Abstract
Poly(benzimidazole/ether/siloxane/amide) (PBESA) having siloxane and ether groups in the backbone has been prepared using 4-(3,4-diaminophenoxy)benzene-1,2-diamine, bis(carboxypropy)tetramethyldisiloxane, and 4,4′-oxydianiline via polyphosphoric acid processes with heating up to 160°C. The sulfonation of polystyrene (PS-S) was conducted using 98% sulfuric acid. Afterward, a series of hybrid membranes using PBESA/PS-S/silica nanoparticles (SiNPs) have been developed with 0.1–2 wt% nanofiller. Later, the membranes were doped with phosphoric acid and subjected to various characterization techniques. Field emission scanning electron micrographs (FESEMs) showed gyroid-like patterning of nanoporous membranes with uniform ionic pathways. Fine water retention capability and higher proton conductivity of new hybrids, owing to consistent porous membrane structure, were observed. Increasing the amount of nanoparticles (0.1–2 wt%) also enhanced the tensile stress of acid-doped PBESA/PS-S/SiNPs nanocomposites from 64.9 to 68.1 MPa. There existed a relationship between nanofiller loading and thermal stability of the membranes. The glass transition temperature of phosphoric acid–doped PBESA/PS-S/SiNPs nanocomposites increased from 202 to 214°C. The membranes also had fine ion exchange capacity (IEC) of around 2.5–3.7 mmol g−1. Novel membranes with high IEC value achieved high proton conductivity of 1.26–2.74 S cm−1 in a wide range of humidity values at 80°C, which was higher than that for perfluorinated Nafion® 117 membrane (1.1 × 10−1 S cm−1 at 80°C, 94% relative humidity (RH)). The fuel cell (hydrogen/oxygen) using PBESA/PS-S/SiNPs 2 (IEC 3.7 mmol g−1) showed better performance than that of Nafion® 117 at 40°C (30% RH).
Keywords
Introduction
In recent decades, proton exchange membrane (PEM) fuel cells have gained considerable attention owing to their application as a replacement for batteries and in portable power sources. In this regard, various types of mechanically stable polymer membranes with fine proton conductivity have been developed. Commercially available perfluorosulfonate ionomer membranes such as Nafion (DuPont, Delaware, USA), Flemion (Asahi Glass Co., Tokyo, Japan), and Acipex (Asahi Chemical, Tokyo, Japan) have been widely used in fuel cells. 1 Nevertheless, use of perfluorosulfonic acid (PFSA) membranes has found to be inappropriate on commercial scale because of high cost, low conductivity at higher temperature, and low humidity. 2,3 A well-known thermally stable polybenzimidazole doped with acid (usually phosphoric acid) has been recognized as an alternative for high-temperature proton exchange fuel cell membrane. 4 Likewise, alkali-doped polybenzimidazole has been known to enhance the conductivity of ions. 5 In addition to their moderate conductivity at higher temperature, the acid–base blend membranes have also been reported to have reduced methanol permeability and thermal and chemical stability. 6 Acid doping has been more popular as the acid-doped polybenzimidazoles show fine proton conductivity at high temperature of up to 200°C. 7 Thermal stability, mechanical properties, water drag coefficient, kinetics of oxygen reduction, and other properties of the acid-doped polybenzimidazole membranes have also been focused in literature. 8,9 Consequently, the conductivity of these membranes depends on the level of phosphoric acid doping and water content. Generally, the high doping level influences the high proton conductivity of the membranes. On the other hand, the mechanical properties of the acid-doped membranes have often been found to be lower on increased doping level. 10 Moreover, the water content has also been found to influence the membrane properties. 11
Polybenzimidazole-based high-temperature PEMs present much compensation in the basic fuel cell design including the confrontation to carbon monoxide and other fuel impurities, and operational temperature above 120°C permits simplified water and thermal management.
12
Consequently, polybenzimidazole has emerged as a prime candidate for low-cost membranes with fine heat resistance and proton conductivity.
13
The synthesis of polybenzimidazole involving dicarboxylic acid and tetraamine via polyphosphoric acid (PPA) process (190–220°C; 16–24 h) has been reported.
