Abstract
The long-term stress-accelerated photothermal oxidative aging behavior of polyamide 6 (PA6) was studied in terms of the creep behavior, mechanical properties, chemical structure, crystallization, and orientation behavior. It was found that the creep deformation of PA6 under stress with ultraviolet (UV) irradiation was lower than that of the sample aging without UV irradiation. Due to stress-induced orientation, the tensile strength of PA6 aging under stress was higher than that of the sample aging without stress. For samples aging with stress, the variation in the content of gel tended to slow down, and the reduced viscosity was lower than that of the sample aging without stress. The oxidation of PA6 can be inhibited by orientation, leading to a relatively low content of carboxylic group; however, stress would accelerate the degradation of PA6, resulting in the strengthening of UV absorption. A decrease in crystallinity of PA6 after aging was observed, which was generated by chain scission in crystalline region and the cross-linking of PA6. Stress would accelerate the degradation of PA6, resulting in further decrease in crystallinity. The orientation factor increased obviously in the case of PA6 aged with stress indicating that a clear orientation of molecules induced by stress formed.
Introduction
Photothermal oxidation is the most common cause of polymer degradation in outdoor conditions. Light/oxygen/heat–polymer interactions lead to drastic changes in the physical and chemical properties of the material. Many studies have been devoted to the comprehension and the prediction of complex phenomena occurring upon the photothermal aging of the polymer. 1 –3
Polyamide 6 (PA6) has been one of the most widely used engineering thermoplastics, since it has been commercially developed first in around 1940s by a German company I.G. Farbenindustrie. 4 It has been largely applied as fibers and molded parts due to its excellent mechanical properties and high thermal resistance. However, PAs are sensitive to oxygen and continuously oxidize in ambient atmosphere resulting in the subsequent deterioration of properties such as gradual increase in yellowing and eventual embrittlement.. The thermal oxidation of PA occurs predominantly through the abstraction of hydrogen atom on the methylene groups close to the nitrogen of amide group –CO–NH–CH2–. 5 –9 In the case of photooxidation, radiation with a certain wavelength can initiate the oxidation of PAs by direct cleavage of the C–N bond and cause chain scission and cross-linking of PA, and therefore weaken the amorphous zone between the crystalline lamellae. Considerable research has been focused on the thermooxidative and photooxidative aging behavior of PAs. 10 –18
However, in the course of its use, the external mechanical stressing can also cause molecular degradation, physical and chemical changes in the molecular structure, which affect aging behavior, performance, and service lifetime of PAs. Therefore, it is significantly important to simulate the aging behavior of PA under stress. To our knowledge, till date, only little information is available for investigating stress-accelerating aging behavior of PA6. Karin Jacobson et al. studied the effect of stress on the oxidation degradation of PA6 with the method of stress–chemiluminescence, and on the basis of their results they concluded that the stress up to 80% of load at yield stress did not change the rate of oxidation of PA6 film. 19 The aging mechanism of PA6 under stress remained relatively unexplored.
In our previous work, the long-term stress-accelerated aging behavior of PA6 has been studied by exposure to ultraviolet (UV) irradiation, and in this work, the long-term stress-accelerating aging behavior of PA6 under UV irradiation and high temperature was studied. The aging behavior and mechanism were investigated in terms of creep behavior, mechanical properties, molecular structure, orientation, and crystallization behavior. Based on these results an attempt was made to get a better understanding of the influence of stress and temperature on the thermooxidative degradation of PA6.
Experimental
Materials
PA6 used in this work was a commercial grade granular product (YH800) without any additives and supplied by Yueyang Petrochemical Co., Ltd (Hunan, China) with relative viscosity of 2.85 ± 0.03 in formic acid. The samples of PA6 were prepared by injecting the mold onto standard dumbbell and rectangular splints (length: 155 mm, thickness: 4.2 mm).
Stress–thermal oxidative aging of PA6
The standard dumbbell-shaped samples of nylon 6 were imposed with tensile stress at 110°C on the self-made stretching apparatus fitted up with mercury lamp as UV source (500 W, 300–450 nm). The samples were taken out at regular time intervals and used for mechanical and chemical characterization.
Measurement
Mechanical properties
The tensile and bending performance of PA6 samples were measured with a 4302 material testing machine (Instron Corporation, Norwood, Massachusetts, USA) according to the ISO 527-1993 and ISO 178-1993 standards, respectively. The tensile test speed was 50 mm min−1, and the sample length between bench marks was 50 ± 0.5 mm.
The notched Charpy impact strength of the samples was measured with a ZBC-4A impact testing machine (Xinsansi Co., Shenzhen, China) according to the ISO 180 standard.
Reduced viscosity
Samples of PA6 of about 0.5 g were dissolved in 100 mL of formic acid (88 wt%). The time of outflow of the solution was measured in a Ubbelohde-type viscometer in a water bath at 25°C, according to ISO 307-1984 standard. Then, the reduced viscosity can be calculated using the following equation
where t is the time of outflow of the PA6 solution in seconds, t 0 is the time of outflow of the solvent in seconds, and C is the concentration of the PA6 solution in gram per milliliter.
End-group analysis
Carboxylic acid groups
Samples of PA6 of about 0.3 g were dissolved in 20 mL of phenylcarbinol at 150°C.
20
Propyl alcohol of about 3 mL were added and the hot solution was titrated with sodium hydroxide (NaOH; of about 0.02 mol L−1) using phenolphthalein as the indicator. The content of the carboxylic acid can be calculated using the following equation
where a is the volume of NaOH solution used by the sample in milliliter, b is the volume of NaOH solution used by the solvent in milliliter, n is the molar concentration of NaOH solution in moles per liter, and W is the weight of the sample in grams.
