Abstract
To expand the potential applications of polylactic acid (PLA), it is essential to incorporate a highly flexible polymer into the blend. Polycaprolactone (PCL) is an ideal choice due to its ductility and biodegradability. However, blending PLA with PCL resulted in weak mechanical properties. To address this issue, glycidyl methacrylate (GMA) and nano calcium carbonate (NCC) were introduced to enhance the adhesion at the interface between PLA and PCL. Scanning Electron Microscope (SEM) images provided clear visual evidence of the impact of GMA and NCC on the morphology of the blend. Both components were effective in reducing the size of the dispersed PCL phase, shrinking it to approximately half the size of the original blend. Spectroscopic analysis revealed that GMA caused a reaction between its epoxy group and the hydroxyl and carboxyl groups of PLA and PCL. This reaction led to the formation of strong peaks in the 6.5 to 7.5 range in 1H NMR, as well as peaks at 76 and 139 ppm in 13C NMR. These findings were further corroborated by FT-IR, which demonstrated that NCC, despite its surface coating, did not create any new bonds. Rheological studies further demonstrated the positive effects of GMA and NCC. Both the storage modulus (G′) and complex viscosity (η*) of the blends increased, showing improved post-processing performance. Investigation into the shear-thinning behavior of the uncompatibilized blends revealed that NCC caused a significant decrease in complex viscosity at higher frequencies, indicating the disruption of the nanoparticle network. The power-law slope was measured to be 0.62. In contrast, the blend containing the compatibilizer demonstrated a moderate decrease in viscosity, with a power-law slope of 0.36. To analyze the behavior of the PLA/PCL blends in the presence of compatibilizers and nanoparticles at intermediate frequencies, the Palirene model was utilized. The superior integrity of the compatibilized blend was effectively demonstrated by the model, which showed enhanced stress transfer and phase relaxation.
Highlights
• The compatibility of polylactic acid/polycaprolactone blend was investigated by incorporating glycidyl methacrylate compatibilizer and nano calcium carbonate nanoparticle. • Glycidyl methacrylate increased impact strength and elongation at break of the blend, while nano calcium carbonate improved modulus and tensile strength. • The glass transition temperature decreased more in the blend containing glycidyl methacrylate, attributed to the improved compatibility of polymers. • The molecular weight and distribution of the compatibilized blend are roughly 30% greater than those of the uncompatibilized blend. • The addition of nano calcium carbonate resulted in an increase in the storage modulus during rheology testing, while simultaneously reducing the Newtonian region.
Introduction
Over the past few decades, the excessive use of nonbiodegradable polyolefin-based plastics has caused significant environmental issues, including increased waste generation and the accumulation of commercial and industrial waste in landfills.1,2 In an effort to address this problem, numerous attempts have been made to replace non-degradable polymers with biodegradable alternatives. 3 One such polymer that has garnered attention is poly(lactic acid) due to its desirable properties such as excellent gloss and clarity, high tensile strength, good processability, and low coefficient of friction. 4 These properties make PLA well-suited for various medical and packaging applications. 5 However, the brittleness of PLA limits its use to specific applications, and PLA also exhibits slow crystallization kinetics.6,7 To overcome these drawbacks, much research has been focused on modifying PLA through methods such as blending with other polymers and filling with inorganic particles.8,9
The effect of nanoparticles on the compatibility level of polymer blends refers to the ability of nanoparticles to enhance the compatibility and interaction between two polymers that would be either incompatible or have poor mixing behavior. When two polymers with different chemical structures or polarities are blended, they typically exhibit phase separation, leading to the formation of distinct domains or phases within the blend. This phase separation can result in poor mechanical properties and limited functionality for many applications. 10 Nanoparticles, particularly those that are surface-modified or functionalized, can act as a compatibilizer in polymer blends. These nanoparticles are designed to have affinity for both polymers in the blend, acting as bridges or mediators between the two phases, promoting their compatibility. The compatibilizing effect of nanoparticles can be attributed to several mechanisms. 11 Firstly, steric stabilization is a notable effect in which nanoparticles with specific surface modification (e.g., polymer brushes) form a layer around the nanoparticles, reducing interfacial tension and preventing phase separation. 12 Secondly, nanoparticles can create strong adhesion at the interface between the two polymer phases, improving the interfacial strength and preventing phase separation. Moreover, nanoparticles can act as physical barriers hindering chain motion and promoting interdiffusion between polymer chains. This can lead to the formation of a more uniform and mixed structure, enhancing compatibility. 13 Finally, nanoparticles serve as nucleating agents, enhancing the dispersion of one polymer phase within the other and reducing the size of phase-separated domains (in the sea-island morphology).
In addition, inorganic particles are commonly used to enhance the strength of polymers. 14 The research conducted by Ercan et al. 15 successfully showed that the incorporation of rare earth oxide nanoparticles at the interface of PLA and PCL resulted in a significant increase in crystallinity of the blend. Consequently, overall entropy of the polymer blend decreased by reducing chain mobility. Additionally, intermolecular interactions between polymer chains at the interface made considerable improvement in both mechanical properties and thermal stability.
One of the most common nanoparticles is nanosized calcium carbonate, which is widely employed in rubbers, plastics, and paints due to its cost-effectiveness. NCC can improve PLA’s thermal and processing properties while also reducing costs.16,17 Mechanical properties, including tensile properties, impact strength, and flexural properties, are crucial indicators of material performance. 18 In a study conducted by Liang et al., 16 the addition of NCC to PLA resulted in an increase in the elastic modulus but a decrease in tensile strength and elongation at break. Moreover, an increase in NCC content from 1 to 4 wt.% led to a linear increase in the elastic modulus of 52%. However, the non-linear decrease in tensile strength and elongation at break was limited to 12% and 19% respectively.
One approach to improve the unfavorable properties of PLA, such as brittleness, is to blend PLA with ductile polymers.19,20 Blending can significantly impact the resulting mechanical properties, which depend on the mechanical properties of the components, blend microstructure, and the interface between phases.21,22 However, one challenge in formulating new polymeric blends is predicting how additives will influence the overall phase behavior and performance, especially when the blend involves incompatible polymers with weak interfacial adhesion and hence poor mechanical properties.23,24 To enhance interfacial adhesion between the phases and reduce interfacial tension, the use of suitable compatibilizers is appropriate. Compatibilizers are typically macromolecules, such as block copolymers, which manipulate interfacial properties.25,26
In contrast to PLA, poly(ε-caprolactone) offers high flexibility, but has relatively low strength and a low melting point of 60°C, which restricts its use to certain applications. Therefore, blending PLA with PCL has the potential to improve flexibility or increase strength compared to each individual component.27,28 In recent years, blends of PLA with more flexible biodegradable polymers, such as PCL, have been explored and developed. 29 Although these blends may not exhibit desired properties due to their incompatibility, several interesting results have been observed. 30
Various compatibilizers, such as PLA-co-PCL, have been investigated to improve the compatibility of PLA/PCL blends and enhance their mechanical properties, such as impact strength. 31 Harada et al. 32 demonstrated that lysine triisocyanate (LTI) was more effective as a reactive processing agent for PLA/PCL blends compared to lysine diisocyanate (LDI) and other isocyanates. However, LTI is not suitable for biocompatibility.
Glycidyl methacrylate is a suitable material for enhancing the interfacial adhesion between components in blends. The epoxy groups in GMA can react with the carboxyl or hydroxyl groups of polyesters. 33 Therefore, toughness is a result of better interfacial adhesion. In another study, an ethylene-methyl acrylate-glycidyl methacrylate terpolymer was employed to reduce interfacial tension between PLA and PCL. The incorporation of 8 phr of compatibilizer through melt blending in PLA/PCL (90/10) achieved the expected toughening effect, leading to a notch impact strength of 64.31 kJ/m2. Recently, researchers have successfully used a glycidyl methacrylate PLA compatibilizer to improve the compatibility of PLA/PCL blends. This approach resulted in higher thermal stability, a lower melting point, and an elongation at break 60 times greater than that of PLA/PCL without compatibilizer. 34
Improvement in compatibility of PLA/PCL blend can lead to exclusive applications, such as in packaging materials, medical devices, and drug delivery systems. This current study aimed to demonstrate that even though there have been preconceived notions about the effectiveness of multi-functional compatibilizers or nanoparticles with specific surface modifications in enhancing the compatibility of polymer blends, using traditional single-factor compatibilizers or nanoparticles with simple surface modification can still lead to improvement in the blends mechanical and other properties. Additionally, a careful consideration of other aspects such as molecular weight, morphology, and rheology in the polymer system can achieve a suitable balance in the conclusion of the effect of these two types of additives. Moreover, rheological test results were used to evaluate behaviors of two polymers at their interface using the Palierne model before and after compatibilizing. Therefore, the objective of this study is to investigate the individual effects of the GMA compatibilizer and NCC nanoparticle on the properties of PLA/PCL blend.
Experimental
Materials
Poly (lactic acid) with a grade of 2003D, obtained from Nature Works, USA, has a melt flow rate of 6 g/10 min (190°C/2.16 kg) and a density of 1.24 g/cm3. The Poly (ε-caprolactone) used is Capa™6800, purchased from Perstorp, Sweden, with a melt flow rate of 4 g/10 min (160°C/5 kg) and a density of 1.13 g/cm3. Glycidyl methacrylate, obtained from Alfa Aesar, Germany, has a density of 1.08 g/cm3 and a boiling point of 189°C. Nano calcium carbonate with a mean particle diameter of 80 nm and a density of 2.65 g/cm3 was purchased from Shiraishi (Austria) and treated with stearic acid.
Nano particle surface treatment
Surface treatment of nano calcium carbonate with stearic acid involves coating the particles with a layer of stearic acid. This process is commonly used to improve the dispersion of nano calcium carbonate in polymer matrices, as well as to enhance its compatibility with various polymer systems. The surface treatment is typically carried out using a solution of stearic acid in a solvent like ethanol or toluene. The nano calcium carbonate particles are then added to the solution and stirred or sonicated to ensure uniform coating of the particles with stearic acid. The solvent is then evaporated, leaving behind a thin layer of stearic acid on the surface of the nano calcium carbonate particles. The stearic acid coating can improve the dispersion of the nano calcium carbonate in polymer matrices by reducing the agglomeration of the particles and promoting better interaction between the filler and the polymer. This can lead to improvements in mechanical properties, thermal stability, and other performance characteristics of the composite materials.
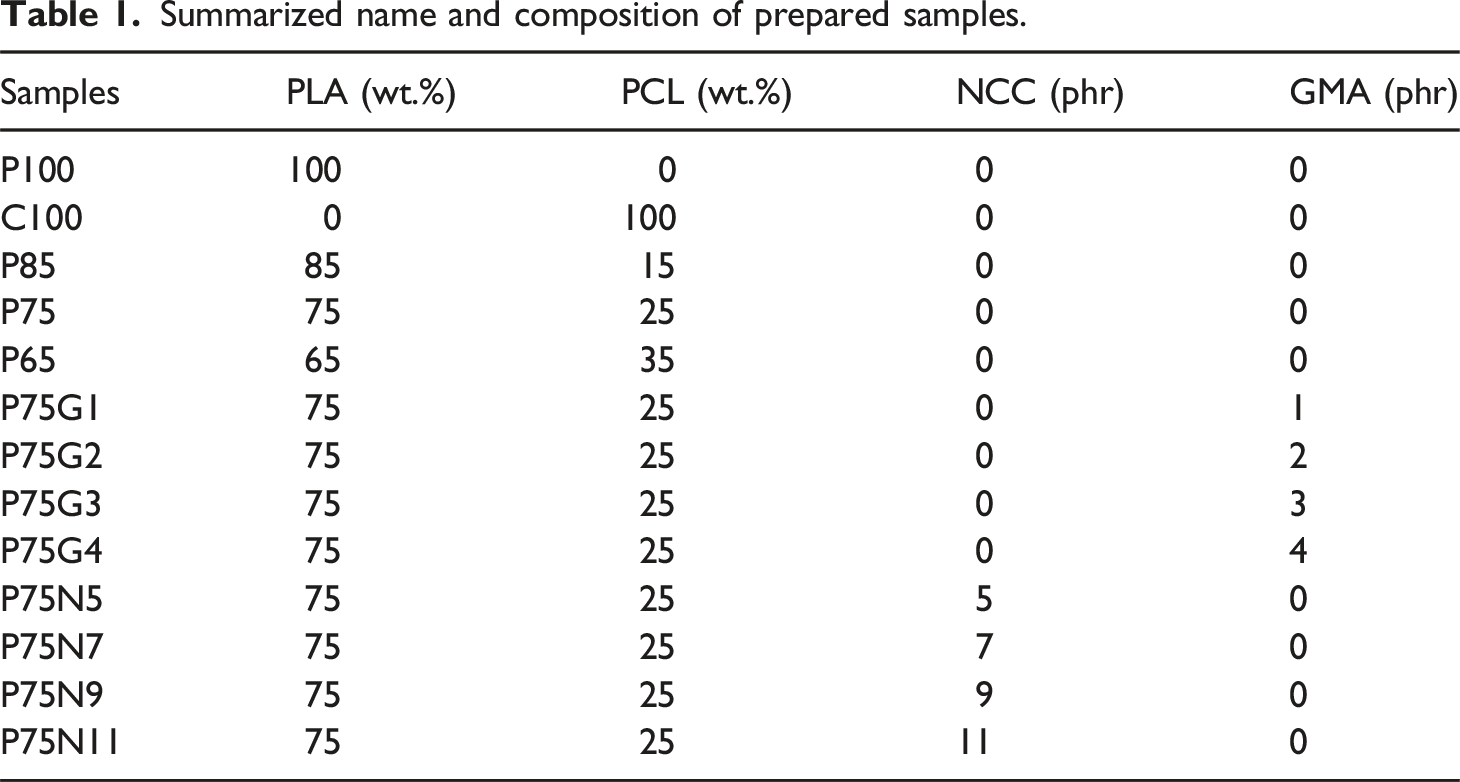
Samples preparation
To prepare materials for compounding, they were dried in a vacuum oven at 70°C for 16 h. The compounding process was performed using an internal mixer (Brabender Plasticorder W50) with a capacity of 60 cm3. The components were mixed simultaneously in a single step at 170°C and 50 rpm for 10 min (However, to prepare nanocomposites, NCC was added after both polymer components were poured in the brabender). The resulting mixture was then compression molded at 190°C to form sheets of 2 and 3 mm thickness for subsequent measurements.
