Abstract
Maleated thermoplastic starches (MTPS) were prepared by reactive extrusion, with corn starch as raw material, glycerol as the plasticizer, and maleic anhydride (MAH) as esterification agent. The influence of the amount of MAH on MTPS properties was studied. With thermoplastic starch (TPS) as contrasting sample, Fourier transform infrared spectroscopy results displayed that esterification reaction successfully occurs between corn starch and MAH. The degree of substitution (DS) results showed that the DS of MTPS increased along with the increasing amount of MAH. X-Ray diffractometry results showed that esterification reaction resulted in the decrease of crystallinity of TPS, indicating that the thermoplastic property of starch was improved. The greater the proportion of MAH, the more decreased is the degree of crystallinity of MTPS. Differential scanning calorimetric results showed that the melting temperature and enthalpy of melting decreased with increasing the amount of MAH. The thermal weight loss initial temperature of MTPS was below TPS, and thermal weight loss rate was greater than TPS. The contact angle of MTPS was greater than TPS, while the water absorption of MTPS was less than TPS. Further, with increasing of MAH content, the contact angle increased, while water absorption decreased, which indicated that the hydrophobic was gradually improved.
Keywords
Introduction
Starch is a type of natural polymer material, which has many advantages such as extensive sources, low price, biodegradable, and renewable. 1 However, the thermoplastic of natural starch is poor, hard to be processed into starch plastics. 2,3 Moreover, starch is a hydrophilic polymer, 4 which makes thermoplastic starch (TPS) susceptible to moisture attacks and significant changes in dimensional stability and mechanical properties. 5,6 Sagar and Merril’s research 7 suggested that esterification modification made side chain of starch grow, and thermoplastic property was improved. On the one hand, the introduction of ester groups plays a role of internal plasticization and reduces the degree of crystallinity of starch, so that the small molecule of plasticizer can penetrate well into the starch molecule for plasticization. On the other hand, ester group can improve the hydrophobic of starch-based materials. Maleic anhydride (MAH) has high reactivity on preparing esterified starch.
Common methods for the preparation of MAH esterified starch are wet and organic solvent method. In the wet method, the subsidiary reaction of hydrolysis made lower efficiency of esterification reaction. Degree of substitution (DS) of the product prepared by organic solvent method is not so high, while the production cost is high; what is more, the environment could be polluted more seriously.
Reactive extrusion is a new technology used for preparing modified starch in recent years. Although reactive extrusion has not been widely used, it can make native starch be of diversification, functionalization, and continuous production. At the same time, it is economic and simple for operating. Therefore, this technology has great potential in the production of modified starch, and 8 people pay more and more attentions on it. In order to improve the reaction efficiency and reduce environmental pollution, this article introduced reactive extrusion to prepare maleated thermoplastic starch (MTPS). Reaction equation of starch and MAH via reactive extrusion