Terminal amine groups
Samples of PA6 of about 0.3 g were dissolved in 25 mL of a mixed solution of phenol–methyl alcohol (volume ratio 1:1) at 55°C. The solution was titrated with hydrochloric acid (HCl; of about 0.005 mol L−1) using thymol blue as the indicator. The content of the terminal amine groups can be calculated using the following equation
where A is the volume of HCl solution used by the sample in milliliter, B is the volume of HCl solution used by the solvent in milliliter, n is the molar concentration of HCl solution in moles per liter, and W is the weight of the sample in grams.
UV spectrum analysis
Samples of PA6 of about 0.2 g were dissolved in 50 mL of a mixed solution of sulfuric acid/methanol (0.4 mol L−1). The UV absorption measurements for the solutions for characterizing the structure during aging were carried out with a U3010 spectrophotometer (Hitachi, Japan).
Nonisothermal crystallization analysis
The nonisothermal crystallization was performed with a 204 Phoenix differential scanning calorimeter (Netzsch, Germany). The temperature scale of differential scanning calorimetry (DSC) was calibrated with indium. The granulated samples of about 10 mg were heated from ambient temperature to 250°C with a heating rate of 10°C min−1 under nitrogen atmosphere. The quantity of heat absorbed during the melting of the polymer is substantively equivalent to that required to break down the crystal structure. The higher the crystallinity (X
c), the higher the melting heat. X
c can be calculated using the following equation
where H m is the melting enthalpy, and H 0 is the balance melting enthalpy, that is, the melting enthalpy of 100% crystallization of PA6, which is 190 J g−9.
WAXD analysis
Wide angle x-ray diffraction (WAXD) analysis of PA6 at the beginning and end of aging was conducted with a D8 diffractometer (Bruker, Japan) with copper-Kα radiation generated at 40 kV and 30 mA.
Results and discussion
Creep behaviors of PA6 during aging
The creep behaviors of PA6 subjected to stress of 24.5 MPa at 110°C with and without UV radiation were investigated. Figure 1 showed the variation in creep deformation of PA6 with aging time. In the initial stage of aging, under two aging conditions, creep deformation of the samples was found to increase rapidly. Then after about 200–300 min, a platform was observed, and the creep deformation tended to be stable. The crystallinity of PA6 is about 30% and there exists a lot of amorphous area and free volume for molecular chains to move at the beginning of aging, resulting in the remarkable increase in creep deformation of PA6 under stress. With increase in aging time, molecular chain arrangement became orderly under stress and the free volume decreased, and the movement of molecular chain became slow and difficult, resulting in decrease in the creep rate. Besides, the creep deformation of PA6 under stress with UV irradiation was lower than that of the sample aging without UV irradiation, which may be due to the cross-linking of molecules under UV irradiation, limiting the mobility of molecular chains.
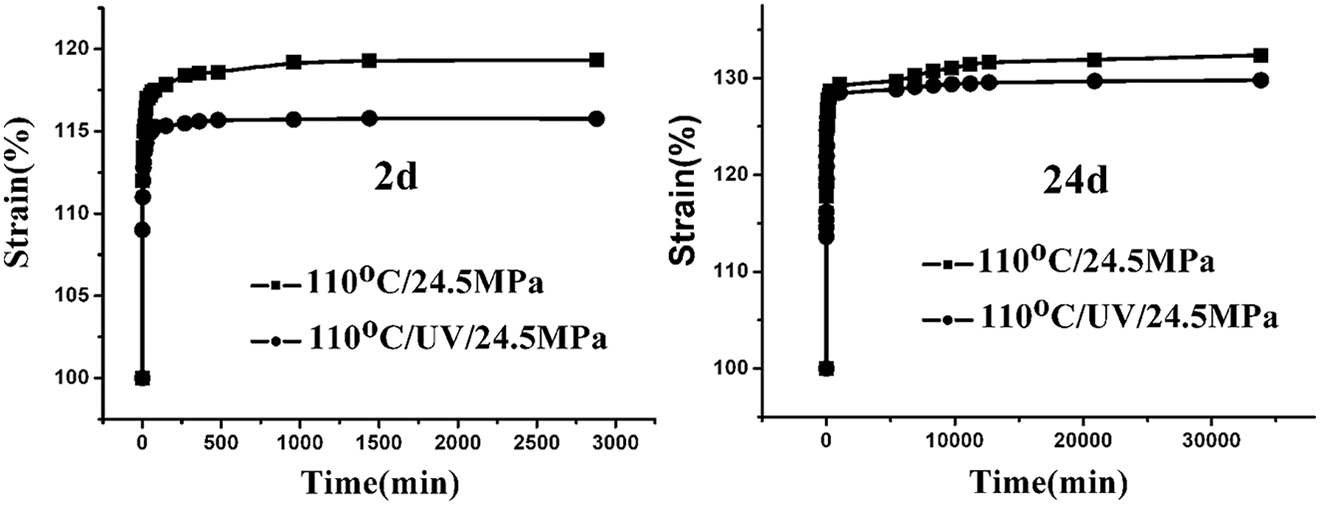
Strain of PA6 as a function of aging time under different conditions. PA6: polyamide 6.
Mechanical properties of PA6 during aging
The comparison of mechanical properties of PA6 under different aging conditions was shown in Figure 2. The tensile strength of PA6 aging both with and without stress continued to increase during the first 6 days and decreased afterward. However, during the final period of aging, the tensile strength increased slightly resulting from UV-induced cross-linking. Due to stress-induced crystallization and orientation, the tensile strength of PA6 aging under stress was higher than that of the sample aging without stress. The elongation at break of PA6 both with and without stress decreased rapidly to a stable value.