For the sample preparation, three steps were taken:
Simple blends of PLA/PCL with 15, 25, and 35 wt.% of PCL were prepared to study their mechanical and thermal properties. The blend with 25 wt.% of PCL (P75) showed the best mechanical properties and was selected for further investigations.
Compatibilized PLA/PCL (75/25) blends with varying amounts of GMA compatibilizer (1, 2, 3, and 4 phr) were prepared and their properties were studied.
PLA/PCL nanocomposites with different amounts of NCC (5, 7, 9, and 11 phr) were made and their properties were investigated as well. (The reason why these composition were chosen for NCC was the previous studied that had shown the proportion of NCC in which filler aggregation happened was higher than 15 wt. %). We started making sample from 3 phr which was about 2 wt.% and TEM images showed no aggregate. Then we increased amount of NCC to 11 phr (9.7 wt,%) and there was no aggregated particle). So, the aggregate formation can be negligible, evet in sample containing 11 phr of NCC)
Summarized name and composition of prepared samples.
Characterization
Mechanical characterization
Tensile tests were conducted using a SUN 2500, Galdabini universal testing machine according to ASTM D638 with a strain rate of 5 mm/min at ambient temperature. Five dumbbell-shaped specimens with a thickness of 3 mm were tested for each sample, and the mean values were reported.
Notched Izod impact tests were performed using the Izod Impact Tester (Ueshima Seisakusho, Tokyo, Japan) in accordance with ASTM D256. Three specimens in dimensions of 64 × 12.7 × 6.4 mm were prepared and tested for each sample.
Preparation of the specimens for testing involved molding the samples obtained from the internal mixer into rectangular sheets with a nominal thickness of 0.5 mm using a Carver press with a flat window frame mold. Compression molding was conducted at a pressure of 5 MPa and temperature of 180°C for a duration of 10 min.
Differential scanning calorimetry (DSC)
Nonisothermal crystallization behavior of the samples was determined using a DSC1, Mettler Toledo differential scanning calorimeter. The samples were heated from room temperature to 200°C at a rate of 10°C/min and held at this temperature for 5 min to eliminate thermal history. Subsequently, the samples were cooled to room temperature at the same rate and then heated to 200°C at a rate of 10°C/min again.
The crystallinity of the samples was calculated using the cooling process, and melting behavior was assessed through the second heating process. The crystallinity (Xc) was calculated according to equation (1).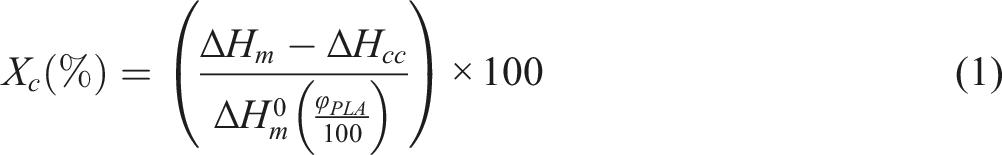
In this equation, ΔHm and ΔHcc refer to the melting and cold crystallization enthalpy of the samples respectively, determined from the DSC thermogram. ϕPLA represents the weight percentage of PLA in the blends, and
Morphology
The morphology of the blends was examined using a scanning electron microscope (SEM) (DSM 960 A, Germany) operated at 10 kV accelerating voltage. The samples were cryogenically fractured after being stored in liquid nitrogen for 15 min. The fracture surface of the samples was coated with gold using a sputter coater to enhance conductivity.
Spectroscopy
Fourier transform infrared analysis (FT-IR) of PLA, PCL, and the prepared samples was conducted over a range of (400–4000) cm−1 with 64 scanning numbers using Spectrum Bomem MB102 and the Attenuated Total Reflectance (ATR) method.
The sample for nuclear magnetic resonance (NMR) analysis was first dissolved in CDCl3 and sealed in an NMR tube with a 10 mm O.D. Subsequently, after degassing, the analysis was conducted using a Bruker AMX400 1H NMR and 13C NMR spectrometers with conditions of 100 MHz, 30 pulse, and 4xs cycle time.
Dynamic mechanical analysis (DMA)
Dynamic Mechanical Analysis was performed on the samples using (a MK11, Polymer Laboratory). The frequency of testing was set at a fixed rate of 1 Hz, while the dynamic strain applied was 0.025%. The temperature range for the analysis spanned from 60 to 180°C, and the scanning rate was precisely controlled at 2°C per minute.
Molecular weight measurements
Gel Permeation Chromatography (GPC) was carried out using a Waters 2414 refractive index detector, equipped with a Styragel HR5E 7.8 × 300 mm column with a molecular weight resolving range of 2000–4,000,000. The melt-blended samples were dissolved in distilled tetrahydrofuran (THF) at a concentration of 3.33 mg/mL, and THF was eluted at a rate of 1.0 mL/min at 40°C and calibrated with polystyrene standards.
Rheology
A rheo mechanical spectroscopy (RMS) apparatus, specifically the Anton-Paar 302 model, with parallel plates set at 1 mm distance with a diameter of 25 mm operating at 180°C, was used to study the small amplitude oscillatory shear rheology (SAOS) of the unblended individual polymers, including neat samples, blends with and without compatibilizer, and nanocomposite blends.
Results and discussion
Mechanical properties
