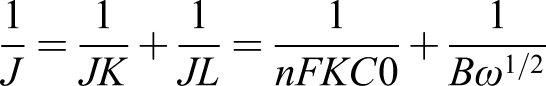Abstract
Dinuclear metallophthalocyanines Fe2Pc2(CP)4 containing carboxyl substitutes were wrapped with amino-functionalized carbon nanotubes (MWCNTs-NH2) to enhance electrocatalytic activity for oxygen reduction reaction (ORR) using a facile “in situ” amidation reaction. The morphological characteristics and chemical environment of the Fe2Pc2(CP)4/MWCNTs-NH2 composites were characterized by scanning electron microscope (SEM), X-ray diffraction, Ultraviolet–visible (UV-Vis), Fourier Transform infrared (FTIR), and X-ray photoelectron spectroscopy. The electrocatalytic activity of ORR was tested and analyzed by cyclic voltammetry and linear sweep voltammetry. The results showed that the π–π interactions between the Fe2Pc2(CP)4 and MWCNTs-NH2 dramatically enhanced the π electron density in the conjugated structure, and oxygen could be reduced much more easily. Moreover, the oxygen reduction reactions mainly proceeded a one-step four electron process for Fe2Pc2(CP)4/MWCNTs-NH2 catalysts. The dispersion and electrocatalytic performance of M2Pc2Rn had be enhanced after being loaded on functionalized carbon nanotubes.
Introduction
In recent years, with the exhausting of traditional fossil energy and the aggravation of environmental pollution, the development and utilization of new energy sources are urgent. Direct methanol fuel cells (DMFCs) have become the research focus because of their simple structure, high energy density, low cost and other merits.1–3 Nevertheless, the energy efficiency of DMFCs systems is greatly affected by the slow kinetics of the oxygen reduction reaction (ORR) as similar to the other air-based ones. 4 Platinum-based materials contribute significantly in decreasing the overpotential of oxygen reduction reaction because of the high catalytic activity. However, platinum-based materials are limited in large-scale applications due to their scarce reserves, lack of excellent methanol resistance, and high price.5,6 In that case, non-platinum catalysts with high catalytic activity and methanol-tolerant performance are needed.
Researchers have carried out many experiments to explore new non-platinum catalysts. The electrocatalytical activity of PtTe2 is similar to that of Pt/C catalysts, and the cost of PtTe2 is significantly lower than the Pt/C catalysts. 6 Specially, many reports have pointed out that the ability to carry oxygen molecules of the conjugated macrocyclic compounds plays a key role in the ORR process. 7 Conjugated compounds with a high π electron density can be oxidized and reduced much more easily, because of the delocalization and weakly binding properties of the π electron cloud.8,9 Transition metal-N4-chelates like metalloporphyrin (MPs) and metallophthalocyanine (MPcs) have been investigated as electrocatalysts for ORR.10–12 MPc molecules have an 18 π electrons conjugated structure, which is similar to naturally occurring MPs, are widely used as biomimetic catalysts for oxygenase. 13 Compared with MPs, the benzene rings of metallophthalocyanines can be modified by different substituents. It is possible to synthesize different functional compounds such as monomeric phthalocyanines, dimer phthalocyanines, and polymerized phthalocyanines.14,15 Noteworthily, many reports have pointed out that the catalytic activity of MPcs for oxygen reduction reaction is strongly affected by a strong ligand–field effects and the formation of adduct MPc−O2-. 16 The properties of MPc−O2- adducts are related to the radius, the valence, and the d filling of the metal central ions. Considering π electron density of the strong ligand–field effects, metallophthalocyanines have excellent catalytic activity for ORR.17,18
However, MPcs materials are limited in large-scale applications because the aggregation of MPcs nanoparticles decreased the stability of electrocatalysts. Reports have also demonstrated that carbon-based materials are conducive to improve the dispersion and stability of metallophthalocyanines catalysts.19–23 An electrocatalysts of iron phthalocyanine (FePc) supported on graphene oxide (GO) prevented the aggregation of FePc, demonstrating a good electrochemical performance in ORR process. Moreover, MPcs loaded on carbon-based materials are good for the electron transfer to improve the catalytic activity. 19 Carbon nanotubes (CNTs) have been widely used in various fields due to their excellent physical and chemical properties, such as an excellent electrical conductivity and a large specific surface areas, establishing itself as a good candidate to provide a pathway for preventing the aggregation of nanoparticles and accelerating the electron transfer. 24 Carbon nanotubes are mainly prepared by chemical vapor deposition and arc discharge.25–28 Chemical modifications further improve the bonding strength between carbon nanotubes and polymers to expand the application range of carbon nanotubes. Amino functionalization for the carbon nanotubes is one of the most effective modification methods to further optimize the performance because amino substituents have high reaction activity with many compounds. 29 In this work, Dinuclear metallophthalocyanines Fe2Pc2(CP)4 containing carboxyl substitutes were wrapped with amino-functionalized carbon nanotubes (MWCNTs-NH2) to enhance electrocatalytic activity for ORR using a facile “in situ” amidation reaction. The electrocatalytic activity and stability for oxygen reduction reaction were tested and analyzed by a cyclic voltammetry (CV) and linear sweep voltammetry (LSV).