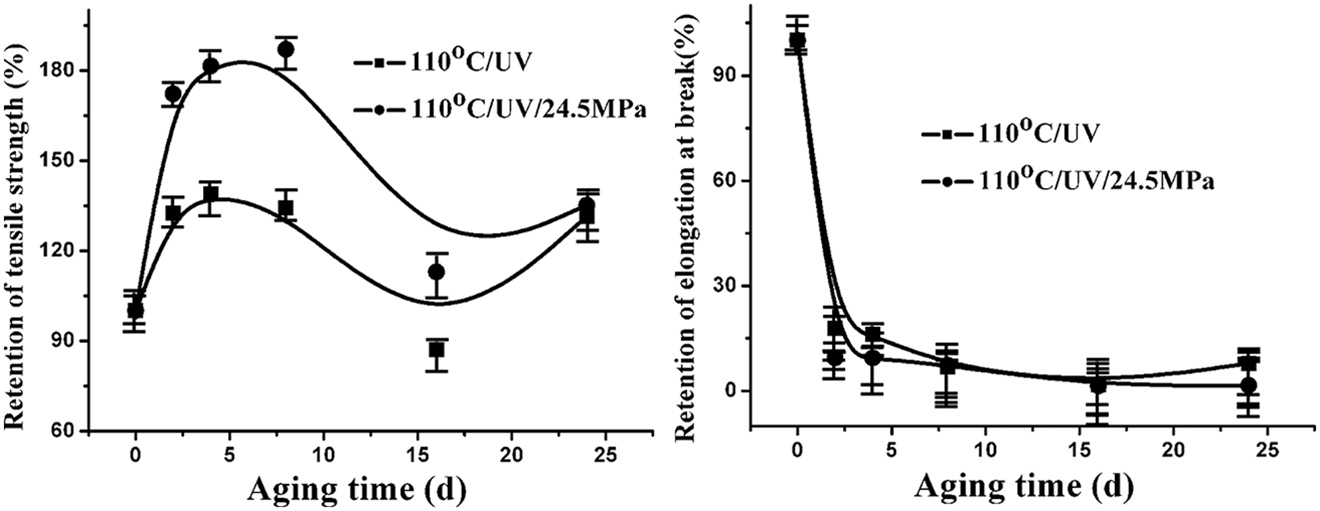
Mechanical properties of PA6 as a function of aging time under different conditions. PA6: polyamide 6.
Cross-linking and degradation of PA6 during aging
To confirm the cross-linking of PA6 under UV irradiation, the gel content of PA6 as a function of aging time was measured, as shown in Figure 3. The gel content of the sample aging under thermophoto oxidative aging increased at the initial stage of aging and then decreased. However, when samples aging are subjected to stress, the variation in the gel content tended to slow down, resulting from the combined action of molecular cross-linking formed under UV irradiation and the molecular degradation under stress and light.
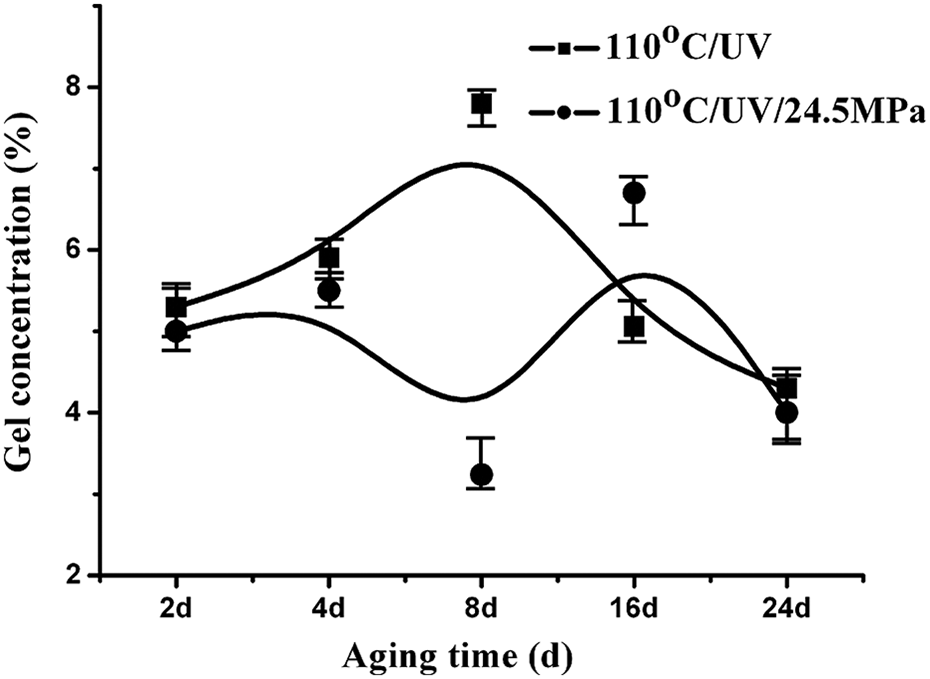
Gel content of PA6 as a function of aging time under different conditions. PA6: polyamide 6.
As shown in Figure 4, the reduced viscosity of PA6 under different aging conditions was plotted as a function of aging time. The variation tendency of the reduced viscosity of PA6 aging with and without stress was similar. Initially, it increased, then declined, and finally reached a stable value with aging time. The PA6 samples aging under stress had lower reduced viscosity than that of the sample aging without stress indicating that the stress may promote PA6 chain scission during aging, which was coincident with the result of gel content analysis.
