Abstract
Commingled composites of polylactide (PLA) and seed flax fibers were prepared by carding and needle pressing of PLA and flax fibers to nonwoven mats followed by compression molding. The nonwoven mats were treated using two different types of cellulolytic enzymes, namely cellobiohydrolases and endoglucanases. Both the enzymes changed the properties of the seed flax fibers surface. The resulting composites were studied using scanning electron microscopy, mechanical testing and thermal analysis. The effects of these modifications were minor on the thermal and mechanical properties measured. However, in the scanning electron micrographs, a difference in the fiber pull-out behavior of the untreated and treated fibers was observed. Treated seed flax fibers showed an improvement in adhesion with PLA.
Introduction
The use of natural fibers as reinforcement in composites has been growing rapidly during the recent years. Especially, interest to combine natural fibers with bio-based polymer matrices is increasing since these composites can be biodegradable and independent of the availability of petroleum resources, but can still maintain similar mechanical and barrier properties as the existing petroleum-based polymers. 1 These benefits and concerns regarding the shortage of landfills are driving researchers to seek for alternative renewable resources to replace conventional composites, such as polyolefin and polyamide matrix composites.
Polylactide (polylactic acid (PLA)) has potential for use as bio-based thermoplastic matrix, which can be processed with conventional melt-processing equipments, 1 thus making its utilization easy. PLA is an aliphatic polyester derived by the fermentation of renewable sources, such as hydrolyzed corn or potato starch. The basic monomer of PLA is lactic acid (2-hydroxy propionic acid), which can occur in two different optically active enantiomers, the proportion of which determines the properties of the PLA formed and allows tailor-made adjustments of the polymer to meet the particular technical requirements of various products. 1 PLA matrix composites have been successfully manufactured with different plant-based natural fiber reinforcements, such as flax, hemp, kenaf and ramie. 2 –5 Some products have already entered successfully into the market. 3 Clearly less data exist on the utilization of fibers originating from agricultural residues, such as wheat straw, corn stovers and soy stalks, 6 which could offer further economical and environmental benefits, to reinforce PLA. The properties of reinforced PLA composites appear to vary considerably depending on the characteristics of the fiber raw materials. 3 Seed flax, a by-product of agro industry, is an easily exploitable natural fiber source. The fiber fraction often remains unutilized as raw material for the textile industry, due to its coarseness and short fiber length. However, this residue offers excellent possibilities due to its strength, which is comparable with flax fibers as technical-grade fiber for the production of composites. 7
Biocomposite performance depends both on the properties of the single components as well as the interfacial compatibility between the matrix and the fibers. 8 Although biocomposites have a number of attractive properties, such as possible biodegrability, low density, high specific strength and stiffness, the weak adhesion between the fibers and the polymer matrix is one of the main disadvantages. Natural fibers are typically hydrophilic, whereas most thermoplastics are hydrophobic. This leads to inherent incompatibility of the different components and thus to poor mechanical properties of resulting biocomposites. Especially, the adhesion between flax fiber and PLA matrix has been reported to be weak. 9,10 Improvement in adhesion properties by preprocessing of the fibers is one of the most important steps in increasing the mechanical properties of bio-based composites. Fiber treatments by chemical coupling agents, such as silane or alkali, have been observed to improve the mechanical properties of PLA composites. 4,11 –14 Also coupling agents, such as di-isocyanate, mixed with the matrix material have been shown to improve the mechanical properties of composites. 15
