Abstract
The aim of this study is to simultaneously improve the mechanical strength and fracture toughness properties of recycled poly(ethylene terephthalate) (r-PET). For this purpose, Joncryl® was used as chain extender and Lotader® was used as impact modifier. The combined effect of chain extender and impact modifier on the chemical, fractural, mechanical, and thermal properties of r-PET was investigated. Fourier transformed infrared spectroscopy (FTIR) analysis, EWF analysis, tensile test, and differential scanning calorimetry (DSC) analysis were performed. FTIR analysis revealed that all the epoxy groups in the Joncryl® were consumed during the compounding. EWF results showed that while toughness of r-PET decreased with the addition of Joncryl®, toughness was increased with addition of impact modifier Lotader®. It was found that 2.5% Lotader® usage at the same time with Joncryl® increased the tensile strength of r-PET as well as toughness. It was observed from DSC analysis that chain extender and impact modifier addition did not change the thermal transition temperatures of r-PET.
Introduction
Poly(ethylene terephthalate) (PET) is widely used in the manufacturing of soft drink and mineral water bottles, fibers, video tapes, insulating materials, etc. because of its properties such as good tensile and impact strength, transparency, thermal stability, and chemical resistance. 1 –5 Because of the wide spread usage area of PET (especially in the beverage industry), there is a need to arise for waste management of PET. 2 Recycling processes are the most proper way to overcome the management of PET waste. Generally two methods are used for recycling of PET. These methods are chemical recycling and mechanical recycling. 2
Mechanical recycling is more preferred than chemical recycling because of its simplicity and lower cost. 2 On the other hand, there are some disadvantages in this method. The most important disadvantage is the decreasing molecular weight and viscosity of PET during the recycling process. 2,3 In addition to this, contaminants which originates from recycling process of PET is another important disadvantage. Poly(vinyl chloride) labels, polypropylene or polyethylene caps and adhesives for labels fall in to this contaminant category and cause to deterioration of physical and chemical properties of PET. The change in these properties of PET with the recycling process affects the mechanical performance and restricts the usage area of it. In order to overcome the disadvantage due to decreasing molecular weight, there are some chemical methods. The most commonly used method is the usage of chemicals which include di- or multifunctional groups. These chemicals act as bridges between degraded chains of polymers by means of their functional end groups. This is called “chain extension.” This chain extension reaction results in increased molecular weight, viscosity, hence mechanical properties of polymer. 6 Di- or multiepoxides, diisocyanates, dianhydrides, and bis-oxazolines are commonly used chain extenders for recycled PET (r-PET) and in the literature there are many studies which investigated the effects of chain extenders on the properties of r-PET. 6 –19 Villalobos et al. 6 used the epoxy functional styrene-acrylic oligomer as a chain extender for recycled PET, poly(butylene terephthalate), polyamide 6, and polycarbonate. Their results revealed that mechanical and rheological properties of chain-extended polymers were similar to or greater than virgin polymers at small loading levels of chain extender (<1.5 %). Awaja and Daver 7 investigated the influence of chain extender (pyromellitic dianhydride (PMDA)) concentration and extruder residence time on the intrinsic viscosity and carboxyl content of r-PET. Their study proved that PMDA is a proper chain extender for r-PET. In addition to this, chain extended r-PET exhibited higher intrinsic viscosity and lower carboxyl content than virgin r-PET. This study also revealed that although chain extender content strongly affects the chain-extension process, extruder residence time has lower effect on the intrinsic viscosity of the final product. Bimestre and Saron 16 investigated the chain-extension performance of secondary stabilizer Irgafos 126 (IRG) on variable concentrations. Their results revealed that IRG increased molecular weight, decreased crystallinity and changed processing behavior of the recycled PET. They also compared the efficiency of IRG with that of well-known chain extender PMDA and they found that IRG can also act as a chain extender for the recycled PET. Zhang et al. 17 studied the chain-extension process of recycled PET by using bisphenol-A dicyanate (BADCy) chain extender. Their results revealed that increasing amount of BADCy increased the viscosity and storage modulus of recycled PET. They also found that melting and crystallization temperature of chain-extended PET were shifted to low temperatures. This result was interpreted as the reduced chain mobility of modified recycled PET and correspondingly difficulty in crystallization. Cavalcanti et al. 19 investigated the use of triphenyl phosphite (TPP) as a chain extender of virgin and recycled PET and they prepared 1% and 3% TPP include compounds. They found that the maximum chain extension and the minimum subsequent degradation were observed for 1% TPP chain extender include compounds. Their work also proved that the recycled PET is much less able to react with TPP in comparison with the virgin polymer.
While the chain extender addition improves the molecular weight, viscosity, and mechanical strength of r-PET, and the fractural properties and toughness of r-PET can be improved by melt blending of r-PET with some kind of impact modifiers such as glycidyl methacrylate (GMA)-modified polyolefin elastomer, maleic anhydride-grafted SEBS triblock copolymer, poly(butylene adipate-co-terephthalate). 1,20,21 In the literature, it appears that especially GMA-based impact modifiers are widely used for polyesters (such as PET) because of the reaction tendency between glycidyl functionality and terminal groups of polyesters. 22,23 Xanthos et al. 22 used diimidodiepoxide as a novel chain extender for PET and compared reactivity of diimidodiepoxide with that of GMA copolymer. Their results indicated that the reactivity of diimidodiepoxide chain extender is higher than GMA copolymer even at lower loading level. Xanthos et al. also proved that chain-extended PET exhibited increased viscosity and storage modulus value with respect to processing time and chain extender loading level. Mouzakis et al. 1 evaluated the static fracture toughness of PET which include GMA-grafted ethylene-propylene rubber by using essential work of fracture (EWF) method. They found that essential, nonessential, and plastic work decreased with increasing GMA-grafted ethylene-propylene rubber loading level.
