Abstract
A series of maleic anhydride grafted poly(lactic acid) (PLA-g-MAH) was prepared by mixing PLA, dicumyl peroxide (DCP) and maleic anhydride (MAH). Effects of DCP and MAH concentration on the grafting percentage were determined. PLA composites were prepared via melt mixing with halloysite clay (HNC; 3 wt%) and various amount of PLA-g-MAH (4–10 phr) using internal mixer followed by compression molding. Properties of the PLA composites were characterized using three-point bending flexural tests, scanning electron microscopy and differential scanning calorimetry. It was found that the grafting percentage of PLA-g-MAH was influenced by both DCP and MAH concentrations; however, DCP showed more profound effect. By the addition of PLA-g-MAH, the adhesion between PLA and HNC improved, which can be manifested by the enhancement in flexural properties. Degree of crystallinity of PLA/HNC increased significantly by the addition of PLA-g-MAH.
Introduction
Poly(lactic acid) (PLA) derived from renewable resources (e.g. sugar beets, sugarcanes, corn, wheat and etc.) has become popular owing to their sustainability, biocompatibility, biodegradability, fabric ability and superior transparency. 1 These properties make it to be a potential material in the packaging, medical devices and tissue engineering applications. However, there are some limitations of PLA, for example brittleness, slow crystallization and low thermal stability.2–5 Basically, there are two approaches to improve the properties of PLA, which include addition of fillers (micro- and nano-), and thermoplastic blending.6–8
Halloysite is a naturally occurring aluminosilicate (Al2Si2O5(OH)4·2H2O) with a predominantly hollow tubular structure mined from natural deposits in countries such as America, China, New Zealand, France and Belgium. The structure and chemical composition of halloysite are similar to those of kaolinite, but the unit layers in halloysite are separated by a monolayer of water molecules giving a basal (d 001) spacing of approximately 10 Å. 9 Halloysite exhibits unique surface chemical property due to the multilayer structure with only a few hydroxyl groups located on the surface. 10
Organic/inorganic hybrid composites have received significant attentions from both academic and industrial area. Recently, various PLA-based composites, prepared by the addition of nanofillers such as organo-montmorillonite,5,11,12 mica, 13 nanocalcium carbonate,14–16 titanium oxide nanoparticles,17,18 cellulose nanofiber19–21 and carbon nanotube22,23 exhibited remarkable improvement in mechanical and thermal properties, dimensional stability, gas permeability, physicochemical behaviors and biodegradability compared to that of neat PLA.
Though the hybridization of PLA and halloysite clay (HNC) can be a potential class of materials, the incompatibility between the two phases and the embrittlement of the resulting nanocomposites always remained the top priority issue. One of the common strategies to improve the compatibility between the polymer and filler is by the addition of compatibilizer and/or coupling agent. It has been documented that the addition of maleic anhydride–grafted polymer such as maleated polypropylene,24,25 maleated polyethylene–octene elastomer, 26 maleated PLA5,27 and maleated polyethylene28,29 has improved the interfacial adhesion of the respective polymer–filler systems, and consequently enhanced their mechanical properties.
In this work, we prepare a series of maleic anhydride–grafted PLA (PLA-g-MAH) by blending PLA, dicumyl peroxide (DCP) and maleic anhydride (MAH). The effects of DCP and MAH on the grafting percentage of the PLA are investigated. We also aim to report the effects of PLA-g-MAH on the mechanical and thermal properties of PLA/HNC composites.
Experimental
Materials
PLA (IngeoTM 3051D) was purchased from NatureWorks LLC®, USA. The specific gravity and melt flow index of the PLA are 1.25 and 25 g/10 min (2.16 kg loads, 210°C), respectively. The glass transition temperature (T g) and melting temperature (T m) of the PLA are approximately 65°C and 165°C, respectively. HNC with diameter of 30–70 nm, length of 1–3 μm and surface area of 64 m2/g was purchased from Sigma-Aldrich (M) Sdn Bhd, Malaysia. MAH and DCP were supplied by Sigma-Aldrich (M) Sdn. Bhd, Malaysia.
Preparation of PLA-g-MAH
PLA was premixed with the MAH and DCP prior to the extrusion process. At the first stage, the MAH content was varied from 4 phr to 10 phr with fixed amount of DCP (see Table 1, formulation for PLA-g-MAH (A–D)). At the second stage, the amount of DCP varied by fixing the amount of MAH (i.e. 10 phr), which was labeled as PLA-g-MAH (D–F). The melt mixing of PLA, DCP and MAH was performed using a single-screw extruder (Brabender, Melchers, Germany). The extrusion temperature was set in the range of 165–180°C, while the screw speed was set at 40 r/min.
Composition of PLA-g-MAH.
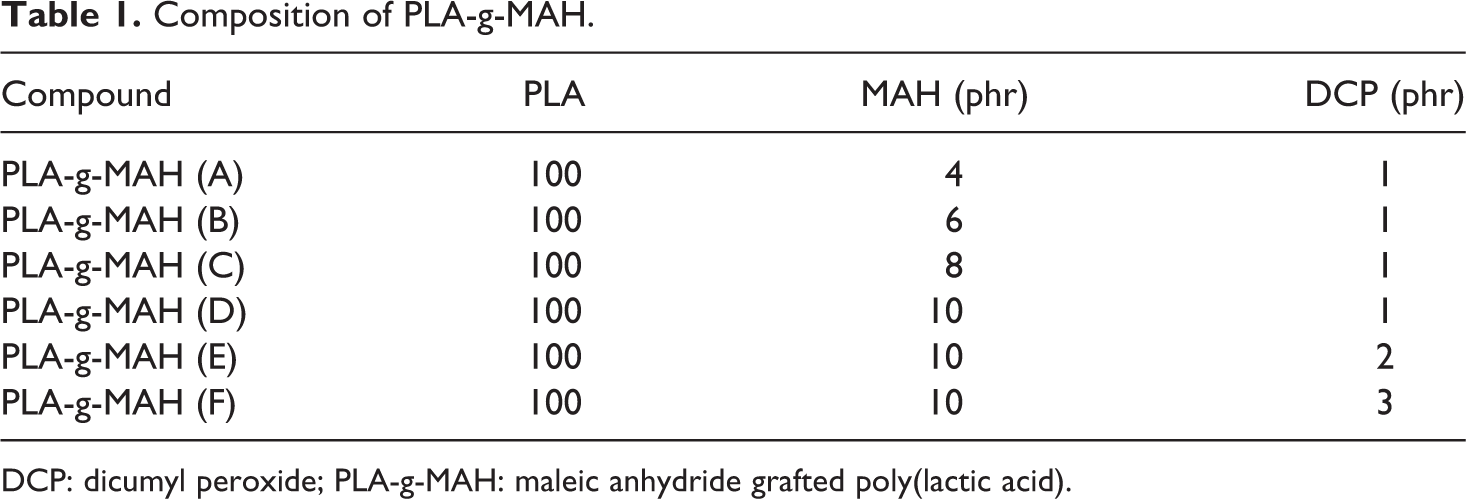
DCP: dicumyl peroxide; PLA-g-MAH: maleic anhydride grafted poly(lactic acid).
