Abstract
“Sea-island” structure silver/polyaniline (Ag/PANI) nanocomposites were synthesized through sonication of an aqueous solution with silver nitrate and aniline at the temperature of 20 ± 1°C under nitrogen atmosphere. The nanocomposites were characterized by X-ray diffraction, Fourier transform infrared spectroscopy, ultraviolet–visible absorption spectroscopy, field emission scanning electron microscopy, and energy dispersive spectrometry, respectively. Furthermore, Ag/PANI nanocomposites were immobilized on the surface of glassy carbon electrode (GCE) and electroactivity behavior was investigated by cyclic voltammetry and differential pulse voltammetry. The obtained sea-island structured Ag/PANI nanocomposite-modified GCE showed high electrocatalytic activity for the oxidation of
Introduction
Among these modifiers, nanocomposites of conjugated CPs and inorganic nanoparticles have attracted considerable attention recently due to their unique physical properties and have been widely used in technological applications. 20 –25 These hybrid nanomaterials not only combine the advantageous properties of metals and polymers but also are expected to exhibit several synergistic properties between the polymer and the metal nanoparticles, making them potential candidates for applications in numerous fields like biosensors 26 and catalysis. 27 PANI is the most important electrically conducting polymers and has a wide spectrum of applications in the fields of electrochemical sensors, biosensors, conductive coating, and anticorrosion materials. 28 On the other hand, since Ag exhibits the highest electrical and thermal conductivities among all the metals, the combination of PANI with Ag nanoparticles seems to be the best switch to prepare the important electro-active and thermostable nanocomposites. 28,29
In previous studies, Ag/PANI nanocomposites can be obtained by reduction of Ag salt with pre-synthesized PANI, 30 polymerization of aniline in Ag nanoparticles containing medium, 31 in situ photoredox reaction, 32 and electrodriven deposition of silver crystals in polyaniline. 33 However, each of these methods has advantages and disadvantages from preparation facility and morphological view. Therefore, it is significant to develop a facile method to obtain Ag/PANI nanocomposites with a novel morphology. Ultrasound radiation has turned out to be a very attractive and efficient tool for many researchers. The main advantages of ultrasound waves in the polymerization process are the absence of external chemical initiators and possibility. de Barros and de Azevedo have prepared fibril Ag/PANI nanocomposites via ultrasound radiation. 34
In this article, we demonstrate, for the first time, that “sea-island” structured Ag/PANI nanocomposites were synthesized in the presence of aniline and silver nitrate (AgNO3) via ultrasonic technique. The obtained Ag/PANI nanocomposites showed considerably high electrocatalytic activity for the oxidation of
Experimental
Materials
Aniline, ethanol, and
Preparation of sea-island structured Ag/PANI nanocomposites
In a typical synthesis, a solution of 5 × 10−3 M aniline and a solution of 5 × 10−3 M AgNO3 were mixed with 1 M nitric acid in a glass vessel, keeping the molar ratio of aniline to AgNO3 as 1:1. The covered vessel was immerged into a water bath and kept at 20 ± 1°C and was irradiated with ultrasonic waves using a multiwave ultrasonic generator (Xianou XO-1800D multifunction ultrasonic cell disruptor, China; 6 cm diameter titanium horn, 20 kHz, 200 W output power). The solutions to be sonicated were purged with nitrogen and were kept under nitrogen throughout the experiment. After being irradiated for 30 min, Ag/PANI nanocomposites were obtained. The product was precipitated and washed with ethanol three times, until the remaining solution became clear, and dried under vacuum at 40°C for 18 h.