In this study, it was aimed to simultaneously improve the mechanical strength and fracture toughness properties of r-PET. For this purpose, epoxy functionalized, multifunctional polymer Joncryl® was used as chain extender and ethylene-acrylic ester-GMA terpolymer Lotader® AX8900 was used as impact modifier. The originality of this work is that it investigates the simultaneous effect of chain extender and impact modifier on the fractural, mechanical, chemical, and thermal properties of r-PET. In addition to these, EWF was used to evaluate the fracture toughness of r-PET that includes chain extender and impact modifier.
Experimental
Materials
Mixed clear and light-blue recycled PET flakes (Figure 1) were supplied from a local recycling company in Kocaeli (Turkey). Supplied recycled PET flakes were obtained from beverage bottles and their type is APET. Contaminant values and some properties of supplied recycled PET flakes were given in Table 1. Commercial styrene acrylic emulsion Joncryl® was purchased from BASF with Joncryl® ADR-4300 trade name. Lotader AX8900 random terpolymer of ethylene, acrylic ester, and GMA was used as impact modifier and obtained commercially from Arkema Functional Polyolefins.

Appearance of r-PET flakes.
Contaminant values and properties of supplied r-PET.

Melt processing
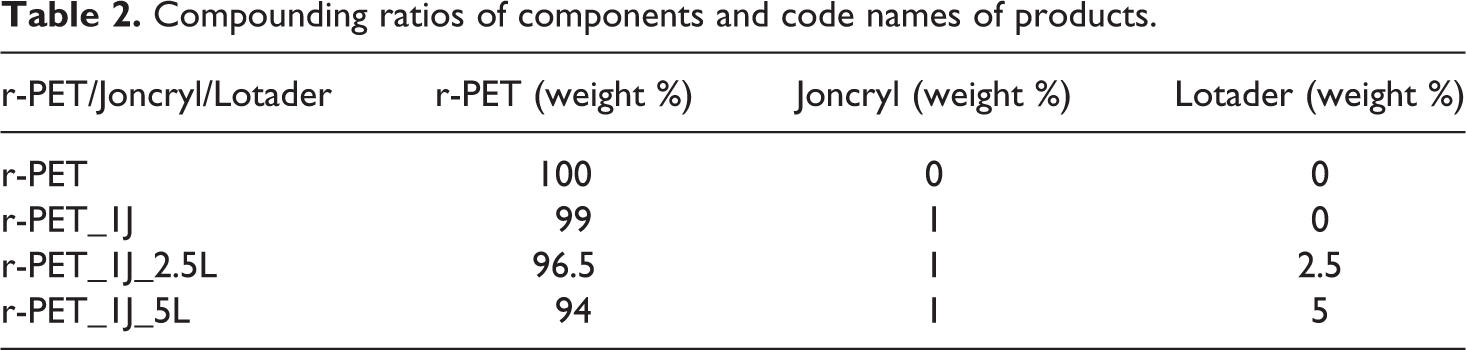
r-PET flakes were dried in a vacuum oven at 80°C for 12 h before compounding process. A laboratory scale twin screw corotating extruder was used for preparing compounds. The compounding ratios of components and code names of products were given in Table 2. Chain extender amount in the compounds was fixed at wt. 1%. This value was chosen based on literature. 6,22 Compounding was conducted at 275°C barrel temperature and at a constant residence time of 3 min. Also during the compounding process, extruder screw rotation speed was kept constant at 100 rpm. Extruded compounds were subsequently injection molded with a laboratory scale injection molding machine. The barrel temperature and mold temperature were 275 and 25°C, respectively; and injection pressure was 10 bars.
Compounding ratios of components and code names of products.
Characterization
Fourier transformed infrared spectroscopy
Fourier transformed infrared spectroscopy (FTIR) analysis was performed by using a Perkin-Elmer Spectrum 100 spectrometer in order to analyze the intensity of carboxylic group of r-PET with and without chain extender and impact modifier. The infrared spectra of products were recorded in the range of 650–4000 cm-1 by using an Attenuated Total Reflectance apparatus.
Essential work of fracture
In order to evaluate the essential work of fracture (EWF) parameters of products, double edge notched tension (DENT) specimens with 80 mm length, 40 mm width, and 2 mm thickness were used (Figure 2). Initially, dual V-shaped notches were drilled into the DENT specimens and after that these notches were sharpened by a razor blade in order to form a crack with crack tip radius of <2 µm. The depths of notches were varied from 6 to 14 mm so it was allowed to the length of the ligament would vary in 2 mm increments according to the ESIS (Test Protocol for Essential Work of Fracture) recommendations. 24 After that, the exact ligament lengths were measured by an optical microscope. Finally, tensile tests of V-shaped notched DENT specimens were performed in an Instron 4411 universal testing machine at a constant crosshead speed of 2 mm min−1.