Figure 1 depicts the stress-strain plots of tensile; also, data regarding tensile properties and impact strengths are presented in Table 2. As can be seen PCL does not change the brittleness of PLA because of their incompatibility. However, GMA swaps the fracture mode of PLA/PCL from brittle to ductile due to stress transfer along the interface. Moreover, the addition of NCC in uncompatibilized blend results in a more brittle behavior compared to neat PLA. For example, 15 wt.% PCL increased the impact strength of neat PLA from 21.6 to 38.2 J/m (P85). Brittle fracture observed in neat PLA indicated a lack of yield phenomenon.
36
However, the presence of PCL introduced a shear yielding mechanism that enhanced the toughness of PLA. Interestingly, the sample containing 25 wt.% PCL exhibited the highest impact strength and elongation at break among the blends. This can be attributed to the intense phase separation in blend containing 35 wt.% PCL, resulting in poor interfacial adhesion and a decrease in mechanical properties in the blend containing 35 wt.% of PCL (P65), as supported by previous studies.
37
The PLA/PCL blend demonstrates a lower critical solution temperature (LCST), in which the optimum PCL composition for the initiation of phase separation was estimated to be around 36% by volume. Consequently, the PLA/PCL blend with a ratio of 75/25 was selected for further experimentation. Stress-strain graphs of: (a) uncompatibilized blends, (b) compatibilized blends, (c) nanocomposites, and (d) P100, C100, P75, P75G3, P75N9. Mechanical properties of neat PLA and PCL and their blends and nanocomposites.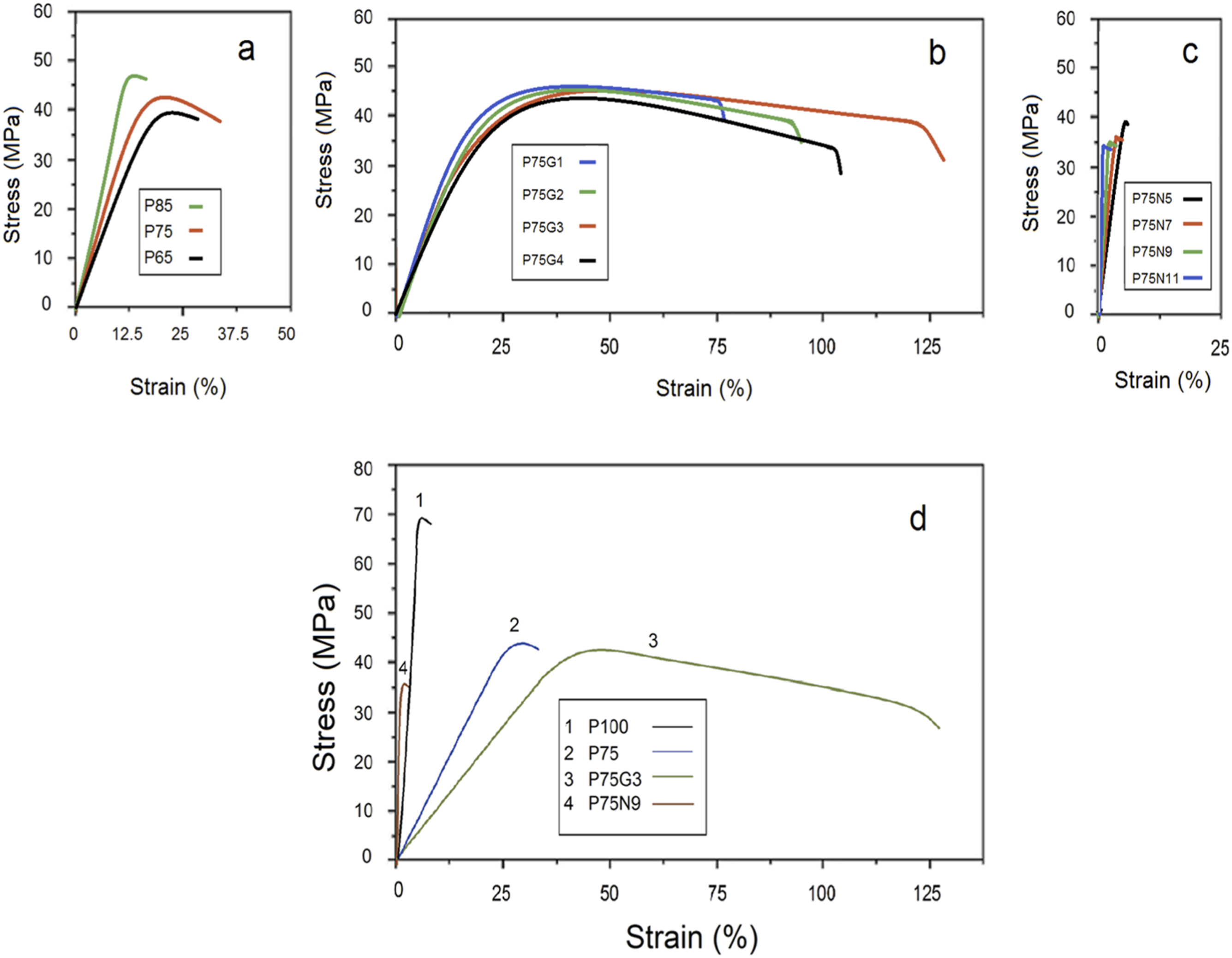
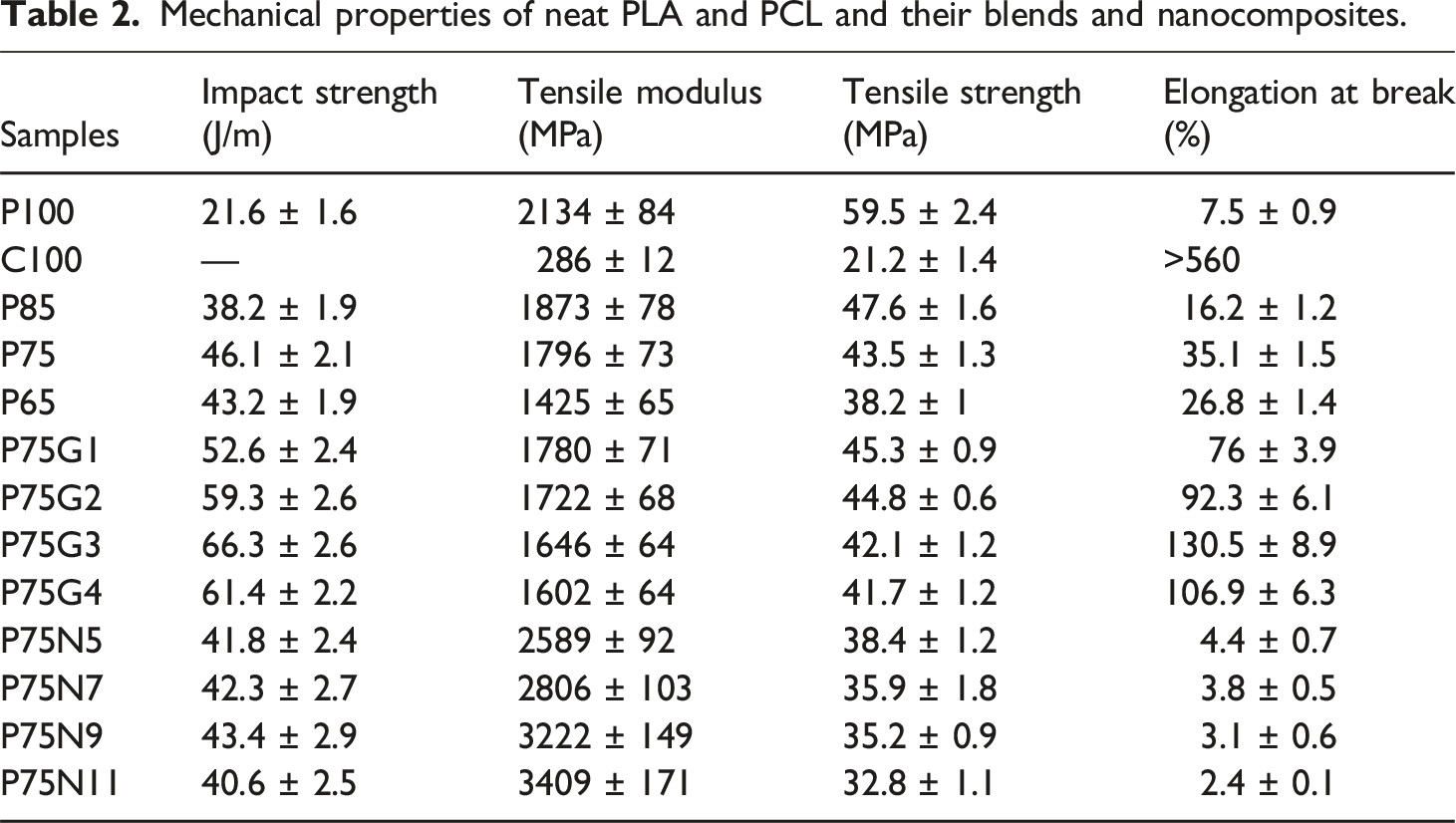
To improve compatibility of PLA and PCL, GMA was employed as a reactive compatibilizer in the blend. Notably, the addition of 1 phr of GMA to P75 increased the elongation at break from 35.1 to 76%. This improvement can be attributed to enhanced compatibility between PLA and PCL, as well as the uniform distribution of PCL droplets facilitated by GMA because of the reduction of interfacial tension.38,39 The positive impact of GMA on the mechanical properties of polymer blends has been reported previously.40,41 The results presented in Table 2 reveal that increasing content of GMA up to 3 phr further improves the elongation at break and impact strength of the samples. However, exceeding the optimum amount of compatibilizer (3 phr) had an adverse effect on the mechanical properties, potentially due to phase separation. This behavior signifies that an extra quantity of compatibilizer within the blend did not undergo any reaction; therefore, its influence on softening of polymer phases decreased the mechanical properties. In the regions where this unreacted compatibilizer existed, the movement of polymer chains accelerated, as the induced free volume within the system increased. Moreover, a significant rise in elongation at break of polymer blends is attributed to the lubricating effect of GMA, and thus improved fluidity of molecular chain. 42
In a previous paper, incorporating more than 3 wt.% of GMA into PLA/PCL blends resulted in a decrease in flexural modulus, elongation at break, and impact strength. 33 Also, Zhang et al. 43 found that increasing the T-GMA content initially improved the elongation at break, but subsequent increases ultimately reduced this property.
In addition to investigating the effect of NCC on PLA/PCL blends, the impact of different amounts of NCC on uncompatibilized PLA/PCL (75/25) blend was examined. A decrease in tensile strength and elongation at break was seen as the quantity of NCC increased; however, the tensile modulus and impact strength of the samples were increased. The decline in tensile properties can be attributed to either reduced chain mobility or the presence of voids, which arose from the discontinuity in load transfer across the chains. Despite this, the presence of NCC led to a decrease in the impact strength of the P75 sample, potentially due to crack growth initiation. Notably, the use of 9 phr NCC yielded the most preferable mechanical properties amidst nanocomposites, as the higher concentration of NCC increased rigidity. Liang et al. 16 also observed that the tensile yield strength and un-notched Izod impact strength of PLA/PCL/NCC nanocomposite experienced minimal changes as the weight fraction of filler particles exceeded 3 wt.%. Generally, in the presence of nanoparticles, the tensile strength and elongation at break of the sample may be affected. The increase in brittleness can lead to a decrease in strength, causing samples to undergo brittle fracture at a lower elongation.
Thermal properties
Thermal properties of neat PLA and PCL and their blends and nanocomposites on the second heating run.
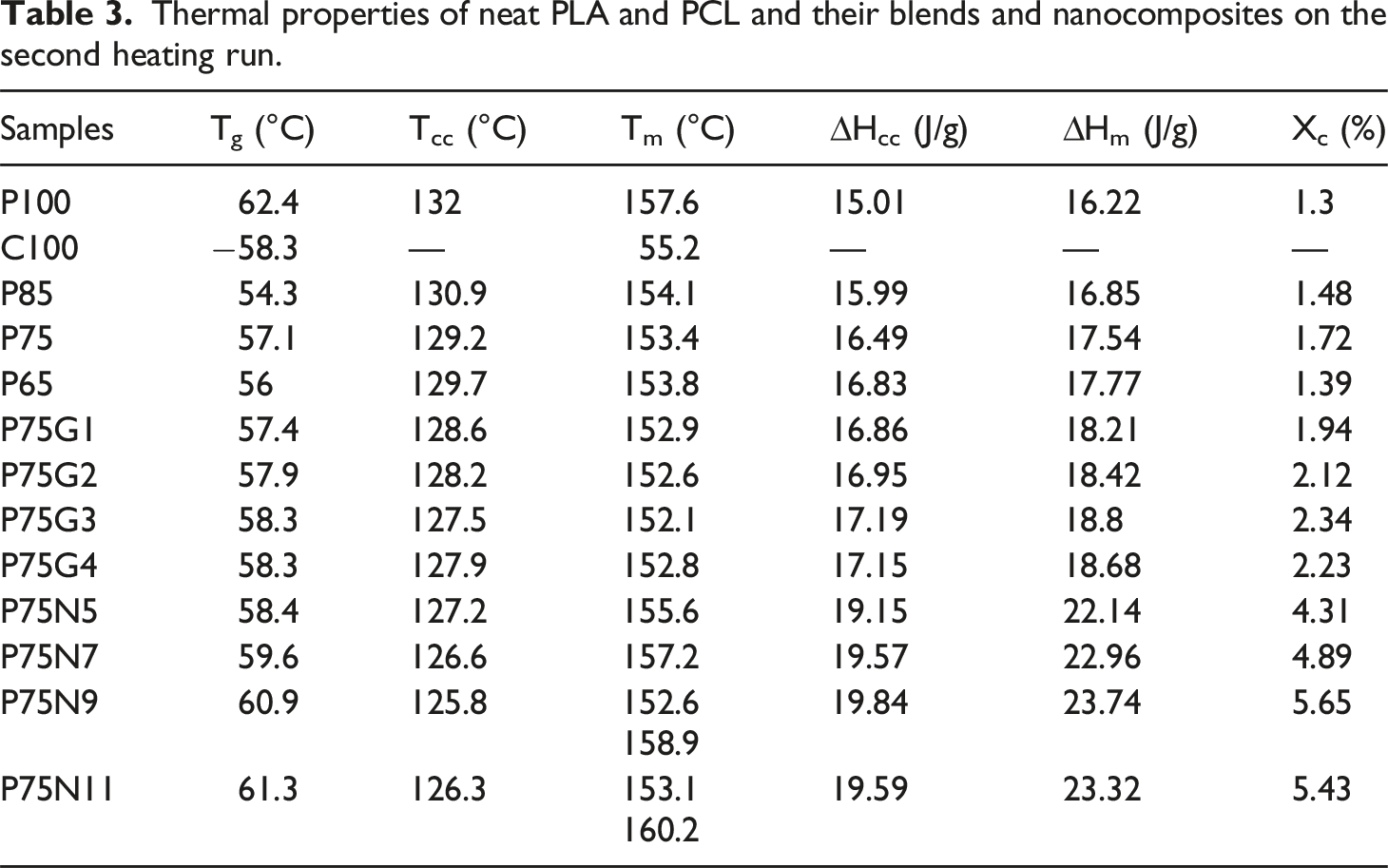
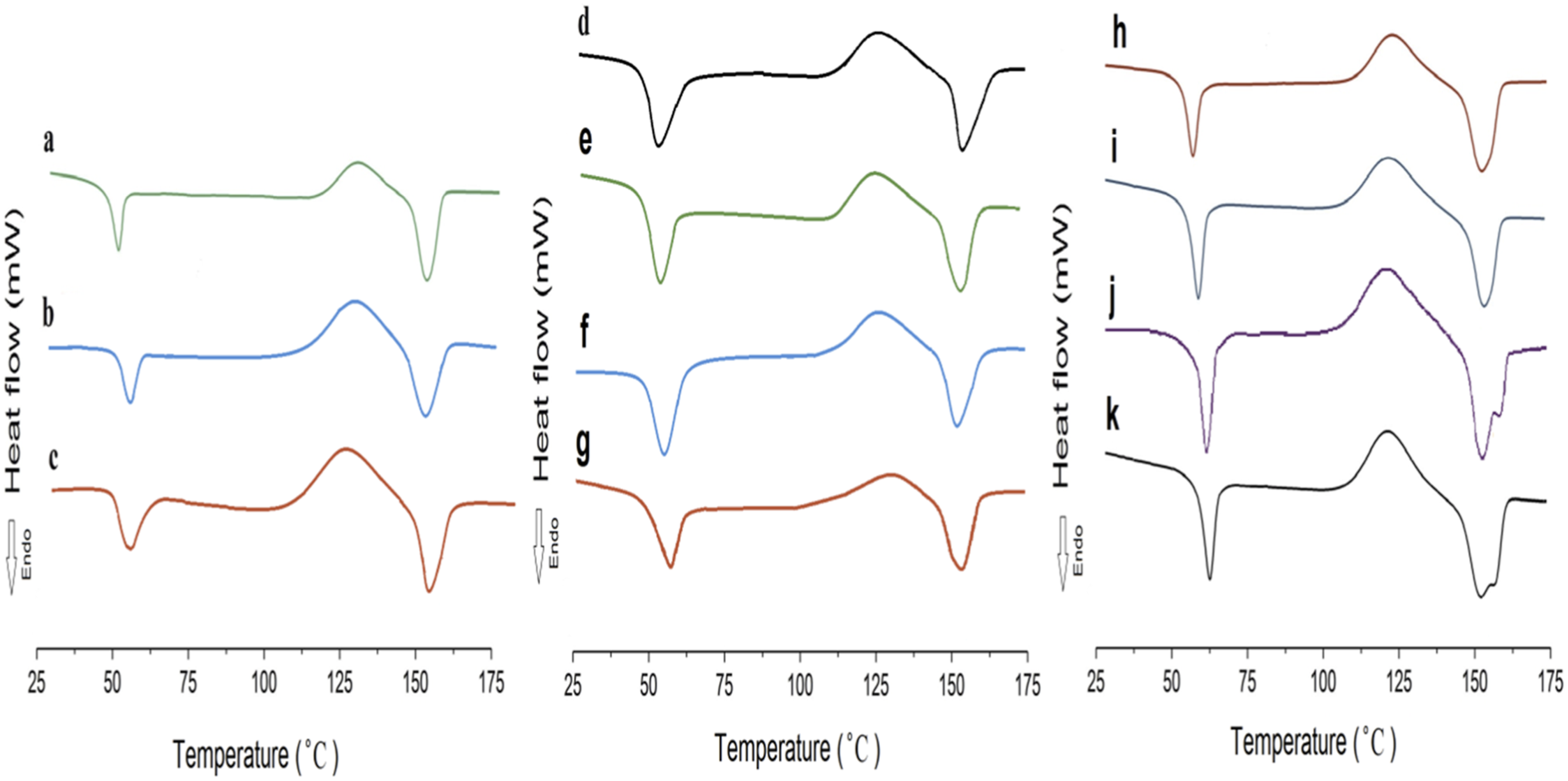
The second heating DSC curves of: (a) P85, (b) P75, (c) P65, (d) P75G1, (e) P75G2, (f) P75G3, (g) P75G4, (h) P75N5, (i) P75N7, (j) P75N9, (k) P75N11.
