Abstract
Cocoa pod husk (CPH)-filled polypropylene (PP) composites were prepared via melt compounding. The effect of filler content and chemical treatment using 3-mercaptopropyltrimethoxysilane (MPS) and sodium dodecyl sulfate (SDS) on properties of composites were investigated. The results indicated that the treated composites with MPS and SDS improved the tensile strength, tensile modulus, thermal stability, stabilization torque, water resistivity, and crystallinity of composites. The treated composites with SDS show better tensile properties and water resistivity than composites treated with MPS. Scanning electron microscopic results show that the interfacial bonding between CPH and PP matrix improved with the presence of MPS or SDS.
Introduction
Cocoa is an important agricultural export commodity and the most widely planted crop in several tropical countries including Malaysia. 1–3 Currently, cocoa pod husk (CPH) is a major by-product of the cocoa industry. In the cocoa industry, CPH is a nonfood part of the cocoa pod, and it accounts for as much as 76% of cocoa pod by weight. 2,3 For every ton of dry cocoa bean produced, there are 10 tons of CPH generated as waste. 4 The CPH is readily abundant but does not have any marketable value; therefore, the utilization of CPH as natural filler could provide a new and sustainable source of filler for plastic industry. Meanwhile, the waste levels of cocoa industry could be reduced.
In recent years, composites from agrowaste and thermoplastic materials have garnered interest among researchers and industries due to today’s environmental issues and economic advantages as well as the accumulation of agricultural waste by-product. 5–8 Nowadays, various combination of agricultural waste and thermoplastic material have been successfully produced commercial products. IKEA injection-molded furniture is a well-known example of commercial product, which is made from polypropylene (PP)/wood flour composites. 9 In Malaysia, a series of eco-tableware was made from rice husk-filled thermoplastic eco-composites by Melsom Biodegradable Enterprise. 6 Hence, the present research was comprehensively to utilize CPH as filler for PP composites. The development of such composites has a potential to replace current forest product, such as wooden fittings, fixtures, deck, and furniture as well.
Generally, the compounding of natural filler in thermoplastic materials would not produce a good composite due to the poor interfacial compatibility between the hydrophilic natural filler and the hydrophobic matrix. 10,11 Thus, filler treatment using silane coupling agent is one of the effective methods used to modify the hydrophilic properties of natural filler. Silane is able to form covalent linkage with the hydroxyl groups of natural filler.The alkyl chains from silane provided hydrophobic properties to natural filler and improved the interfacial compatibility. 12,13 Thus, many researchers reported that the silane coupling agent does remarkably to improve the water resistivity and mechanical and thermal properties of composites. 12–21 Moreover, some literatures reported that the use of fatty acid for filler treatment will also give a coupling effect to the composites. Fatty acid is made from sustainable resources, and it is inexpensive compared to commercial silane coupling agent. Consequently, fatty acid is an alternative choose of coupling agent besides silane. In our previous study, sodium dodecyl sulfate (SDS) was used as coupling agent to enhance the filler–matrix adhesion between coconut shell–polylactic acid 6 and chitosan–polypropylene. 22 The use of SDS shows a remarkable result in improving water resistance and tensile and thermal properties of composites.
Currently, the studies on PP/CPH composites were reported in our previous research, 1–3 and the use of 3-mercaptopropyltrimethoxysilane (MPS) in natural filler-based composites is not found in the literature. Thus, the present work was undertaken to compare the effect of filler treatment using MPS and SDS on torque development, tensile properties, water absorption, thermal properties, and morphology of PP/CPH composites.
Methodology
Materials
The discarded CPH was obtained from cocoa plantation, Perak, Malaysia. The CPH was dried in an air circulatory oven at 80 C for 24 h. Then, the dried CPH was crushed into small pieces and ground into fine powder. The average particle size of CPH produced was 22 µm, which is analyzed by Malvem Particle Size Analyzer Instrument. PP, type copolymer, grade SM 340, was supplied by Titan Petchem (M) Sdn. Bhd (Malaysia). The melt flow index value of PP was 4.0 g/10 min at 230 C and density 0.9 g cm−3, respectively. MPS (97%) and SDS (98%) were supplied by Sigma Aldrich (St Louis, Missouri, USA). Ethanol (Fluka, Penang, 95%) was used as the solvent in filler modification.
Filler treatment
Firstly, MPS (3% based on weight of filler) was dissolved into ethanol. The CPH powder was added into MPS solution and stirred continuously for 1 h. The CPH was soaked in MPS solution and left overnight. The soaked CPH was filtered and dried in an oven at 80 C for 24 h. The similar procedure was carried out for filler treatment using SDS.
Melt compounding and molding procedures
The formulation of PP/CPH composites are listed in Table 1. All composites were compounded using a Brabender® Plastrograph intermixer (model EC PLUS, Germany) in counterrotating mode at 180 C and a rotor speed of 50 r min−1. The compounding procedures involved were as follows: (i) the PP pellets were added into the mixing chamber for 3 min until it melted homogeneously; (ii) the unmodified or modified CPH was incorporated to the melted PP and continuously mixed for 5 min. Finally, the composites compound was collected from mixing chamber. All the compounds were molded into 1-mm thick sheets using a compression molding machine (Gotech, Taiwan, Model GT 7014A) at 180°C. The compression sequences involved were as follows: (i) preheat the compound for 4 min; (ii) compress the compound under a pressure of 100 kgf cm−2 for 1 min; and (iii) cooling under the same pressure for 5 min. Then, the PP/CPH composite sheets were cut into tensile specimens using a dumbbell cutter with dimensions according to ASTM D638 type IV. 23
Formulation of PP/CPH biocomposites.
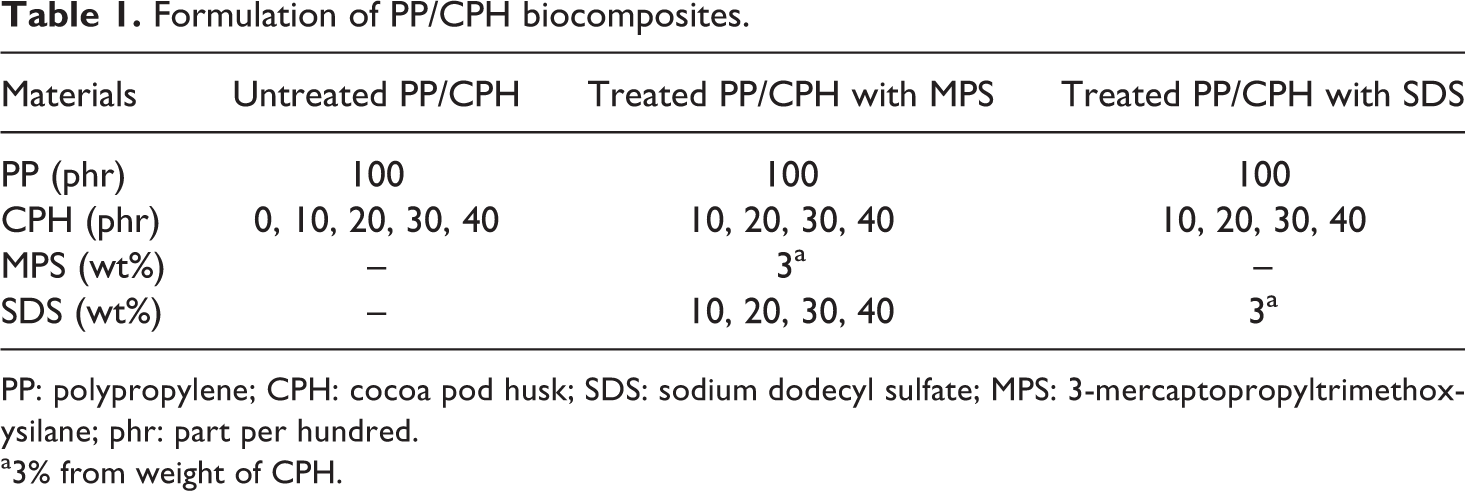
PP: polypropylene; CPH: cocoa pod husk; SDS: sodium dodecyl sulfate; MPS: 3-mercaptopropyltrimethoxysilane; phr: part per hundred.
a3% from weight of CPH.
Processing torque measurement
The processing torque was measured during the compounding of composites using Brabender Plastrograph internal mixer. The processing characteristics of composites compound with the time were recorded and the torques versus time curves was plotted by Brabender Mixer Program (WINMIX). The torque values at the end of processing time were taken as stabilization torque.
