Abstract
Photonic crystals were fabricated using self-assembled polystyrene (PS) submicrospheres with different content of nanospheres with different glass transition temperatures (T g: 100 to −54°C) and their opalescent and mechanical properties were investigated. PS spheres (185.4 nm) and nanospheres were prepared by soap-free emulsion polymerization. Nanospheres were prepared using one or two types of monomers, styrene, n-butyl methacrylate, and butyl acrylate with sodium p-styrenesulfonate. Surface morphology, particle size (D n), and wavelength (λ) of photonic crystals were determined from scanning electron microscopy (SEM), effective refractive index equation, and modified Bragg’s law, respectively. The reflection wavelength (λ max) was measured from ultraviolet-visible spectroscopy. SEM results showed that hard nanospheres were distributed around PS spheres in their photonic crystals. On the other hand, soft nanospheres were coated onto PS spheres in their photonic crystals. With increasing weight fraction of hard or soft nanospheres in the photonic crystals, Dn and λ were increased. The value of λ max corresponded to that of λ. The mechanical property of the photonic crystals was measured by the pencil hardness (PH) test. The result showed that by decreasing the T g and increasing the weight fraction of nanospheres, PH was increased. The opal film prepared from PS spheres with 20% N-54 nanospheres had the highest PH (H).
Introduction
Photonic bandgap (PBG) materials have emerged as promising materials during recent decades because of their unique light reflection properties. These materials are periodicity constructions of two kinds of materials with different refractive indexes or permittivity. 1,2 Therefore, the propagation of light waves can be strongly confined and controlled within certain wavelength ranges because of the specific architectural construction of the material, which allows periodicity in the refractive index and thereby gives rise to the PBGs. 1 –4 Based on these features, extensive development is underway for many prospective applications of optical equipment 5 –7 and electronic products 8,9 such as smart windows, chemical sensors, optical devices, and memory devices.
In general, there are two ways to classify the numerous approaches for three-dimensional construction of PBG materials: top-down and bottom-up methods. 10–11 The top-down approaches are often carried out using traditional microfabrication techniques, such as photolithography and etching, 12 –15 to produce periodic structures with the desired shape from bulk materials. The bottom-up approach generally exploits the characteristic of molecular species or colloidal spheres of self-assembly into one-, two-, and three-dimensional periodic structures. 16–17 The most commonly used strategy for large-scale production of PBG materials involves a relatively simple and cost-efficient procedure of assembling colloidal spheres by self-assembly. 18–19
Colloidal crystals are typically ordered arrays of highly monodispersed oxides or polymer spheres because these species can readily be synthesized with the desired diameter. 20 –22 Because of Van der Waals force, the colloidal spheres have been fabricated by themselves as face-centered cubic (FCC) structures with the lowest free energy. 23,24 The sol-gel process is typically used to synthesize oxide materials such as silicon dioxide, titanium dioxide, and zinc oxide, whereas the conventional polymer spheres of polystyrene (PS), poly(methyl methacrylate), and their cross-linked products are generally synthesized by soap-free emulsion polymerization (or using surfactants). 25 –28 However, such colloidal spheres are always “hard” beads. Consequently, using these spheres to construct photonic crystal films result in less satisfactory mechanical properties.
Some interesting approaches for the construction of PBG films with high toughness have been developed in order to resolve these issues. 29 –35 These strategies can be categorized into three methods. The first method uses various means, such as construction of colloidal spheres with embedded periodicity into polymer materials to form PBG film and achieve enhanced linkage among the colloidal spheres. 36 For example, Fortes’s group embedded submicrospheres PS into elastic poly(imide) substrate to enhance the film-forming properties of photonic crystal construction. 37 The second method achieves enhanced linkage among the colloidal spheres by various means, such as coating the polymer PBG with a thin silica layer and then sintering the PBG film at high temperature, 38 –40 or removing the colloidal spheres from the polymer substrate. 41 –43 The third method involves direct arrangement of modified narrow-size or core–shell spheres in the PBG film. For example, Leyrer’s group synthesized various modified spheres with different functional groups on their surfaces and subsequently assembled them to generate covalent or physical cross-linking among the PBG films. In this way, a 10-fold enhancement of the chemo mechanical stability of the PBG films was achieved. 33
Successful construction of tough PBG films has also been reported using rigid-core/soft-shell spheres such as polystyrene/poly(butylmethacrylate), polystyrene/poly(methyl methacrylate-acrylic acid), and polystyrene/poly(N-isopropylacrylamide) 18,29,44 by arrangement of the rigid cores into periodic lattices, while the soft shell provides the binding force between the surfaces of the spheres. In the third approach, prepolymers or monomers that are subsequently cured or polymerized are introduced directly into the voids of the colloidal crystal to replace the air voids with elastic polymer instead of simply enhancing the binding force between the contact areas of the spheres. Thus, the binding area is increased. For example, Finlayson’s group used the low glass transition temperature (Tg ) of the photonic crystal shell layer and prepared artificial photonic crystal fibers by a hot extrusion method, which were successfully used in contexture. 45
The first and second methods involved complex and expensive synthesis processes; the third method required the reaction monomers’ ratio, time, and temperature to be controlled during the process when the core–shell colloids were fabricated. The core–shell spheres are then used to construct opal films with improved film properties. The hard cores function to generate the periodic structure of the opal film, while the soft shells act as a binder between the interfaces of the spheres. In this study, PS submicrospheres and nanospheres with various T gs were mixed to prepare PBG materials. Introducing hard nanospheres could reveal the morphology, and soft nanospheres could improve the mechanical properties without sacrificing the PBG property contributed by the PS submicrospheres. The results of this study are expected to provide a simple approach for the synthesis of PBG films with improved film properties.