Characterization of sea-island structured Ag/PANI nanocomposites
X-ray diffraction (XRD) pattern was recorded on a XD-3 type X-ray diffractometer employing copper K α radiation with 36 kV and 30 mA. Fourier transform infrared (FTIR) absorption spectra, between 4000 cm−1 and 400 cm−1, were obtained using a Nicolet spectrometer model 380 FTIR (Waltham, Massachusetts, USA) on potassium bromide pellet of the samples. Ultraviolet-visible (UV-Vis) absorption spectroscopy of the prepared suspension was obtained by an UV-Vis spectrophotometer from Shimadzu (Japan) with quartz cuvettes over wavelengths from 250 nm to 800 nm. The field emission scanning electron microscopy (FESEM) images and energy dispersive spectrometry (EDS) were performed with a Sirion 200 instrument (FEI, USA).
Electrochemistry tests
Electrochemical measurements were performed with a CHI 660E electrochemical workstation (CH Instruments, Chenhua Co., Shanghai, China) and conducted on a conventional three-electrode cell, which includes a platinum wire as counter electrode, a saturated calomel electrode as reference electrode, and the Ag/PANI-modified glassy carbon electrode (GCE, 3 mm in diameter) as working electrode. The GCE was polished with alumina slurry followed by a rinse with deionized water, then allowed to dry at the room temperature. Ag/PANI were dispersed in distilled water to form a 0.5 mg/mL solution, and 6 μL of a suspension containing Ag/PANI nanomaterials was dropped on the clean electrode surface and dried in air. The solutions were deaerated thoroughly for at least 30 min with pure nitrogen and kept under a positive pressure of this gas during the experiments. All experiments were performed at room temperature.