The neat PLA demonstrates an orthorhombic structure with a crystallization peak at Tc = 132°C and a melting peak at Tm = 157.6°C (Figure 3). On the other hand, neat PCL only exhibits a melting peak at Tm = 55.2°C. P75 blend exhibits lower degree of incompatibility between PLA and PCL among other blend samples. For samples without NCC or GMA, Tcc values for all blends are lower than that of neat PLA, with the P75 blend having the lowest value, suggesting the nucleation effect of PCL. Therefore, the P75 sample composition is selected for subsequent samples. The second heating DSC curves of superior samples in each category: (a) P100, (b) P75, (c) P75G3, (d) P75N9, and (e) C100.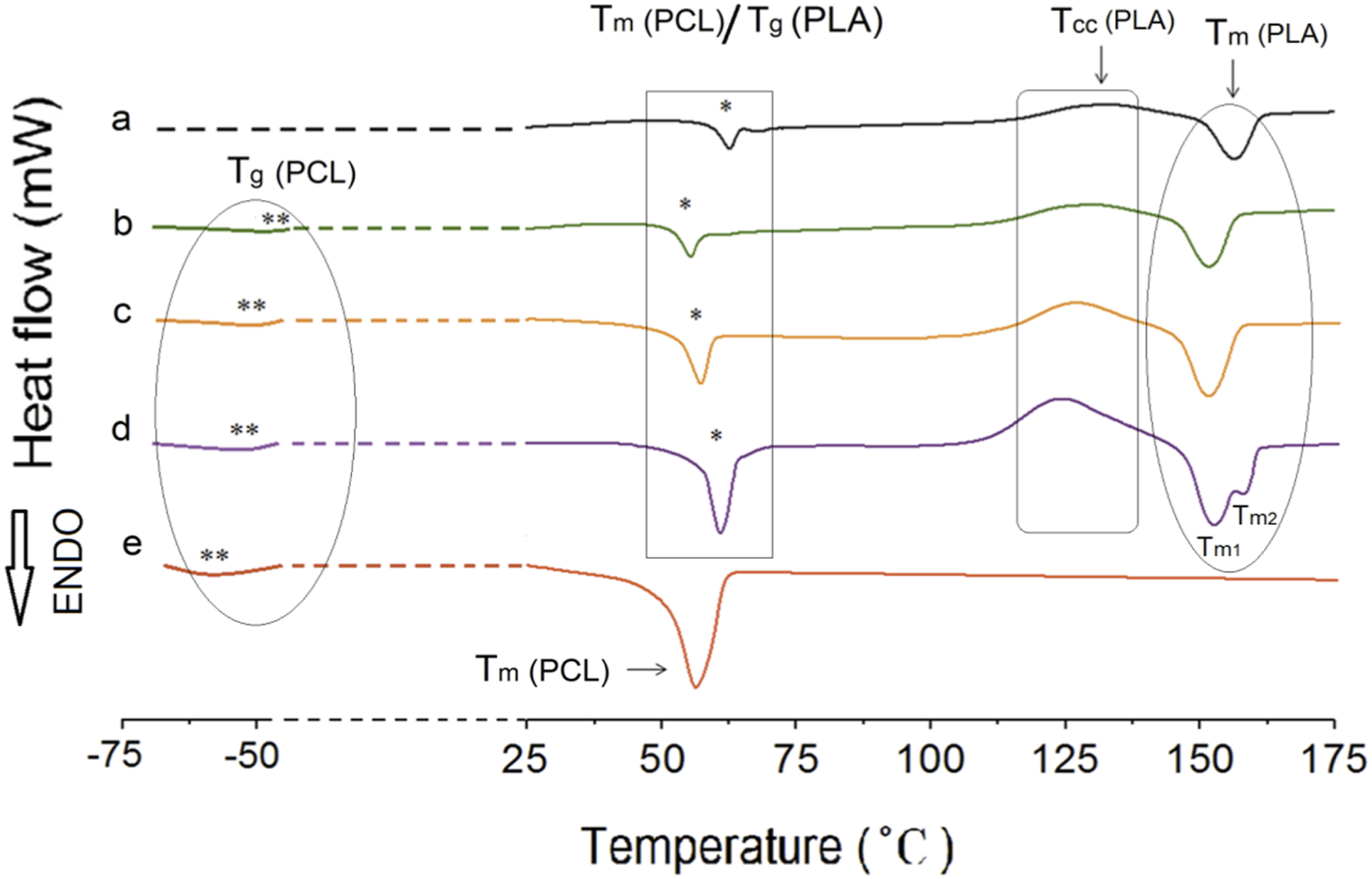
The impact of varying amounts of GMA compatibilizer on the thermal properties of P75 blend was investigated. GMA decreases the Tg and Tm of P75 due to an increase in polymer chain mobility. This is attributed to the reactive epoxide group present in GMA, which can react with both PLA and PCL, facilitating the creation of covalent bonds that bridge the two polymer phases. Consequently, this covalent bonding strengthens the interfacial adhesion and promotes efficient stress transfer between the polymers, resulting in improved chain mobility. 45 Additionally, as the Tg of GMA is lower than both PLA and PCL, its incorporation into the blend leads to an overall reduction in the blend’s Tg. This decrease in Tg enhances chain mobility, enabling polymer chain rearrangement and reducing chain entanglements; however, it had been expected that GMA acted as a plasticizing agent and caused an increase chain mobility.
In addition, the cold crystallization of PLA in the blend was accelerated after addition of GMA as evidenced by a decrease in Tcc and an increase in ΔHm, in agreement with the findings of Sugih et al. 46 GMA reduces the size and increases the number of PCL domains and the total area of PLA/PCL interface increases. In addition, the acceptable effect of GMA on thermal properties of blends took place in the sample containing 3 phr of GMA. The limited interaction capacity between the polymer and compatibilizer led to unreacted GMA, resulting in incompatibility in the P75G4 sample. 33
Furthermore, the addition of NCC to the P75 increased the Tg and Tm. The Tcc of P75 decreased significantly, indicating that NCC enhances the rate of PLA crystallization. This can be attributed to the nucleating effect of the nanoparticle. NCC in 9 phr enhanced thermal properties, as evidenced by a distinct increase in the percentage of crystallinity compared to P75.47,48 Besides Tcc changes, it was understood that the P75N9 and P75N11 samples exhibit two distinct melting peaks. According to Figure 3(d), low temperature melting peak (Tm1) should be associated with the fusion of the crystals grown by normal primary crystallization, whereas the high-temperature melting peak (Tm2) is related to the melting peak of the most perfect crystals after rearrangement during the second heating process.49,50 This phenomenon happened because of the increment in NCC composition, which prompted reduction of chain mobility and prevented the lamellas from being completed. Therefore, some imperfect crystals with lower lamella’s thickness are formed; consequently, double peaks for Tm are seen. Moreover, it is found that the use of 11 phr of NCC in the P75N11 did not make favorable changes in Tcc and Xc compared to the P75N9. It seems that the nucleating effect of NCC is restricted due to nanoparticle aggregation. It is important to take into consideration that the process of crystallization is divided into two stages: nucleation and growth of lamellas. 6 Upon examining the selected samples from each category (Figure 3), it is evident that the peak associated with the melting temperature of PLA is significantly higher in the P75N9. This can be attributed to the presence of nanoparticles, which hinder the mobility of polymer chains due to NCC rigid nature. This trend is also noticeable in the peak melting temperature of PCL and the glass transition temperature of PLA. Conversely, P75G3 exhibits a lower melting temperature, likely due to its softer nature. As the changes of Tcc shows, the addition of NCC and GMA enhances nucleation, but the effectiveness of NCC is superior. However, NCC hinders the growth of lamellas by restricting molecular chain movement and resulting in the formation of imperfect lamellas. On the other hand, GMA increases chain mobility and facilitates the arrangement of chains within the lamellas. Therefore, GMA counteracts the formation of imperfect crystals and alters the characteristics of the samples at their melting point.
Morphology
In Figure 4, SEM images of all samples are displayed. All samples exhibit droplet-matrix morphology. The circular structures in the dispersed phase represent the PCL domains. Table 4 presents the results of particle size determined using Image J software. SEM micrographs of: (a) P85, (b) P75, and (c) P65, (d) P75G1, (e) P75G2, (f) P75G3, (g) P75G4, (h) P75N5, (i)P75N7, (j) P75N9, and (k) P75N11. PCL droplets sizes for PLA/PCL blends and their nanocomposites.
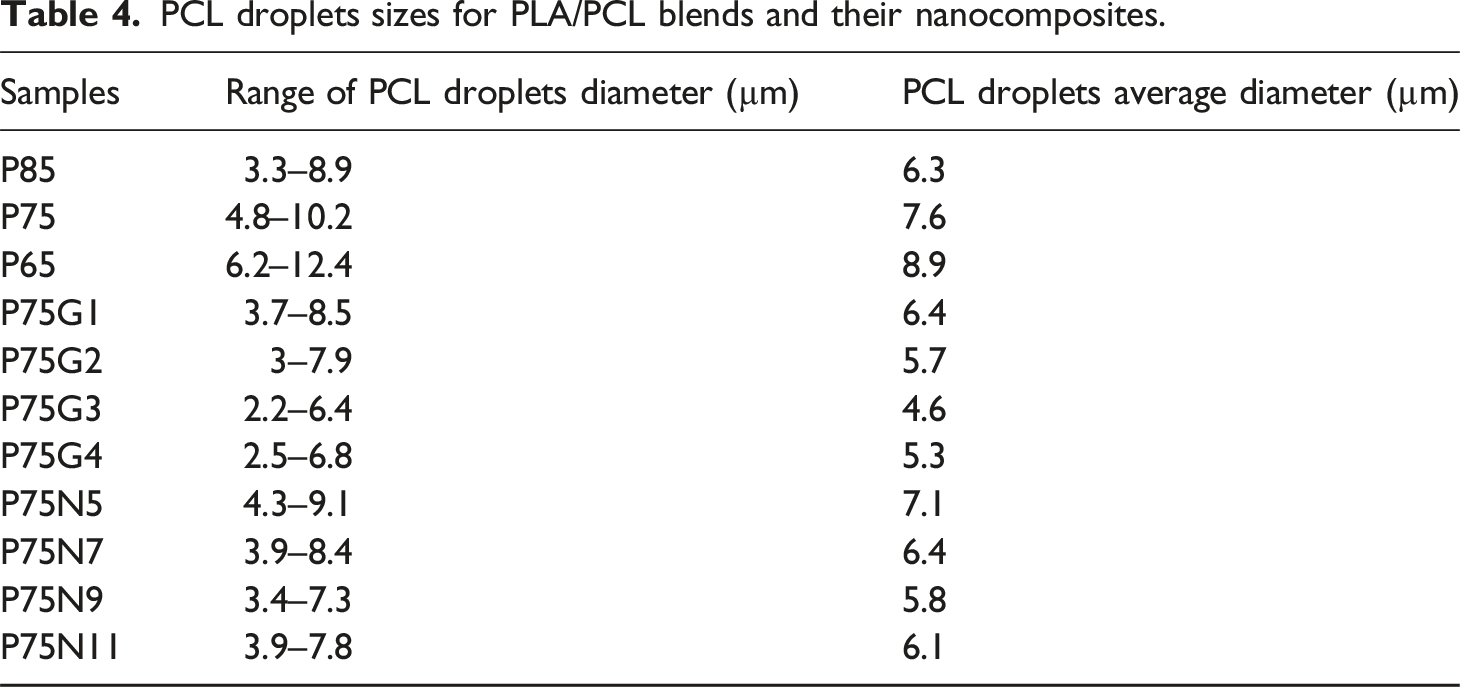
The images reveal that in uncompatibilized blends, as the amount of PCL increased, the droplet size also increased. However, with the addition of the GMA, the size of the this droplets decreased up to 3 phr. Beyond this value, the trend reversed due to increased chain mobility from the plasticization of GMA, resulting in expansion of the PCL domains. In nanocomposites, NCC particles prevented droplet coalescence by creating physical barriers. In P85N11, the formation of NCC aggregates led to fewer particles surrounding the droplets, allowing them to grow dimensionally.