Experimental
Materials
The reagent-grade monomers, styrene (St) and butyl acrylate (BA), were purchased from SHOWA (Showa Chemical Co., Ltd., Tokyo, Japan). n-Butyl methacrylate (BMA) was purchased from Acros (Belgium). Methacrylic acid (MAA) and sodium p-styrenesulfonate (NaSS) were purchased from TCI (Japan). Tetraethoxysilane was purchased from Merck (Germany). The reagent-grade initiator, potassium persulfate (KPS), was purchased from SHOWA.
Preparation of PS sphere
Monodisperse PS sphere was synthesized via soap-free emulsion polymerization of St at the boiling point. Initially, 85.4 g of deionized water was mixed with 9.6 g St and 490 µl MAA in a 250 ml three-necked flask equipped with a reflux condenser. The reaction mixture was mechanically stirred at 480 r/min, and the reaction temperature was then increased to reach the boiling point. After 15 min, 0.0876 g of KPS dissolved in 5 g of deionized water was added to the reaction mixture. The polymerization reaction was terminated after 2 h.
The particle diameter was estimated from scanning electron microscopy (SEM) observation of more than 100 particles to ensure accuracy. The monodispersity of the spheres was determined on the basis of the coefficient of variation (C v), which can be derived as follows:
where the terms D n, Di , ni , D v, and σ represent the number-averaged diameter, the particle diameter, amount of particles, volume-averaged diameter, and standard deviation, respectively.
Preparation of nanospheres with different T g
Based on the Fox equation, monodisperse nanospheres with different T gs were designed by varying the feeding ratio of St, BMA, and BA. The formulations are shown in Table 1. Monodisperse nanospheres were synthesized via soap-free emulsion polymerization of St, BMA, and BA at boiling point. Initially, 85 g of deionized water was mixed with 10 g total monomer, unique or mixture with various contents, with various ratios NaSS in a 250 ml three-necked flask equipped with a reflux condenser. The reaction mixture was mechanically stirred at 560 r/min, and the reaction temperature was then increased to reach the boiling point. After 15 min, 0.0876 g of KPS dissolved in 5 g of deionized water was added to the reaction mixture. The polymerization reaction was terminated after 2 h.
The reaction conditions and results of nanospheres.
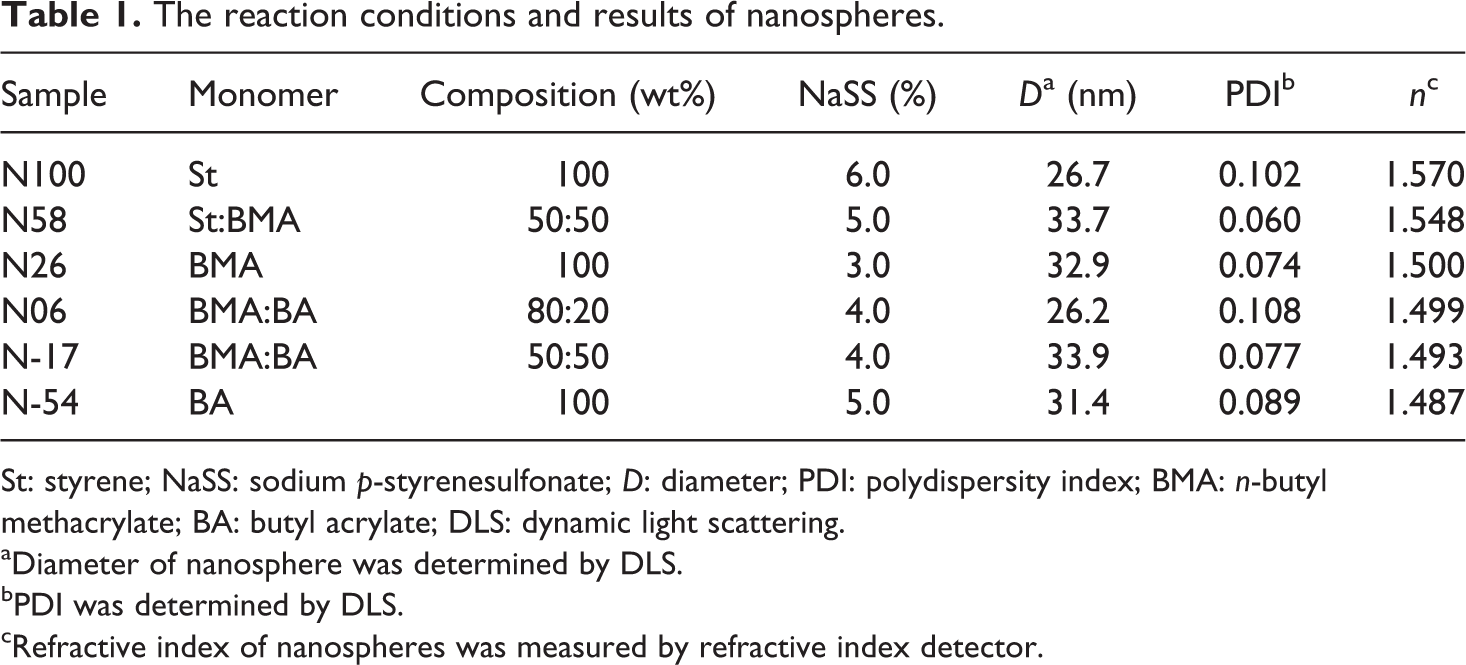
St: styrene; NaSS: sodium p-styrenesulfonate; D: diameter; PDI: polydispersity index; BMA: n-butyl methacrylate; BA: butyl acrylate; DLS: dynamic light scattering.
aDiameter of nanosphere was determined by DLS.
bPDI was determined by DLS.
cRefractive index of nanospheres was measured by refractive index detector.
Preparation of photonic crystals with soft nanospheres
PS sphere-suspensions with equal solid content (10 wt%) were blended with the required amount of nanosphere, and magnetically stirred for 5 min. This dispersion was ultrasonically treated for 2 min, and 400 μl of suspended particles was subsequently spread onto a glass plate and dried at 50°C for 2 h. The water of latex was evaporated, during which the spheres in the latex were self-assembled into the ordered lattice.