Results and discussion
XRD study
The XRD pattern of the Ag/PANI nanocomposites obtained in the above typical experiment is shown in Figure 1. Four characteristic peaks of palladium at 2θ = 38.2°, 44.4°, 64.5°, and 77.4° corresponding to the (111), (200), (220), and (311) lattice planes, are observed. All the diffraction peaks can be well-indexed to fcc-Ag according to the JCPDS card no. 4-0783, indicating that the as-prepared Ag nanomaterials have a high purity and high crystallinity. According to Scherrer’s equation, the reflecting peaks at 2θ = 38.2°and 77.4° were chosen to calculate the average diameter, and the average size of Ag nanoparticles was about 48 nm.
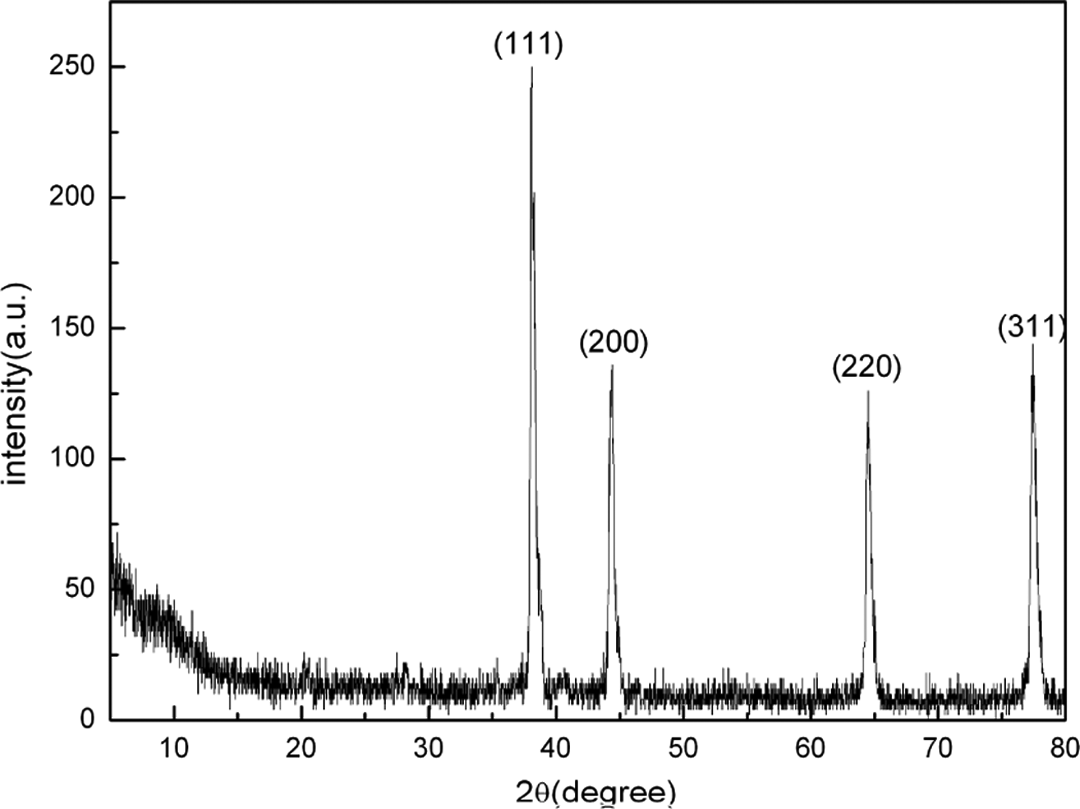
XRD patterns of Ag/PANI nanocomposites. XRD: X-ray diffraction; Ag: silver; PANI: polyaniline
UV-–Vis absorption study
Figure 2 shows the UV–Vis absorption spectra of Ag/PANI nanocomposites. Characteristic peaks of Ag/PANI nanocomposites were obtained at 271 and 370 nm, which are attributed to π–π* transitions. 35 The characteristic peaks for the surface plasmon resonance (SPR) peak of the Ag and the polaron–π* and π–polaron transitions of PANI are all absent, which could be explained on account of the strong interaction between Ag and PANI. 36 It is well known that the SPR bands of metal nanoparticles are sensitive to a number of factors, such as shape and particle size, and can also be changed as in the surrounding media of different nature. 19,37
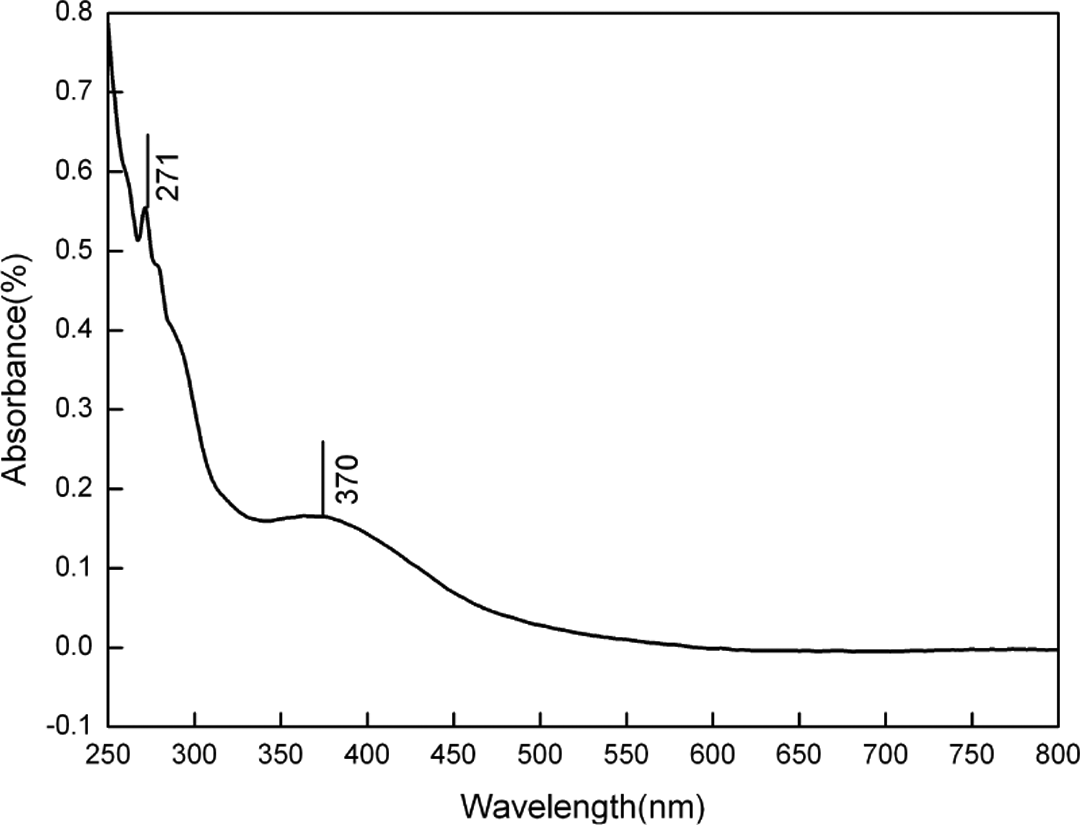
UV–Vis spectra of Ag/PANI nanocomposites dispersed in ethanol solution. UV-Vis: ultraviolet–visible; Ag: silver; PANI: polyaniline.