14,15
Poly[2,2′-(
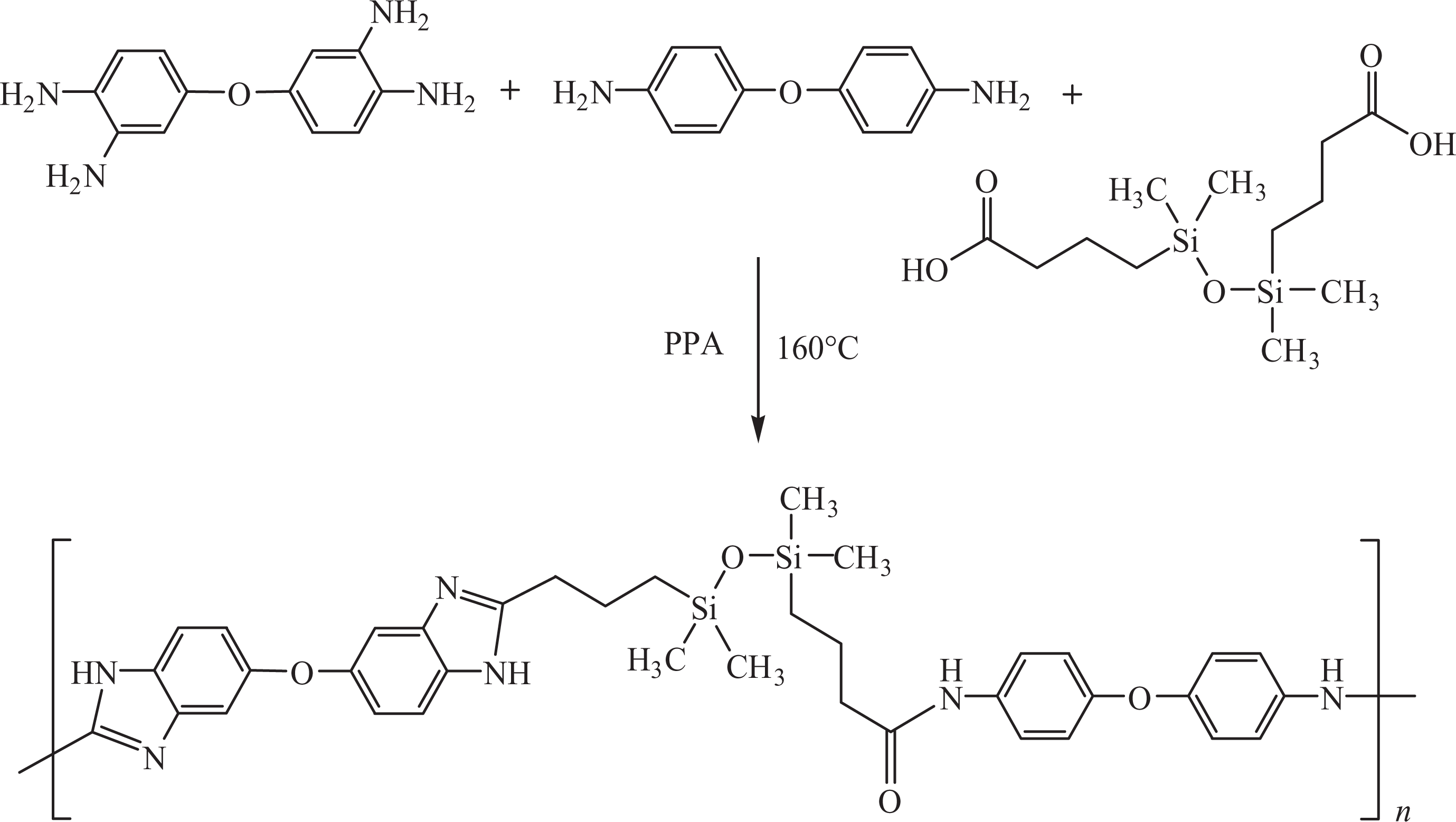
Synthetic route to PBESA. PBESA: poly(benzimidazole/ether/siloxane/amide).
Experimental
Chemicals
DupontTM Nafion® 117 PFSA membrane (nonreinforced films based on chemically stabilized PFSA/polytetrafluoroethylene copolymer in the acid (H+) form), polystyrene (
Instruments
Infrared (IR) spectra were recorded using Fourier transform IR (FTIR) spectrometer (model no. FTSW 300 MX; Bio-Rad, California, USA) (4 cm−1 resolution). Molar mass was calculated through gel permeation chromatography using dimethylformamide/THF. FESEM of freeze-fractured samples was performed using field emission scanning electron microscope (FESEM) (JSM5910, JEOL, Tokyo, Japan). Differential scanning calorimetry (DSC) was performed by Mettler Toledo DSC 822 differential scanning calorimeter) taking 5–10 mg of samples at 10°C/min under nitrogen. Tensile measurement was performed with a mechanical testing Instron-1211 instrument (Instron Co., Norwood, MA, USA) at a speed of 2 mm min−1 at 60% relative humidity (RH) (25°C). The humidity of water uptake was measured by placing the membrane in a thermo-controlled humidity chamber for 4 h. Then the membrane was taken out and quickly weighed on a microbalance. Water uptake was calculated using the following equation:
where
Dimensional change in the hydrated membrane was investigated by placing the membrane in water at 20°C or 80°C for 4 h, and the changes in length were calculated using the following equation:
where 
where
Synthesis of SiNPs
A solution containing 200 mL of methanol, 20 mL of 28 wt% ammonia (NH3), 2 mL of deionized water, and 16.64 g of TEOS was completely mixed by stirring. The final concentration in the mixture was 0.33 M for TEOS, 1.2 M for NH3, 8 M for water and 13.6 M for methanol. The above mixture was continually stirred at 60°C for 3 h. Afterward the suspension was centrifuged at 13,000 r/min for 15 min and SiNPs precipitate was obtained. The precipitate was filtered and dried. 23
Functionalization of PS-S
Four grams of polystyrene were dissolved in 40 mL THF in a 250-mL conical flask equipped with mechanical agitation, vertical condenser, and thermometer. Fifteen milliliter of concentrated H2SO4 was slowly added dropwise to the above mixture. The reaction flask was then placed in an ice–salt mixture for 2 h to be cooled to 0°C. Then the mixture was brought to room temperature. The mixture was heated to 70°C for 2 h and again cooled to room temperature. The polymer was neutralized with 10% sodium hydroxide (NaOH) solution and afterward with distilled water to maintain the neutralized pH. FTIR: 3394 cm−1 (OH stretching), 3082 and 3059 cm−1 (aromatic C–H stretch), 2925 and 2850 cm−1 (asymmetric and symmetric CH2 vibrations), 1190 cm−1 (asymmetric SO2 stretch), 1010 and 1040 cm−1 (symmetric SO2 stretch), and 670 cm−1 (S–OH) (Figure 2).