Tensile testing
Tensile testing was performed by using an Instron Testing Machine (model 5569, Norwood, Massachusetts, USA) according to ASTM D638 standard. 23 The test was carried out at 25 ± 3 C. A crosshead speed of 30 mm min−1 was used and the load cell selected was 50 kN. A minimum of five specimens were tested for each composite.
Morphological analysis
The fracture surface of tensile specimens were examined using scanning electron microscope (SEM; model JEOL JSM-6460 LA, Japan). The samples were coated with a thin layer of palladium for conductive purpose before they were analyzed at 5 keV.
Water absorption test
All composite samples with dimension 30 × 25 × 1 mm3 were prepared and dried before immersed in water. The specimens were immersed in distilled water at room temperature and the water absorption was determined by record sample weight at regular intervals. A Mettler balance (model AJ150; Columbus, Ohio, USA) with precision of ± 1 mg was used to measure the weight of sample. The water absorption at time t (W
a) was calculated by formulation below:
where W d and W n are original dried weight and weight after exposure, respectively.
FTIR spectroscopy
Perkin Elmer Paragon 1000 Fourier transform infrared (FTIR) spectrometer (Waltham, Massachusetts, USA) was used to characterize chemical functional groups in unmodified and modified CPH. The attenuated total reflectance method was selected. The sample was recorded with eight scans in the frequency range of 4000–600 cm−1 with a resolution of 4 cm−1.
DSC analysis
Differential scanning calorimetric (DSC) analysis was evaluated using DSC Q10 (TA Instruments, USA). The specimen was cut into small pieces and placed into closed aluminum pan with a weight in range of 7 ± 2 mg. The specimen was heated from 30°C to 200°C at a heating rate of 10°C min−1 under nitrogen atmosphere. The nitrogen gas flow rate was 50 ml min−1. The degree of crystallinity of composite (X c) can be calculated from DSC data by following equation:

where ΔH
f is the heat fusion of the PP composites, and
The crystallinity of PP matrix (X PP) was calculated using following equation:

where W f PP is the weight fraction of PP matrix.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was carried out using TGA Pyris Diamond Perkin Elmer apparatus. The samples were about 7 ± 2 mg in weight and were placed into platinum crucible. Then, the weight loss against temperature was measured at a heating rate of 10°C min−1 and range of thermal scan from 30°C to 700°C.
Results and discussion
Processing torque
Figure 1 illustrated the processing torque versus times curves for the neat PP, untreated PP/CPH composites with different CPH content. The first processing torque rose rapidly due to the shearing action from the solid PP pellets. Then, the processing torque gradually reduced indicating decreasing viscosity as PP pellets melt when subjected to high temperature and continuous shearing. The second torque development can be found after 3 min. This is because the addition of the CPH which interrupted the flow of melted PP. The processing torque decreased gradually and achieved stabilization torque after the compound in homogenously mixed. Similar trends of processing torque have also been reported by other researchers. 1,16,24,25 The stabilization torque was increased with the increasing of CPH content (as shown in Figure 2). This is due to the resistance from CPH particles which increased the viscosity of PP. 1,3 From Figure 2, the processing torque of treated PP/CPH composites with MPS or SDS were higher as compared to untreated PP/CPH composites. This might be because CPH treated with MPS or SDS have better filler dispersion and adhesion with matrix that led to increase the viscosity of the composites.
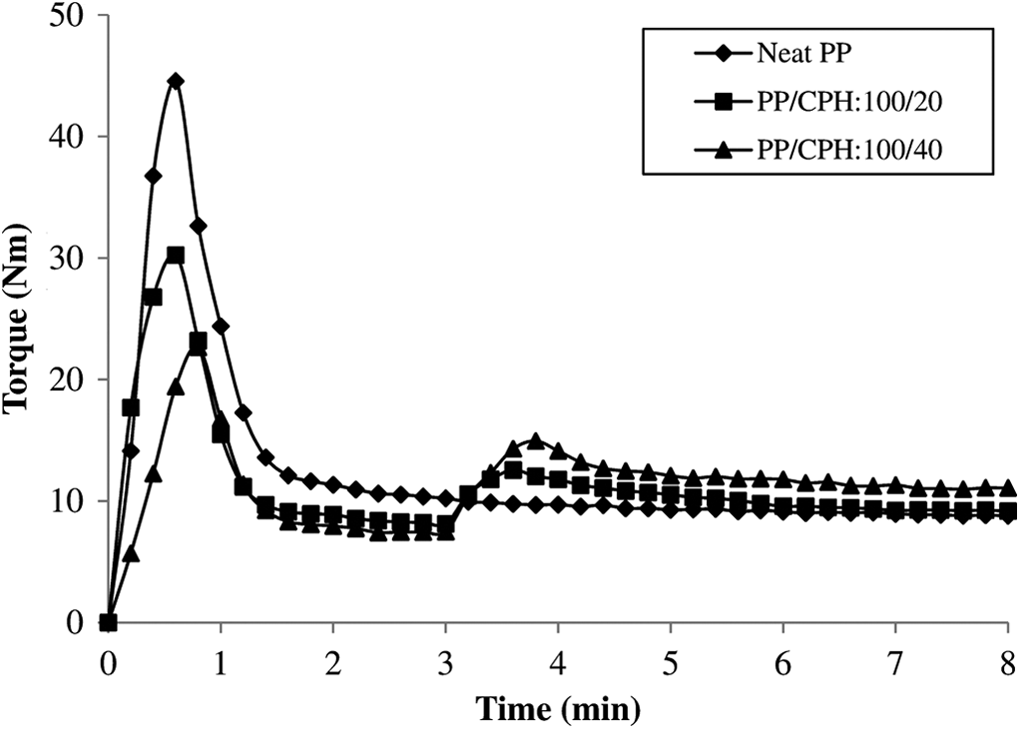
The torque–time curves of neat PP and PP/CPH composites with different filler content. PP: polypropylene; CPH: cocoa pod husk.
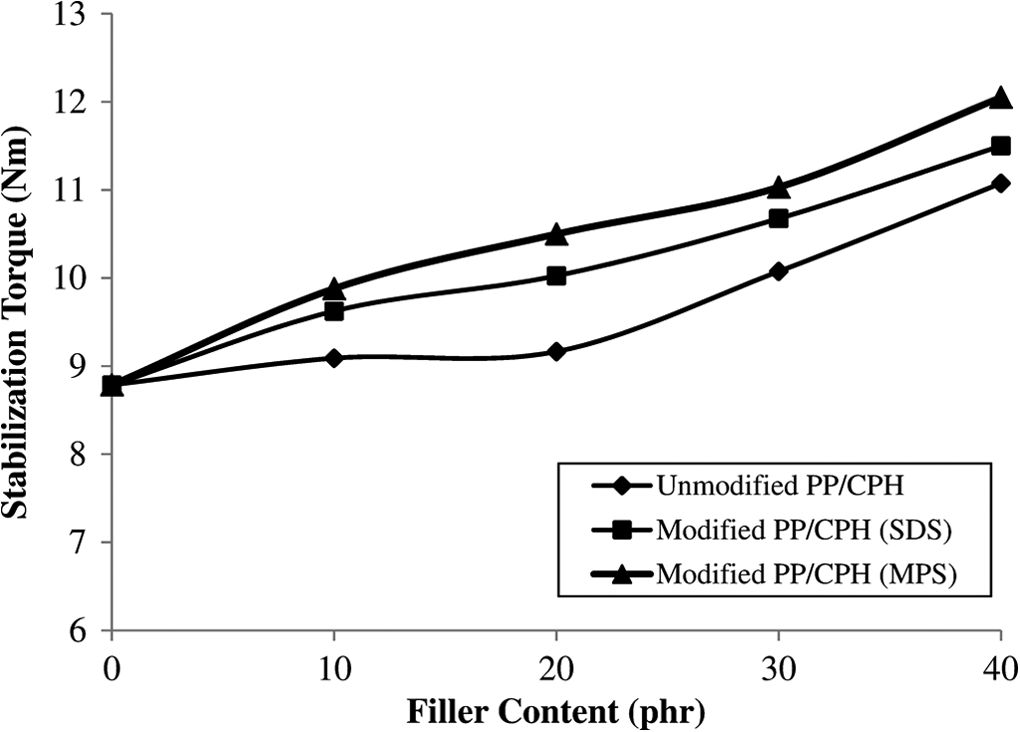
Stabilization torque of untreated and treated PP/CPH composites with MPS and SDS. PP: polypropylene; CPH: cocoa pod husk; MPS: 3-mercaptopropyltrimethoxysilane; SDS: sodium dodecyl sulfate.