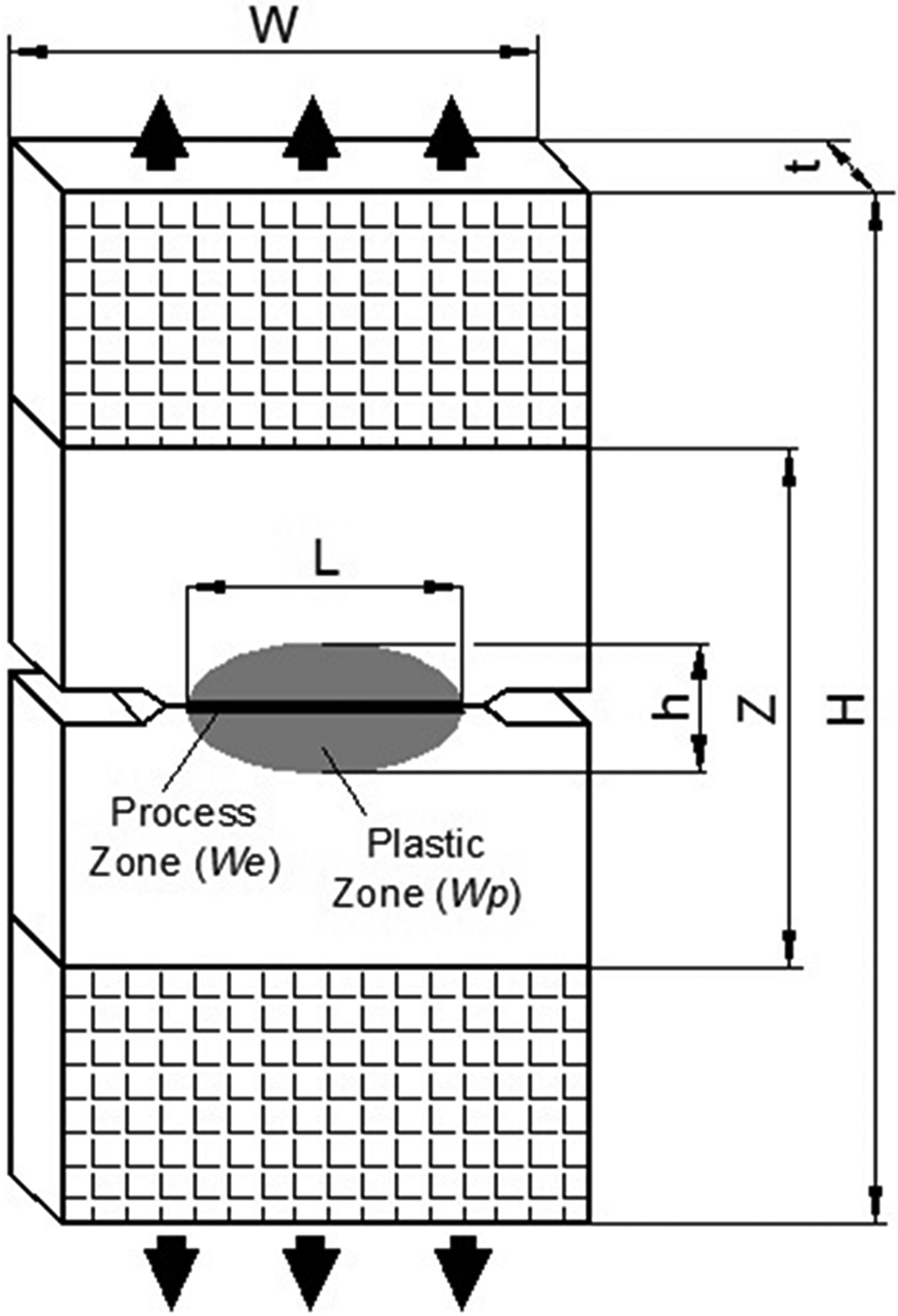
Double edge notched tension specimen.
Tensile test
Tensile tests were performed according to ASTM D 638-10 by using Instron 4411 universal testing machine. Average tensile strength values of compounds were determined using five dumbbell-shaped samples for each composition of product at a constant crosshead speed of 5 mm min−1. Dimension of the test samples were 4 mm width, 2 mm thickness, and 30 mm length.
Differential scanning calorimeter analysis
Glass transition temperature (Tg), crystallization temperature (Tc), melting temperature (Tm), and melting enthalpy (ΔHf) of the products were measured with a Mettler Toledo DSC 1 model differential scanning calorimeter under nitrogen atmosphere. The differential scanning calorimetry (DSC) analysis was carried out in the temperature range from 25 to 300°C at a heating rate of 5°C min−1. The relative degree of crystallinity of the samples was calculated with the following expression:
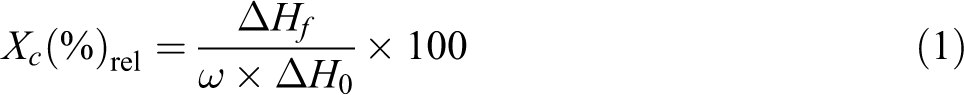
where, ΔHf is the experimental heat of fusion of product, ω is the weight fraction of the r-PET in the product, and ΔH0 is the heat of fusion of virgin r-PET. 25
Results and discussion
FTIR
FTIR spectra of the r-PET, Joncryl®, and their compounds were given in Figure 3. The peak at 758 cm−1 on the spectra of Joncryl® was attributed from the C-O stretching the epoxy groups in the Joncryl®. On the other hand, this peak did not occur on the spectra of compounds. This result indicates that all the epoxy groups in the Joncryl® consumed during the compounding process.