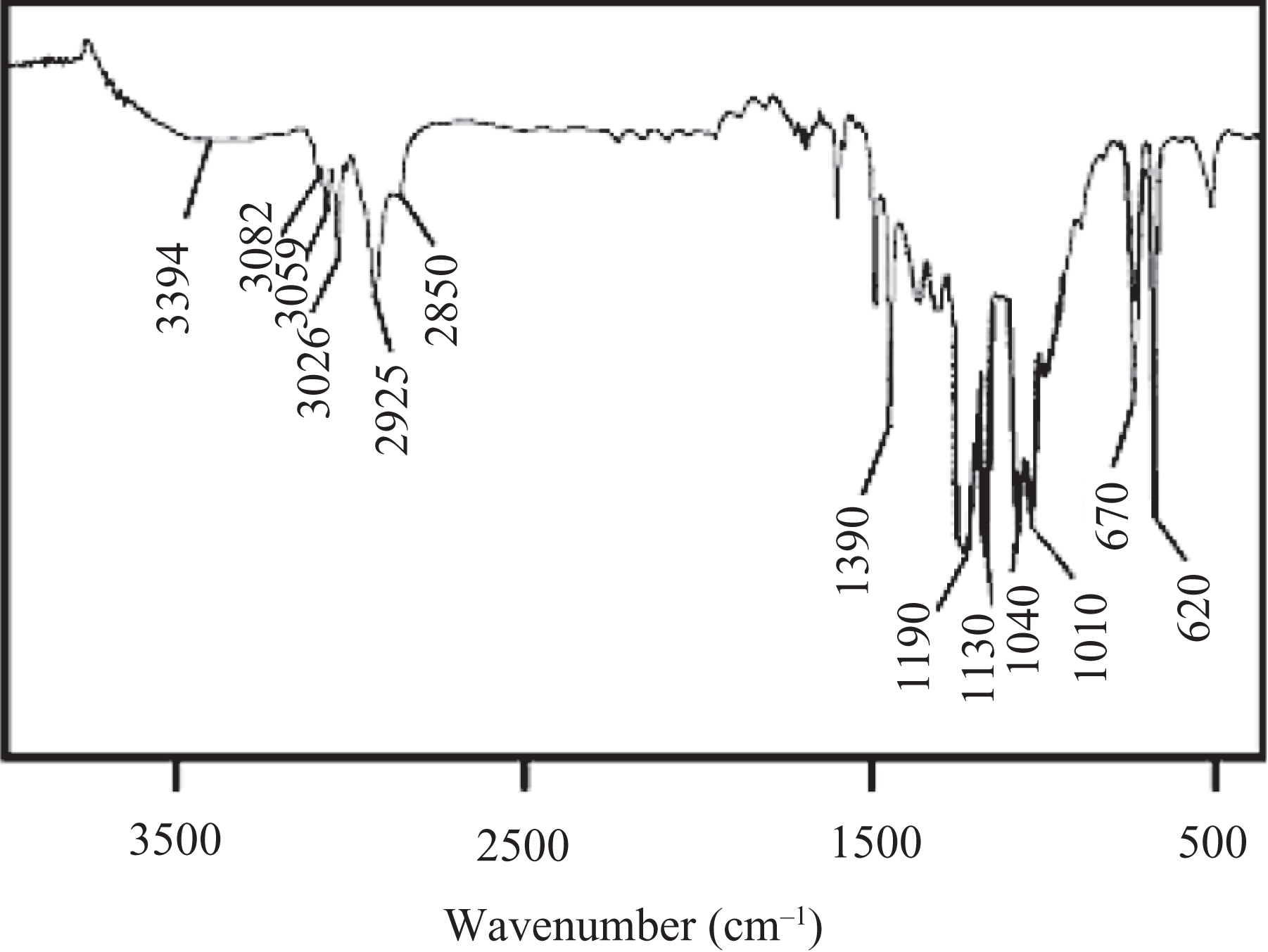
FTIR spectrum of PS-S. FTIR: Fourier transform infrared; PS-S: sulfonated polystyrene.
Synthesis of PBESA
To a 500-mL three-necked round-bottomed flask equipped with a mechanical stirrer and a condenser, 2.87 g (12.5 mmol) of 4-(3,4-diaminophenoxy)benzene-1,2-diamine, 1.916 g (6.3 mmol) of BTMDS, 1.26 g (6.3 mmol) of 4,4′-oxydianiline, 12.97 g of phosphorus pentoxide, and 134 g of PPA (preheated at 120°C) were added. The solution was heated up to 160°C and stirred for 8 h under nitrogen. The resulting viscous solution was added dropwise to 5 L of deionized water by stirring continuously. The precipitated polymer was collected by filtration and washed with 10 wt% NaOH. The polymer was further washed several times with deionized water. The polymer was dried under vacuum at 100°C for 24 h. Molecular weight of PBESA was found to be
Synthesis of PBESA/PS-S blend
Stock solutions of PS-S and PBESA were prepared separately in DMAc. The desired amounts of both the solutions were then mixed and stirred for 24 h to prepare PBESA/PS-S blend (50:50). FTIR: 3378 cm−1 (N–H stretching), 1633 cm−1 (N–H bending), 3003 cm−1 (aromatic C–H stretch), 2945 and 2891 cm−1 (asymmetric and symmetric CH2 vibrations), 1132 and 1188 cm−1 (asymmetric SO2 stretch), 1016 and 1068 cm−1 (symmetric SO2 stretch), and 699 cm−1 (S–OH) (Figure 3).
