Abstract
The disposal problems associated with the conventional plastic have imposed a long-standing quest of developing the degradable material. Indeed, blending of conventional plastic with renewable resources as the base materials is an attempt of imparting some level of biodegradability on the resulting composites. Thus, for this study, the effect of plasticized sago starch (plasticized) incorporation on the properties of low-density polyethylene (LDPE)-reinforced with kenaf core fiber (KCF) was evaluated. The ratio of LDPE/KCF was fixed at 80/20 and blended with the thermoplastic sago starch (TPSS) content ranging from 10 to 40 wt%. The blended samples were characterized by means of mechanical performance, Fourier transform infrared analysis, thermogravimetric and differential scanning calorimetry behavior, water uptake, and morphological properties. The experimental result shows that there is a gradual decrease in tensile strength, modulus, and elongation at break with an increase in TPSS loading. The degree of dispersion and adhesion of TPSS in LDPE/KCF composites revealed by scanning electron micrograph supports the findings of tensile properties. The thermal stability of the composite was clearly improved with the addition of TPSS. However, water uptake and hydrophilic character of the composite system tended to augment as the TPSS imparted to the composites.
Introduction
Tremendous global awareness and environmental concerns have been paid on the disposal problems associated with conventional petroleum-based plastic considering the amount churned out as waste.
It is undeniable that the continuous use of plastic is due to their low cost, lightweight yet high mechanical performance, and ease of production. However, the ever-growing production and use of plastic have led to a waste disposal problem because they are inherently inert to the microorganisms or the chemicals in the environment. 1 Therefore, in the current revolution taking place it is crucial to develop plastic material comparable to the conventional one together with the benefit of biodegradability, which will then reduce the environmental impact.
In order to impart degradability, biopolymers such as starch are commonly incorporated into conventional plastic, which will then boost the accessibility of the plastic to oxygen and microorganism. Starch is a good biodegradable filler candidate because it possesses satisfactory thermal stability and causes minimum interference with melt flow properties of most materials used in the plastic industry. 2 Basically, the method used to combine the hydrophilic starch and hydrophobic polyolefin plays an important role in determining the performance. The processing and properties of starch/polyethylene (PE) blend has been described by Griffin previously. 3 Besides the mentioned advantages, starch is cheap, easy to be gelatinized, high in viscosity if properly extracted and easy to be molded. 4 So far, many researchers have focused on starch from maize, tapioca, and rice-filled PE5 and very few reports are available on the properties of sago starch blend. Malaysia is one of the countries with a large cultivation of sago palm. The rather excessive production with regards to the current needs make sago starch attractive and promising filler for PE. 5 –7 However, in the absence of additives, products from starch are brittle and sensitive to water (hygroscopic). Therefore, various approaches can be utilized to design starch materials with enhanced structural and functional stability during use, which then could broaden their application range. In the presence of plasticizers to turn into thermoplastic starch (TPS) at high temperature and under shear, it can readily melt and flow, allowing for its use as an extruded or injected material, similar to most conventional synthetic thermoplastic polymers. In the previous work, it was found that higher amount of starch can be incorporated with low-density PE (LDPE) when it used in a thermoplastic (plasticized) form. 8 Generally, addition of TPS as a minor phase into a synthetic thermoplastic matrix as a major phase causes a reduction in the mechanical properties. 9 It was reported, for example, that a blend of TPS from wheat starch (0–22% w/w) with LDPE exhibited a decrease in the Young’s modulus. 8 To preserve renewability, biodegradability, and to improve mechanical properties of the final products, association between natural fiber and blend has been investigated. 10,11 Apart from that, TPS has been blended with synthetic polymers such as high-density PE (HDPE) and linear LDPE; however, the degradation duration is still very high due to the nonbiodegradable synthetic polymers. 12 Thus, the blend might be reinforced with natural fibers in order to improve degradability.
Natural fiber is the preference in light of the fact that it can be completely biodegraded in the natural environment and fully sustainable. Recently, many studies have investigated biodegradable polymers filled with natural fibers. For instance, different natural fibers (flax, ramie, jute, palm, and sisal, among others) are described in the literature as reinforcement of starch-based thermoplastic polymers. 13 –17 Most natural fibers are composed of cellulose, hemicellulose, and lignin in different relative amounts. 18 Thus, when natural fibers are mixed with TS and its blends, their mechanical properties become notably enhanced. This fact has been attributed to the chemical similarity of starch and fibers, providing good compatibility between them. 19 Kenaf-reinforced composites are one of the most researched areas. Kenaf is an herbaceous annual plant that is available in a wide range of weather conditions; it has been actively cultivated since it grows quickly and can be harvested at a very low cost. 20
Yet, there is little literature regarding the combination of TS and kenaf fibers with polyolefin (i.e. PE). Therefore, this work focuses on the investigation of the mechanical, thermal, water uptake, and morphological properties of this composite. Likewise, the properties were determined by means of several parameters: plasticization of sago starch, fiber loading, and processing factors.
Experimental
Materials
Sago starch (13% moisture) was obtained from the Land Custody Development Authority (LCDA), Sarawak, Malaysia. It had an average particle size of 20 µm and a decomposition temperature of 230°C. Glycerol (plasticizer) was an analytical grade reagent, purchased from Merck Chemicals (Selangor, Malaysia). LDPE (LDF 260GG) with a melt flow index of 5 g/10 min was obtained from Titan (M) Sdn. Bhd. (Malaysia). Kenaf fiber (core) with an average length of 5 mm was supplied by National Kenaf and Tobacco Board, Malaysia.
Sample preparation
Sago starch powder was vacuum dried by heating at 80°C for 24 h before blending and processing. The dried sago starch was then premixed with glycerol using a high speed mixer. The weight ratio of sago starch and glycerol was maintained at 65:35 (wt%). The blend was stored overnight to allow the diffusion of glycerol into starch granules and would help the melt mixing process. Thermoplastic sago starch (TPSS) was melt-blended with LDPE and kenaf fiber using an internal mixer (Haake Rheomix Mixer, Model R600/610, Selangor, Malaysia) at a temperature of 150°C and a speed of 50 r/min in order to obtain a homogeneous sample. Different loadings of TPSS (i.e. 10–40 wt%) were used. The processed samples were then compression molded in an electrically heated hydraulic press (Kao Tieh Go Tech Compression Machine, Taichung City, Taiwan) at a temperature of 150°C into a 1 mm thickness sheet. Table 1 shows the formulation of the composites.