Purification of PLA-g-MAH
PLA-g-MAH pellets were first dissolved in toluene at 110°C under reflux until a clear solution with no precipitate was obtained. These solutions were then precipitated in methanol and filtered in order to obtain the purified PLA-g-MAH. The precipitated PLA-g-MAH was then further washed with methanol to remove residual MAH and DCP. Further, the obtained PLA-g-MAH was dried in a vacuum oven for 24 h.
Determination of grafting percentage
The grafting percentage analysis was performed according to the method adopted from Nabar et al. 30 The PLA-g-MAH was dissolved with toluene. Five milliliters of distilled water were added to the hot solution to hydrolyze the anhydride groups. The hot solution was then titrated immediately against 0.025 N of ethanolic KOH in the presence of 4 drops of 1% phenolphthalein in ethanol as an indicator. The blank titration was also performed as a control. The grafting percentage was calculated using equation (1).

G r is the grafting percentage of MAH, M is the molecular weight of MAH (98.06 g/mol), C is the concentration (mol/L) of ethanolic KOH, V 0 is the volume (ml) of ethanolic KOH used to titrate the blank sample, V is the volume (ml) of ethanolic KOH used to titrate the sample and m is the weight (g) of the sample.
Preparation of PLA composite
Prior to melt compounding, PLA, HNC and PLA-g-MAH (F) were dried in an oven at 80°C for 24 h. The compounding of PLA, HNC and PLA-g-MAH (F) were performed using an internal mixer (Haake Rheomic Polydrive R 600/610, USA). The mixing temperature and time are 180°C and 15 min. respectively. Table 2 shows the designation and composition of PLA composites. Compression molding of PLA composites into 1 mm thickness were carried out using a hot plate hydraulic press (Gotech, Taiwan) at 180°C. The compression and cooling time are 4 min and 3 min, respectively.
Material designation and composition of PLA and its composites.
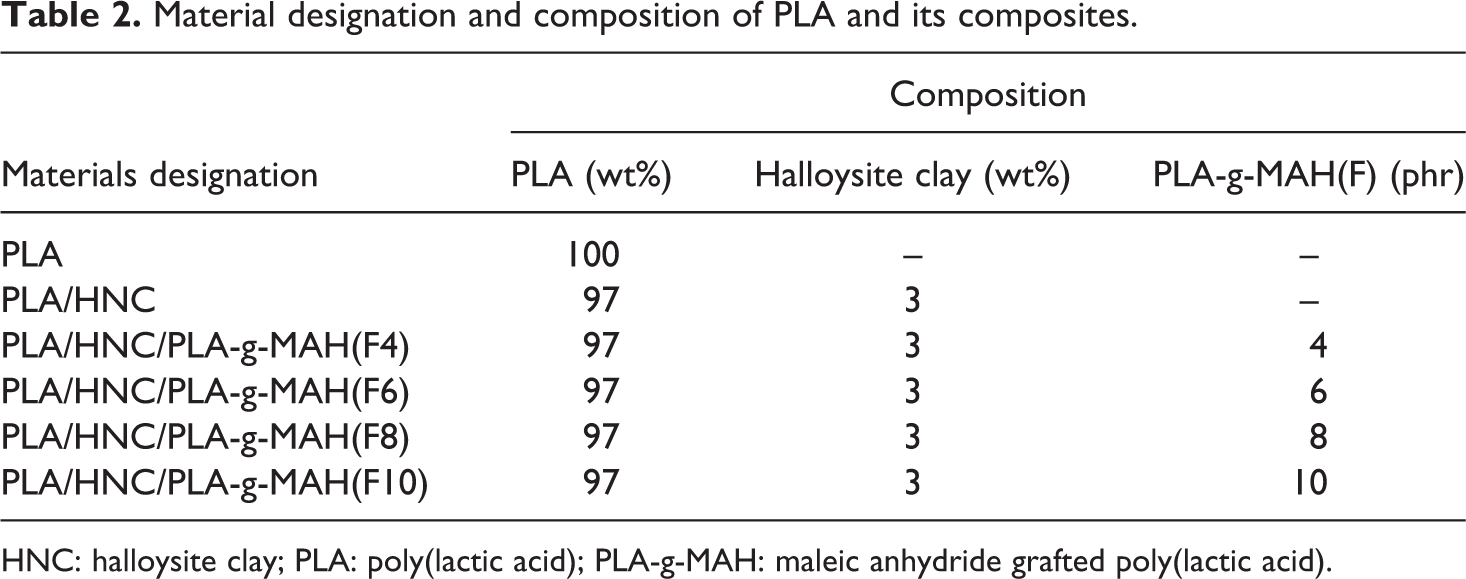
HNC: halloysite clay; PLA: poly(lactic acid); PLA-g-MAH: maleic anhydride grafted poly(lactic acid).
Measurements
Three-point bending flexural tests were conducted with an Instron testing machine (model 3366, USA) according to American Society for Testing and Materials (ASTM) D-790. Flexural tests were performed at a testing speed of 5 mm/min and a span length of 32 mm. Flexural modulus and flexural strength were determined.
Fourier transform infrared (FTIR) spectroscopy was performed using Spectrum One FTIR Spectrometer (Perkin Elmer, USA) from 400 cm−1 to 4000 cm−1 with a resolution of 2 cm−1 for 32 scans.
Differential scanning calorimetry (DSC) was conducted using Mettler-Toledo DSC 1 (Mettler-Toledo, USA). The specimens were scanned from 30°C to 190°C at a heating rate of 10°C/min and held for 1 min at 190°C. Then, they were cooled from 190°C to 30°C at a cooling rate of 10°C/min and held for 1 min at 30°C. Second scanning was performed similar to the first scanning in order to erase the thermal history. The degree of crystallinity (χ c) of PLA composites was calculated using equation (2)

where χ c is degree of crystallinity; ▵H m is the heat of fusion of the sample; ▵H f corresponds to the heat of fusion for 100% crystalline material and w PLA is the net weight fraction of the PLA. The heat of fusion of 100% crystalline PLA (▵H f) is approximately 93.6 J/g.31,32
Morphological properties of PLA composites were investigated using a field emission scanning electron microscope (FESEM, Zeiss Supra 35VP, Carl Zeiss, Germany). The accelerated voltage used was 5 kV. Prior to FESEM examination, the specimen surfaces were coated using aurum/palladium alloy in order to prevent electrostatic discharge.
Results and discussion
Grafting percentage on PLA
Figure 1 shows the grafting percentage of PLA as a function of MAH. In general, grafting percentage indicates the percentage of MAH grafted to the PLA chain. From Figure 1, it can be observed that increasing the concentration of MAH leads to an increase in the grafting percentage. The grafting percentage increases from 0.5% to 2.0% as the MAH concentration increased from 4 to 10 phr. From Figure 2, it can be seen that the grafting percentage increased as the DCP content increased. Note that, at 3 phr of DCP, the grafting percentage achieved 3.8%. This is attributed to the fact that, the higher amount of initiator (DCP) will generate higher amount of macroradicals, which could promote more reactive sites in PLA macromolecule chain for the MAH grafting. Accordingly, higher DCP content gives higher grafting percentage. Similar observations have been reported by Bettini and Angnelli, 33 Li et al. 34 and Li et al. 35 Note that the concentration of DCP has more profound effects on the grafting percentage than the MAH concentration (c.f. Figures 1 and 2). This indicates that the initiator concentration directly affects the total free radical concentration.