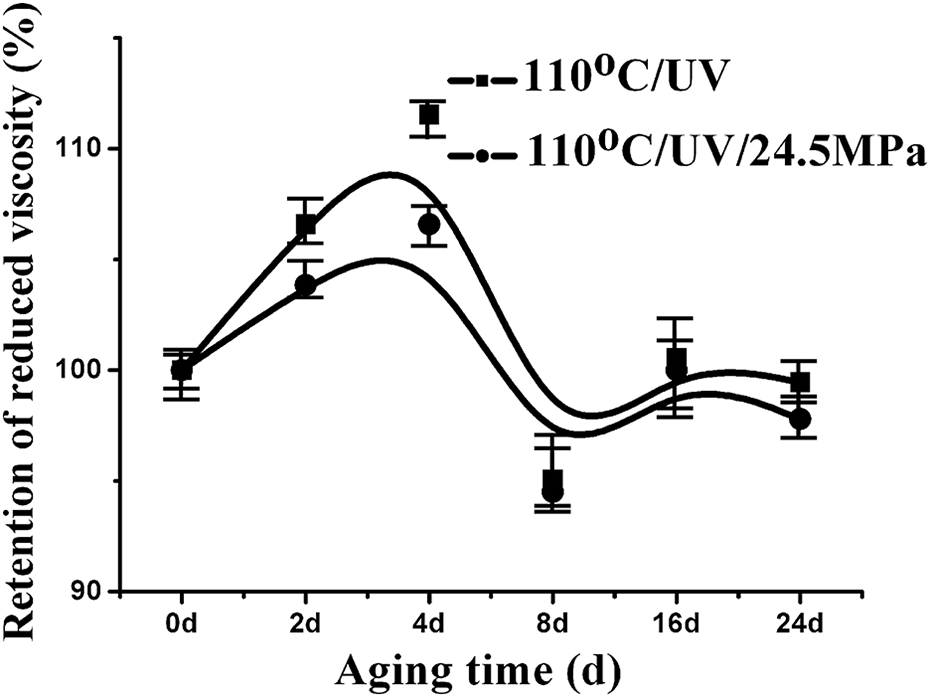
Reduced viscosity of PA6 as a function of aging time under different conditions. PA6: polyamide 6.
Chemical structure of PA6 during aging
The variation in the chemical structure (the carboxylic acid and the end amine group) of PA6 with aging time was shown in Figure 5. With increase in the aging time, the carboxylic acid concentration rose and the end amine group concentration dropped monotonically for PA6. The carboxylic acid concentration of PA6 aging with stress was lower than that of the sample aging without stress, and its end amine group concentration was relatively higher during the whole aging process.
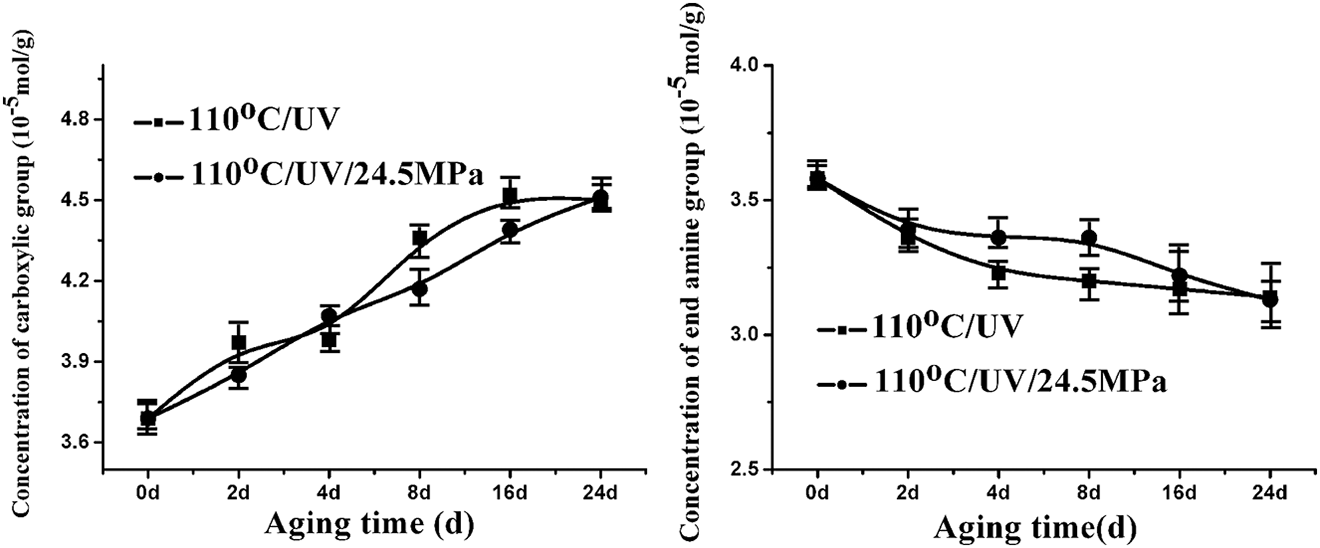
Concentration of carboxylic group and amine end group of PA6 as a function of aging time under different conditions. PA6: polyamide 6.