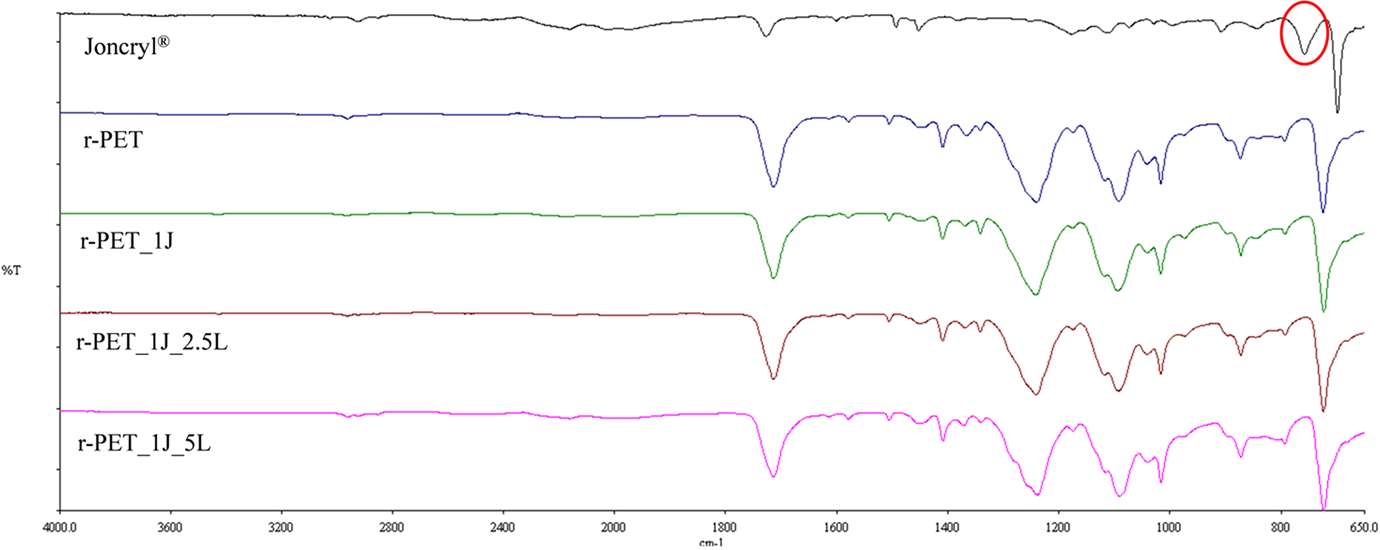
FTIR spectra of samples.
In the case of using epoxy-based chain extenders, both the esterification and etherification reactions occur between carboxyl and hydroxyl end groups of polyester, respectively. 26 However, it was assumed that epoxy groups prefer to react with carboxyl groups than hydroxyl groups at the beginning of the mixing period. 22,27 For this reason, carboxyl group content of r-PET gives clues about the efficiency of chain-extension reaction.
In the literature, it was reported that amount of carboxyl chain ends decrease with increasing molecular weight of PET. 2 According to this result, it can be concluded that decreasing amount of carboxyl groups can be interpreted as efficient chain-extension reaction between r-PET and Joncryl®. In this study, carboxyl peak height which was observed from FTIR spectrum was used for assessment of approximate carboxyl group amount and results were given in Figure 4(a)–(d). These figures reveal that the minimum peak height was observed for r-PET_1J_2.5L sample and its value is 42. It can be said that while Joncryl® reacts with carboxyl end groups of r-PET, GMA-based Lotader® also reacted with carboxyl groups of it. This case resulted from the reaction tendency of glycidyl group of GMA with terminal groups of PET. 22 Besides, Figure 4(d) also shows that peak height increased with increasing Lotader amount. This increase may result from the high amount of Lotader and we can conclude that 2.5% Lotader is optimum value for obtaining better property products.
