Abstract
In the present study, solution casting method was used for the preparation of nanocomposite films. Primarily, the surface of copper (II) oxide (CuO) nanoparticles was modified with biosafe molecules such as citric acid and ascorbic acid for better dispersion in the polymer matrix. Then novel nanocomposite films were fabricated by loading various percentages of modified CuO nanoparticles in the poly(vinyl chloride) matrix. The ultraviolet (UV)-blocking effects of nanocomposites and their optical properties were studied using UV-visible spectroscopy. Also, other analyses, including Fourier transform infrared spectroscopy, mechanical tensile test, thermogravimetric analysis, and X-microscopy, were performed for investigating thermal, mechanical, and morphological properties of the hybrid materials.
Keywords
Introduction
In nanotechnology, particle is defined as a small object that behaves as a whole unit with respect to its transport and properties. The particles with an average size of 1–100 nm are called nanoparticles (NPs). 1,2
Copper (II) oxide (CuO) as a kind of NP has the monoclinic crystal system that can be shaped in different forms, such as nanowires and nanorods. 3 The main advantage that makes the CuO NPs unique for electronic and photonic applications such as one-dimensional p-type semiconductor and optical equipment polisher is its narrow bandgap (1.2 eV). 4 In addition, it is used as heterogeneous catalysts, gas sensors, optical switch, magnetic storage media, field-emission devices, solar cells, and rectifiers as well as in different acoustic applications. 5 –12 In recent years, different attempts were made to enhance the physical characteristics and photocatalytic activity of nanocomposites (NCs) by loading CuO NPs to various polymeric matrixes. 13 –17
Pure poly(vinyl chloride) (PVC) is a white and brittle solid polymer. The main two advantages of this polymer are its relatively low cost, biological, and chemical resistance. 18 On the other hand, PVC is a thermoplastic polymer and its properties are usually categorized based on the rigidity and flexibility. The rigid form of PVC is used for some applications such as doors, windows, bottles, other nonfood packaging, and cards (such as bank cards). PVC can be softer and more flexible by adding the plasticizers. 19 The flexible PVC can be used as an alternative for rubbers, electrical cable insulations, artificial leathers, and inflatable products. 20,21
Ascorbic acid (AA) is a white solid powder with the γ-lactone structure. The
Citric acid (CA) with the C6H8O7 formula is a weak organic acid, which is produced naturally in citrus fruits as a preservative. 24 It is an inexpensive and nontoxic acid that can be used as a flavoring agent, a chelating agent, an emulsifier, a pH adjusting agent, and in pharmaceuticals. As a special application, CA can enhance the performance of starch, polyvinyl alcohol/starch, cellulose, and thermoplastic starch/polylactic acid blend by acting as a cross-linking agent. 25,26
In the present study, CuO NPs were modified with biosafe molecules including CA and AA, and their effects on NCs films were studied. Polymer-based NCs and modified CuO were fabricated under ultrasonic irradiation. The properties of the prepared samples were characterized by different techniques such as Fourier transform infrared (FTIR) spectroscopy, ultraviolet–visible (UV-Vis) spectroscopy, X-ray diffraction (XRD), thermogravimetric analysis (TGA), and mechanical tensile test. Dispersion of NPs in PVC matrix was studied using field-emission scanning electron microscopy (FESEM) and transmission electron microscopy (TEM).
Experimental
Materials
CuO nanopowder was obtained from Neutrino Co. (Tehran, Iran), with average particle size <50 nm and specific surface area >80 m2 g−1. CA and AA were supplied from Merck Chemical Co. and used as received. PVC was a commercial polymer produced by LG Chem (Korea Origin), with weight-average molecular weight = 78,000 g mol−1. Tetrahydrofuran (THF) was purchased from JEONG Wang, Siheung, South Korea.
Characterization
FTIR spectra on a Jasco-680 (Tokyo, Japan) spectrophotometer in the range of 400–4000 cm−1 were characterized to identify the chemical bonds of the prepared samples in potassium bromide pellets for powder materials, and spectrograms of NC films were acquired directly. Preparation of NCs was accomplished using MISONIX (Raleigh, North Carolina, USA) ultrasonic liquid processors, XL-2000 SERIES (USA), with frequency wave 2.25 × 104 Hz and power of 100 W. TGA data were taken on STA503 instrument (TA Instruments, New Castle, Delaware, USA) at a heating rate of 20°C min−1 from 25°C to 800°C under argon atmosphere. XRD spectra were collected on a Philips X’pert MPD diffractometer (Netherlands) with a copper (Cu)
Surface modification of CuO NPs with CA and AA
For the surface treatment of CuO NPs, 0.01 g of CA was dissolved in 7 mL of deionized (DI) water and sonicated for 30 min (solution 1). Also 0.01 g of AA were dissolved in 7 mL of DI water and sonicated for 30 min (solution 2). Then, 0.10 g of CuO NPs were dispersed in 7 mL of DI water and the obtained mixture was ultrasonicated for 30 min (solution 3). In the next step, solutions 1 and 2 were mixed and irradiated under sonication for 30 min. At last, solution 3 was added to the previous mixture and irradiated under ultrasonic for 30 min. Ultimately, this solution was dried at room temperature for 24 h to give the CuO-CA-AA (modified CuO) NPs.
Preparation of the NC films
In this procedure, 0.10 g of PVC was dissolved in 5 mL of THF. Then various amounts of the modified CuO NPs (4, 8, and 12 wt%) were added to PVC solutions. The PVC/modified CuO suspensions were ultrasonicated for 1 h and then stirred for 14 h at room temperature. Once again the suspensions were sonicated for 1 h, and the obtained suspensions were casted onto uncovered glass petri dishes for 1 day in order to evaporate the THF solvent. After removal of THF, films were fabricated and could be easily separated from the glass petri dishes.
Results and discussion
Preparation of the NC film
Recently, NPs have received a great attention due to the enhancement of NC properties. 27 –29 Good dispersion of NPs in the polymer matrix plays an important role in the development of NC properties. On the other hand, surface modification of NPs can improve the dispersion process. According to the recent articles, molecules such as dicarboxylic acid, chiral diacid, and CA have been used in order to modify the surfaces of NPs. 30 –32 In this study, biosafe molecules including CA and AA were used for double-layer surface modification. Coating of the modification layers (CA and AA) on the CuO NPs substrate is due to the possible interactions between the functional groups presented on the NPs surface, especially hydroxyl groups and the modifiers. The effect of various amounts of prepared NPs on PVC in THF was investigated. The schematic of the process is shown in Figures 1 and 2.