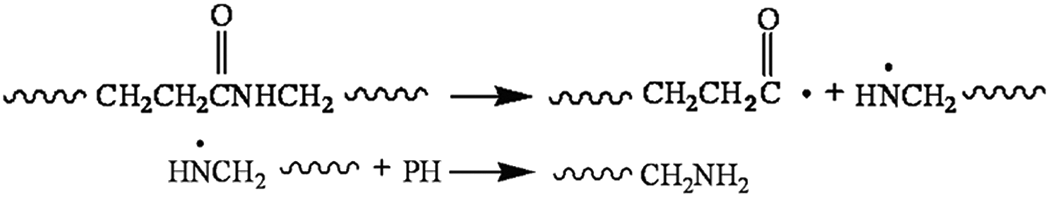
The amide bond is considered to be the weakest bond in the molecule of polyamide. The rupturing of the amide bond and the adjacent C–C bond resulted in the formation of the carboxylic acid, carbonyl, and the end amine group. The decline of the concentration of the end amine group of PA6 perhaps resulted from the reaction between end amine groups and carboxylic acids, or producing the compounds such as tertiary amine, or producing the compounds escaped from the matrix by aging degradation. During the thermooxidative aging of PA6, the higher the concentration of the carboxylic acid, the more quickly the oxidation aging of the sample, and further degradation of the polymers may be promoted. However, the degradation reaction became very complicated in the presence of stress.
The stress-induced orientation may inhibit the oxidation of PA6, leading to a relatively low content of carboxylic group. However, under UV irradiation, stress would also accelerate the degradation of PA6, resulting in the rupture of the amide bond as shown below, and eventually causing relatively high end amine group concentration for the sample aging under stress.
UV spectra of PA6 under different aging conditions were shown in Figure 6. UV absorption of PA6 at the band of 220 nm and approximately 250–290 nm increased after aging, while UV absorption of PA6 aging under stress was significantly stronger than that of sample aging only under UV irradiation and heat.
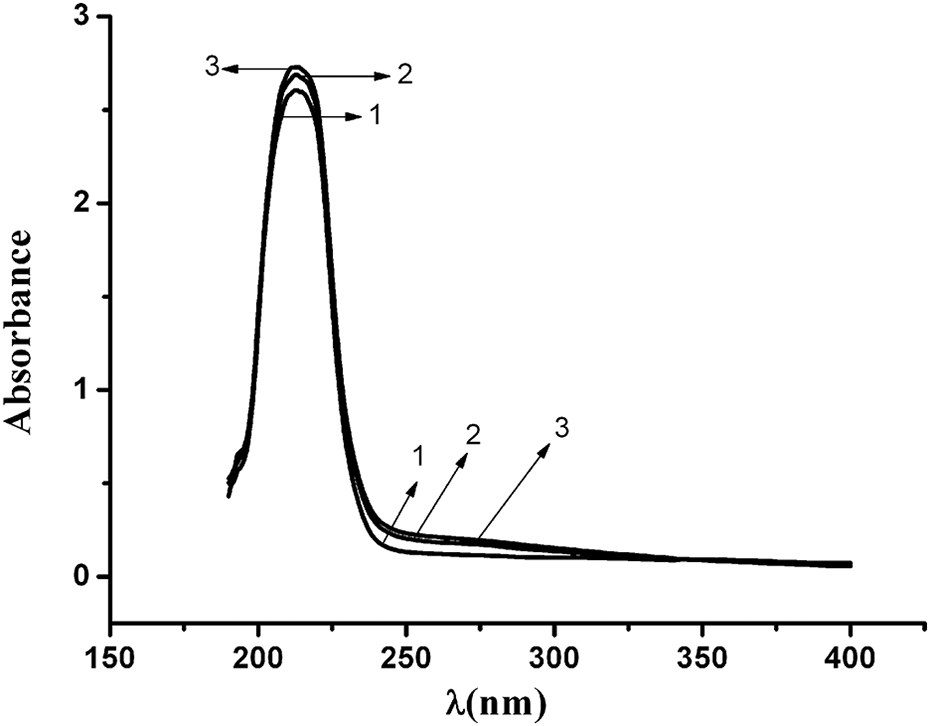
UV spectrum of PA6 under different aging conditions (in 0.4 mol L−1 sulfuric acid/methanol; 1: PA6 at 0 days; 2: PA6 at 110°C under UV irradiation at 24 days; 3: PA6 at 110°C under UV irradiation with 24.5 MPa stress at 24 days). PA6: polyamide 6; UV: ultraviolet.
After aging, degradation products such as unsaturated aldehydes, ketones, carboxylic acids, and other compounds were generated, and π→π* or n→π* electron transition of carbonyl group in these compounds may result in an increase in UV absorption at about 205 nm. However, the auxochrome group in the vicinity of carbonyl group led to the n–π conjugation effect and resulted in the red shift of the corresponding UV absorption to 220 nm. The UV absorption at 260–290 nm was enhanced due to the formation of isolated carbonyl group during aging. A series of degradation reactions were initiated as shown below:
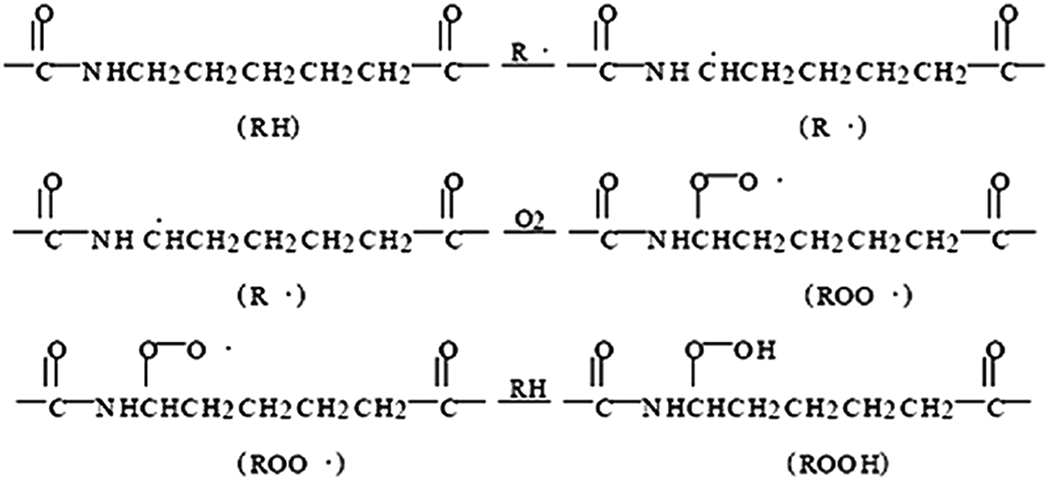
Moreover, stress may accelerate chain scission during thermooxidative aging, resulting in the strengthening of UV absorption of PA6 during aging.