The formulation of LDPE/KCF/TPSS composites.

LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
Measurement of tensile properties
Tensile tests were carried out with a Universal Testing Machine (Instron 3366) (Norwood, USA) according to ASTM D638. Dumbbell specimens of 1 mm thickness were cut from the compression-molded sheets with a Wallace die cutter. A crosshead speed of 5 mm/min was used and the test was performed at a temperature of 25 ± 3°C and a relative humidity of 60 ± 5%. Five specimens were used to obtain the average values of tensile strength, elongation at break, and Young’s modulus.
Fourier transform infrared spectroscopy
The functional groups and chemical characteristics of LDPE/kenaf core fiber (KCF)/TPSS composites were obtained by Fourier transform infrared spectroscopy (FTIR, Perkin Elmer System 2000, Selangor, Malaysia) with a resolution of 4 cm−1 in a spectral range of 4000–600 cm−1 using 32 scans per sample.
Thermogravimetric analysis
LDPE/KCF/TPSS composite samples were cut into small pieces (5–10 mg), which were then tested using thermogravimetric instruments (Perkin Elmer, Pyris Diamond TG/DTA, Selangor, Malaysia) under a nitrogen atmosphere within a temperature range from room temperature to 450°C at a heating rate of 15°C/min. Thermal degradation temperature was reported by the onset degradation temperature where the weight loss started to occur.
Differential scanning calorimetry
The differential scanning calorimetry (DSC) measurements were performed in a Perkin-Elmer DSC 7 (Selangor, Malaysia). About 10 mg of the samples was first heated from room temperature to 190°C at 5°C/min and held there for 5 min to eliminate the thermal history, cooled to room temperature, and then reheated to 190°C at 5°C/min to trace the melting behavior. Each run was performed under a nitrogen atmosphere. The melting temperature (T m) and heat of fusion (ΔH m) of the samples were determined from the peak maximum and the area under peak, respectively. The degree of crystallinity of the composites was calculated using the following equation
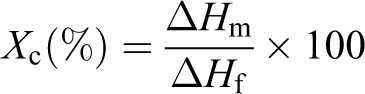
where ▵H m is the melt enthalpy of the semicrystalline LDPE blends and ▵H f is the heat fusion for crystalline LDPE.
Morphology evaluation
Scanning electron micrographs of tensile fracture surfaces of LDPE/KCF/TPSS composites were obtained using a scanning electron microscope (model ZEISS Supra 35 VP, Carl Zeiss, Oberkochen, Germany). The samples were sputter coated with a thin layer of carbon to avoid electrostatic charging during the examination. The image results were analyzed to investigate the distribution of natural fibers in the polymer matrix and their interaction.
Water absorption
Water uptake measurement was carried out as per ASTM D570. Newly prepared samples were first dried in an oven at 70°C for 24 h until a constant weight was attained and then dipped in the distilled water at ambient temperature. After immersion for a specific interval, the samples were then removed from the water, gently dried by wiping with a clean cloth, and immediately weighed to the nearest 0.001 g. The percentage of water absorption was calculated as follows
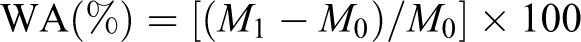
where M 0 and M 1 are the dried weight and final weight of the sample, respectively.
Results and discussion
Processing characteristics
The melt processing characteristics of the LDPE/KCF/TPSS composites have been studied with processing torque–time curves. Figure 1 shows the processing torque of the melt mixed blends with different TPSS loadings. It was observed that the torque decreases as the amount of TPSS increases in the blend. This behavior arises from the lower melt viscosity that TPSS has when compared with LDPE. In the composite with 10 wt% of TPSS, the torque decreases continuously until it becomes stable, while in the blend with 20 wt% TPSS, there is a small increase after the initial melting of the materials. Such an increase in torque is expected in the reactive blends. 8 The first peak refers to the shear torque before melting of LDPE and the next peak corresponds to the TPSS addition before the torque stabilized. The third peak appears at around 13 min, corresponding to the introduction of kenaf fiber. After 15 min, the processing torque stabilized demonstrating that a good level of mixing was achieved.
