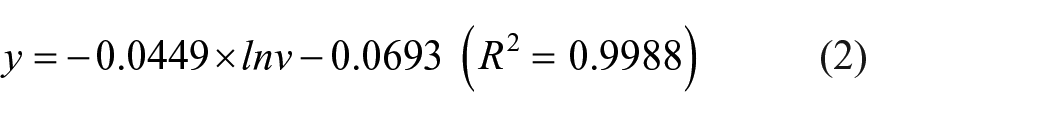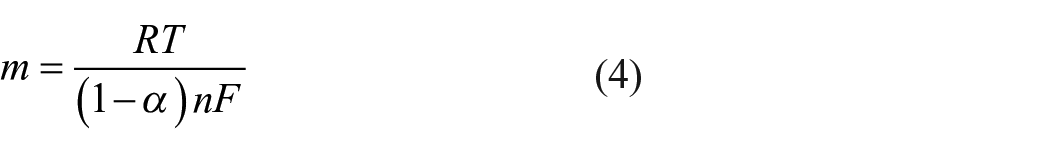Abstract
Polyaniline nanowires are directly synthesized on a glassy carbon electrode (3 mm diameter) by an electrochemical process. The polyaniline nanowires, of uniform size, a diameter of 85–95 nm, and high conductivity, distribute evenly throughout the surface of the working electrode. Electrochemical measurements are conducted in order to determine the electrochemically effective surface area of the obtained glassy carbon electrode modified with polyaniline nanowires, and an investigation of the electrocatalytic activity of polyaniline nanowires for the oxidation of methanol under alkaline conditions is carried out. The electrochemically effective surface area of the glassy carbon electrode modified with polyaniline nanowires is nearly 27 times larger than that of a glassy carbon electrode. In a cyclic voltammetry curve of the glassy carbon electrode modified with polyaniline nanowires measured in a 3.0 M CH3OH and 0.5 M KOH solution, an anodic peak corresponding to the oxidation of methanol under alkaline conditions appears at 0.17 V with a peak current of 34.4 μA.
Keywords

Introduction
Nanostructured conducting polymers, especially nanostructures of polyaniline (PANi), are important due to their ability to combine the properties of low-dimensional organic conductors with high surface areas.1–3 PANi exists in various oxidation states ranging from the most reduced leucoemeraldine form, through the half-oxidized emeraldine base form, to the fully oxidized pernigraniline form (Figure 1), so it is a unique and interesting polymeric material. 4 It is used in diverse applications such as sensors, nano-electronic devices, electron-field emitters, data storage devices, actuators, membranes, rechargeable batteries, supercapacitors, and catalysts.5–8
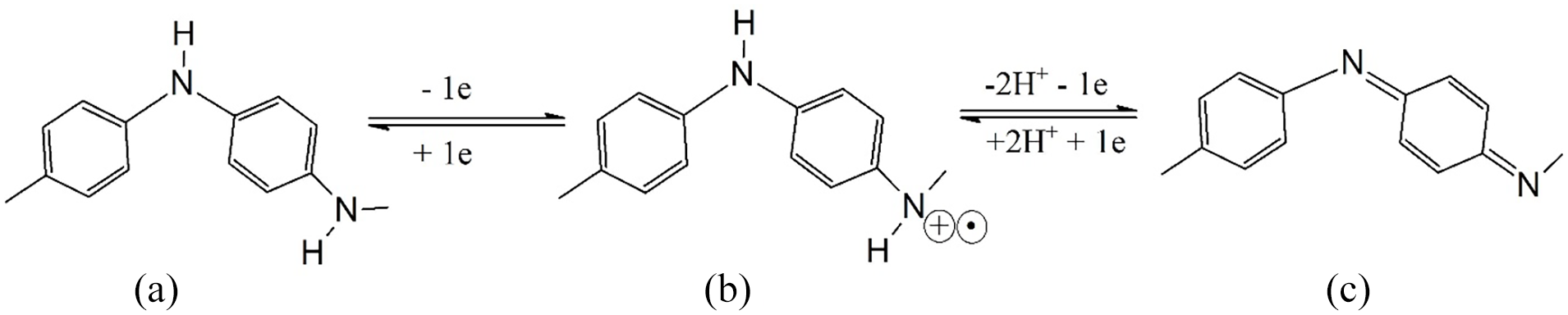
The three oxidation states of PANi: (a) the most reduced leucoemeraldine form; (b) the half-oxidized emeraldine base form; and (c) the fully oxidized pernigraniline form.