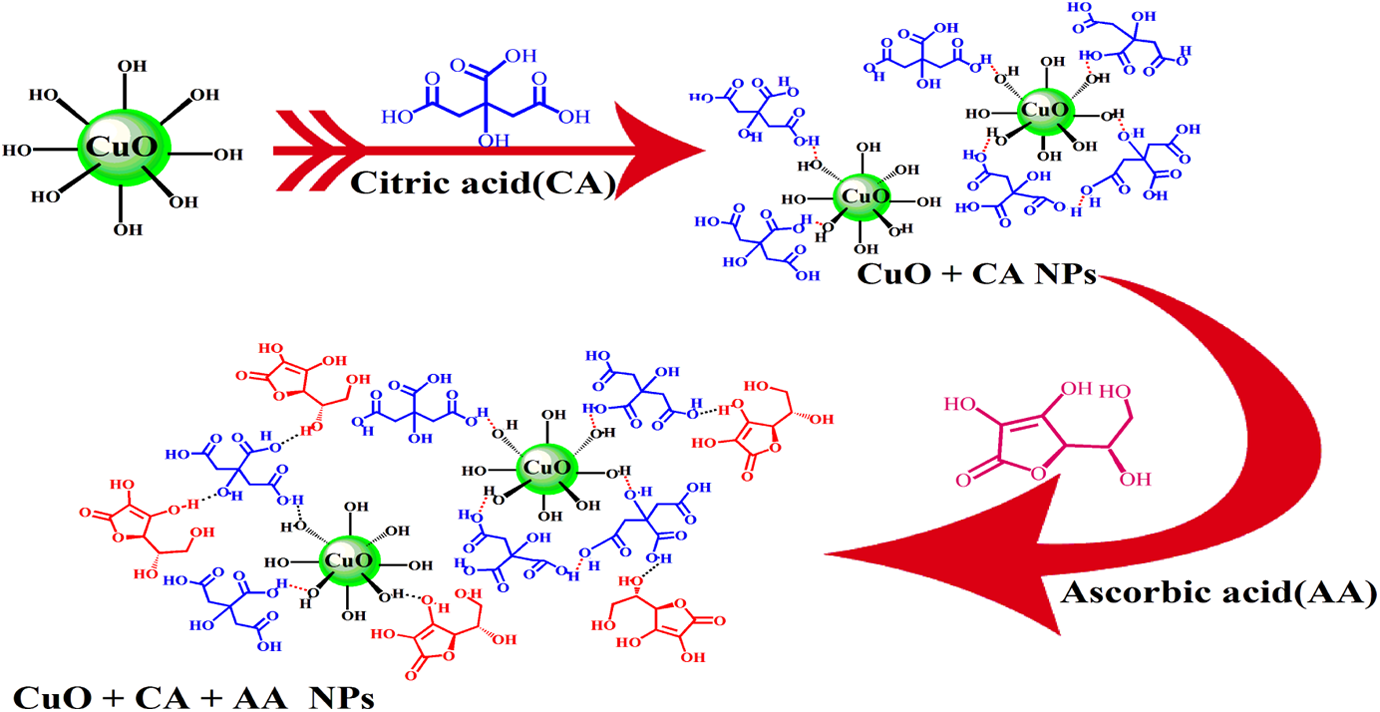
Surface modification of CuO NPs with CA and AA under ultrasonic irradiation. CuO: copper (II) oxide; NPs: nanoparticles; CA: citric acid; AA: ascorbic acid.
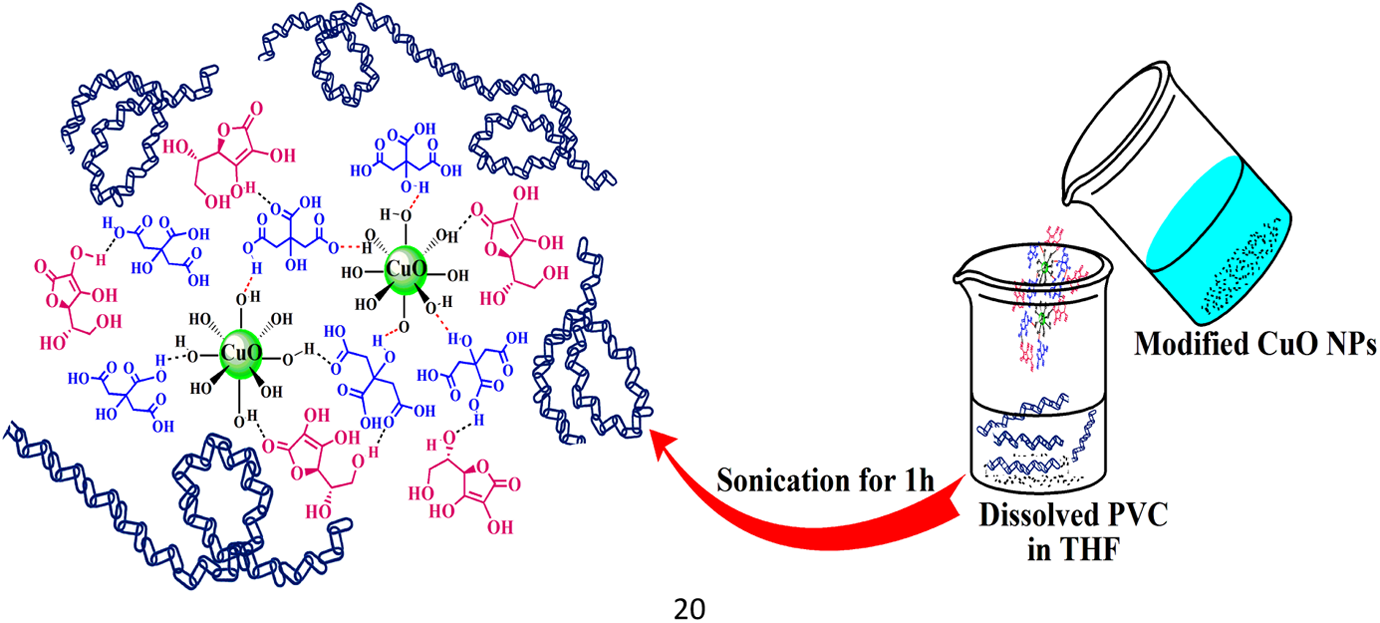
The schematic image of the formation of PVC/modified CuO NC. PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite.