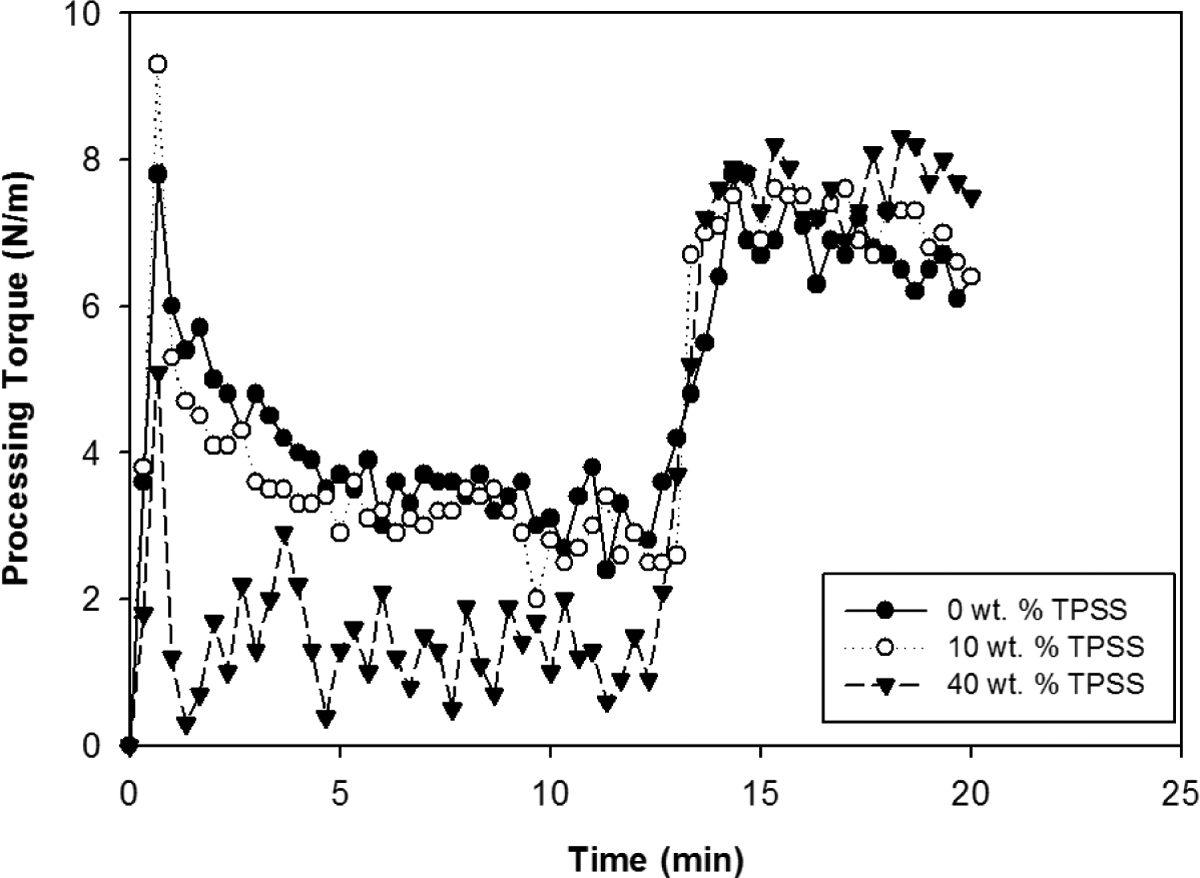
The processing torque of LDPE/KCF/TPSS composites. LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
Tensile properties
Tensile properties of LDPE/TPSS/KCF composites at different TPSS loadings are shown in Figure 2(a) and (b). It was found that the addition of 10 wt% TPSS into the LDPE/KCF composite caused an increase in the tensile strength and Young’s modulus compared to the LDPE/KCF composite without TPSS loading. The LDPE/KCF composite filled with a minor amount of TPSS showed higher strength and modulus due to the chemical similarity of both TPSS and kenaf fiber. The results were in accordance to the findings reported by Prachayawarakorn et al. 21 It is expected that the TPSS and KCF are compatible due to the similarity of the main cellulose structures composed of hydroxyl functional groups. However, the presence of high starch content (>10% by weight) had an adverse effect on the mechanical properties of the blends. 22 Both strength and modulus begin to decrease with an increase in TPSS loading. Addition of 20–40 wt% of TPSS into the system caused a reduction in its properties due to the phase incompatibility between highly hydrophilic macromolecules TPSS and nonpolar hydrophobic LDPE. Basically, LDPE can flow easier than TPSS; however, by increasing the TPSS content, the flow of LDPE might be interfered, resulting in phase separation as well as cracks at the interfacial boundary. Theoretically, the tensile strength could probably result from more dominant property of LDPE, since LDPE shows higher tensile strength than TPSS21 and the incorporation of starch into LDPE increases the stiffness of the sample. However, a high surface energy between the hydrophobic polymers (LDPE) and hydrophilic starch yields a low degree of adhesion. A weak interfacial adhesion provides site for failure initiation. Loading stresses are transferred to the filler phase with reduction in the effective surface area. 2 By virtue of having hydroxyl groups, the starch is hydrophilic. However, the formation of strong interfacial bonds like hydrogen bonds with PE is not feasible due to hydrophobicity of polymer. As a result, the tensile strength of composites was not improved. 5 The drop in tensile strength becomes more drastic at higher filler loading. This is due to the fact that filler–filler interaction becomes more pronounced than filler–matrix interaction with increasing filler content. The applied forced cannot be transferred from the polymer matrix to rigid filler particles due to the reduction in effective cross-sectional area of the composites caused by the particles. Starch granules are irregular in shape and have the tendency to form aggregates, especially at higher loading.
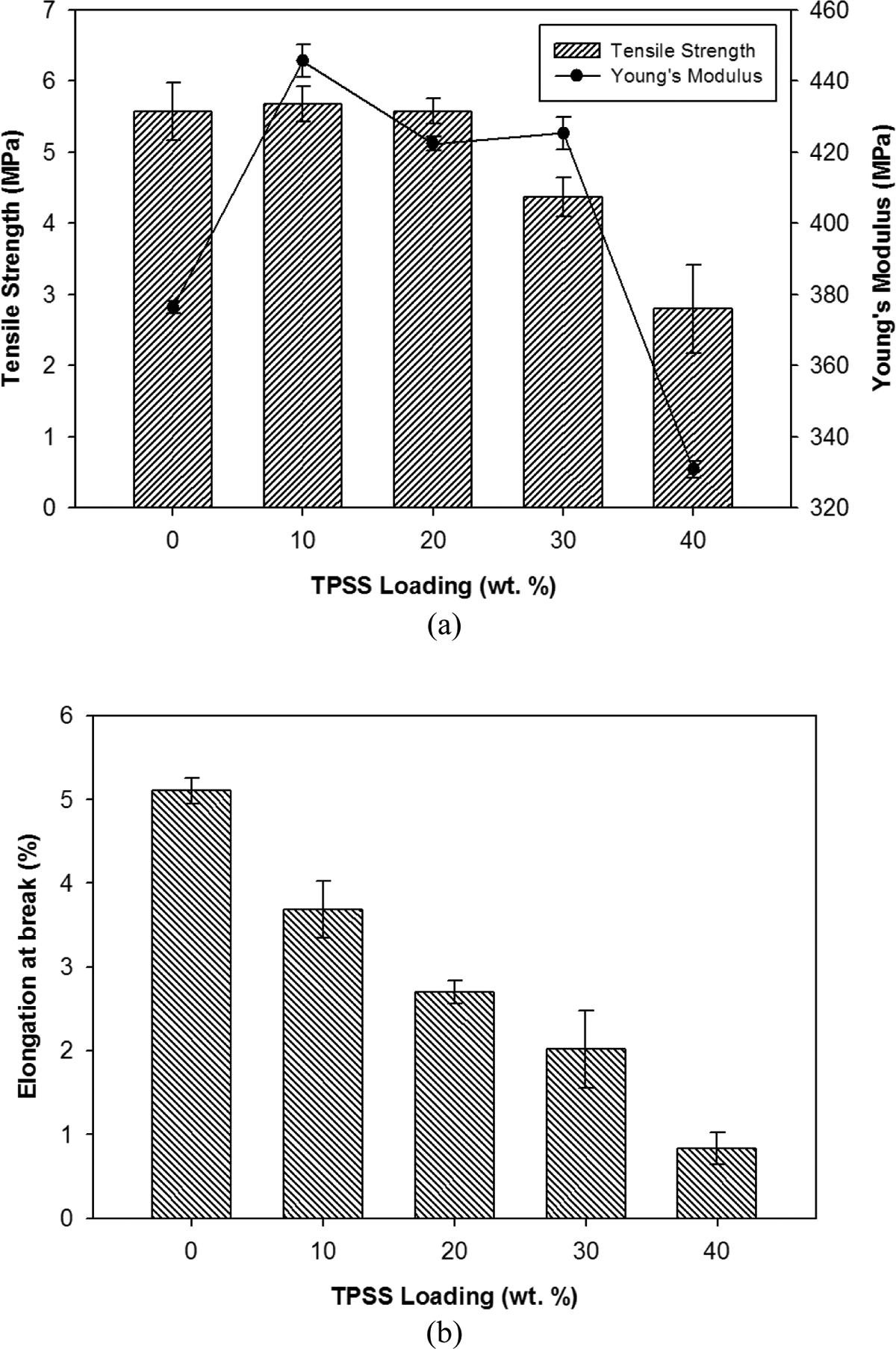
(a) Tensile strength and Young’s modulus and (b) elongation at break properties of LDPE/KCF/TPSS composites at different TPSS loading. LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
As expected, the elongation at break for LDPE/TPSS/KCF composites decreased from 5.1 to 0.8%. This is due to physical incorporation of starch into the LDPE that weakens the forces between LDPE layers and the fact that starch (a low molecular weight polymer) has lower elongation compared to LDPE. Since there is no chemical interaction between both the constituents, starch inclusion formed discontinuity, resulting in lower elongation. 23 In the synthetic polymer blend, the addition of second immiscible phase to a ductile matrix material usually significantly decreases the elongation at break. 24