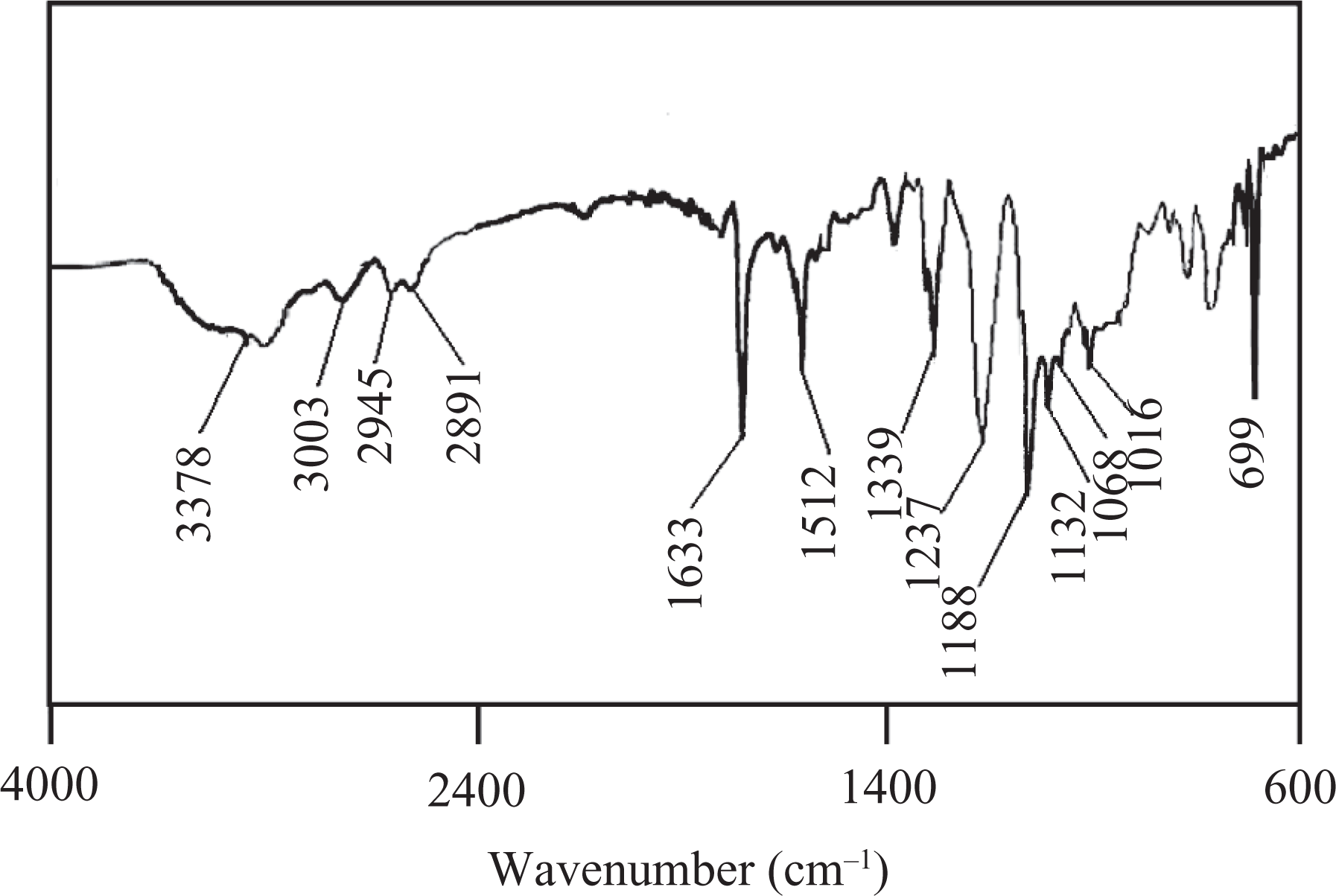
FTIR spectrum of PBESA/PS-S. FTIR: Fourier transform infrared; PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene.
Preparation of PBESA/PS-S/SiNPs nanocomposites
Stock solution of polymer was first prepared by dissolving the desired amount of PBESA/PS-S in DMAc. Nanocomposites were prepared by mixing SiNPs with 0.1–2 wt% with appropriate amount of the stock solution. Thin films were cast in glass Petri dishes by evaporating solvent at 70°C. Thin films were dried at 80°C for at least 24 h under vacuum.
Preparation and doping of PBESA/PS-S/SiNPs membranes
PBESA/PS-S/SiNPs powder, 0.5 g, was dissolved in 25 g of DMAc to make a 2% (w/w) solution. Films were cast from the viscous solution on glass plates by a doctor’s knife. The films were dried under vacuum at 80°C for 24 h. The films were doped by immersion in 9 M phosphoric acid solution for 100 h. The films were white in color and transparent (Figure 4).

(a) PBESA/PS-S and (b) PBESA/PS-S/SiNPs 1 nanocomposites. PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles.
Results and discussion
Mechanical properties
Mechanical analysis data for PBESA/PS-S/SiNPs nanocomposites with 0.1–2 wt% nanoparticles are shown in Table 1. PBESA/PS-S was selected for the PBESA/PS-S blend, while in nanocomposites the number in the sample designation referred to weight percentage of SiNPs. Stress–strain curves of PBESA/PS-S/SiNPs 0.1–2 showed the mechanical trends of nanocomposites with various filler loading (Figure 5). Ultimate tensile stress value for pure blend PBESA/PS-S was 53.5 MPa. Afterward, the mechanical strength was increased with an increase in filler content. PBESA/PS-S/SiNPs 0.1 showed the ultimate tensile stress of 64.9 MPa, and it enhanced to 66.9 MPa in PBESA/PS-S/SiNPs 0.5. Accordingly, the inclusion of 1 wt% silica nanotubes in PBESA/PS-S/SiNPs 1 increased the strength up to 67.7 MPa, which was further raised to 68.1 MPa in PBESA/PS-S/SiNPs 2. The enhanced mechanical property values signified an enhancement in strength of materials because of fine interaction between the filler and blend. The elongation at break for pure blend PBESA/PS-S was 0.52%. PBESA/PS-S/SiNPs 0.1 with 0.1 wt% SiNPs nanocomposites showed the elongation at break value of around 0.88%, which faintly increased to 1.51% for PBESA/PS-S/SiNPs 2 (2 wt% SiNPs). Tensile modulus for PBESA/PS-S/SiNPs 0.1 was 7.2 GPa, which was superior to PBESA/PS-S (6.1 GPa). The tensile modulus property was further increased up to 8.9 GPa in PBESA/PS-S/SiNPs 1 and to 10.1 GPa in PBESA/PS-S/SiNPs 2. Toughness of the membrane was also found to increase with the nanofiller, as pure PBESA/PS-S blend had a value of 32.7 MPa, which was further enhanced to 47.4 MPa in PBESA/PS-S/SiNPs 0.1. The addition of 0.5, 1, and 2 wt% SiNPs increased the toughness to 50.6 MPa, 58.9 MPa, and 61.9 MPa, respectively. There was an overall enhancement in the mechanical properties from 0.1 to 2 wt% possibly due to the fine compatibility between the polymer blend and nanofiller. Furthermore, fine contact between the matrix and nanoparticles was attained because of the homogenous dispersion of filler in the matrix domains. 24
Mechanical properties of PBESA/PS-S and phosphoric acid–doped PBESA/PS-S/SiNPs membranes.
