Abstract
In this study, a feasibility for developing window films for energy efficiency in building based on poly(vinyl chloride) (PVC) composite was explored, using two different types of thermochromic pigments, such as vanadium dioxide (VO2) and thermoreversal-40. Using VO2, tensile properties of the PVC film improved, accompanied with an increase of percentage gelation of PVC. On the other hand, thermochromic behaviors, in the visible light region of the PVC/thermoreversal-40 film, were more obvious than those of the PVC/VO2 analog. The percentage of transmittance of the near infrared wave through the PVC/VO2 film was much lower than that transmitted through the PVC/thermoreversal-40 film. The above improvement was achieved at the expense of the thermal stability of the PVC film.
Introduction
Energy efficiency in a building is considered to be one of the most important issues in the design and development of modern buildings for a smart living society. The energy consumption in buildings contributes significantly to the total energy consumption. 1 For example, in the United States in 2016, the energy consumption in both residential and commercial buildings accounted for about 40% of total energy consumption. 2 Likewise, in Thailand, the majority of the electricity was consumed by the industrial sector, which accounted for 41.8% of total consumption in 2013; whereas the commercial and public sector represented 35.2% of consumption. Residential electricity consumption accounted for the remaining 23.0%. In this regard, for the sake of energy efficiency for building and human thermal comfort, there has been a considerable interest in the development of materials with excellent optical and heat insulation properties. From the material science and technology viewpoints, doped tin oxides such as indium tin oxide (ITO) and antimony-doped tin oxide (ATO) are known to be good candidates for this purpose, taking into account their high reflectivity in the near infrared (NIR) wave. 3 In general, the ATO can be prepared using several techniques including a sol–gel process, 4,5 chemical precipitation, 6,7 and chemical vapor deposition (CVD). 8 ATO has also been used commercially for coating onto glass or windows in existing buildings using a spraying technique. 9
Apart from tin oxide-based materials, other interesting and promising materials capable of exhibiting heat insulation properties have been discovered and have yet to be further explored and developed. These include the vanadium dioxide (VO2) in the form of a monoclinic phase, which is known to be a kind of thermochromic material. The synthesis of VO2 for smart glasses and energy efficient windows has been reported. The VO2 films were synthesized by either gas phase processes (such as sputtering and CVD) 10,11 or solution-based processes (such as sol–gel 12 and polymer-assisted deposition). 13 –15 The VO2 films, with and without doping, obtained from these techniques, are well characterized and their thermochromic and NIR reflectance properties have been demonstrated. However, scaling up the production process of VO2 films based on these techniques still has some limitations in terms of both the production cost and the uniformity of the film. Alternatively, VO2 may be synthesized into a powder form, using techniques such as hydrothermal 16,17 and reduction-hydrolysis processes. 18 To further apply the synthesized VO2 powder for use as a heat reflective coating on an industrial scale, our research group came up with the idea of compounding the VO2 particles with polymer resins to produce polymer composite films. In fact, this idea has been demonstrated in the case of ethylene vinyl acetate copolymer (EVA)/VO2 composite films for laminated glasses. 19 In that case, the thermochromic properties of the EVA composite films exist, whereas the transparency of the films can be compromised and optimized using a suitable concentration of the VO2 particles. However, since the EVA film used as a binder for laminated glass has to be cured in an assembly line in the factory prior to transportation and installation, this means that the EVA/VO2 film cannot be used or applied to the glass or windows which have already been installed in an existing building. In this regard, other types of polymer-based window films have to be considered. In this study, plasticized poly(vinyl chloride) (PVC) film was selected for the study and development of this application, owing to the good rubbery, transparency, and process ability of the PVC. The development of PVC composite films mixed with ITO and/or ATO particles has been reported by Qi et al. 20 It was found that PVC film containing 2 phr of ITO was superior to the PVC/ATO analog, in terms of NIR reflectance. Nevertheless, both types of the PVC/tin oxide composite films were capable of reducing the temperature by about 3–5°C inside a model house.
In relation to this study, the spectral response to solar radiation and the optimum concentration of VO2 in the PVC composite films is still unknown and has yet to be clarified. For comparison purposes, a commercial thermochromic pigment, thermoreversal-40, was used for compounding with PVC. The aim of this work is to investigate the effects of various types and concentrations of the pigments on the mechanical, thermal, optical, chromic, and insulation properties of PVC film. Thermal stability of the PVC composite films, assessed by a thermal aging test, is also of interest.
Experimental
Materials
PVC powder (K-66) was supplied by the Thai Plastic and Chemicals Co. Ltd, Thailand. The barium–cadmium–zinc-based thermal stabilizer (CS-1368 N) and dioctyl phthalate (DOP) were supplied by The Cosmos Chemicals Co. Ltd, Thailand. The VO2 powder was synthesized in our lab, via hydrothermal and calcination processes. Vanadium pentoxide was used as a raw chemical, and hydrazine was used as a reducing agent. From X-ray photoelectron spectroscopy and differential scanning calorimetry (DSC) analysis techniques, the monoclinic phase with the average crystal size of 26.9 nm and a transition temperature of 65°C of the synthesized VO2 was confirmed. According to the results from a particle size analysis, the size of the VO2 is in the range of 2.8–30 µm. More details concerning the synthesis procedure, conditions, and characterization results can be seen from our earlier report. 19 A thermochromic pigment (thermoreveral-40, particle size in the range of 2.5–52.5 µm) was purchased from the Tanaka Chemical Co. Ltd, Thailand.
Compounding of PVC
PVC compounds were prepared using an internal mixer (MX7 S from the Charoen Tut Co. Ltd, Thailand). Basically, the compounding formula contains 100 phr of the PVC powder, 50 phr of the DOP plasticizer, 3.5 phr of thermal stabilizer, and a given type and amount of thermochromic pigments. In this study, the pigment concentration was varied, ranging from 1 phr to 4 phr. The internal mixing was carried out at 140°C for 3 min, using a fill factor of 70%.
Fabrication of PVC films
The PVC compound obtained from the internal mixer was then fabricated into a 0.2-mm-thick film, using a hydraulic compression mold (Lab Tech Engineering Co. Ltd, Thailand). The mold was preheated at 160°C for 3 min. After that, the PVC compound was transferred into the mold cavity, and the compression molding process was carried out at 160°C for 3 min, under a pressure of 2000 psi. Finally, the sample was solidified by cooling it in the mold for 2 min.
Mechanical properties test
The mechanical properties of the PVC composite films were determined from tensile testing, using the LLOYD-LR50 K instrument equipped with a 500-N load cell. The test was carried out in accordance with the ASTM D638 standard method, at a crosshead speed of 15 mm min−1. At least three specimens were tested for each sample, and the average values of tensile strength, percentage elongation, and the modulus at 100% strain were then calculated and reported.
Morphological analysis
The morphology of the neat pigments and the various PVC composite films were examined by a scanning electron microscopy (SEM) technique, using the Hitachi, S-3400 N microscope, equipped with a secondary electron detector, a backscattering electron detector, and an energy dispersive X-ray analyzer (EDX). Practically, the prepared specimens (pigment powder and/or a square piece; 5 × 5 mm2) of the various PVC composite films were put onto the SEM stub prior to coating with gold, using a sputtering unit. This was to eliminate the charging effect. The SEM images and EDX patterns of the various specimens were then examined and recorded.
Optical properties tests
The optical properties of the PVC composite films were determined using the ultraviolet (UV)–visible (Vis)–NIR spectrometer (UV-3100; Shimadzu, Japan). The spectra were recorded over wavelengths ranging between 400 nm and 1200 nm. Light transmittance was determined in accordance with the ISO 9050 standard method. Specifically, the transmission of light through the polymer film was integrated over a wavelength range of 400–2500 nm. Spectral reflectance in the NIR solar region (ranging from 780 nm to 2500 nm) was also measured at an incident angle of 15°. The spectral data were integrated against an air mass 1.5 global spectrum (ASTM E891) to yield weighted ordinates over the total spectral bandwidth. Five measurements were made of each sample and the weight-averaged values were reported.
Color index test
Changes in the color of PVC composite films were quantitatively analyzed by a color index test. The color, described in terms of CIE lab scale (
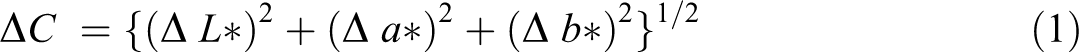
Thermal analysis
The thermal behavior of the PVC composite films was investigated using a DSC (DCS 204; Netzsch, Germany) over temperatures ranging between 45°C and 200°C, under a nitrogen gas atmosphere, and at a heating rate of 10°C min−1. In this study, apart from the glass transition, the gelation of the PVC compound was also determined using the following equation (2). 21 More details concerning the theoretical aspect and typical DSC curves used for a calculation of the gelation can be studied in the literature 22
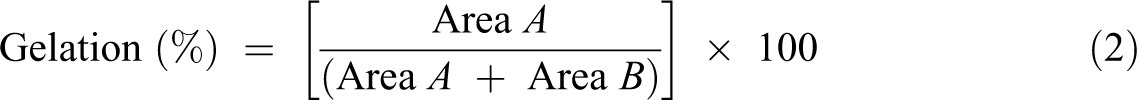
where Area
Insulation property test
In order to evaluate the capabilities of various PVC composite films in blocking heat waves from penetrating a window, a demo system containing PVC and PVC thermochromic films coated on a window of a model house was developed. In this system, an infrared lamp (IR 250 RH IR2 250 V 250 W) was used as a heat source to activate phase changes of the thermochromic pigments. The actual temperature in front of the window, measure by a thermocouple, was 100 ± 1°C. This is well above the transition temperature of both the thermoreversal and the monoclinic VO2. In this study, changes in temperature behind the window film on both sides of the model house were recorded as a function of time for every 30 s, until an equilibrium was reached. This took about 12 min in total.