Crystallization and orientation behavior of PA6 during aging
The nonisothermal DSC curves of PA6 aging under different conditions with a heating rate of 10 K min−1 were shown in Figure 7. Table 1 presents the onset melt temperature (T onset), the end melt temperature (T end), the peak melt temperature(T m), and the X c. It can be seen that the crystallinity of PA6 decreased and T m changed slightly after aging.
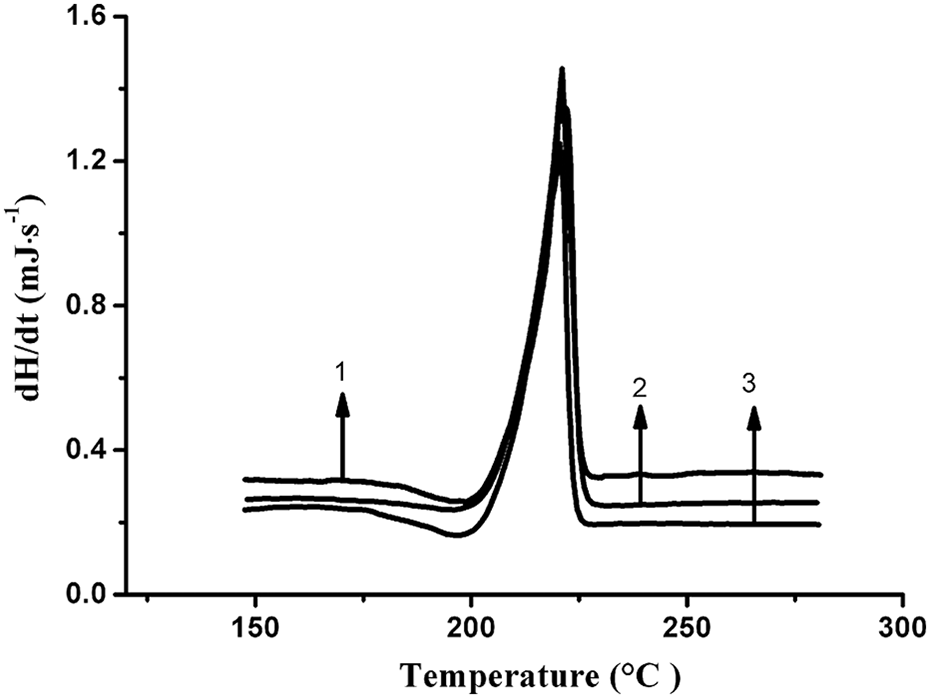
DSC curves of PA6 with a heating rate of 10 K min−1 under different aging conditions (1: PA6 at 0 days; 2: PA6 at 110°C under UV irradiation at 24 days; 3: PA6 at 110°C under UV irradiation with 24.5 MPa stress at 24 days). DSC: differential scanning calorimetry; PA6: polyamide 6; UV: ultraviolet.
Melting parameters of PA6 under different aging conditions.

T onset: onset melt temperature; T end: the end melt temperature; T m: the peak melt temperature; X c: crystallinity; PA6: polyamide 6; UV: ultraviolet.
The decrease in crystallinity of PA6 after thermophoto oxidation may be generated by chain scission in the crystalline region and the cross-linking that would limit the mobility of molecular chains and hinder the crystallization of PA6. In the case of samples aging subjected to stress, stress would accelerate the degradation of PA6, resulting in further decrease in crystallinity.
The WAXD patterns of varying aging samples of PA6 were shown in Figure 8. The diffractions of (200) planes of PA6 appeared as two strong circular arc on the equator for all samples, indicating a preferential orientation along the injection flow direction. However, there was no obvious difference between samples with different aging time after photothermal oxidative aging, which demonstrated that the variation in orientation may not be caused by UV and thermal aging. When subjected to stress, with increase in aging time, (200) reflection of PA became narrower in their spread and more prominent, suggesting that the crystal axis was oriented.

2D-XRD patterns of PA6 under different aging conditions. 2D-XRD: two-dimensional x-ray diffraction; PA6: polyamide 6.
Orientation factor and crystallinity of PA6 under different aging conditions.
