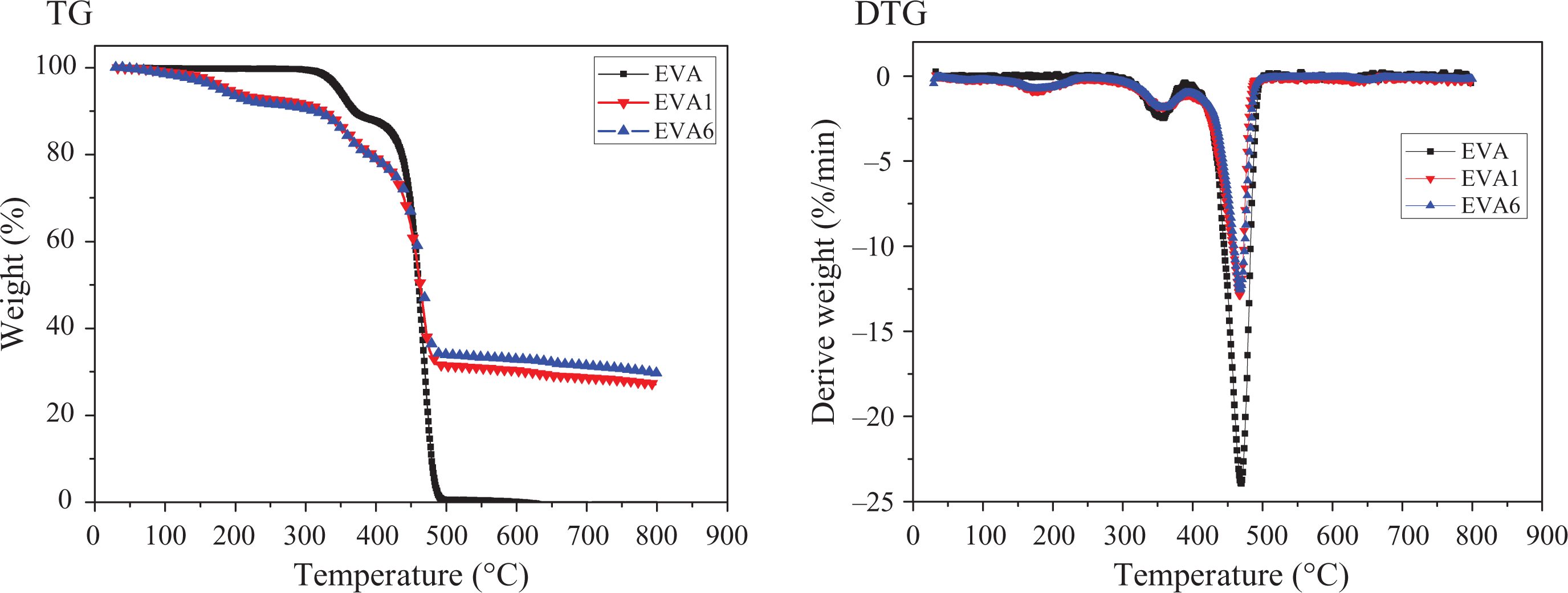
The TG-DTG curves of the EVA composites. TG-DTG: thermogravimetry and differential thermogravimetry; EVA: ethylene vinyl acetate.
It can also be noticed that EVA1 and EVA6 show higher decomposition rate in the first step but lower decomposition rate in the third step than EVA. The incorporation of LDHs lowers the decomposition rate of the third step but accelerates the loss of carboxylic acid. It is obvious that the –OH groups on the fillers can assist β-hydrogen leaving. That is to say that the loss of carboxylic acid can be catalyzed by LDHs. This has been also reported by Costache and Riva. 41,42 When the temperature goes above 600°C, EVA leaves no residue. However, EVA1 and EVA6 can leave about 30% residue. In addition, EVA6 with both nano-SiO2 and LDHs shows higher thermal stability in the test than the EVA1. This should be the main reason that EVA6 has better flame retardant effects than EVA1. This result confirms the synergistic effects between nano-SiO2 and LDHs, which correlates well with the CCT results.
Conclusion
LDHs were synthesized and characterized by XRD. The XRD result showed that the LDHs were synthesized successfully. It was obvious that nano-SiO2 had a synergistic effect on the flame retardancy with LDHs in EVA composites. When nano-SiO2 was added into the composites, LOI values of the EVA5 containing 2% nano-SiO2 increased significantly. The CCT data indicated that the HRR, THR, SPR, TSR, SF, and mass loss of the EVA6 containing 2.5% nano-SiO2 were almost the lowest. The SEM results showed that a relatively compact and complete char layer was obtained from burning EVA6. The TGA data show that the nano-SiO2 can improve the thermal stability of the flame retardant EVA composites.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: The work was financially supported by the National Natural Science Foundation of China (grant no. 51372129 and 51572138) and the Projects of Science and Technology from Shandong Province (no. 2013GSF11608), China.