Results and discussion
Characteristics of monodisperse nanospheres
Monodisperse nanospheres were fabricated by soap-free emulsion polymerization in a boiling state. In order to prepare nanospheres with different T g, one or two kinds of monomers, St, BMA, and BA, were mixed with NaSS in a boiling state and polymerized. Preparation conditions and characterization of six kinds of nanospheres are shown in Table 1.
The T g of nanospheres was from 100°C to −54°C. The nanospheres were named according to their T g values. For example, the nanosphere with a T g value of 100°C was named as N100. NaSS, a hydrophilic monomer, was added to increase the number of nucleation sites and reduce the size of the nanospheres. As shown in Table 1, the diameters of the nanospheres were reduced to approximately 30 nm (26.2–33.9 nm) by adding a suitable amount of NaSS. The polydispersity indexes of these six nanospheres are close to or less than 0.1 and the polydispersions are narrow, which means that the sphere sizes are highly uniform.
However, when the refractive indexes of nanospheres were measured by a refractive index detector, the refractive indexes of N26 poly(butyl methacrylate) and N54 poly(butyl acrylate) were smaller than that of N100 (PS). Therefore, the refractive index was decreased by increasing the BMA or BA content. N100 with the highest refractive index (1.57) was prepared with 100% St composition. N-54 with the lowest refractive index (1.487) was prepared with 100% BA composition.
Characteristics of photonic crystal construction with PS spheres and N100 (PS/N100)
Photonic crystal construction with optical characteristics showed specific color due to reflecting light of a specific wavelength, and the optical characteristics could be shown by Modified Bragg’s law. In general, photonic crystals were fabricated by PS spheres with an FCC structure, so the modified Bragg’s law can be represented as equation (5).
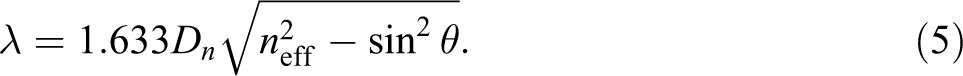
The value of n eff can be calculated by the effective refractivity formula using equation (6), where f ps is the PS volume fraction, n ps is the refractive index of PS, f air is the air volume fraction, and n air is the refractive index of air. The value of n eff could be set by adjusting the hard or soft nanosphere content.
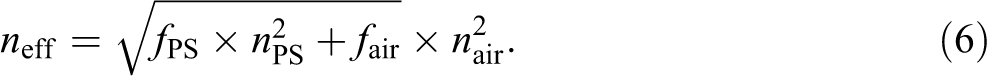
In this study, the photonic crystal was made by casting a blended polymer suspension onto glass substrate at 50°C for 2 h. The blended polymer suspension consisted of two categories of spheres distinguished by sphere diameter. The latex of submicrosphere PS, (Dn = 185.4 nm) called PS spheres for short, were blended with nanospheres (Dn = 30 nm) to produce a blended polymer suspension. These nanospheres surrounded the PS spheres during the evaporation process. Thus, the monodisperse PS spheres are tightly packed after self-assembling, and the voids between the polymer spheres are filled with nanospheres forming a three-dimensional polymer–polymer ordered structure.
The architectural arrangement of the PS/N100 films prepared from PS spheres with various N100 contents were observed using SEM, and the images are shown in Figure 1. Highly ordered, closely packed lattices with perfect FCC arrays are observed in each of the samples. The PS spheres are hexagonally arranged with the N100 nanospheres occupying the interstices. These observations demonstrate that the PS spheres can self-assemble into three-dimensional periodic structures with the aid of N100 nanospheres. The SEM images also revealed that the surface morphologies of the PBG films were significantly altered with different N100 nanosphere content. In the case of the PBG film constructed using PS spheres with 1.0 wt% of N100 [Figure 1(c)], PS spheres and N100 nanospheres were clearly observed. The diameter of the PS spheres (185.4 nm) was the same as that of the PS spheres assembled by themselves without adding N100 (Figure 1(a)). The high T g of N100 nanospheres could be easily observed in photonic crystal construction by SEM. In addition, in the case of the film with a low N100 nanosphere content, the N100 nanospheres could not fully surround the PS spheres, so PS spheres were not fully shielded by N100 nanospheres.

SEM of PS submicrospheres prepared with various contents of N100: (a) 0 wt%, (b) 0.5 wt%, (c) 1.0 wt%, (d) 5.0 wt%, (e) 10 wt%, and (f) 20 wt%. SEM: scanning electron microscopy; PS: polystyrene.
When the appropriate N100 nanosphere content (more than 10 wt%; Figure 1(f)) was present, the PS spheres were precisely surrounded by N100 nanospheres, giving rise to ordered structures. Therefore, with the progressive increase of the weight fraction of N100 nanospheres from 1.0 wt% to 20 wt%, the number of N100 nanospheres increased and the diameter of PS spheres decreased from 185.4 nm (Figure 1(c)) to 146.1 nm (Figure 1(f)) because of being shielded by the N100 nanospheres. However, by increasing the amount of N100 nanospheres from 1.0 wt% to 20 wt%, the PS spheres center-to-center distance (D) increases from 185.4 nm to 192.8 nm to make more interspace for the N100 nanospheres. The value of D corresponds to the value of Dn in equation (5). Conversely, equation (6) can be modified as equation (7), and n (air + nanosphere) can be calculated by equation (8). Then λ is obtained by equation (5), as shown in Table 2.
The characteristics of photonic crystals of PS spheres with different N100 nanospheres contents.