Other than that it is believed the inclusion of KCF also caused the decrease in percentage of elongation at break gradually due to the high crystallinity of the fibers. The elongation at break decreased dramatically as the starch content increased up to 40% (by weight). The decrease in elongation at break values with increasing starch loading may be attributed to the fact that the starch particles do not elongate coherently with LDPE. Therefore, less strain is required to cause the sample to fail. Thus, the composite fails at lower elongation at break with increased starch loading. 5
When starch is present in small quantities, the polymer matrix is weakened by starch because the scattered starch particles do not aggregate in the polymer matrix. This means that more weak interfacial regions are formed that permit easier crack propagation. 25 In general, lower concentrations of starch result in higher values of tensile strength. However, an interesting observation is that all the starch/LDPE blends show higher modulus values though the trend was that increasing starch content reduces the modulus. This result might be due to the stiffening effect of the starch granules. The stiffening effect is because the modulus of starch is higher than that of semicrystalline LDPE due to the presence of hydrogen bonding in starch not in LDPE. 26 On the other hand, elongation at break mainly depends on the interfacial adhesion.
Infrared spectroscopic study
Infrared (IR) spectra of LDPE/KCF composites filled with different TPSS loadings are shown in Figure 3. The observed peaks are the functional groups of LPDE, starch, and fiber; however, there are some slight changes in the band positions and intensities. It is known that, if two polymers are compatible, a distinct interaction (hydrogen bonding or dipolar interaction) exists between the chains of one polymer and those of the other, causing the IR spectra of composites to change. It can be seen in Figure 3(a) that the LDPE/KCF composite exhibits IR main peak positions of 2915, 1465, and 719 cm−1, representing C–H stretching, –CH3 bending, and –CH2 vibration, respectively. However, in the case of LDPE/KCF composite filled with TPSS, IR peaks related to starch materials become prominent. It is evident in Figure 3(b) that starch is characterized by two strong and broad absorption peaks occurring at 3000–3900 (O–H) and at 1000–1250 cm−1 (C–O–C) stretching. The presence of OH group peak at 3345 cm−1is related to the presence of amylose and amylopectin in sago starch. The characteristic peaks appeared at 3200–3500 cm−1, which can be assigned to C–O–H bond stretching slightly broadened due to the increase in intermolecular hydrogen bond formation occurring when TPSS was incorporated into the composite. It is also worth noting that a slight increase in the peak at 1260–900 cm−1attributed to the stretching of the C–O group could be observed in the presence of kenaf fibers in the composite system. 27 These changes could be interpreted in terms of the rearrangement of hydrogen bonds between starch and fiber. The change in IR spectra indicates a distinct interaction and compatibility between the chains of polymers. All the characteristic peaks of LDPE and TPSS have appeared in the blends. These results indicate that the TPSS is distributed within the LDPE matrix. This is further justified with the obtained scanning electron micrograph (SEM) morphology in Figure 7.