Aging test
The long-term stability of the PVC composite films was evaluated using an accelerated weathering test in accordance with an ASTM G154-06. A QUV tester, equipped with a ultraviolet lamp (emitted UV light at 313 nm) was used. The exposure cycle was established at 8 h of UV radiation, at 70°C, followed by 4 h of dark condensation at 50°C. The specimens for each composite sample, prepared in accordance with ASTM4412-06a, were collected every 50, 100, 200, and 400 h. After that, changes in the tensile properties of the aged specimens, compared with those of the original samples, were measured and reported.
Results and discussion
Mechanical properties
Changes in the tensile properties of the various PVC composite films with types and concentration of the pigments are presented in Figures 1 to 3. Overall, it can be seen that the types of pigment significantly affected the tensile properties of the composite films. Specifically, when either VO2 or the mixed pigments were added to the compounds, the tensile strength and percentage elongation values of the PVC films rapidly increased and tended to reach a plateau. The same effects did not occur when the thermoreversal-40 pigment was used. In the latter case, both tensile strength and percentage elongation values of the PVC film were inferior to those of the neat PVC film (without any pigment), regardless of the pigment concentration. This implies that the efficacy of the thermoreversal-40 in acting as a reinforcing agent for PVC is unsatisfactory. In terms of the tensile modulus of the composite films, the values changed with the pigment concentration in a nonlinear fashion. A kind of bell-shaped curve was observed (Figure 3). The modulus values initially increased to the maximum with the concentration of the VO2-based pigment. The optimum concentration of VO2 and the mixed pigments were about 3 phr, beyond which the values decreased again, with a further increase of the pigments content. This can be ascribed to some agglomeration of the pigment particles at a high percentage of pigment loading. The tensile modulus values of both the PVC/VO2 and the PVC/mixed pigment films are greater than that of the PVC/thermoreversal-40 system, regardless of the pigment concentration. It is also noteworthy that the tensile strength and modulus of the PVC composite films (both PVC/VO2 and PVC/mixed pigments) were increased without sacrificing their percentage elongation values (see Figure 2). This could be related to the presence of hydroxy (OH) groups on the surface of the synthesized VO2 particles. 23 In this regard, a polar interaction between OH groups of VO2 and chlorine (Cl) atoms of the PVC molecules might contribute to a better compatibility and greater tensile properties of the composites.
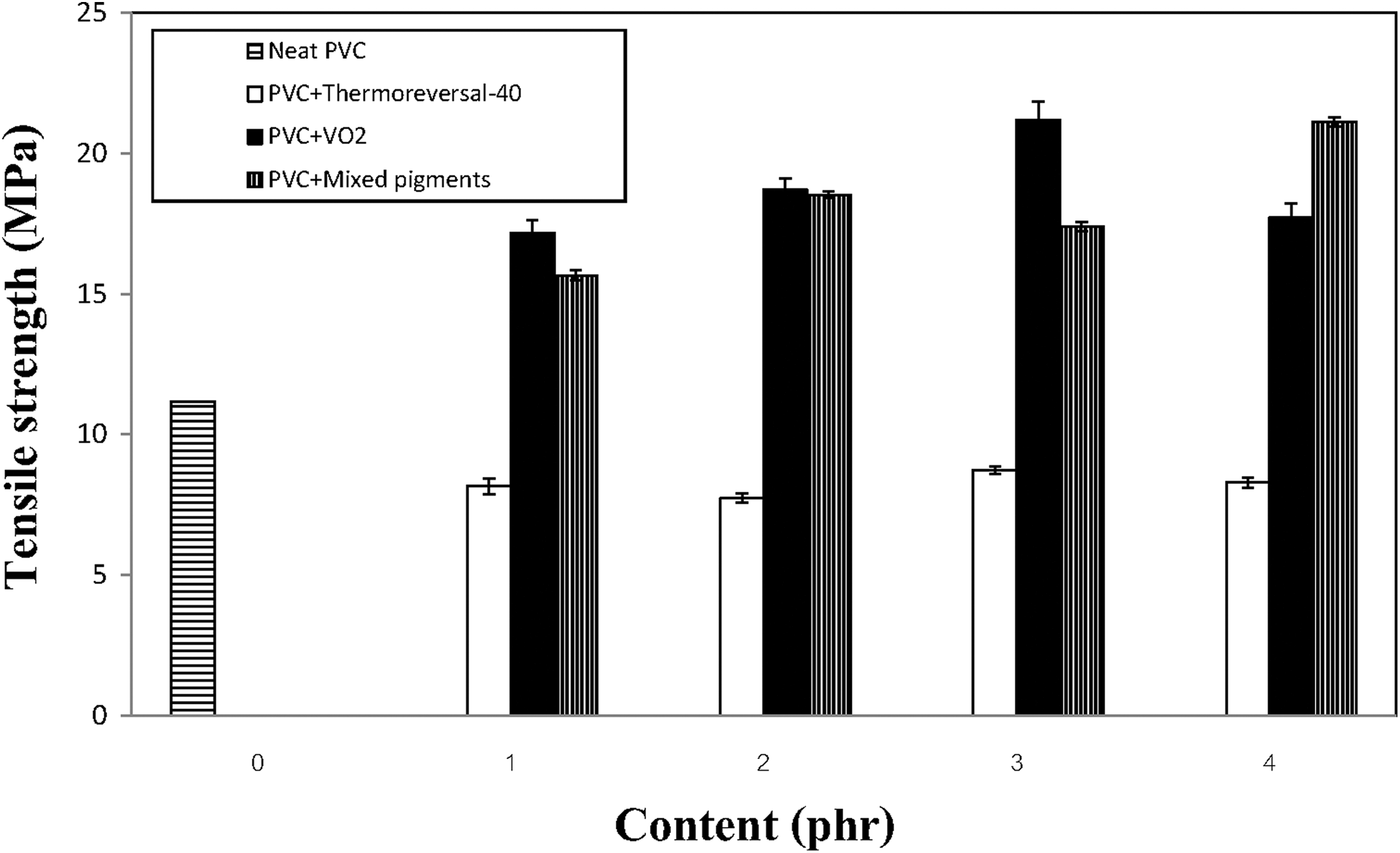
Changes in tensile strength of the various PVC composite films containing different types and concentration of the pigments.
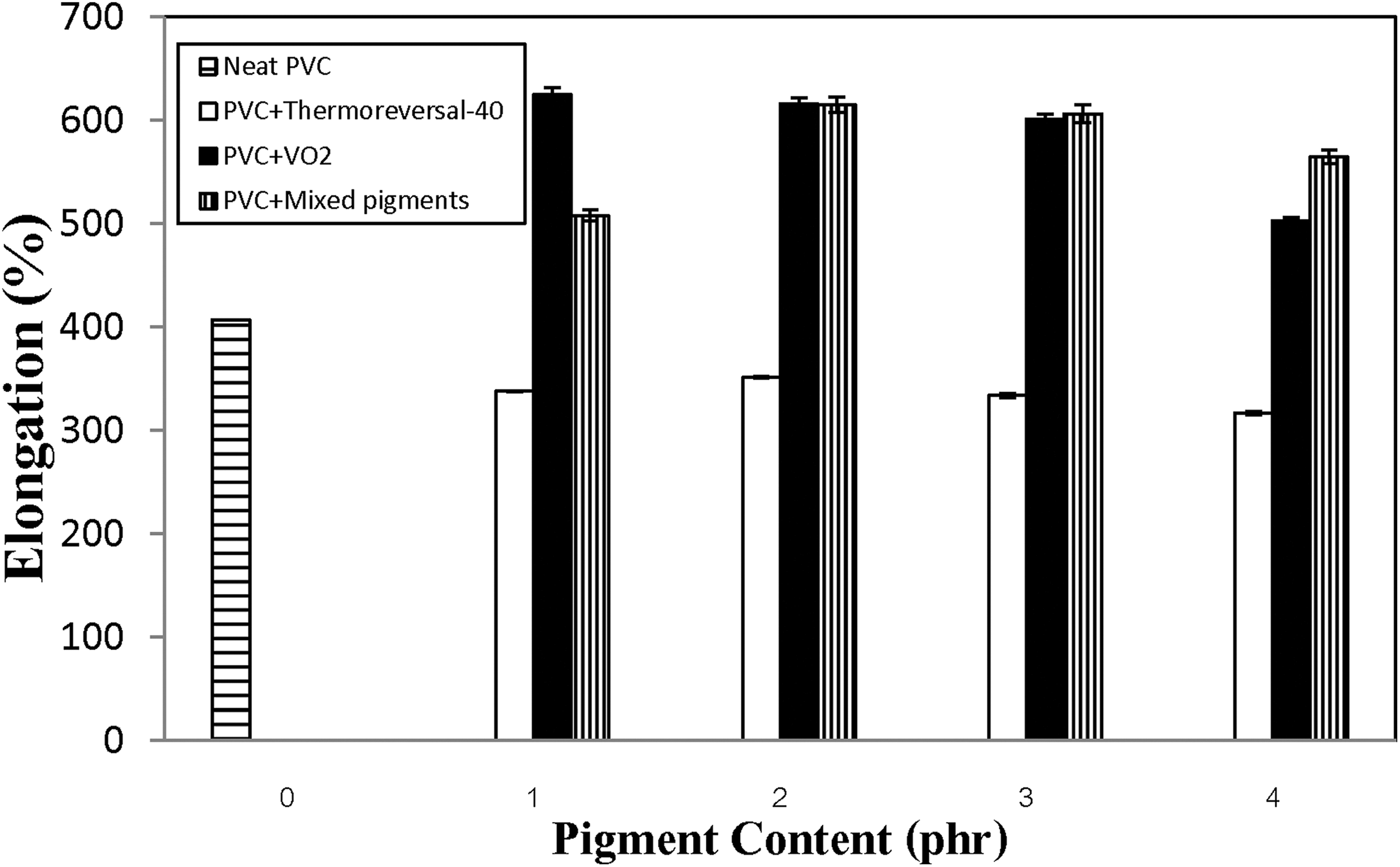
Changes in percentage elongation at break of the various PVC composite films containing different types and concentration of the pigments.