Due to the environmental concerns, sustainable enzyme-based technologies, that can replace hazardous chemicals, have been given increasing interest in the processing of natural fibers. Enzymes acting on the main carbohydrates in fibers have been used to obtain targeted modifications in the fibers. Pectinases have long been known to improve the retting process of flax stem, 16 and pectinase pretreatment has been used on bast fibers used as composite reinforcement. With flax, pectinase treatment with notable xylanase activity was observed to weaken the mechanical properties without improvement in the strength properties of the epoxy–flax composites. 17 However, improvements in tensile and textural characteristics of pectinase-treated hemp fibers in polypropylene matrix have been obtained, 18 possibly due to improved adhesion between the two components. Previous enzymatic work on flax has focused on bioscouring of linen fabrics by hydrolytic 19 –22 and oxidative 23,24 enzymes. Also chelating agents, such as ethylene diamine tetra-acetic acid, have been studied to extract the calcium ions that bind with the pectic polymers into a tight matrix in order to improve the properties of composites. 17,25 Other hydrolytic enzyme treatments can clearly alter the mechanical performance of flax fibers, and especially high cellulolytic enzyme loadings have been found to weaken the strength properties of flax. 19 In order to achieve targeted changes in the fiber properties, the pattern of cellulolytic enzymes can be selected. Two major groups of cellulases can be differentiated both by hydrolyzing the β-1,4 glycosidic linkages of cellulose and by liberating glucose, cellobiose and oligosaccharides. Endoglucanases (EGs) act on the amorphous areas of cellulose in fibers, whereas cellobiohydrolases (CBHs) also act on the crystalline cellulose. EGs preferentially depolymerize the easily accessible short fibrils on fiber surfaces and have been frequently used, for example, in pulp and paper and textile industries to polish the fiber surfaces or for biofinishing of cotton fabrics. 26 CBHs, on the other hand, have been observed to increase the outer fibrillation and delamellation of plant fibers and thus improve the refining of coarse mechanical fibers or depilling in biofinishing. 27,28 It can be expected that these treatments would affect the area of bonding between fiber and matrix.
The aim of the current study was to investigate the effects of two different types of cellulolytic enzymes, CBHs and EGs, on the properties of composites based on PLA and seed flax fibers. We targeted the modifications on the surface of the seed flax fibers, thus potentially promoting the adhesion between the natural reinforcement and the polymeric PLA matrix. Commingled biocomposites were prepared by carding and needle pressing followed by compression molding. Before compression molding, carded and needle-pressed nonwoven fabrics were treated with different enzymes. Prepared composites were characterized by mechanical testing (flexural test), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA) and scanning electron microscopy (SEM).
Experimental
Materials
Thermoplastic, biodegradable PLA was chosen as matrix of the composites. PLA was produced by Far Eastern Textile Ltd (Taipei, Taiwan) in fiber form. It has linear mass density of 1.65 dtex, fiber length of 3.8 mm, tensile strength of 3.5 cN/dtex and elongation at break of 50%. The seed flax (Linum usitatissimum) fibers were used as reinforcing fibers. The fibers of laser cultivar were cultivated on 2005 in the research farm of the University of Helsinki in Siuntio, Finland. The fibers had been field retted, decorticated and carded. The unwashed fibers were characterized with Lenzing Vibroskop to determine the fineness of the fibers and with Lenzing Vibrodyn to determine the tensile strength and elongation. The fibers were conditioned prior testing for a minimum of 48 h at 20°C and relative humidity of 65%. Deformation rate used was 2 mm/min and gauge length was 20 mm. For each strain, 20 single fibers were tested. Based on these measurements, the studied fibers had a linear density of 4.57 dtex, tensile strength of 4.59 cN/dtex and elongation at break of 2.27%. The enzymes studied for fiber pretreatments were a CBH I-rich preparation (CB10096A3), originating from Acremonium thermophilum, and an EG (Biotouch C800)-enriched preparation, both obtained from ROAL Ltd (Rajamäki, Finland).
Fabrication of nonwoven fiber mats
The fibers were first washed before composite processing (ISO 6330, procedure 1B). The nonwoven mats of PLA/linseed fiber were prepared using a carding machine followed by needle punching. Before carding, the flax fiber content was 50 wt%. In the final product, the flax content was lower than 50 wt% because during carding, the wooden parts and shives were separated. During carding, the fibers were mixed and formed into a uniform carded web.