is shown in Figure 1. Under the conditions of plasticizer and heating, corn starch was gelatinized. At the same time, melting temperature of MAH is 52.8°C. In the process of reactive extrusion, gelatinized starch and molten MAH was easier to react. The study on the preparation of MTPS by reactive extrusion process played an important role in improving the thermoplastic and water resistance of starch. Furthermore, it could provide an important theoretical basis for the development and application of starch-based biodegradable plastics.
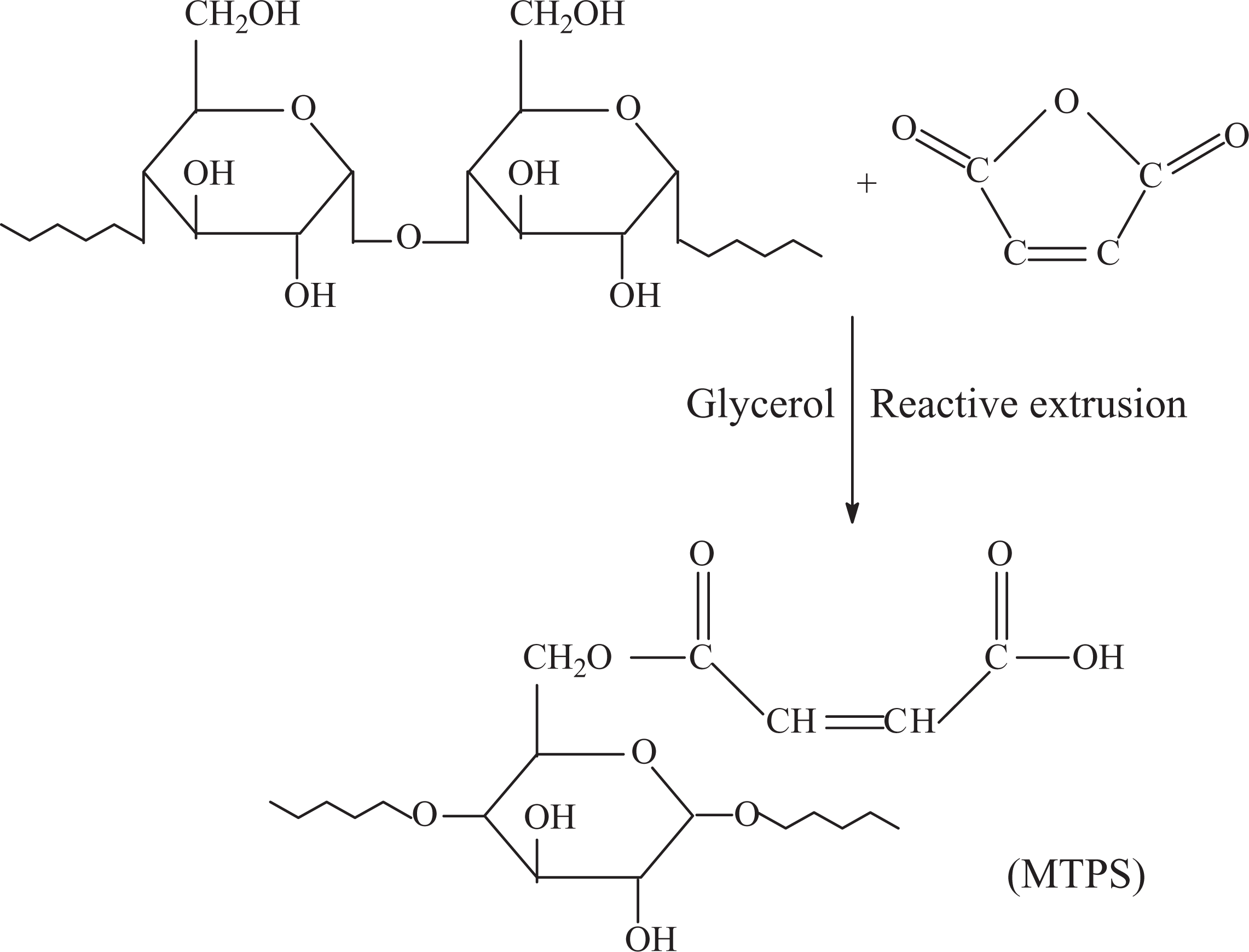
Reaction between starch and maleic anhydride by reactive extrusion.
Experimental
Materials and methods
Materials
Corn starch was obtained from Dacheng Corn Development Co. Ltd (Changchun, Jilin, China). MAH and glycerol were purchased from Tianjin Hengxing Chemical Preparation (Tianjin, China). All chemical products were analytical reagent grade.
Preparation of MTPS
Corn starch was dried at 50°C for 24 h to eliminate the moisture. MAH (according to the ratio in Table 1) was dissolved in a small amount of acetone, respectively sprayed into 100 g dry corn starch, and then evenly dispersed by stirring. After acetone volatilization, add 30 g glycerol to starch, mix evenly and place in a sealed plastic bag for 18 h. The plasticization starch was manually fed into a single-screw plastic extruder (SJ-25(s), Harbin Special Plastic Products (Harbin, China); screw ratio length/diameter = 25:1) with a screw speed of 15 r min−1. The temperature profile of the extruder barrel from the feed zone to the die was 100, 110, 120, and 90°C. The die was a rectangular sheet with one hole of 2 × 3 mm2. The extruded samples were conditioned in sealed polyethylene bags to maintain stable moisture content. Under the same conditions, mixing native starch and glycerol to prepare TPS as a contrasting sample.
The addition proportion of experimental reagents.

MAH: maleic anhydride.
Performance characterization
Fourier transform infrared spectroscopy
MTPS was dried, crushed, and washed with acetone to wash away the unreacted MAH. After drying, samples were scanned for FTIR analysis using Magna-IR 560 ESP type FTIR (USA) spectrometer produced by Thermo Nicolet Corporation had a resolution of 4 cm−1 was used to characterize MTPS from 500 to 3800 cm−1. The samples were made up by pressing along with potassium bromide and scanned 40 times using the transmission method.
Determination on the DS
The unreacted MAH were removed according to test by FTIR method.
9
The DS of MTPS was determined according to the method of Siggia. Dry MTPS (1 g) was weighed accurately and placed in a 250 ml conical flask. Then 10 ml of 75% ethanol solution in deionized water was added, and 10 ml of 0.5 M aqueous sodium hydroxide solution was added. The stoppered conical flask was agitated, warmed to 30°C, and stirred for 30 min. The excess alkali was back titrated with a standard 0.5 M aqueous hydrochloric acid solution. A blank titration using native starch was carried out. The DS was calculated as follows:
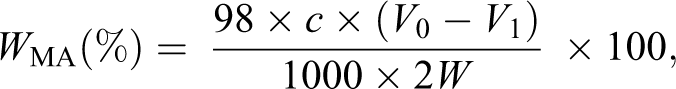
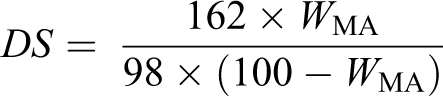
X-Ray diffractometry
The XRD patterns of the native starch, TPS, and MTPS were obtained using an x-ray diffractometer (Rigaku D/max220, Japan) at 40 kV and 30 mA with 0.154 nm copper Kα radiation (nickel filter). The scanning range was 2θ = 10–40° at 0.02° step size with a scan speed of 4 s step−1.