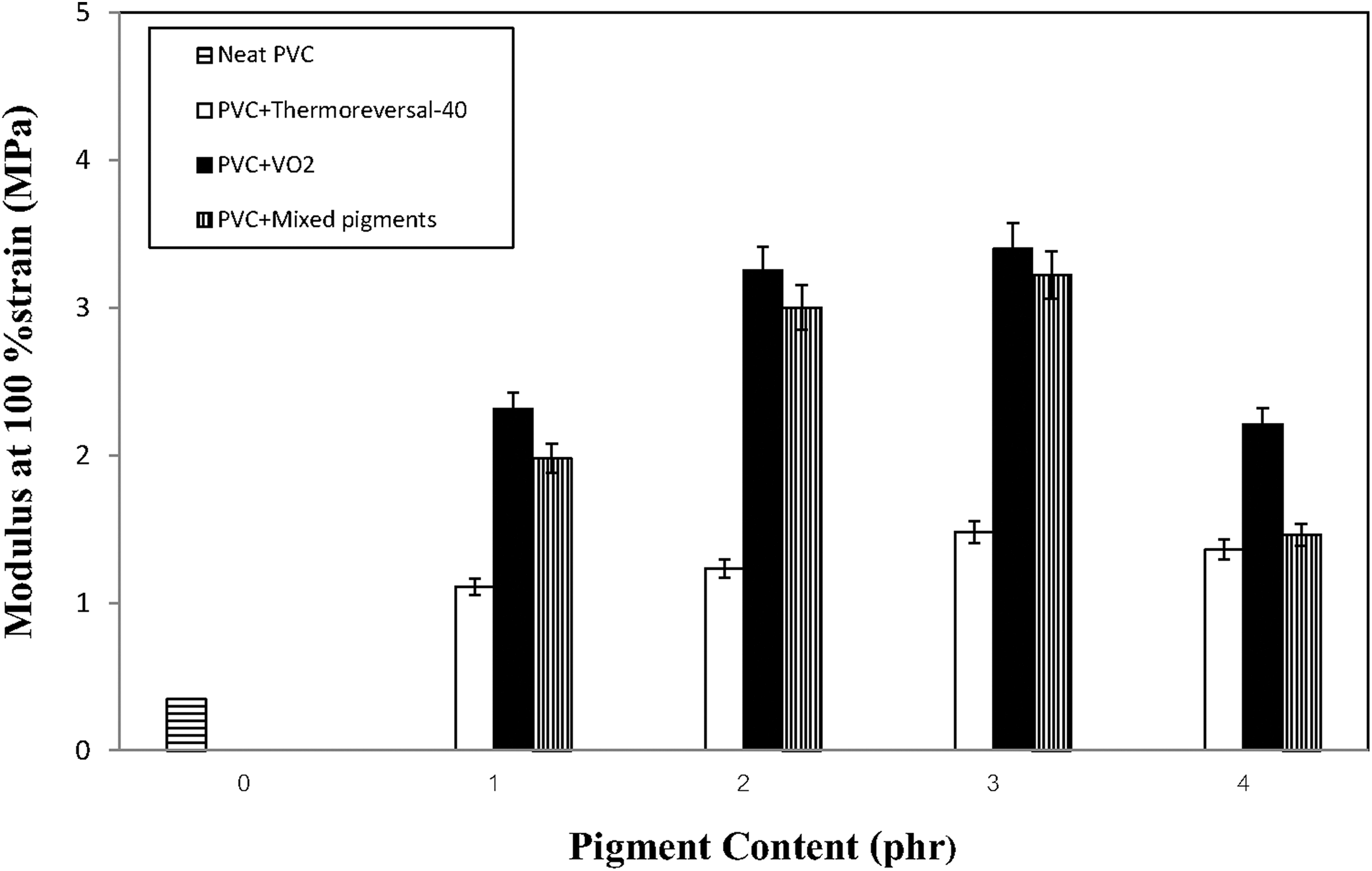
Changes in tensile modulus of the various PVC composite films containing different types and concentration of ther pigments.
Morphology
Figure 4(a) shows a backscattering SEM image of PVC/VO2 film. The bright domain phase represents the VO2 particles as were verified by EDX dot maps (Figure 4(b) and (c)). Some agglomeration of the VO2 particles can also be observed and confirms the above discussion concerning the decrease of the tensile modulus of the composite films at a high VO2 loading. From the SEM backscattering electron image of the PVC/thermoreversal film (Figure 5), it is noteworthy that the pigment appeared dark. This implies that the pigment lacks heteroatoms or heavy elements. In other words, the thermoreversal pigment could be a kind of hydrocarbon material. The result from an Fourier-transform infrared analysis of the neat thermoreversal-40 pigment shows the absorption peaks at 3302, 1449–1551, and 1060 cm−1, representing the vibrational frequencies of O–H, C=C, and C–O bonds, respectively.
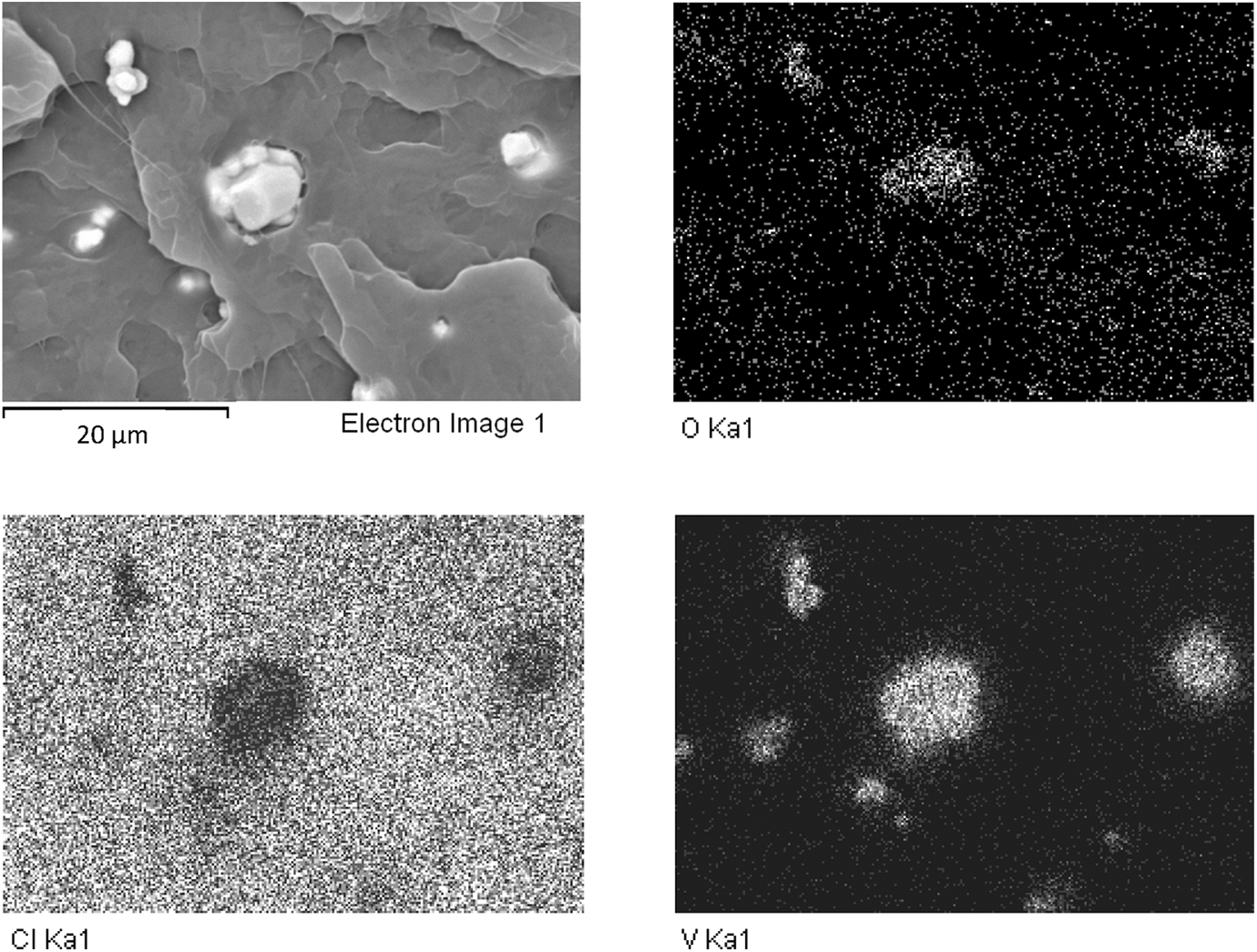
SEM micrograph (backscattering electron image; a) and EDX dot maps (b to d) from the tensile fracture surface of PVC composite film containing 4 phr of the VO2 pigments (magnification = 2500 times).