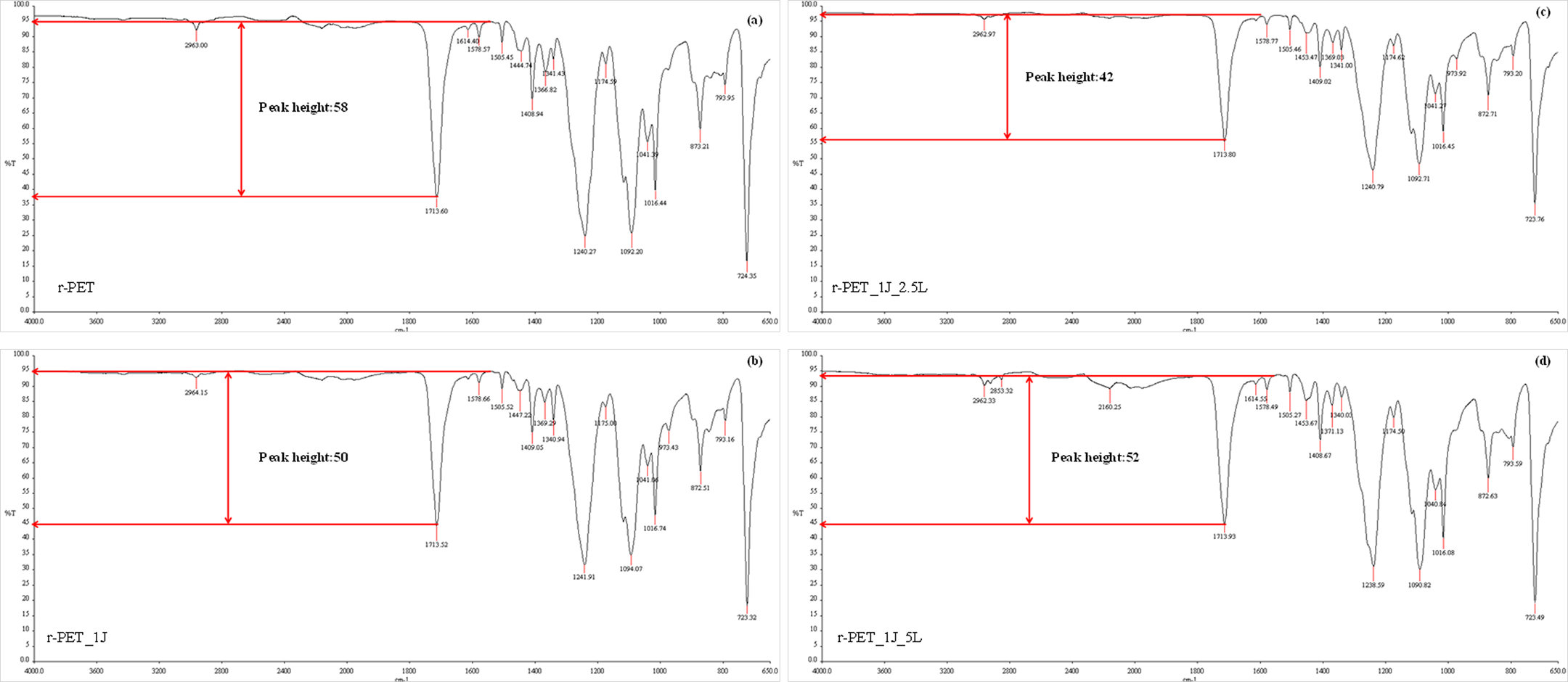
Carboxyl peak height values obtained from FTIR spectra, (a) r-PET, (b) r-PET_1J, (c) r-PET_1J_2.5L, (d) r-PET_1J_5L.
Essential work of fracture
EWF method was firstly proposed by Broberg 28,29 and then developed by Cotterell, Reddel, and Karger-Kocsis. 30 –33 This method gives information about the fracture toughness of ductile materials. According to this theory, when the load is applied to a ductile-type notched specimen, plastic flow occurs around the notch zone and there is a separation between the process zone where the actual crack propagation occurs and the plastic deformation zone that surrounds the process zone. 1 As a result, when these two types of process zones are considered, the total work required to fracture this notched sample can be separated into two components:

where Wf is the total fracture work, W e is the EWF consumed in the process zone where the actual crack propagation occurs, and Wp is the plastic (or nonessential) work consumed in the plastic deformation zone. 34 W e is the surface energy term and its value is proportional to ligament area (Lt). On the other hand, Wp is the volume-dependent energy and its value is proportional to the volume of the yielded zone (L2t). 35 As a result We and Wp terms can be rewritten as follows:


If equations 3 and 4 are inserted in the equation 2 and rearranged, the following equation can be observed:

where w f is the specific total fracture work, w e is the specific EWF, w p is the specific plastic (or nonessential) work, L is the ligament length, t is the specimen thickness, and β is the shape factor related with plastic deformation zone. 35 It can be deduced from equation (5) that specific total fracture work, w f, is the function of the ligament length, L, and it can be determined from the intercept of the linear regression line, fitted to the w f versus L graph; in addition to this the slope of this line gives βw p. 35
In order to evaluate the EWF data, first of all it is wise to investigate the load-displacement curves which are obtained from DENT samples. In Figure 5, load-displacement curves of samples were collated from a ligament of about 12 mm length. It is obviously seen from figure that r-PET_1J sample undergoes a brittle fracture; on the other hand, other samples exhibit a ductile fracture and full ligament yielding.