Differential scanning calorimeter
Effect of MAH and its proportion on starch-pasting properties were determined using DSC. The test samples were mixed native starch, MAH (1, 2, and 3%) and glycerol. Native starch and glycerol mixture as a comparison sample were sealed for 18 h. Sample pans were heated from 30 to 180°C with a heating rate of 5°C min−1 and an argon flow of 30 ml min−1.
Thermogravimetric analysis (TGA)
TPS and MTPS were cut into small pieces for TGA test. The TGA measurements were conducted using a thermal analysis system (TGA 209 F3) produced by NETZSCH Co. (Germany). Samples were loaded into an aluminum oxide pan. Experiments were tested from 40 to 500°C with a heating rate of 10°C min−1 and an argon flow of 30 ml min−1.
Contact angle
The CAs for the surfaces of TPS and MTPS were tested by a contact angle analyzer (Model OCA20, Dataphysics, Germany). The samples were adhered to a movable sample stage horizontally; then about 2 µl probe water was introduced on the surface of the sheet using a microsyringe.
Water absorption
According to National Standard of China GB1034-98, the bars of samples were cut into small pieces with size of 10 × 10 mm2, and the pieces were vacuum dried at 50°C for 48 h. The dried pieces were weighed immediately and then soaked in distilled water at room temperature. After 24 h, the specimens were removed from distilled water, blotted dry with filter paper, and then weighed again. Data were recorded as averages of three specimens. Water absorption of the starch plastic was calculated as follows:
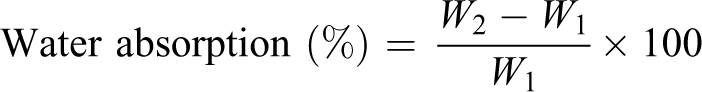
Mechanical properties
The mechanical properties of TPS and MTPS, such as tensile strength and elongation at break, were evaluated. The mechanical testing (ISO 6239-1986 standard) of specimens was determined in a CMT-5504 Materials Testing Machine (Sans, Shenzhen, China) at a crosshead speed of 25 mm min−1. To promote the failure of specimens toward the center of the film, they were cut in a dumbbell shape. The data were averages of five specimens. The tensile strength (TS, in mega pascal) was calculated by dividing the peak force with the cross-sectional area of the sample. The elongation was obtained by dividing the change in length achieved at breakage by the original specimen length (0.025 m).
Results and discussion
Proof of esterification reaction
It can be known form Figure 1 that starch and MAH could be occur esterification reaction, resulting in introduction of the ester bond and carbon–carbon double bonds into starch. In order to prove this reaction, FTIR was used to analyze the group transformations between TPS and MTPS, the result is shown in Figure 2. 10,11 MTPS not only contained the characteristic absorption peaks of TPS but also appeared the absorption peak of ester bond (C=O) at 1720 cm−1 12 and the absorption peak of C=C at 1580 cm−1. The sample was washed with acetone before the treat, so the unreacted MAH had been removed from MTPS sample. This showed that the absorption peaks C=O and C=C were derived from MTPS. Therefore, the FTIR result suggested that the esterification reaction of starch and MAH had successfully occurred by reactive extrusion.
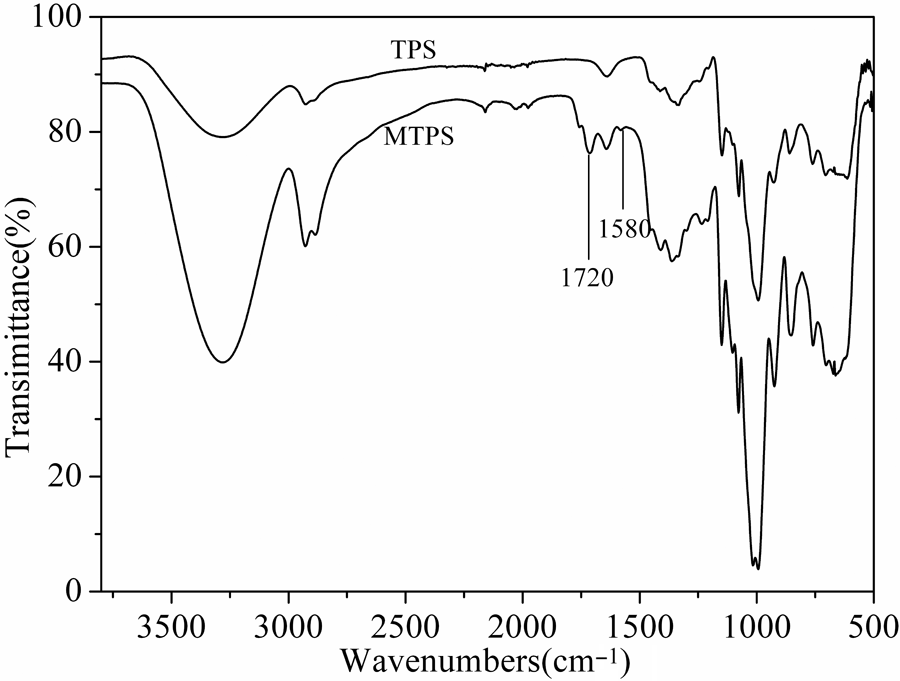
The FTIR spectrum of TPS and MTPS.