SEM (BE) images of the cryogeinc fractured surfaces of PVC composite films containing different types of the pigments; thermoreversal-40 (a), VO2 (b), and the mixed pigments (c).
From Figure 6, some agglomeration of the VO2 particles can obviously be seen. The particle size of the VO2 is also relatively smaller than that of the thermoreversal. It is noteworthy that some voids at the interface between the PVC matrix and the pigment particles can be observed from the SEM images of the tensile fracture surfaces of the PVC composite films, regardless of the pigment types. These voids could be induced from the deformation of the plasticized PVC phase, during tensile testing. This caused a de-bonding of pigments from the polymer matrix. The above results suggest that an interfacial adhesion between the pigments and the PVC polymer matrix has yet to be further improved, if the greater mechanical properties of the composite films are expected. In our opinion, one possible strategy includes the use of VO2 core–shell particles, instead of using normal VO2 powder. In fact, the development of VO2 over-coated with titanium dioxide (TiO2) in a form of core–shell nanorods has been reported by Li et al. 24 The presence of TiO2 shell could enhance both the chemical stability against oxidation and luminous transmittance of VO2 based on an antireflection effect of the VO2. It seems that a kind of synergistic effect was achieved. It was also claimed that both thermochromic and photocatalytic properties exist in this system and can offer significant potential for creating a multifunctional smart coating. In relation to our work, however, the use of VO2 core–shell particles for compounding with PVC window film has yet to be developed and explored.
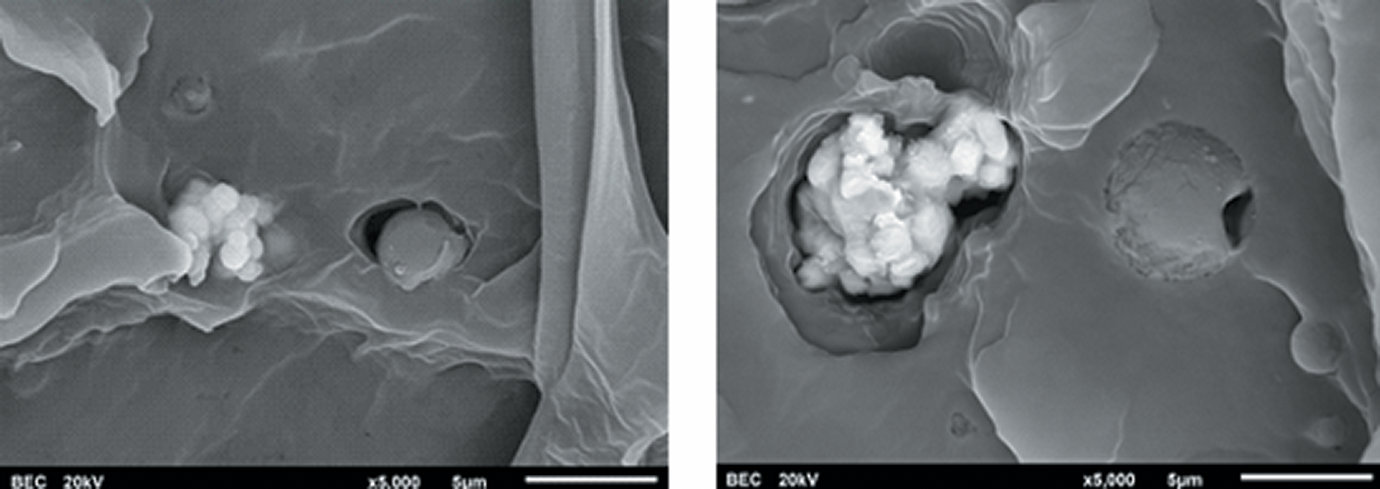
SEM micrographs (secondary electron images) from the tensile fracture surfaces of PVC composite films containing 1 phr (left) and 4 phr (right) of the mixed pigments.
Thermal behaviors
Examination of the DSC thermograms of the PVC and PVC composite films (Figure 7) reveals that the exothermic peaks of PVC increased with the presence of either VO2 or the mixed pigments. This peak could be attributed to gelation of the plasticized PVC. 25,26 The percentage of gelation of the various PVC composite films was calculated and is summarized in Table 1. It seems that the effects of the types and concentration of pigments on the gelation values of the PVC composite films resemble the mechanical properties of the system. For example, percentage of gelation of the PVC/thermoreversal-40 film hardly increased, compared to that of the neat PVC film (gelation = 50%). On the other hand, the maximum percentage of gelation was obtained when 3 phr of the VO2 was applied. The gelation value dropped again, however, when a higher content of the pigments was used. Upon an induction and growth of the gelation process, the greater mechanical properties of the fabricated PVC products or specimens can be expected. 27 In relation to this study, it is possible that the enhanced percentage of gelation of the PVC composite films containing VO2-based pigments contributed to the greater tensile properties of the system. The above results also imply that VO2 is more compatible with PVC than thermoreveral-40. Again, this could be attributed to the polar interaction between OH groups of VO2 and Cl atoms of the PVC molecules as was discussed above.
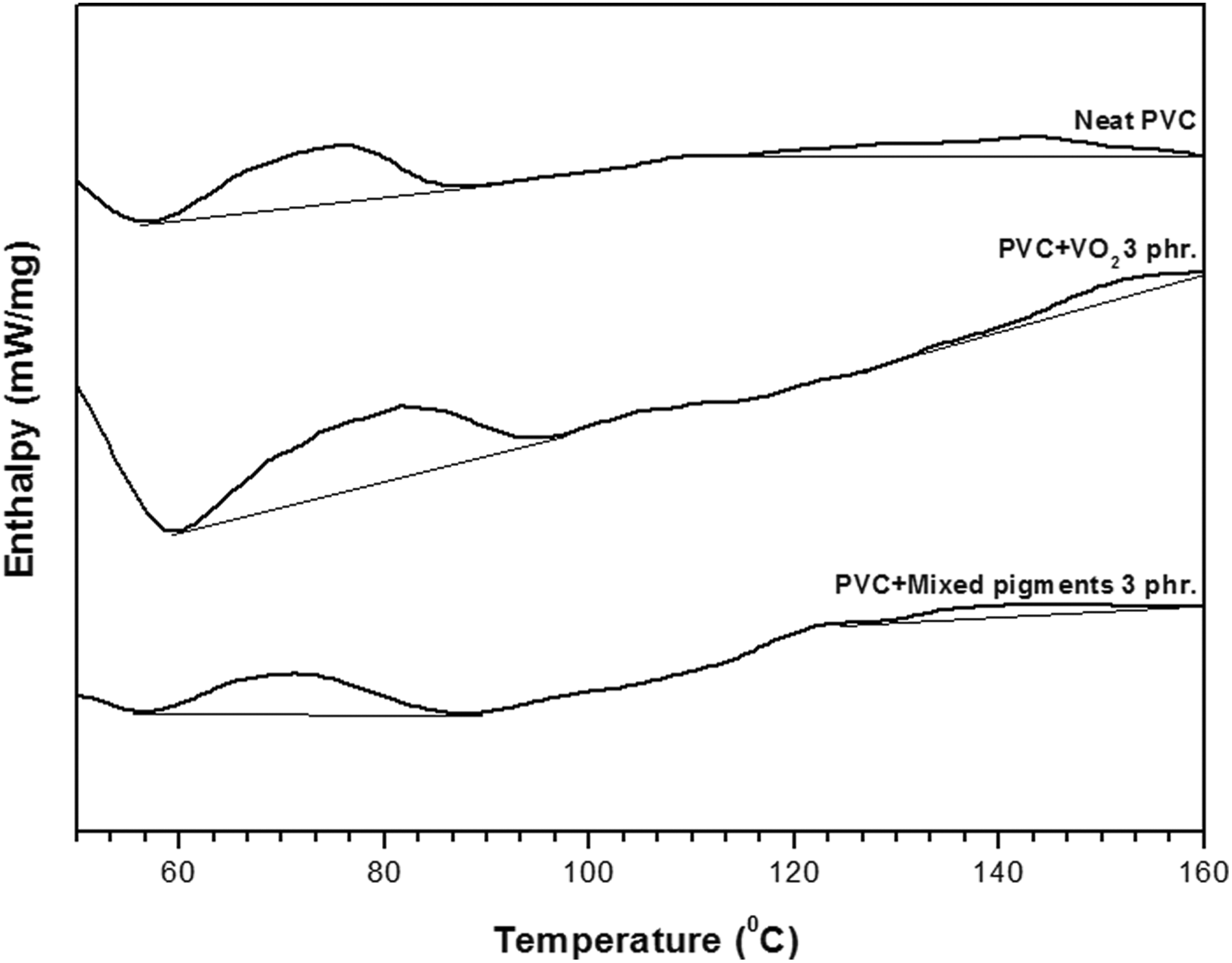
DSC thermograms of the neat PVC film, the PVC/VO2 (3 phr) film, and the PVC/mixed pigments (3 phr) film.
