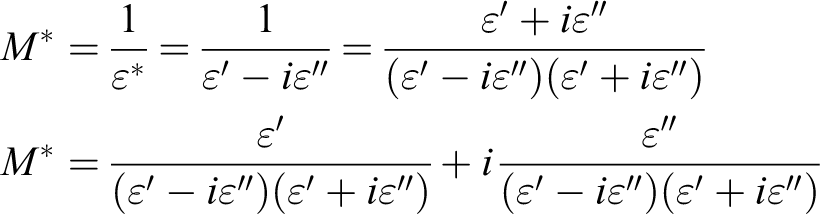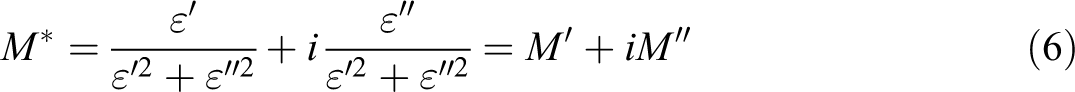Abstract
The effect of β-hydroxynaphthoic acid (β-HNA) addition on electrical and mechanical properties of multiwalled carbon nanotubes (MWCNTs)–polystyrene (PS) composite at a fixed amount of MWCNTs (0.85 wt%) are delineated. Obtained electrical, mechanical, morphological, and Fourier transform infrared results showed that addition of β-HNA up to 1 wt% to MWCNTs-PS composite enhanced the dispersion of MWCNTs in the neat PS, formation of MWCNTs-PS network, relative permittivity, dielectric loss, and the electrical conductivity increased by about three orders of magnitude for the prepared composite. Dielectric relaxation results showed that at the domain frequency range, the relaxation process below 1.0 wt% β-HNA was mostly due to polymer molecular relaxation process, while at 1.0 wt% β-HNA, the composite relaxation behavior was mostly due to charge conductivity relaxation. Mechanical results showed that addition of β-HNA to composite system up to 1.0 wt% increased the elastic modulus, yield stress, and tensile strength of the composites by about 12.2, 17.1, and 10.0%, respectively, which indicated that the addition of β-HNA to composite system improved MWCNTs dispersion in PS matrix and enhanced the interfacial bond in the β-HNA-MWCNTs-PS network.
Keywords
Introduction
Many studies were focused on carbon nanotubes (CNTs) as nanofillers in polymers due to their very attractive and unique properties to create new materials with attractive properties that are very useful for industrial applications. 1 –7 At the same time, the dispersion factor of CNTs in the neat polymers is still up to date represents the big challenge to improve the physical properties of CNTs-polymer composites and to obtain new materials with attractive properties for the industrial applications. One of the well-known widely used methods by many researchers to improve the dispersion of CNTs in the neat polymer matrix is the wet chemical method by functionalizing CNTs using different strong acids. At the same time, many studies showed that acid treatment will cause morphological damages to CNTs and create a significant physical damage in addition to severe degradation of CNTs. 5 –16 On the other hand, the use of ozone in the presence of ultraviolet (UV/O3) at ambient atmosphere have been an alternative treatment to resolve the issues associated with the wet oxidation. 2,3,9 Moreover, some studies appear to investigate the effect of pyrene pretreatment on the electrical properties of single-walled CNTs-polystyrene (PS) 17 composites or multiwalled CNTs (MWCNTs)–Polymethyl methacrylate composites. 18 Additionally, MWCNTs-PS composites were previously investigated with different preparation conditions such as coating MWCNTs with polypyrrole, 19 nickel-filled MWCNTs, 20 MWCNTs/carbon foams using polyurethane foam as a template, 21 microwave irradiation of MWCNTs, 16 UV/O3-pretreated MWCNTs. 2,3 Yu et al. 15 prepared MWCNTs-PS composites using the latex technique. Initially, they dispersed MWCNTs in aqueous solution of sodium dodecyl sulfate driven by sonication and then mixed them with different amounts of PS latex. They found that the percolation threshold was about 1.5 wt% MWCNTs. Mathur et al. 22 prepared the as-produced MWCNTs-PS using casting technique. They demonstrated that the electrical conductivity appear to increase significantly only at high level of MWCNTs content (up to 10 vol.%). However, to the best of our knowledge, there are no reported investigation on the effect of β-hydroxynaphthoic acid (β-HNA) addition on the dielectric properties and mechanical properties of MWCNTs-PS nanocomposites. As a part of our ongoing studies into the multiwalled CNTs-PS nanocomposites, 3,23 we recently reported a novel dispersion behavior of MWCNTs in tetrahydrofuran (THF) solvent induced by the addition of β-HNA. It was observed that β-HNA molecules adsorb onto the surface of MWCNTs due to inherent hydrophobic/hydrophilic sites and create partial electrostatic charge on the surface. Such an adsorption was confirmed using Fourier transform infrared (FTIR) spectroscopy and found to markedly influence the dispersion of MWCNTs. However, in the continuation of our previous work, the present work introduces additional and interesting effects of β-HNA addition on the dielectric properties, dielectric relaxation behavior, and mechanical properties of MWCNTs-PS composites.
The present work aims to study the effect of β-HNA addition on the electrical conductivity, dielectric properties, dielectric relaxation behavior, and mechanical properties of MWCNTs-PS composites at fixed amount of MWCNTs. The effect of β-HNA addition on the direct current (DC) electrical conductivity in addition to alternating current (AC) dielectric parameters such as relative permittivity (ε′), impedance (Z), dielectric loss (ε″), and AC electrical conductivity (σ AC)will be investigated as a function of β-HNA concentration at constant loading of MWCNTs-PS composite (near the percolation threshold of MWCNTs-PS). Measurements such as FTIR and morphology also will be considered and correlated with dielectric properties.
Experimental
Materials
The MWCNTs (purity: >95 wt%, outside diameter (OD): <8 nm, length: approximately 30 μm) used in this work were purchased from Chengdu Organic Chemicals Co. Ltd., Chinese Academy of Sciences (China). PS was kindly provided by Saudi Basic Industries Corporation (SABIC; Riyadh, Saudi Arabia) with a brand name of PS 125. β-HNA was obtained from Sigma-Aldrich Chemical Co. (St Louis, Missouri, USA) with a purity of >98 wt%. All the chemicals were used as received without further purification.