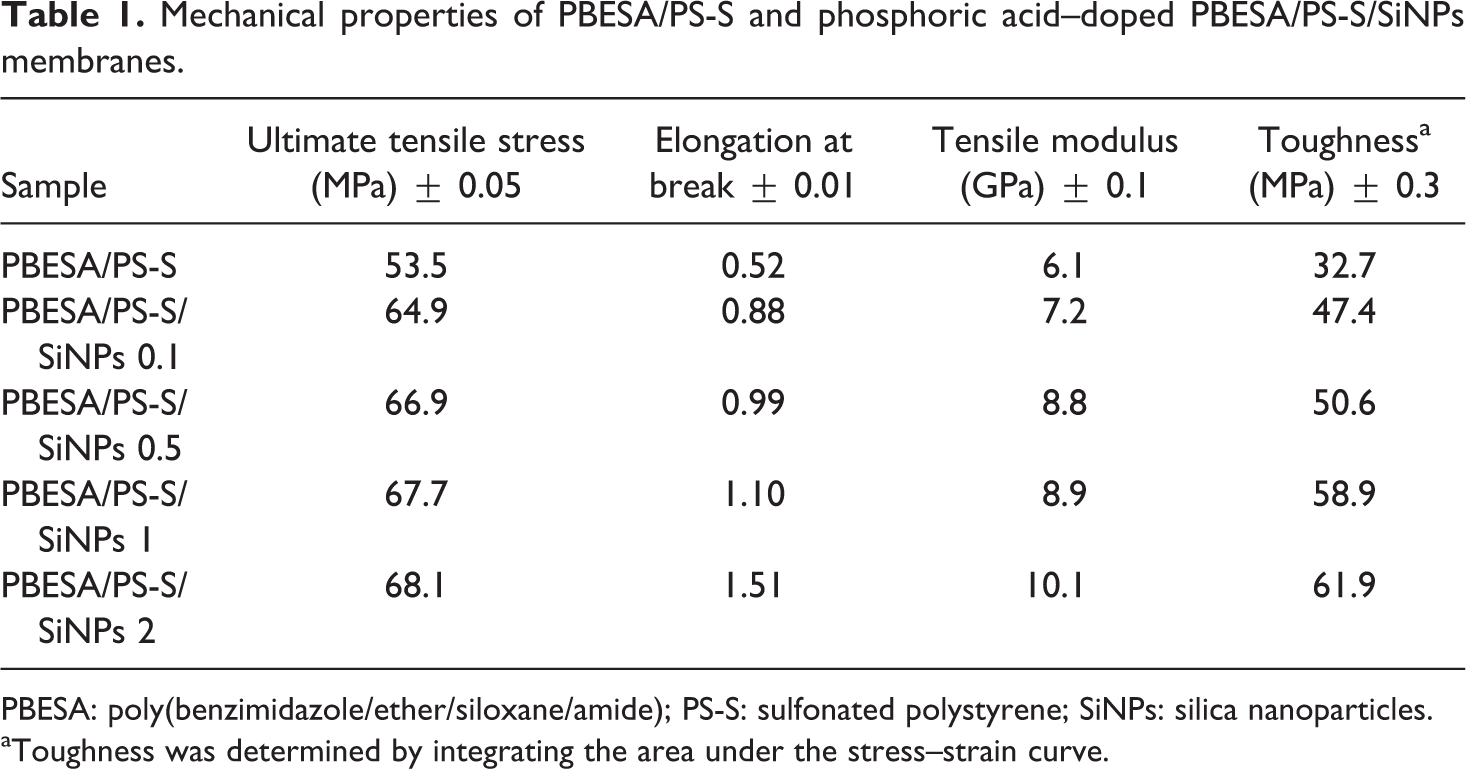
PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles. aToughness was determined by integrating the area under the stress–strain curve.