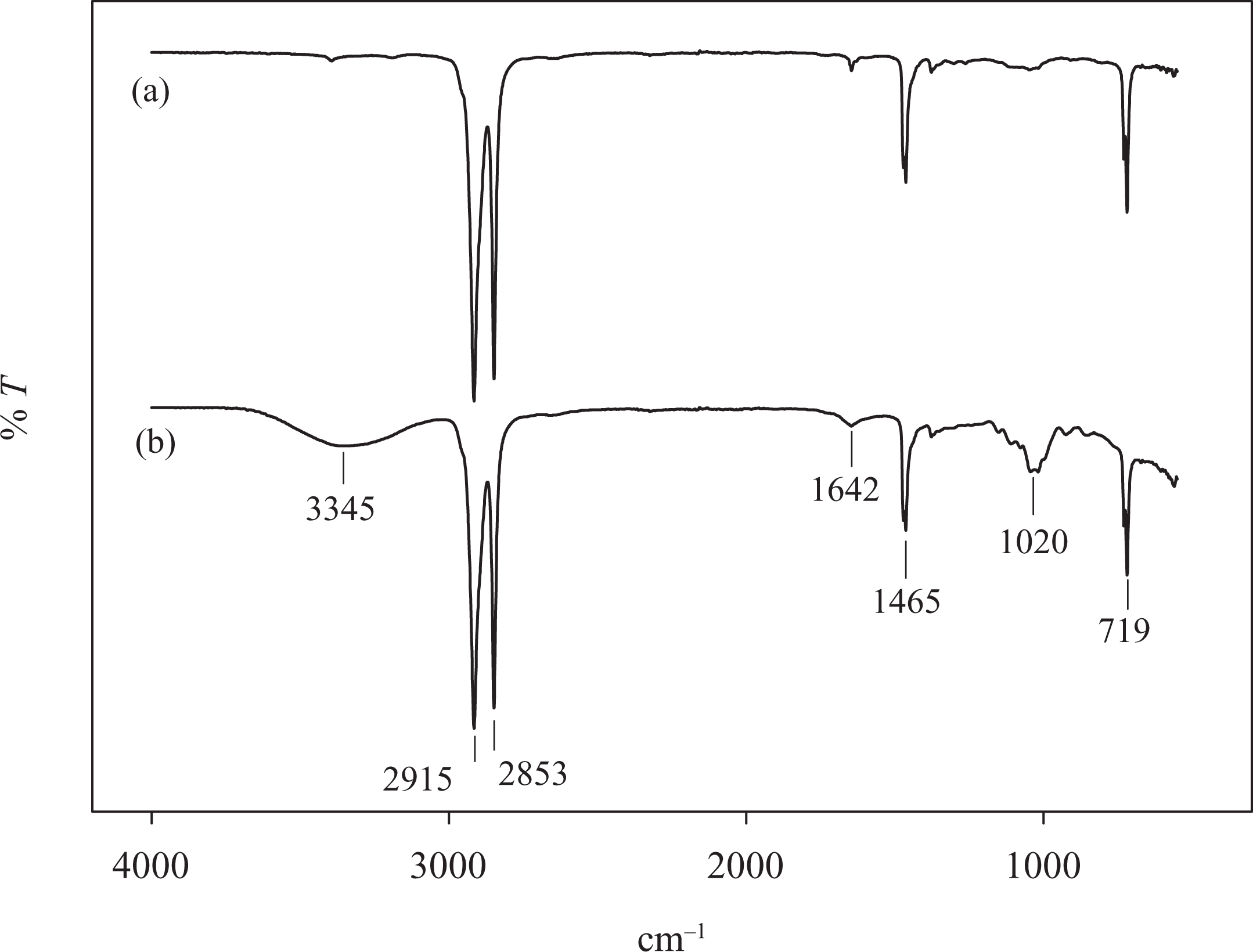
Infrared spectra of LDPE/TPSS/KCF composites at different TPSS loadings: (a) 0, (b) 10, and (c) 40% (by weight). LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
Thermogravimetric analysis
Thermogravimetric analysis of the composite was carried out to assess its thermal stability and degradation temperature. The onset degradation temperature and thermogravimetric curves of different LDPE/KCF/TPSS composite samples are shown in Figure 4. It can be observed that thermal degradation of the samples occurred in three steps: first region (250–280°C), second region (335–400°C), and third region (above 400°C). The thermal degradation of LDPE/KCF composite occurred in the two-step degradation process. The LDPE/KCF exhibited initial mass loss from approximately 302 to 370°C, which was mainly due to the decomposition of KCF. After that, second thermal degradation step of LDPE was observed. 28 The thermal degradation at around 450°C was associated with the greatest weight loss, and was due to thermal degradation of the LDPE backbone. 29
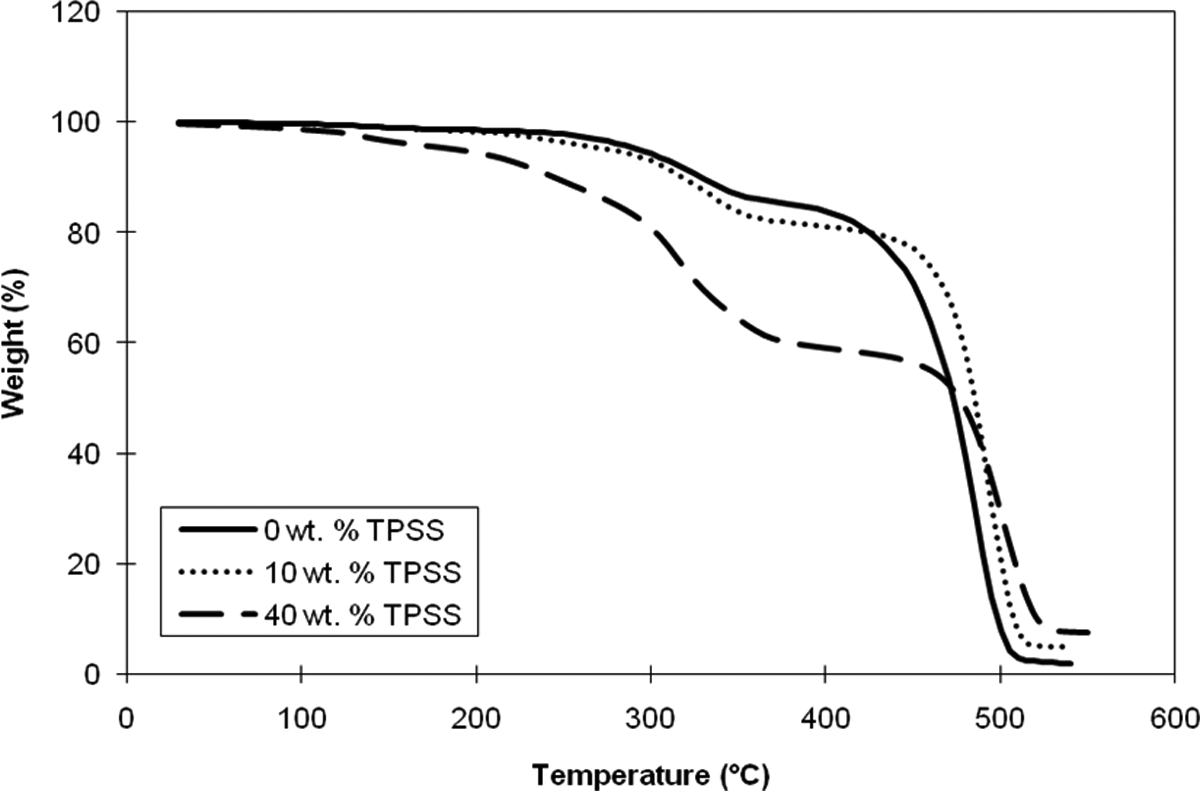
Thermogravimetric analysis thermogram of LDPE/KCF/TPSS composites at different TPSS loadings. LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
With the incorporation of TPSS into the LDPE/KCF composite, it was observed that the onset decomposition temperature (T d) of 176.2 and 295.8°C was due to the decomposition of glycerol and starch, respectively. The process continues gradually up to 300°C, where thermal degradation of starch occurred. 30 The T d value of the TPSS was 303.4°C when 10 wt% of TPSS was incorporated into the LDPE/KCF composite system. This is because of higher thermal stability of fiber, and because of the phase compatibility between TPSS and KCF due to their similar chemical structures, the two structures was linked through hydrogen bonds. It is noted that the T d of LDPE component was obtained at a temperature of 457.8°C. It was also found that the percentage weight loss at 400°C was 81.17 and 59.237% for the TPSS loading of 10 and 40 wt%. The results clearly indicated that the thermal stability could be improved by the addition of TPSS. 9 According to the content of TPSS, the reduction in weight loss was the result of the improvement in thermal stability. 30