Effect of the amount of MAH on the DS of MTPS
The influence of the amount of MAH on the DS of MTPS was tested, and the result was shown in Figure 3. Each anhydroglucose (except the branched part) units of starch molecular chains had three hydroxyl groups, which could react with three MAH molecule in theory. And increasing the amount of MAH could promote the reaction equilibrium to the product orientation. It can be seen from Figure 3, the DS of MTPS were very small due to a smaller proportion of MAH. However, with the increasing amount of MAH, the DS increased gradually. It means that more hydroxyl groups were substituted by MAH molecule.
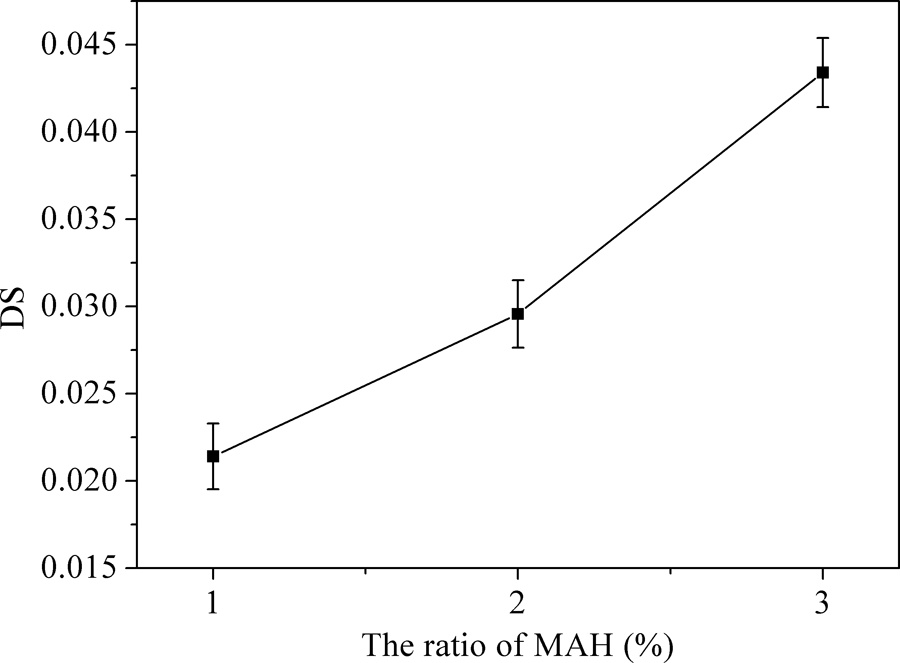
The degree of substitution of MTPS with different amount of MAH.
The change of crystalline structure
Starch is a natural semicrystalline biopolymer of
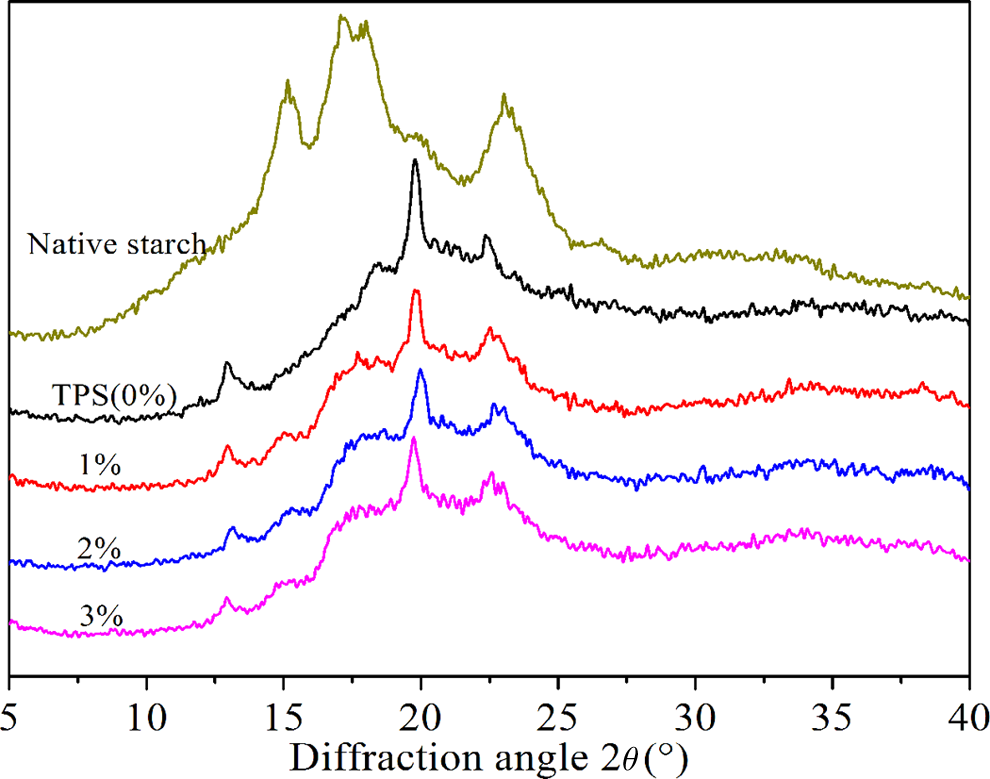
XRD diffraction patterns of TPS and MTPS (1, 2, and 3%).
