Abstract
This work focuses on the combination of the complementary physical properties of multiwalled carbon nanotubes (MWCNTs) and poly(3-octylthiophene) (P3OT)/polystyrene (PS) polymer blend. The characterization of MWCNTs/P3OT/PS hybrid system performed by Fourier transform infrared spectroscopy, ultraviolet (UV)–visible absorption, and electrical measurements show interesting effects. Optical results demonstrate that the addition of MWCNT to P3OT/PS matrix blend will enhance the UV–visible absorption and insignificantly affect the optical energy gap of P3OT/PS matrix; this can be explained in terms of a reduced photo-induced charge transfer in such composite system. This study shows that incorporation of 5 wt% functionalized MWCNTs onto P3OT/PS polymer blend will convert this material from insulator to conductor and at this level of MWCNTs content, MWCNTs will form a network inside blend matrix with low capacitor element and high resistor element effects.
Introduction
Due to the unique properties of the carbon nanotubes (CNTs), polymer/CNT nanocomposites have received intense attention in recent years to create new material systems with superior properties. 1,2 Scientists and researchers are attracted to the unique shape, size, and remarkable physical performance of the nanotubes that possess a very high tensile strength, high thermal stability, and high electrical conductivity in a cylindrical shape with a diameter of around 10 nm and a length of upto 100 μm. Due to these attractive properties, nanotubes have found applications in various areas such as electrochemical devices, hydrogen storage, field emission devices, sensors, and probes. Particularly, multiwalled nanotube (MWNT)-based polymer nanocomposites have the potential to enable development of unique lightweight materials. 2 –6 On the other hand, use of nanotube reinforcements in polymer composites has been a challenge because of the difficulties in optimizing the processing conditions to achieve good dispersion and load transfer. Such difficulties in dispersion refer to the fact that the nanotubes are held together as bundles and ropes; thus they have very low solubility in solvents and tend to remain as entangled agglomerates. Such a problem can be solved using several techniques such as the use of surfactants, sonication, and other mixing methods. Recent work has demonstrated superior dispersion of MWNTs in polymers by functionalizing the nanotubes to make them compatible with solvents and the matrix material.7–17 The most commonly used method to functionalize CNTs is the wet chemical method using different strong acids. At the same time, recent research showed that such a method will cause a morphological damage to CNTs, and also found that the oxidation process with strong acids will create a significant physical damage and will cause severe degradation of CNTs. 18,19 Besides, dry oxidation, such as the use of ozone in the presence of ultraviolet (), or exposing CNTs to UV radiation at ambient atmosphere has been an alternative treatment to resolve the issues associated with the wet oxidation. 20 –23 Moreover, many studies showed that UV-modified CNTs induced a higher electrical conductivity in polymer matrices and will not cause morphological damages to CNTs. 22,23
Previously, increasing emphasis in the research of conjugated polymers is placed on the engineering and processing of conjugated polymers in blends and composites with conventional polymers. For these blends, popular conventional polymers with excellent optical and mechanical properties like polystyrene (PS), polyethylene (PE), poly(vinyl carbazole) (PVK), or poly(vinyl benzyl chloride) (PVBC) are used as host matrices. 24,25 These studies, however, focused on the investigation of the photoexcited states in conjugated polymer (donor)/fullerene (acceptor) interpenetrating networks embedded into conventional polymer hosts like PS, PVK, or PVBC. At the same time, it was found that no other studies investigated optical or electrical properties of multi-walled carbon nanotube (MWCNT)/poly(3-octylthiophene) (P3OT)/PS composites. However, this study will show very interesting results in the electrical and optical properties of such composites.
The extreme surface to volume ratio of the nanofillers, several orders of magnitude larger than that of micron-sized fillers, and the proximity of size of the nanofiller to the radius of gyration of the host polymers lead to interphase volume fractions that can easily exceed the volume fraction of the filler phase. By increasing the dispersion of the nanofillers in the polymer matrix, the influence of this interphase zone is increased. Previous study 26 has introduced visual evidence of the interphase in a MWCNT/polycarbonate system, with scanning electron microscopic images illustrating a significantly adsorbed polymer layer on nanotubes protruding from fracture surfaces. This study also demonstrated an increased size of this region for systems in which the nanotubes had been functionalized before processing the composite. This evidence, combined with mechanical property data, indicates that functionalization leads to an increased interaction zone in polymer nanocomposite systems. 11,12
Recently, some success has been reported in improved electrical, optical, and mechanical properties of MWNT-reinforced polymers as well as enhanced crystallization and orientation of nanotubes. 5,7 –11,13,15,27 –32 All these studies show that the physical properties of MWCNT-reinforced polymers are postulated to be heavily dependent on the specific MWCNT/polymer systems and different processing methods. Among them, the promising method is functionalization of MWNTs.
In the present work, we prepared MWCNT-P3OT-PS composites through the addition of different functionalized MWCNT loadings to polymer blend host composed of 30% P3OT polymer and 70% PS. Their electrical and optical properties are characterized and correlated.
Experimental
Materials
MWCNTs were purchased from Chengdu Organic Chemicals Co. (Chengdu, China). The samples were synthesized by natural gas catalytic decomposition over nickel-based catalyst and were advertized as having a diameter of > 50 nm, length of 10–20 μm, and purity of >95%. Figure 1(a) and (b) shows the scanning electron and transmission electron micrographs of MWCNTs as received with out further purification. MWCNTs were functionalized with UV radiation using a UV–ozone generator (Model # 42-220, Jelight Co., California, USA) in ambient laboratory air for a period of upto 180 min.