Tensile properties
Figure 3 shows the effect of filler content on tensile strength of untreated and treated PP/CPH composites with MPS or SDS. The incorporation of CPH decreased the tensile strength of PP matrix. This was a common trend for the particular natural filler containing thermoplastic composites, whereas the similar trend was found in previous work. 5,6 Usually, the irregular shape CPH filler has low aspect ratio and poor efficiency of transferring tensile stress from matrix to filler. Thus, the incorporation of CPH reduced the tensile strength of PP matrix. The decreased tensile strength was also supported by the weak interfacial adhesion and the presence of filler agglomeration. Alternatively, the tensile strength of treated PP/CPH composites increased compared with the untreated PP/CPH composites, but the tensile strength was still lower as compared to neat PP. The treated CPH with MPS or SDS had long alkyl chains covalent bonded on its surface that lead to increase the wettability with PP matrix and it enhanced the interfacial bonding between CPH and PP matrix. Chun et al. 6,12 reported that treated coconut shell with silane coupling agent and SDS improved the tensile strength of the composites.
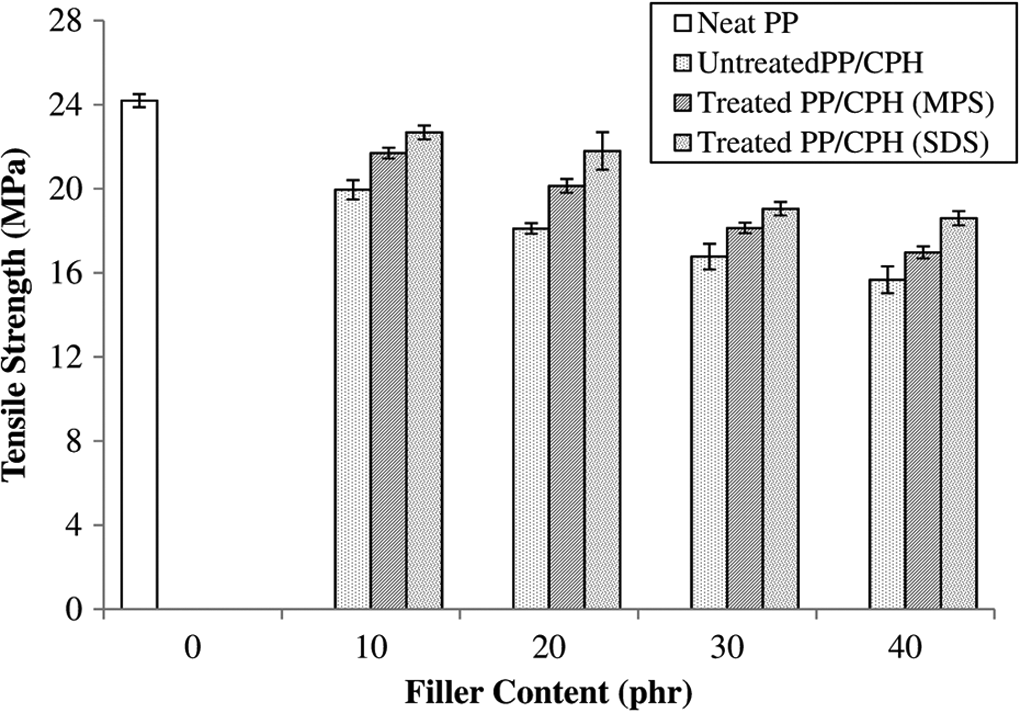
Effect of filler content on tensile strength of untreated and treated PP/CPH composites with MPS and SDS.
The elongation at break of untreated and treated PP/CPH composites is displayed in Figure 4. The results indicated that the elongation at break of PP/CPH composites had decreasing trend as the CPH content increased. The addition of rigid CPH particles restricted the PP chains mobility, which increased brittleness of composites. This is a common trend found by other researchers. 13,14,16,21,24 Treated PP/CPH exhibited lower elongation at break values compared to untreated PP/CPH. The filler modification with MPS or SDS altered the CPH properties, making it less hydrophilic and increased the filler–matrix adhesion. Thus, the flexibility of PP matrix was reduced by the stronger filler–matrix adhesion. Other researchers reported a similar influence of silane coupling agent 20 and SDS 22 on the elongation at break of PP/chitosan composites.
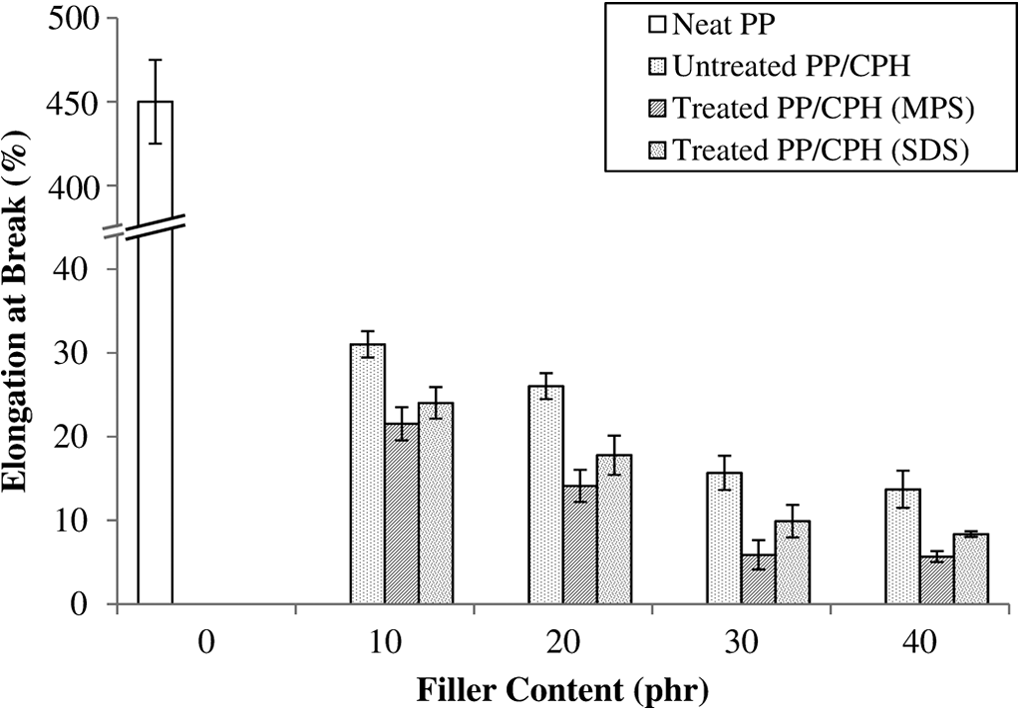
Effect of filler content on elongation at break of untreated and treated PP/CPH composites with MPS and SDS.
However, the increase of CPH content has increased the tensile modulus of treated and untreated PP/CPH composites (as illustrated in Figure 5). This is due to the fact that the friction at interface region between CPH particles and PP matrix led to a rigid interface which restricted the polymer chain mobility. This increased the rigidity and stiffness of composites. A similar observation also reported by other researchers. 26,27 Furthermore, the tensile modulus of PP/CPH composites increased by filler treatment using MPS and SDS. It can be seen that the treated CPH with MPS or SDS had better interfacial adhesion with PP matrix, which led to increase tensile modulus. A similar result was also reported in the literature for treated composites with silane coupling and fatty acid. 5,19,24 –27
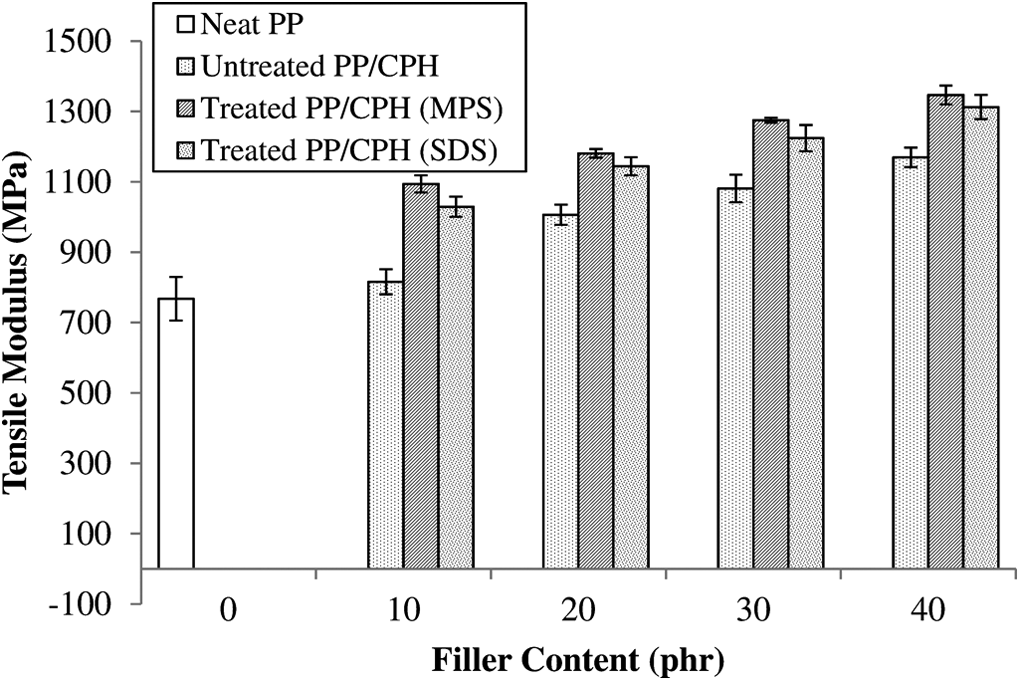
Effect of filler content on tensile modulus of untreated and treated PP/CPH composites with MPS and SDS.