Preparation of MWCNTs-PS nanocomposite films
A casting technique was employed to prepare β-HNA-MWCNTs-PS nanocomposites at fixed amount of MWCNTs (0.85 wt% by weight) and different weight fraction of β-HNA. Set of solutions containing 0.85 wt% MWCNTs were probe ultrasonicated together with β-HNA (0–1 wt%) in THF solvent for 20 min using Heilscher Ultrasonic Processor (Germany) at an amplitude of 50% and a cycle of 0.5. PS was dissolved separately in THF under stirring conditions for 3 h. The solutions of sonicated MWCNTs were then added to the PS solutions followed by stirring of the resultant mixture for 24 h. The mixtures were carefully casted into glass petri dishes (diameter of 5 cm), enclosed with a glass cover, and allowed to dry for 4 days at ambient conditions. The thickness of the resulting nanocomposite films was found to be around 0.08 mm. To confirm the dispersion of MWCNTs in the neat PS and to observe the effect of β-HNA addition on the morphology of the resulting composites, a field-emission scanning electron microscope (JSM-7600, JEOL, Tokyo, Japan) was used.
The influence of β-HNA on the dispersion of MWCNTs in the neat PS matrix is shown in Figure 1. As can be seen from these figures, the dispersion of MWCNTs depends on the amount of β-HNA content in MWCNTs-PS composite. As the amount of β-HNA increases from 0 wt% to 1.0 wt%, the dispersion of MWCNTs in the neat PS is significantly improved.

SEM micrographs of β-HNA-MWCNTs-PS composite films: (a) 0.0 wt.% β-HNA (b) 0.25 wt.% β-HNA, and (c) 1 wt.% β-HNA. SEM: scanning electron microscopic; β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
Electrical measurements
Electrical measurements with AC and DC applied electric fields were performed using 4200-SCS Semiconductor Characterization System (KEITHLEY Co., Cleveland, Ohio, USA). Samples were shaped into circular discs having an area of 1.2 cm2 and thickness of 0.08 mm and placed between two copper electrodes. The DC electrical conductivity (σ
DC) was measured at room temperature and calculated using the formula
Z value and phase angle (φ) were obtained directly from the instrument and ε′, ε″, and σ
AC were determined from the known Z and φ values at room temperature and frequency range 1 kHz–1 MHz. The real (Z′) and imaginary (Z″) components of the Z can be calculated using the following relations:
3,24,25
The ε′ and ε″ values were calculated from the following equations:
26
where d is the sample thickness, A is the cross-sectional area of the sample, ε 0 is the permittivity of free space, and f is the frequency, in hertz.
The σ
AC value was calculated using the following equation:
Mechanical measurements (tensile test)
The tensile test was carried out on a dynamic mechanical analysis Q800 (TA Instruments LLC, New Castle, Delaware, USA) instrument using the tension film clamping arrangement. Specimens in the form of films (dimensions: length = 15 mm, width = 4 mm, thickness = 0.1 mm) were used. The tensile test on film samples were carried out at 25°C with a force rate of 3 N min−1.
FTIR analysis
FTIR spectra for the required samples were recorded using a PerkinElmer 100 FTIR spectrophotometer (Waltham, Massachusetts, USA) in the transmission mode at wave number range of 4000–400 cm−1. For the convenience, a careful adjustment of the Y-axis was carried out while keeping the original information intact and unchanged.
Results and discussion
Electrical properties
The effect of β-HNA addition on the DC electrical conductivity of the prepared MWCNTs-PS nanocomposites at room temperature and 0.85 wt% MWCNTs is presented in Figure 2 as a function of β-HNA weight fraction. It is obvious that addition of β-HNA to composite increases the electrical conductivity of the neat PS by about three orders of magnitude near the percolation threshold of MWCNTs-PS. 3 This indicates that the addition of β-HNA to composite system enhances the formation of MWCNTs-PS network structure that facilitates the electron transport through tunneling throughout the polymer or by electron hopping along CNTs interconnects. 1,2,27 It is well known that the electrical conductivity of CNTs-polymer composites depends on the polymer layer in the inter-nanotube connections, which presents highly resistive section in the electrical pathway, acting as a barrier to efficient carrier transport between the nanotubes. When CNTs concentration in the CNTs-polymer network marks insulator–conductor transition, then the CNTs weight fraction in neat polymer is referred to as the percolation threshold (P c). 1,2,27 We found in our previous work 3 that the electrical P c of UV/O3-treated MWCNTs-PS composites was around 0.8 wt% and the electrical conductivity increased by only about six orders of magnitude above P c value. While we showed in our recently published article 23 that the addition of 1.0 wt% β-HNA to MWCNTs-PS composites elevate the electrical conductivity to about eight orders of magnitude above P c value (P c was also around 0.8 wt% MWCNTs). As can be seen from the above results, addition of β-HNA enhances the dispersion of MWCNTs in the neat PS, the formation of MWCNTs-PS network, and electrical conductivity of prepared composites.