Experimental part
Materials
4-nitrophthalonitrile (99%) and 4,4-bis(4-hydroxyphenyl)pentanoic acid (98%) were purchased from Aladdin. The other reagents were of analytical grade and were used without further purification. The M2Pc2(CP)4 compounds were synthesized by the template reaction of a bisphthalonitrile and the metal salts in the dimethylaminoethanol (DMAE). This was done according to the literature. 30 Amino-functionalized carbon nanotubes (MWCNTs-NH2) were prepared by amino modification of carbon nanotubes (MWCNTs COOH) in the ethylenediamine (EDA).31,32
[2’,10’,16’,24’-{Tetrakis-4,4’-bis(4-(3-cyano-4-isocyanophenoxy)phenyl)pentanoate diphthalocyaninato) diiron (II)] preparation Fe2(II)Pc2(CP)4 (0.2250 g, yield 39.12%). olive green solid, m.p.>300°C. UV-Vis (DMF) λmax/nm: B band: 327; Q band: 556, 654. IR (KBr)νmax/cm−1: 1635 (νC=N); 944 (νM-N); 1711 cm−1 (νC=O); 1400, 1224, 797 (νPc). Anal. Cald. for C32H16N8Fe: C, 72.17; H, 4.22; N, 9.43; Found: C, 71.59; H, 4.37; N, 9.37. 1H NMR (300 MHz,DMSO)δ 10.21 (s, 4H, COOH), 8.09–7.90 (m, 18H, Ar-H),7.70–7.30 (m,9H,Ar-H), 7.02–6.91 (m, 9H, Ar-H).
Characterization
JEM-2010 electron microscope was performed on a SEM analysis at 200 kV voltage. X-ray diffraction was measured by Rigaku D/max-2400 Diffractometer (Tokyo, Japan), equipped with a Cu Kα radiation source with an X-ray wavelength (λ) of 1.5406Å. The IR spectra are recorded on a Germany Bruker Vertex70 spectrometer. The UV−VIS spectrophotometer was recorded on a UV/visible spectrophotometer (UV-1600, China) using a quartz cell at room temperature. Thermogravimetric Analysis (TGA) was recorded on a TGA/DSC Star System (Mettler Toledo) in N2 with a heating rate of 10°C/min. X-ray photoelectron spectroscopy (XPS) was analyzed by Axis Ultra spectrometer and Al (Mono) Kα X-ray source (1486.6 eV). Electrocatalytic performance were tested by electrochemical workstation CHI 660E (Shanghai Chenhua Company) and a Pine Instrumennt Company AF-MSRCE modulator rate rotator in a 0.1 KOH, separately.
Synthesis
Preparation of Fe2Pc2(CP)4/MWCNTs-NH2
The Fe2Pc2(CP)4 was synthesized by the template method in the DMAE using a bisbenzonitrile(1.02 mmol), metal salt (0.52 mmol) and 4 mL DMAE were poured into a Teflon-lined autoclave at 160°C for 4 h. The reaction mixture was then poured into methanol to produce a precipitate, and the precipitate was washed sequentially with acetic acid, water, and methanol. The crude product was then dissolved in DMF and reprecipitated by gradually adding methanol to the solution. The precipitate was washed again, as in the previous method, followed by centrifugation, and dried at 100°C in an oven.