In Figure 4(b), the micrograph of P75 suggests poor interfacial adhesion between the PLA matrix and PCL domains, indicating complete incompatibility of these two polymers, which is supported by previous studies.51,52 A similar structure is observed in P75G3 (Figure 4(f)), with smaller PCL droplets compared to those in P75, showing improved compatibility of polymers. The positive effect of GMA on modifying the interfacial adhesion of PLA/PCL was also reported. 33 In addition, SEM analysis demonstrated that the incorporation of GMA into PLA/PCL resulted in enhanced compatibility by promoting multiple crazing and cavitation mechanisms. The same conclusion according to SEM analysis was made as an evidence of improved compatibility between maleated SEBS and PLA/cellulose nanocrystal. 53
In the case of the P75N9 sample (Figure 4(j)), the size of PCL domains is smaller compared to P75, but larger than those in P75G3. These results suggest that NCC can also act as a compatibilizer for the PLA/PCL. This incident may be due to the steric hindrance of nanoparticles in the merging of PCL droplets.
Spectroscopy
Fourier transform infrared analysis
Fourier Transform Infrared Analysis (FT-IR) was carried out and spectra of the samples are shown in Figure 5(A). The peaks in the range of 3400–3600 cm−1 correspond to O-H group stretching vibrations, while the peaks around 2800–3000 cm−1 are associated with the symmetric stretching vibration of CH groups in saturated hydrocarbons. The intense peak at around 1760 cm−1 is attributed to C=O stretching vibrations. Several weaker peaks in the range of 1050–1250 cm−1 are assigned to C-O from carboxyl groups and C-O-C stretching vibrations. Three peaks at 1300–1500 cm−1 may be attributed to the vibration of C-H in CH3 groups. FT-IR curves (A): (a) P100, (b) C100, (c) P75, (d) P75G3, and (e) P75N9, (B): normalized FT-IT spectra with baseline correlation in the wavelength range of C-O bond stretching vibration.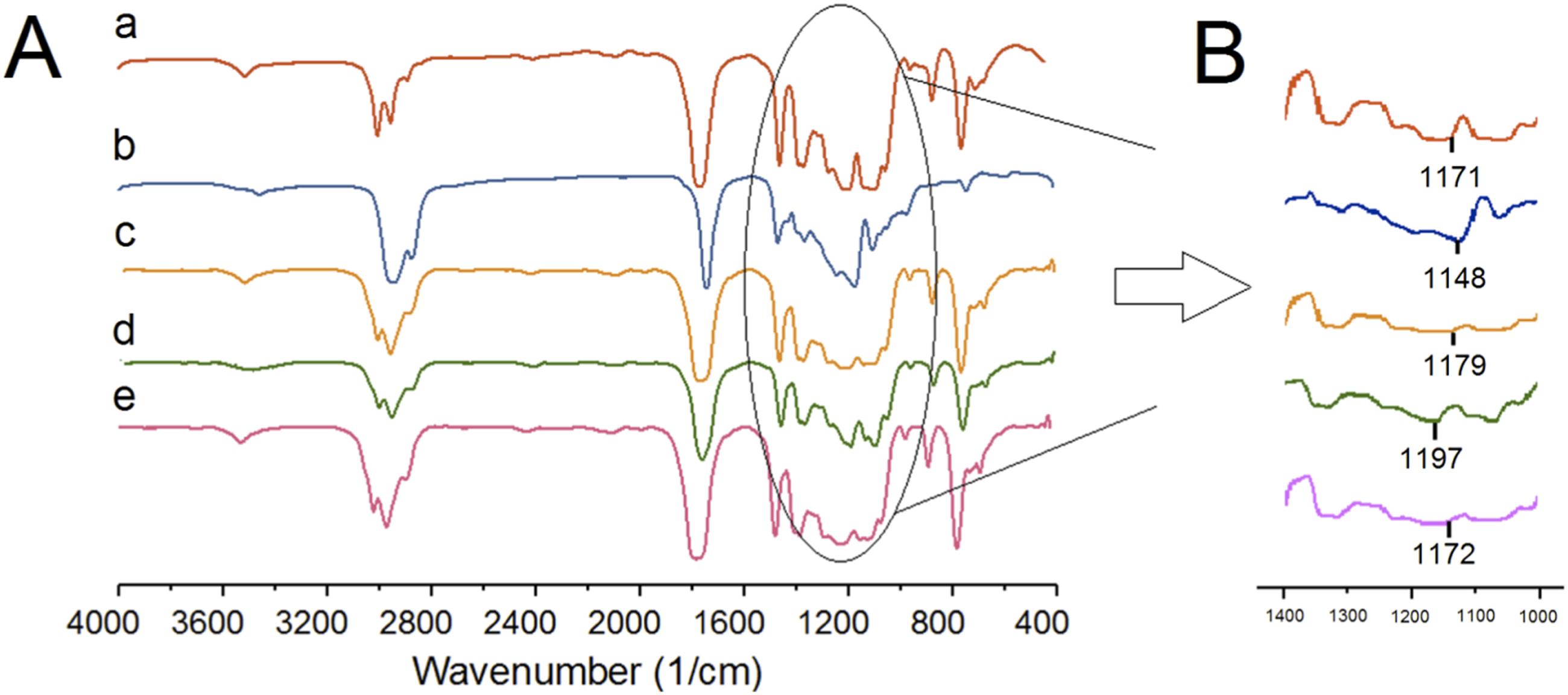
The absence of significant shifts in wavenumbers between P75 and P100 suggests no interactions between PLA and PCL upon blending. Figure 5(B) shows that compared to P75, the C=O stretching vibration of P75G3 remained nearly unchanged, indicating no reaction on the C=O bonds of PLA and PCL backbone chains. Figure 5(B) illustrates the C-O bond stretching vibration, indicating a noticeable shift when GMA is introduced into blend, according to peaks depicted in Figure 4. This suggests reactions between the hydroxyl groups of PLA and PCL with the epoxy group of GMA (Figure 6), potentially increased compatibility. Furthermore, the FT-IR peaks of P75N9 and P75 exhibit subtle changes. In addition to the steric hindrance effect caused by NCC, it is deduced that the presence of stearic acid on the surface of NCC slightly absorbed polymer chains. This observation corresponds with previous works suggesting that steric hindrance plays a role in morphology stabilization in nanocomposites in the presence of nanoparticles.54,55 It is also highlighted that the localization of nanoparticles at the polymer interface leads to higher compatibility between the two polymers. FT-IR curve of GMA.
Nuclear magnetic resonance
Nuclear Magnetic Resonance (NMR) is a highly efficient technique for determining the composition of unknown materials and accurately identifying reactions between different materials during the mixing process. In this study, NMR was performed solely on the PLA, PCL, and compatibilized blend, as there were no expected reactions during the mixing of PLA/PCL with nano calcium carbonate. As depicted in Figure 7, the reactions between the hydroxyl and carboxyl groups of PLA and PCL with the epoxy group of GMA were observed in two separate schemes, 1 and 2, with peaks at 76.3 and 139.1 ppm in 13C NMR, and 6.7 and 7.1 ppm in 1H NMR. These peaks are believed to originate from a methylene unit (-O-CH2-CH3) produced during the melt-blending process, primarily through chain extension due to potentially involving esterification. GMA also caused noticeable changes in peaks related to the carbon functional groups of both polymers, bringing them closer together compared to their original state, as observed in previous study.
56
The longer chains of PLA and PCL played a beneficial compatibilizing role by providing only one possible reaction site. In contrast, chain extender agents such as Joncryl ADR with a higher number of reaction sites can sometimes lead to phase separation due to uncontrolled cross-linking reactions, while chain scission can also deteriorate chain extension process and cause a decrease in properties.
57
The (a)-13C NMR, and (b)-1H NMR of P75G3, (c)-13C NMR and 1H NMR of PLA and PCL, and (d)-possible bond formation during esterification.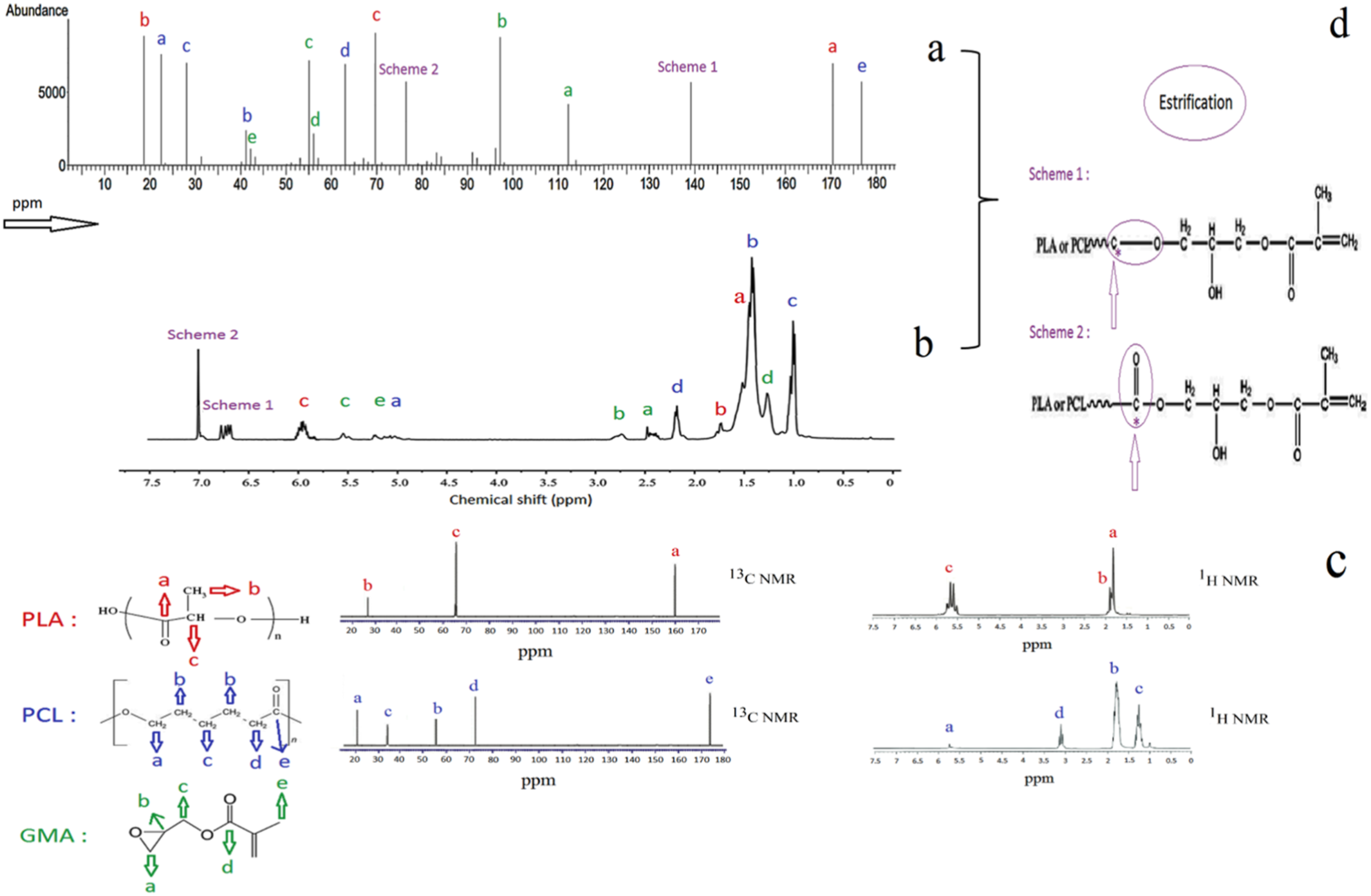
In 1H NMR, PLA exhibits peaks at 5.13 and 1.56 ppm, corresponding to protons in the methyne unit and methyl unit, respectively. PCL shows peaks at 4.07, 2.29, and 1.38 ppm, representing protons in methylene units at different positions on the backbone. GMA displays peaks at 2.33, 1.64, and 0.93 ppm, representing methylene, methylene, along with a peak at 2.11 ppm for the methyl unit in the carboxyl side group and peaks at 3.29–5.47 ppm for the hydroxyl backbone.