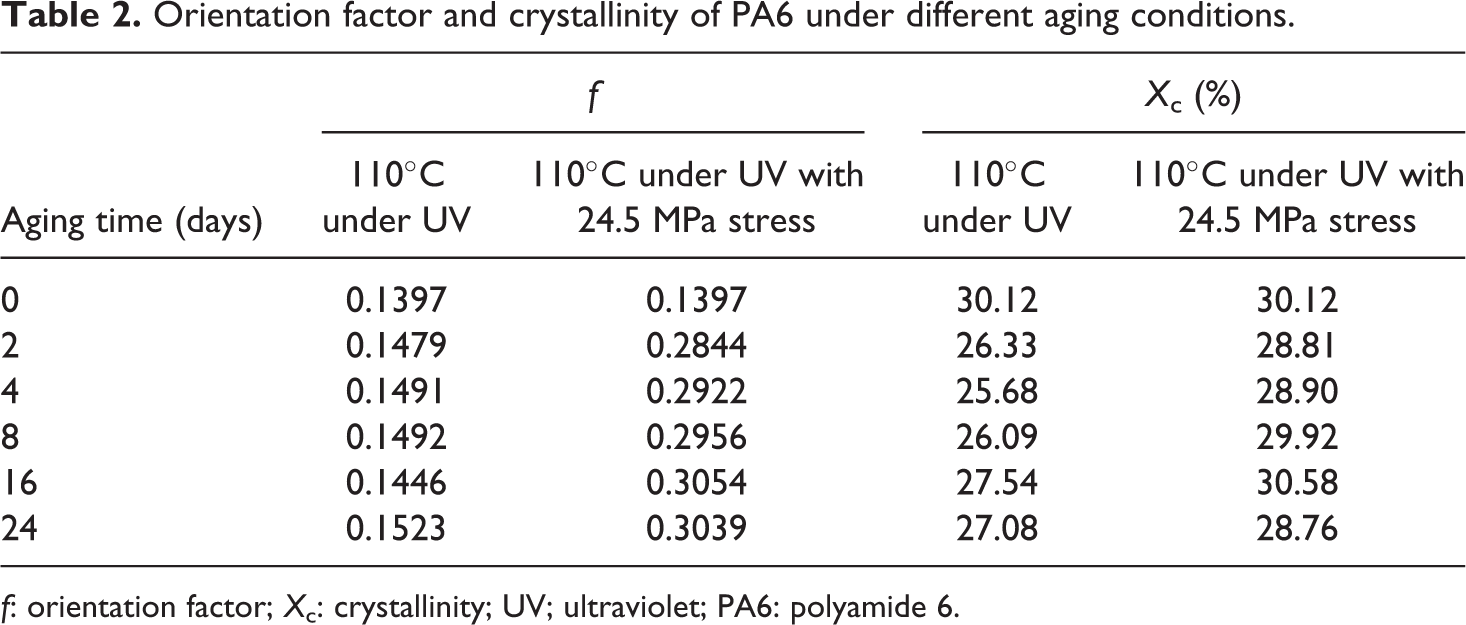
f: orientation factor; X c: crystallinity; UV; ultraviolet; PA6: polyamide 6.

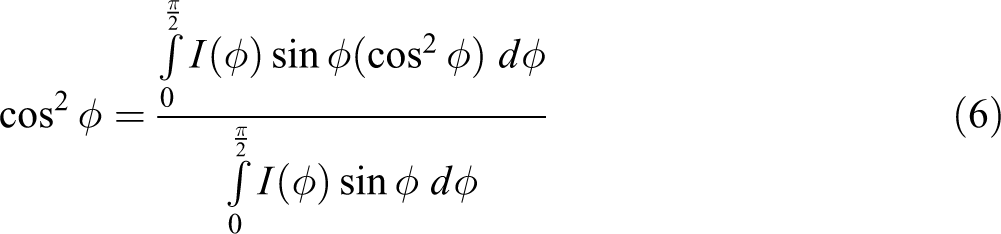

where θ is Bragg’s diffraction angle, u is the angle between the normal crystal surface and the tensile direction.
One-dimensional x-ray diffraction curve corresponding to the two-dimensional diffraction pattern was shown in Figure 9. The intensity of the diffraction peak represented the degree of the order in the material, including crystallization and orientation. It can be seen that the diffraction peak position of aged PA6 had no change; however, there were significant differences in intensity, indicating that stress-accelerated aging did not affect the crystal type, but it can significantly affect the crystallization and orientation of PA6.
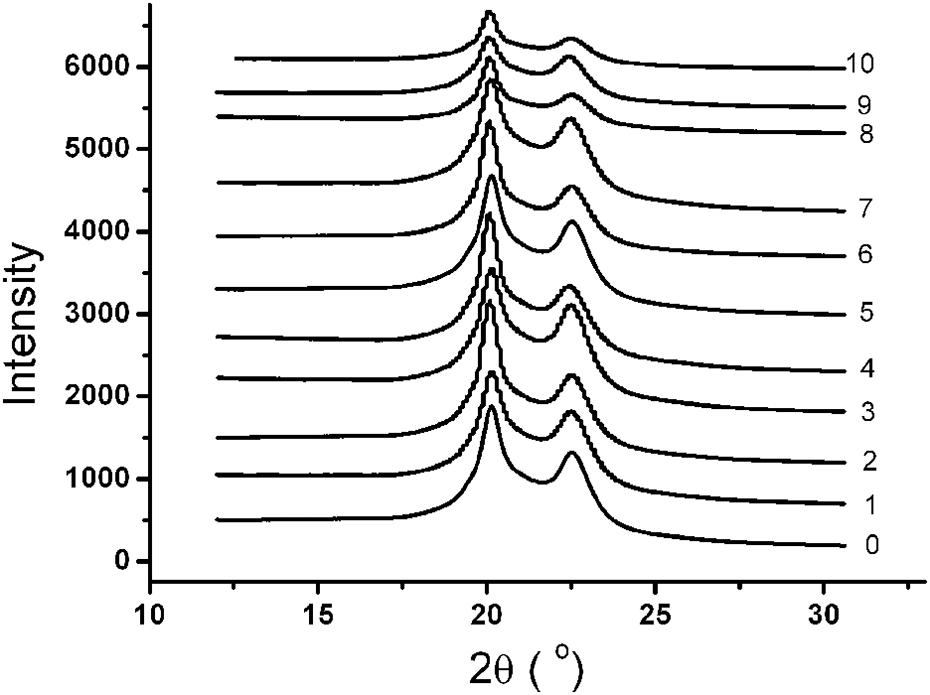
XRD curves of PA6 under different aging conditions (0: 0 day; 1: 110°C under UV irradiation at 2 days; 2: 110°C under UV irradiation with 24.5 MPa stress at 2 days; 3: 110°C under UV irradiation at 4 days; 4: 110°C under UV irradiation with 24.5 MPa stress at 4 days; 5: 110°C under UV irradiation at 8 days; 6: 110°C under UV irradiation with 24.5 MPa stress at 8 days; 7: 110°C under UV irradiation at 16 days; 8: 110°C under UV irradiation with 24.5 MPa stress at 16 days; 9: 110°C under UV irradiation at 24 days; 10: 110°C under UV irradiation with 24.5 MPa stress at 24 days). XRD: x-ray diffraction; PA6: polyamide 6; UV: ultraviolet.