Fe2Pc2(CP)4/MWCNTs-NH2 composites were synthesized by a facile “in situ” amidation reaction, using the MWCNTs-NH2 and Fe2Pc2(CP)4 as the precursors (Figure 1). In brief, 0.0400 g (0.020 mmol) of Fe2Pc2(CP)4 powder was dispersed in 10 mL of dimethylformamide solution, followed by the addition of 0.0340 g (0.164 mmol) Dicyclohexylcarbodiimide (DCC). The resulting solution was reacted for 48 h under room temperature. 10 mg MWCNTs-NH2 and 0.0200 g (0.164 mmol) 4-Dimethylpyridine (DMAP, a super nucleophilic acylation catalyst) were then poured into the mixture gradually, and was stirred for 48 h under room temperature. All of the crude products were washed with ethanol, then centrifuged and drying overnight in an oven at 60°C. Formation of M2Pc2(CP)4/MWCNTs-NH2 composites.
Evaluation of the electrocatalytic activity
The electrochemical properties of Fe2Pc2(CP)4/MWCNTs-NH2 composites were investigated by CV and rotating disk electrode (RDE) in an O2-saturated 0.1
Results and discussion
Characterization of Fe2Pc2(CP)4/MWCNTs-NH2 composites
The surface morphology of M2Pc2(CP)4/MWCNTs-NH2 composites was investigated by SEM images. As shown in Figure 2, numerous dark particles of Fe2Pc2(CP)4, as indicated by white arrows, were observed on the MWCNTs-NH2 nanotubes from the SEM images of Fe2Pc2(CP)4/MWCNTs-NH2 composites. The results showed that the Fe2Pc2(CP)4 nanoparticles were uniformly dispersed on the surface of MWCNTs-NH2. Fe2Pc2(CP)4 nanoparticles with a diameter of about 500 nm were coated on the surface of the MWCNTs-NH2 by π-π interactions and amide bond after “amidation reaction”. SEM images of MWCNTs-NH2 and Fe2Pc2(CP)4/MWCNTs-NH2.
The UV-VIS spectra of Fe2Pc2(CP)4/MWCNTs-NH2 composites is shown in Figure 3(a). N,N-dimethylformamide (DMF) solution was used as the solvent for the test. There are two characteristic absorption bands for Fe2Pc2(CP)4, the Q band and the B band. The Q band is located at 654 nm, which belongs to the electronic transition of 2a1u (LUMO)→6eg (HOMO); The B-band is located in the wavelength range of 327 nm, which belongs to the electronic transition of 4a2u→6eg. The absorption at 556 nm is mainly due to the agglomeration of Fe2Pc2(CP)4.