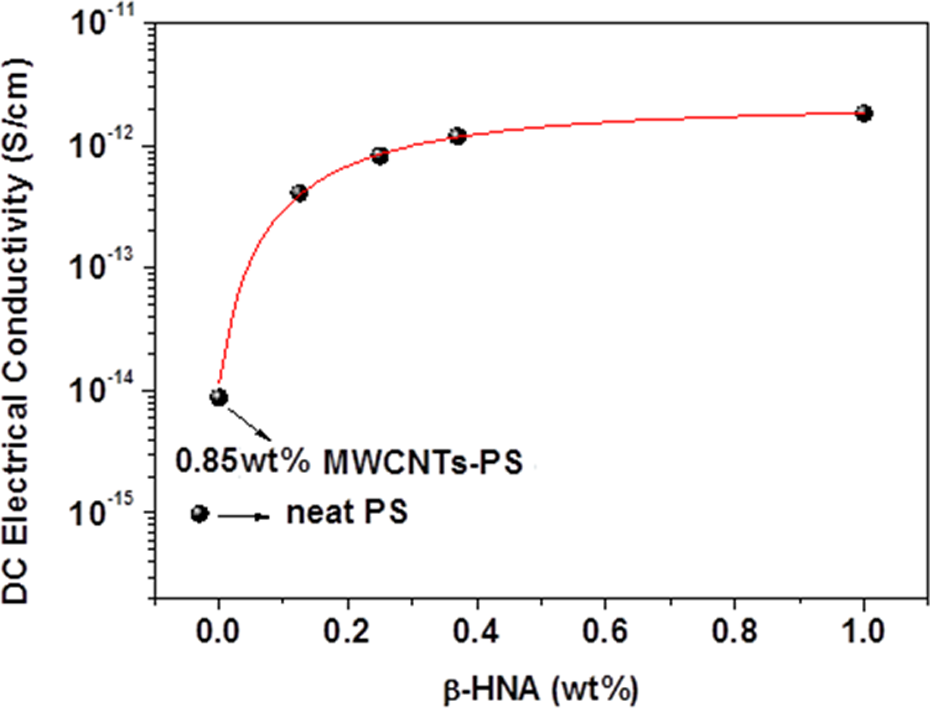
DC electrical conductivity versus β-HNA weight fraction of β-HNA-MWCNTs-PS composite films. DC: direct current; β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
The variations of Z, ε′, ε″, and σ AC with β-HNA weight fraction and applied frequency at room temperature are shown in Figures 3 to 5, respectively. Figures 6 and 7 illustrate the variation of Z and σ AC with β-HNA weight fraction, respectively. It is clear from Figures 3, 4, and 6 that addition of β-HNA to 0.85 wt% MWCNTs-PS composite reduces the Z values while increases ε′ and ε″ values of 0.85 wt% MWCNTs-PS composite. Besides, addition of β-HNA to 0.85 wt% MWCNTs-PS composite increases σ AC values up to three orders of magnitude, as shown in Figures 5 and 7. The obtained results, however, are well correlated with previous results indicating that addition of β-HNA to 0.85 wt%MWCNTs-PS composite enhances the dispersion in addition to the formation of MWCNTs-PS network.
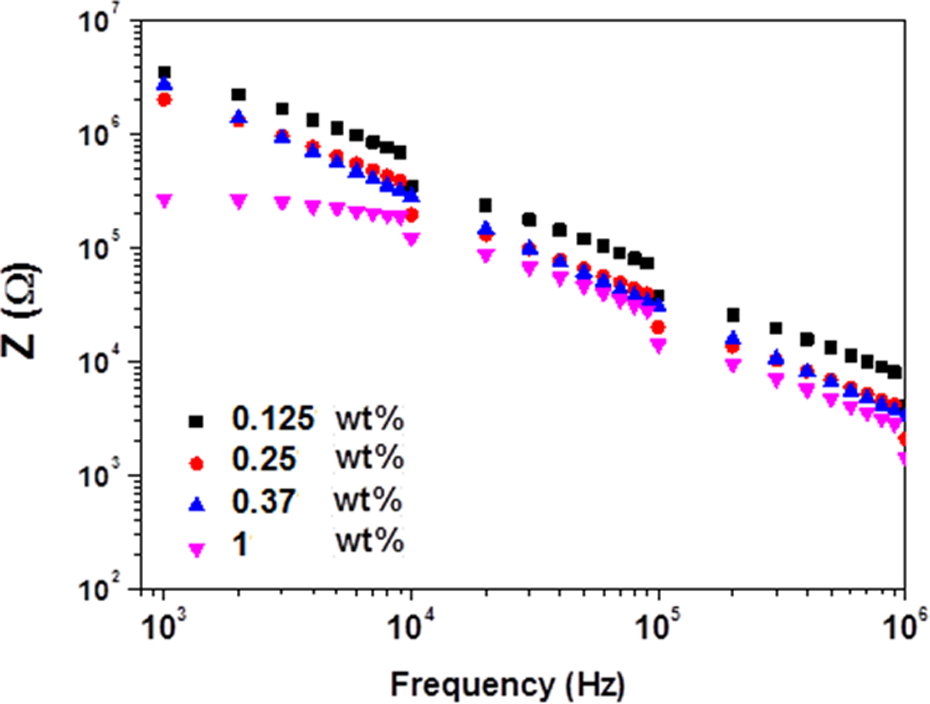
Dependence of impedance (Z) on applied frequency of β-HNA-MWCNTs-PS composite films. β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
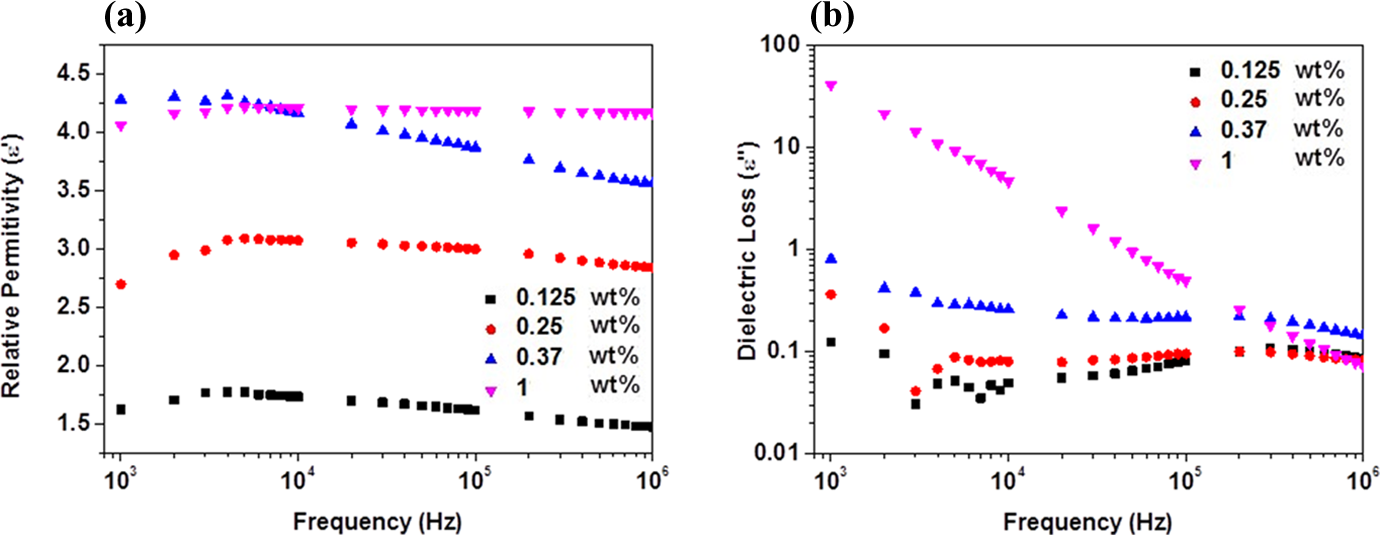
Dependence of (a) relative permittivity (ε′) and (b) dielectric loss (ε″) on applied frequency of β-HNA-MWCNTs-PS composite films. β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