In 13C NMR, PLA exhibits peaks at 16.7, 69.1, and 177.3 ppm for methyl, methylene, and carbonyl carbons, respectively. PCL displays peaks at 24.7–25.6, 28.5, 34.2, and 64.2 ppm for four methylene carbons and a peak at 173.8 ppm for the carbonyl carbon. GMA shows peaks at 100.2, 73.0, 72.5, 76.8, and 71.9 ppm for the backbone, 62.3 ppm for the methylene carbon, 170.1 and 35.9 ppm for the carbonyl and methyl carbons in the acetate side group, and 172.6, 21.0, 18.7, and 13.8 ppm for the carbonyl carbon, methylene carbons, and methyl carbon in the epoxy side group. Chen et al. 34 in another study reported that in PLA/PCL/PLA-g-GMA blend, besides the PLA peaks, PLA-g-GMA exhibited 13C signal peaks at 49.74 and 44.98 ppm, as well as 1H signal peaks at 3.25 and 2.85 ppm. These peaks corresponded to the chemical shift of epoxy groups, suggesting successful grafting of GMA onto the PLA structure.
Dynamic mechanical analysis
The main goal of the DMA is to study Tg and investigate the molecular interactions within the amorphous and crystalline regions of polymers.
58
As can be seen in Figure 8, nanoparticles in P75N9 led to a higher modulus compared to P75G3. However, the modulus of P75N9 experienced a quicker decrease as the temperature increases. This can be attributed to the rearrangement of NCC particles and transformation of nanoparticle network. The introduction of nanoparticles served to restrict the mobility of polymer chains, thus impeding their movement. Consequently, this resulted in a decline in molecular motions, which in turn contributed to a rise in the overall modulus of the nanocomposite. As the temperature increased, the role of nanoparticles as physical barriers became less prevalent, hindering the deformability and movement of polymer chains. Thus, this led to a significant decrease in modulus. Generally, nanoparticles often possess a considerable surface area and can form robust interactions with polymer chains. These interactions facilitate load transfer between the nanoparticles and the matrices, boosting their bond formation. However, due to the increased temperature, polymer internal chain segments tend to detach from the surface of nanoparticles due to higher relative motional freedom.
59
Moreover, because of conformational changes at higher temperatures, well-dispersed nanoparticles become less abundant within the polymer matrix, giving rise to formation of defects and regions of lower stiffness. Differences between steric (entropic) states with nanoparticle, does not allow polymer chains to replenish nanoparticles previous position completely. These localized regions characterized by reduced modulus contribute to an overall decline in modulus during DMA. Lastly, as the temperature increases, nanoparticles tend to aggregate or cluster, particularly if they possess a strong mutual attraction, such as NCC particles. These clusters of nanoparticles are susceptible for stress concentration, leading to a drop in the overall modulus.
10
DMTA plots of P75, P75N9, and P75G3.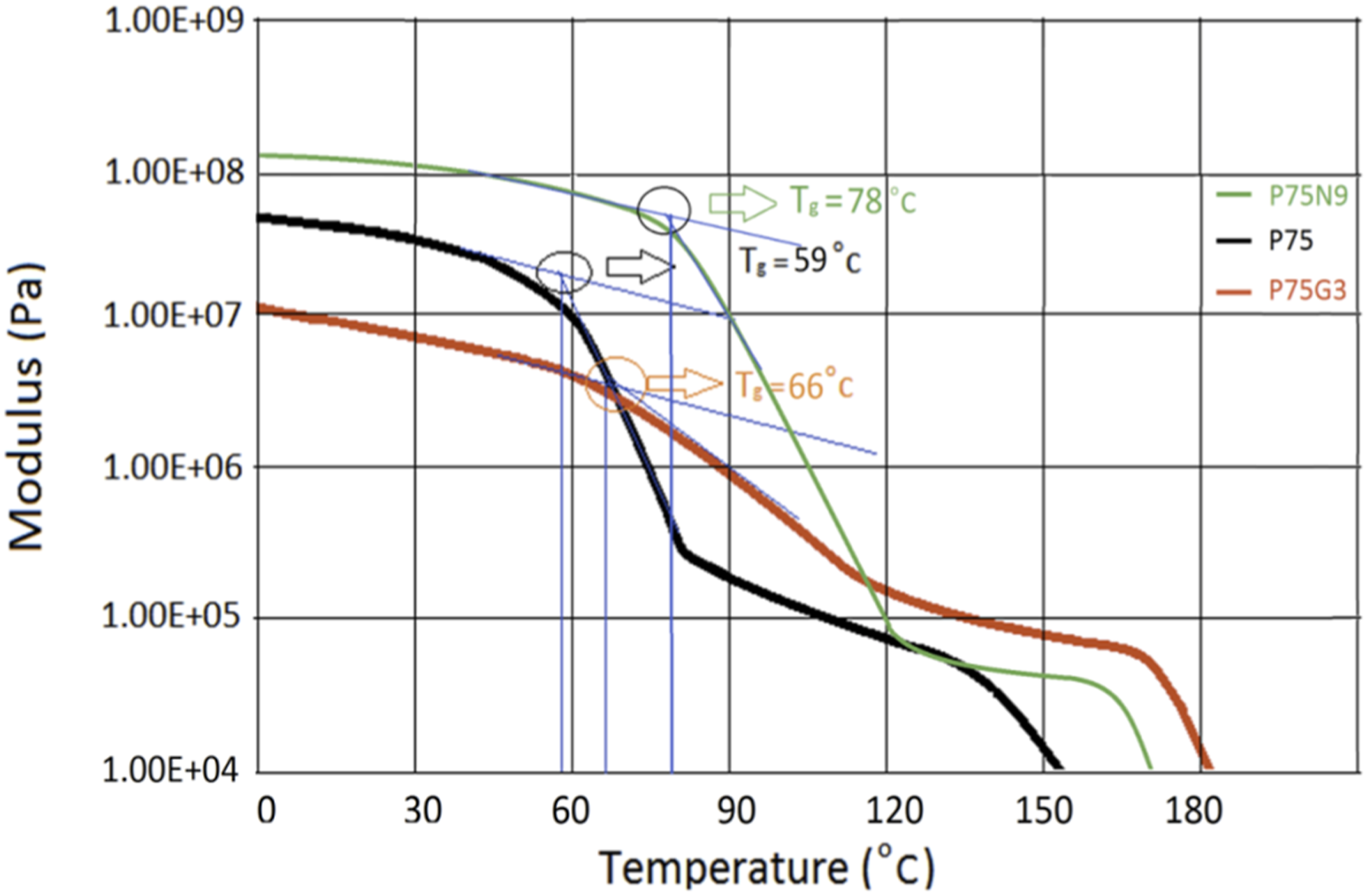
In contrast, the glass transition region of P75G3 appeared in mild slope due to the increased compatibility and notable interaction between PLA and PCL compared to the P75N9. Additionally, the P75G3 exhibited a higher modulus and length in the rubbery region, which can be attributed to the effective performance of GMA in increasing chain extension, enhancing intermolecular entanglements, and promoting mutual attractions among the ends of PLA and PCL chains. (These are solid evidence to prove the formation of longer chains in P75G3).
Gel permeation chromatography
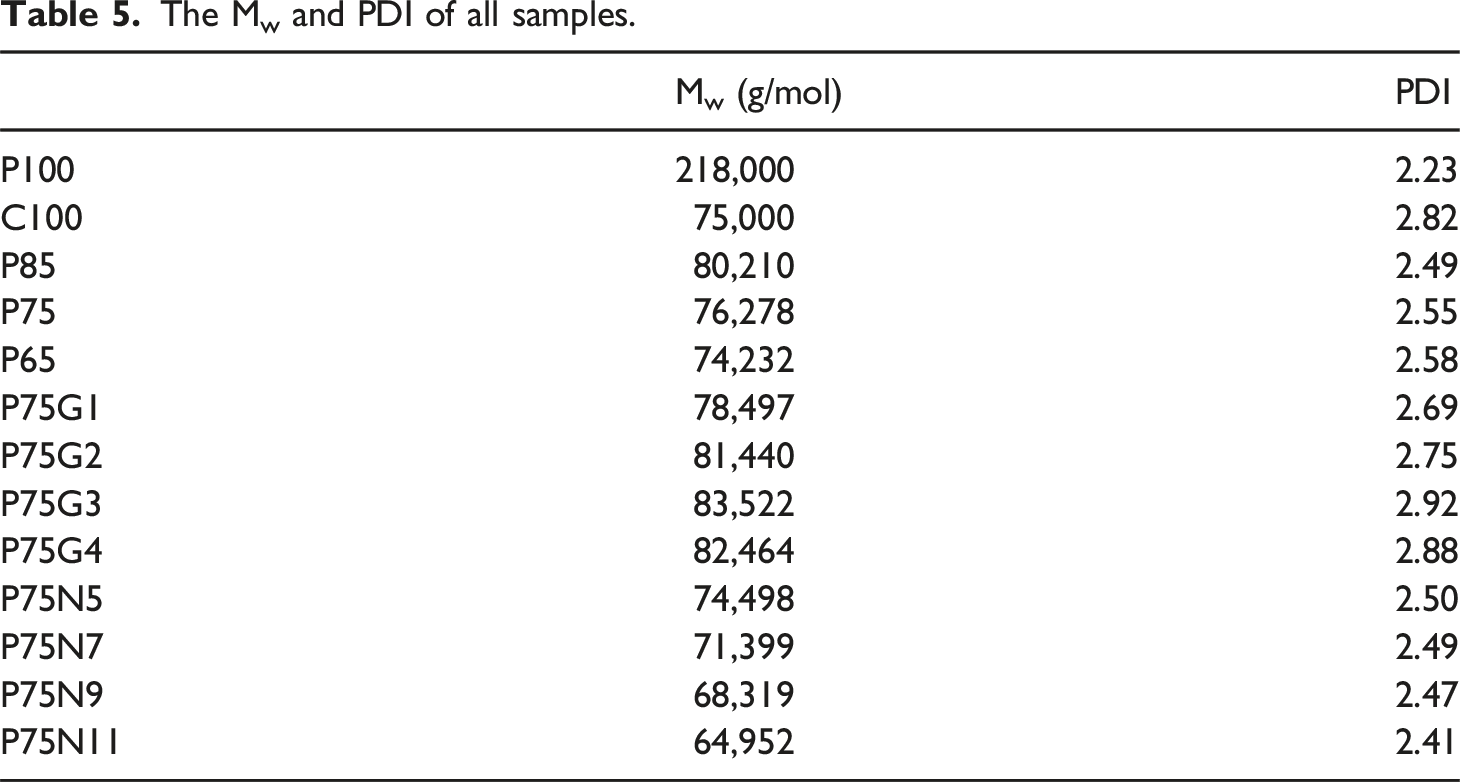
The molecular weight distribution was determined using GPC with the Mark-Houwink-Sakurada parameters provided by Huang et al. 60 The average molecular weight of polylactic acid is approximately 218,000 g/mol, while the average molecular weight of polycaprolactone is around 75,000 g/mol. In addition, weight-average molecular weight (Mw) and polydispersity index (PDI) of all samples were measured.
In uncompatibilized blends, as the percentage of PCL in the composition increases, the lower molecular weight of this polymer and its incompatibility with PLA result in a decrease in both Mw and PDI. However, between compatibilized blends it is evident that GMA in P75G3 has a greater impact on these elements. This increase can be attributed to the reaction of the polymers in the presence of the GMA, while the higher amount of this compatibilizer has the negative effect as it plasticizes the blend and making more free volume and more distance which It destroys the opportunity to create higher bonds between PLA and PCL. In nanocomposites, however, the story is very different as nanoparticles increase the likelihood of polymer chain rupture during mixing. While the surface absorption of the chains on the NCC aids in enhancing structural integrity of chains, the former outweighs this effect Figure 9. Molecular weight distribution curves of: (a) uncompatibilized blends, (b) compatibilized blends, (c) nanocomposites measured by 3D-GPC using a multi-angle light scattering detector.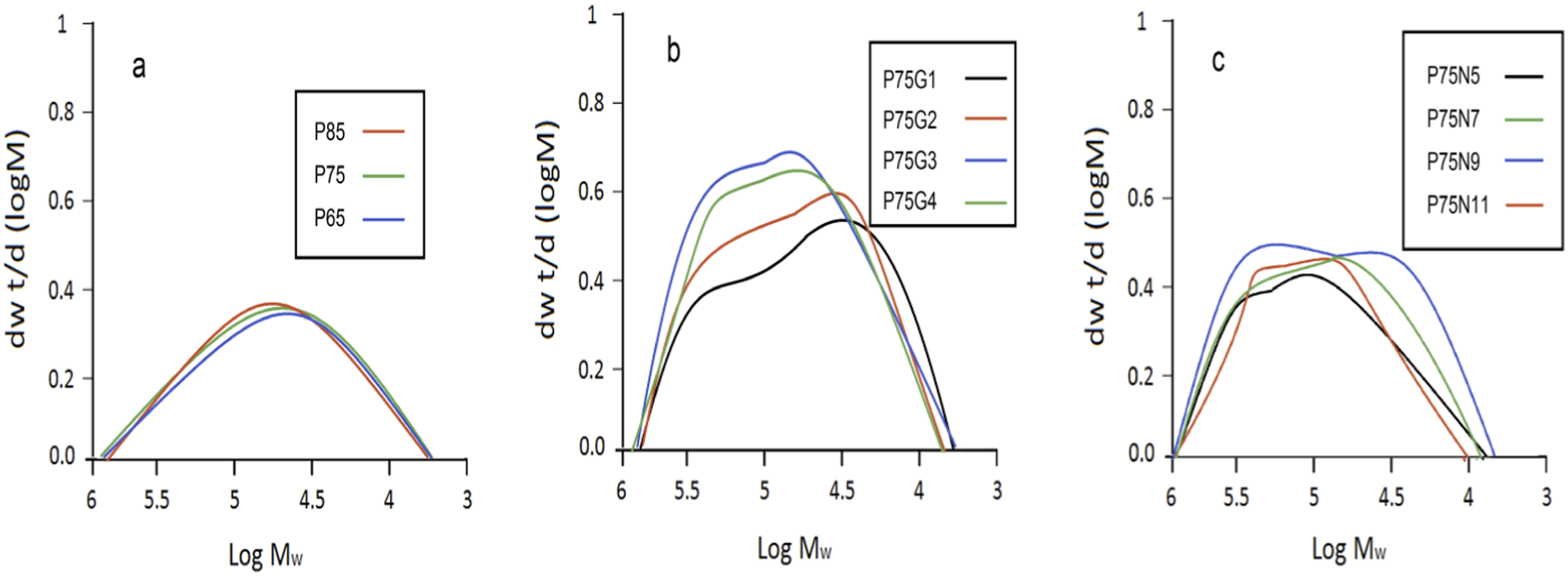
The Mw and PDI of all samples.