After carding, the web was mechanically bound to a nonwoven form by needle punching, carried out with a needle punch machine, type MPR600 (Automatex Nonwoven, Pistoia, Italy). During the needle punching, the carded web became stiffer and tighter. The piston stroke of the needles was 150 push/min and the feeding rate was 200 cm/min.
The surface weight of the obtained carded and needle punched webs varied between 180 g/m2 and 240 g/m2, the average weight of the obtained carded and needle punched webs being approximately 200 g/m2.
Enzymatic treatments of the nonwoven mats
Hydrolysis experiments of nonwoven mats were performed in 2 L flasks (working volume 50 mL) in 50 mM Na–citrate buffer, pH 5.0, with a fiber consistency of 4%. The treatment time was 2 h and the temperature was 45°C. Enzyme loadings were based on total protein content, measured by the Lowry method. 29 Preliminary determination of the degree of hydrolysis (DH) was based on the treatments of flax fibers by measuring the amount of sugars released during the treatment, analyzed as reducing compounds with the 3,5-dinitrosalicylic acid reagent, using glucose as standard. 30 The DH obtained per dry matter of flax fibers was used to calculate the loading of enzymes for the nonwoven PLA–flax mats. After hydrolysis, the substrates were boiled for 2 min in order to inactivate the enzymes. Finally, the fiber mats were filtered and washed with distilled water. For the reference, PLA–flax mat incubated with 50 mM citrate buffer without enzyme was used.
Fabrication of composites
The composites from PLA/seed flax fiber mats were compression molded in a hydraulic press type MKH E60M (manufactured by K-S Teräsrakenne, Jämsänkoski, Finland). The molding temperature was 180°C, the molding time was 8 min and the molding pressure was 4 MPa. To control the thickness, aluminum strips of 1 mm were used as spacers.
Analysis of biocomposites
The prepared composites were characterized by flexural tests, DSC and TGA, and the fiber and the fracture surfaces were studied by SEM.
The flexural strength of the composites was measured by a three-point bending test method according to ASTM D 790 using Messphysik (midi 10-20/4×11, Messphysik Materials Testing Gmbh, Fürstenfeld, Austria) universal testing machine with 1 kN load cell. The thickness, width and length of flexural test specimens were 1 mm, 13 mm and 51 mm, respectively, and the span length was 25.4 mm. The crosshead speed used was 5 mm/min. At least five specimens were tested for each different material. Before testing, the samples were conditioned for at least 24 h at 23°C and 50% relative humidity.
The thermal properties of the composites were determined using a Netzsch DSC 204 (NETZSCH Gerätebau GmbH, Selb, Germany) F1 differential scanning calorimeter. Heating rate of 10°C/min and flow of inert gas (nitrogen) were applied. The samples were scanned from 20°C to 200°C. Glass transition (T g) and melting temperatures (T m) were determined from the data.
Thermal decomposition of the composites was analyzed using thermogravimetric analyzer TGA6 (The Perkin-Elmer Corporation, Norwalk, CT, USA). The heating rate was 20°C/min over the temperature range from 30°C to 1000°C under nitrogen atmosphere.
Analysis of the enzyme-treated fiber surfaces and the fracture surfaces of the composites were carried out by SEM (Philips Electron Optics, Eindhoven, The Netherlands). Before SEM analysis, the samples were sputtered with thin gold layer to avoid sample charging under the electron beam.
Results and discussion
Enzymatic treatments of flax fibers
Enzymatic treatments were performed on composite mats commingled of seed flax fibers and PLA in a ratio of 50:50. To adjust the DH, the dosages of enzymes used were first compared with pure linseed fibers. CBH-rich and EG-rich enzymes were then applied on the composite mats for 2 h, which resulted in various DHs between very low (below detection level) to about 4% of the dry matter of fibers. The hydrolysis products were mainly cellobiose and glucose and minor amounts of oligosaccharides (results not shown).