With the increasing amount of MAH, XRD diffraction peak intensity of MTPS decreased. According to the report of Frost et al., 20 it can be calculated that the degree of crystallinity of TPS was 11.03%, whereas 10.11% (1%), 9.32% (2%), and 8.61% (3%) for MTPS. This rule showed that the degree of crystallization of esterified starch decreased with gradual increase in DS. That was to say, with the increasing amount of MAH, the thermoplastic of starch increased gradually. By increasing the amount of MAH, the DS of MTPS increases that result in increasing the distance between the starch molecular chains, and starch molecular chains arranged looser.
Effects of esterification on starch thermoplastic
Plasticization of starch can be regarded as melting under the condition of a shearing action in the presence of a plasticizing agent. DSC is a common method for researching melting properties. 21 The starch melting can also be used to characterize the degree of difficulty for starch plasticization. By comparing the DSC endothermic peak area, the change of melting enthalpy after added different proportion of MAH can be obtained to analyze MAH and its proportion effect on melting degree of difficulty of corn starch. Starch/MAH(1, 2, and 3%)/glycerol were mixed evenly, sealed and placed for 18 h, and then scanned by DSC, with starch and glycerol mixture as contrasting sample. The results were shown in Figure 5. The thermal transitions temperature of the samples were defined as To (onset), Tp (peak), and Tc (conclusion), and the enthalpy of melting was referred to as ΔH.
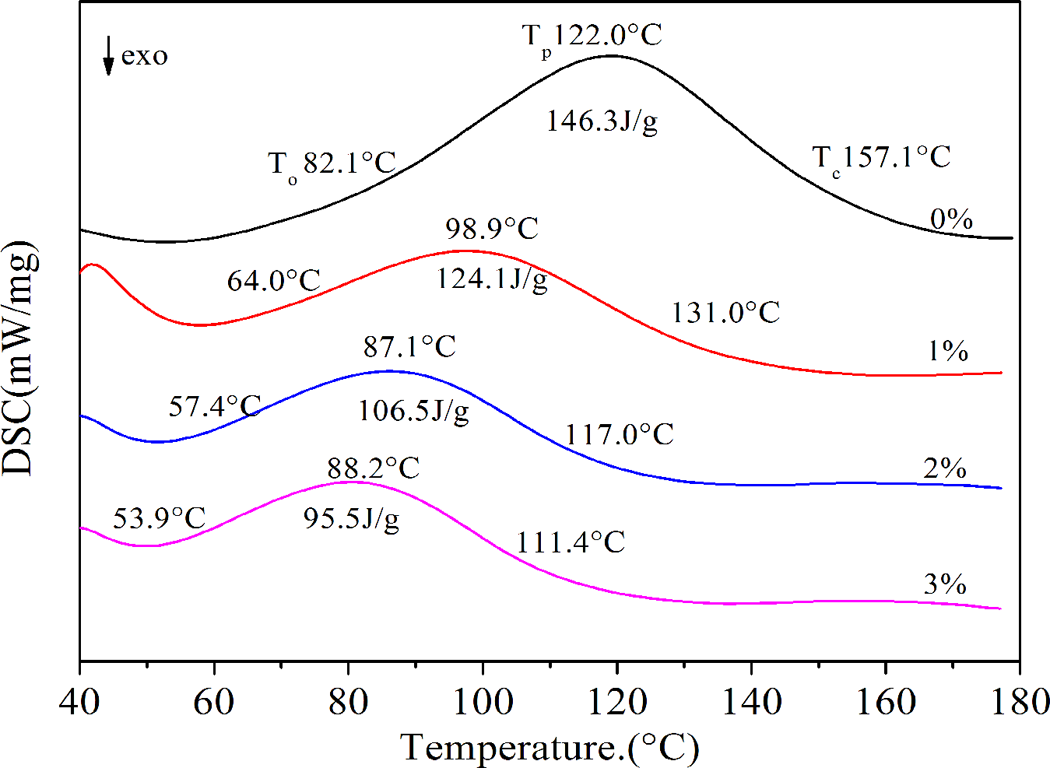
The DSC curves of plasticizing starch powder.