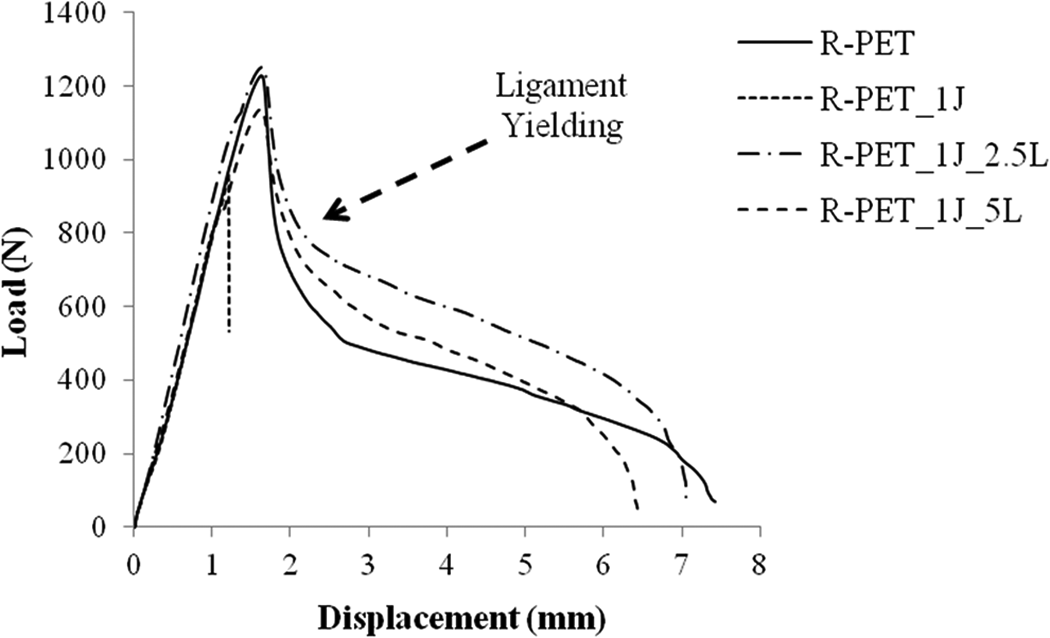
Load-displacement curves of all DENT samples at the about 14 mm ligament length.
Specific EWF curves versus ligament lengths for the prepared samples were shown in Figure 6(a)–(d). Also the values of w e and w p β, which were obtained from the interception and the slope of the straight lines extrapolated to zero ligament length, were listed in Table 3 with the regression coefficients. Figure 6(a)–(d) show that all the samples exhibited a linear increase in the specific work of fracture with increasing ligament length. According to Figure 6(a)–(d) and Table 3, results show that we decreased with the addition of Joncryl® to the r-PET. This result means that addition of chain extender decreased the toughness and crack resistance of r-PET. In case of the reaction between epoxy-based chain extender and carboxylic acid end group of polyester, there could be formed gelation because of the hydroxyl and carboxyl groups react with the more than two epoxide groups per chain extender molecule and this case may result in the decreasing of toughness. 27
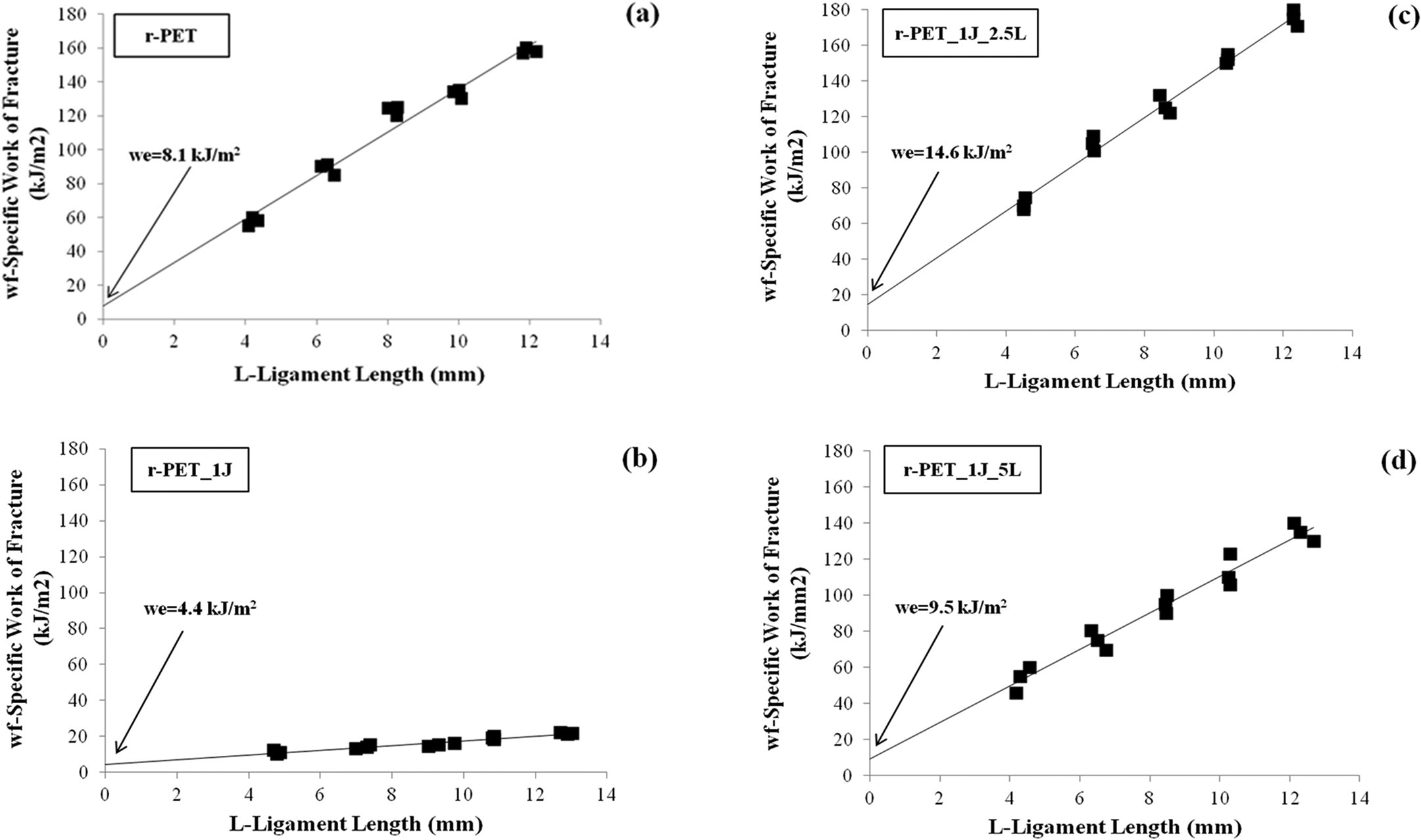
Specific essential work of fracture curves versus ligament lengths.