The treatment with CBH was shown to alter the surface morphology of the fiber distinctively, and the fibers were observed where the cellulose fibrils seemed to be loosened and partially hydrolyzed, thereby forming a “delamellated” structure. PLA fibers, visible in the background in Figure 1, showed no detectable effect on the surface after the treatment, as expected. When the DH remained below 1%, morphological alteration of fiber surfaces remained low (Figure 2). When the EG preparation was used resulting in a relatively high DH (3.7%), polishing or smoothening of the fiber surface was clearly visible and was a characteristic feature of EG-treated seed flax fibers (Figure 3). No visual damage, such as breaking of fibers, could be observed at this stage of hydrolysis. However, the DH obtained with dilute enzyme solutions targeting at 0.1% and 0.5% level of hydrolysis remained even lower than expected (below detection limit and 0.05%, respectively), possibly due to the interference of PLA, which may have partially covered the flax fiber surfaces, thereby diminishing the hydrolytic efficiency.

Visualization of the surface of seed flax fiber bundle by SEM. Reference material without enzyme treatment intensely covered by waxy material with polylactide fibers is visible in background (polylactide fiber marked with arrow). SEM: scanning electron microscopy.

Visualization of the surface of seed flax fiber after CBH treatment (degree of hydrolysis 0.6%). Arrow indicates “delamellated” feature of the fiber surface. CBH: cellobiohydrolase.

Visualization of the surface of seed flax fiber by SEM, polished surface of seed flax fiber after EG treatment (DH 1.3%). EG: endoglucanase; SEM: scanning electron microscopy; DH: degree of hydrolysis.
SEM observations of PLA–flax composites
The fracture surfaces of PLA composites, as investigated by SEM, gave a good indication of the adhesion between the fiber and the matrix. The fracture surface of PLA composites with untreated flax fibers can be seen in Figure 4 and that of treated fibers is shown in Figure 5. The fracture surface of the untreated composite is characterized with many fibers with no or very little polymer coating and the average pull-out length is longer than in the case of the treated fibers. For treated flax fibers, there is more evidence on the fibers of a polymer layer and pull-out length is shorter. These findings indicate a weaker adhesion between untreated fibers and matrix. Enzyme-treated flax fibers, pulled out from the matrix, were more intensely coated with the PLA matrix, especially when EG was used. The SEM analysis indicates that adhesion between matrix and fibers was improved especially by the EG treatment of the fibers but also to some extent by the CBH treatments. The positive improvement of adhesion between PLA and EG-treated fiber correlated well with the polishing effect of fiber surfaces by EG.

The SEM image of the fracture surface of a flexure test specimen of the PLA composite of untreated fibers. PLA: polylactide; SEM: scanning electron microscopy.

The SEM images of fracture surface of a flexure test specimen of the PLA composite of enzyme-treated fibers. PLA: polylactide; SEM: scanning electron microscopy.
Properties of PLA–flax composites
Three-point bending test of the composites
The flexural strength of the tested flax/PLA composites is shown in Table 1. Enzymatic treatment marginally improved the flexural strength of the composites, when low doses of CBH were applied to linseed fibers. When the EG- and CBH-enriched enzymes were dosed in excessive amounts, flexural properties remained at the same level as without enzymatic treatments, with a minor loss of strength. The highest flexural strength was obtained with PLA–flax nonwoven treated with CBH resulting in a low DH of 0.5%.
Flexural strength of PLA-based composites.

aDenotes the degree of hydrolysis (% of dry weight of fibers).
PLA: polylactide; CBH: cellobiohydrolase; EG: endoglucanase.
Differential scanning calorimetry
The melting temperatures (T m) and glass transition temperatures (T g) were determined with DSC. The results of all PLA–flax composites and neat PLA are summarized in Table 2. No clear differences in the melting temperatures between the composites could be observed. However, the melting temperature of PLA-based composites was decreased (1–5°C) when compared to PLA matrix. After enzyme treatments with EG and the highest dose of CBH treatments, the melting temperature was decreased less (1–2°C) in the treated samples than in the composites with untreated fibers.