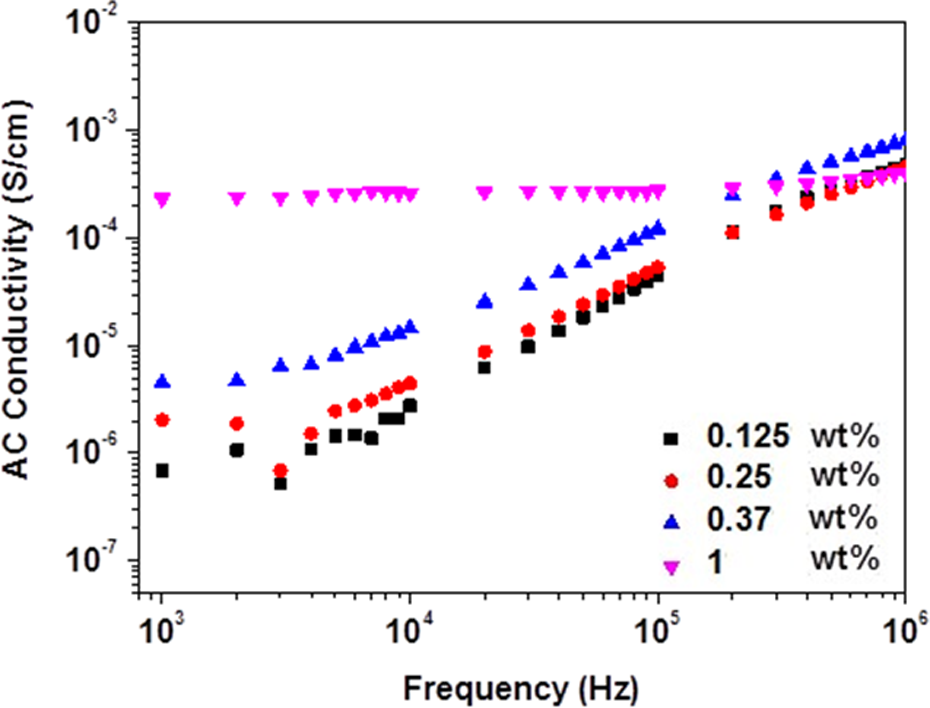
Dependence of σ AC on applied frequency of β-HNA-MWCNTs-PS composite films. σ AC: alternating current electrical conductivity; β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
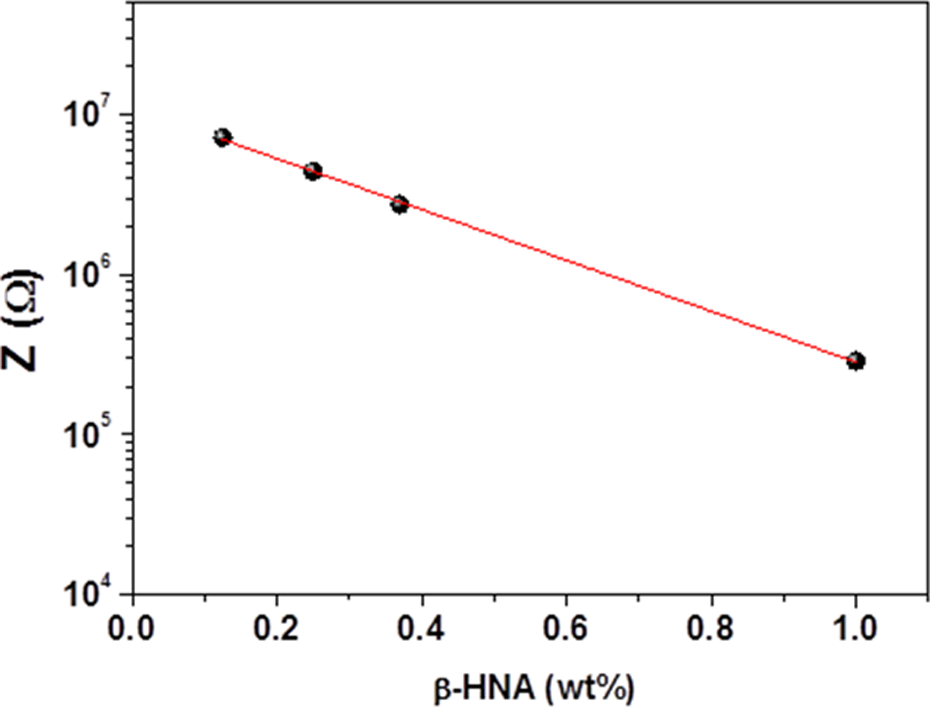
Variation of impedance (Z) with β-HNA weight fraction at 1 kHz of β-HNA-MWCNTs-PS composite films. β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
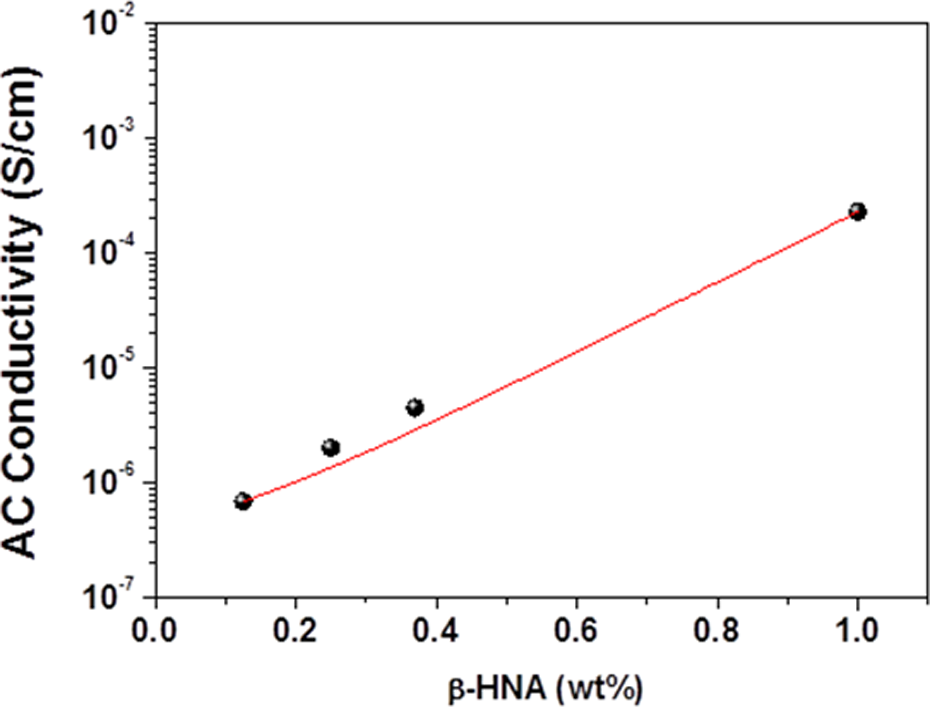
Variation of σ AC with β-HNA weight fraction at 1 kHz of β-HNA-MWCNTs-PS composite films. σ AC: alternating current electrical conductivity; β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
Total Z value that was measured for prepared composites was resolved into real (Z′) and imaginary (Z″) parts to construct effect of β-HNA addition on behavior of the Cole–Cole Z plot which will provide a strong evidence about the role of β-HNA addition on the formation of MWCNTs-PS network.
Figure 8 represents the Z Cole–Cole plot of β-HNA-MWCNTs-PS composite at fixed amount of MWCNTs (0.85 wt%) and different weight fractions of β-HNA at room temperature. It is obvious that the radius of the Cole–Cole plot of composite decreases with increasing β-HNA weight fraction which indicates that the distance between CNTs-CNTs in the β-HNA-CNTs-polymer network decreases with the β-HNA addition. 