It can be seen from Figure 5 and Table 2 that MAH has important influence on the endothermic peak type of DSC curve. After adding the MAH, T0, Tp, and Tc of starch melting temperature all shifted to lower temperature. And with the increase in the ratio of MAH, DSC melting temperature also gradually shifted to lower temperature. This result showed that starch melting temperature decreased along with the increasing amount of MAH.
The DSC analysis dates.

DSC: differential scanning calorimeter; T0: thermal transition temperature of the samples at onset; Tp: thermal transition temperature of the samples at peak; Tc: thermal transition temperature of the samples at conclusion; ΔH: enthalpy of melting.
DSC endothermic peak width and area were obviously difference. After adding the MAH, both melting peak width and area were decreased. Adding MAH can diminish melting enthalpy of corn starch, made starch easier melting. The melting temperature of MAH is 52.8°C, so MAH would be melted and reacted with corn starch when scanned by DSC. The crystal structure of starch was destroyed. At this time, small molecular plasticizer can penetrate into the starch molecules easily, so the thermoplastic property of corn starch was improved. With the increasing amount of MAH, starch melting peak area gradually decreases. This was because the increasing of MAH content, the degree of crystallinity of MTPS decreased more, under the action of heat, resulting in easier melting. It also showed that, the thermoplastic performance of starch was improved with increasing the content of MAH.
Thermal analysis
The TGA curves of TPS and MTPS (1, 2, and 3%) were shown in Figure 6. TGA curves were basically the same before the temperature of 200°C, 22 which mainly was the water and plasticizer partial loss in the system. 23 When achieving the decomposition temperature, the decomposition initiation temperature of MTPS was lower than TPS. Finally, the thermal weight loss of MTPS was greater than TPS. This phenomenon was attributed to the fact that the crystalline structure of MTPS has been damaged to some extent after esterification reaction. The MTPS chains arranged were looser than TPS, which weaken the force between the chains and extended the distance between the molecules. Therefore, under the action of heating, MTPS was easier decomposed than TPS. This result was in common with that of XRD analysis. With increasing the amount of MAH, thermal decomposition temperature of MTPS decreased. This was also related to the crystalline structure of MTPS, because the degree of crystallinity decreased with the amount of MAH increasing.
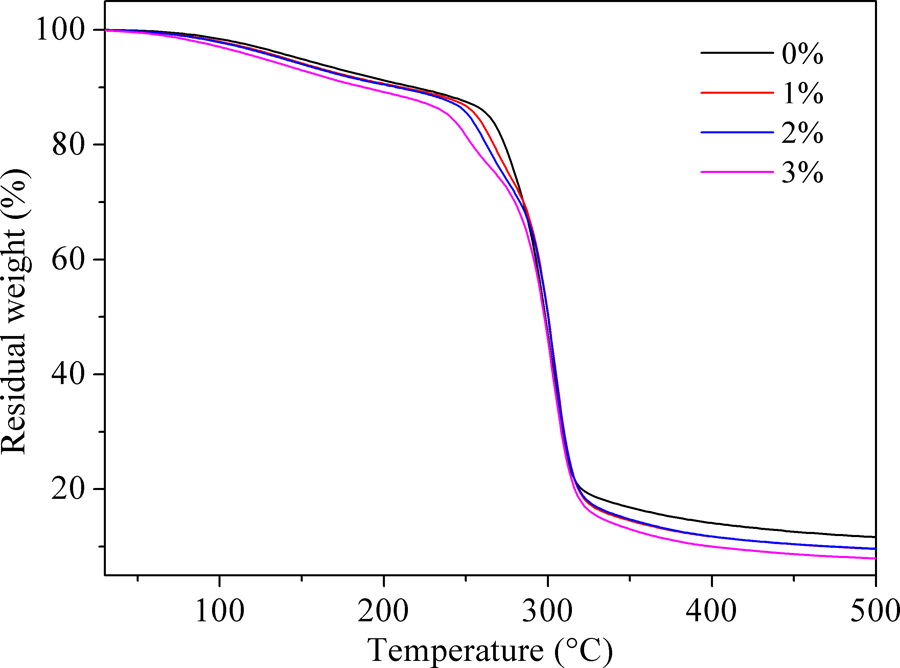
The TGA curves of TPS and MTPS (1, 2, and 3%).