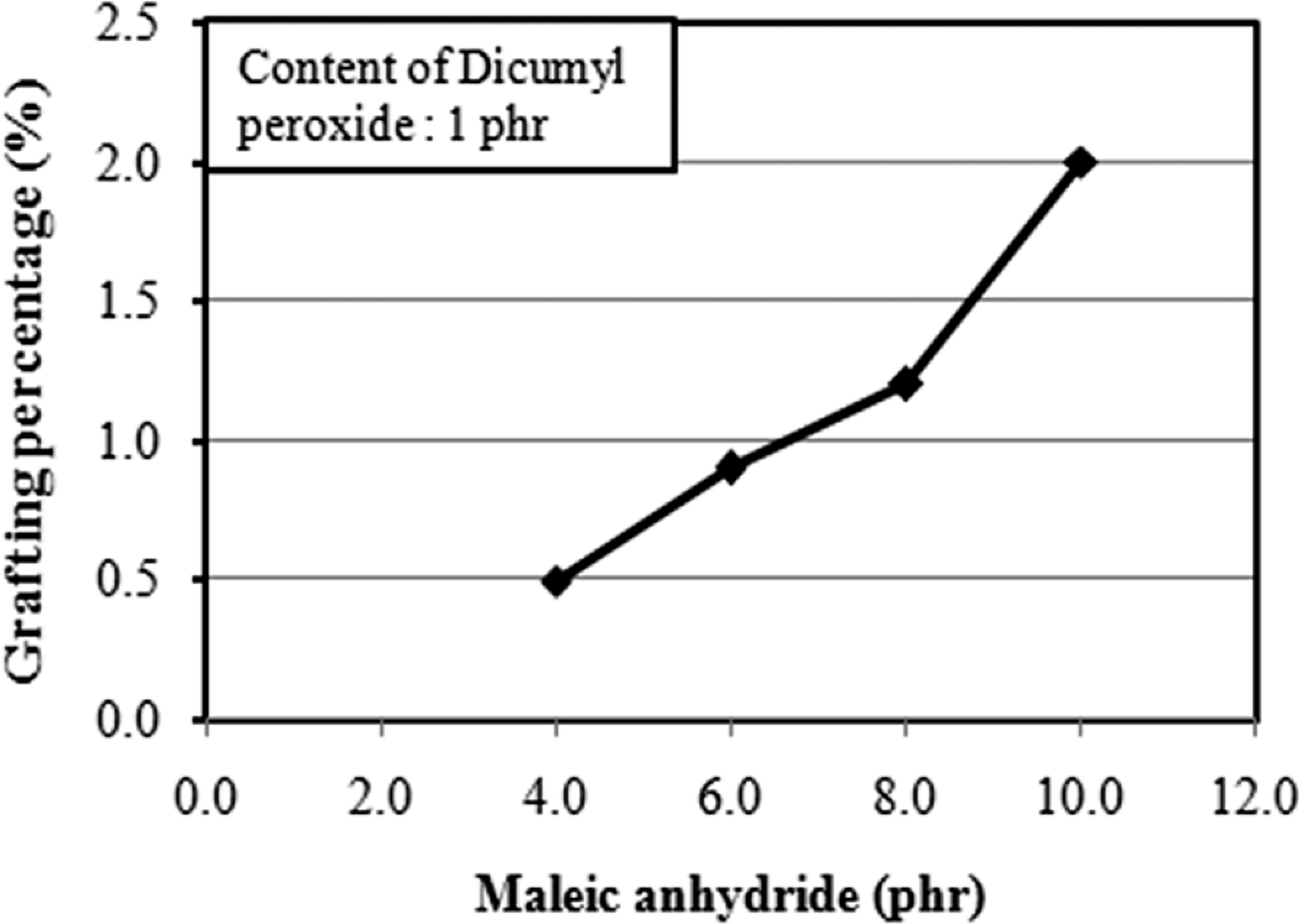
Effect of maleic anhydride (MAH) concentration on the grafting percentage of poly(lactic acid) (PLA).
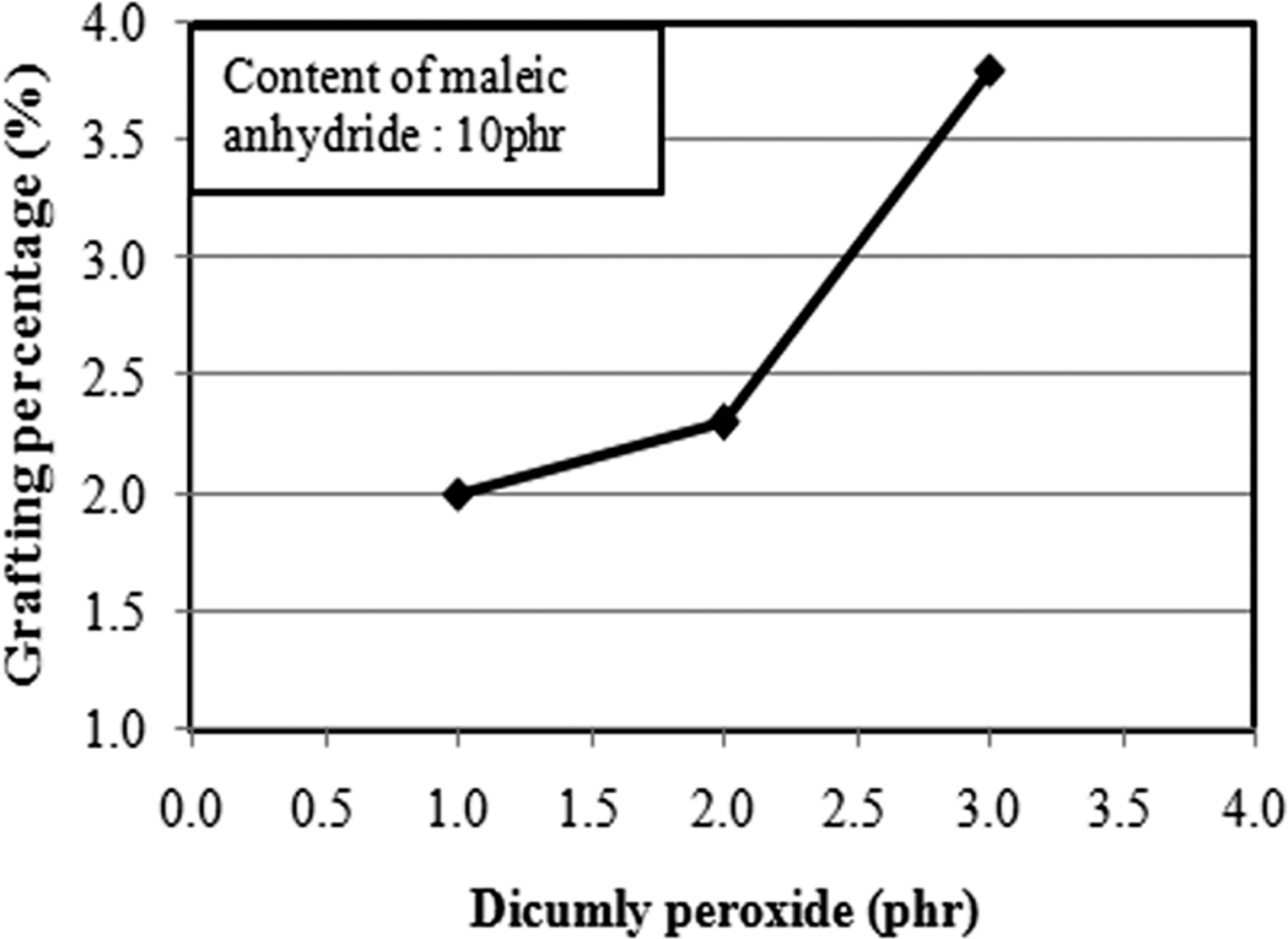
Effect of dicumyl peroxide (DCP) content on the grafting percentage of poly(lactic acid) (PLA).