1 The obtained results, however, suggest that the addition of β-HNA in to composite system enhances the interfacial bonds in the β-HNA-MWCNTs-PS network system through the adsorption of β-HNA on MWCNTs surface and the creation of noncovalent bonds as will be confirmed through FTIR spectra in the coming paragraph.
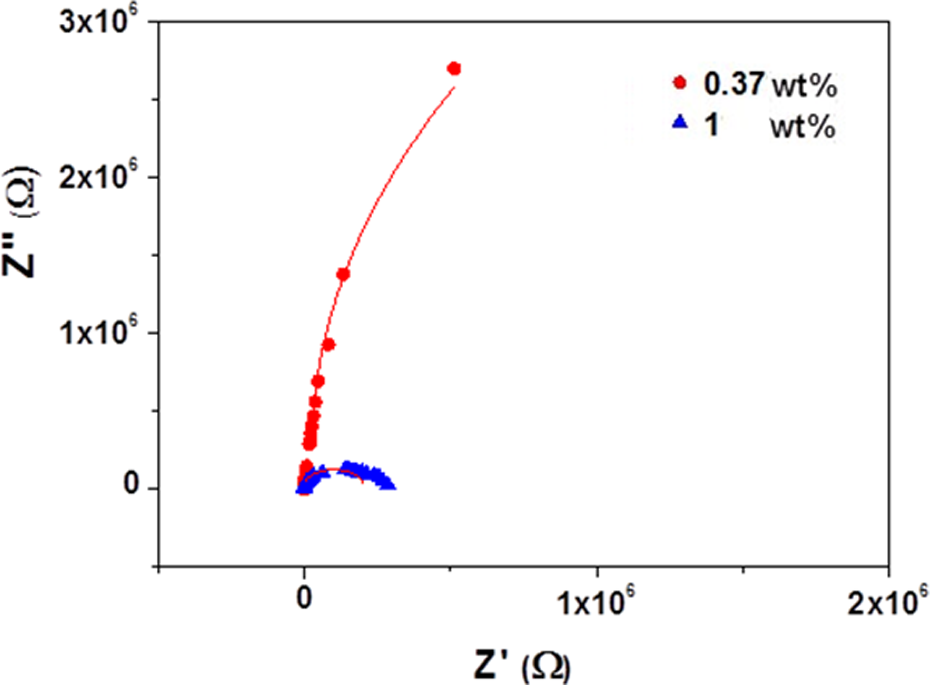
Impedance Cole–Cole plot of β-HNA-MWCNTs-PS composite films. β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
The relaxation behavior of the prepared composites show in all cases the existence of capacitor element and as β-HNA content increases the resistance element in these composites becomes more pronounced as shown in Figure 3. However, it will be very interesting here if we consider the Argand plot between imaginary part (M″) versus real part (M′) of electric modulus to recognize effect of β-HNA addition on the relaxation behavior of β-HNA-MWCNTs-PS nanocomposites. It was reported previously
25,28
–30
that if the Argand plot between imaginary part (M″) versus real part (M′) of electric modulus has semicircular behavior then the relaxation is due to conductivity relaxation process, if not, then it is due to viscoelastic relaxation (or polymer molecular relaxation). In the present work, the values of M′ and M″ were determined from:
where M′ is the electric modulus and M″ is the electric loss modulus.