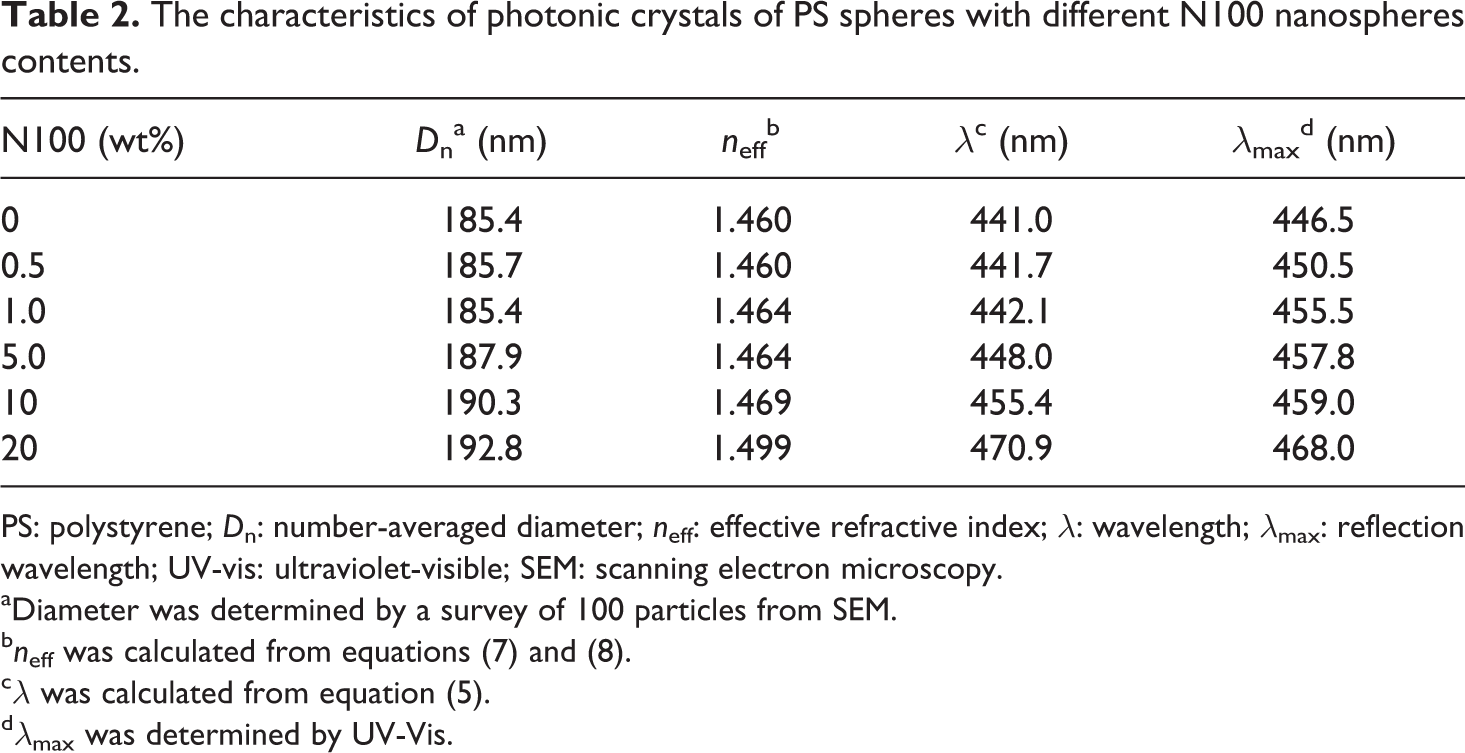
PS: polystyrene; D n: number-averaged diameter; n eff: effective refractive index; λ: wavelength; λ max: reflection wavelength; UV-vis: ultraviolet-visible; SEM: scanning electron microscopy.
aDiameter was determined by a survey of 100 particles from SEM.
b n eff was calculated from equations (7) and (8).
c λ was calculated from equation (5).
d λ max was determined by UV-Vis.
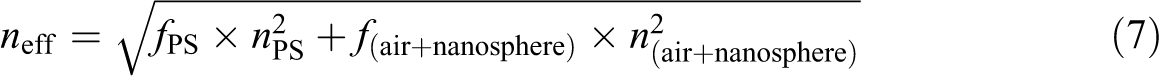
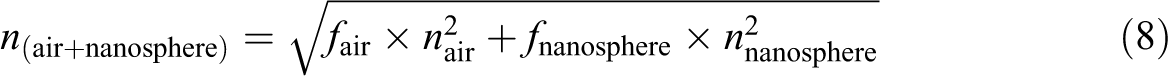
The reflection spectra of a series of films prepared with PS/N100 spheres at 50°C is shown in Figure 2. All of the films exhibited brilliant colors from violet to blue (λ max = 446.5–468.0 nm), resulting from the diffraction of light inside the periodic structure comprising the core–shell spheres. The sharpness of the reflection peaks indicated the high quality of the opal film. The λ max detected by ultraviolet-visible (UV-Vis) spectroscopy and the λ calculated by equation (5) had the same tendency, which was increased as N100 content increased, as shown in Figure 3. The same results were also observed in the PS/N58 and PS/N26 series.