33
The spectrum of the MWCNTs-NH2 composites did not exhibit any obvious absorption peaks from 300 to 900 nm. Compared with Fe2Pc2(CP)4/MWCNTs-NH2 composite, a new absorption peak appeared at 683 nm, which indicated that Fe2Pc2(CP)4 was successfully loaded on the surface of MWCNTs-NH2. It was found that the absorption peaks of Fe2Pc2(CP)4/MWCNTs-NH2 composites are red shifted from 654 nm to 683 nm, and the absorption peak at 556 nm disappeared. The result indicates that a strong p - π interaction between Fe2Pc2(CP)4 and MWCNTs-NH2, which makes Fe2Pc2(CP)4 nanoparticles uniformly dispersed on MWCNTs-NH2. The strong p - π interaction promoted the electron transfer for the oxygen reduction reaction. Structure and composite of Fe2Pc2(CP)4/MWCNTs-NH2; (a)UV-vis spectra; (b) Thermogravimetric analysis; (c), (d) FTIR spectra.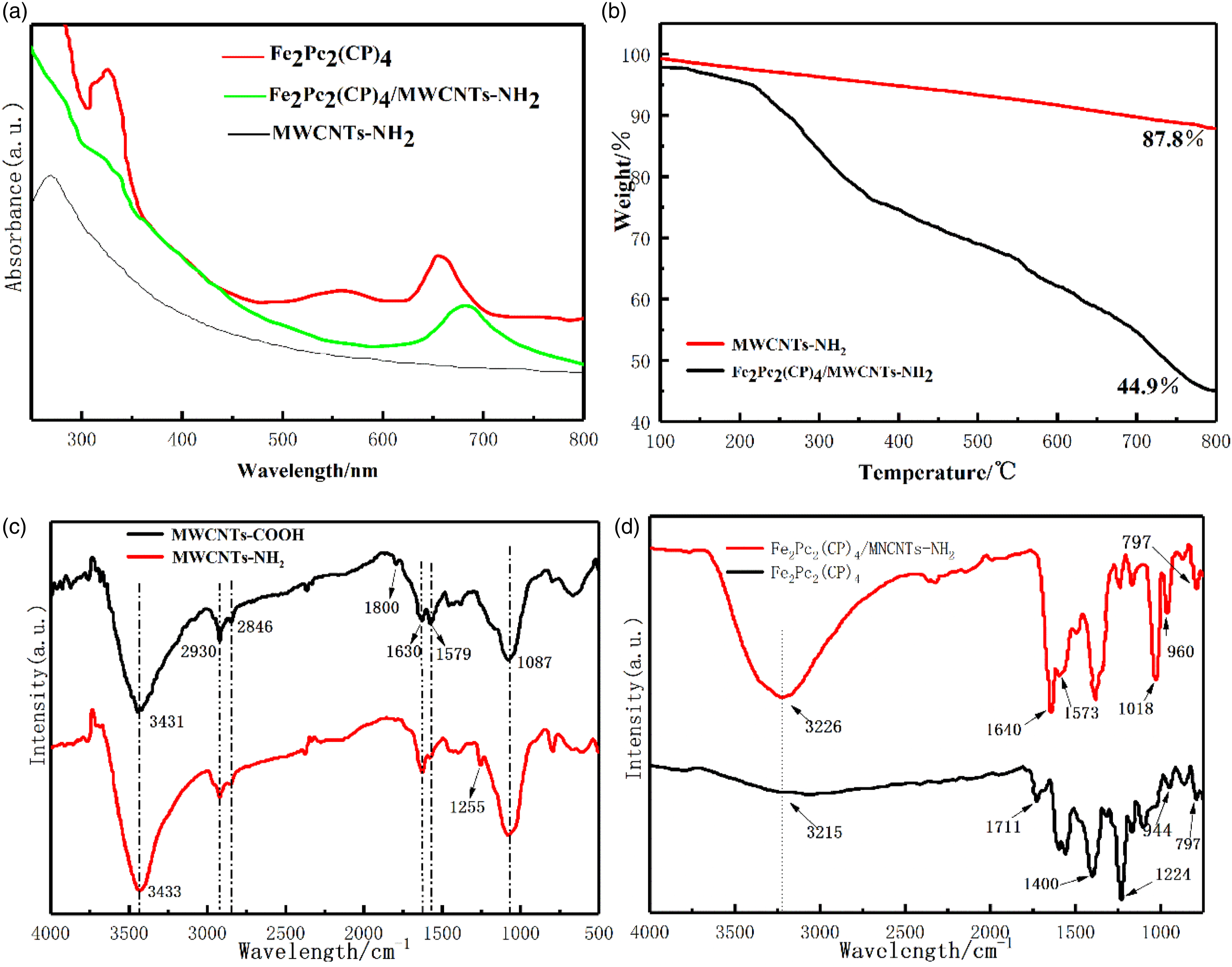
The FTIR spectra of the MWCNTs-NH2, Fe2Pc2(CP)4, Fe2Pc2(CP)4/MWCNTs-NH2 samples are shown in Figure 3(c) and (d). As can be seen from Figure 3(c), the vibration peaks at 1630 cm−1 attributed to the stretching vibrations of C=O for MWCNTs-COOH. Specially, the intensity of the absorption peak at 1579 cm−1 decreased significantly and the characteristic absorption peak at 1255 of amide appeared for MWCNTs-NH2. 8 The absorption peaks at 1255 cm−1 and 799 cm−1 assigned to the C-N and N-H stretching vibrations, respectively. The peaks at 2930 cm−1 and 2846 cm−1 assigned to the vibration of -CH2. 11 The results indicated that MWCNTs-NH2 particles were prepared by amino modification of MWCNTs-COOH with ethylenediamine. In Figure 3(d), the strong absorption bands at 797 cm−1 and 960 cm−1 attributed to the vibration of Pc ring and the Fe-N bond of Fe2Pc2(CP)4/MWCNTs-NH2. 33 Compared with MWCNTs-NH2, the C= O absorption peak at 1711 cm−1 is effectively eliminated, and the peak of H2N-C=O was observed at 1640 cm−1. The results showed that Fe2Pc2(CP)4/MWCNTs-NH2 catalysts were successfully prepared.