FTIR spectroscopy
The FTIR spectrum of pure CuO is shown in Figure 3(a). The characteristic peak around about 475 cm−1 refers to Cu–O bond stretching. The appearance of a broad absorption band at 3298 cm−1 confirms the presence of stretching mode of water molecules and/or surface hydroxyls. 33 Also, the absorption bands at 1600–1700 cm−1 can be related to the bending vibrations of O–H. Pure CA has strong intermolecular and intramolecular H bonding that create expansive peaks in the region of 2500–3500 cm−1 (Figure 3(b)). In addition, the peaks around 1700 and 1755 cm−1 corresponds with CA carbonyl vibration peaks. 34 For pure AA (Figure 3(c)), the peak at 1672 cm−1 attributes to the vibration of C=O and also the broad band peak in 2500–3000 cm−1 ascribes the hydroxyl groups of pure AA. 35 Figure 3(d) represents the spectrum of modified CuO with CA and AA. The peak at 1621 cm−1 is owing to the carbonyl groups of modifiers. Besides, broad band peak in area of 2800–4000 cm−1 is observed which confirms different hydroxyl groups of CA and AA. Figure 4 illustrates the FTIR spectra of pure PVC and its NCs. For pure PVC, peaks under 3000 cm−1 characterize the C–H stretching mode. In addition, peaks at 1430, 1252, 616, and 692 cm−1 are related to CH2, CH2–Cl, C–Cl, respectively. 36,37 The presence of modified CuO in the polymer matrix creates changes in FTIR spectrum of pure PVC which is observable in the region of 430–540 cm−1. Also the peak around 1560 cm−1 shows a rise in intensity with increasing the percentage of modified filler.
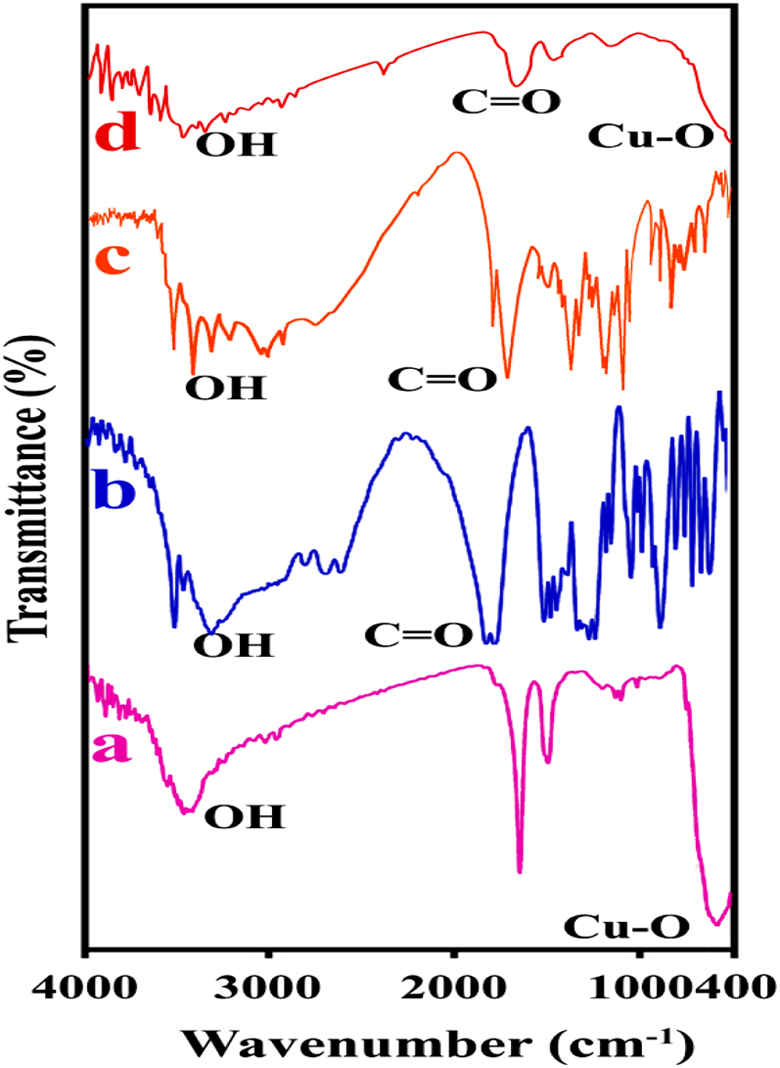
FTIR spectra of (a) pure CuO NPs, (b) CA, (c) AA, and (d) modified CuO NPs. FTIR: Fourier transform infrared; CuO: copper (II) oxide; NPs: nanoparticles; CA: citric acid; AA: ascorbic acid.
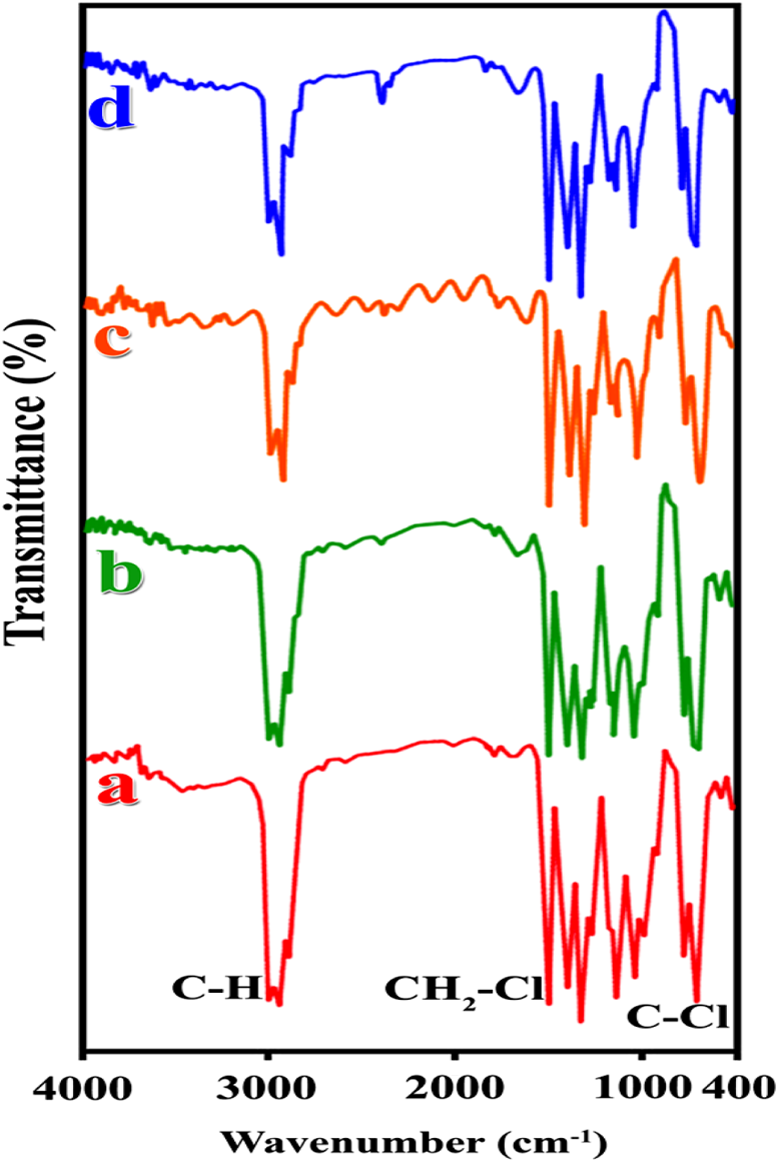
FTIR patterns of (a) pure PVC, (b) PVC/modified CuO NC 4 wt%, (c) PVC/modified CuO NC 8 wt%, and (d) PVC/modified CuO NC 12 wt%. FTIR: Fourier transform infrared; PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite.