(a) SEM and (b) TEM of MWCNTs. SEM: scanning electron micrograph; TEM: transmission electron micrograph; MWCNTs: multiwalled carbon nanotubes.
PS was supplied by Saudi Basic Industries Corporation (KSA, Jobail) with brand name PS 330. P3OT polymer was purchased from Aldrich Co. (USA) with a molecular weight of 34,000 g mole−1 .
Films preparation
A casting technique was used to prepare the composites. In this technique, first, the P3OT and PS polymers were dissolved in chloroform at a weight ratio of 30% P3OT and stirred with a magnetic stirrer at room temperature (RT) for 2 h. In another vial, functionalized MWNTs were dispersed in the same solvent (chloroform) by bath sonication for 30 min, added to the polymer solution, and again bath sonicated for 1 h, then stirred for 24 h at 30°C to disperse the MWNT in the polymer mixture. The mixture was casted into a glass petri dish (diameter of 5 cm). The obtained products were dried at RT for 24 h. The thickness of the resulting composite films was 0.1 mm. The composites used in this study are 0, 0.5, 1, 3, and 5 wt% of functionalized MWNTs. Control sample of pure P3OT/PS with 30% P3OT was prepared following the same procedures.
Fourier transform infrared analysis
Fourier transform infrared (FT-IR) spectra of composite films were recorded using a PerkinElmer 100 FT-IR spectrophotometer in the transmission mode at a wavenumber range of 4000–1000 cm−1.
Electrical measurements
Electrical measurements with alternating current (AC) and direct current (DC) applied electric fields were performed using the 4200-SCS Semiconductor Characterization System (Keithley Co., USA). Samples were shaped into circular disks of area 1.2 cm2 and thickness 0.1 mm. The DC electrical parameters (current–voltage (
Optical properties
UV–visible absorption spectra of the prepared composites were measured using a Shimadzu UV-VIS spectrophotometer (Model UV-3600; Japan) in the wavelength range of 230–700 nm. The scan step was 0.5 nm. The optical absorbance (
Results and discussion
Results of FT-IR spectra
The FT-IR spectra of the as received MWCNTs and those treated with UV–ozone (UVO) for 120 min are compared in Figure 2(a). The peak at 1170 cm−1 corresponds to the presence of ester groups on the treated CNTs surface. Furthermore, the peaks at 1636 and 3442 cm−1 are assigned to the stretching of C=O and a hydroxyl group, respectively. The presence of these peaks of the UVO-treated CNTs indicates that UVO treatment led to the production of carboxylic acid functional group on the CNTs surface. 21 –23,38
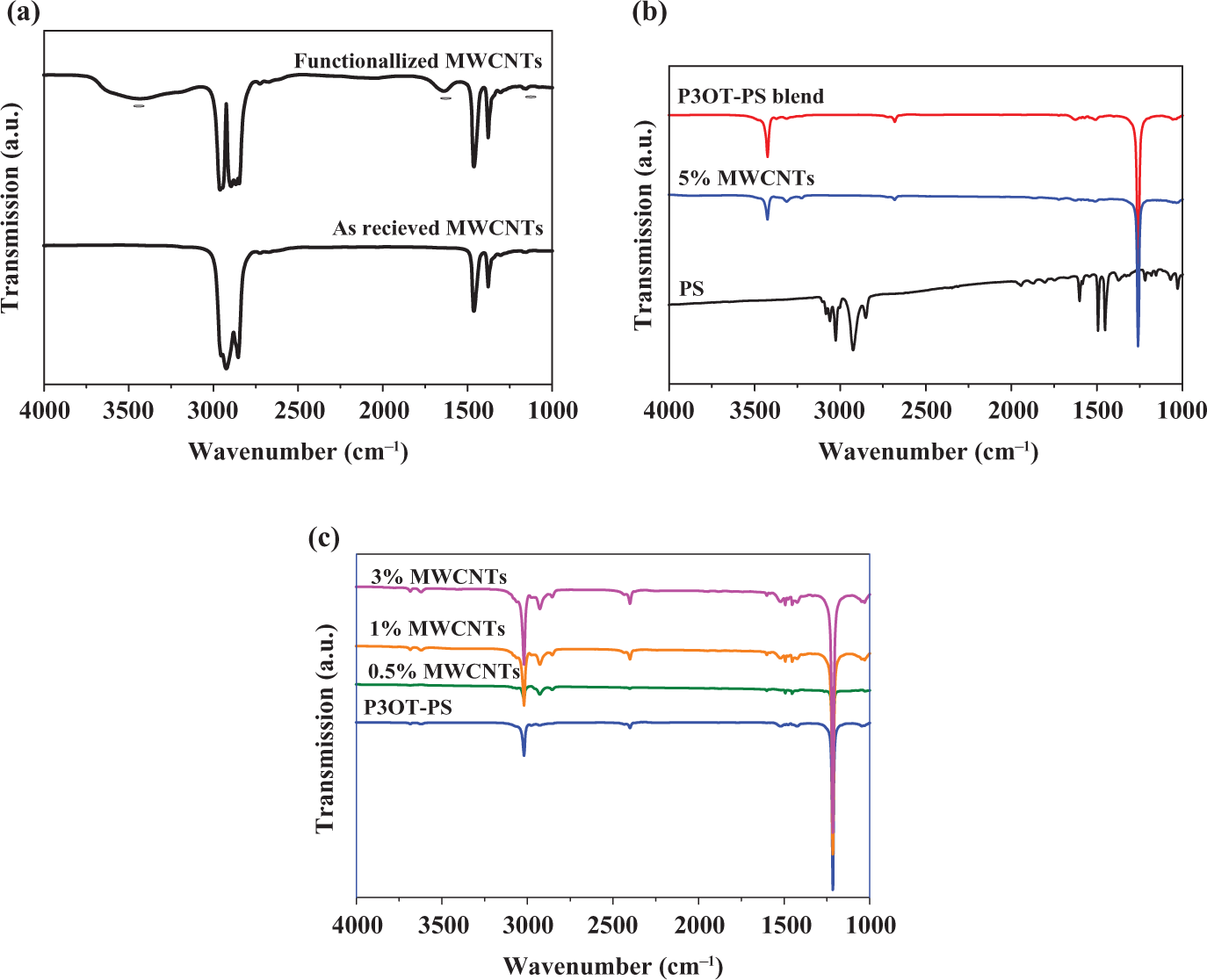
FT-IR spectra of MWCNT (a); blend (b) and composites (c). FT-IR: Fourier transform infrared; MWCNT: multiwalled carbon nanotube.