TGA analysis was investigated to further explore the Fe2Pc2(CP)4 loading amount on the MWCNTs-NH2. As shown in Figure 3(b). The thermogravimetric process was performed on an inert atmosphere at the rate of 10°C/min. The weight loss of MWCNTs-NH2 powder reached 12.2% in the 100–800°C range. However, the Fe2Pc2(CP)4/MWCNTs-NH2 samples exhibited up to one step of mass losses of 55.1% from 250°C to 800°C range. The results indicated that the weight loss of Fe2Pc2(CP)4/MWCNTs-NH2 composites was obviously higher 42.9% than MWCNTs-NH2 powder. Considering Fe2Pc2(CP)4/MWCNTs-NH2 composites, the mass losses mainly attributed to the decomposition of Fe2Pc2(CP)4.
As shown in Figure 4, the composition and chemical state of Fe2Pc2(CP)4/MWCNTs-NH2 were studied by XPS. Figure 4(a) displays the XPS survey spectra of Fe2Pc2(CP)4, MWCNTs-NH2 and Fe2Pc2(CP)4/MWCNTs-NH2 catalysts. The results indicated that Fe2Pc2(CP)4/MWCNTs-NH2 composites are composed of O, C, Fe and N, which showed that Fe2Pc2(CP)4 molecules were successfully supported on the surface of carbon nanotubes. The C\O content rate (13.51) of Fe2Pc2(CP)4/MWCNTs-NH2 composites are significantly lower than that of MWCNTs-NH2(35.87), which further indicated that Fe2Pc2(CP)4 was successfully supported. The high-resolution N1s spectra is shown in Figure 4(c) and (d). The two asymmetric broad peaks at 398.5ev and 400.2ev corresponded to the signals of C-N and C=N of pyrrole ring in phthalocyanine macrocycle Fe2Pc2(CP)4.
34
Moreover, the new peak located at 400.4 eV in Fe2Pc2(CP)4/MWCNTs-NH2 composites, which is higher than that in pure Fe2Pc2(CP)4, corresponded to the signals of H2N-C=O in the Fe2Pc2(CP)4/MWCNTs-NH2 composites. The results further confirmed the amide bonds between Fe2Pc2(CP)4 and MWCNTs-NH2. XPS spectra of Fe2Pc2(CP)4/MWCNTs-NH2; (a) XPS survey spectra Fe2Pc2(CP)4, MWCNTs-NH2, Fe2Pc2(CP)4/MWCNTs-NH2; (b), (c), (d)High resolution of Fe2p and N1s XPS spectra of Fe2Pc2(CP)4 and Fe2Pc2(CP)4/MWCNTs-NH2. XPS: X-ray photoelectron spectroscopy.
The high resolution Fe2p spectra were shown in Figure 4(b). The peaks of Fe2p for Fe2Pc2(EP)4/MWCNTs-NH2 are located at 712.9 eV and at 727.3.8 eV, which corresponded to the Fe2p1/2 and Fe2p3/2 signals in the bivalent oxidation state, respectively. It can be found that the binding energy of Fe2Pc2(EP)4/MWCNTs-NH2 composites are higher than that of pure Fe2Pc2(CP)4. The results further indicated that there is a p-π conjugate interaction of HN-C=O between Fe2Pc2(CP)4 and MWCNTs-NH2.
Electrocatalytic activity of Fe2Pc2(CP)4/MWCNTs-NH2
The electrocatalytic activity of Fe2Pc2(CP)4/MWCNTs-NH2 composites for ORR was first tested by the technique of CV. The results were shown in Figure 5. The reduction peaks for Fe2Pc2(CP)4/MWCNTs-NH2 composites at around −0.20 V in N2- and O2-saturated solutions are reversible and overlaps with the signal of O2 molecules. The Fe2Pc2(CP)4/MWCNTs-NH2 composite has a well interaction with O2 because of the redox activity of the M2+ cores. The π electron conjugated structure of the MN4-type phthalocyanine macrocycle system has a significant effect on the redox activity of the M2+ cores. Furthermore, the Fe(II)/Fe(III) reduction peak current of O2 at positive potential (−0.20 V vs. SCE ) is close to that of Pt/C (- 0.18 V vs. SCE). The redox transition of Fe(II)/Fe(III), which occurs at a lower positive potentials, provide relatively low overpotential for ORR.