Percentage gelation of various PVC composite films containing different types and concentration of thermochromic pigments.
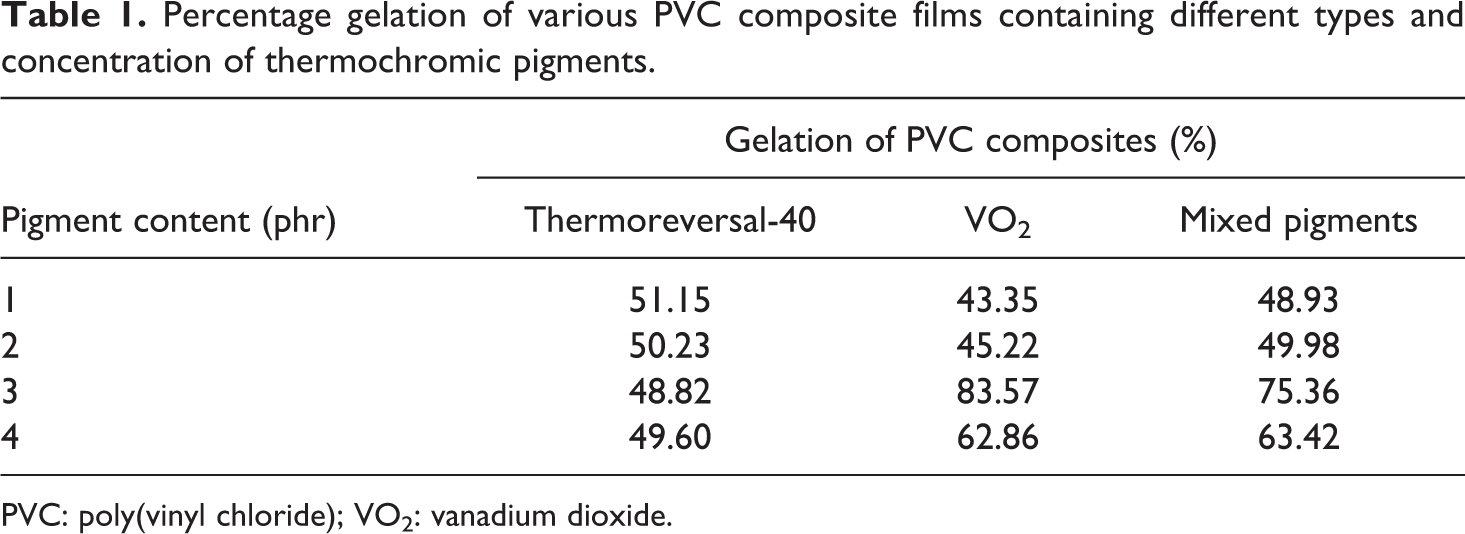
PVC: poly(vinyl chloride); VO2: vanadium dioxide.
Chromic properties
In terms of chromic properties, it was found that the color of the PVC/thermoreveal-40 film changed obviously from darkness to semitransparent, when the specimen was heated to above its transition temperature (40°C; see Figure 8). After cooling, the process was rapidly and completely reversible. This implies that the composite materials might be used as an effective thermal sensor and indicator. The above observations were further confirmed and quantified by the results from a color index test. The results from Table 2 show that, at room temperature, the parameter

Photographs of the PVC/composite films containing 1 phr (top) and 4 phr (bottom) of thermoreversal-40 pigment both before (left) and after heating (right) to 75°C.
Changes in color index (
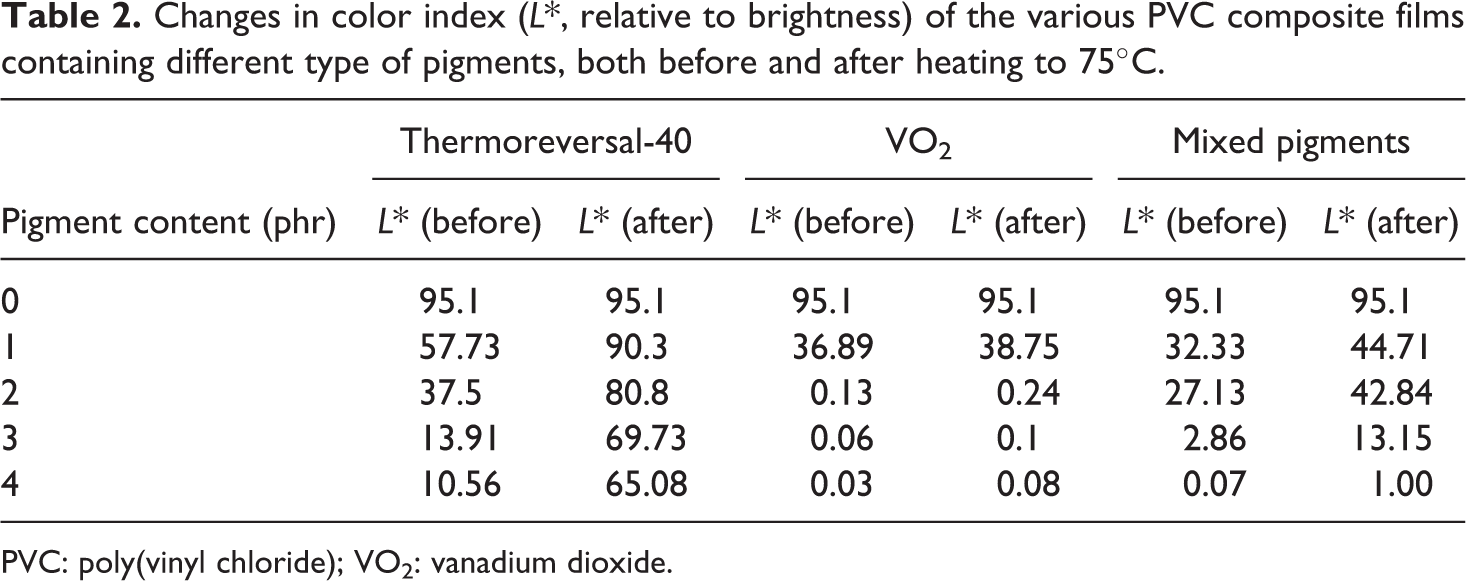
PVC: poly(vinyl chloride); VO2: vanadium dioxide.
Color differences (Δ
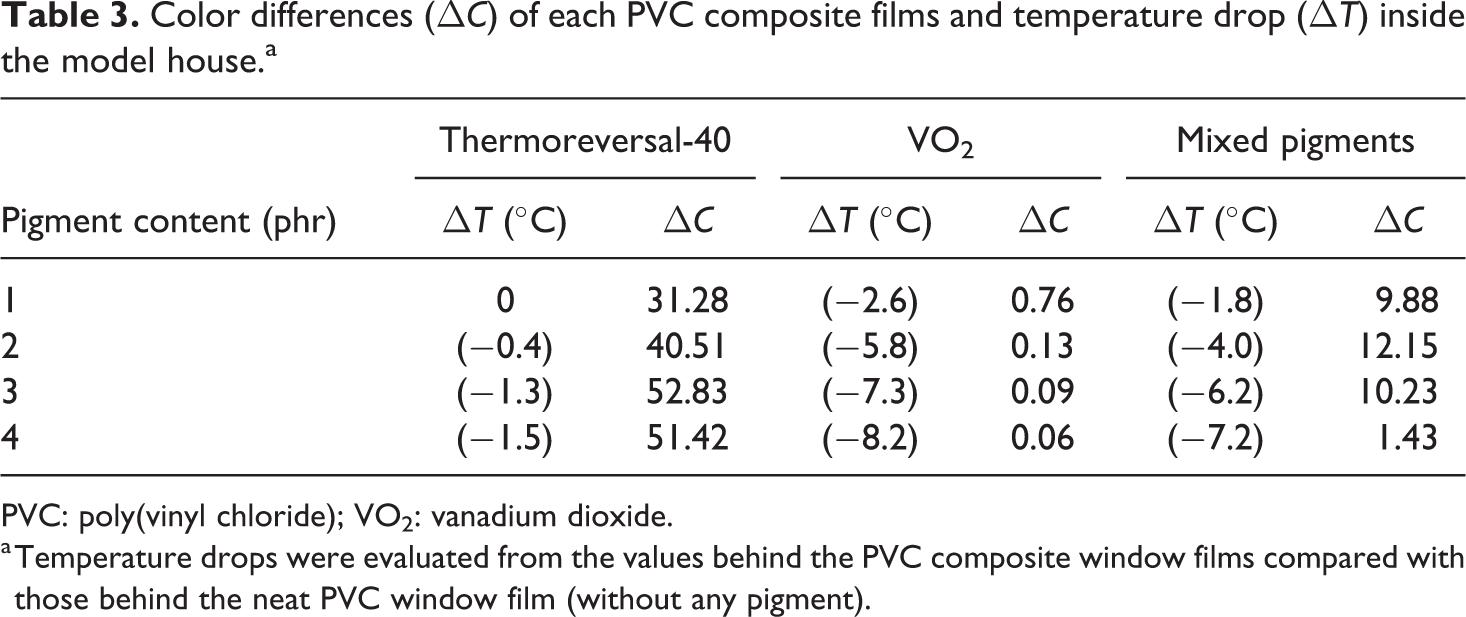
PVC: poly(vinyl chloride); VO2: vanadium dioxide.
a Temperature drops were evaluated from the values behind the PVC composite window films compared with those behind the neat PVC window film (without any pigment).