X-Ray diffraction
The XRD patterns of CuO NPs, CA, AA, and modified CuO NPs are shown in Figure 5. The diffraction peaks in Figure 5(a) can be corresponded to the monoclinic structure of single-phase CuO. 38 Due to the usage of slight amount of CA and AA, diffraction peaks did not appear in the curve of modified CuO NPs. The neat and modified CuO NPs almost show similar XRD patterns, indicating that crystalline structure of CuO NPs has no changes after surface modification with CA and AA.
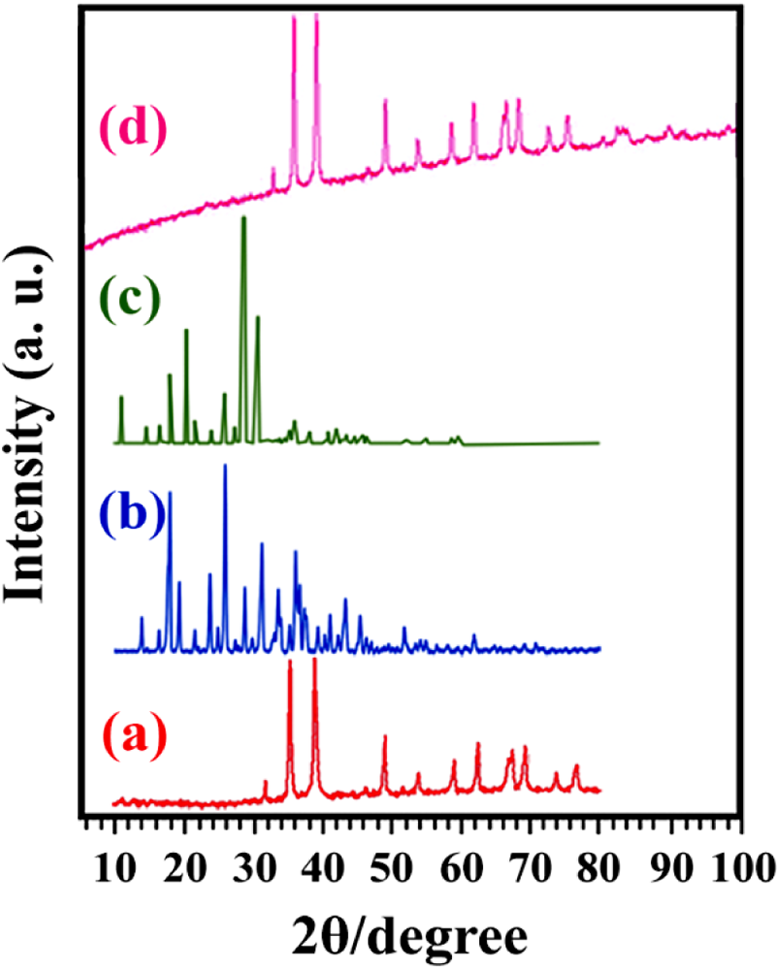
XRD spectra of (a) pure CuO NPs, (b) CA, (c) AA, and (d) modified CuO NPs. XRD: X-ray diffraction; CuO: copper (II) oxide; NPs: nanoparticles; CA: citric acid; AA: ascorbic acid.
The XRD results of the PVC, PVC/modified CuO NC 4, 8, and 12 wt% are demonstrated in Figure 6. The diffraction pattern of PVC illustrates that this polymer is amorphous. According to the XRD patterns of the NCs, intensity of peaks rises with increasing the amount of CuO NPs in PVC matrix.
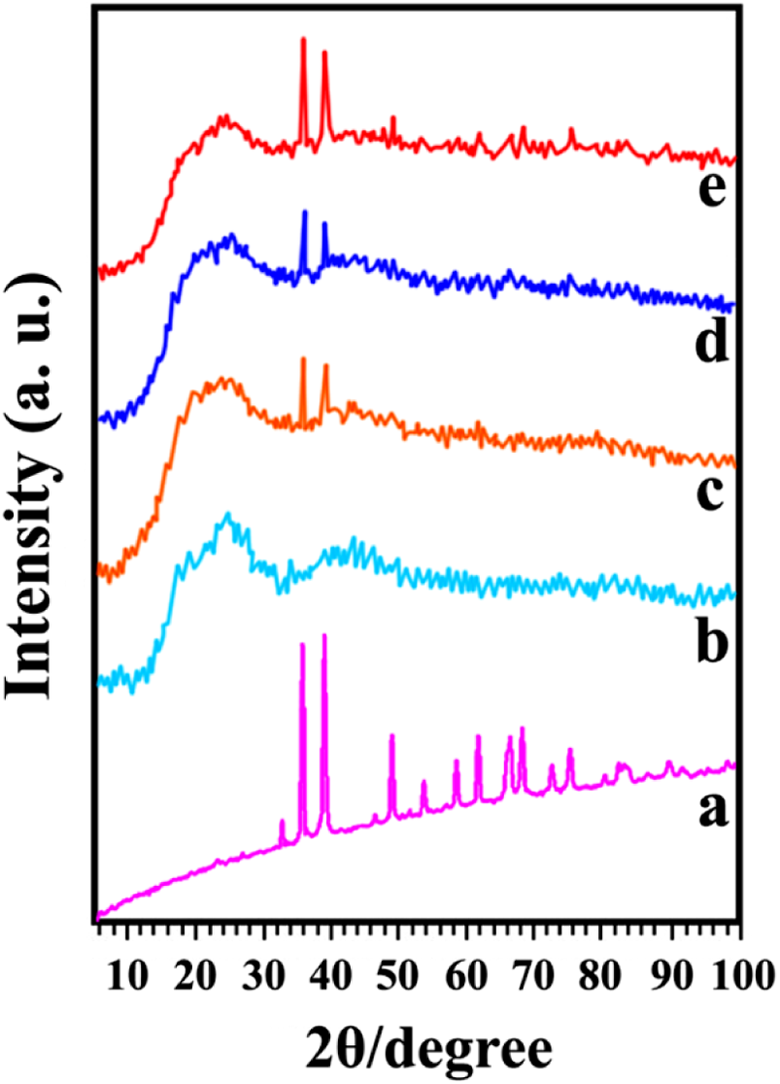
XRD curves of (a) modified CuO NPs, (b) pure PVC, (c) PVC/modified CuO NC 4 wt%, (d) PVC/modified CuO NC 8 wt%, and (e) PVC/modified CuO NC 12 wt%. XRD: X-ray diffraction; CuO: copper (II) oxide; NPs: nanoparticles; PVC: poly(vinyl chloride); NC: nanocomposite.