EWF data of samples.

Figure 6(a)–(d) and Table 3 also shows that we increased with addition of impact modifier and the highest we value was observed for the r-PET_1J_2.5L sample. It can be concluded that while 2.5% impact modifier addition increased the crack resistance behavior of chain extended r-PET, 5% impact modifier addition decreased it. It is known from the literature that impact modifiers can act in two different roles. In one of roles they act as craze initiators and in the other role they act as craze terminators. 35 In our case, while GMA-based impact modifier acted as a craze terminator at the 2.5% loading level, it started to act as a crack initiator after this amount. For this reason, it can be concluded that 2.5% Lotader is optimum value for obtaining samples with higher impact resistance and toughness (we and wpβ terms both had high values).
Tensile test
As mentioned before, mechanical recycling is simple and low-cost method for recycling of PET. 2 However, there are some disadvantages for this method. One of these disadvantages is the presence of some contaminations in recycled PET which originate from recycling process. These contaminants generate some problems such as chain scission, molecular weight reduction, decrease in intrinsic viscosity, and correspondingly decrease in mechanical performance of material. 36 Therefore, contamination amount in the recycled PET is one of major causes of deterioration of its physical and chemical properties. 2 For this reason, it is important to minimize the adverse effect of these contaminants for obtaining better quality of r-PET. As a result, it can be concluded that chain extender usage increases the molecular weight of recycled PET as well as minimize the adverse effect of contaminants.
Tensile strength values of samples were given in Figure 7. According to tensile test results, while 1% Joncryl® addition to r-PET increased its tensile strength value as 6%, 1% Joncryl®, and 2.5% Lotader addition simultaneously, increased this value as 5.5%, 1% Joncryl®, and 5% Lotader addition simultaneously, decreased this value as 14%. When EWF and tensile test results are considered, it can be said that Joncryl® chain extender addition to r-PET increased the tensile strength but it decreased the toughness of r-PET. On the other hand, 2.5% Lotader impact modifier usage at the same time with Joncryl® chain extender not also increased the toughness of r-PET, it also increased the tensile strength of it. As it is mentioned before, this result originated from the reaction of Joncryl® with carboxyl end groups of r-PET and reaction of GMA-based Lotader® with carboxyl groups of r-PET. 22 However, 5% Lotader impact modifier usages at the same time with Joncryl® chain extender did not make the same effect on the toughness and especially tensile strength of samples. This result reveals the importance of impact modifier content on the properties of samples.
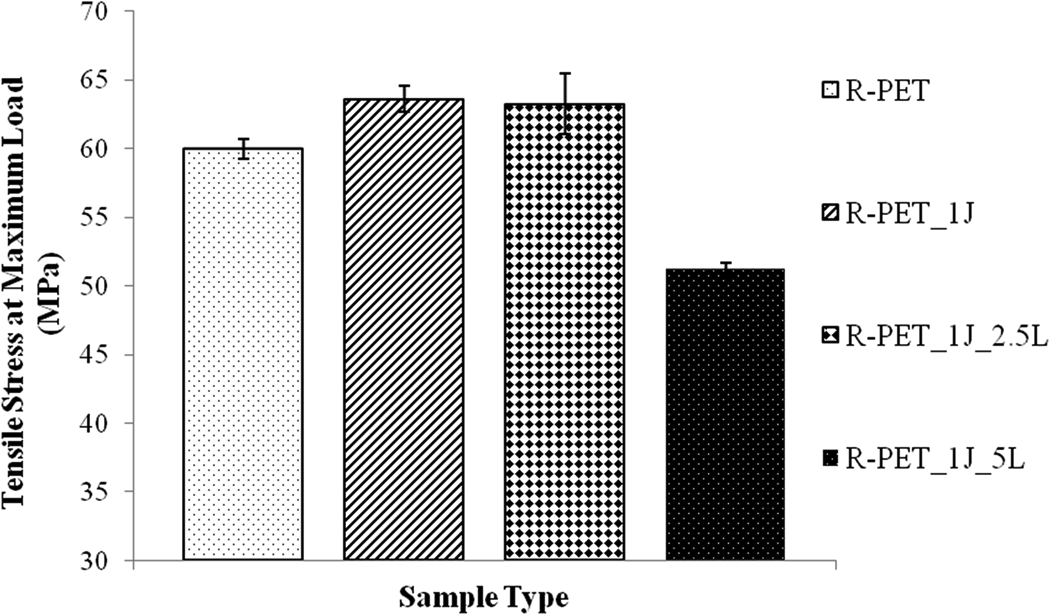
Tensile strength values of samples.