For comparison, the FT-IR spectra of PS, P3OT-PS blend, and composites are included in Figures 2(b) and (c). The main bands of PS (Figure 2(b)) are located over the 2800–3100 cm−1 range, attributed to C–H stretching vibrations in the main chain and in aromatic rings, and the peaks at 1601, 1492, 1451, 1215 and 1027 cm−1 are due to deformation and skeletal vibrations of C–H in PS. 39 In the case of P3OT polymer, it was reported that the main peaks in the P3OT infrared (IR) spectrum appear at 3054 and 1370–1550 cm−1, which are attributed to aromatic CH stretching, methyl deformation, ring-stretching modes or the aromatic CH stretching, and C=C stretching. 40 However, upon blending of 30 wt% P3OT with PS, the IR spectra of blends or composites do not show any new peaks or shift in their peak positions. This indicates that the presence of such IR bands in the spectra of composites only reflect the superposition of P3OT and PS bands, which means that there are no interactions between P3OT and PS or between MWCNTs and blend host. 41
UV–visible absorption
Figure 3 shows the normalized absorption spectra of pure PS, P3OT-PS, and MWCNT-P3OT-PS composites with different nanotube concentrations. The subsidiary frame in this figure represents the absorption spectrum of MWCNTs. PS has a strong absorption below 300 nm and the peak at 262 nm arises from the phenyl ring in each repeat unit. 39 Besides, the P3OT polymer has an absorption peak centered at around 512 nm. 42 It is known that P3OT polymer films themselves can show different absorption spectra when prepared using different solvents. 27,42,43 Figure 4 shows the absorbance of MWCNT-P3OT-PS composites as a function of MWCNTs weight fraction at 262 and 512 nm. Upon blending of 30 wt% P3OT with 70 wt% PS, no new absorption peaks are observed for the P3OT-PS blend or even in MWCNTs-P3OT-PS composite materials. Usually, new absorption peaks are expected if there is a ground-state electronic interaction between the components in a blend composite matrix. 42 In P3OT-PS blend and MWCNT-P3OT-PS composites, the absence of new absorption peaks indicates that there is no significant electronic interaction in the ground state between MWCNTs, PS, and P3OT. A peculiar observation shown in Figure 3 is the variation in PS absorbance at 262 and 512 nm when P3OT polymer and MWCNTs are blended with PS neat. The decrease in the absorbance of P3OT-PS blend at 262 nm could be attributed to the screening effects of P3OT on the absorbance of PS. This indicates, however, that P3OT-PS blend is a partially miscible blend, such an effect can also be seen in the FT-IR spectra (Figure 2). MWCNTs have a strong absorption below 300 nm in addition to their absorption in the visible region; consequently, addition of MWCNTs upto 5 wt% to P3OT-PS blends will enhance their UV–visible absorbance. It can also be seen that the increase in MWCNTs weight fraction on P3OT-PS blend will cause a slight blueshift within 2–4 nm in the peak maximum at 512 nm, this may signify that the effective π-conjugation length of P3OT becomes extended in the composite, because the local nanoscopic structure of P3OT becomes modified by PS and MWCNTs. 42
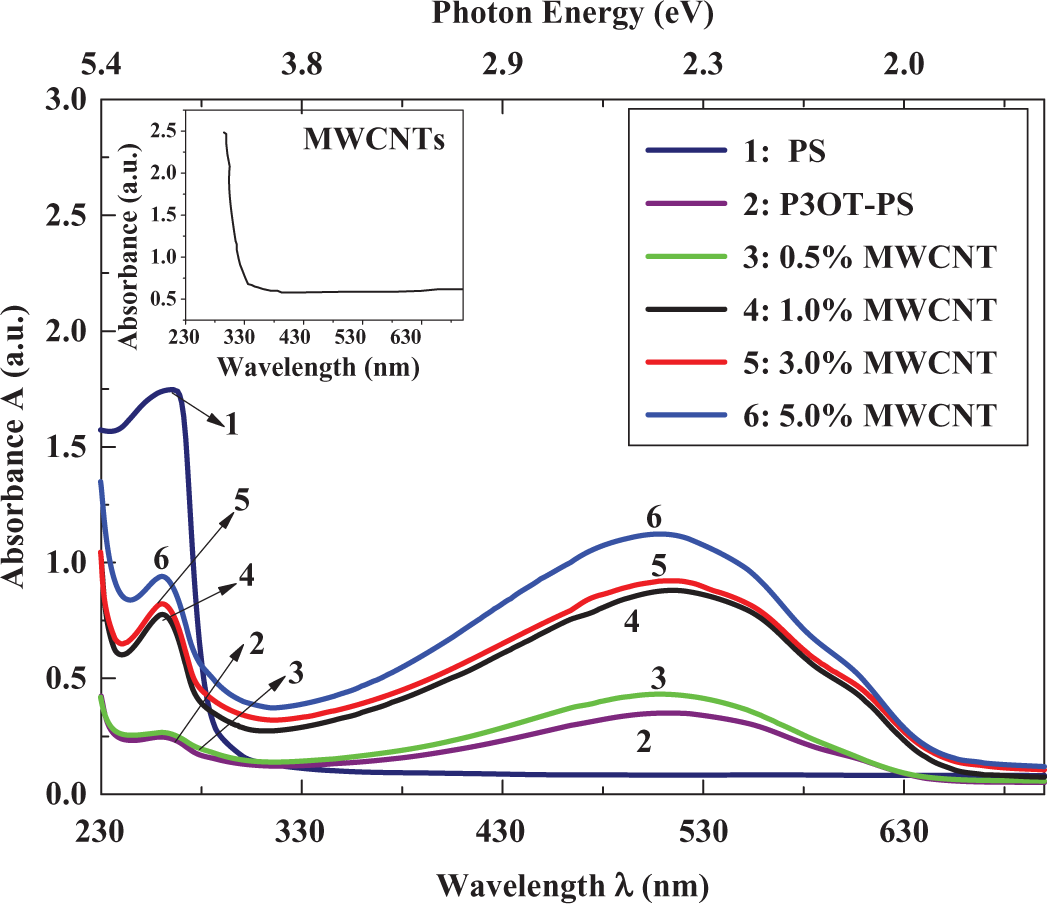
Ultraviolet–visible absorption spectra of polymer composite systems.