FTIR study
Figure 3 shows FTIR spectra of the pure PANI powders and Ag/PANI nanocomposites in 400 to 4000 cm−1 region. The FTIR measurement of Ag/PANI represents peaks at 1592 and 1500 cm−1, which can be assigned to quinoid and benzenoid unit stretching modes. The peak at 1290 cm−1 is due to the C–N stretch from benzenoid unit stretching mode of PANI. The band at 1143 cm−1 can be assigned to the quinoid unit of doped PANI, confirms the identity of the polymer, and is in agreement with published articles in literature. 38 The strong band that appears at 1384 cm−1 is assigned to nitrate ions. 39 The peak at 821 cm− 1 is attributable to C–H bending vibration out of the plane of the paradisubstituted benzene rings. The broad absorption band above 3451 cm−1 is assigned to N–H stretching vibrations. The FTIR spectra of Ag/PANI nanocomposites are similar to the pure PANI, which confirms the formation of PANI in nanocomposites. 40 However, the quinoid peaks in Figure 3(b, curve b) are found to be shifted by 30 cm−1 compared to Figure 3(b, curve a; from 1592 cm−1 to 1562 cm−1). The shift toward lower wave number indicated a decrease in the conjugation length as a result of the formation of H-bonding between the –OH group of both the amine and imine group of PANI, which suggested positive interactions between the Ag molecules and the PANI chains according to earlier studies. 41
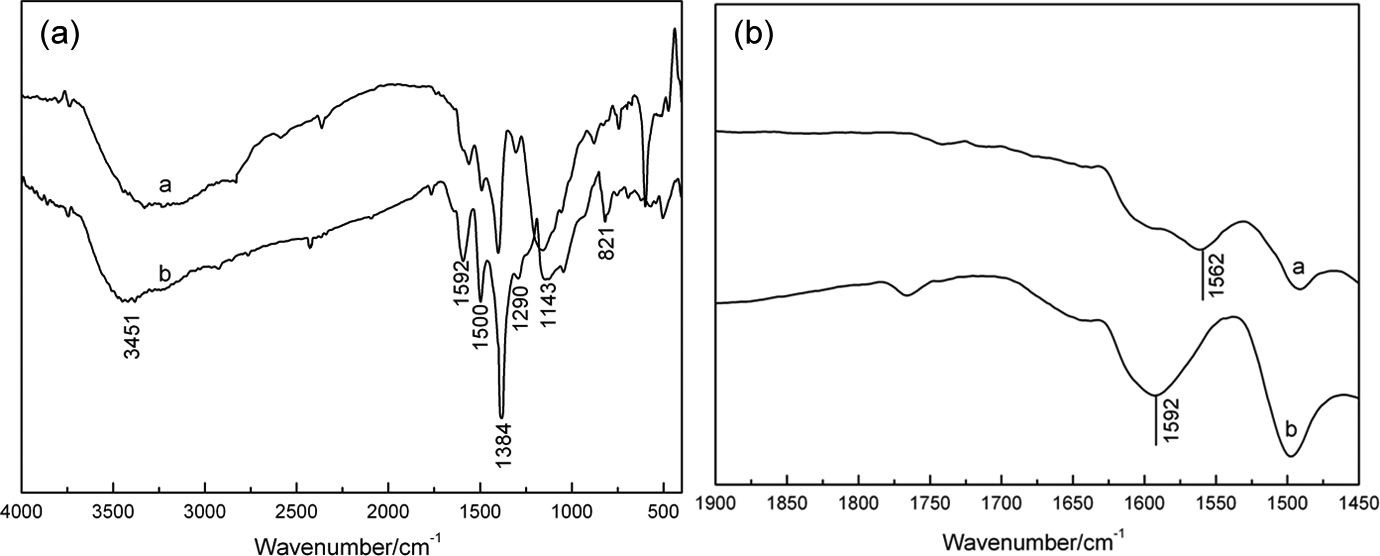
FTIR spectra of (a′) pure PANI and (b′) Ag/PANI nanocomposites. (a) 4000–400 cm−1 and (b) 1900–1450 cm−1. FTIR: Fourier transform infrared; Ag: silver; PANI: polyaniline.
Morphological study