The change of CA
The CA is the included angle formed by the tangent line of the droplet out layer and the solid surface. CA of water usually reflects the hydrophilic/hydrophobic character of the surface sample. Generally, 24,25 the larger the CA, the better hydrophobicity of materials. In theory, if MTPS was connected to the hydrophobic of ester bond, hydrophobic will improve. In order to verify this hypothesis, the surface contact angle of TPS and MTPS (1, 2, and 3%) were tested, with results as shown in Figure 7. It can be obviously found in Figure 7 that CAs of MTPS were larger than those of TPS. The results of CA showed that native starch was grafted to the hydrophobic ester bond by esterification, namely replacing part of the hydrophilic hydroxyl groups in the starch molecular chains. Therefore, the hydrophobic of MTPS was improved. With increasing the amount of MAH, the number of C=O connected to starch was increased instead of hydroxyl groups, so that the CA gradually increases.
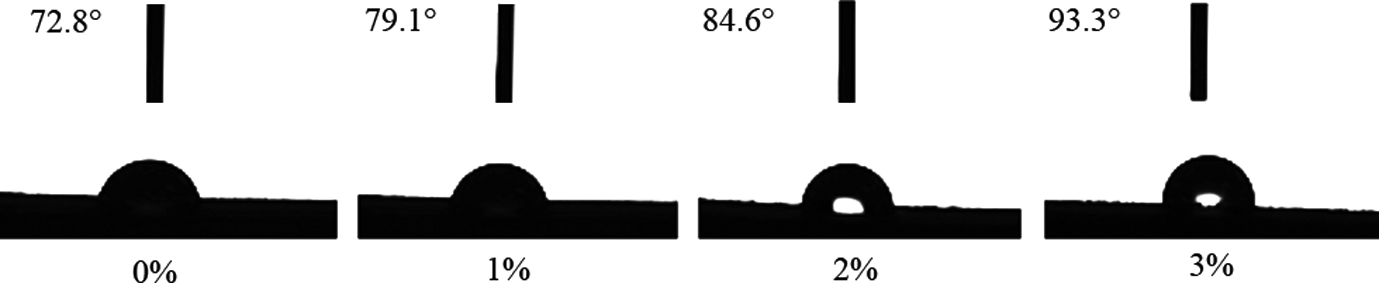
The contact angle changes of TPS and MTPS (1, 2, and 3%).
Water absorption
In order to further prove that the esterification modification can improve the hydrophobic of MTPS, the water absorption of TPS and MTPS (1, 2, and 3%) were measured. Results of water absorption were presented in Figure 8. The water absorption of MTPS was significantly less than TPS, proving that esterification modification can significantly improve the water resistance of TPS. Starch is a natural hydrophilic polymer, with a large number of hydroxyl groups in the macromolecular internal. TPS was hydrophilic because of the hydroxyl groups exited in the molecule. MAH molecules contained hydrophobic ester bond (C=O). When esterification reaction via reactive extrusion occurs in starch and MAH, part of the hydroxyl groups in starch molecule was substituted by hydrophobic ester bond. Hydrophilic hydroxyl groups decreased, and connected up the ester hydrophobic. Therefore, MTPS decreased the water absorption of starch material, improving the water resistance of TPS. It was the same as the CA analysis, that increasing the amount of MAH improved the hydrophobic property of MTPS, so water absorption was decreased gradually.
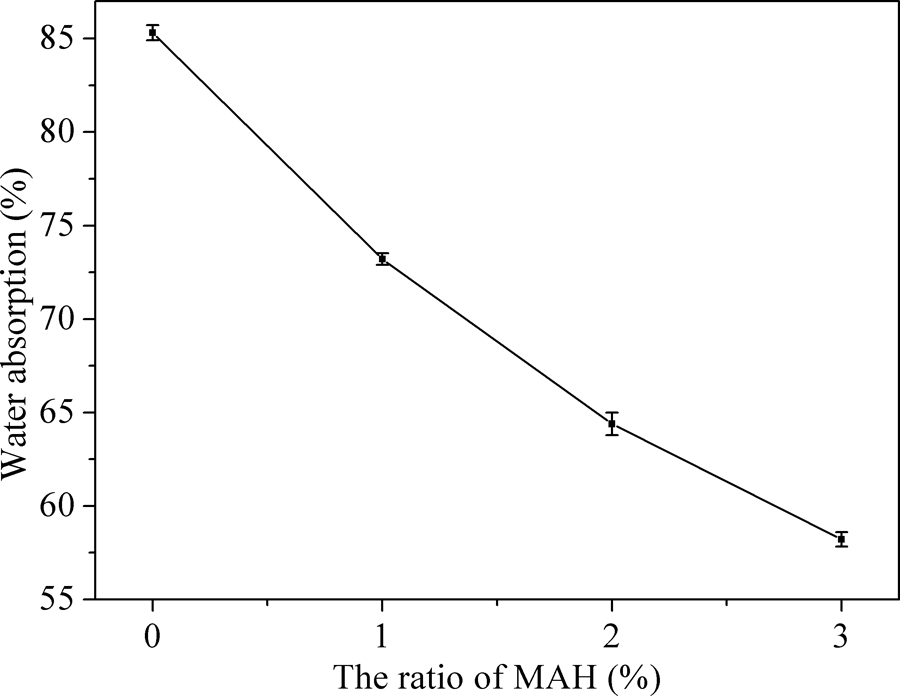
Water absorption of TPS and MTPS (1, 2, and 3%).