Direct methanol fuel cells (DMFCs) are essential alternative power sources for portable and small-sized electronic devices. Composites based on platinum nanoparticles (PtNPs) are the most active electrocatalysts for the methanol electro-oxidation reaction in DMFCs.9–12 However, their high cost, limited availability, and poor durability hinder their commercial applications.13,14 Therefore, carrying out studies to explore effective electrocatalysts in order to replace Pt-based electrocatalysts is necessary.
PANi and PANi-based nanocomposites are considered as potential alternatives to Pt-based catalysts due to their low cost, high surface areas, high electrical conductivities, excellent chemical and physical stabilities, unique redox properties, ease of preparation, and strong adhesion with electrodes.9,15 Therefore, in this work, PANi nanowires (NWs) have been directly electrosynthesized on a glassy carbon electrode (GCE). In addition, the electrochemically effective surface area of the GCE/PANi NWs electrode has been determined, and the electrocatalytic activity of the PANi NWs for the oxidation of methanol under alkaline conditions has been investigated.
Results and discussion
Characterization of the PANi NWs
The chronoamperometry (CA) method was used to electropolymerize directly PANi NWs on the GCE electrode, and the corresponding CA curve is shown in Figure 2(a). When the time for electropolymerization increases from 0 to 550 s, the current increases due to the formation of a conductive polymer layer on the GCE electrode. The cyclic voltammetry (CV) scans measured at a scan rate of 25 mV s−1 in a 0.1 M HCl solution using the GCE and GCE/PANi NWs electrodes are shown in Figure 2(b). The CV curve of the GCE electrode (Figure 2(b), curve a) has no redox peak, and the corresponding current is very small due to the absence of materials on the electrode surface. On the contrary, the CV curve of the GCE/PANi NWs electrode (Figure 2(b), curve b) has two pairs of redox peaks that are characteristic of the PANi material. The first pair of redox peaks (b1 and b1′), occurring between 0 and 0.25 V, are related with the conversion of the fully reduced leucoemeraldine base to the partially oxidized emeraldine. 16 The second pair of redox peaks (b2 and b2′), appearing between 0.6 and 0.8 V, are attributed to the conversion of the emeraldine into the fully oxidized pernigraniline form. 16 The CV scans measured at a scan rate of 25 mV s−1 in a K3Fe(CN)6/K4Fe(CN)6 (0.005 M) and 0.1 M KCl solution using the GCE and GCE/PANi NWs electrodes are shown in Figure 2(c). The peak current of the GCE/PANi NWs electrode (Figure 2(c), curve b) is higher than that of the GCE electrode (Figure 2(c), curve a). The use of PANi NWs with high conductivity to modify the GCE electrode led to an increase in the peak current of the GCE/PANi NWs electrode. The results in Figure 2 prove that PANi NWs are formed directly on the GCE electrode by using the CA method.

(a) Chronoamperometric curve during electropolymerization of PANi NWs on the GCE electrode in a 0.1 M aniline and 0.5 M H2SO4 solution over 550 s. (b) CV scans conducted at a scan rate of 25 mV s−1 in a 0.1 M HCl solution using: (a) GCE and (b) GCE/PANi NWs electrodes. (c) CV scans conducted at a scan rate of 25 mV s−1 in a K3Fe(CN)6/K4Fe(CN)6 (0.005 M) and 0.1 M KCl solution using: (a) GCE and (b) GCE/PANi NWs electrodes.
A scanning electron microscopy (SEM) image of PANi NWs electrosynthesized on the GCE electrode is shown in Figure 3. The PANi NWs with a uniform size and a diameter of 85–95 nm distribute evenly over the surface of the working electrode (WE). The obtained PANi NWs are homogeneous and less agglomerated. The homogeneous distribution of the PANi NWs on the electrode surface is expected to enhance the electrochemically effective surface area of the GCE/PANi NWs electrode.

SEM image of PANi NWs electrosynthesized on the GCE electrode.