Microscopy characterization
FESEM and TEM techniques were used to investigate the morphology and distribution of modified CuO NPs in PVC matrix. Figure 7 displays the FESEM photographs of modified CuO and NCs with different magnification. These images indicate that modified CuO NPs are well dispersed in the polymer matrix, and the aggregation is reduced. FESEM image of NC 12 wt% shows little agglomeration. The homogeneous dispersion of CuO NPs might be due to the presence of capping agents on their surfaces, and ultrasonic irradiation technique leads to a decrease in surface energy of NPs.

FESEM micrographs of (a) modified CuO NPs, (b) pure PVC, (c) PVC/modified CuO NC 4 wt%, (d) PVC/modified CuO NC 8 wt%, and (e) PVC/modified CuO NC 12 wt%. FESEM: field-emission scanning electron microscopic; CuO: copper (II) oxide; NPs: nanoparticles; PVC: poly(vinyl chloride); NC: nanocomposite.
TEM micrographs of pure CuO NPs, modified CuO, and PVC/modified CuO NCs 8 wt% are demonstrated in Figure 8. The spherical shape and nanoscale-modified CuO are seen in these pictures, and the average particle size is 10.80 ± 2.91 nm. It was expected that the particles to be quantum dots, but after performing the fluorescence test, a sufficient result did not achieve. TEM observations confirm the modification surface of CuO NPs, since the average particle size is reduced from 30 nm (for pure CuO) to around 10 nm (for modified CuO). The size distribution histograms for pure CuO NPs, modified CuO, and PVC/modified CuO NCs 8 wt% showed mean particle sizes of 29.12, 10.80, and 5.87 nm, respectively (Figure 9).

TEM micrographs of pure (a) CuO NPs, (b, c) modified CuO NPs, and (d, e) PVC/modified CuO NC 8 wt% at different magnifications and their histograms. TEM: transmission electron microscopic; CuO: copper (II) oxide; NPs: nanoparticles; PVC: poly(vinyl chloride); NC: nanocomposite.
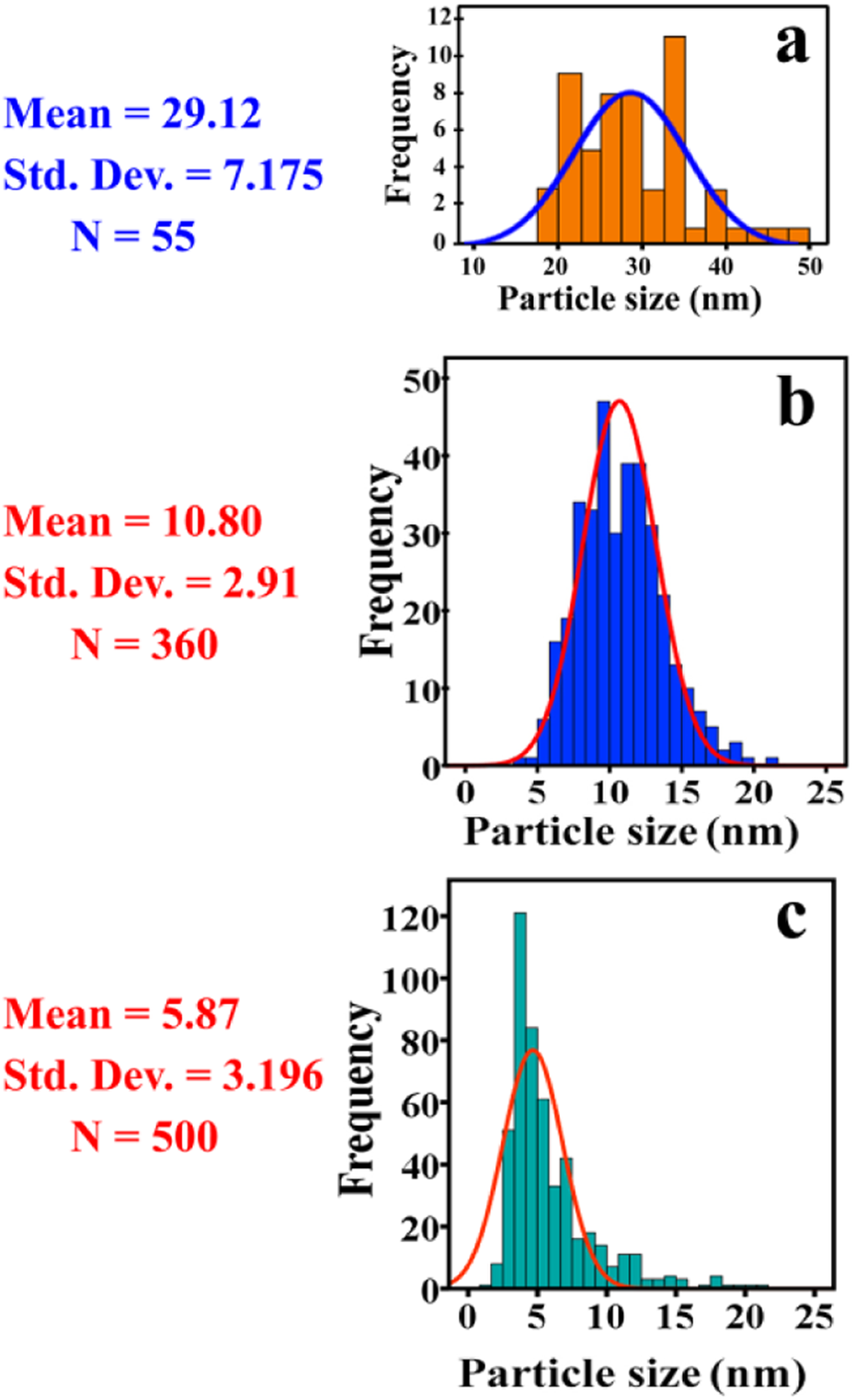
The size distribution histograms for (a) pure CuO NPs, (b) modified CuO NPs, and (c) PVC/modified CuO NC 8 wt%. CuO: copper (II) oxide; NPs: nanoparticles; PVC: poly(vinyl chloride); NC: nanocomposite.