The effect of filler treatment on interfacial interaction can be expressed quantitatively by a simple model developed by Pukánsky.
28
This model (Equation (4)) takes into account the most important factors influencing the tensile strength, such as (i) λ
n—the change of specimen dimensions during the deformation and the rise of tensile strength due to strain hardening; (ii)
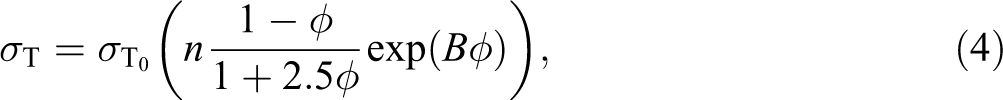
where σT and 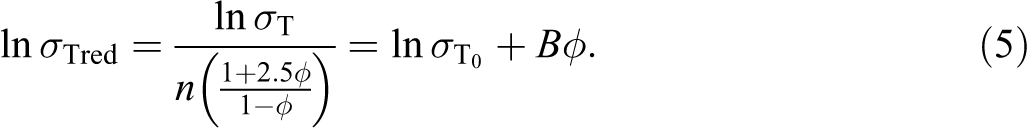
The plot of ln-reduced tensile strength (σ Tred) as a function of filler content should give a straight line with a slope of B. Figure 6 displayed the plot of reduced tensile strength of untreated and treated PP/CPH composites versus filler content. A good linear correlation is observed; therefore, parameter B expressing stress transfer and interfacial adhesion can be determined with more accuracy. The slope is changed after filler treated with SDS or MPS. The parameter B of treated PP/CPH composites with SDS or MPS were 2.44 and 2.11, respectively, which is higher than untreated PP/CPH composites (1.55). This confirms the interfacial adhesion was enhanced by filler treatment with SDS or MPS. Meanwhile, the parameter B of treated PP/CPH composites with SDS was larger than treated PP/CPH composites with MPS. The results indicated that the treated CPH with SDS was better in enhancing interfacial adhesion and improved stress transfer. The efficiency between SDS and MPS might be related to the molecular orientation on the filler surface. The SDS molecules are possibly perpendicular attached on filler surface, which result in better wetting of matrix to filler surface as shown in Figure 7(a). From literature, it is found that the coupling agent like stearic acid would form a perpendicular oriented structure on filler surface as illustrated in Figure 7(a). However, the molecules of silane coupling agent might resulting in flat or bridge-like structure rather than perpendicular on filler surface as illustrated in Figure 7(b)- and (c). 30 This probably influences the wettability between filler and matrix. Furthermore, the better wettability yields a stronger interfacial bonding between filler and matrix. As a result, the treated CPH with SDS shows a better adhesion with PP matrix compared to the treated CPH with MPS. Demjen and Pukánszky 31 also reported that the interfacial bonding was enhanced when the molecules of coupling agent form a perpendicularly orientation on the filler surface which improves the tensile strength and tensile modulus.
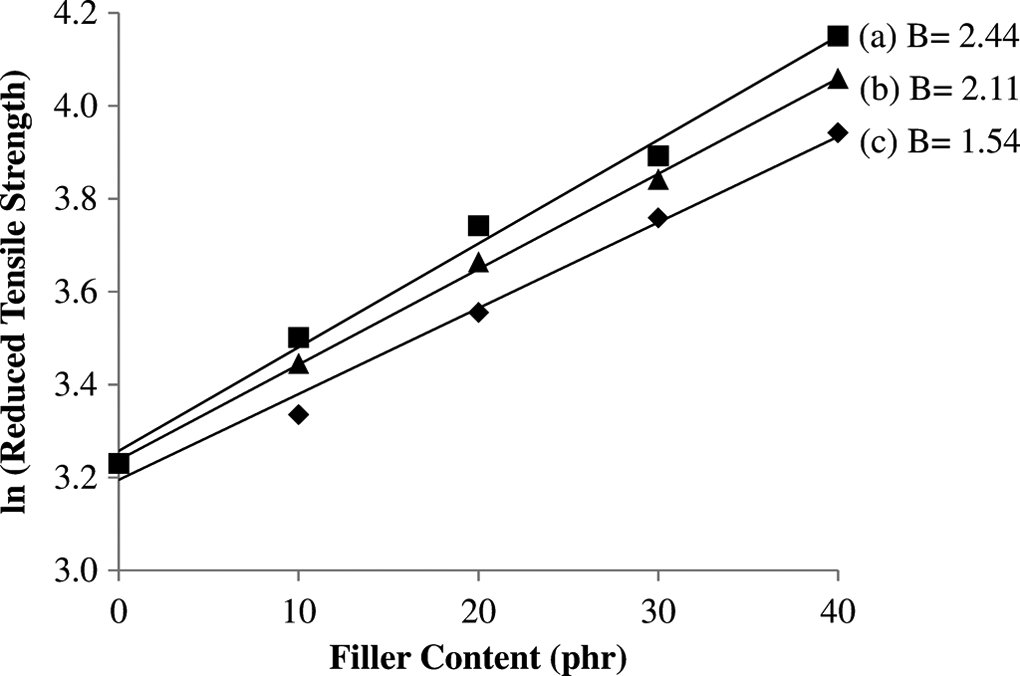
Reduced tensile strength of untreated and treated PP/CPH composites plotted against filler content in the linear form of equation (4). Symbols: (a) untreated, (b) treated with MPS, and (c) treated with SDS.
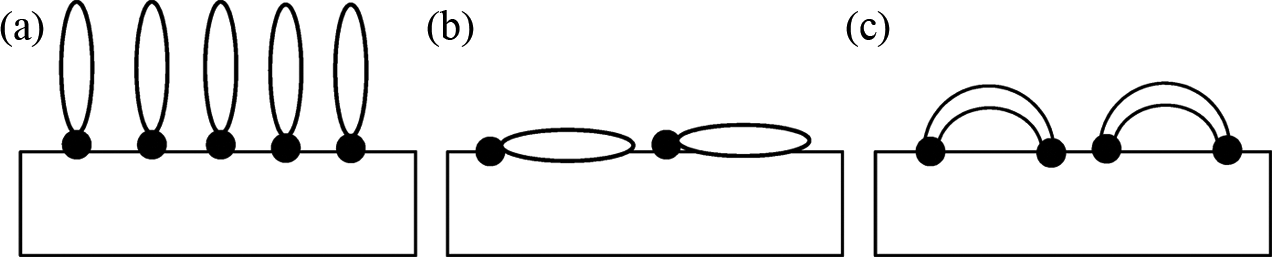
Different idealized molecular orientation of coupling agent on filler surface: (a) perpendicular oriented, (b) flat laying, and (c) bridge-like structure. 30
Morphology study
Figure 8(a) and (b) show SEM micrograph of tensile-fractured surface of untreated PP/CPH composites with 20 and 40 wt% of CPH content. The SEM micrograph show that the untreated CPH had poor dispersion and easily to form agglomeration, especially at 40 phr of filler content. The presence of holes due to filler pull out and detached CPH particles can be observed. This indicated the weak interfacial bonding between untreated CPH and PP matrix. In contrary, SEM micrographs of treated PP/CPH composites with MPS or SDS illustrated a brittle fracture surface as shown in Figures 9(a) and (b) and 10(a) and (b). CPH treated with both MPS and SDS was embedded and well covered by PP matrix. This evidenced the treated CPH had better adhesion with PP matrix.

SEM micrograph of untreated PP/CPH composites at (a) 20 phr and (b) 40 phr of filler content.

SEM micrograph of treated PP/CPH composites with MPS at (a) 20 phr and (b) 40 phr of filler content.