Optical properties
A spectral response to the solar radiation of the various thermochromic PVC composite films deserves consideration, taking into account the fact that the PVC composite films were designed and developed for use with windows or glasses in existing buildings. In this regard, three main parameters, such as transmittance, absorbance, and reflectance, shall be considered. Figure 9 shows a typical UV/Vis spectra of various PVC films. Herein, changes in light transmittance, both in the visible range and in the NIR range, can be noted. The extent of spectral changes, described in terms of peak intensity (or percentage transmittance), varies depending on the type and concentration of the pigments. Figure 10 shows that the transparency of the PVC film dropped rapidly with the presence of the pigments. Specifically, the light transmittance of the film containing VO2 dropped faster compared to the PVC/mixed pigments and the PVC/thermoreversal-40 films. However, beyond 2 phr of the pigment loading, the discrepancy is marginal. In relation to the use of PVC composite films as a window film for chromic glass, it is recommended that both the concentration and the state of dispersion of the pigments in PVC matrix should be improved and optimized.
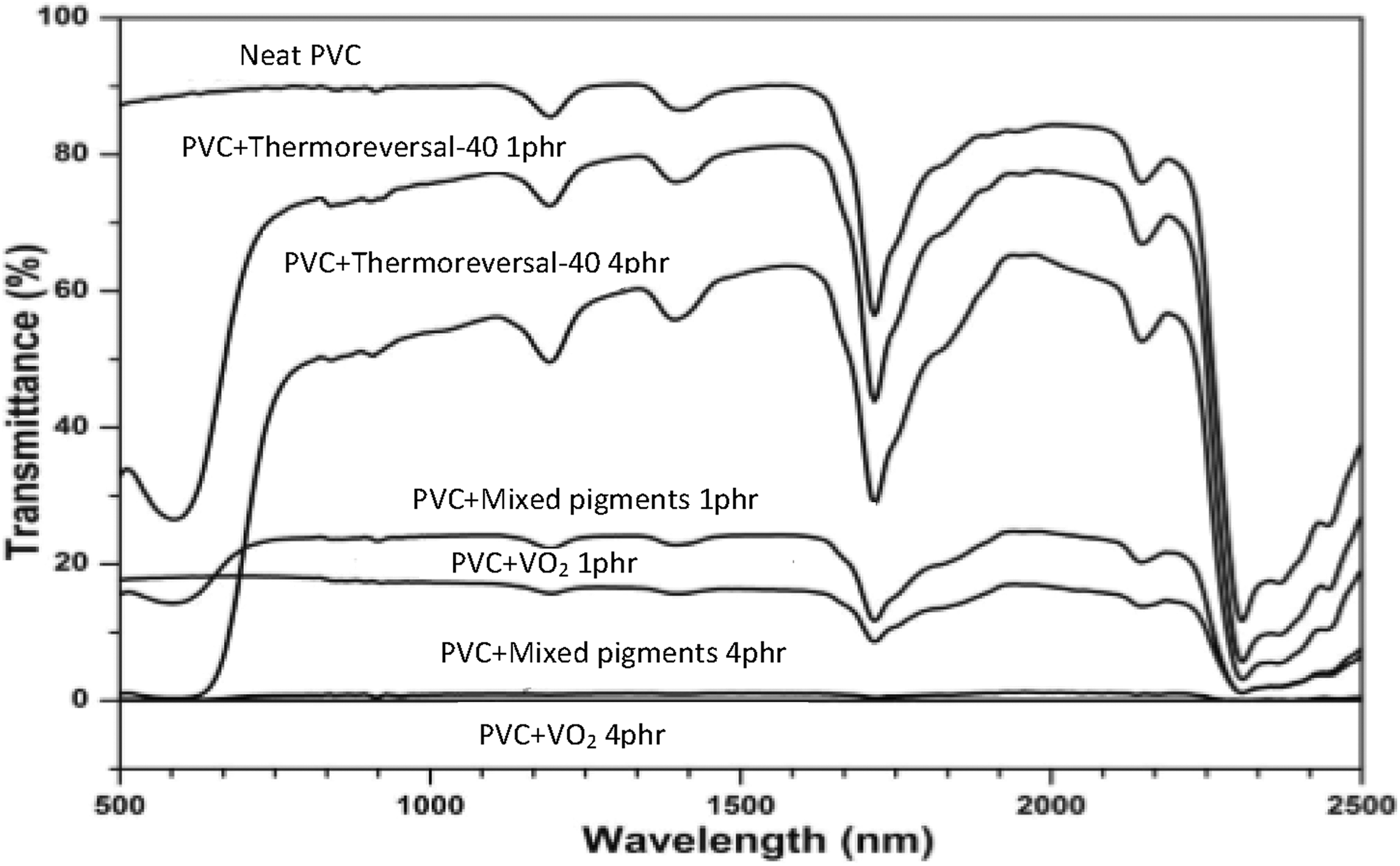
UV/Vis spectra of the various PVC composite films containing different concentration of thermoreversal-40 pigment.
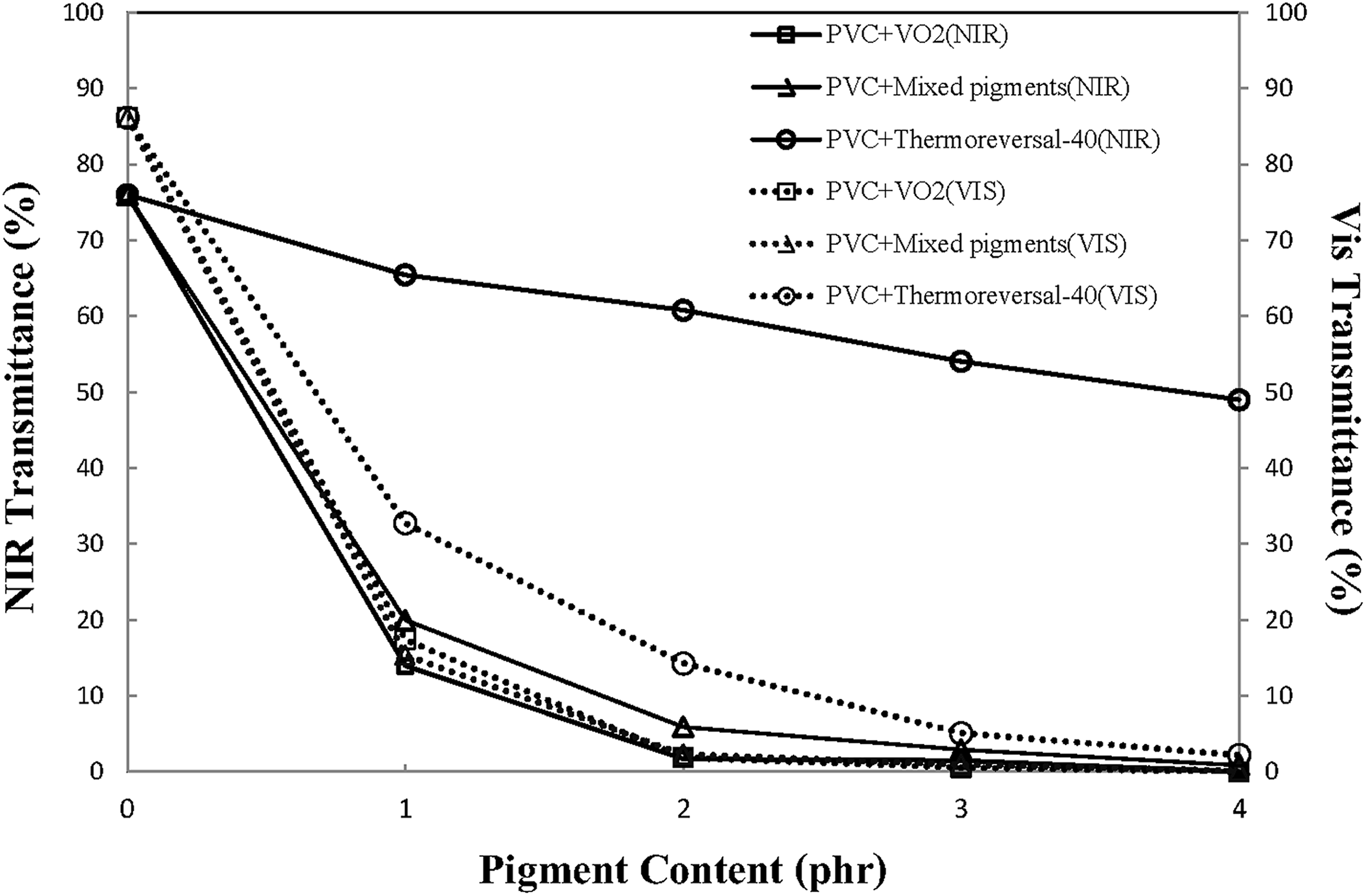
Percentage transmittance in the visible and NIR region of the PVC films containing different types and concentration of thermochromic pigments.