Thermal properties of PLA and the flax composites measured by DSC.

PLA: polylactide; CBH: cellobiohydrolase; EG: endoglucanase; DSC: differential scanning calorimetry; DH: degree of hydrolysis.
Thermogravimetric analysis
The weight loss as a function of temperature is shown in Figure 6. The neat PLA underwent thermal degradation in a single step between 310°C and 390°C. The residual ash content of neat PLA in 400°C was 3 wt%. The PLA–flax composites had two weight loss steps: the first step below 120°C was obviously due to the removal of absorbed moisture and the second step of degradation occurred from 290°C to 400°C. The residual ash content of the composites at 400°C was about 11–15 wt%, arising from the flax component. The thermal degradation of PLA–flax composites began about 20°C earlier than in neat PLA (Figure 6). This is due to the lower thermal stability of flax fibers. The TGA data of different PLA–flax composites and PLA are given in Table 3. The results show the decomposition temperature at the weight loss of 1%, 10%, 30%, 60% and 90%. There is a weak trend of composites with treated fibers showing a smaller amount of residue at higher temperatures. This result is consistent with the knowledge of enzyme treatments removing some chemical entities from the surface.
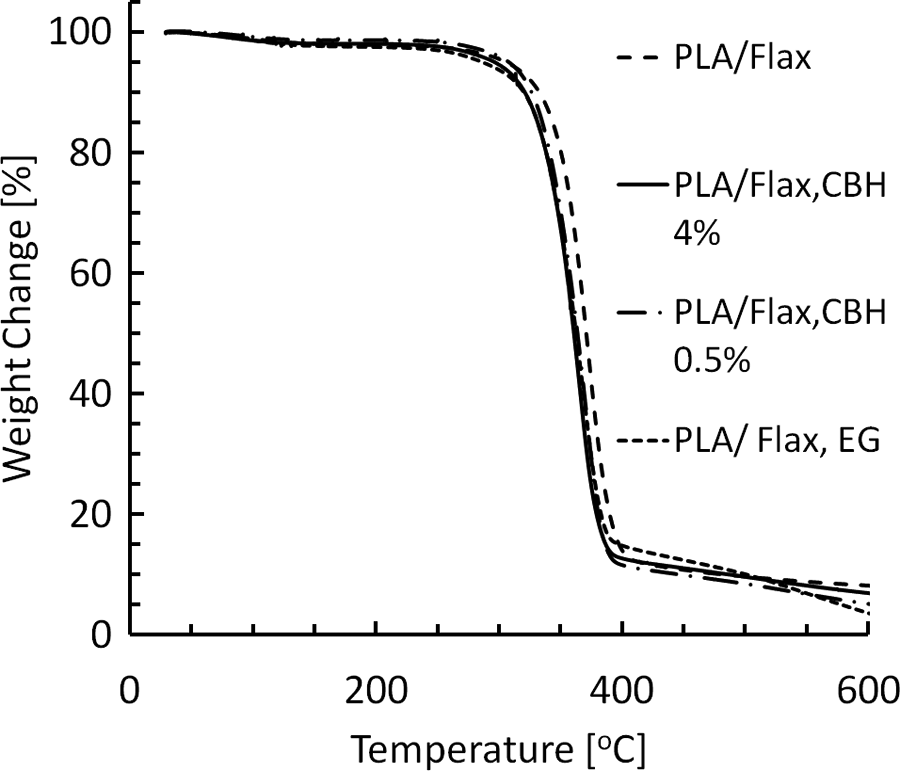
Thermogravimetric results showing the decomposition temperatures of PLA and PLA–flax composites. PLA: polylactide.
Thermal stability data of the samples.