FESEM was carried out to display the morphology of the Ag/PANI nanocomposites. A clear sea-island structure indicating the phase-separated morphology of the Ag/PANI nanocomposites is shown in Figure 4. The dispersed phase (island) is aggregates of Ag nanoparticles and the continuous phase (sea) is the PANI phase. To determine whether these aggregates of Ag nanoparticles detected were “real” Ag nanoparticles, EDS was analyzed, demonstrating EDS spectra from the framed region in Figure 5(a). The EDS spectra with significant peaks at 2.64 keV and 3.0 keV were observed in Figure 5(b) indicating that Ag atoms in the framed region really existed and definitely detected. Ultrasound radiation of high frequency is able to reduce Ag ions and polymerize aniline monomer simultaneously in earlier reports. 34 The mechanism to explain the formation of silver nanoparticles and aniline polymerization takes into account radical species that are generated when ultrasound wave interacts with an aqueous solution. Thus, silver ions are reduced and PANI is polymerized at the same time via ultrasonic irradiation. The reduced Ag ions grow up into Ag clusters and finally form aggregates in the PANI matrix. The chemical reactivity of Ag atom clusters is larger than bulk Ag. 42 Based on the analysis above, we consider that there is the physical–chemical interaction between the aggregates of Ag nanoparticles and PANI matrix. As shown in Figure 5(a), the aggregates of Ag nanoparticles are clearly composed of individual Ag nanoparticle. Every nanoparticle with a size of 48 ± 1 nm, which is excellently consistent with the statistic calculation by XRD, appears as a discrete entity in the aggregates. The aggregates of Ag nanoparticles have diameter of about 420 nm (see Figure 5(a)) and are uniformly distributed in the PANI matrix, which may enhance the electrochemical activity of Ag/PANI nanocomposites.

FESEM images of Ag/PANI nanocomposites with different magnification. (a) ×30,000 and (b) ×100,000. FESEM: field emission scanning electron microscopy; Ag: silver; PANI: polyaniline.