Figure 10 also shows that the percentage of NIR transmittance through the PVC/VO2 film and PVC/mixed pigment film is lower, as compared to those of PVC/thermoreversal film. It is noteworthy that when the PVC resin was compounded with more than 2 phr of the VO2 pigment, NIR transmittance values of the composite films as low as 1–2% can be achieved. These values are lower than that of PVC/ATO composite film (24.46% of NIR transmittance was obtained when 2 phr of ATO was mixed with PVC). 20 In this study, the lower NIR transmittance of PVC/VO2 corresponded to the greater NIR absorbance of the composite system (Figure 11). Similarly, NIR absorbance of the PVC/mixed pigments film was greater than those of the PVC/thermoreversal film that can be attributed to the presence of VO2 in the system. It was also noted that the percentage of NIR absorbance of PVC/VO2 and PVC/mixed pigments film increased with the pigment concentration. The above results indicate that, at room temperature, the VO2 particles could act as a kind of IR absorbing material. These factors contribute to a lower percentage of NIR transmittance of the film containing VO2. In this regard, the energy absorbed could be dissipated through the polymer and emitted into the atmosphere. Both phenomena could affect the thermal stability of the polymer, and the human thermal comfort indoors shielded by window film.
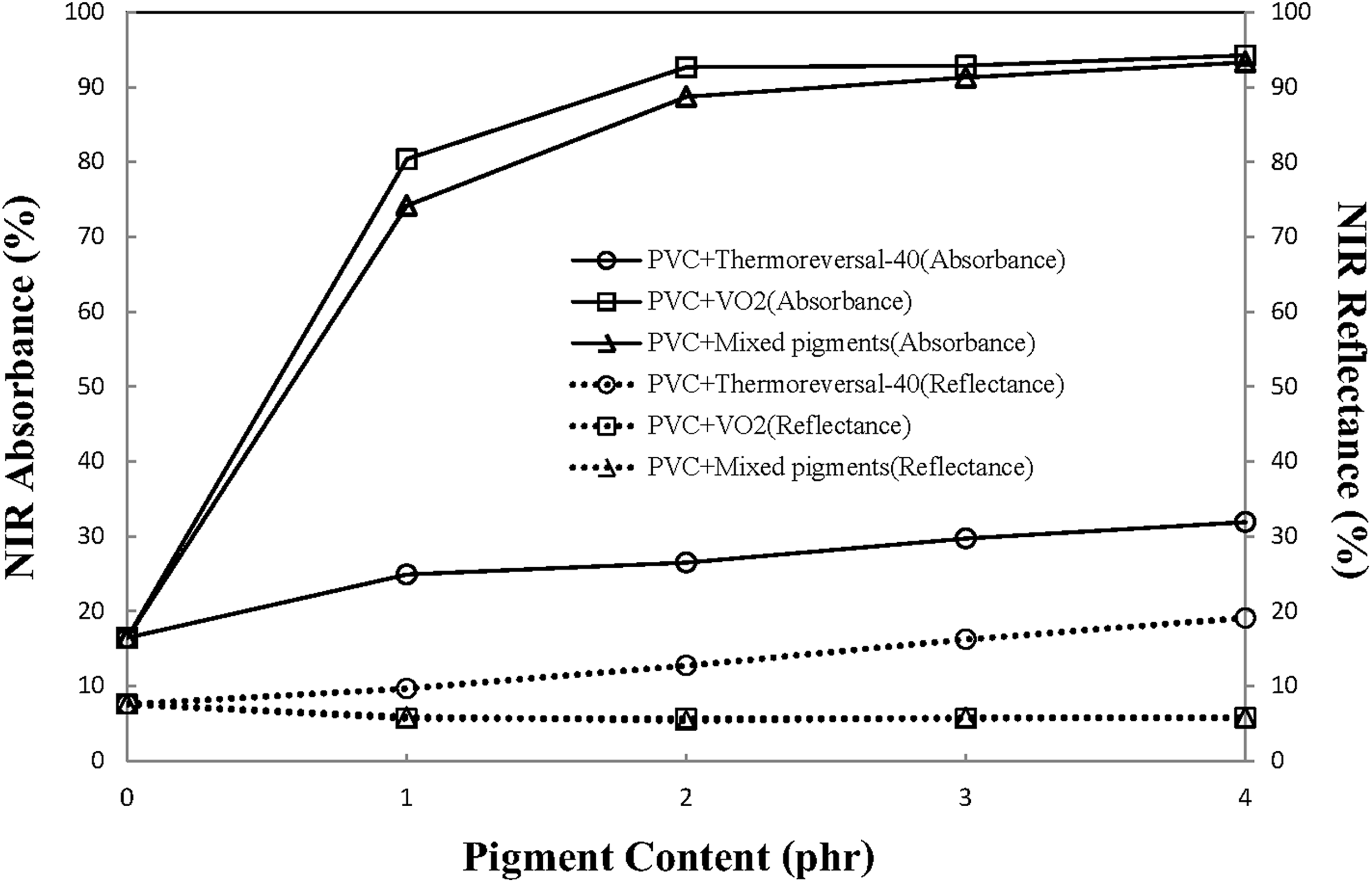
Percentage absorbance and reflectance in the NIR region of the various PVC composite films containing different types and concentration of thermochromic pigments.
In terms of spectral reflectance, Figure 11 also shows that the percentage of NIR reflectance of both the PVC/VO2 and the PVC/mixed pigment films is inferior to that of the PVC/thermoreversal-40 film. In other words, thermoreveral-40 behaves as an NIR reflective material, whereas the VO2 particles hardly reflect the NIR wave. This is because the UV/Vis spectra were recorded at room temperature, which is below the phase transition temperature of monoclinic VO2 (65°C). 19 In this regard, a spectral reflectance in the NIR region of VO2 cannot be expected. In this study, attempts were made to examine the thermochromic behaviors and insulation properties of the PVC composite films, using a model house-containing window film, equipped with an IR lamp and thermocouples. The results are described and discussed in the next section.
Heat insulation properties of window films
In this part, the temperature inside the model house equipped with PVC composite films was recorded as a function of time (Figure 12). The values were also compared to a system equipped with the neat PVC window film (without any pigment). In general, the temperature gradually increased with the exposure time and reached a plateau within 12 min. The actual temperature inside the model house, containing the neat PVC window film (without any pigment), was about 68°C. On the other hand, when the PVC composite films were used, the temperature dropped significantly, depending on the type and concentration of the pigment. At a low pigment loading (1 phr), the temperature behind the neat PVC window film was only slightly different from that behind the PVC composite films. In other words, the temperature drop (presented in Table 3) was only marginal. However, as the concentration of the pigments was increased to 4 phr, the temperature drop became more apparent. After 12 min, the temperature drop values, for the PVC/thermochromic, PVC/VO2, and the PVC/mixed pigments films, were 1.5°C, 8.2°C, and 7.2°C, respectively. In this case, the properties of PVC composite films containing VO2 pigments were outstanding. This was probably due to the fact that the formation of the rutile VO2 phase was induced by heating the monoclinic VO2 particles above their transition temperature. Since the rutile VO2 is a kind of thermochromic material, it is capable of reflecting an NIR wave. 29 Consequently, the thermal insulation properties of the PVC/VO2 can be expected. Likewise, the temperature drop for the PVC/mixed pigments system was satisfactory and only second to those of the PVC/VO2 system.