Electrochemically effective surface area of the GCE/PANi NWs electrode
The CV curves of the GCE/PANi NWs electrode were measured in a K3Fe(CN)6/K4Fe(CN)6 (0.005 M) and 0.1 M KCl solution at different potential scan rates from 15 to 40 mV s–1 in order to determine the electrochemically effective surface area (Figure 4(a)).

(a) CV scans of the GCE/PANi NWs electrode recorded in a K3Fe(CN)6/K4Fe(CN)6 (0.005 M) and 0.1 M KCl solution, at potential scan rates of 15, 20, 25, 30, 35, and 40 mV s–1; (b) the relationships between the anodic and cathodic peak potentials and lnv, and their corresponding linear fit lines: Epa, the green line with square symbols, and Epc, the red line with star symbols; and (c) the relationship between the cathodic peak current (Ipc) and the square root of the scan rate (v1/2), and its corresponding linear fit line.
On the basis of the results in Figure 4(a), the relationships between the anodic and cathodic peak potentials (Epa and Epc) and the natural logarithm of the potential scan rate (lnν) were determined and are shown in Figure 4(b), that is, Epa versus lnν
and Epc versus lnν
In addition, the relationships between Epa and Epc and lnν are represented by the following equations17,18
where E0 (V) is the formal standard potential, D (cm2 s–1) is the diffusion coefficient, k0 (cm s–1) is the electron transfer rate constant, α is the charge transfer coefficient, n is the number of transported electrons, R is the gas constant (R = 8.314 J mol–1 K–1), T is the working temperature (T = 298 K), and F is Faraday’s constant (F = 96,480 C mol–1).
The values of n and α are calculated from the slopes of Epa versus lnν and Epc versus lnν, and as a result, the value of n is 0.944 and that of α is 0.303. The value of n proves that the oxidation of Fe(CN)64– and the reduction of Fe(CN)63– happening on the GCE/PANi NWs electrode surface are through monoelectronic steps. Moreover, as can be seen in Figure 4(b), when the potential scan rate ν increases, the peak potential difference ΔE (ΔE = Epa – Epc) increases. This result confirms that the electrochemical processes occurring on the GCE/PANi NWs electrode surface are quasi-reversible. 17
On the other hand, on the basis of the CV results in Figure 4(a), the relationship between the cathodic peak current (Ipc) and the square root of the potential scan rate (v1/2) is plotted in Figure 4(c):
Figure 4(c) shows that the dependence of Ipc on v1/2 is linear. Besides, the electrochemically effective surface area of the GCE/PANi NWs electrode (A (cm2)) is determined by using the Randles–Sevcik equation for quasi-reversible reactions as follows 19
where ν (V s–1), α, n, and D (cm2 s–1) are mentioned above in equations (3)–(6), C0 (mol cm–3) is the concentration of the redox species, and Ipc (A) is the cathodic peak current. The D value of Fe(CN)63– in 0.1 M KCl medium at 25 °C is 7.20 × 10–6 cm2 s–1. 20 On the basis of equation (8) and the slope of Ipc versus v1/2 (Figure 4(c)), the A value of the GCE/PANi NWs electrode is calculated to be 1.913 cm2. Thus, the electrochemically effective surface area of the GCE/PANi NWs electrode is nearly 27 times larger than that of the GCE electrode (0.071 cm2). This result could be an important factor leading to the electrocatalytic activity of the PANi NWs for the oxidation of methanol under alkaline conditions.
Electrocatalytic activity of the PANi NWs for the oxidation of methanol under alkaline conditions
In order to investigate the possibility of using the electrosynthesized PANi NWs material as an electrocatalyst for the oxidation of methanol under alkaline conditions, the CV curves of the GCE and GCE/PANi NWs electrodes measured in a 3.0 M CH3OH and 0.5 M KOH solution were recorded (Figure 5). In the case of the GCE electrode (Figure 5, curve a), there is no redox peak. On the contrary, in the case of the GCE/PANi NWs electrode (Figure 5, curve b), the anodic peak corresponding to the oxidation of methanol under alkaline conditions appears at 0.17 V.

CV scans conducted at a scan rate of 50 mV s−1 in a 3.0 M CH3OH and 0.5 M KOH solution using: (a) GCE and (b) GCE/PANi NWs electrodes.