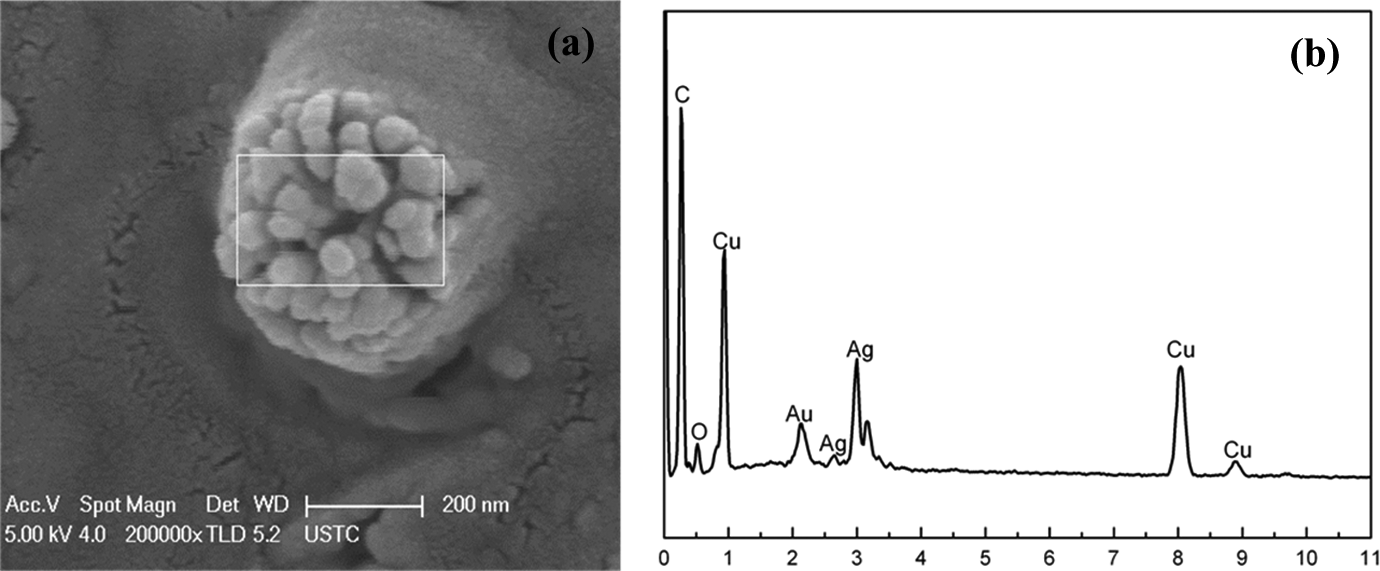
FESEM image of Ag/PANI nanocomposites and EDS spectra of the selected areas in the framed region of (a). (a) FESEM image and (b) EDS spectra. FESEM: field emission scanning electron microscopy; EDS: energy dispersive spectrometry; Ag: silver; PANI: polyaniline.
Electrocatalytic oxidation and determination of l -Tyr using sea-island structured Ag/PANI nanocomposites
Cyclic voltammograms were recorded for
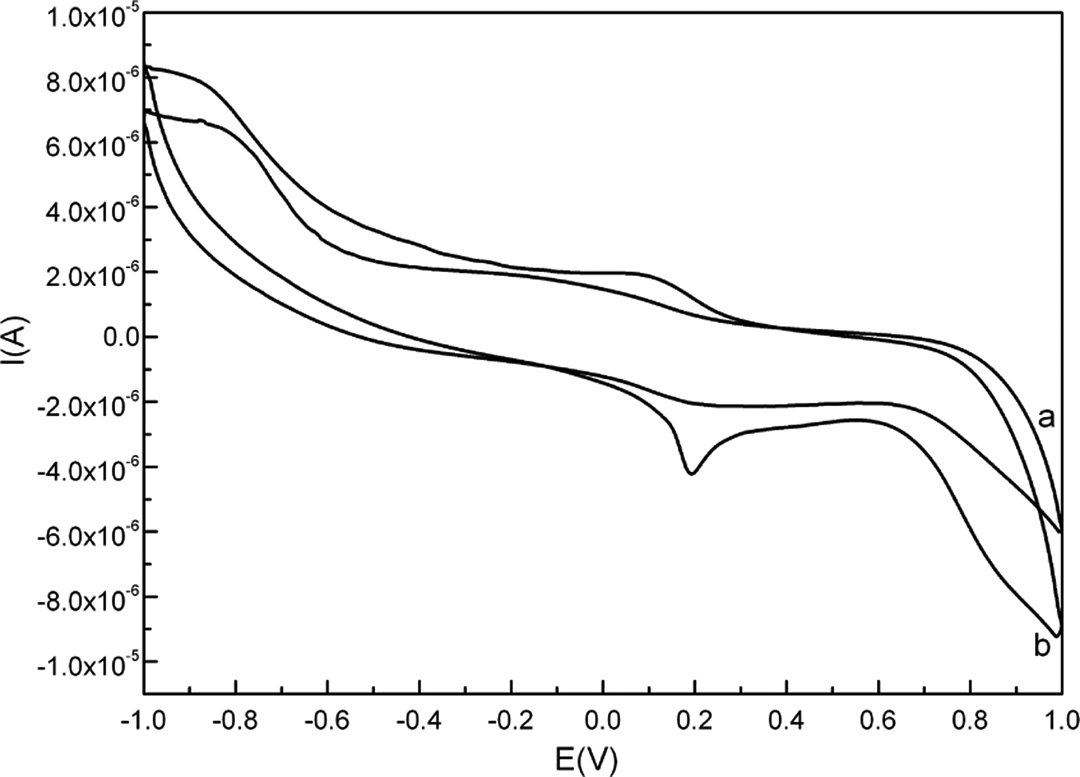