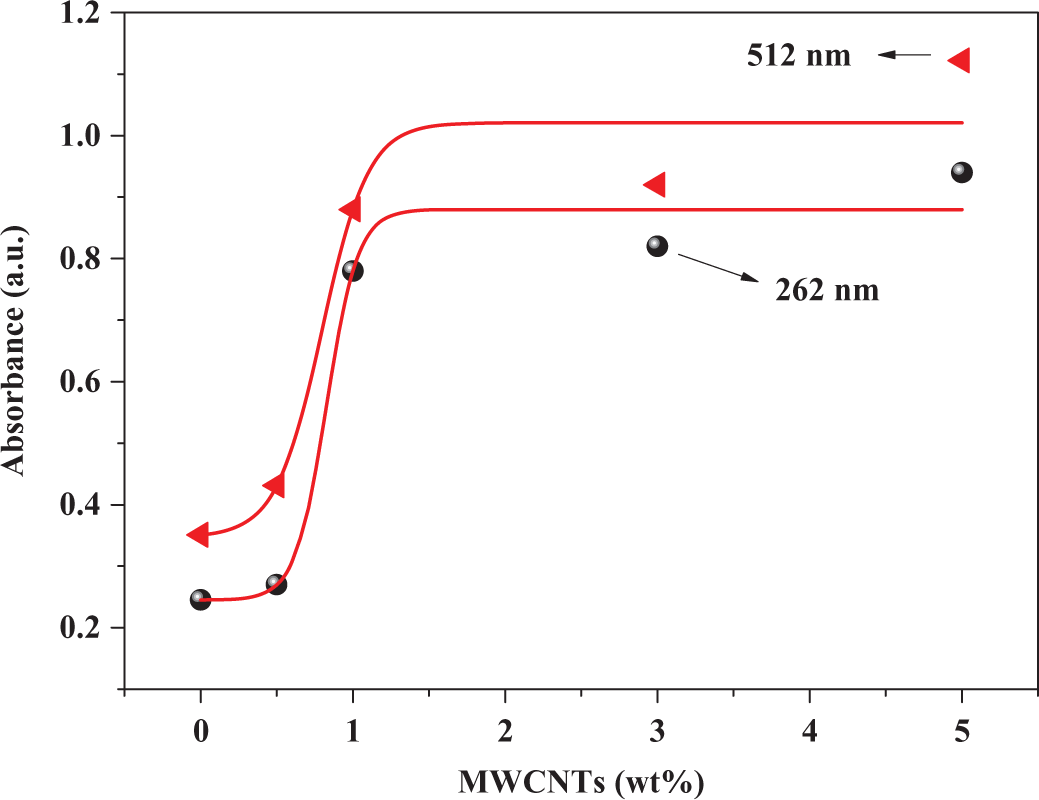
Absorbance versus MWCNTs weight fraction. MWCNTs: multiwalled carbon nanotubes.