Thermal properties
Thermal properties of modified CuO, pure PVC, and the prepared films were measured by TGA under argon atmosphere at a heating rate of 20°C min−1. Before TGA, the NC films were dried at 70°C to remove the solvent. The thermograms of neat and modified CuO are exhibited in Figure 10. In decomposition processes, the physisorbed water molecules eliminate around 120°C. According to the TGA curve of modified CuO, degradation process around 200°C can be corresponded to the decomposition of both modifiers, because the thermal degradation of AA and CA are in the temperature range of 190°C and 197°C, respectively. 39 From this graph, it is found that about 12% of CA and AA are grafted, which confirm the presence of capping agents on the surface of CuO NPs.
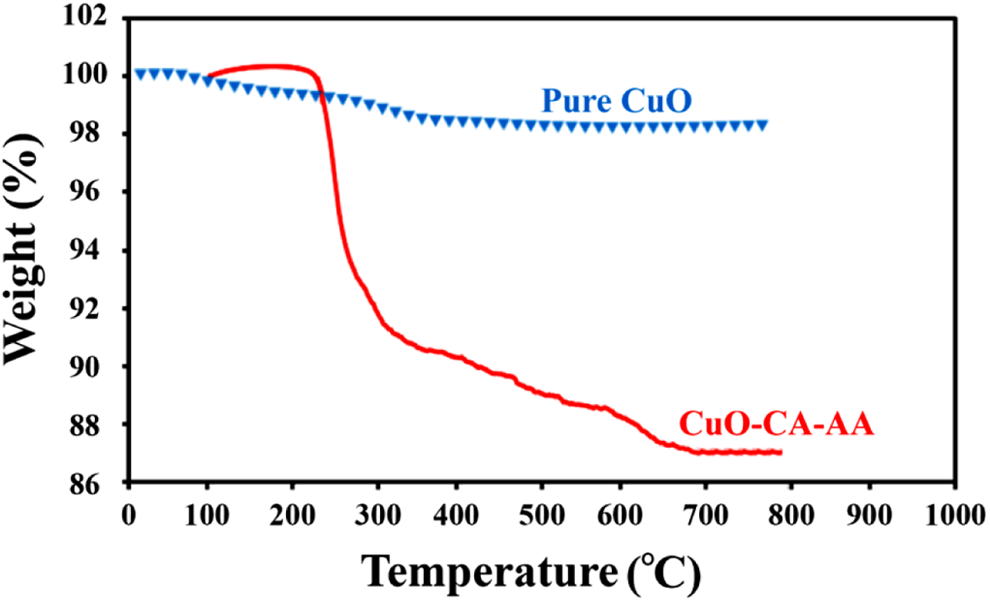
TGA thermograms of pure CuO and modified CuO NPs. TGA: thermogravimetric analysis; CuO: copper (II) oxide; NPs: nanoparticles.
The weight loss of 5 wt% (
Thermal properties of the PVC and PVC/modified CuO NCs.
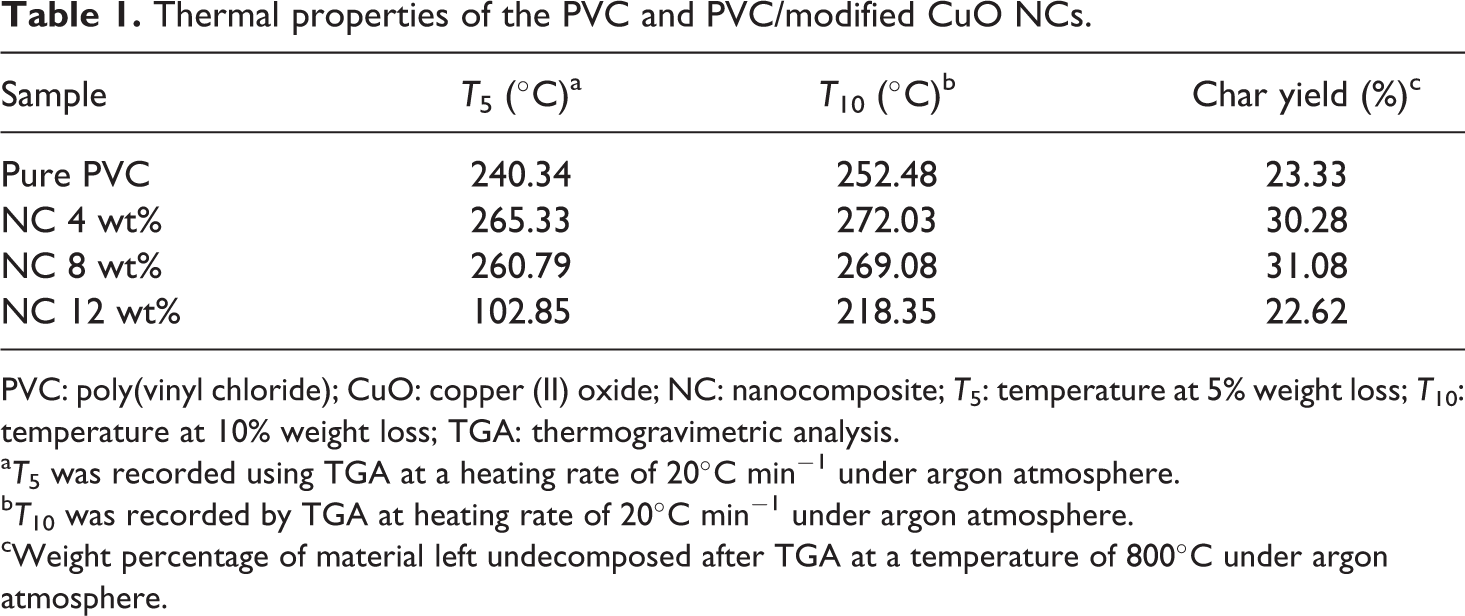
PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite;
a
b
cWeight percentage of material left undecomposed after TGA at a temperature of 800°C under argon atmosphere.