FTIR study
Figure 3 shows the FTIR spectra of PLA, MAH and various PLA-g-MAH. The 2998 cm−1 and 2949 cm−1 are associated with –C–H– stretching. The peak at 1750 cm−1 is the assignment of –C=O carbonyl stretching. The peaks at 1451 cm−1 are assigned as –C–H– deformation, while 1381 cm−1 and 1359 cm−1 are corresponding to C–O–H band. The peaks in the range of 1200–1040 cm−1 and 955–867 cm−1 are attributed to –C–O– stretching and –C–C– stretching, respectively. It has been documented by Sclavons et al. 36 that the MAH is a symmetric molecule in structure with symmetrical C=O stretching which can be assigned at 1850–1700 cm−1. In this study, the peak of 1704 cm−1 is corresponding to –C=O stretching of MAH. While, the peaks detected at 1632, 1585 and 1563 cm−1 are associated with –C=C stretching. The infrared absoprtion frequency at 1457 and 1428 cm−1 can be assigned to the –CH2– deformation and C–O stretching, respectively. The 1260 cm−1 and 1217 cm−1 peaks are attributed to the –C–O stretching of anhydrides group from MAH. The bands of 1780 cm−1 was corresponding to the succinic anhydride group, while 1850 cm−1 can be assigned to the symmetric (strong) and asymmetric (weak) stretching vibrations of C=O.31,34 From Figure 3, referring to the FTIR spectra of various PLA-g-MAH, one may observe that the intensity of the absorption band at 1750 cm−1 increased from PLA-g-MAH (D) to PLA-g-MAH (F). This indicates that the increase in -C=O carbonyl stretching for the PLA-g-MAH are attributed to the increasing of DCP content. Several new peaks were detected in the range of 1640–1610 cm−1, 1586 cm−1 and 1460 cm−1 for these PLA-g-MAHs. The absorption bands at 1640–1610 cm−1 are corresponding to the C=O stretching which are originated from the anhydride group of MAH. The peaks at 1586 cm−1 and 1460 cm−1 are assigned to the C=C stretching and –CH2– deformation, respectively. Also, the peak at 1205 cm−1 is observed for the PLA-g-MAH, which is corresponding to the C–O–C stretching from the anhydride group of MAH. Based on these results, it is believed that the MAH is successfully grafted on the PLA chains.
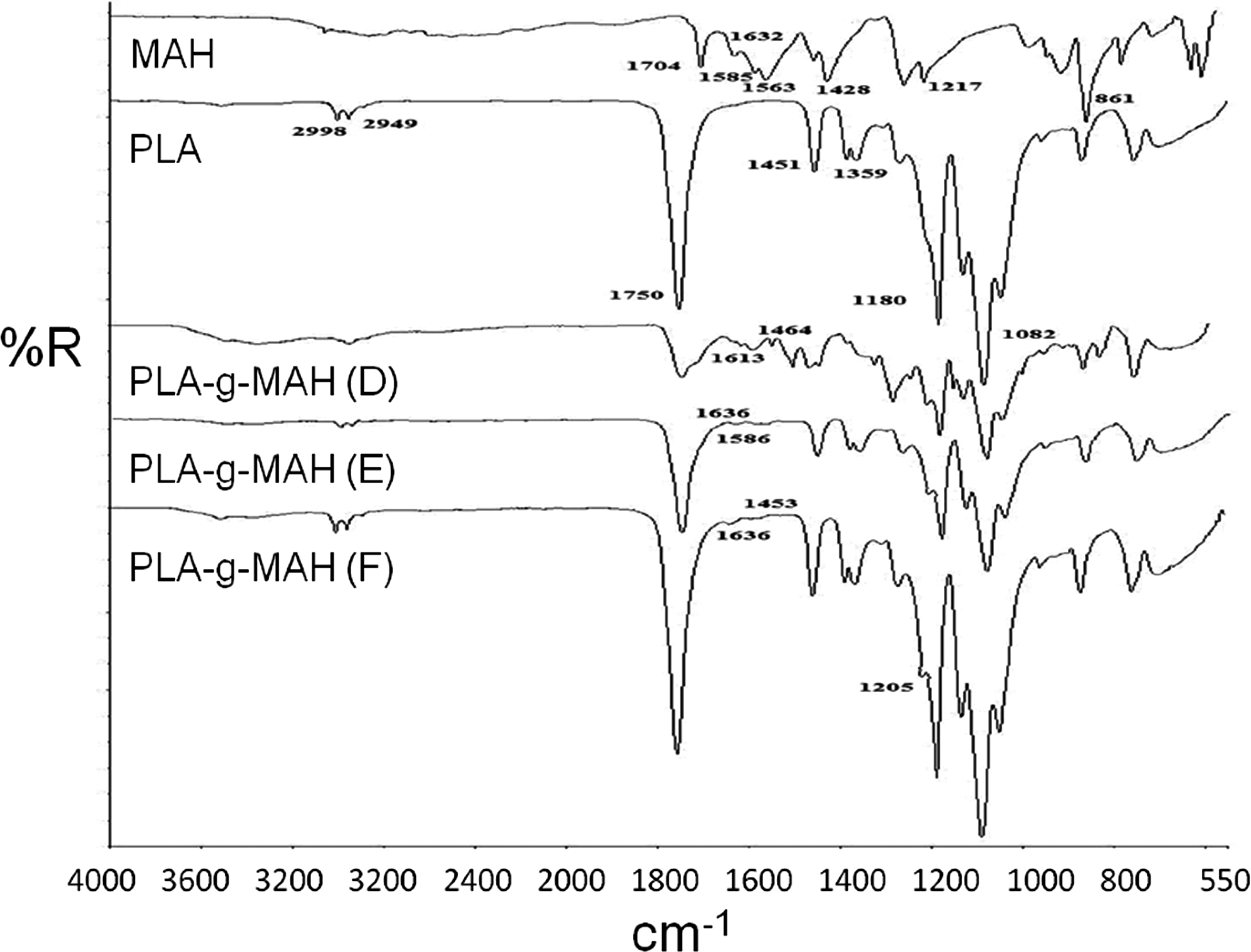
Fourier transform infrared (FTIR) spectra of poly(lactic acid) (PLA), maleic anhydride (MAH) and various PLA grafted PLA (PLA-g-MAH).