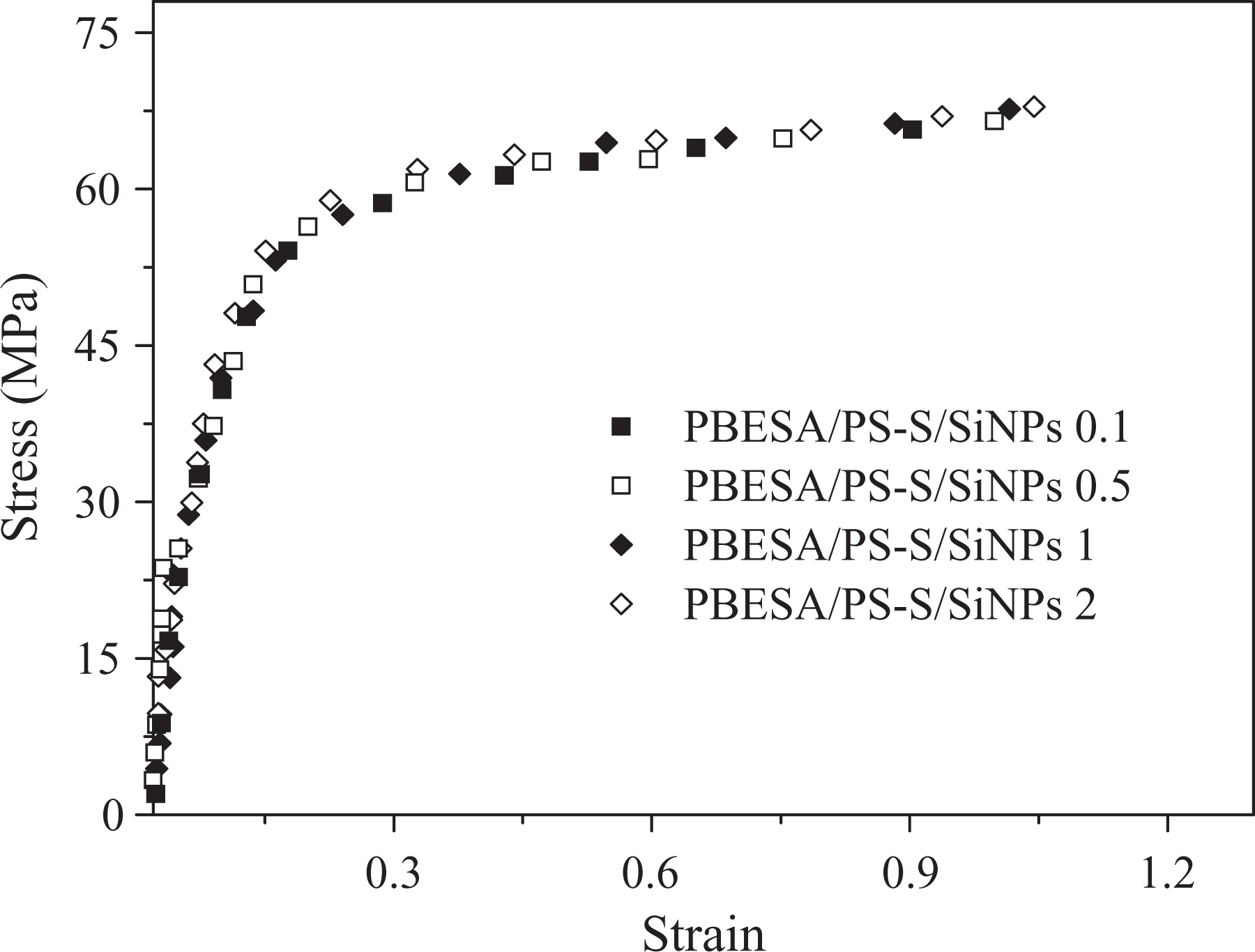
Stress–strain curves of PBESA/PS-S/SiNPs nanocomposites. PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles.
Morphology
FESEMs were used to study the morphology of cryo-fractured samples of blend membranes (Figure 6). All membranes depicted a symmetric porous membrane structure with fully developed micropores. The pores were assembled in somewhat gyroid-like morphology. Excellent water retention capability and higher proton conductivity of the new membranes were observed due to the porous membrane structure. Moreover, the addition of increasing nanoparticle content did not suppress the formation of macropores in the membrane structure (Figure 6(a) to (d)). However, there was a slight increase in the pore size of membranes with the addition of 0.1–2 wt% SiNPs in PBESA/PS-S/SiNPs. Figure 6(a) shows the alignment of nanoparticles in the macroporous walls without disturbing the gyroid porous structure. The porous structure was sustained with the addition of 0.5 wt% filler in PBESA/PS-S/SiNPs 0.5 as shown in Figure 6(b). Nevertheless, with the increasing amount of nanoparticles, the porous gyroid structure became wider with larger pore structure owing to the phase separation. Figure 6(d) represents the morphology of PBESA/PS-S/SiNPs 2 with larger pores relative to PBESA/PS-S/SiNPs 0.1. The exceptional morphology was surely due to the compatibility between the SiNPs and the blend components. The porous PBESA/PS-S bend/SiNP membrane structure was accountable for fine water retention representing higher proton conductivity of the nanocomposites.

FESEM images of (a) PBESA/PS-S/SiNPs 0.1, (b) PBESA/PS-S/SiNPs 0.5, (c) PBESA/PS-S/SiNPs 1, and (d) PBESA/PS-S/SiNPs 2. FESEM: field emission scanning electron micrography; PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles.
Thermal analysis
Glass transition temperature (
Thermal analysis data of PBESA/PS-S and phosphoric acid–doped PBESA/PS-S/SiNPs membranes.

PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles;
DSC thermograms of PBESA/PS-S/SiNPs at a heating rate of 10°C min−1 in N2. DSC: differential scanning calorimetry; PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles; N2: nitrogen.
Water uptake and dimensional change
Table 3 shows the water uptake and water stability of PBESA and PS-S blend membranes at 50 or 80°C. There subsisted an association between the water uptake and ion exchange capacity (IEC) since the membranes with higher IEC values tended to uptake more water. The stability of membranes was examined by soaking the membranes into distilled water (50 or 80°C) and was characterized by time spent until the hydrated membranes lose their mechanical properties. The standard for the judgment of the loss of mechanical properties was that the membrane was broken when bowed. Both the PBESA/PS-S blend and PBESA/PS-S/SiNPs 0.1–2 membranes were stable at high temperature (80°C) and were not dissolved in water. In addition, all the membranes were insoluble at elevated temperature (e.g. 80°C) when tested in water at 80°C for more than 600 h. The increase in temperature and IEC influences an increase in water uptake and swelling ratio for the membranes. PBESA/PS-S/SiNPs 0.1 showed water uptake of 1.9 and 17.9% at 34 and 94% RH, respectively, at IEC value of 2.5 mmol g−1. With the increasing filler content in PBESA/PS-S/SiNPs 0.5, water uptake was 2.7 and 18.6% at 34 and 94% RH, respectively, at 2.6 mmol g−1 IEC. The water uptake was further increased up to 8.3 and 25.8% at 34 and 94% RH, respectively, at 3.7 mmol g−1 IEC in PBESA/PS-S/SiNPs 2. As a result, the swelling ratio of PBESA/PS-S and PBESA/PS-S/SiNPs 0.1–2 at 20°C and 80°C, respectively, in water was also measured. The PBESA/PS-S/SiNPs 0.1–2 membranes with IEC 2.5–3.7 mmol g−1 showed rapid increase in swelling ratio at high temperature due to the formation of a large and continuous ion network in the phosphoric acid–doped matrix. The swelling ratio of PBESA/PS-S/SiNPs 0.1–2 increased from 5.8 to 18.3 at 20°C and 12.3 to 26.1 at 80°C compared with pristine PBESA/PS-S blend. The blend membranes also demonstrated excellent dimensional stability relative to reported sulfonated poly(aryl-ether-sulfone)s with analogous IEC value frequently dissolved in water or lost their mechanical properties because of serious swelling. 25,26 Moreover, the IEC value of PBESA/PS-S/SiNPs 2 membrane, that is, 3.7 mmol g−1 was superior than previous poly(aryl-ether-sulfone)s and other membranes. 27,28
Water uptake, swelling ratio, and conductivity of PBESA/PS-S and phosphoric acid–doped PBESA/PS-S/SiNPs membranes.