Figure 6 shows the CV scans of the GCE/PANi NWs electrodes measured in solutions containing only 0.5 M KOH (Figure 6, curve a) and containing 0.5 M KOH and 3.0 M CH3OH (Figure 6, curve b). In the presence of CH3OH (Figure 6, curve b), the electro-oxidation of methanol in an alkaline electrolyte background occurs on the GCE/PANi NWs electrode surface, and the corresponding anodic peak current is 34.4 μA. The methanol electro-oxidation mechanism is shown as the reaction (9)9,21

CV scans of the GCE/PANi NWs electrodes conducted at a scan rate of 50 mV s−1 in: (a) 0.5 M KOH and (b) 3.0 M CH3OH and 0.5 M KOH solutions.
The results in Figures 5 and 6 indicate that the electrosynthesized PANi NWs material has electrocatalytic activity for the oxidation of methanol under alkaline conditions. The nanowire structure with large electrochemically effective surface area is an important factor leading to the electrocatalytic activity of the fabricated PANi. On the contrary, in other studies in the literature, the PANi film, without the nanowire structure, exhibits no activity towards the oxidation of methanol in alkaline media.22,23
Conclusion
PANi NWs have been directly electrosynthesized on a GCE electrode by using the CA method. The electrochemically effective surface area of the obtained GCE/PANi NWs electrode is nearly 27 times larger than that of the GCE electrode. The electrochemical results initially showed that PANi NWs are electrocatalytically active for the oxidation of methanol under alkaline conditions. However, this study needs to be further developed in order to evaluate the stability of the PANi NWs electrocatalyst.
Experimental
Chemicals
Aniline (C6H5NH2, 99.5 wt%), potassium hexacyanoferrate(III) (K3Fe(CN)6, 99 wt%), potassium hexacyanoferrate(II) trihydrate (K4Fe(CN)6·3H2O, 99.95 wt%), potassium chloride (KCl, 99 wt%), and methanol (CH3OH, 99.8 (v/v)%) were purchased from Sigma Aldrich. The supporting chemicals including hydrochloric acid (HCl, 37 wt%), sulfuric acid (H2SO4, 98 wt%), and potassium hydroxide (KOH, 85 wt%) were of analytical grade.
Instrumentation
The SEM images of the PANi NWs were investigated by using a Nova NanoSEM 450 microscope. Electropolymerization of PANi NWs and electrochemical measurements were carried out by using a PGSTAT302N AutoLab electrochemical workstation.
Electrosynthesis of the PANi NWs
A three-electrode system, consisting of the GCE electrode (3 mm diameter) as a WE, a Pt electrode as a counter electrode (CE), and an Ag/AgCl electrode in 3 M KCl solution as a reference electrode (RE), was immersed in an electrolyte solution containing 0.1 M aniline and 0.5 M H2SO4. CA with an applied voltage of 0.9 V versus Ag/AgCl RE was used over 550 s to electropolymerize the PANi NWs on the GCE WE. Next, the GCE/PANi NWs electrode was gently washed with deionized water and dried at room temperature.
The electrochemically effective surface area of the GCE/PANi NWs electrode and the electrocatalytic activity of the PANi NWs for the oxidation of methanol under alkaline conditions
Electrochemical measurements were conducted by using a three-electrode system consisting of the fabricated GCE/PANi NWs electrode as a WE, the Pt electrode as a CE, and the Ag/AgCl electrode in 3 M KCl solution as a RE. The CV curves of the GCE/PANi NWs WE in a K3Fe(CN)6/K4Fe(CN)6 (0.005 M) and 0.1 M KCl solution at potential scan rates of 15, 20, 25, 30, 35, and 40 mV s–1 were recorded to calculate the electrochemically effective surface area of the GCE/PANi NWs WE. The CV curves of the GCE and GCE/PANi NWs WEs in a 3.0 M CH3OH and 0.5 M KOH solution at a scan rate of 50 mV s−1, and the CV curves of the GCE/PANi NWs WEs at a scan rate of 50 mV s−1 in solutions containing (i) only 0.5 M KOH and (ii) 0.5 M KOH and 3.0 M CH3OH were recorded to investigate the electrocatalytic activity of the PANi NWs for the oxidation of methanol under alkaline conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 104.03-2019.19.