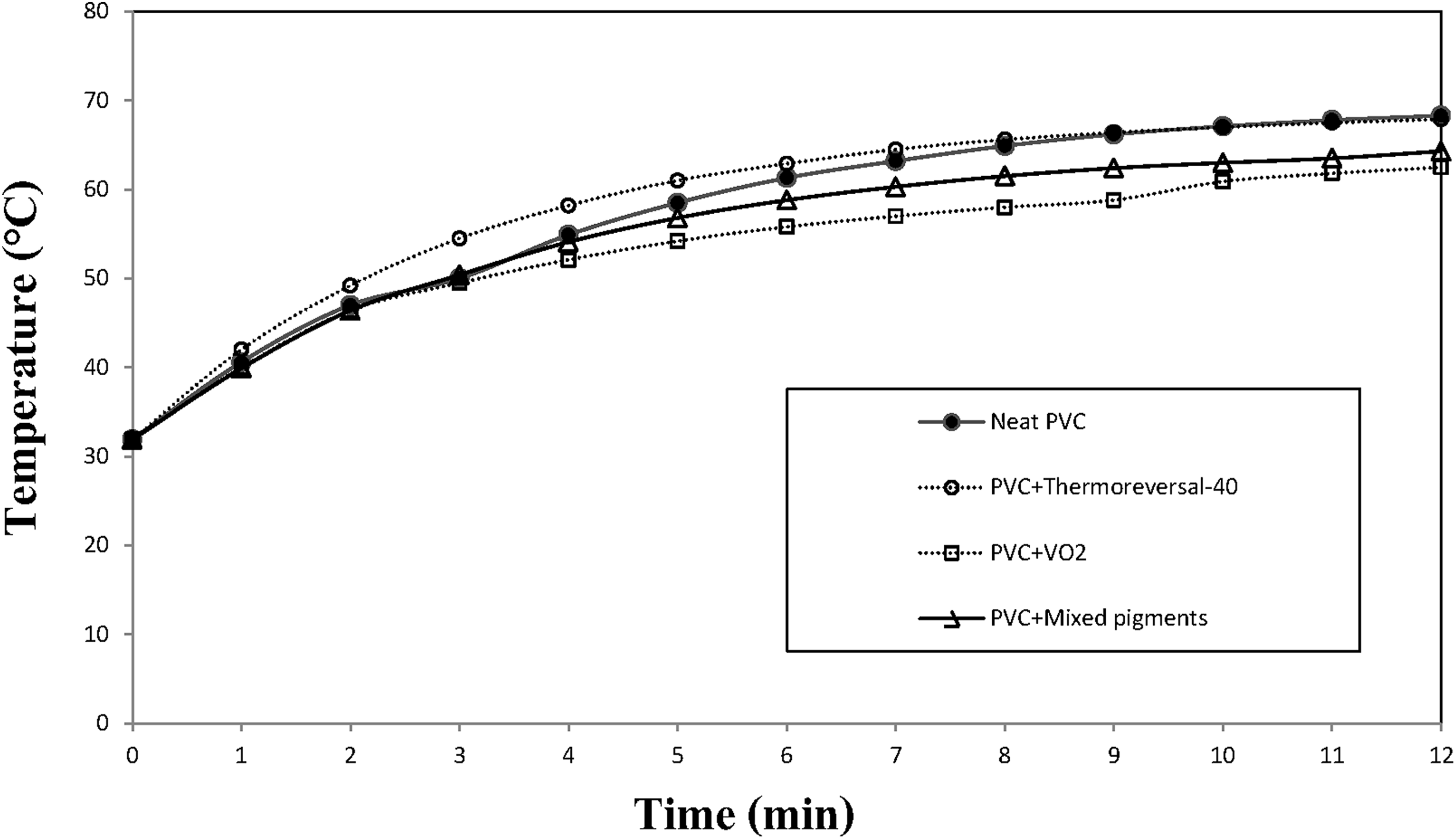
Changes in temperature in a model house, containing PVC window films containing different types of pigment at 2 phr loading.
Aging resistance
Last but not least, the stability of the PVC film containing different types and concentrations of pigments deserves consideration. In this study, the PVC composite films containing 2 phr of three different types of pigment (VO2, thermoreversol-40, and the mixed pigments) were prepared and studied for comparison purposes. The results from aging test, described in terms of the percentage of retention of the tensile strength of the PVC composite films, are presented in Figure 13. It can be seen that the tensile strength of the neat PVC film hardly decreased after aging for 8 days (194 h). This could be due to the presence of some thermal stabilizers in the PVC compound, capable of prolonging the degradation of the PVC film under the accelerated conditions (exposure to ultraviolet lamp at 70°C). However, when the VO2-based pigments were added to the PVC compound, the percentage of retention of the tensile strength of the films dropped significantly over time. The effect was more pronounced when the pure VO2 was used instead of the mixed pigments. The discrepancy could be ascribed to the dilution effect, that is, the higher the VO2 concentration, the greater the effect. It is also noteworthy that the percentage of retention of the tensile strength of the PVC/thermoreversal-40 film is close to those of the neat PVC film, regardless of the aging time. In other words, the presence of the thermoreversal-40 pigment did not have an adverse effect of thermomechanical properties of the PVC composite films. In our opinion, the inferior thermal stability of the PVC/VO2 film could be due to the fact that VO2 is capable of exhibiting a kind of NIR absorbing material (see Figure 11). Consequently, some heat energy stored in the polymer film could be dissipated through the polymer matrix and has induced some thermal degradation of the PVC molecules.
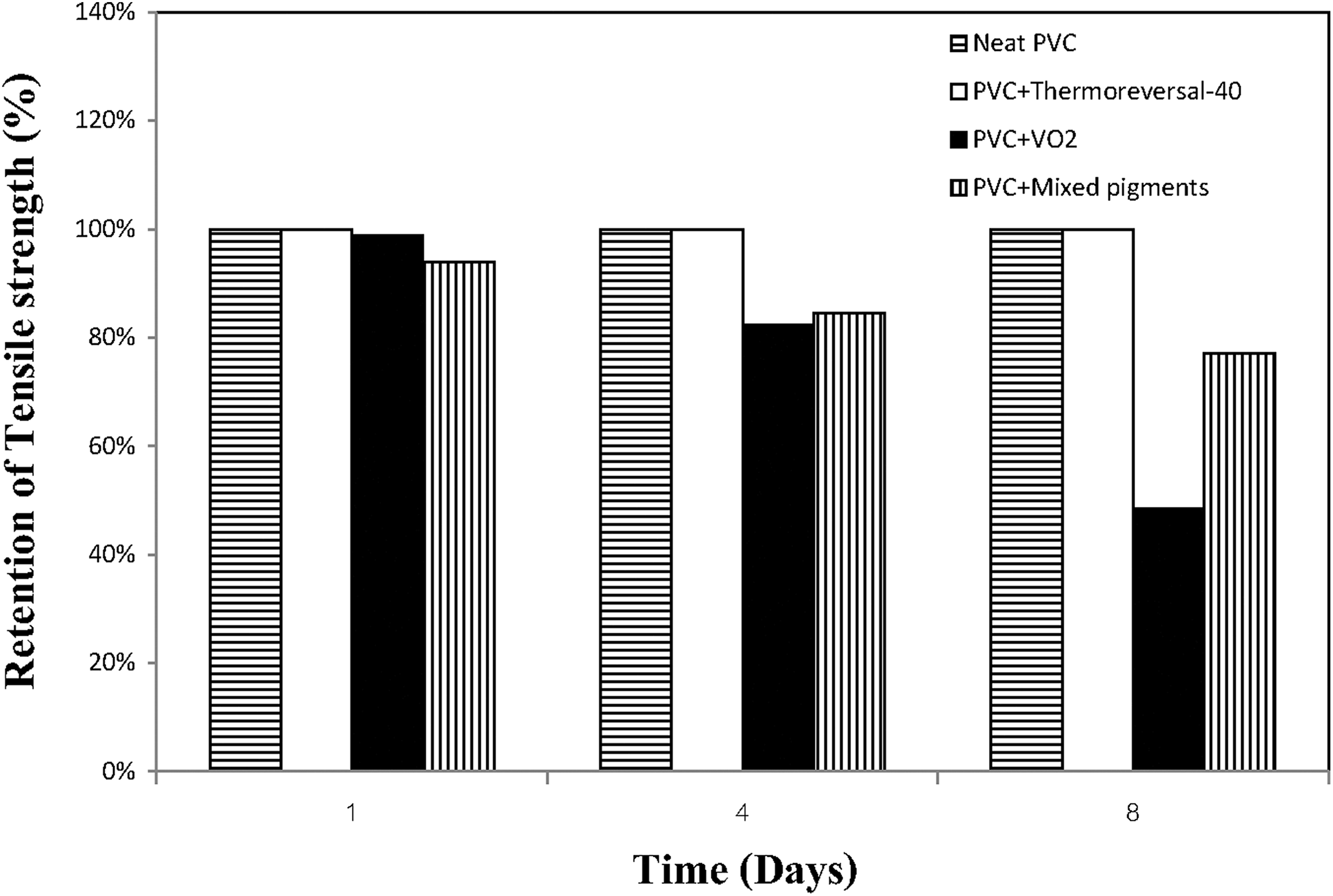
Changes in percentage retention of tensile strength with time, after thermal aging test of the PVC composite films containing 2 phr of three different types of pigment.
Conclusions
Two different types of thermochromic pigments, namely, VO2 and thermoreversal-40, were used for compounding with PVC. It was found that the types of pigment strongly affect the thermomechanical, optical, chromic, and insulation properties of the PVC composite films. The VO2 behaves like a kind of NIR absorbing material. It is also capable of inducing the gelation of PVC, which leads to the greater mechanical properties of the PVC film. In this regard, if mechanical and insulation properties of the composite films are of higher priority, the use of VO2 pigments is recommended. On the other hand, if the color contrast, chromic properties, and thermal aging resistance of the composite films are of greater concern, the use of thermoreversal40 as pigments is preferable. However, the color contrast and thermochromic properties of the PVC films containing thermoreversal-40 pigment are usually obtained at the expense of visible light transmittance at a temperature below the transition (40°C) of the pigment. Properties of the PVC composite films (NIR reflectance, light transparency, color contrast, and thermochromic behavior) could be compromised by optimizing the concentration of the mixed pigments. In overall, this study demonstrated that it was possible to apply thermochromic pigments for compounding with PVC film to develop heat reflective window film, using the suitable type and concentration of pigments. In this study, on the basis of the results from mechanical NIR transmittance tests, the optimum concentration of pigments for compounding with PVC film was 2 phr, regardless of the pigment type.
Footnotes
Acknowledgements
The authors acknowledge the King Mongkut’s University of Technology Thonburi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nanotechnology Center (NANOTEC), NSTDA, Ministry of Science and Technology, Thailand, through its program the Center of Excellence Network. This work also supported by King Mongkut’s University of Technology Thonburi through the “KMUTT 55th Anniversary Commemorative Fund.”