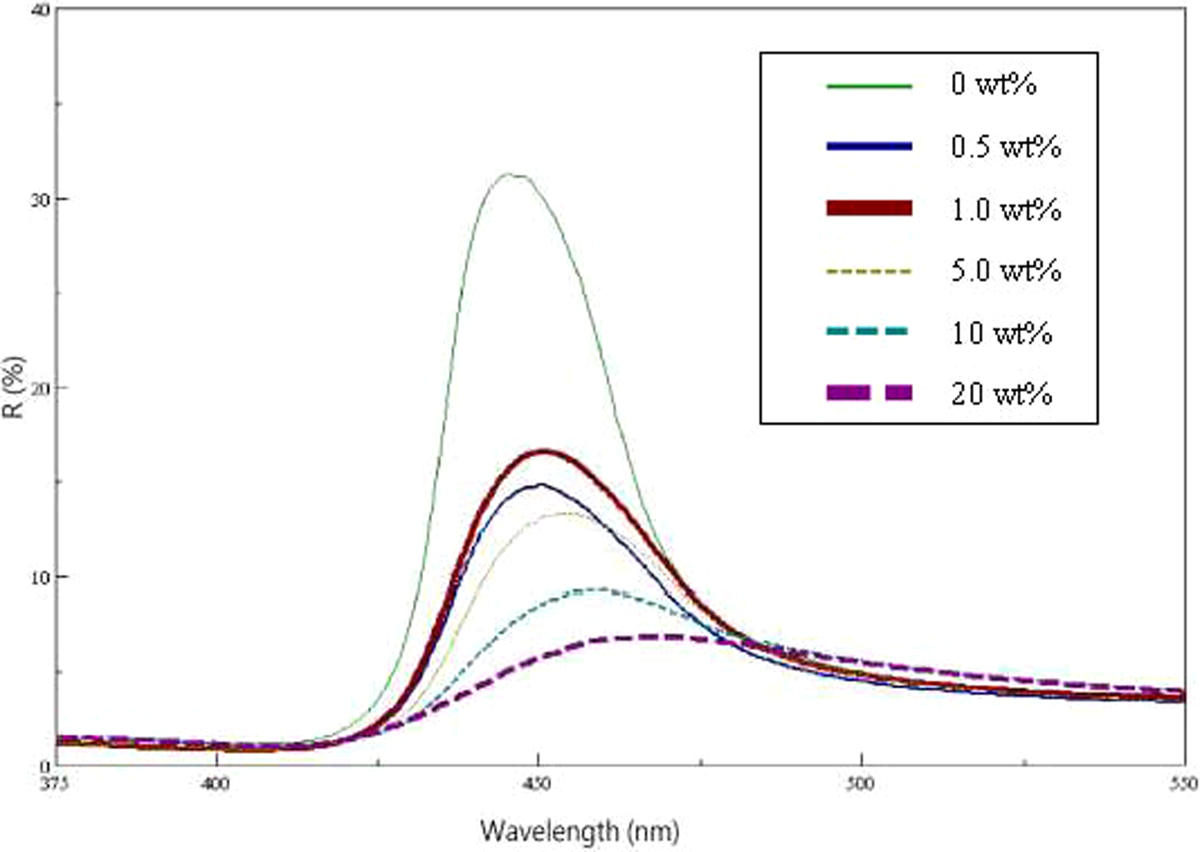
UV-Vis spectra of PS spheres with various contents of N100. UV-Vis: ultraviolet-visible; PS: polystyrene.
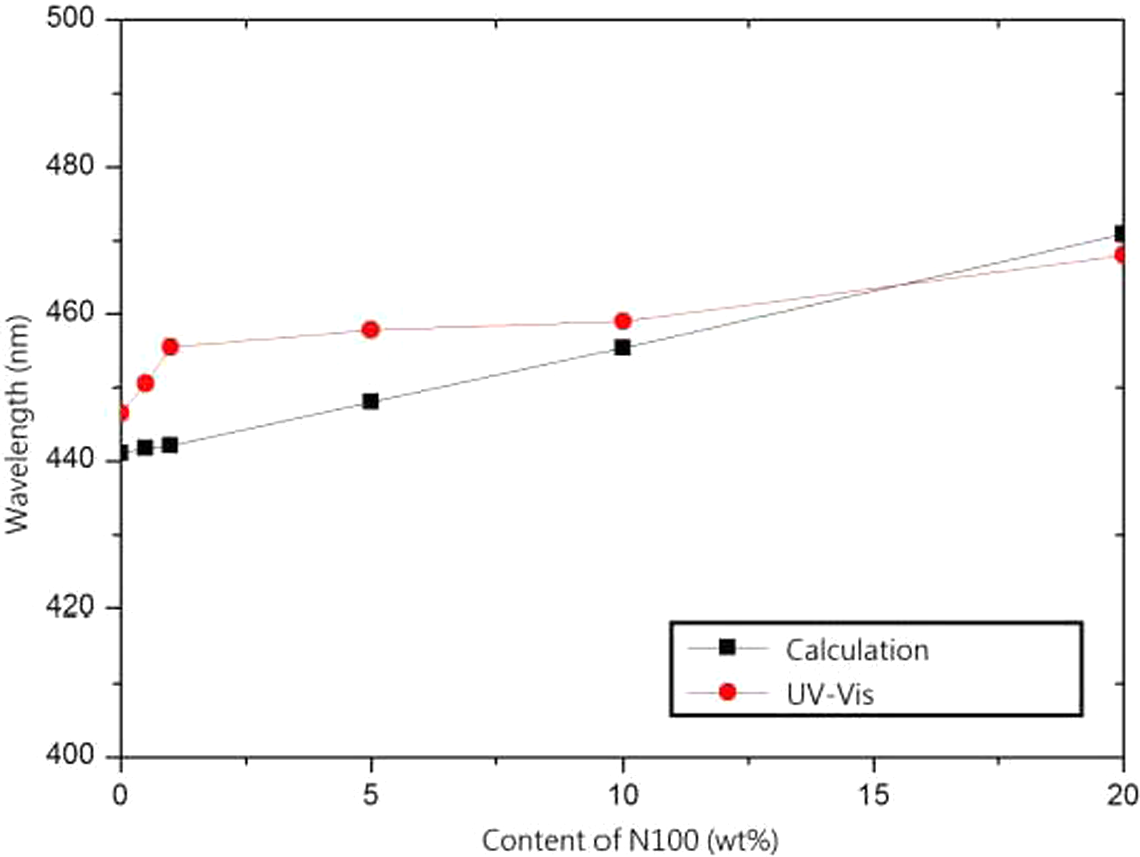
The relationship between wavelength and content of N100.
Characteristics of photonic crystal construction with PS spheres and N-54 (PS/N-54)
PS spheres and N100 nanospheres were clearly observed in the SEM images of the PBG film prepared from PS spheres with more than 1.0 wt% of N100. This phenomenon was also observed in the PS/N58 and PS/N26 series due to the high T g of the nanospheres.