The dependence of the absorption coefficient 
where
The absorption coefficient

where
Figures 5 and 6 show the plot of (
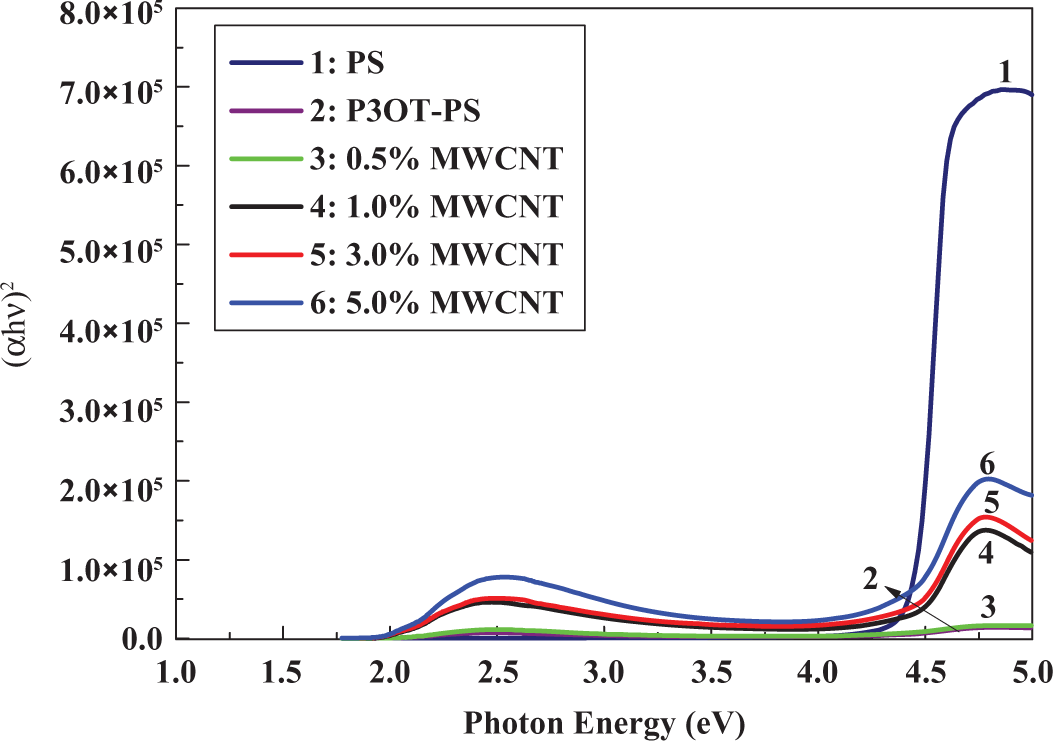
(
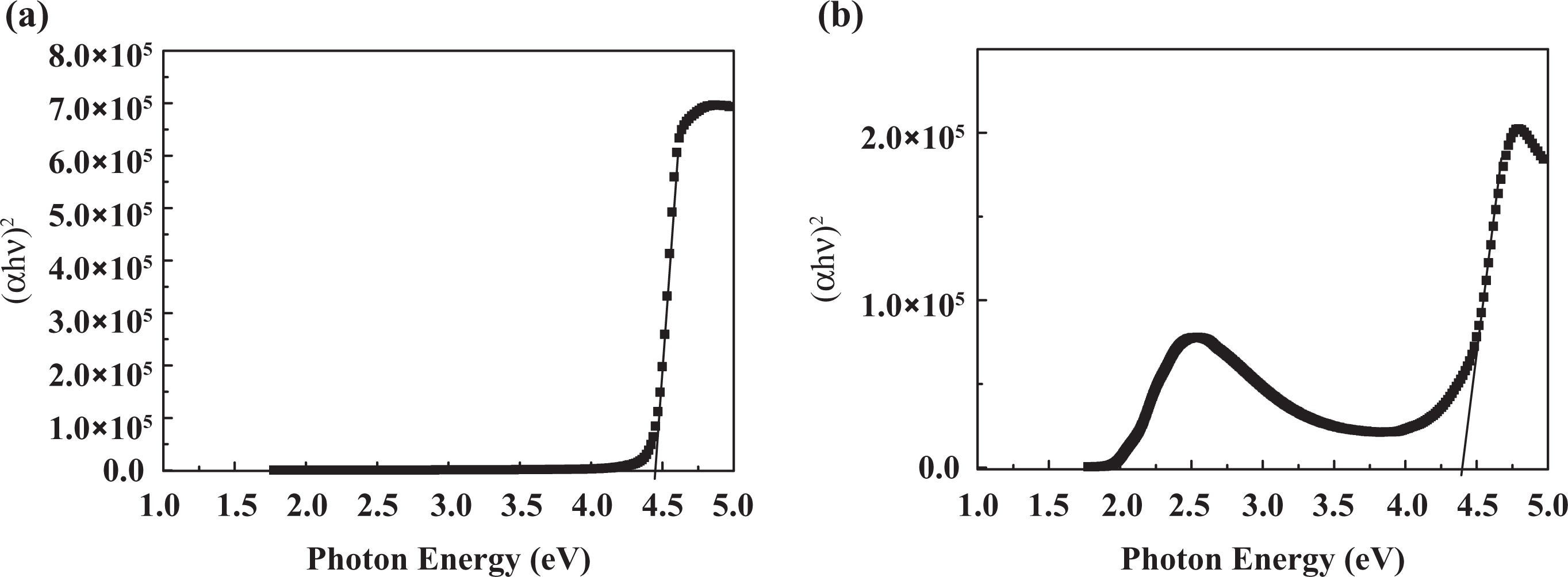
(
Experimental results for polymer composite systems.

MWCNTs: multiwalled carbon nanotubes; PS: polystyrene; P3OT: poly3-octylthiophene.