Flexural properties
The PLA-g-MAH(F) prepared using 10 phr MAH and 3 phr DCP achieved the highest grafting percentage in this work. Thus, the effects of PLA-g-MAH(F) on the flexural properties of PLA/HNC composites are investigated. Figures 4 and 5 show the effects of PLA-g-MAH(F) on the flexural modulus and strength of PLA composites. As expected, the addition of HNC increased the flexural modulus of PLA. This is attributed to the reinforcing effects and rigidity of HNC. Interesting to note that, the flexural modulus of PLA/HNC composites was further increased as the content of PLA-g-MAH(F) increases. The percentage of increment of PLA/HNC/PLA-g-MAH(F10) composites is about 27.6%. This may be attributed to the bulkiness and stiffness of the MAH group in the PLA composites. According to Al Roomi and Hussain, 37 the presence of bulk anhydride moiety group can lead to the increment of the modulus. The improvement in modulus could be also associated with the improvement in interfacial interaction between PLA and HNC in the presence of PLA-g-MAH. According to Modesti et al., 38 the elastic modulus of PP/clay composites were increased by the presence of compatibilizer owing to the greater interaction between filler and polymer. This implies that the stress is much efficiently transferred in between polymer and inorganic filler. Another possible reason for the enhancement in modulus can be related to the increase in crystallinity of PLA composites by the addition of PLA-g-MAH. Figure 5 shows the effect of PLA-g-MAH loading on the flexural strength of PLA/HNC composites. Although HNC fillers could contribute to the improvement in stiffness and modulus, however the weak interfacial adhesion and low compatibility between the HNC and PLA matrix leads to slight reduction in flexural strength. On the other hand, the flexural strength of PLA/HNC consisting of PLA-g-MAH(F) is higher than PLA/HNC composites. The percentage of increment of PLA/HNC/PLA-g-MAH(F10) composites is about 14%. The enhancement in flexural strength is attributed to the improvement in interfacial bonding between PLA and HNC by the incorporation of PLA-g-MAH. According to Fowlks and Narayan, 39 the enhancement of tensile strength for PLA/talc composites by the addition of maleated PLA is attributed to their better interfacial adhesion and wettability.
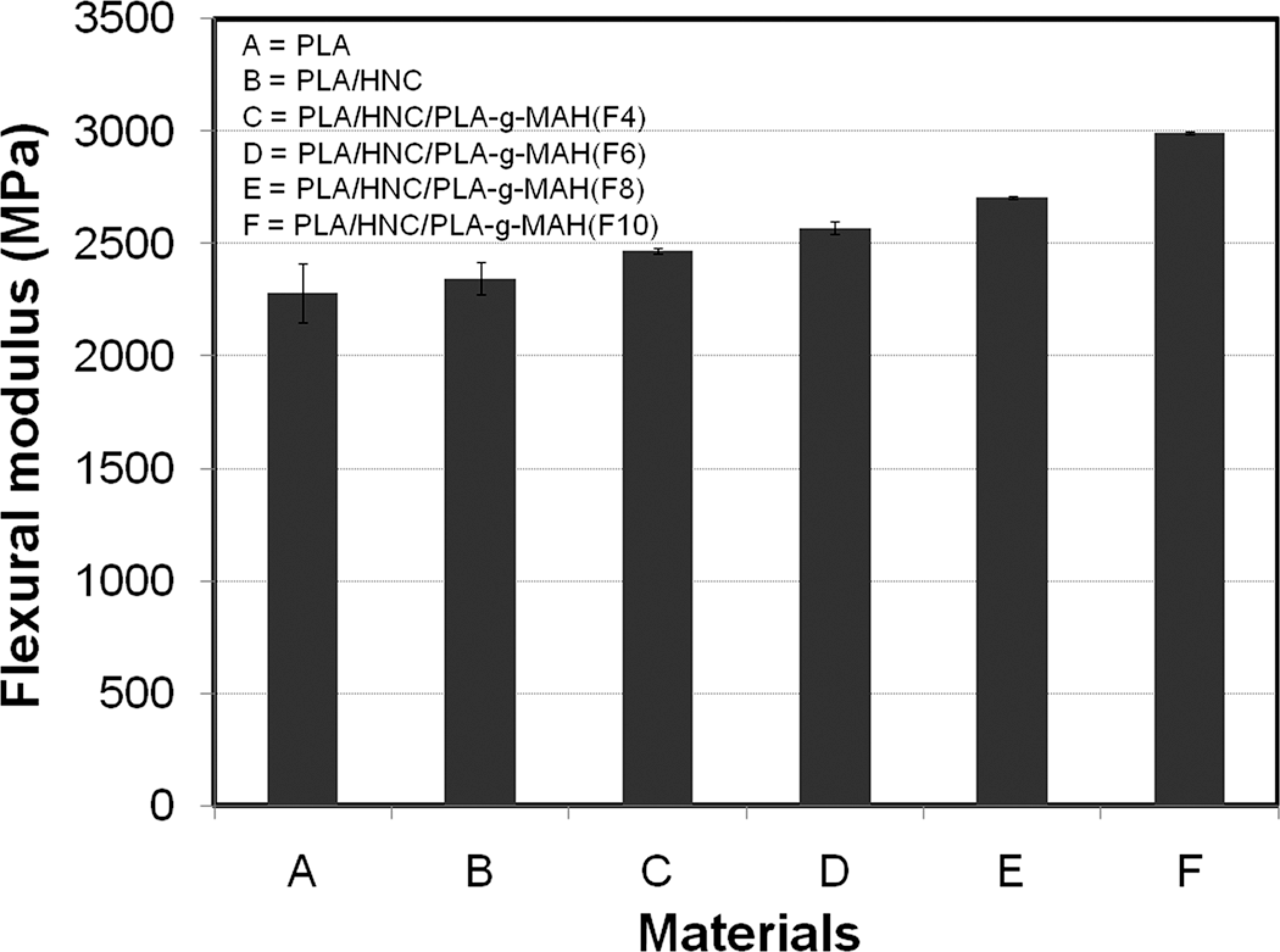
Effects of PLA-g-MAH(F) on the flexural modulus of PLA/HNC composites. HNC: halloysite clay; PLA-g-MAH: maleic anhydride grafted poly(lactic acid).