Soft nanospheres with different content (0–20%) of N-54 (T g = −54°C) were chosen to blend with PS sphere, and SEM images are shown in Figure 4. The observation shows the significant results that the particles had a uniformly spherical morphology with only one diameter. This indicated that N-54 had low T g to coat onto PS spheres to form a core–shell architecture during the self-assembly process at 50°C. The influence of different N-54 content on the diameter of the core–shell spheres obtained from the experiment (SEM) is shown in Figure 5. By increasing the content of N-54, the diameter is increased. This result also corresponds to the calculation (UV-Vis) as shown in Figure 5.

SEM of PS spheres with various contents of N-54: (a) 0 wt%, (b) 0.5 wt%, (c) 1.0 wt%, (d) 5.0 wt%, (e) 10 wt%, and (f) 20 wt%. SEM: scanning electron microscopy; PS: polystyrene.
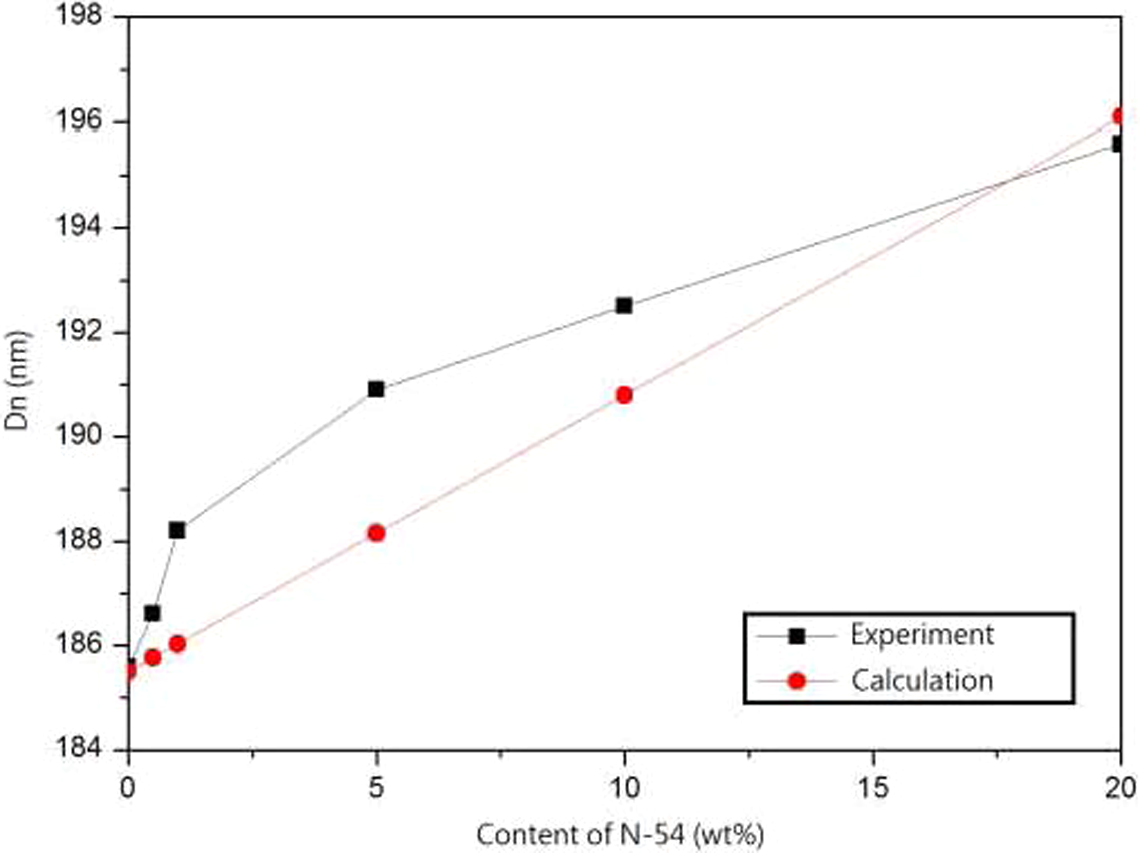
The relationship between D n and content of N-54. D n: number-averaged diameter.
Because the photonic crystal construction had FCC architecture, the periodic structure comprised two different refractive index materials, the core–shell spheres and air. The soft N-54 nanospheres were coated onto the PS spheres to increase the diameter. The slight increase in the diameter by N-54 did not influence the refractive index of core–shell submicrospheres. Therefore equation (6) could be modified as equation (9) giving an n eff of 1.460. However, Dn was obtained from SEM images, and λ was calculated by equation (5). These data are shown in Figure 6.