SEM micrograph of treated PP/CPH composites with SDS at (a) 20 phr and (b) 40 phr of filler content.
Water absorption
The water absorption of untreated and treated PP/CPH composites with MPS or SDS is illustrated in Figure 11. The water absorption of all composites increased with the increasing of the filler content. Clearly, the untreated PP/CPH composites exhibited higher water absorption than treated PP/CPH composites with MPS and SDS. The increment of water absorption of composites is correlated to the ability of hydrophilic CPH to form hydrogen bonding toward water molecules. The water absorption of composites is higher at more filler content. In contrast, the water absorption of composites is reduced with the presence of MPS and SDS. This reason is that the CPH treated with MPS or SDS has hydrophobic aliphatic chains attached on filler surface, which decreases the hydrophilicity of CPH. As a result, the tendency of treated CPH to bond with water molecules was lower. Besides, the treated PP/CPH composites with SDS had lower water absorption than treated PP/CPH composites with MPS. The treated CPH with SDS probably had more efficiency in preventing diffusion of water molecules into CPH due to the presence of longer hydrocarbon chains.
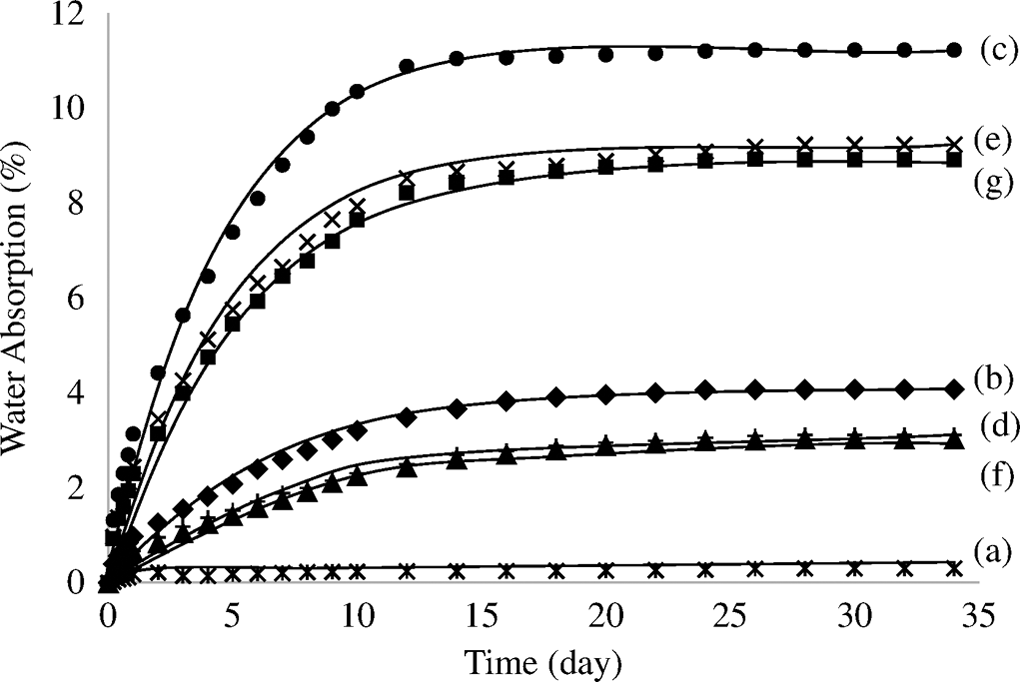
Water absorption of neat PP (a); untreated PP/CPH at 20 phr (b) and 40 phr (c) filler content; treated PP/CPH with MPS at 20 phr (d) and 40 phr (e) filler content; treated PP/CPH SDS at 20 phr (f) and 40 phr (g) filler content.
FTIR spectroscopy analysis
Figure 12 shows the FTIR spectra of untreated and treated CPH. The broad peak at 3291 cm−1 was assigned to the hydroxyl groups (–OH) of CPH. The peak at 2927 cm−1 related to C–H stretching and a small peak at 1732 cm–1 was attributed to C=O stretching of carboxyl groups from hemicellulose. The peak of C=C stretching from hemicellulose and –OH group from embed moisture were detected at 1604 cm–1. The small peak presence at 1518 cm–1 was indicated as conjugated C–O group from aromatic skeletal in lignin. The peak at 1434 cm–1 was corresponding to CH2 deformation vibration of cellulose. Moreover, the peak at 1372 cm–1 was referring to C–H group deformation in cellulose and hemicellulose. A peak found at 1247 cm–1 was assigned as C–O groups from acetyl group in lignin. Another broad peak detected at 1038 cm–1 was C–H group vibration in cellulose. There are small peaks in the 700–900 cm−1 range related to C–H vibration of lignin. The treated CPH with MPS or SDS show less hydrophilic properties then untreated CPH as the peak’s intensity at 3291 and 1605 cm−1 were reduced. The peak intensity at 1736 cm−1 increased after CPH treated with MPS. This is due to the presence of covalent linkage between MPS and CPH. The peak’s intensity at 775 cm−1 significantly increased due to presence of Si–C group from MPS on CPH. Alternatively, the other characteristic peaks of MPS could not be found in modified CPH. This might be due to the overlapping of characteristic peaks between MPS and CPH. Figure 13 shows the schematic diagram of chemical reaction between MPS and CPH. The reaction steps of MPS involved are as follows: (i) MPS undergoes hydrolysis to become silanols and some will be self-condensation to form silanol oligomer; (ii) silanol oligomer is physically absorbed to hydroxyl group of CPH; (iii) condensation of silanol oligomer and form Si–O–C bond between MPS and CPH. Besides, the treated CPH with SDS also shows an increasing of peak intensity at 1737 cm−1 that attributed to the formation of ester linkage between SDS and CPH. The peak intensity at 2923 and 2853 cm−1 increased due to the presence of long alkyl chains from SDS attached on CPH surface via covalent bonding. The schematic reaction between SDS and CPH was shown in Figure 14.
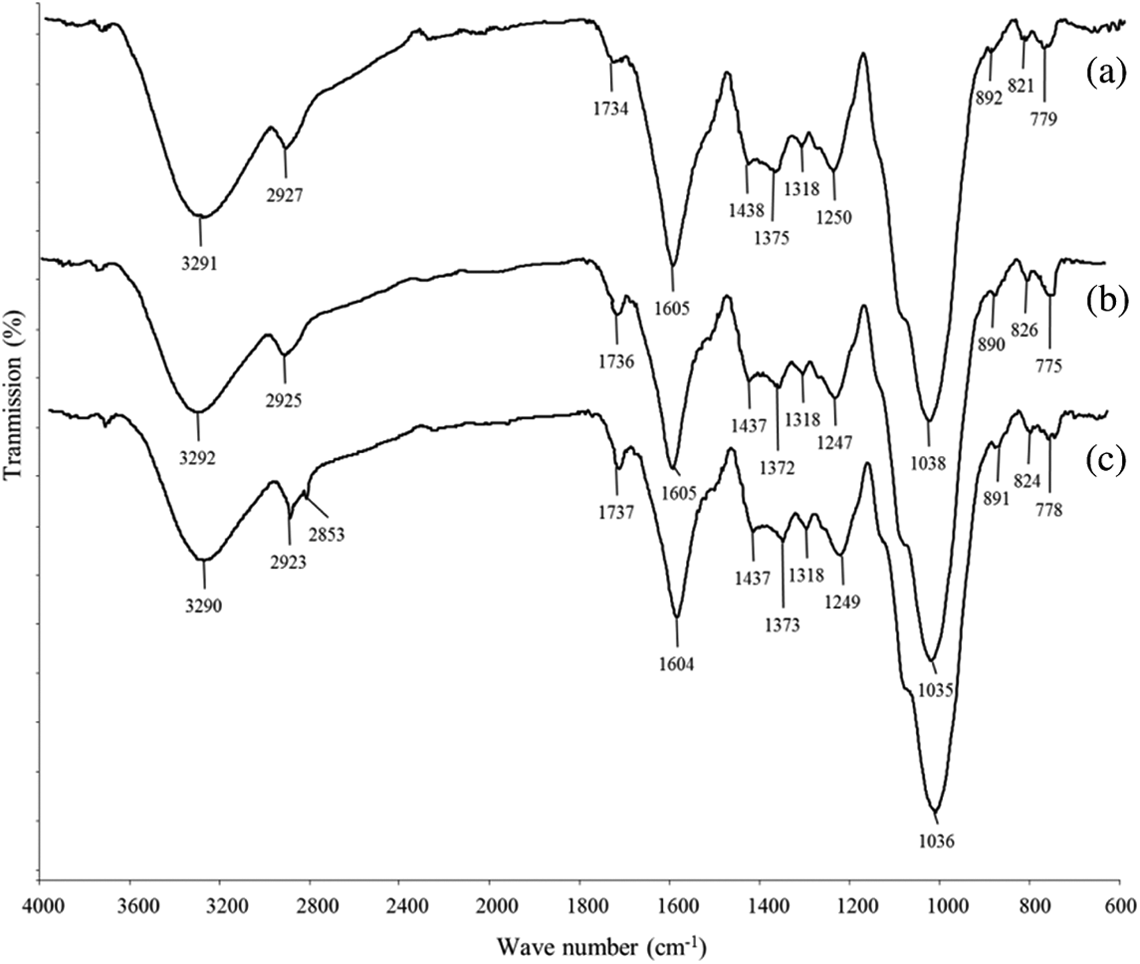
FTIR spectra of (a) neat CPH and treated CPH with (b) MPS and (c) SDS.