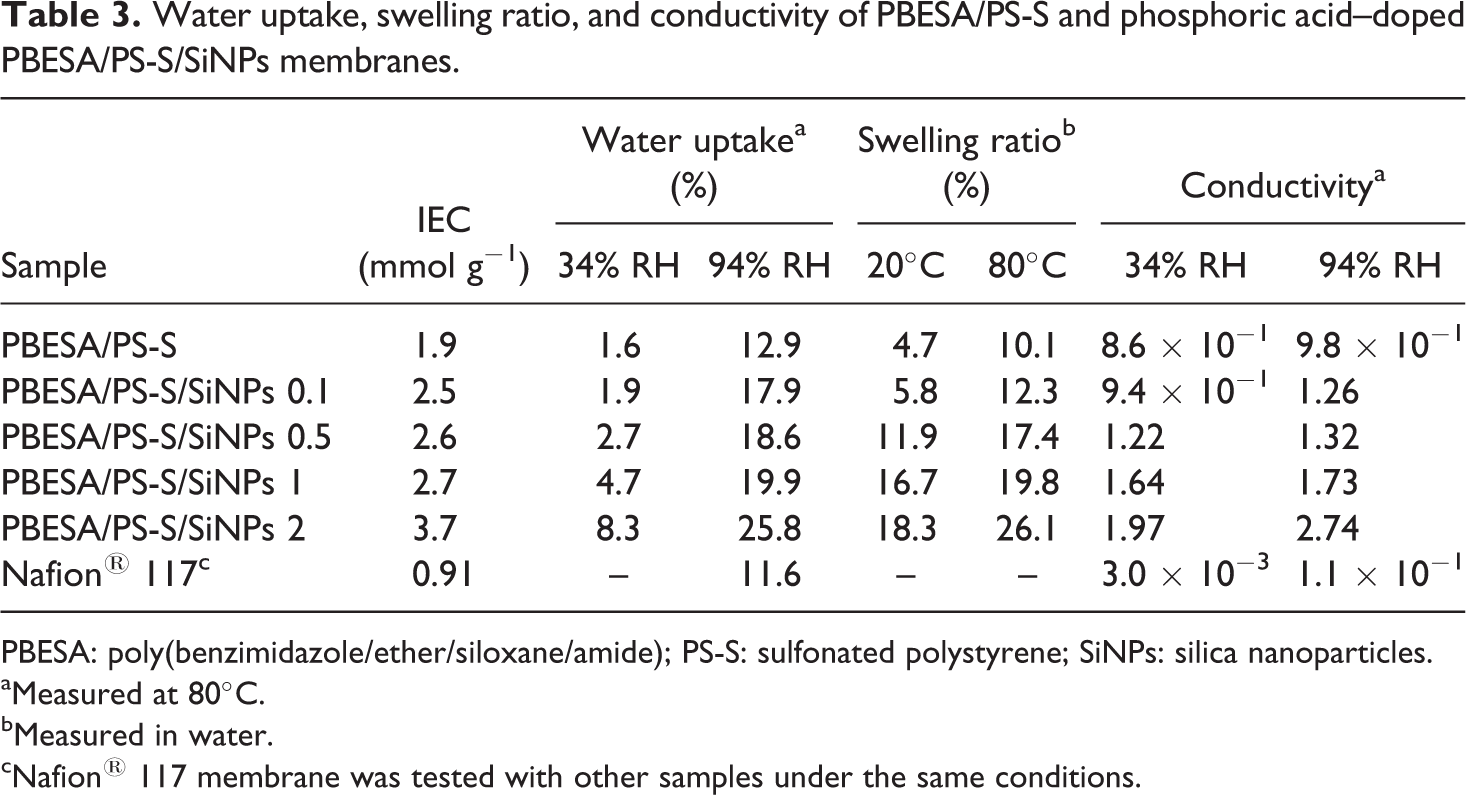
PBESA: poly(benzimidazole/ether/siloxane/amide); PS-S: sulfonated polystyrene; SiNPs: silica nanoparticles. aMeasured at 80°C.
bMeasured in water.
cNafion® 117 membrane was tested with other samples under the same conditions.