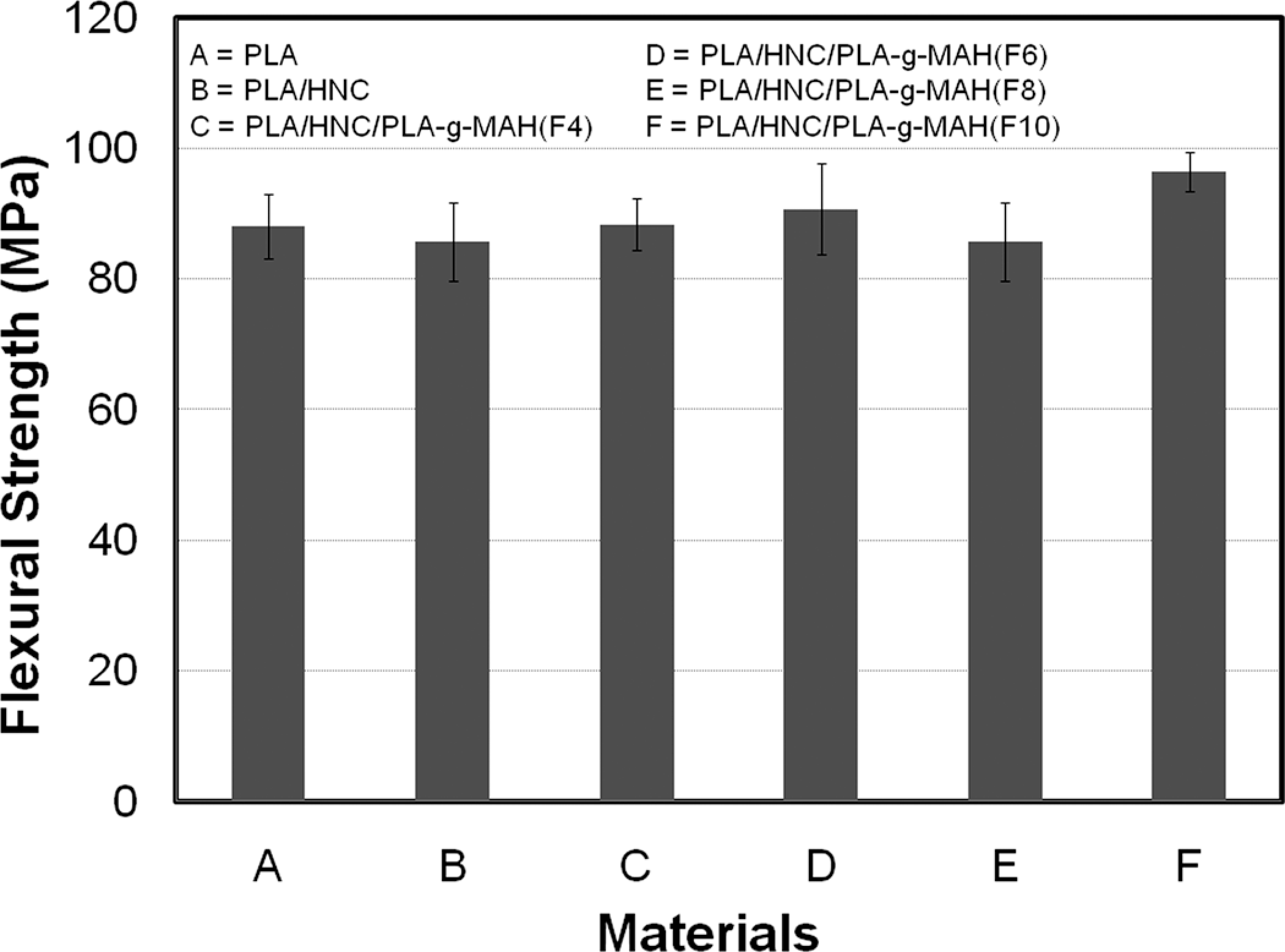
Effects of PLA-g-MAH(F) on the flexural strength of PLA/HNC composites. HNC: halloysite clay; PLA-g-MAH: maleic anhydride grafted poly(lactic acid).
Figure 6 shows the possible mechanism of MAH grafting onto PLA and the formation of PLA-g-MAH. Thermal decomposition of DCP produces radicals (RO•) which can abstract hydrogen from the PLA main chain and subsequently lead to the formation of PLA macroradicals—the reactive sites for the MAH grafting. Figure 7 shows the possible reaction mechanism for PLA, PLA-g-MAH and HNC. As discussed earlier, the interfacial interaction between PLA and HNC improved in the presence of PLA-g-MAH. This can be seen from the schematic drawing in Figure 7 that the PLA-g-MAH acts as a bridge between PLA and HNC. While, the PLA has the affinity and physical entanglement with the PLA moieties in PLA-g-MAH, the MAH moieties could interact with the hydroxyl group (–OH) of HNC.
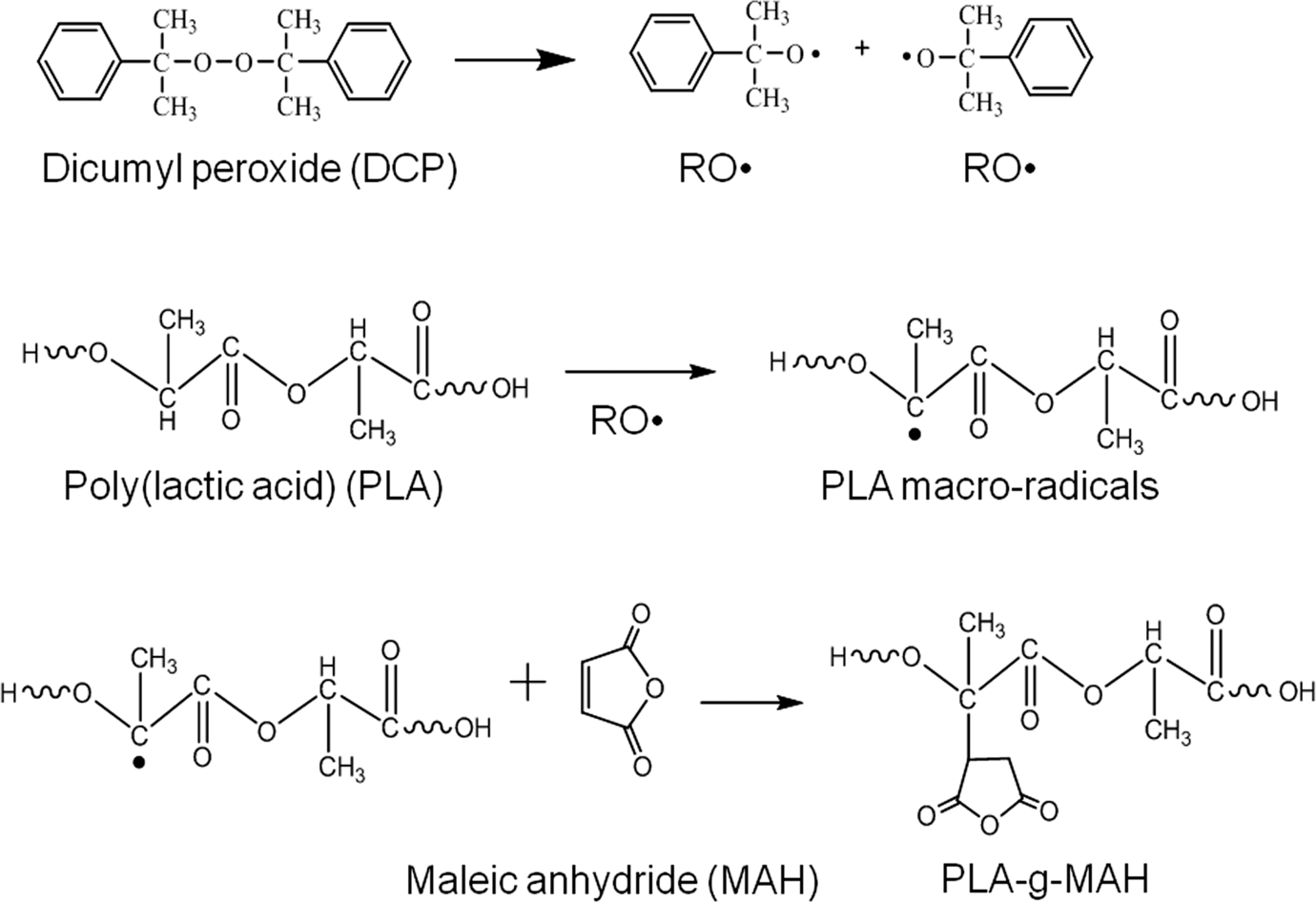
Possible mechanism for maleic anhydride grafting onto poly(lactic acid) (PLA).