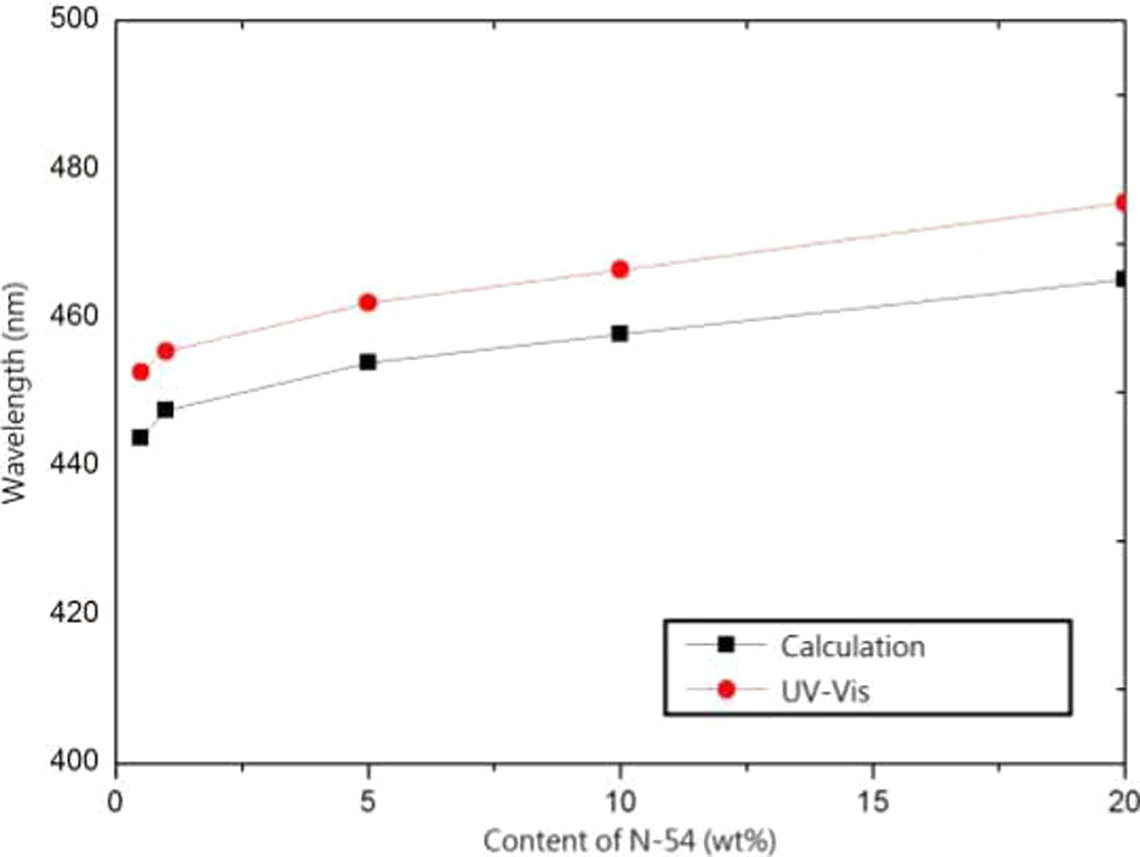
The relationship between wavelength and content of N-54.
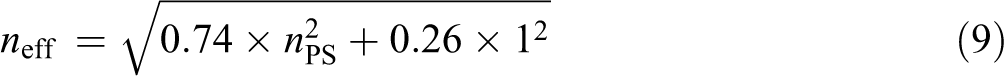
The reflection spectra of the films prepared with a series of PS/N-54 spheres at 50°C is shown in Figure 7. All of the films exhibited brilliant colors from violet to blue (λ max = 446.5–475.5nm), resulting from the diffraction of light inside the periodic structure comprising the core–shell spheres. The sharpness of the reflection peaks indicated the high quality of the opal film. The value of λ max was detected by UV-Vis spectroscopy, and the value of λ was calculated using equation (5) with the same tendency that was increased as N-54 content increased, as shown in Figure 6.
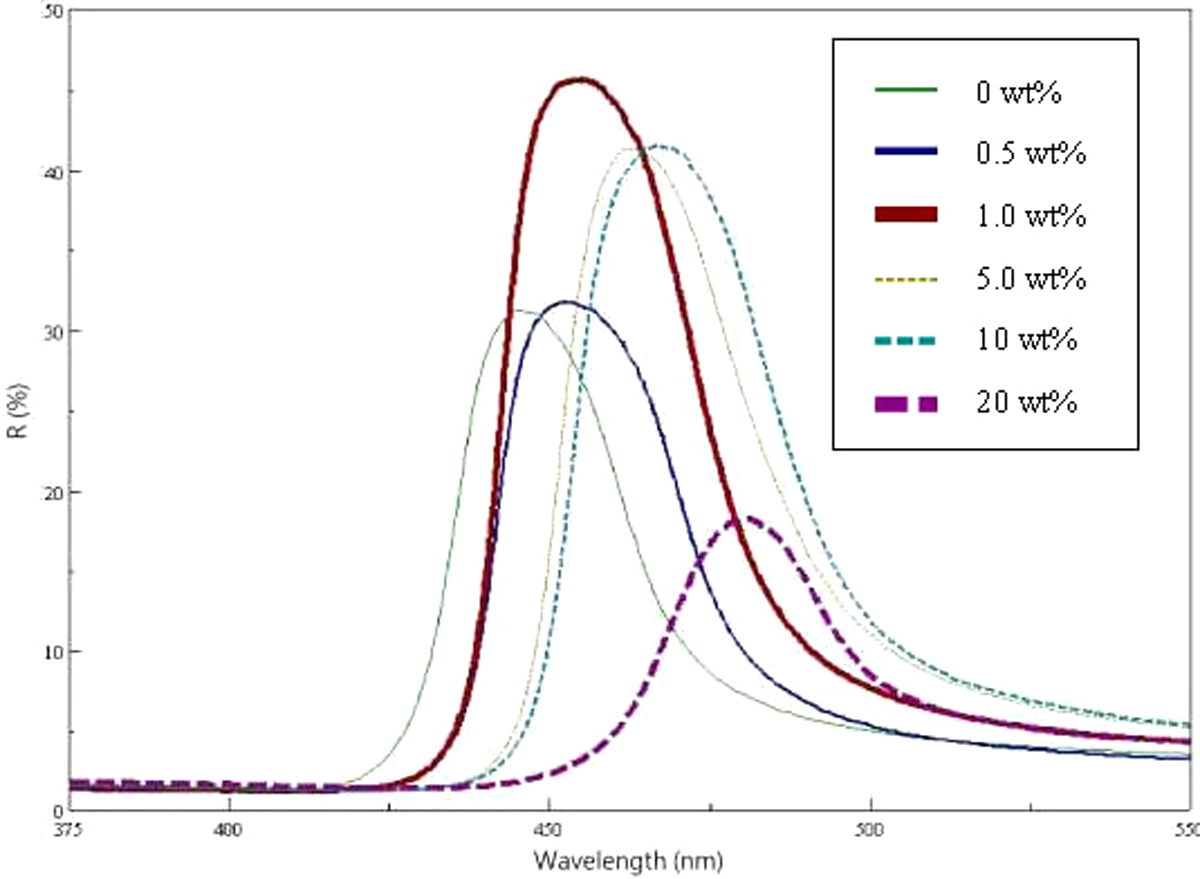
UV-Vis spectra of PS spheres with various contents of N-54. UV-Vis: ultraviolet-visible; PS: polystyrene.