Differential scanning calorimeter analysis
In order to investigate the effects of chain extender and impact modifier on the thermal transition and properties of r-PET, DSC analysis was used. Tg, Tc, Tm, and melting enthalpy values were observed from DSC analysis, also crystallinity of r-PET was calculated and results were given in Table 4. According to Table 4, it can be said that chain extender and impact modifier addition did not have an effect on thermal transition temperatures of r-PET.
DSC analysis results of samples.
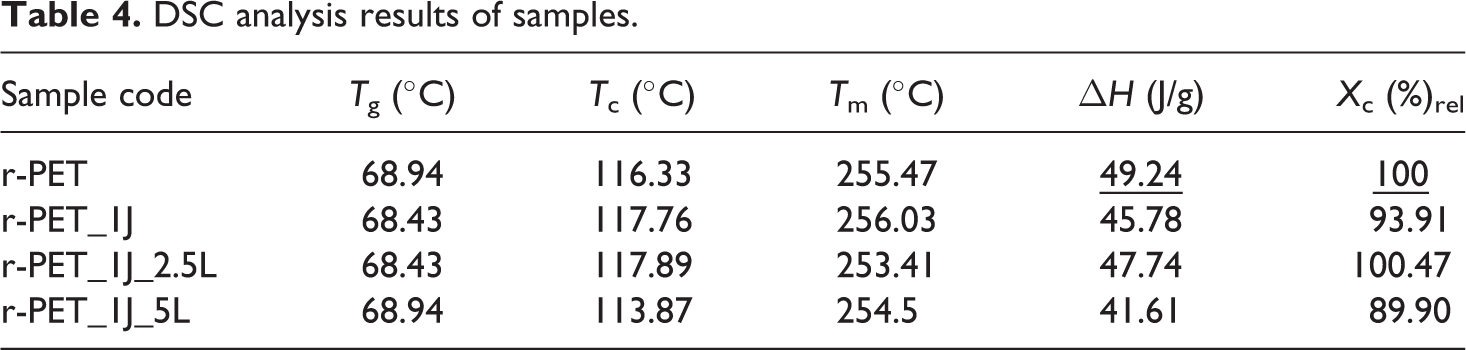
Crystallinity has a strong effect on the end-use application of r-PET and it fundamentally depends on the molecular structure and crystallization conditions. While decrease in crystallinity can be interpreted as chain entanglements and the difficulties in chain folding into a crystal structure due to the high degree of branching, increase in crystallinity can be interpreted as chain disentanglement. 2 When it is investigated the relative crystallinity (Xc (%)rel) values of r-PET are investigated (Table 4), it can be concluded that Joncryl® addition decreased the crystallinity of r-PET. On the other hand, 1% Joncryl® and 2.5% Lotader addition increased the crystallinity slightly. This result reveals that chain extension of r-PET with Joncryl in the presence of Lotader does not hinder the chain folding of r-PET into a crystal structure as well it promote this case. In addition to this, 1% Joncryl® and 5% Lotader addition simultaneously, decreased the crystallinity of r-PET considerably. This result is based on the chain entanglement of r-PET and it is due to the high degree of branching which may resulted from high amount of Lotader impact modifier.
Conclusions
This study is focused on to improve the fractural and mechanical properties of r-PET simultaneously by means of chain extender and impact modifier addition. For this purpose, multifunctional polymer Joncryl® was used as chain extender and ethylene-acrylic ester-GMA terpolymer Lotader® was used as impact modifier. Prepared samples were characterized by FTIR, EWF, DSC analyses, and tensile test. FTIR spectra revealed that all the epoxy groups belong to the Joncryl® consumed during the compounding process. This result attributed to reaction of these groups with carboxyl groups of r-PET. Because decreasing amount of carboxyl groups can be interpreted as efficient chain-extension reaction between r-PET and Joncryl®, carboxyl peak height which observed from FTIR spectrum was used to evaluate the approximate carboxyl group amount in the compounds. Results showed that the minimum peak height was observed for r-PET_1J_2.5L sample. EWF method was used to assess the fracture toughness of samples. According to EWF test results, 1% chain extender addition decreased the toughness and crack resistance of r-PET. On the other hand, toughness and crack resistance increased with simultaneous addition of 2.5% impact modifier and 1% Joncryl®. However this value decreased when the impact modifier amount was increased to 5%. Tensile test results showed that while only 1% Joncryl® addition and simultaneous 1% Joncryl® to 2.5% Lotader addition to r-PET increased its tensile strength value simultaneously, 1% Joncryl® to 5% Lotader addition decreased this value. According to DSC analysis results, it can be said that Tg, Tc, and Tm values of samples did not change with the addition of chain extender and impact modifier. While crystallinity of r-PET decreased with only 1% Joncryl® and 1% Joncryl® to 5% Lotader addition, this value increased with 1% Joncryl® to 2.5% Lotader addition. This result can be interpreted as the chain extension of r-PET with Joncryl® in the presence of Lotader® promotes the chain folding of r-PET into a crystal structure. As a conclusion of all results, it can be summarized that addition of 1% Joncryl as a chain extender and 2.5% GMA-based Lotader as an impact modifier, increases both fracture toughness and tensile strength of r-PET. Thus, usage area of r-PET can be expanding owing to its enhanced properties.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