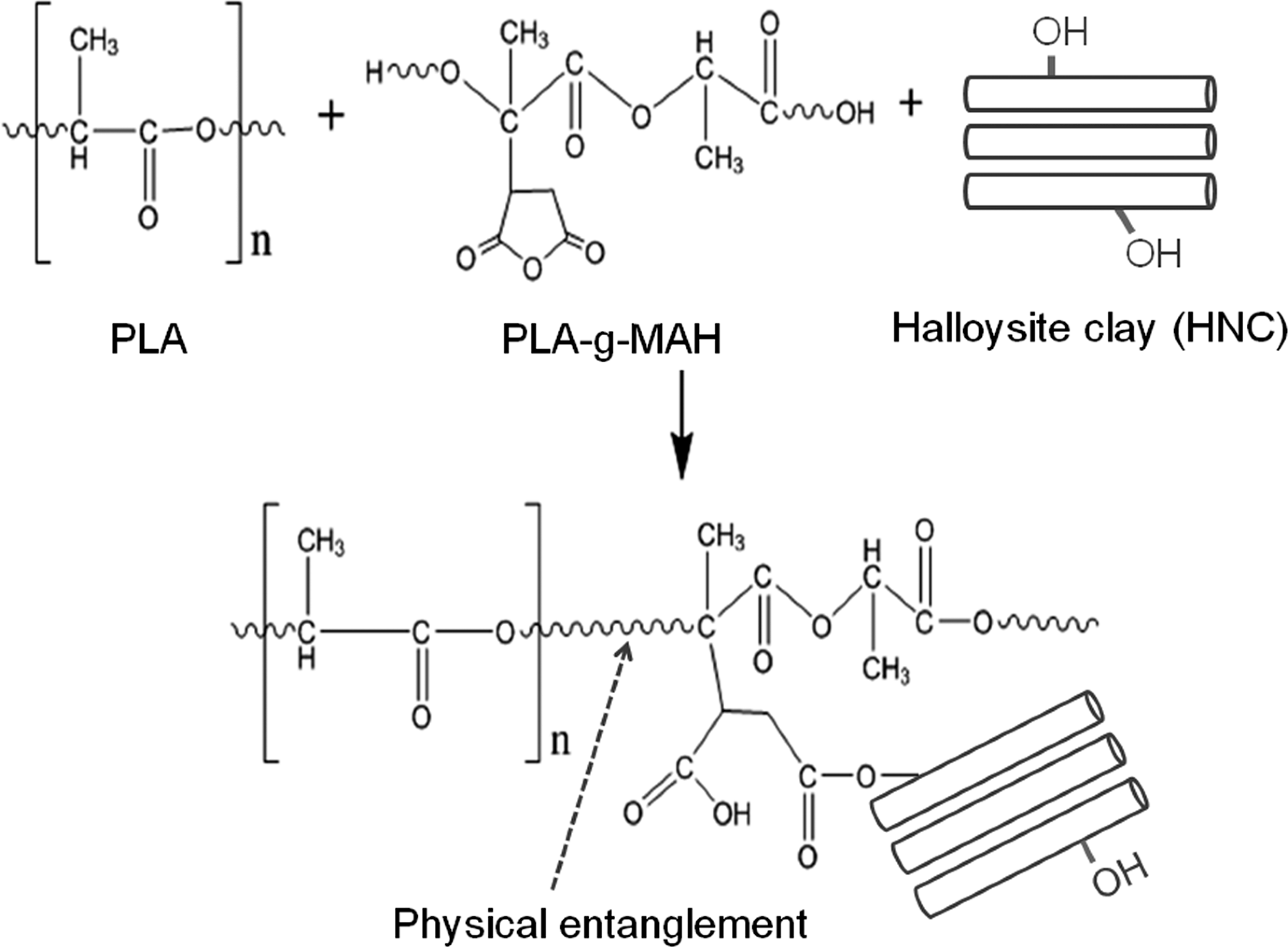
Possible reaction mechanism for poly(lactic acid) (PLA), maleic anhydride grafted PLA (PLA-g-MAH) and halloysite clay.
Thermal properties
Figure 8 shows the DSC thermograms of PLA composites. A summary of the thermal characteristics of PLA and its composites has been tabulated in Table 3. It can be seen that the T m of PLA is approximately 168°C. One may also observe that the T m of the PLA/HNC with and without PLA-g-MAH(F) are in the range of 165–167°C. The T g of PLA is about 60°C. Note that adding HNC and PLA-g-MAH did not change the T g of PLA significantly. However, we can see that the crystallization temperature (T c) and degree of crystallinity (χ c) of PLA were greatly influenced by the addition of HNC and PLA-g-MAH(F). The T c and χ c of the PLA was slightly decreased by the incorporation of HNC. This maybe associated with the fact that the HNC restrict the mobility of the PLA macromolecule, causing their chain arrangement more difficult. Similar finding has been reported by Wu 40 and Wu, 41 although they are studied on polycaprolactone/starch and polylactide/starch composites. According to Jang et al. 31 and Wang et al., 32 the reduction in crystallinity of PLA/starch composites may be due to the fact that the starch particles interfere with the motion of PLA chains. It is interesting to note that the T c of PLA/HNC composites was shifted to lower temperature as the content of PLA-g-MAH(F) increases. In addition, the degree of crystallinity of PLA/HNC composites increased significantly with the increasing content of PLA-g-MAH(F). It is believed that the presence of low-molecular-weight PLA-g-MAH facilitates the crystallization of PLA due to the increase in molecular mobility of the shorter chain. Accordingly, the nuclei can grow by the continued ordering and alignment by the presence of short molecular chain segment (i.e. PLA-g-MAH) in PLA/HNC composites. Another possible explanation can be related to the plasticization effects of MAH, which has been previously reported by several researchers31,39,42,43 who mentioned that PLA crystallizes more easily at lower temperature due to enhanced chain mobility in the presence of plasticizer.