35
Cyclic voltammograms curve of Fe2Pc2(CP)4/MWCNTs-NH2 and Pt/C catalysts on glassy carbon electrodes.
Linear sweep voltammetry) measurement was further performed on a rotating disk electrode (RDE) to test the catalytic performance of the catalysts. The electrodes modified with Pt/C, Fe2Pc2(CP)4 and Fe2Pc2(CP)4/MWCNTs-NH2 were performed in an O2-saturated 0.1
Furthermore, the electrocatalytic performance of one-step four-electron process is better than that of two electron process for an oxygen reduction reaction. The total electron transfers number (n) in the ORR reaction is calculated by the Koutecky-Levich (K-L) equation, listed below
As shown in Figure 6, the K-L points (J−1 vs ω−1/2) of Fe2Pc2(CP)4/MWCNTs-NH2 at different voltages had a good linearity, and the slope is consistent. The linearity of the K-L diagram and the approximate parallelism of the fitting lines indicated that the first-order reaction kinetics of the concentration of dissolved oxygen. The electron transfer number n is similar in the redox process at different potentials. The calculated value of n for Fe2Pc2(CP)4/MWCNTs-NH2 was 3.76 at the voltage range from - 0.7 V to 0.4 V. The results showed that Fe2Pc2(CP)4/MWCNTs-NH2 and Pt-C catalysts mainly carried out redox reaction through one-step four electron process. Firstly, the central metal ion Fe(II)(d filling of t2g6 eg0) of Fe(II)2Pc2(CP)4 macrocycle system bonded to the axial ligand O2 molecule through bridge adsorption.16,17 The electrons on π electron conjugated structure Pc2(CP)4 migrate to central metal ion Fe(II) and molecule O2 to form an adduct Fe(III)2Pc2(EP)4·O2-. The iron (III) - oxygen iron (III) peroxide is obtained. The breaking of O–O linkage for Fe(III)-O-O-Fe(III) peroxide promotes the formation of OH- ions. The electrons on MWCNTs-NH2 then migrate to Fe(III)2Pc2(CP)4, and Fe(III)2Pc2(CP)4 was reduced to Fe(II)2Pc2(CP)4.19,32,34 Moreover, the similar number of transfer electron number n calculated for Fe2Pc2(CP)4/MWCNTs-NH2 and Pt/C at - 0.7 V versus SCE suggests that Fe2Pc2(CP)4/MWCNTs-NH2 composites is a great substitute for Pt/C catalysts. Electrocatalytic activities of Pt/C, Fe2Pc2(CP)4 and Fe2Pc2(CP)4/MWCNTs-NH2 at different rotation rate in O2-saturated 0.1 
The CV curves for Fe2Pc2(CP)4/MWCNTs-NH2 and Pt-C catalyst were further studied to reveal the cross effect in O2-saturated 3 Cyclic voltammetry curves of Fe2Pc2(CP)4/MWCNTs-NH2 in O2-saturated 0.1 Mechanism for oxygen reduction reaction catalyzed by Fe2Pc2(CP)4/MWCNTs-NH2.

Conclusions
Fe2Pc2(CP)4/MWCNTs-NH2 composites were synthesized by a facile “in situ” amidation reaction and the electrocatalytic activity was analyzed by CV and LSV techniques. The results showed that Fe2Pc2(CP)4) complexes were successfully wrapped with the amino-functionalized carbon nanotubes (MWCNTs-NH2) by π-π interactions and amide bond. Amino-functionalized carbon nanotubes enhanced the dispersivity of Fe2Pc2(CP)4) complexes. The π–π interactions between the Fe2Pc2(CP)4 and MWCNTs-NH2 dramatically enhanced the π electron density in the conjugated structure, and oxygen could be reduced much more easily. The electrocatalytic activity of Fe2Pc2(CP)4/MWCNTs-NH2 were enhanced by the HN-C=O bonding interaction between Fe2Pc2(CP)4 nanoparticles and MWCNTs-NH2. It was concluded that carbon-based materials is an effective way to increase the catalytic avtivity of MPc-based materials for oxygen reduction reaction. The results suggests that Fe2Pc2(CP)4/MWCNTs-NH2 composites is a great substitute for Pt/C catalysts.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Natural Science Foundation of Shaanxi Province (No. 2019JM-527), the National Natural Science Foundation of China (61604120).