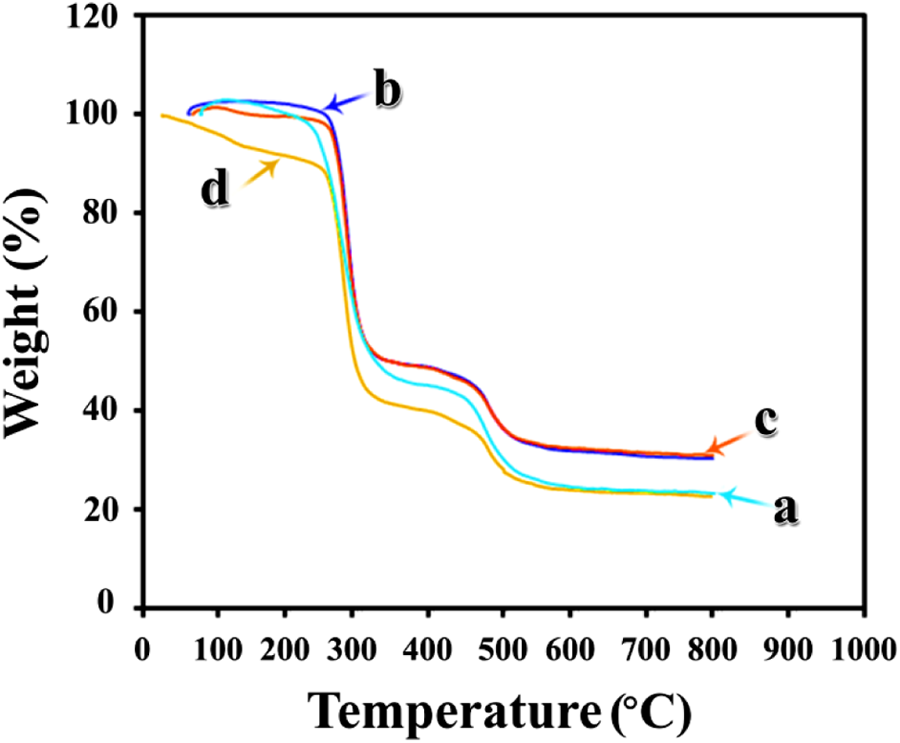
TGA thermograms of (a) neat PVC, (b) PVC/modified CuO NC 4 wt%, (c) PVC/modified CuO NC 8 wt%, and (d) PVC/modified CuO NC 12 wt%. TGA: thermogravimetric analysis; PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite.
The char yield of pure PVC is 23.33%, and the residual weight of the NCs for different weight fractions of modified CuO NPs: 4, 8, and 12 wt% are 30.28%, 31.08%, and 22.62%, respectively.
Optical properties of NCs
UV-Vis spectroscopy was applied to investigate the optical properties of NC films. The UV-Vis absorption spectra of PVC and PVC/modified CuO NCs (4, 8, and 12 wt%) are illustrated in Figure 12. The intensity of peaks increases by adding rising percentages of modified CuO in NCs. In figure 12, d–d transitions are observed due to Cu2+ has d orbital with 9 valance electrons. The d–d transition wavelengths are often placed below 400 nm, but because the particles are in nanosize, these transitions shift to lower wavelengths.
40
The peak in region near 277 nm may be related to
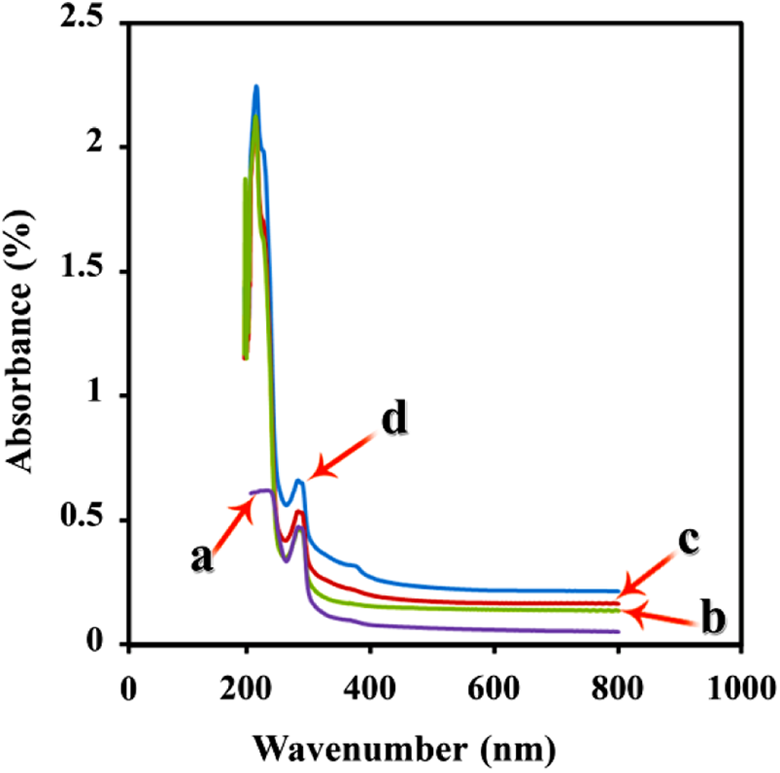
UV-Vis spectra of (a) neat PVC, (b) PVC/modified CuO NC 4 wt%, (c) PVC/modified CuO NC 8 wt%, and (d) PVC/modified CuO NC 12 wt%. UV-Vis: ultraviolet–visible; PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite.

Photographs of (a) pure PVC, (b) PVC/modified CuO NC 4 wt%, (c) PVC/modified CuO NC 8 wt%, and (d) PVC/modified CuO NC 12 wt%. PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite.