GMA led to intermingling of adjacent chains, resulting in a higher molecular weight for P75G3 compared to P75N9. This also made molecular weight distribution wider. However, NCC nanoparticles might interfere with the chain extension process, potentially resulting in their scission during mixing. 63 This could be due to collision of the nanoparticles and chains.
Rheological properties
Small amplitude oscillatory shear (SAOS) test were condocted to study the rheology of samples, with the aim of understanding the relationship between their molecular structures and flow properties. The plots of storage modulus (G′) and complex viscosity (η*) as a function of angular frequency are presented in Figure 10. a) storage modulus, and (b) complex viscosity of all samples.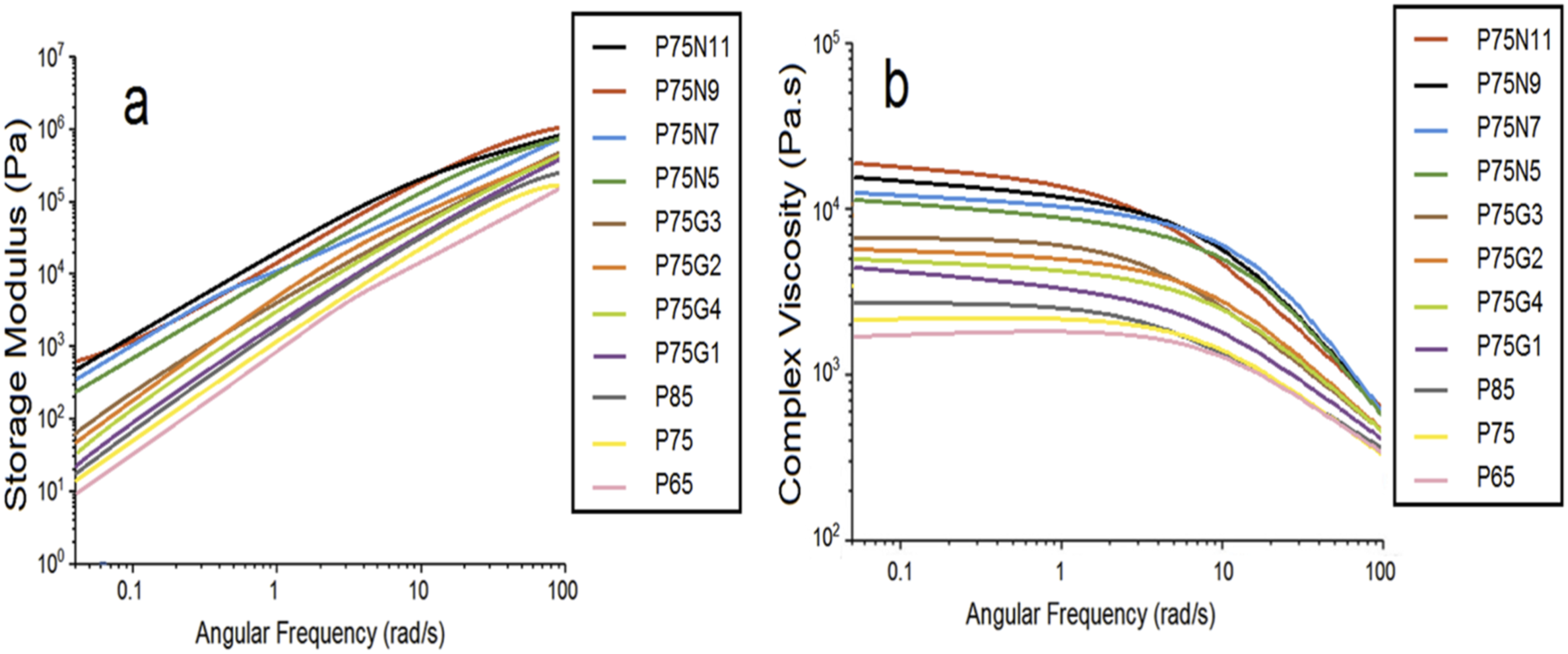
As can be seen, the storage modulus and complex viscosity of uncompatibilized blends decrease as the percentage of polycaprolactone increases, reflecting the softer nature of this polymer. In contrast, compatibilized blends exhibit a similar behavior in the storage modulus and complex viscosity. Increasing the amount of compatibilizer up to 3 phr leads to an increase in both quantities, as the strength of the polymers is enhanced. However, beyond 3 phr, both properties decrease due to excessive amount of GMA acting as a plasticizer. In nanocomposites, a higher percentage of nanoparticles results in an increase in storage modulus and a steeper initial slope in the graph. Furthermore, increasing the amount of NCC in nanocomposites leads to an increase in complex viscosity, accompanied by a decrease in the length of the Newtonian region and a steepening of the modulus drop after this region. Notably, P75N11 exhibits unique behavior at high frequencies, where the breakdown of the nanoparticle network leads to a sudden release of entrapped polymer chains and a lower modulus compared to other nanocomposites. These findings highlight the intricate interplay between nanoparticles and polymers in these composite materials.
Figure 11 demonstrates that both neat PLA and PCL exhibit a terminal behavior, where the storage modulus G′ is proportional to the angular frequency ω2. Additionally, the storage modulus of PCL is significantly lower than that of PLA across all frequencies. When PCL is added to PLA, the storage modulus increases at low frequencies, while the storage modulus of P75 is lower than that of pure PLA at medium and high frequencies, indicating a weak interfacial interaction between polymers. However, GMA leads to higher storage modulus compared to uncompatibilized blend. Nevertheless, beyond 1 rad/s, the storage modulus of P75G3 becomes smaller than that of PLA, suggesting partially enhancement the interfacial adhesion between the polymers. Storage modulus of the nanocomposite (P75N9) is notably higher than that of the other samples, aligning with the findings of previous research on polymer blends incorporating nanoparticles.
64
Moreover, the appearance of G′ plateau at low frequencies in the nanocomposite sample indicates the formation of a three-dimensional network of nanoparticles, resulting in a solid-like behavior. The slope of G′ versus ω plots, denoted as (N), was investigated in the range of 0.01–0.1 rad/s and is shown in Table 6. The results reveal that the blend has a lower initial slope of G′ than PLA, showing the formation of entangled structures in PLA/PCL melts. GMA has a minimal effect on this slope, as the low molecular weight compatibilizer has a negligible impact on increasing the entanglement density. Furthermore, the outset slope of the G′ versus ω plot in P75N9 is much lower than that of neat polymers and compatibilized blends, suggesting nanoparticle aggregation.
65
Storage modulus of samples against angular frequency. Slope of G′ versus ω plots (N), in the range of 0.01–0.1 rad/s and absolute value of degree of shear-thinning (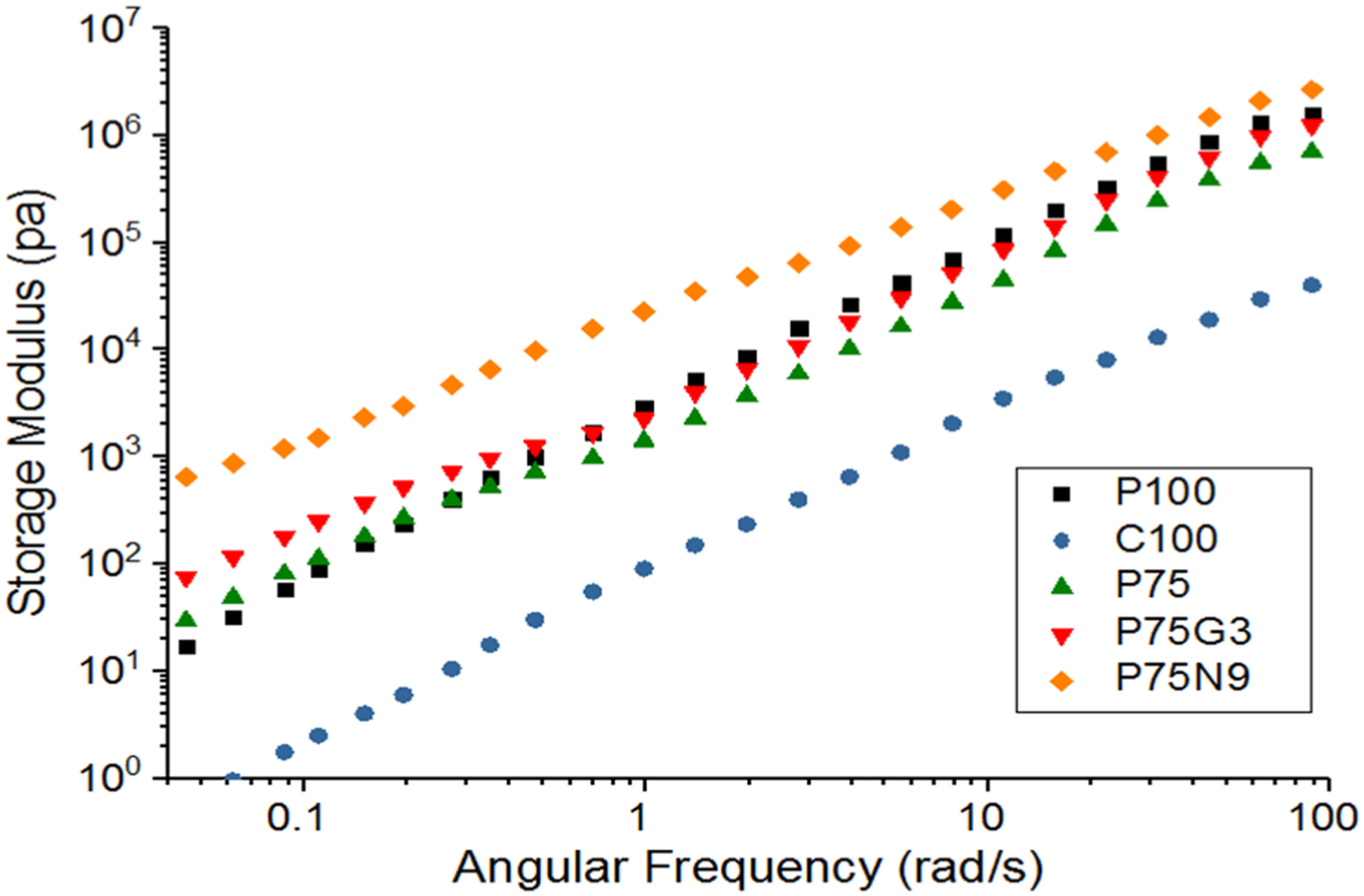

Additionally, the storage modulus plots of the blends exhibit a secondary plateau attributed to the shape relaxation of the PCL dispersed phase PCL.
66
GMA widens this plateau, because of improved compatibility between PLA and PCL. The frequency dependence of complex viscosity for all samples is depicted in Figure 12. It is evident GMA influences the rheological behavior of P75, leading to an increase in complex viscosity due to enhanced connectivity between the PLA and PCL chains. Similarly, the incorporation of NCC particles into PLA/PCL blends significantly alters their rheological properties, resulting in a notable increase in complex viscosity, especially at low frequencies.
67
Some researchers corresponded the observation of this plateau to the location of nanoparticles at the interface between two polymer phases.
68
The outcomes of previous study demonstrated that a huge increase in the quantity of NCC particles present within the interface triggered the formation of aggregates along the polymer interface.