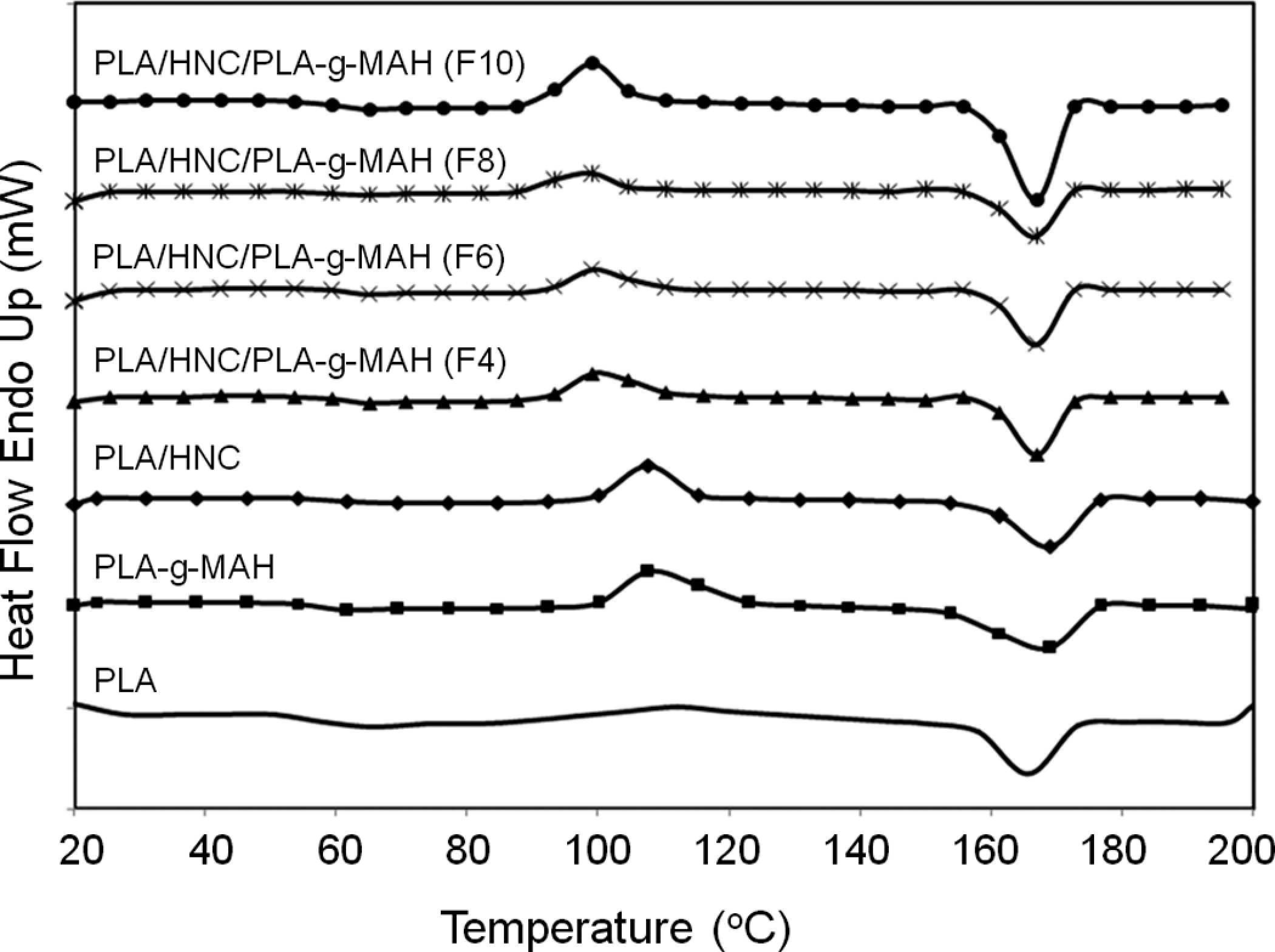
Differential scanning calorimetry (DSC) thermograms of poly(lactic acid) (PLA) and its composites.
Thermal characteristics of PLA and its nanocomposites recorded from DSC.
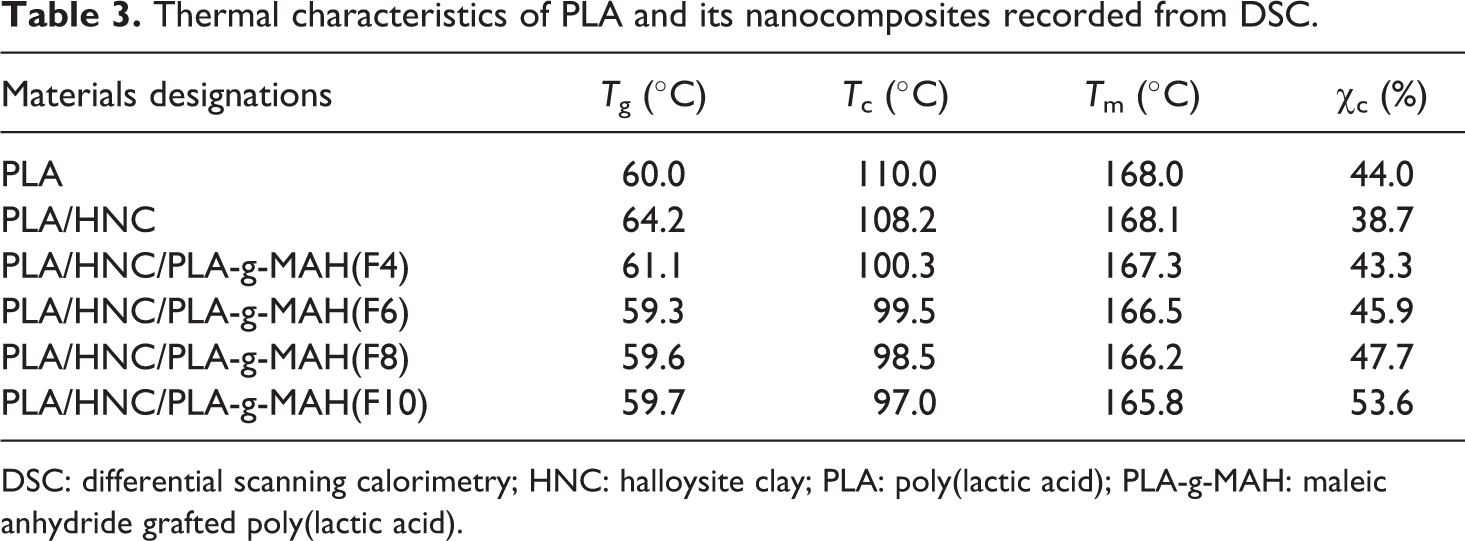
DSC: differential scanning calorimetry; HNC: halloysite clay; PLA: poly(lactic acid); PLA-g-MAH: maleic anhydride grafted poly(lactic acid).
Morphological properties
Figure 9 shows the FESEM micrograph taken from the flexural fractured surface of the PLA/HNC composites. Figure 9(a) shows the randomly propagated smooth fractured surface which indicated the brittle behavior induced by PLA matrix. There are small voids (as shown by white arrows) observed at the fractured surface of PLA/HNC. This can be due to the detachment of HNC particles from the PLA matrix, which indicates the weak interfacial interaction between PLA and HNC. On the other hand, microfibrillated morphology can be revealed for the PLA/HNC/PLA-g-MAH(F10) composites (c.f. Figure 9(b)), which imply that the specimens experienced plastic and yielding deformation. Also, less HNC detachment can be observed for the PLA/HNC composites which containing of PLA-g-MAH. This again proves that the addition of PLA-g-MAH provides better interfacial bonding and wettability of the PLA/HNC composites.

Field emission scanning electron microscopy (FESEM) micrographs of the fractured surface of (a) PLA/HNC and (b) PLA/HNC/PLA-g-MAH(F10). HNC: halloysite clay; PLA: poly(lactic acid); PLA-g-MAH: maleic anhydride grafted poly(lactic acid).
Conclusions
Based on this work devoted to study the effect of PLA-g-MAH on the properties of the PLA/HNC composites, the following conclusions can be drawn:
The grafting percentage increased with the increasing concentration of MAH and DCP. PLA-g-MAH with 3.8% grafting percentage was successfully prepared by blending PLA with 10 phr of MAH and 3 phr of DCP.
The flexural modulus and strength of PLA/HNC composites were increased significantly in the presence of PLA-g-MAH. This is attributed to the enhancement of interfacial bonding between HNC and PLA by the incorporation of PLA-g-MAH.
Although the addition of HNC and PLA-g-MAH did not influence the T m and T g of PLA, the degree of crystallinity of PLA/HNC increased, while their crystallization temperature reduced in the presence of PLA-g-MAH.
Footnotes
Funding
This work was supported by the Universiti Sains Malaysia Research University Grant (USM, Malaysia, grant number 814070) and Universiti Sains Malaysia Incentive Grant (USM, Malaysia, grant number 8021013).