Mechanical properties
To investigate the tensile properties of pure PVC and NC films, mechanical tests were operated. Stress versus strain curves of hybrid specimens are depicted in Figure 14. According to Figure 14 and Table 2, some mechanical behavior of NC films such as elongation and strain are enhanced in comparison with neat polymer, but stress is decreased with increasing the content of modified CuO NPs. The most important reason can be the reduction of polymer matrix uniformity by loading filler through it. These data show that flexibility of the NCs is improved in comparison with the pure PVC. 43
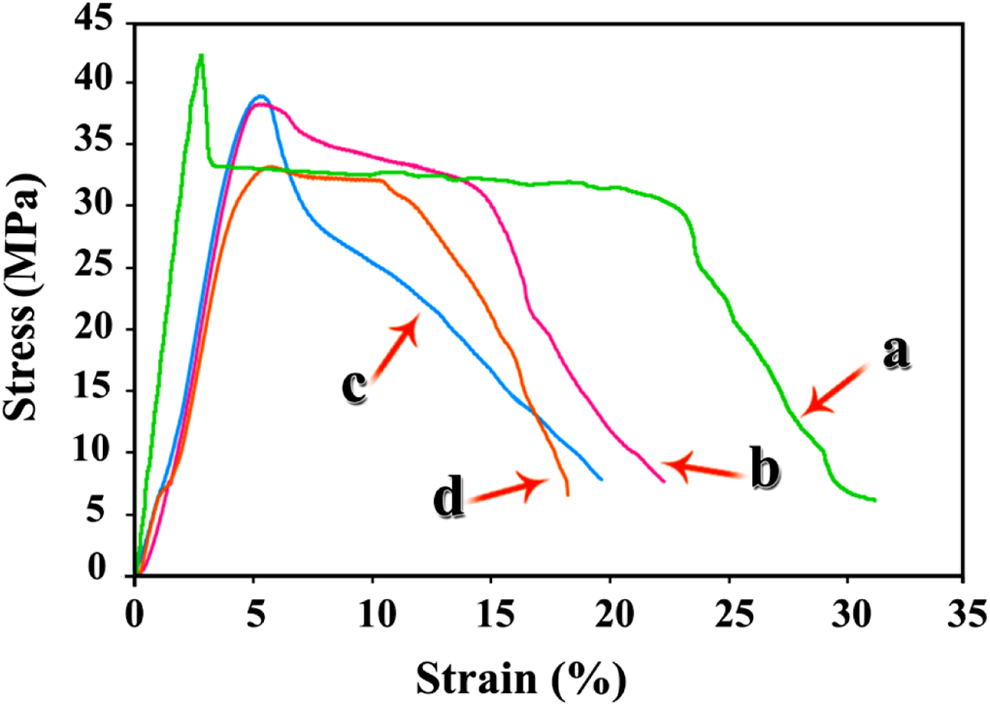
Stress–strain curves of (a) pure PVC, (b) PVC/modified CuO NC 4 wt%, (c) PVC/modified CuO NC 8 wt%, and (d) PVC/modified CuO NC 12 wt%. PVC: poly(vinyl chloride); CuO: copper (II) oxide; NC: nanocomposite.
Summary of mechanical data for the PVC and its composites.
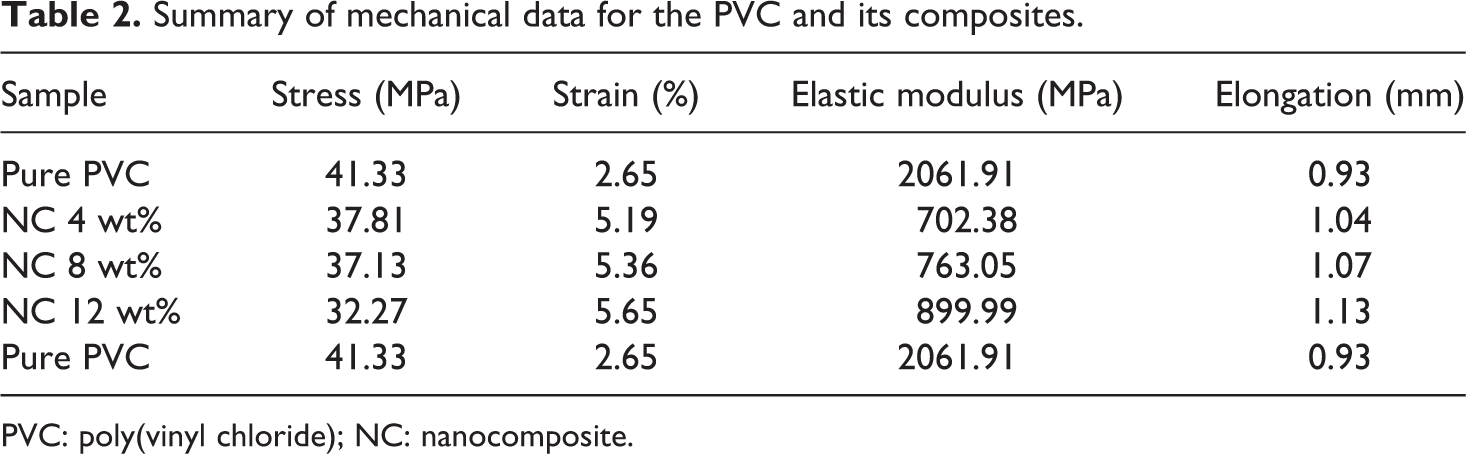
PVC: poly(vinyl chloride); NC: nanocomposite.
Conclusions
PVC is one of the most extensively used forms of plastics. So it is necessary to improve its properties. In this work, before the preparation of PVC NCs, the surface of CuO NPs was grafted with CA and AA as capping agents. Then, various amounts of modified CuO NPs loaded to PVC matrix and the PVC/modified CuO NCs were successfully fabricated under ultrasonic irradiation as a green and an easy method. The properties of these polymeric NCs were investigated by different techniques. The presence of PVC and CuO NPs were confirmed using characterized absorption bands in the NCs FTIR spectrograms. Based on FESEM and TEM results, the average particles size reduced after surface modification, and CuO NPs were uniformly dispersed into PVC backbone. TGA exhibited that thermal resistance of NCs decreased by loading greater than 8 wt%. The optical properties of NC films represented an absorption in the region of UV–C. Also, according to the results of the mechanical tests, the flexibility of the NCs improved with the incorporation of modified CuO NPs in the PVC matrix.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors deliver their gratitude to the Research Affairs Division Isfahan University of Technology (IUT), Isfahan and the National Elite Foundation (NEF) for financial support. Further financial support from Iran Nanotechnology Initiative Council (INIC) and Center of Excellency in Sensors and Green Chemistry Research (IUT) is gratefully acknowledged.