The obtained results are presented graphically in Figure 9. It is clear that when the β-HNA content in the composites is <1.0 wt%, the plot does not follow semicircular behavior. This indicates that the relaxation process below the 1.0 wt% β-HNA is mostly due to polymer molecular relaxation process. While at 1.0 wt% β-HNA, the composite relaxation behavior is mostly due to charge conductivity relaxation. 25,28 –31
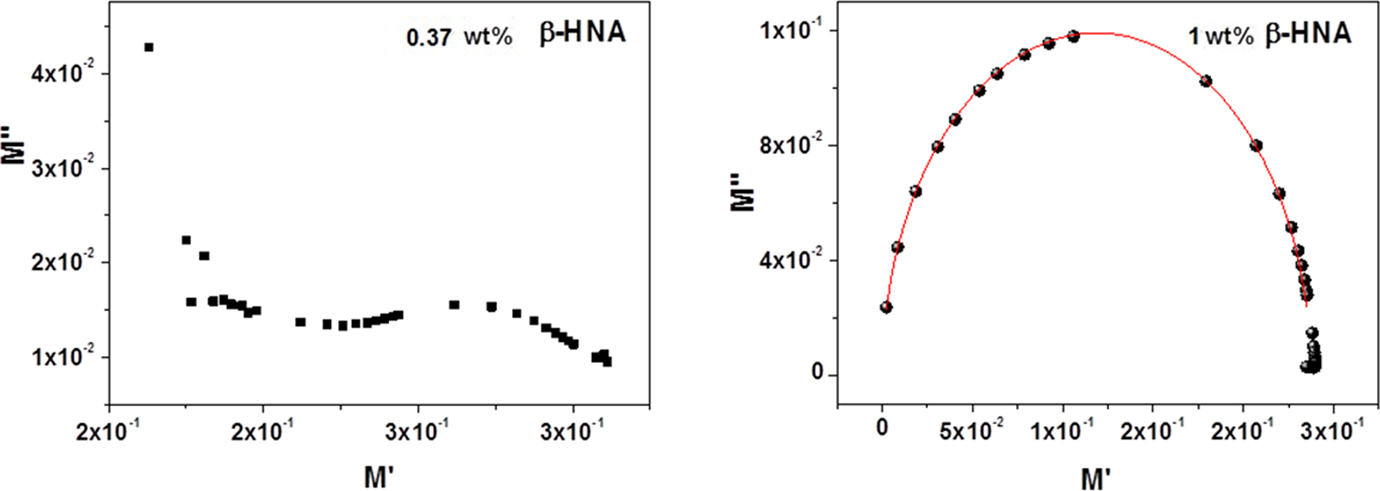
Argand plot of β-HNA-MWCNTs-PS composite films. β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
Mechanical properties
Figure 10 (a) to (c) shows the elastic modulus, yield stress, and tensile strength versus β-HNA weight fraction in MWCNTs-PS composites. Mechanical results indicate that the variation of mechanical properties for prepared composites depends on the amount of β-HNA that is incorporated into MWCNTs-PS composite. Upon addition of 1.0 wt% β-HNA to MWCNTs-PS composite, the elastic modulus, yield stress, and tensile strength of the composites compared with the neat PS increases by about 12.2, 17.1, and 10.0, respectively. Mechanical results are well correlated with our previous results, which means that addition of β-HNA up to 1.0 wt% to MWCNTs-PS composites improves the MWCNTs dispersion in the neat PS and enhances the interfacial bonding in the β-HNA-MWCNTs-PS network. 2,3,26,32 –37
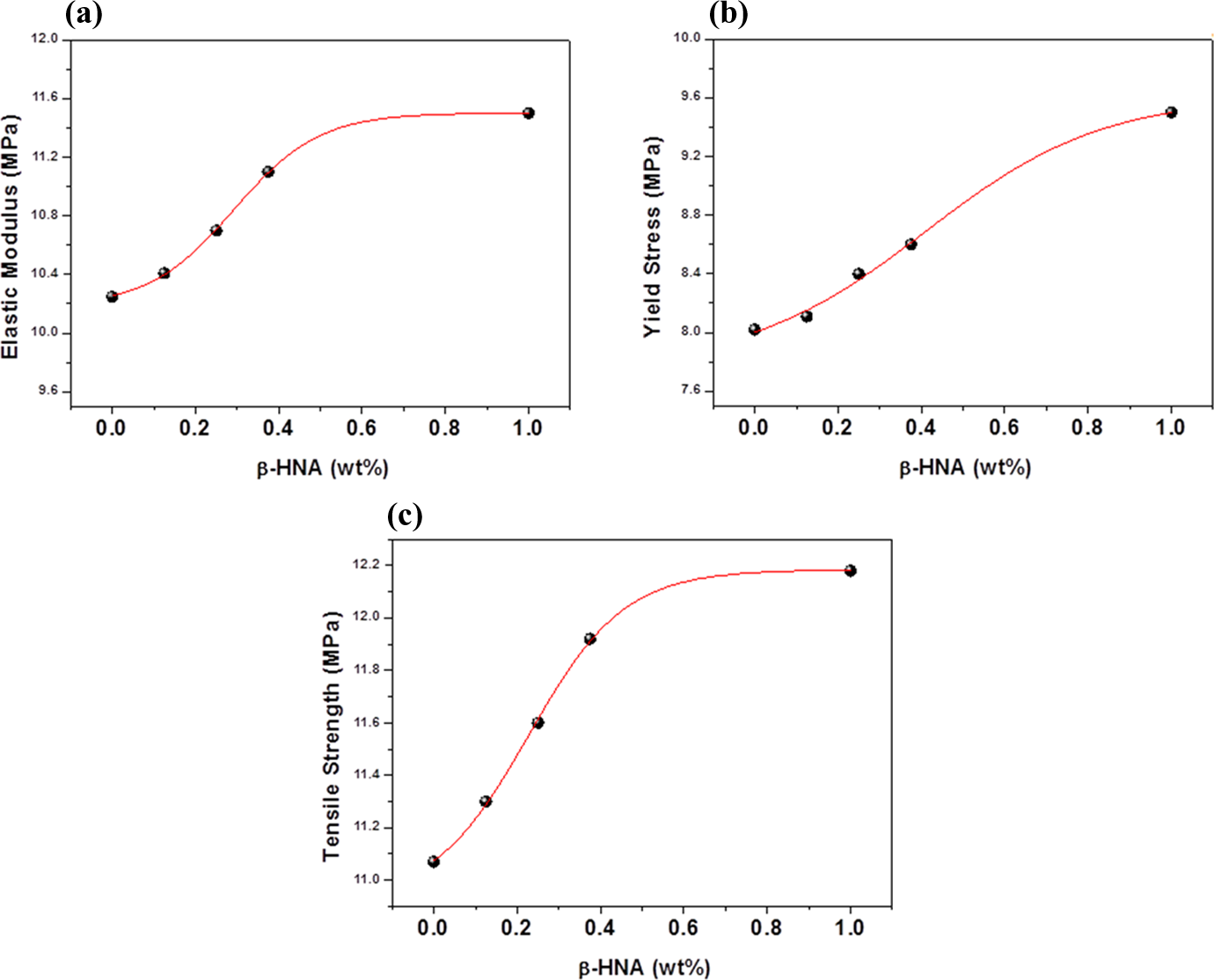
Variations of (a) elastic modulus, (b) yield stress, and (c) tensile strength with β-HNA weight fraction in β-HNA-MWCNTs-PS composites. β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
FTIR analysis
FTIR spectroscopic measurements, in conjunction with other spectroscopic techniques, has been instrumental in the structural elucidation of functionalized MWCNTs 38,39 and the presence of various functional groups in polymers. 1,32,33
The adsorption phenomenon of β-HNA on the surface of MWCNTs was verified by the IR spectrum (Figure 11).