Electrical results
Characteristics
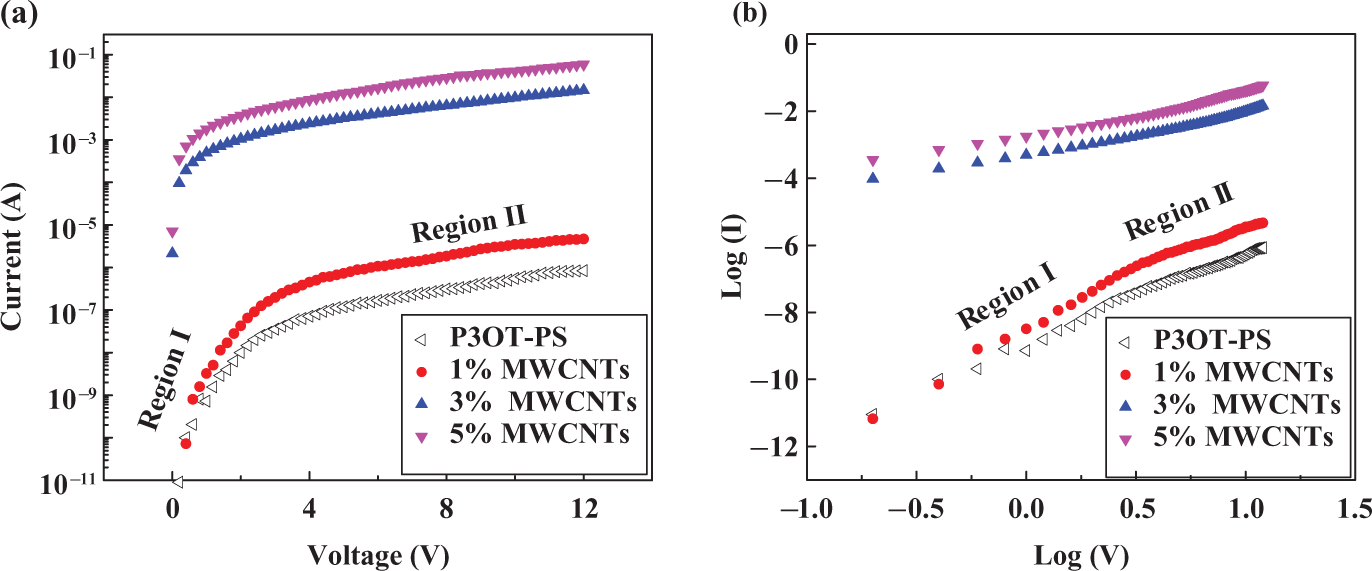
(a) Current (
The RT conductivity of PS and MWCNT-P3OT-PS composites was determined from the

where
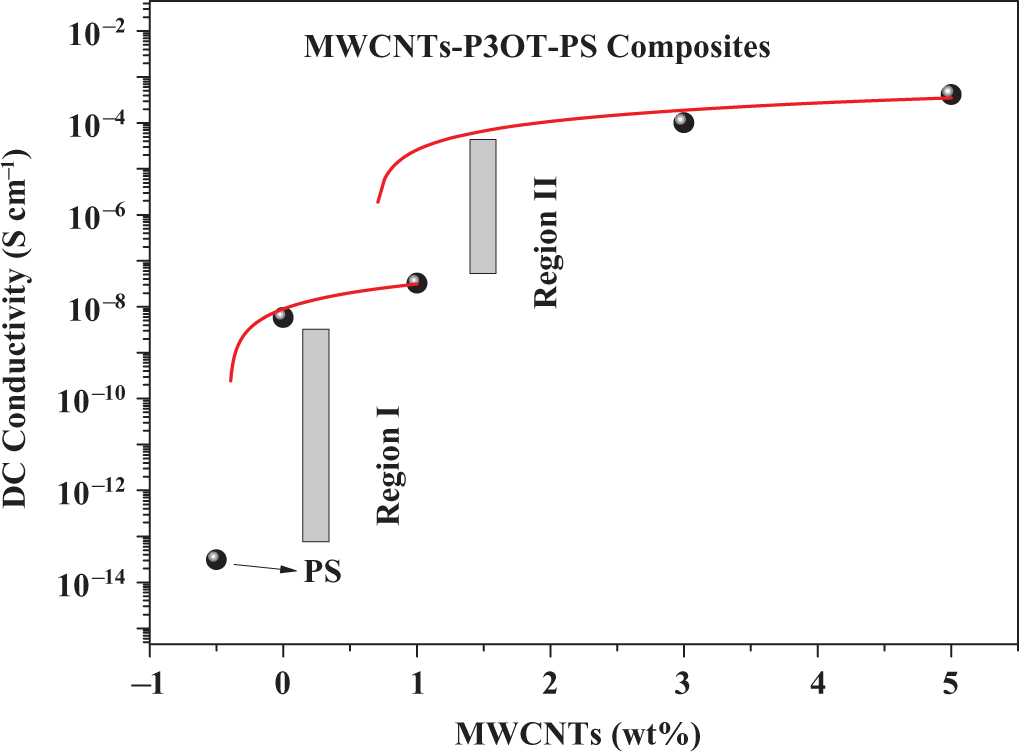
Variations in DC conductivity with MWCNTs weight fraction for MWCNT-P3OT-PS composites. DC: direct current; MWCNT: multiwalled carbon nanotube; PS: polystyrene; P3OT: poly3-octylthiophene.
Generally, conductivity depends on the polymer matrix and the filler materials. With regard to the polymer, it is controlled mostly by the conjugation and the dopants present in them. 27 If the filler material is CNTs, it is decided by its purity, alignment, and the concentration. 3,4,27,47 However, as shown in Figure 8, region I represents the first jump of conductivity upon blending of 30 wt% P3OT with 70 wt% PS; here the conductivity increases upto five orders of magnitude when compared with PS neat. At this region (i.e. region I), the material is converted from the insulator region to near semiconductor region. A peculiar observation shown in Figure 8, in region II, is that MWCNTs will significantly increase the electrical conductivity of the P3OT-PS matrix from the order of 10−9 S cm−1 (as in the case of P3OT-PS matrix) upto 10−4 S cm−1 (as in the case of 3 and 5 wt% MWCNTs-P3OT-PS), which means that addition of 3 or 5 wt% MWCNTs to P3OT-PS matrix will convert the material from near semiconductor limit to near conductor limit. We previously 27 found that the addition of 5 wt% single-walled carbon nanotubes to the P3OT matrix will increase the electrical conductivity of P3OT upto 10−4 S cm−1. In the present work, however, we show that the same limit can be reached even at low applied voltage (of 10 volts) by blending 70% commercial polymer such as PS with 30% P3OT polymer and adding 3 or 5 wt% CNTs.