Cyclic voltammograms of the Ag/PANI/GCE in the absence (a) and presence (b) of 50 μM of
Figure 7 shows that upon increasing
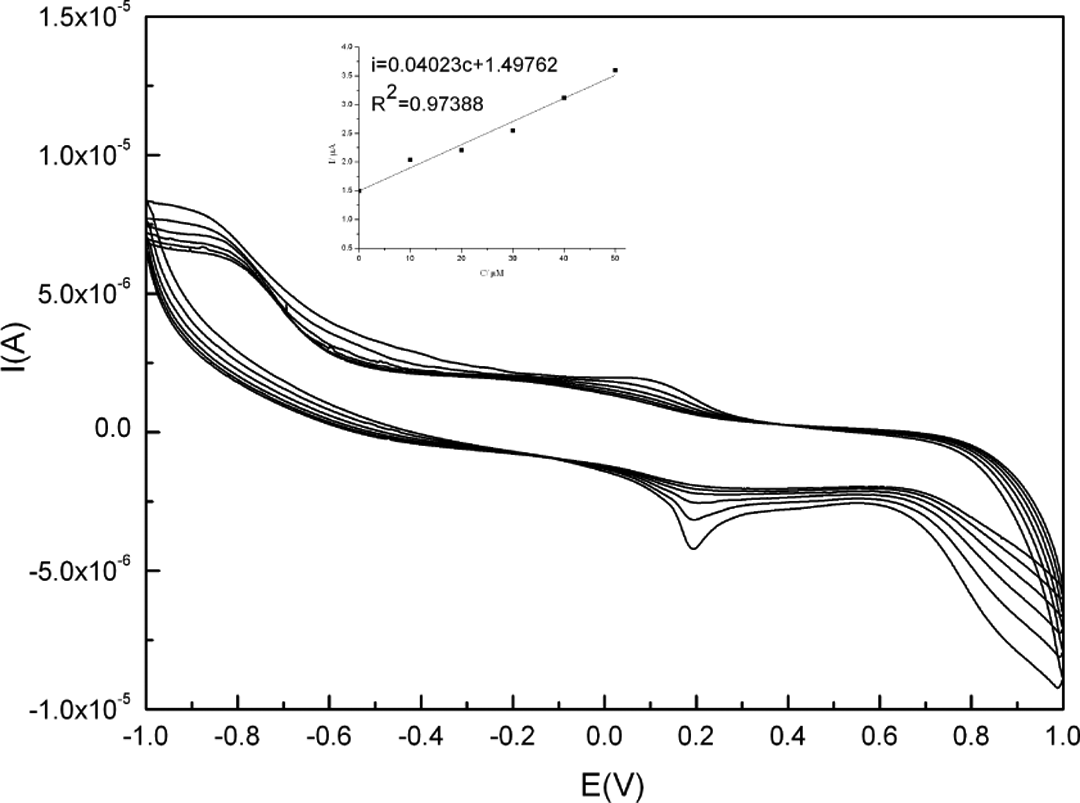
Cyclic voltammograms of Ag/PANI/GCE in the presence of different
It is also observed that in the presence of
Figure 8 shows the differential pulse voltammograms recorded for
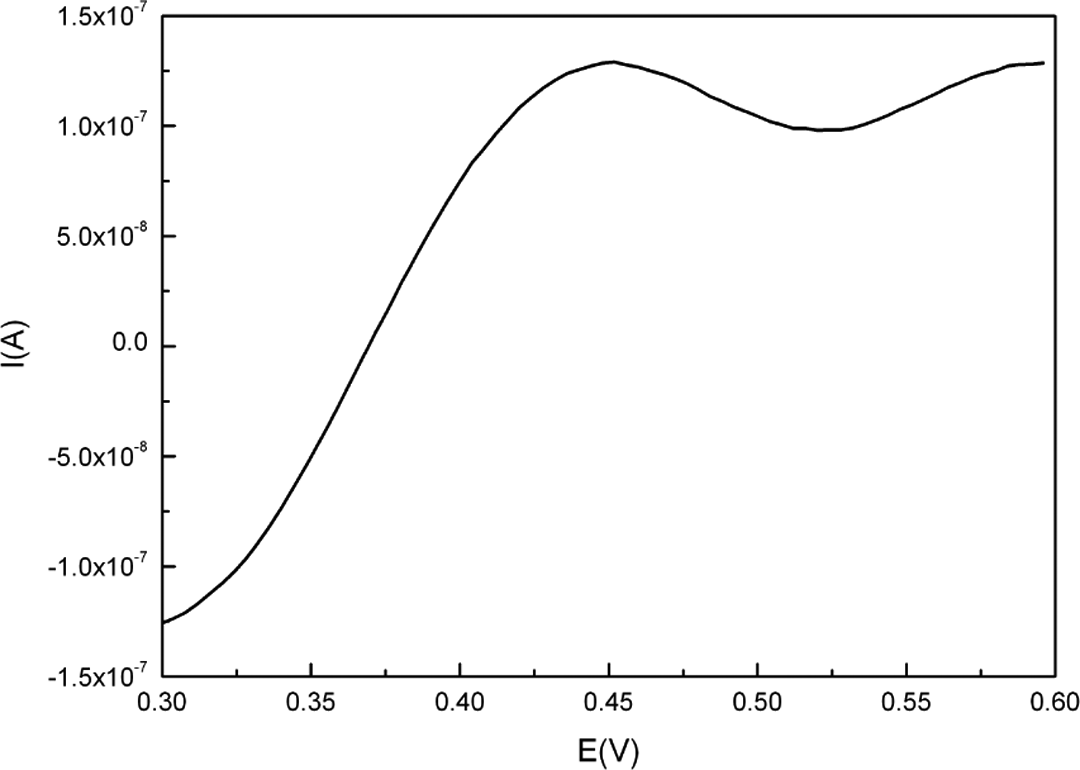
Differential pulse voltammograms of 0.01 μM
Conclusions
In summary, we have synthesized sea-island structured Ag/PANI nanocomposites via ultrasonic technique. The dispersed phase (island) is aggregates of Ag nanoparticles and the continuous phase (sea) is the PANI phase. These nanostructures exhibit a remarkable electrocatalytic activity for
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Nature Science Foundation of China (No. 51303005), the Educational Commission of Anhui Province of China (nos KJ2013A087 and KJ2013A095) and the Doctor Foundation of the Anhui University of Science and Technology.