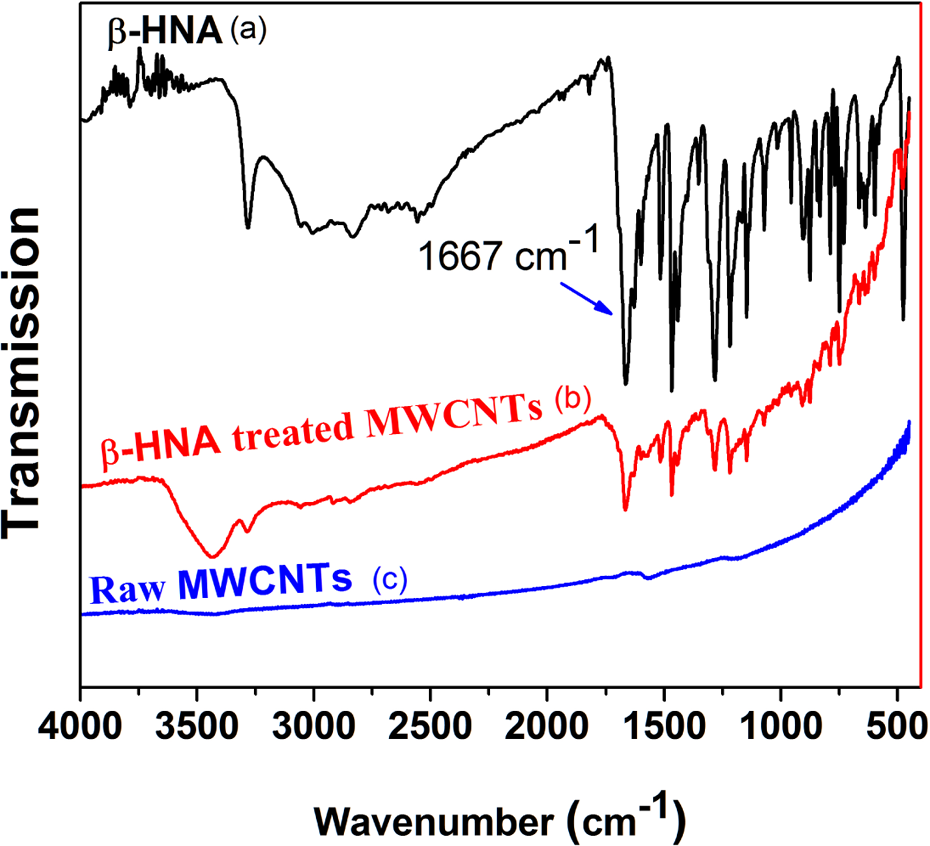
FTIR spectra for (a) β-HNA, (b) β-HNA-treated MWCNTs, and (c) raw (untreated) MWCNTs. FTIR: Fourier transform infrared; β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
Treated MWCNTs with β-HNA (Figure 11(b)) exhibited peaks for various anticipated stretching modes in the IR spectrum. Characteristic IR absorption peaks observed for functional groups in the treated MWCNTs include a broad peak at 3437 cm−1 corresponding to hydrogen bonded O–H stretch, 1669 cm−1 attributed to C=O stretch mode of the carboxylic group, 1601 and 1465 cm−1 assigned to aromatic C=C symmetric and asymmetric stretches respectively, 1281 cm−1 due to O–H bending deformation in the carboxylic group, 1147 cm−1 representing C–O stretching vibration of the carboxylic group and the peaks at 749 and 700 cm−1 indicating C–H deformation vibration band of benzene ring hydrogen and ring deformation vibration, respectively. Similar results for related functionalized MWCNTs have been previously reported in the literature. 39 Notably, absence of a peak in the region of 2400–2700 cm–1 indicates lack of O–H stretch arising from strongly hydrogen-bonded –COOH. 34
To gain further insight and verify our spectrum assignments, we opted to correlate our spectrum with the spectra of β-HNA and raw MWCNTs as shown in Figures 11(a) and 12(c), respectively. Pleasingly, we did not observe the presence of hydroxyl groups (–OH) on the surface of our raw MWCNTs (Figure 12(c)), which is believed to happen as a result of either oxidation during purification or from ambient atmospheric moisture. 35 The IR spectrum of neat β-HNA showed characteristics absorptions peaks at 3279 and 1667 cm−1 corresponding to O–H stretch and C=O stretch mode of the carboxylic group, respectively. The peaks at 1600 and 1467 cm−1 are assigned to aromatic C=C stretches, while at 1281 and 1146 cm−1 are due to O–H bending deformation and C–O stretching vibration of the carboxylic group, respectively. On the basis of comparative spectroscopic analysis (Figure 11(a) to (c)), it becomes evident that the sidewalls of MWCNTs are covered with functional moieties like carboxyl (–COOH) and hydroxyl (–OH) groups, resulting into an enhanced dispersion and electrical properties of MWCNTs.