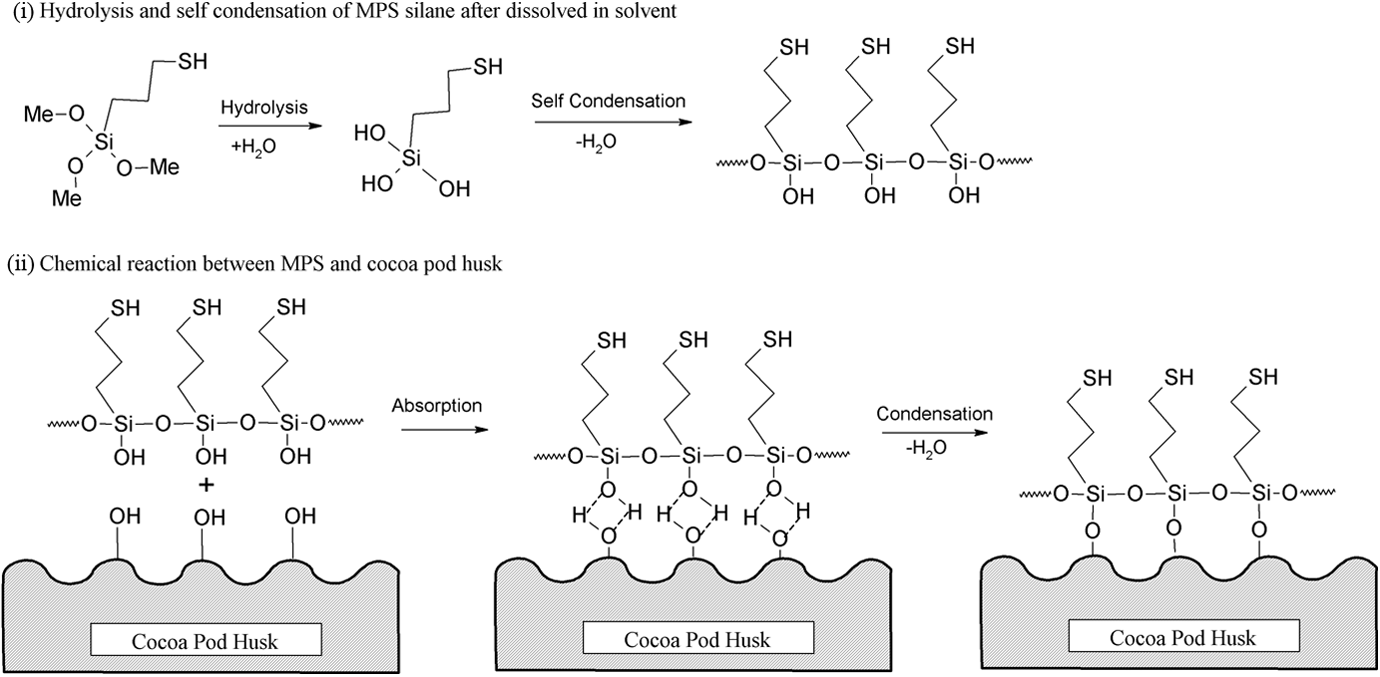
Schematic reaction between MPS and CPH.
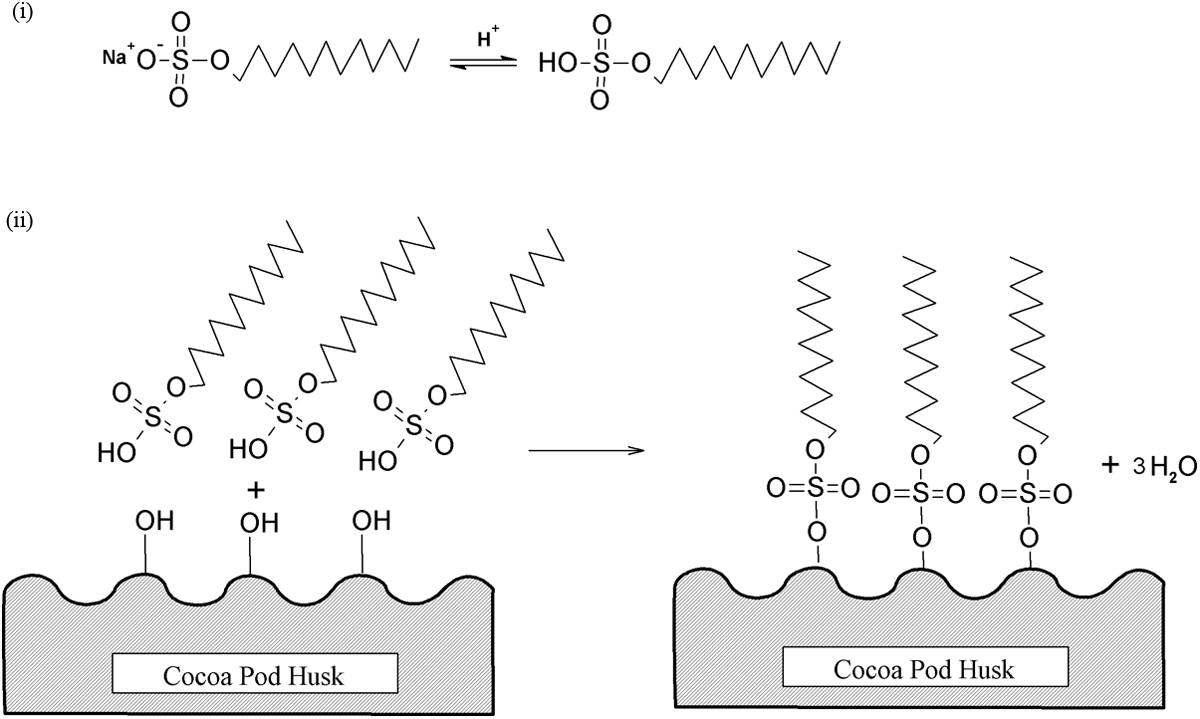
Schematic reaction between SDS and CPH.