As mentioned above, the SEM results of the films prepared with a series of PS/N-54 spheres showed only a uniformly spherical morphology. Because PS was blended with low-T g N-54 nanospheres, and these nanospheres were coated onto the PS sphere, this resulted in one size of sphere. The same results were also observed in the PS/N-17 and PS/N06 series.
Film-forming and hardness of photonic crystal construction with PS spheres and nanospheres
The mechanical properties of opal films could be obtained by PS spheres with nanospheres. As discussed above, soft nanospheres were coated onto PS spheres to adhere to each other and improve the mechanical properties of the films. However, the hard nanospheres retained their shapes, and only surround the PS spheres. It was difficult to improve the mechanical properties of the film without adding any further adhesives. Pencil hardness (PH) is a generally accepted method for measuring the resistance of an opal film. PH is defined as the hardest pencil grade that does not mark the opal film when pressed firmly against it at 45°; the results of these tests are shown in Table 3. Regardless of the self-assembly environment temperature used, opal films made with PS/hard nanospheres exhibited less than satisfactory mechanical resistance (lower than 6B). In contrast, the opal films consisting of PS/soft nanospheres exhibited increasing PH with increasing T g of the soft nanospheres and increasing the amount of soft nanospheres in the opal film.
Pencil hardness of photonic crystals of PS spheres with different kinds and contents of nanospheres.
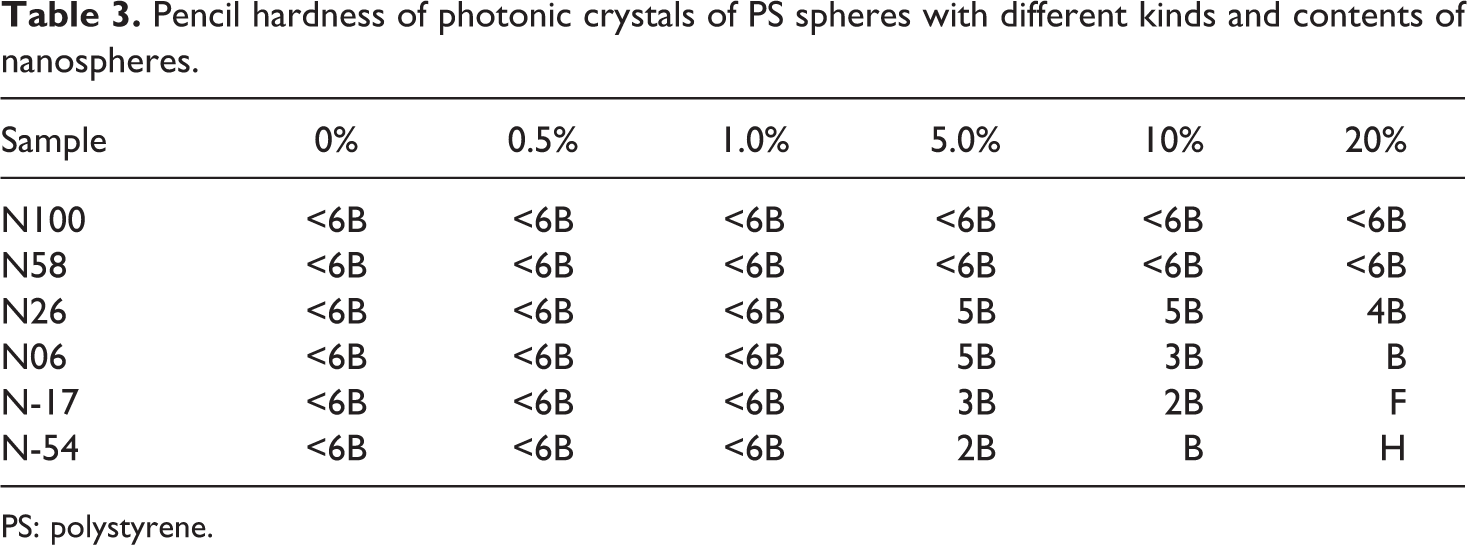
PS: polystyrene.
It is well known that the mobility level of a polymer chain on the molecular scale is reflected in the T g value. The lower the T g value of the nanospheres, the more adhesive is the film. This indicates that the PS spheres remained in the glassy state, and soft nanospheres were coated onto the PS spheres to increase the binding interaction between the spheres. The structure of the PS with soft nanospheres was designed to enhance the properties of the opal film by combining the periodic structure derived from the glassy state of the PS spheres and the binding force between the spheres resulting from the rubbery state of the shell formed by soft nanospheres. In addition, by increasing the amount of soft nanospheres, the hardness of the film was also increased. Consequently, a higher PH (H) could be achieved with the opal film self-assembled from PS with N-54 spheres.
Conclusions
Nanospheres with different T g values (100 to −54°C) were successfully prepared by soap-free polymerization of one or two kinds of monomers, St, BMA, and BA with NaSS. Photonic crystal fabrication could be successfully produced by the self-assembly method of mixing PS spheres and various T gs of nanospheres.
Photonic crystal prepared from PS spheres with hard nanospheres showed that nanospheres were distributed around PS spheres. However, photonic crystal prepared from PS spheres with soft nanospheres showed that soft nanospheres were coated onto the PS spheres. By increasing the weight fraction of hard or soft nanospheres in the photonic crystals, Dn and λ were increased. The value of λ max corresponded to that of λ.
This method is not only simple and feasible, but the obtained photonic crystal fabrications also had good robust properties including their PH value. The color and hardness were tunable by varying the T g and wt% of the nanospheres due to the behavior of the nanospheres during the self-assembly process. Thus, photonic crystals with increased PH were produced when soft N-54 nanospheres were blended with PS spheres.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.