Differential scanning calorimetry
The second heating cycles in DSC thermograms of LDPE and its blends are shown in Figure 5. The melting temperature (T m), enthalpy of melt (ΔH m), and degree of crystallinity (X c) are summarized in Table 2. DSC thermograms showed only one endothermic transition of LDPE and its blends. Other endothermic peaks were not obtained. It can be observed that the endothermic peak at 108.3°C indicates the melting temperature of pure LDPE. In comparison with the LDPE and its blends, the DSC results show that the T m did not change significantly, which suggested that LDPE properties of composites are intact. 29 It is interesting to note that although there might be a slight increase in T m of the LDPE blend with increasing starch content, the endothermic transition that occurred was only attributed to the LDPE phase given that starch was not melted during processing. 1 The addition of starch hinders the mobility of LDPE segments and thus increases the value of T m. It is can also be observed that there was a reduction in enthalpy of melt (ΔH m) for the blends compared to pure LDPE. ΔH m is obtained from the total area of the melting peak. The reduction in ΔH m is correlated with the decrease in the LDPE content in blend specimen. 23 The presence of starch by some means also interfere the nucleation effect of LDPE. In addition, there is a slight decrease in crystallinity of the LDPE and its blends. This suggested that LDPE-starch interactions are weak. 31
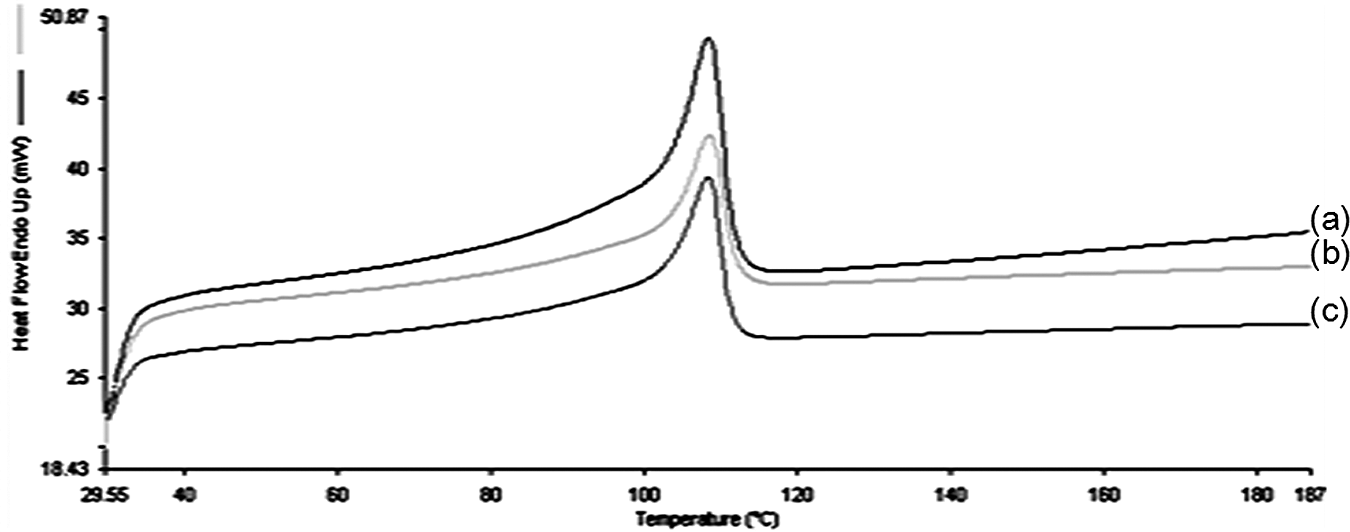
Differential scanning calorimetry thermograms of (a) LDPE, (b) LDPE/KCF, and (c) LDPE/KCF/TPSS composites. LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
Differential scanning calorimetry data for LDPE/KCF/TPSS composites.

LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
Water absorption
Figure 6 illustrates the behavior of the sample on immersion in water at ambient temperature of TPSS at different loadings. The absorption of water is related to its rate of diffusion into the composite. It can be observed that the water uptake of the composites increases linearly within the initial stage of absorption process, then the increasing rates slow down and finally leads to a plateau corresponding to the water uptake at equilibrium. It took about 3 days immersion for different TPSS loading samples to reach absorption equilibrium. Theoretically, the existence of LDPE in the composite could dramatically limit water absorption and reduce water sensitivity due to the hydrophobic characteristics of nonpolar LDPE. However, in this case the rate of moisture absorption highly depended on the TPSS content. Starch is known to take up large amounts of water, which is further exacerbated by glycerol plasticization. It is expected that the amount of water absorbed increases with the increase in the amount of starch. This is because starch possesses hydroxyl groups that enable an interaction with water molecules. At low TPSS loading, the blend absorbed moisture gradually until it reaches equilibrium. However, at high fractions of TPSS, the moisture absorption isotherm is slightly different. The absorption rate was very fast, although every blend reaches their own equilibrium within the same period of time. 1 These behaviors were related to the TPSS, which is more hydrophilic than LDPE, leading to an enhancement of hydrogen bond formation between hydroxyl groups in starch and water molecules. As a result, the water uptake of the composite increases as a function of the TPSS content. Apart from that, it is worth noting that, low degree of adhesion between the hydrophobic LDPE and hydrophilic TPSS resulted in cracks and voids between them, allowing for the easy penetration of moisture in the voids. Thus, the percentage of water uptake arises significantly.
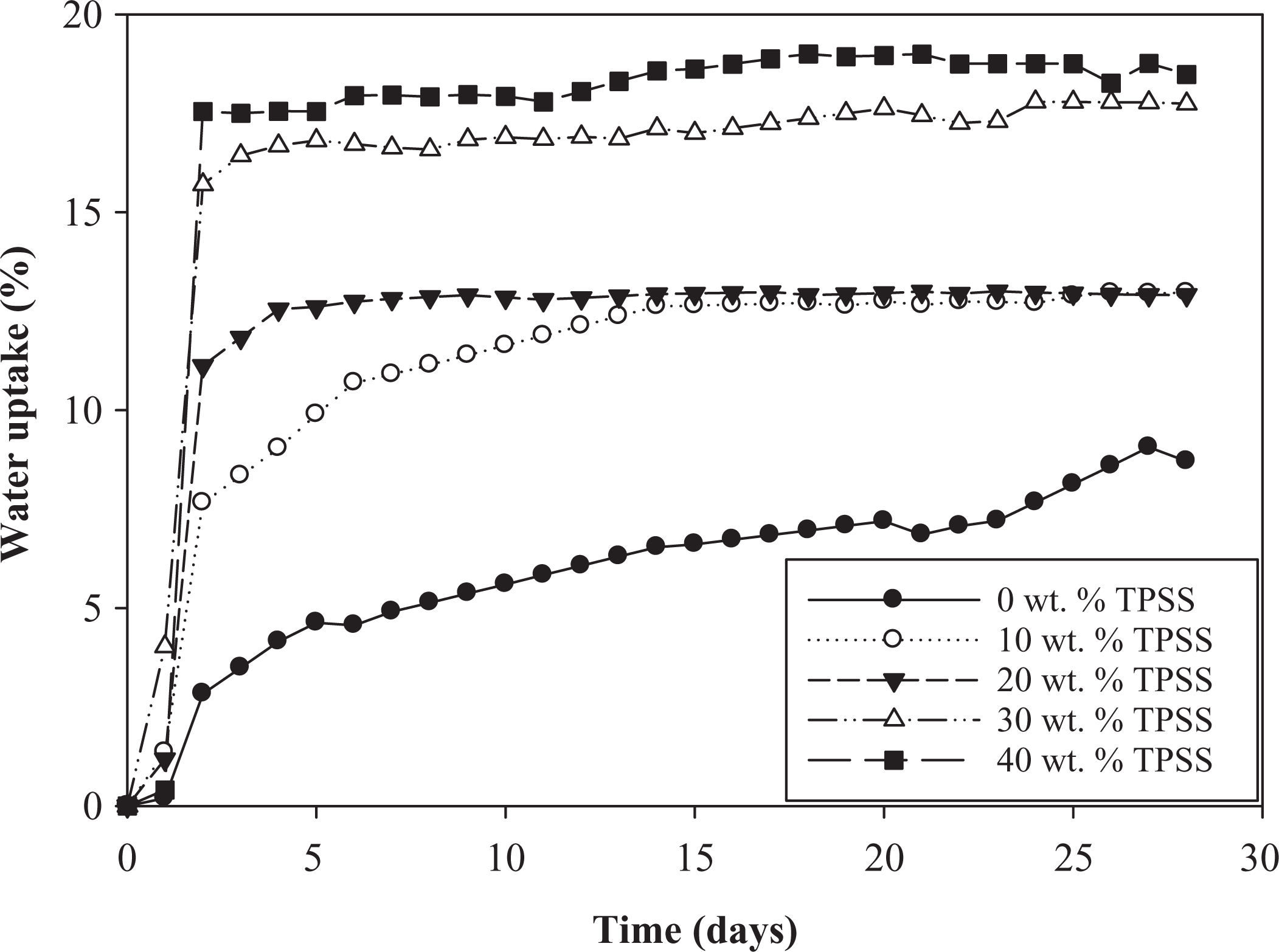
Water uptake of LDPE/TPSS/KCF composites at different TPSS loadings. LDPE: low-density polyethylene; KCF: kenaf core fiber; TPSS: thermoplastic sago starch.