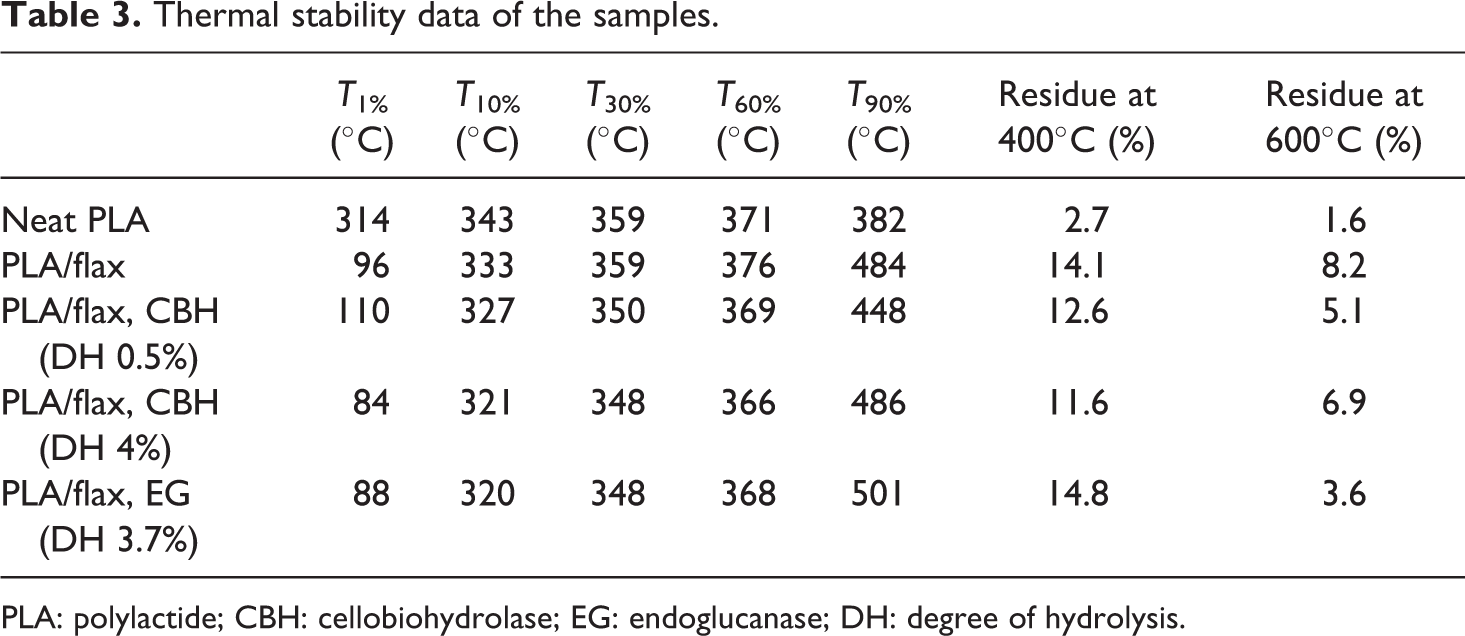
PLA: polylactide; CBH: cellobiohydrolase; EG: endoglucanase; DH: degree of hydrolysis.
Conclusions
Here, we prepared commingled composites of PLA and seed flax fibers. Seed flax fibers were treated using two different types of cellulolytic enzymes, CBH and EG, introducing site-specific changes in the properties of the linseed flax fibers surfaces. The effect of these modifications was minor on the thermal and mechanical properties measured. However, in the scanning electron micrographs, a difference in the fiber pull-out behavior of the untreated and treated fibers was observed, showing that the treatments had an effect on the reinforcements. There is a clear difference in the adhesion between unmodified and modified seed flax fibers and PLA fibers.
Footnotes
Acknowledgement
The authors thank the Suominen Nonwovens (Nakkila, Finland) for supplying PLA fibers.
Funding
This work was supported by the Academy of Finland, project no. 1127961.