Differential scanning calorimetry
The DSC curves of neat PP and untreated and treated PP/CPH composites are shown in Figure 15. The DSC data of neat PP and all composites are listed in Table 2. The melting temperature (T m) of neat PP can be observed at 165 C and the crystallinity of neat PP is 27%. According to Table 2, the increase in CPH content increased the crystallinity of PP/CPH composites and PP matrix as well. The presence of CPH provided a site for nucleation as it promotes the migration and diffusion of PP chain nucleation sites for initiation of spherulites growth. Furthermore, the treated PP/CPH composites with MPS or SDS show a higher crystallinity than untreated PP/CPH composites. This result indicated treated CPH with MPS or SDS that might had better adhesion with PP matrix and resulting a strong nucleating effect to composites. The addition of natural filler gives nucleating effect to composites and the nucleating effect is increased with the presence of silane coupling agent or fatty acid were reported by other researchers. 5,12,15,20 The T m of PP/CPH composites was not influenced by changes of CPH content and SDS or MPS modification. Some literatures also reported that the increase of filler content or filler modification did not tend to change the T m of composites. 6,12,22,32 Generally, the T m of the semicrystalline polymer is increased with the increasing of spherulites dimensions. 33 The presence of particulate filler usually causes a heterogeneous nucleating effect to semicrystalline polymer. 34,35 The number of smaller spherulites are probably growth due to the increase of filler content or filler modification, which increased the crystallinity of composites, but it does not affect the T m. 36,37
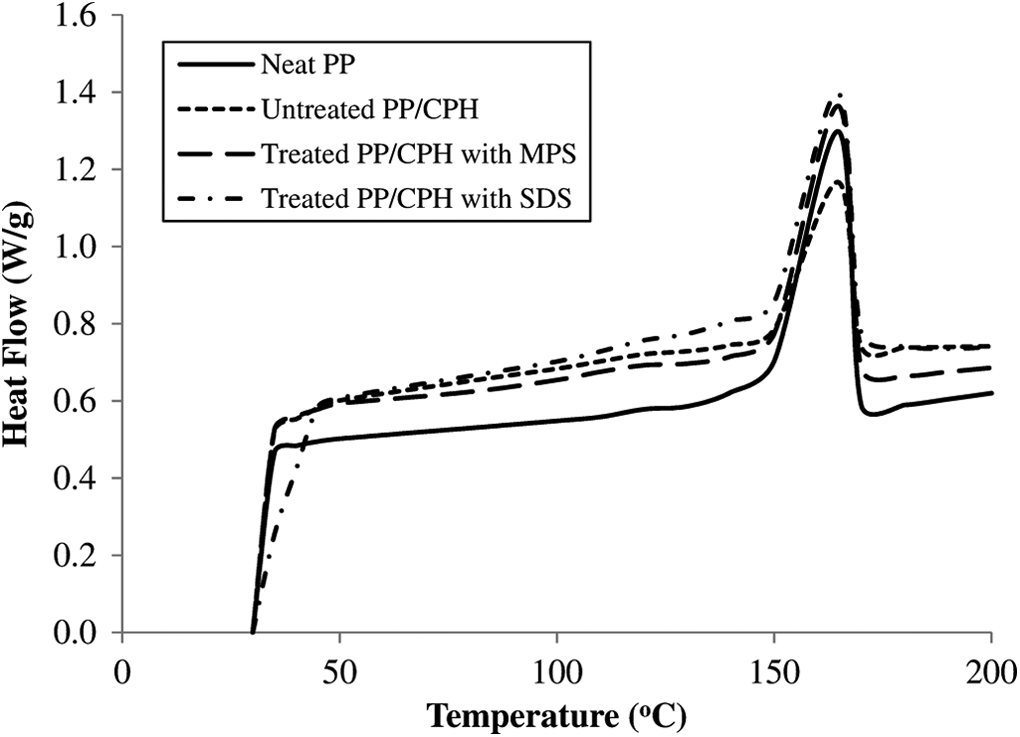
DSC curves of neat PP, untreated and treated PP/CPH with composites MPS and SDS. DSC: differential scanning calorimetry; PP: polypropylene; CPH: cocoa pod husk; SDS: sodium dodecyl sulfate; MPS: 3-mercaptopropyltrimethoxysilane.
DSC data of neat PP, untreated and treated PP/CPH composites with MPS and SDS.
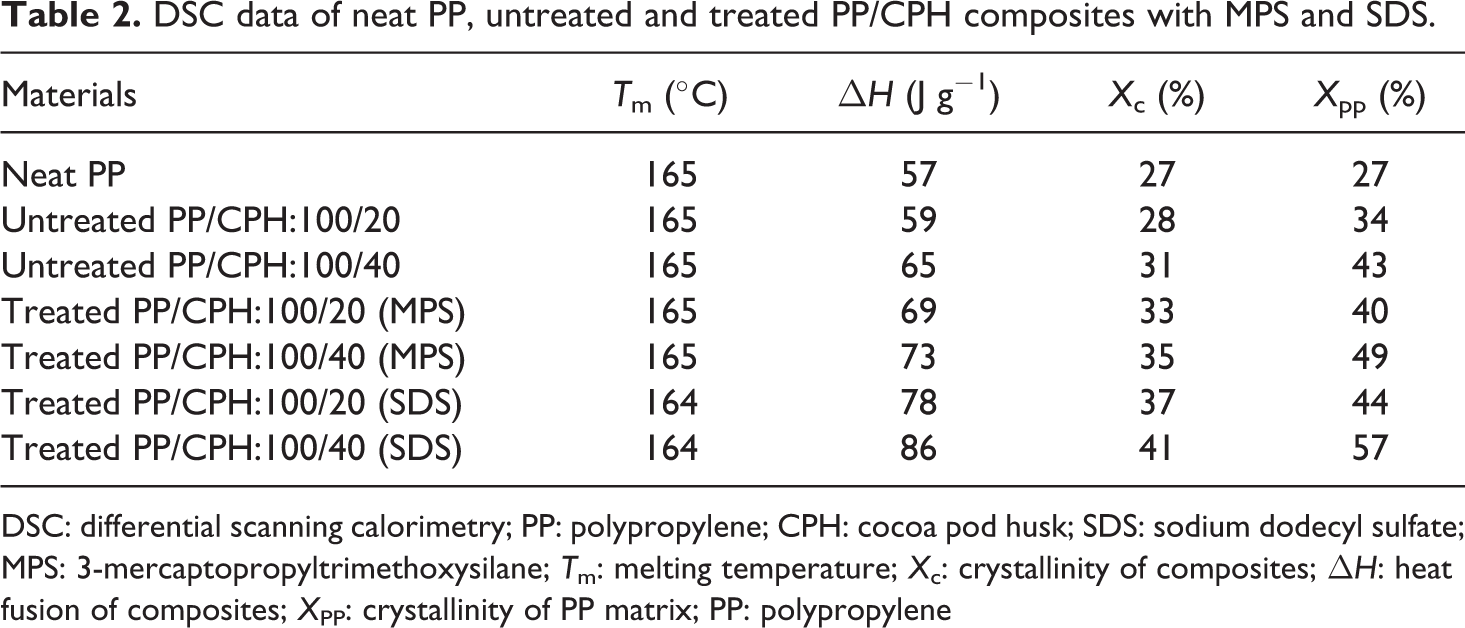
DSC: differential scanning calorimetry; PP: polypropylene; CPH: cocoa pod husk; SDS: sodium dodecyl sulfate; MPS: 3-mercaptopropyltrimethoxysilane; T m: melting temperature; X c: crystallinity of composites; ▵H: heat fusion of composites; X PP: crystallinity of PP matrix; PP: polypropylene
Thermogravimetric analysis
The derivative thermogravimetric (DTG) and TGA thermograms of neat PP, CPH and untreated and treated PP/CPH composites are illustrated in Figures 16 and 17, respectively. The DTG and TGA data of samples are summarized in Table 3. As shown in Figure 16, the CPH exhibited three decomposition steps. First, the dehydration of moisture and other volatile compound in CPH at temperature about 60 C. Then, the weight loss is attributed to the decomposition of hemicellulose at temperature about 250 C, followed by further decomposition of lignin and cellulose at temperature above 350 C. The increased CPH content reduced the thermal stability of PP/CPH composites compared to neat PP as evidenced from the temperature at 5% weight loss (T d5%). This indicated that the PP/CPH composites had undergone an early thermal decomposition and the weight loss at T d5% was attributed to the loss of moisture, volatile material, and hemicellulose in the CPH. However, the temperature at maximum weight loss (T dmax) of the PP/CPH composites shifted to higher temperature and the residue content increased as CPH content increased. This is because the addition of more CPH increased the thermal stability of PP/CPH composites to higher decomposition temperature. This phenomenon occurred due to the formation of char residue from thermal decomposition of hemicellulose, which acts as thermal protecting layer on the composites and delaying the process of thermal decomposition. 38,39 Araujo et al. 40 also reported that increasing the curaua fiber content raised the formation of char residue and inhibited the thermal decomposition of the biocomposites. The thermal stability of treated PP/CPH composites with SDS or MPS was higher than untreated PP/CPH composites, as can be seen from the increase in T d5%, T dmax, and residue content. This was because the filler modification with MPS and SDS improved the filler dispersion and filler–matrix adhesion that further enhanced the thermal stability of composites against thermal decomposition. Arbelaiz et al. 41 found that the PP/flax fiber composites with silane modification had improved the thermal stability of the composites. Arrakhiz et al. 42 reported the PP composites with alfa fiber treated with palmitic acid show a better thermal stability.