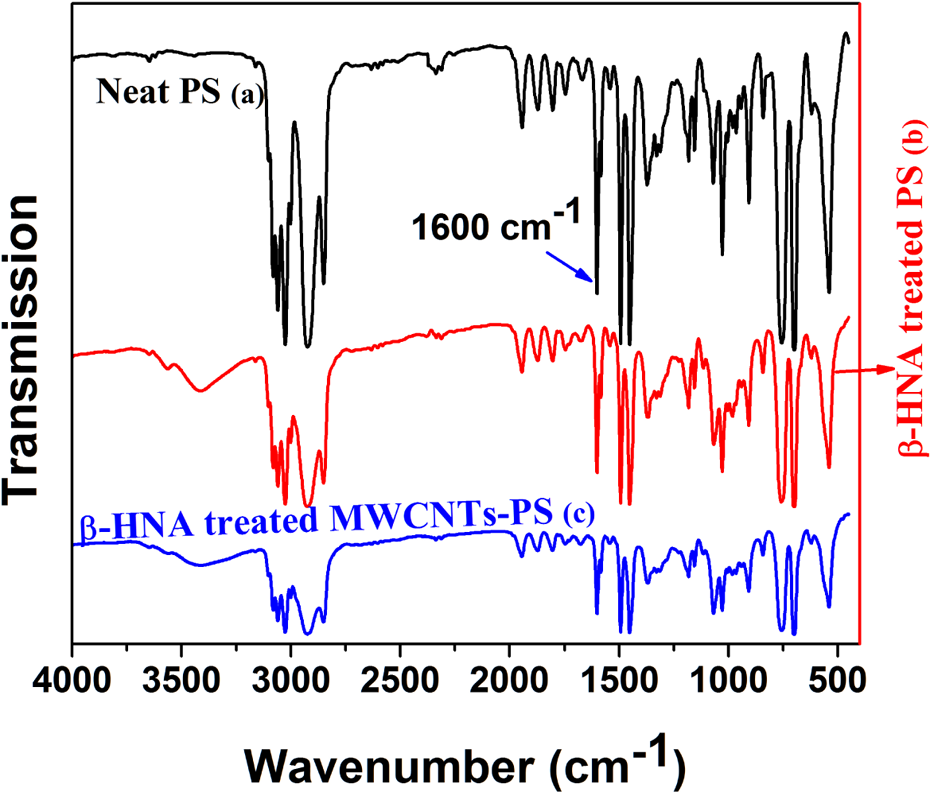
FTIR spectra for (a) neat PS, (b) β-HNA-treated PS and (c) β-HNA treated MWCNTs-PS composite. FTIR: Fourier transform infrared; β-HNA: β-hydroxynaphthoic acid; MWCNTs: multiwalled carbon nanotubes; PS: polystyrene.
The IR spectrum of neat PS sample (Figure 12(a)) showed absorption bands at 3060 and 2926 cm−1 corresponding to aromatic and aliphatic C–H stretching, respectively. The peaks at 1600 and 1493 cm−1 are assigned to aromatic C=C symmetric and asymmetric stretches. The C–H deformation vibration band of benzene ring hydrogen appeared at 753 cm−1, while ring deformation vibration was observed at 703 cm−1. This spectroscopic assignment is in good accordance with the reported in the literature. 36
The FTIR spectrum of PS treated with β-HNA is presented in Figure 12(b). The presence of absorption peaks at 3410, 3060, 2923, 1601, 1493, 1367, 1069, 758, and 701 cm−1 corresponds to various anticipated stretching vibrations as mentioned in the case of neat PS (Figure 12(a)) and β-HNA (Figure 11(a)) samples. It is worth mentioning that the peak at 1660 cm−1 attributed to C=O stretch mode of the carboxylic group present in the neat β-HNA sample (Figure 11(a)) was not observed in the IR spectrum of β-HNA-treated PS composite (Figure 12(b)).
Ab initio quantum–chemical calculations reveal that aromatic moieties are prone to act as proton acceptors and their delocalized π electronic cloud has a tendency to establish noncovalent interactions such as van der Waals clusters with molecules like water and formic acid, acting as proton donors. 26 Considering the documented theoretical studies, it is plausible to rationalize that the aromatic system of PS forms an electrostatically supported complex with the highly conjugated β-HNA molecule that acts as a proton donor. Consequently, an intermolecular complex formation is established between PS and β-HNA. The IR spectrum obtained from β-HNA-treated MWCNTs-PS composite (Figure 12(c)) shows the same trend as found for the spectrum of β-HNA-treated PS (Figure 12(b)), suggesting a noncovalently bonded interaction between β-HNA and PS in nanocomposites.
Conclusions
In this work, role of β-HNA on the electrical conductivity, dielectric properties, dielectric relaxation, and morphology of MWCNTs-PS nanocomposite with 0.85 wt% MWCNTs and different β-HNA (0–1 wt%) contents were studied. Addition of β-HNA to composite system enhances the dispersion of MWCNTs in the neat PS, the formation of MWCNTs-PS network, and the electrical conductivity of prepared composites by about three orders of magnitude. Incorporation of β-HNA into 0.85 wt% MWCNTs-PS composite (near the percolation threshold of MWCNTs-PS) reduces the Z values while increases the ε′ and ε″ of composite. Dielectric relaxation results showed that at the domain frequency range, the relaxation process below 1.0 wt% β-HNA is mostly due to polymer molecular relaxation process, while at 1.0 wt% β-HNA, the composite relaxation behavior is mostly due to charge conductivity relaxation. Mechanical results showed that addition of 1.0 wt% β-HNA to MWCNTs-PS composite, the elastic modulus, yield stress, and tensile strength of the composites compared with the neat PS increases by about 12.2, 17.1, and 10.0%, respectively. Mechanical results are well correlated with electrical and morphology results, which means that the addition of β-HNA up to 1.0 wt% to MWCNTs-PS composites improves the MWCNTs dispersion in the neat PS and enhances the interfacial bond in the β-HNA-MWCNTs-PS network. FTIR spectra of representative samples establish not only the adsorption of β-HNA molecules onto the surface of MWCNTs but also indicate the presence of intermolecular complex formation between PS and β-HNA in nanocomposite films.
Footnotes
Funding
The authors would like to thank the Deanship of Scientific Research at King Faisal University, Al-Ahsa, Kingdom of Saudi arabia, for providing financial support for this research, which has a project ID number: 140011