69
This consequently impeded a consistent shape relaxation within the polymer interface owing to the substantial difference in the degree of molecular mobility between the polymer chains in the bulk and the interface. Subsequently, this phenomenon leads to the formation of a distinct separated phase in the proximity of the interface. Complex viscosity of samples against the angular frequency.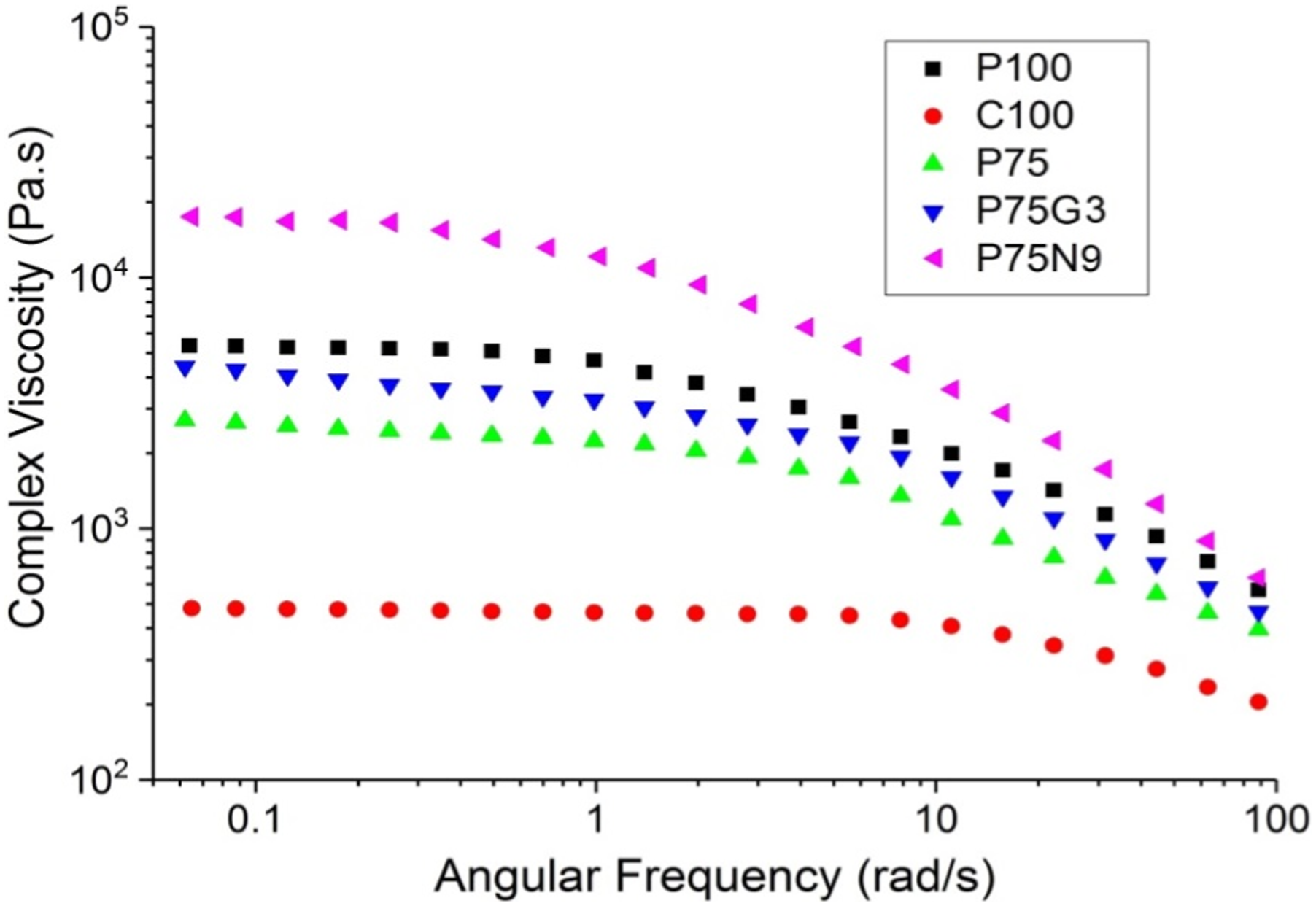
Additionally, it is obvious that interactions between nanoparticle and polymer, as well as particle-particle interactions, making a substantial increase in the complex viscosity of nanocomposite sample, particularly at low frequencies. High frequency measurements revealed that all samples exhibited pseudoplastic behavior, but neat PCL only displayed Newtonian behavior within a limited frequency range (up to 10 rad/s), on the other hand, P75N9 has a smaller frequency range for Newtonian behavior. The viscosity reduction after the Newtonian region, as determined by the degree of shear-thinning (
The value of
Rheological study of the samples was developed using the Palierne model. This model provides valuable information on the interfacial properties of incompatible PLA/PCL blend and nanocomposite. In the Palierne model, complex modulus of matrices and polymer interface have been utilized to predict the complex modulus of samples.
71
The aim of this study is to elucidate the relaxation mechanisms based on polymer chain movements, droplet shape evolution, and relaxation caused by Marangoni stresses.
72
The plots obtained from SAOS test were compared with the plots generated by the Palierne model to assess their overlap. This model is expressed as follows: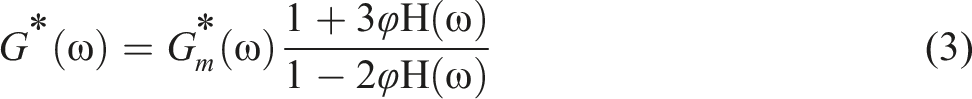
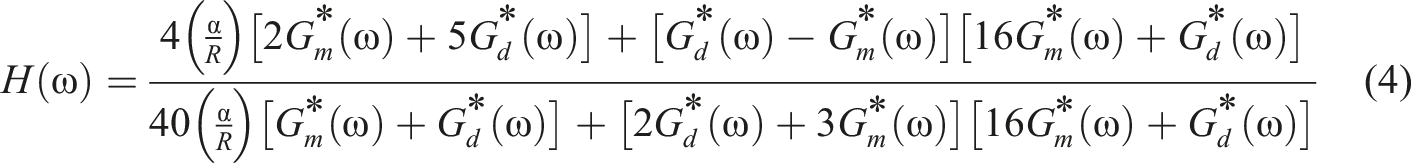
The simplified Palierne model with the approximation of Graebling et al. 73 was used, where the surface dilatation modulus and surface shear modulus were ignored. The fitting program was assigned an initial value of 100 for α/R, and the optimum value was found to be 76.31 N/m2. Using Table 6, the interfacial tension was calculated to be 0.29 mN/m for PCL droplets with an average diameter of 7.6 μm for P75. Also, P75N9 and P75G3 were compared with Rv extracted from Table 6 and the same interfacial tension. 74
As shown in Figure 13, the Palierne predictions for P75N9 and P75G3 showed a discrepancy in the actual value. It is assumed that in the P75N9, the NCC particles are located both at the interface and in polymer phases, which does not mean that particles saturated the interface, but rather that excessive amounts of NCC were located in the droplets, hindering their shape relaxation.
75
In the case of sample P75G3, there was more similarity between the fitted plot and the experimental one. Therefore, the effect of GMA on the compatibility of polymers is more remarkable, partially increased their interfacial adhesion during the mixing procedure. Fitted storage modulus of samples based to palierne model.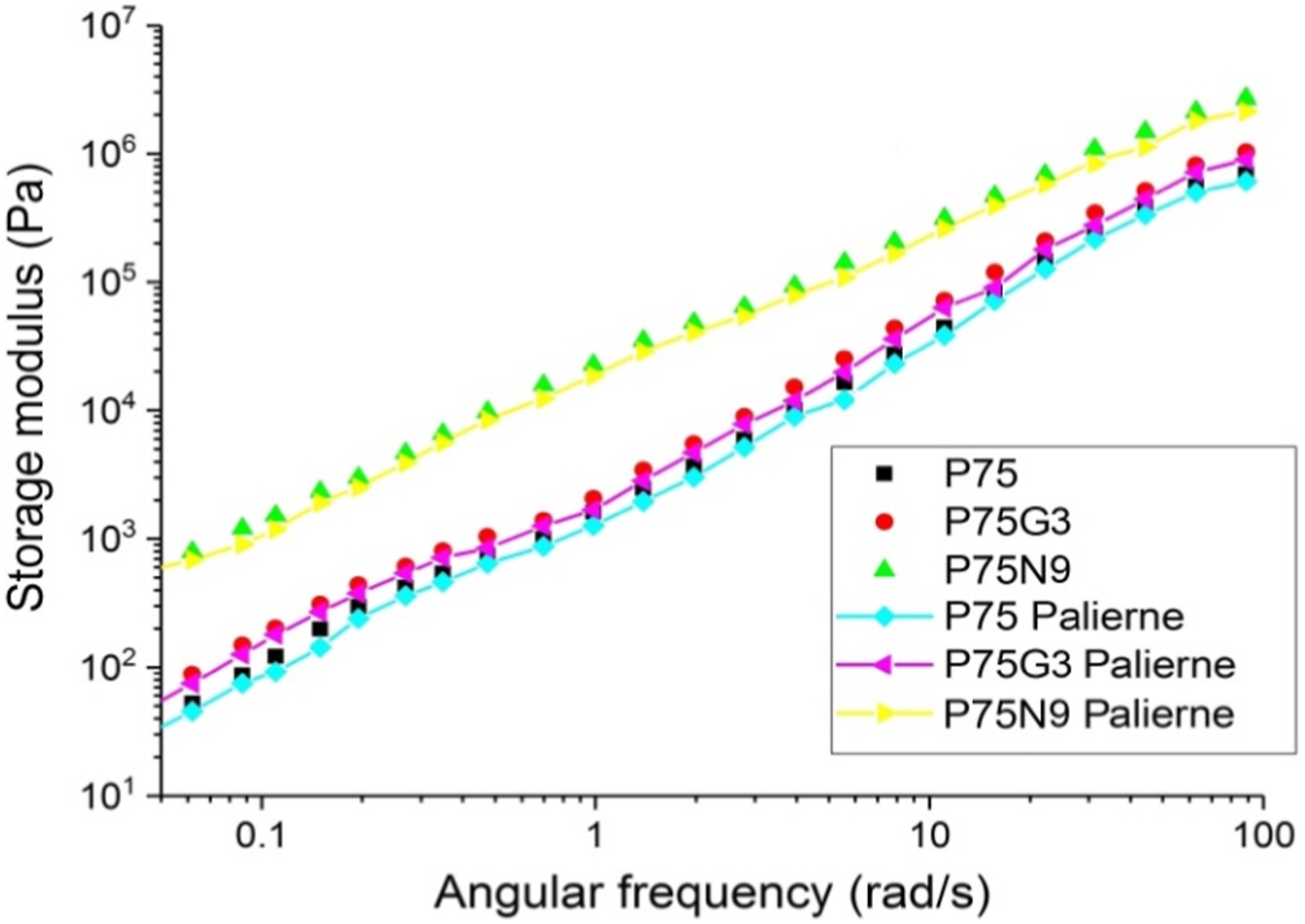
Conclusion
Mechanical, thermal, morphology, and rheological properties of PLA/PCL blends and nanocomposites were investigated with the aim of analyzing the impact of GMA and NCC. GMA enhanced the elongation at break and impact strength of the PLA/PCL. NCC, on the other hand, increased tensile modulus and impact strength. DSC revealed that GMA and NCC altered the degree of PLA crystallinity in the PLA/PCL. While NCC acted as a nucleation agent, GMA enhanced the crystallinity of the blend through better distribution of PCL in PLA and increasing the chain mobility. NMR and FT-IR spectroscopic analyses also unveiled the reaction between the epoxide group of GMA and the hydroxyl and carboxyl groups of the polymeric chains. The effect of GMA was evident in the spectroscopic range of 1150 to 1200 cm−1, which is associated with the stretching vibration of C-O bond. Furthermore, GPC plots demonstrated that GMA increased the average molecular weight of P75 from 76,278 to 83,522 g/mol, whereas NCC caused a reduction in the molecular weight to 68,319 g/mol. Additionally, GMA resulted in a 0.5-fold increase in the molecular weight distribution, indicating the impact of GMA in generating longer chains. Moreover, DMA indicated that Tg was higher in nanocomposite, compared to compatibilized blend. Rheological results showed a significant increase in storage modulus and complex viscosity in the nanocomposite and compatibilized blend compared to the neat polymers, particularly at low frequencies. Nevertheless, according to the Palierne model, localization of NCC particles at the interface of PLA/PCL hindered the elastic response at the intermediate frequencies. In the compatibilized blend, the PCL dispersed phase exhibited a noticeable elastic response, which was different from its state before the addition of GMA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Data is available within the article and can be available on request.