Proton conductivity
The proton conductivity of phosphoric acid–doped PBESA/PS-S blend with 0.1–2 wt% SiNPs and Nafion® 117 was measured at 80°C in the range 34–94% RH. Figure 8 presents the proton exchange mechanism in phosphoric acid–doped PBESA/PS-S/SiNPs membranes. PBESA/PS-S/SiNPs 0.1–2 membranes showed the proton conductivity of 1.26–2.74 S cm−1 at 94% RH and 9.4 × 10−1 to 1.97 S cm−1 at 34% RH. The proton conductivity increased with IEC values. To better understand the relationship between RH and proton conductivity, humidity dependence of the proton conductivity of PBESA/PS-S, PBESA/PS-S/SiNPs 0.1–2, and Nafion® 117 membranes was also examined. It was found that the conductivity was dependent on the humidity for IEC membranes as the conductivity of PBESA/PS-S/SiNPs 0.1–2 (IEC = 2.5–3.7 mmol g−1) increased from 1.26 to 2.74 S cm−1 (94% RH) at 80°C. This behavior is typical for the membranes with well-developed microphase separated structure as compared to perfluorinated ionomers. 28,29 Though IEC and conductivity were slightly lower for pure PBESA/PS-S blend, screening the effect of the addition of SiNPs.
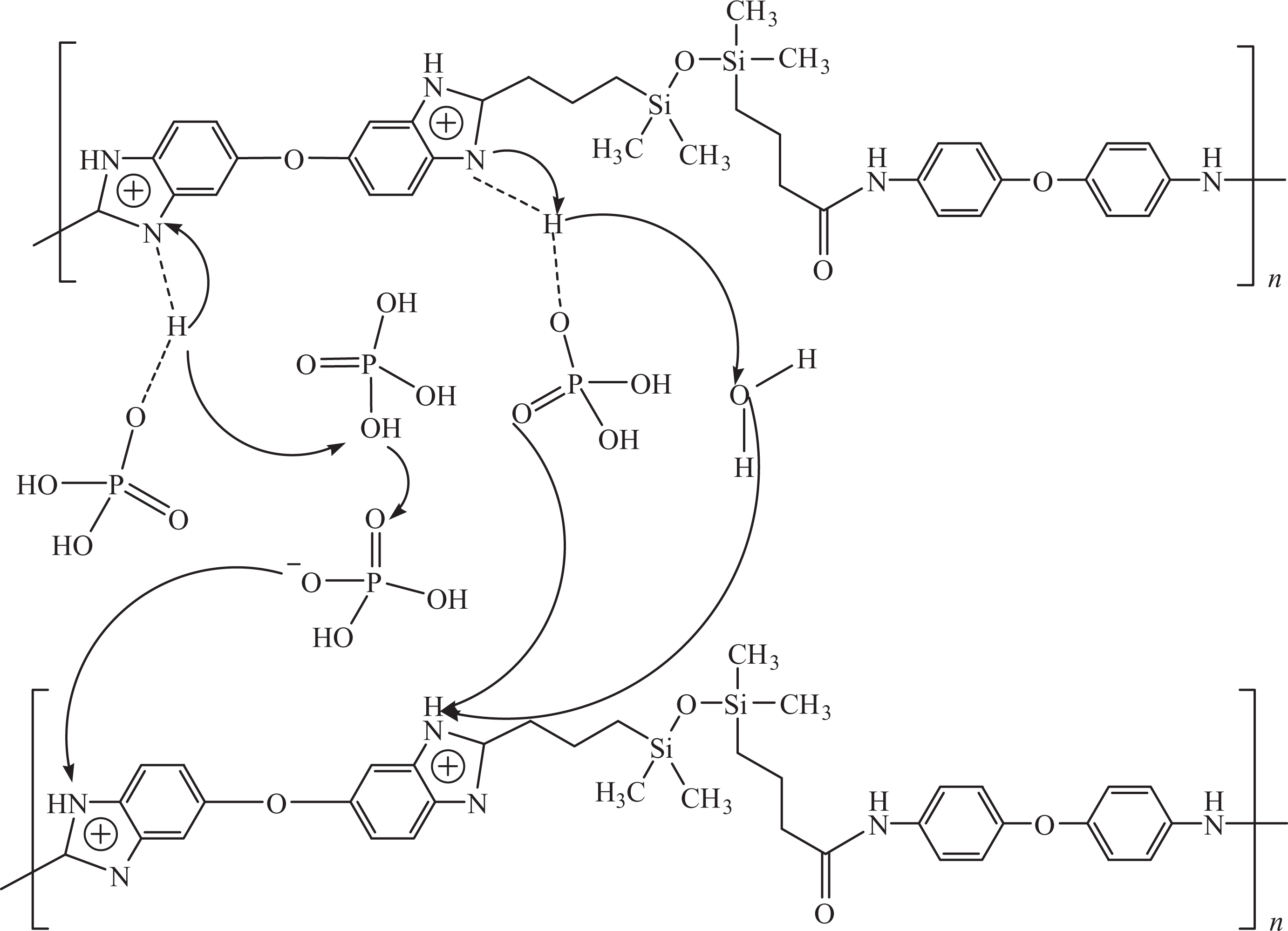
Proton transport mechanism in PBESA. PBESA: poly(benzimidazole/ether/siloxane/amide).
Conclusion
In this effort, high-molecular-weight PBESA was prepared and utilized for the fabrication of a series of blend membranes of PBESA, PS-S, and SiNPs doped with phosphoric acid. Owing to flexible siloxane and ether groups, the PBESA copolymers have good solubility in common organic solvents and have high molar mass of 24 × 103 g mol−1. PBESA/PS-S/SiNPs membranes were easily prepared from the copolymers by solution casting in DMAc. The phosphoric acid–doped membranes revealed fine thermal and mechanical profile. The membranes also had good water swelling resistance. FESEM study revealed distinct porous network structure with uniform ionic pathways in all membranes (PBESA/PS-S/SiNPs 0.1–2). The good water holding capability and porous structure render novel membranes with high IEC values and high proton conductivity in a wide range of humidity. Furthermore, PBESA/PS-S/SiNPs 2 may act as functional PEM materials as tested in H2/O2 fuel cell (IEC = 2.5–3.7 mmol g−1) and found to have superior performance relative to Nafion® 117.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