Mechanical properties
The tensile strength and elongation at breakage of TPS and MTPS (1, 2, and 3%) were shown in Figure 9. The tensile strength of MTPS was lower than TPS, but the elongation at break was larger than TPS. From XRD analysis, we know that the degree of crystallinity of MTPS decreased after esterification modification, which result in intermolecular force decreases, and leads to the decreasing of tensile strength. The intermolecular distance of MTPS was increased with the decreasing of degree of crystallinity, which results in flexibility of MTPS molecules improved, so the elongation at break was increased. With the increasing MAH amount, the degree of crystallinity of MTPS decreased gradually. So, the tensile strength decreased and the elongation at break increased with the increasing of MAH amount. We had found from other studies, the introduction of hydrophobic ester groups into MTPS can be obviously improved the interfacial compatibility between MTPS with other hydrophobic materials, thus greatly improved its mechanical properties.
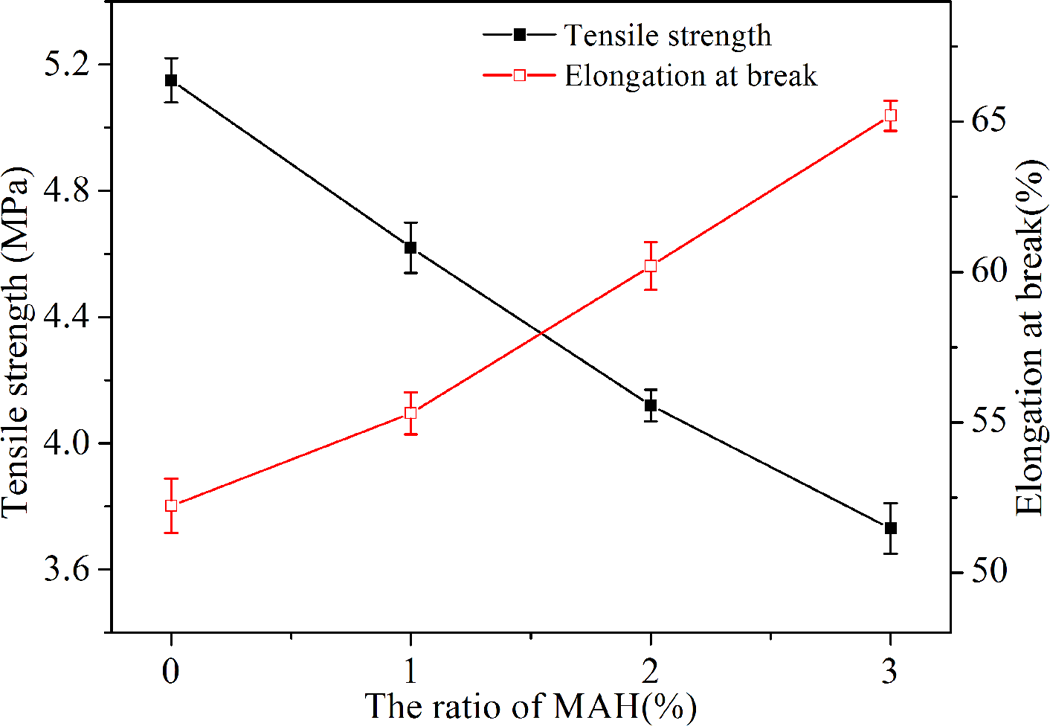
Mechanical properties of TPS and MTPS (1, 2, and 3%).
Conclusions
In summary, MTPS can be successfully prepared by reactive extrusion. Under the effect of temperature and pressure of extruder, melted starch, and the molten MAH could occurred esterification reaction. The DS of MTPS increased with increasing the amount of MAH. Esterification reaction made the crystalline structure of TPS was subject to a certain degree of damage, while degree of crystallinity decreased and thermoplastic improved. The addition of MAH was conducive for starch melting, so starch was easier to plasticize. Both XRD and DSC results showed that the thermoplastic of esterification starch was improved with the increasing DS. Esterification made hydrophilic hydroxyl of starch molecular chains replaced by hydrophobic ester bond, so that the CA of the MTPS was significantly greater than TPS, and water absorption was smaller than TPS. Therefore, the esterification reaction can significantly improve the water resistance of starch materials. Water absorption and CA test also showed that the hydrophobic of MTPS was increased with DS increasing. With the increasing MAH amount, the tensile strength of MTPS was decreased, while elongation at break was increased.
Footnotes
Acknowledgements
Special thanks are given to outstanding doctoral dissertation foster support and National Natural Science Foundation of China (No. 31200442).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was supported by Fundamental Research Funds for the Central Universities (DL12EB06-04).