Figure 10 shows the corresponding decomposed WAXD curves obtained using PeakFit software (Jandel Scientific, San Rafael, CA), from which the crystallinity of PA6 can be calculated with the peak area of crystalline and amorphous regions. The calculated data were summarized in Table 2.
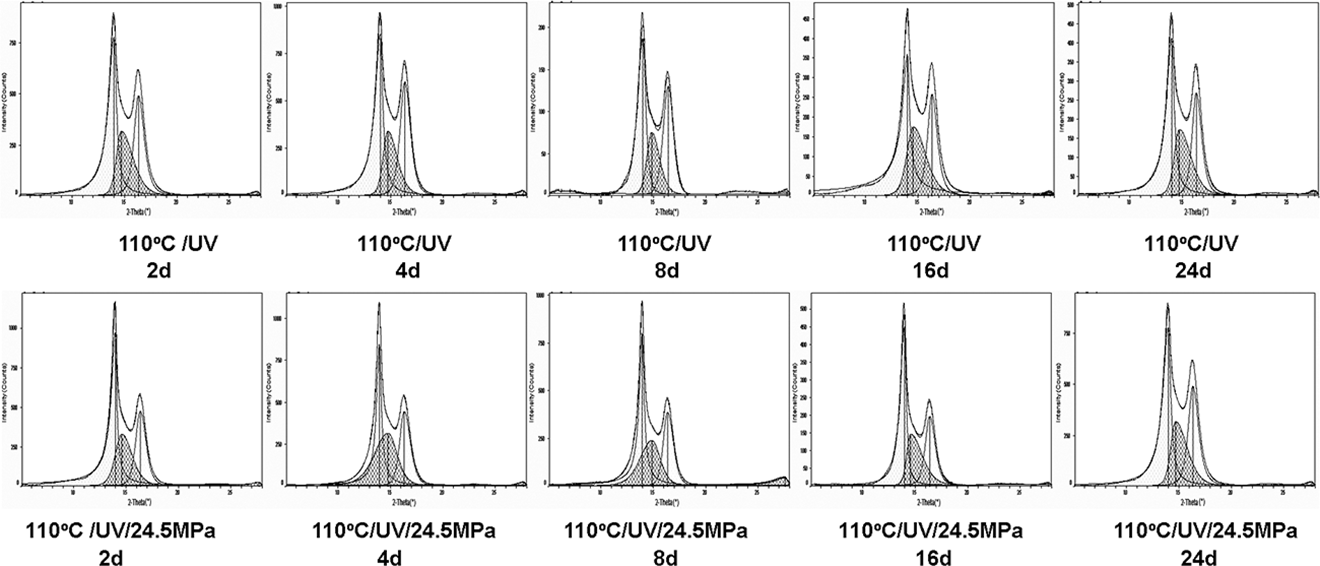
Deconvolution of WAXD curves of PA6 under different aging conditions. WAXD: wide-angle x-ray diffraction; PA6: polyamide 6.
As shown in Table 2, the photothermal oxidative aging caused a slight increase in the value of f, which was more pronounced in the case of PA6 aged with stress indicating that a clear orientation of molecules induced by stress formed. However, the crystallinity decreased after aging, demonstrating that degradation and cross-linking occurred, and the crystallization of PA6 was weakened, which was coincident with the results of DSC analysis.
Conclusion
The long-term stress-accelerating photothermal oxidative aging behavior of PA6 was studied. The results showed that a remarkable increase in creep deformation of PA6 under stress was observed at the beginning of aging, and the creep deformation of PA6 under stress with UV irradiation was lower than that of the sample aging without UV irradiation. Due to stress-induced crystallization and orientation, the tensile strength of PA6 aging under stress was higher than that of the sample aging without stress. The gel content of the sample aging under thermophotooxidative aging increased at the initial stage of aging and then decreased. For samples aging under stress, the variation in the gel content tended to slow down, and the reduced viscosity was lower than that of the sample aging without stress, resulting from the combined action of molecular cross-linking formed under UV irradiation and the molecular degradation under stress and light. The stress-induced orientation can inhibit the oxidation but accelerate the chain scission of PA6, leading to a relatively low content of carboxylic group, high end amine group concentration, and strengthening of UV absorption. DSC and WAXD results showed that a decrease in crystallinity of PA6 after aging was observed, which was generated by chain scission in crystalline region and the cross-linking of PA6. In the case of samples aging when subjected to stress, stress would accelerate the degradation of PA6, resulting in further decrease in crystallinity. The f value increased obviously in the case of PA6 aging with stress indicating that a clear orientation of molecules induced by stress formed.
Footnotes
Funding
This work is financially supported by Key Natural Science Fund of China (51133005).