Morphology
SEMs at 100× magnification of the tensile-fractured surface of LDPE/KCF composite filled with different TPSS loadings are shown in Figure 7. The morphology structure of polymer blends is very important because it ultimately determines the properties of the composites. For the surface and fracture view of the LDPE with the addition of 20 wt% kenaf fibers (Figure 7(a)), it can be seen that LDPE formed a continuous phase with KCF randomly distributed. However, there are pull-out traces on the fracture surfaces, which might be due to the opposite nature of the components (hydrophilic KCF and hydrophobic LDPE). For the composite filled with 10 wt% TPSS, the SEM in Figure 7(b) revealed spots of starch granules within the LDPE continuous phase. Starch particles seem to be embedded in LDPE matrix, although very few surface gaps, cracks, and separate starch particles were visible in LDPE matrix. LDPE/TPSS were immiscible but behaved as polymer blends composed of two deformable phases as seen by the TS domains in LDPE. Fibers are thoroughly dispersed within the polymer matrix and the fracture surface shows intimate mixing of the fiber with the matrix. This corroborates the presence of chemical bonding between fiber and TPSS, probably due to the occurrence of hydrogen bonding. 32 This phase morphology is the evidence of phase compatibility and it is well accepted that tensile strength and modulus of composites are influenced by the nature of matrix and the interaction between fiber and matrix. 33 Whereas, Figure 7(c) illustrates that the starch was still granular in structure and agglomerates, which was an indication of heterogeneous dispersion of starch in LDPE. However, there is phase separation between LDPE and TPSS because of the differences in polarity. The distinct interfacial appearance between starch and LDPE suggested that there was little interaction but large interfacial tension between them. The fracture surface revealed poor interaction (poor wettability) between the starch granules and the polymer matrix and a nonuniform distribution of the granules, a situation that gave rise to the formation of stress concentration points. 2 The fractured surface micrographs showed that the number of voids during fracture due to poor interfacial adhesion keeps on increasing with increasing starch content (Figure 7(d)). The poor interfacial adhesion was reflected in the mechanical properties. It is seen that increasing starch loading decreases the interfacial adhesion and homogeneity. The mechanical properties already discussed also support these findings. 26
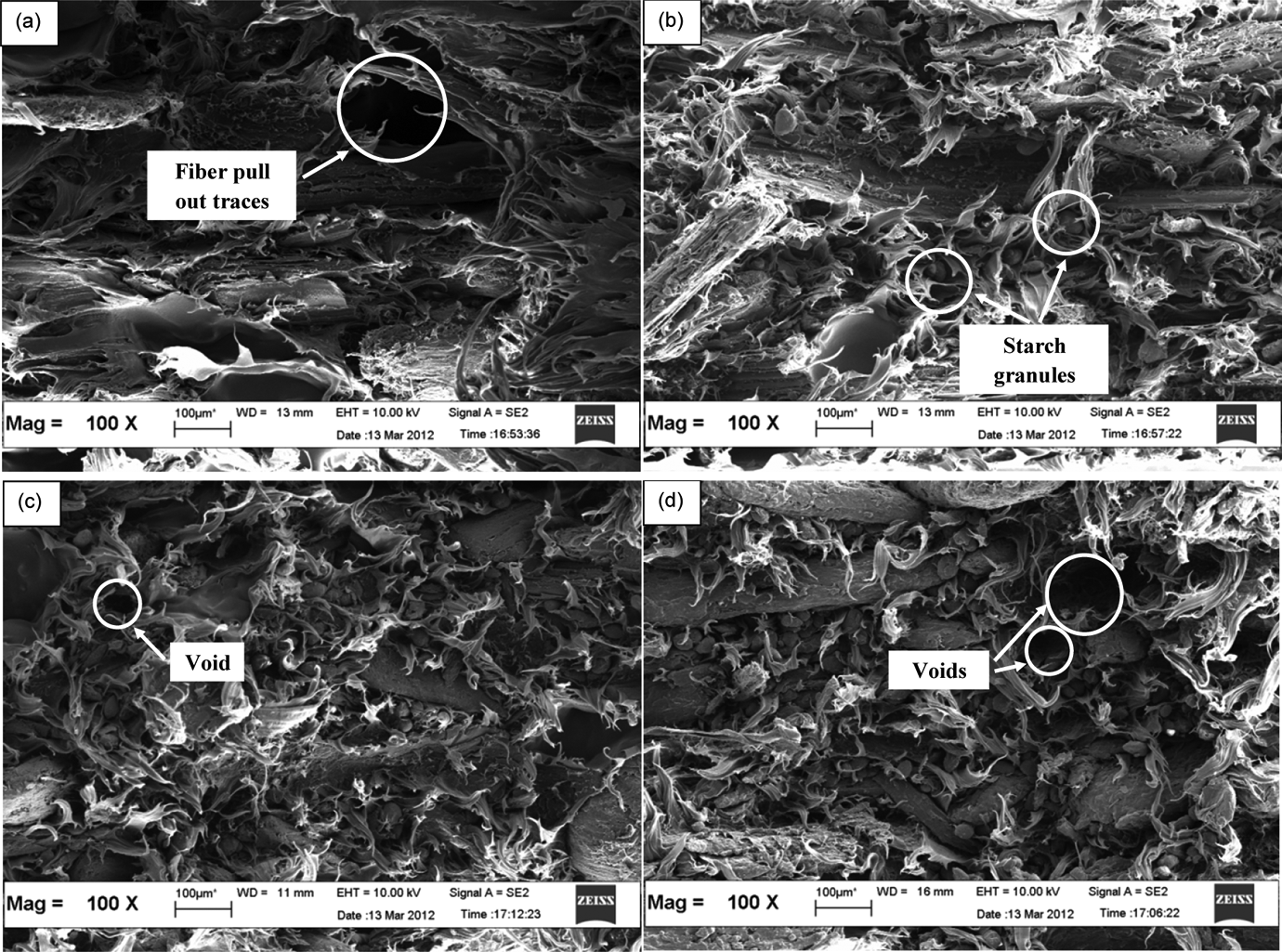
Scanning electron micrographs of LDPE/KCF/TPSS composites at (a) 0, (b) 10, (c) 20, and (d) 40% (by weight) of TPSS loading.
Conclusions
The incorporation of 10 wt% TPSS into the blend caused better improvement in strength and modulus. Whereas, the elongation at break decreased tremendously with increased amount of TPSS loaded into the system. FTIR analysis showed the occurrence of bonding between functional groups of LDPE, TPSS, and kenaf fiber. LDPE/KCF composite with 10 and 40 wt% TPSS had a significant improvement in thermal stability. Water uptake tended to increase when a greater amount of TPSS incorporated into the system. It seems that water absorption increases with immersion time reaching a certain value at saturation point, where no more water was absorbed and the water content remained constant. This polymer composite with excellent moisture stability has the potential to create new development in the construction of materials for food packaging or various application products suitable in the hot–wet environment. From SEMs, it can be seen that kenaf fibers were embedded in the matrix, however, there are phase separations between TPSS and LDPE. These findings significantly correlate with the mechanical properties. Interfacial adhesion between all components (i.e. LDPE, starch, and kenaf) was found to be the major factor that contributes to the performance of the composite.
Footnotes
Acknowledgements
The authors would like to thank National Kenaf and Tobacco Board, Malaysia for supplying kenaf used in this study.
Funding
This work was financially supported by Research University Grant (Grant No.