In CNTs–polymer composites, the polymer layer in the internanotube connections presents highly resistive section in the electrical pathway, acting as a barrier to efficient carrier transport between the nanotubes and models fluctuation-induced tunneling for the conductivity.
27,46
Additionally, direct connection and overlapping of the CNT are not necessary, that is, nanotubes do not need to physically touch each other for conductivity. Nanotubes can just be close enough to allow for a hopping/tunneling electron effect; these mechanisms require the CNT–CNT distance to be less than 5 nm. As a result, a higher volume fraction of CNTs filler is needed to achieve electrical percolation threshold.
2
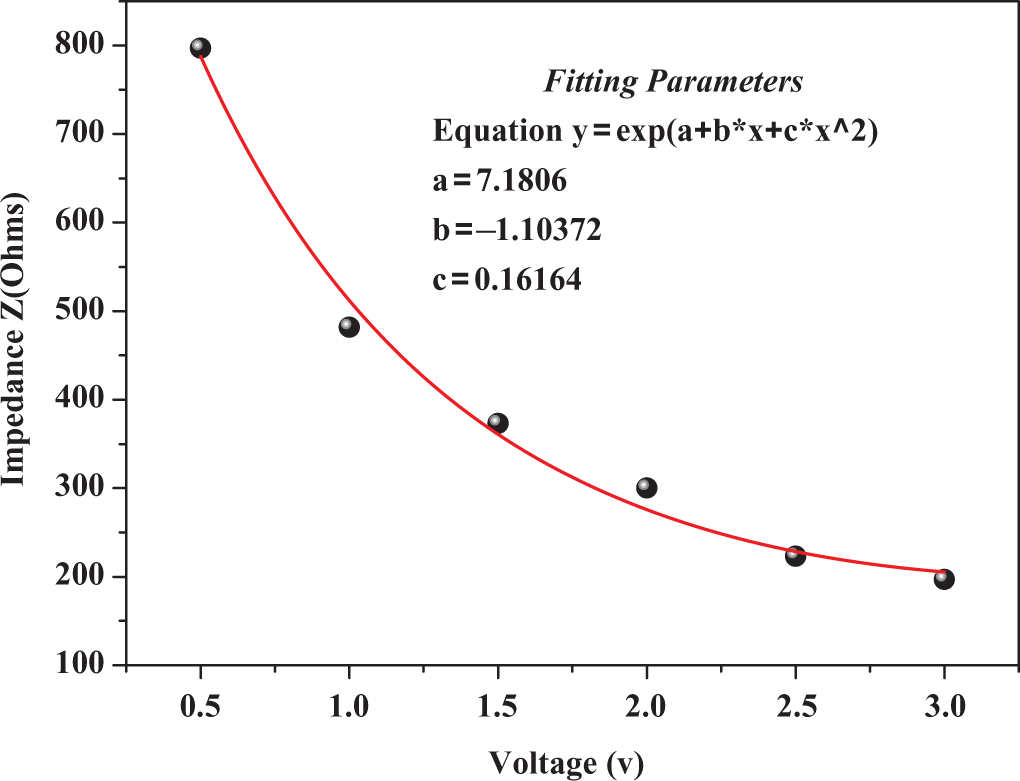
This, however, indicates that in addition to the above-mentioned conduction mechanisms, other mechanisms such as hopping and tunneling mechanisms are also included in the electrical conduction of composites. However, it is interesting here to study the effects of cyclic voltage on the
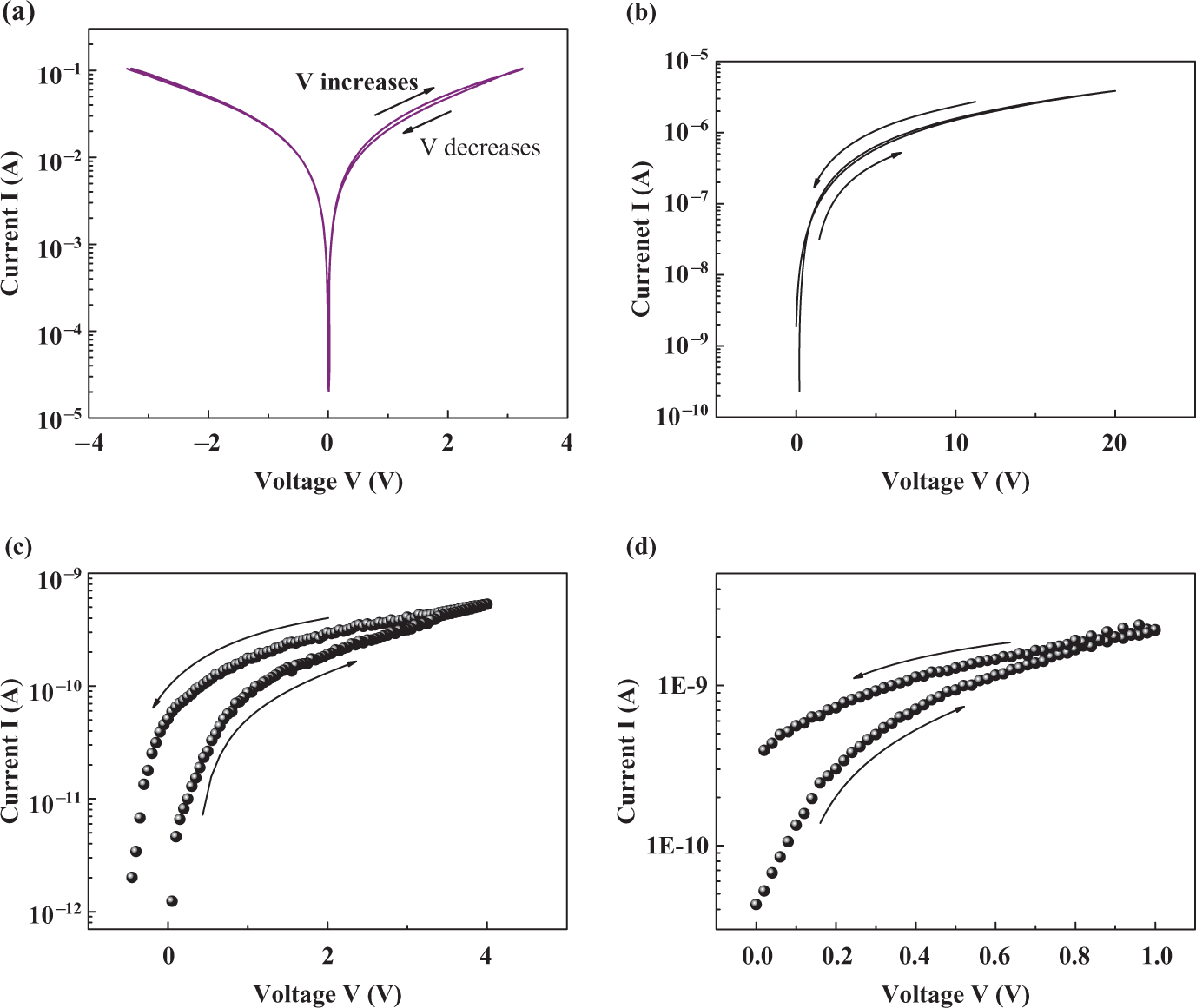
Effect of cyclic applied biasing voltage on the
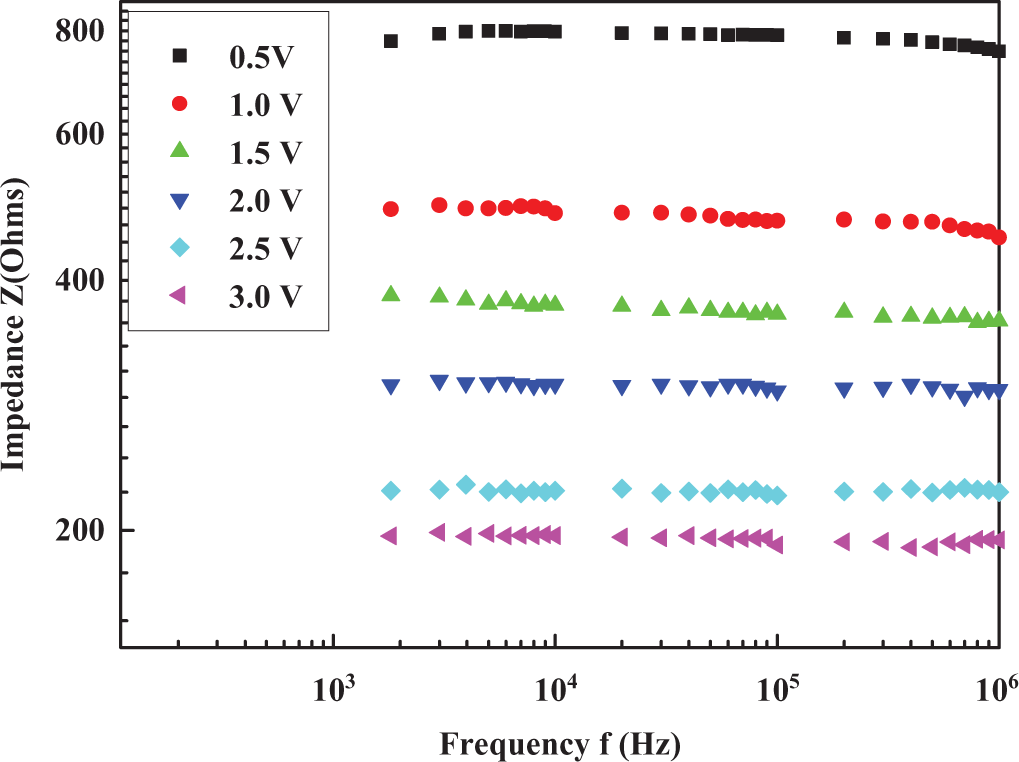
Impedance (
Impedance (
Conclusions
MWCNT-P3OT-PS composite films were prepared by casting technique. The dispersion of MWCNTs in 30 wt% P3OT-PS blend is improved through the functionalization of MWCNTs using UV–ozone. FT-IR results reveal the existence of carboxylic acid at the MWCNTs surface and there are no detectable interactions between composite components. Incorporation of MWCNTs upto 5 wt% into the blend matrix will enhance the UV/visible absorption of the P3OT-PS polymer blend and will not highly influence the optical energy gap of the blend matrix. Electrical results show that there are more than two mechanisms in the electrical conduction of these composites and their contributions depend on the applied voltage and composite compositions. At low MWCNTs content, capacitor element is the predominant one, while at high MWCNTs content resistor is the most effective element. This work shows that blending of 30 wt% P3OT with PS and incorporating 3 or 5 wt% MWCNTs into P3OT-PS matrix will increase electrical conductivity upto 10 order of magnitudes compared with PS and will convert composite material to conductor.
Footnotes
Funding
The authors would like to thank the Deanship of Scientific Research at King Faisal University, Al-Ahsa, KSA, for the financial support for this research, with the project ID 130168.