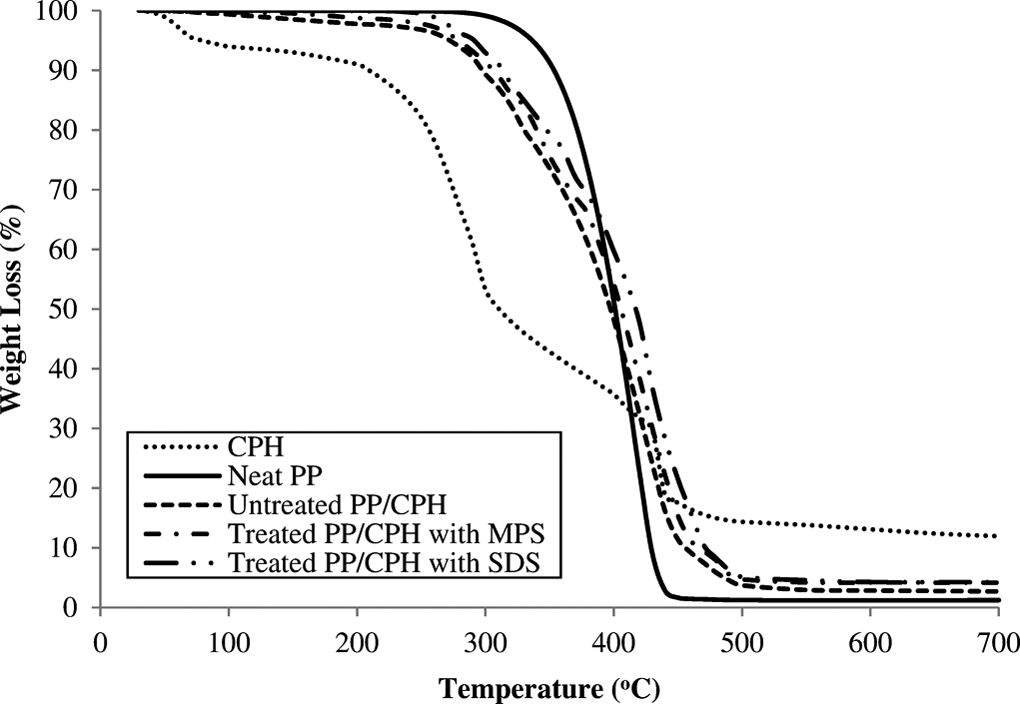
DTG curves of CPH, neat PP, untreated and treated PP/CPH composites with MPS and SDS.
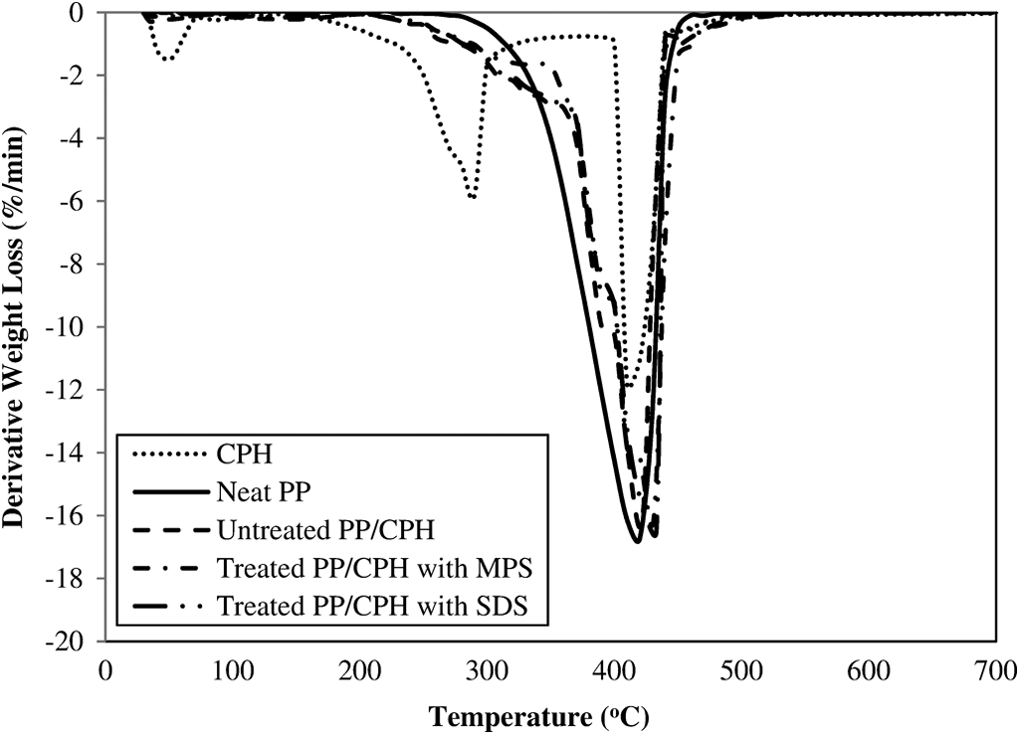
TGA curves of CPH, neat PP, untreated and treated PP/CPH composites with MPS and SDS.
TGA data of unmodified and modified PP/CPH biocomposites.
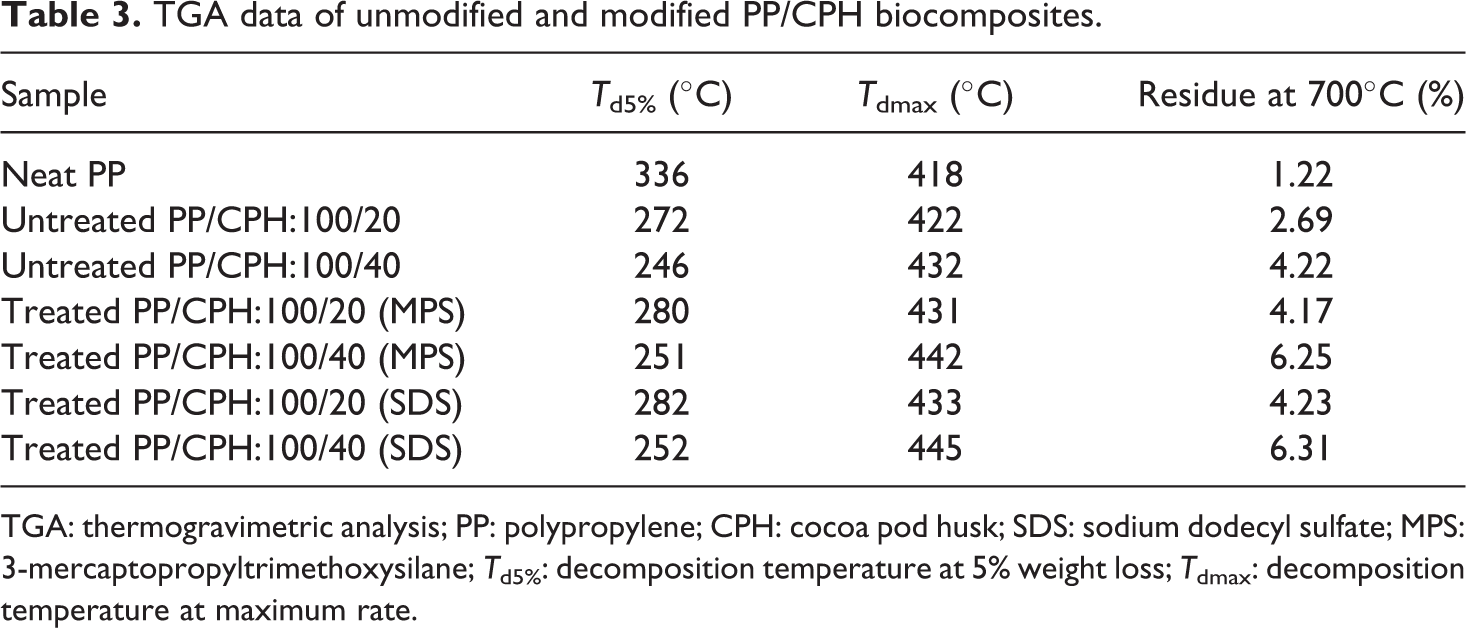
TGA: thermogravimetric analysis; PP: polypropylene; CPH: cocoa pod husk; SDS: sodium dodecyl sulfate; MPS: 3-mercaptopropyltrimethoxysilane; T d5%: decomposition temperature at 5% weight loss; T dmax: decomposition temperature at maximum rate.
Conclusions
The tensile strength and elongation at break of PP/CPH composites decreased with increasing CPH content. However, the processing torque, tensile modulus, water absorption, and crystallinity of PP/CPH increased with increasing CPH content. The incorporation of CPH causes early thermal decomposition to PP/CPH composites. The processing torque, tensile strength, tensile modulus, water resistivity, crystallinity, and thermal stability of PP/CPH composites increased with the presence of MPS or SDS. The properties of PP/CPH composites improved due to the enhanced filler–matrix adhesion by filler treatment with MPS or SDS. The SEM results evidenced that the CPH treated with MPS or SDS has better filler dispersion and interfacial adhesion with PP matrix. Overall, PP/CPH composites treated with SDS show better performance in enhancing tensile properties and water absorption than PP/CPH composites treated with MPS, but PP/CPH composites treated with both MPS and SDS show similar improvement in the thermal properties.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
