Abstract
Microporous layers of 0.3 µm α-alumina powder were deposited on the surface of porous zirconia substrates (average pore size of about 1 µm) using suspension–sedimentation method. The characteristics of the deposited alumina microporous layer was evaluated using different types of dispersants, namely, citric acid, sodium dodecyl sulfate and Triton X-100. The effect of these dispersants on the stability and settling characteristics of alumina suspension was evaluated by sedimentation tests. Scanning electron microscopy analysis of the microporous layers (prior to sintering) revealed that sodium dodecyl sulfate dispersant produced uniform layers with least agglomeration of alumina particles, whereas citric acid showed more agglomeration of alumina particles and the use of Triton X-100 developed holes on the surface of alumina layer. The layers deposited using sodium dodecyl sulfate dispersant were then sintered at 1400 °C in air. Scanning electron microscopy observations revealed that a uniform porous alumina layer with average pore size in the range of 0.5–1.0 µm was produced with this method. The results of sedimentation tests indicated that alumina powder suspension prepared with sodium dodecyl sulfate dispersant yielded very good stability with lowest particle settling rate, whereas citric acid exhibited the highest particle settling rate.
Keywords
Introduction
Ceramic materials such as alumina, zirconia, silicon carbide and titania have been widely used for fabrication of porous structures for filtration applications due to their good chemical resistance, bio-inertness and structural integrity at high temperatures. Porous ceramic structures are generally fabricated by powder consolidation and sintering. Powder compaction1,2 and colloidal processing3–6 are two important consolidation procedures. Powder compaction and sintering method is generally restricted to single-layer filters. For multiple-layer filters, successive layers are deposited on the base layer (or substrate) by colloidal processing. The base layer acts as a support that provides the mechanical strength/integrity. The deposited layers act as the active layers for filtration fineness. Different methods of colloidal processing of ceramic components include slip casting/dip coating, 3 tape casting 4 and solgel.5,6 These colloidal processes require preparation of suspension of ceramic particles in water with processing additives. It is important to have a well-dispersed suspension to prepare green bodies and layers with homogeneous and uniform structure with desired porosities. The properties of the green bodies and layers are crucial to achieve low and uniform shrinkage of the ceramic component during drying and sintering. If shrinkage is not uniform, distortion of the body can occur, which may lead to cracking of the component and delamination and/or cracking of layers during drying and sintering. Interparticle interactions and particle size are some of the important factors that control the suspension behavior of particles including suspension stability, settling rate and final consolidation. 7 The interaction between particles in suspension may be repulsive or attractive. Particles that have repulsive forces between them remain as individual particles in the liquid, whereas the presence of attractive forces between particles produces larger and more massive aggregates. Settling rate of particles depends upon the particle (or aggregate) size. When the particles are small, they have very little mass and will settle very slowly under the influence of gravity. Large particles lead to unstable suspension due to increased settling rates. Processing additives such as surfactants and dispersants are used to improve suspension stability by increasing the interparticle repulsive forces and surface tension. Citric acid has been used as dispersant for TiO2 particles in aqueous medium. It was observed that flocculation occurs at lower pH levels; 8 in contrast, the presence of citric acid was shown to have increased the stability of alumina suspension in the presence of dimethylformamide (DMF). 9 The stability of alumina suspension was high at lower pH due to increased adsorption of citric acid on the surface of alumina particles. A comparative study of citric acid–based and polyacrylic acid–based dispersant, duramax D-3005, was performed to analyze the effect on the stabilization of alumina suspension. 10 It was observed that duramax had higher stabilizing ability compared to citric acid. Addition of 0.5% wt/vol sodium dodecyl sulfate (SDS) solution to zirconia suspension in water was shown to reduce the agglomeration of zirconia particles. 11 In another study, the presence of 10% SDS was shown to produce stable aqueous suspension of ZrO2 nanoparticles at all pH values of suspension. 12 The nonionic surfactant Triton X-100 was found to prevent the agglomeration of particles during the synthesis of zinc phosphate nanoparticles. 13 It was also reported that Triton X-100 helped in depositing TiO2 film on glass substrate for fabrication of photoelectrode, due to its sterical repulsion characteristics that provided suspension stability. 14
Ceramic filters are generally made as stand-alone units with single layer. For applications where filtration of a wide range of particle sizes is needed, single-layer filters pose problems such as difficulty in maintaining good throughput. Filters with graded porosity should alleviate these problems. Filters with graded porosity can be fabricated by depositing multiple porous layers of graded porosity on the porous support. In this work, a simple but efficient method of suspension–sedimentation was utilized to deposit microporous α-alumina layer onto a porous zirconia substrate. Suspension stability plays an important role in the success of suspension–sedimentation method. Based on data published in literature, a preliminary study on the selection of dispersant, dispersant/suspension concentration was performed and a combination of these parameters that suited our objective was utilized and reported. Three different dispersants, namely, citric acid, SDS and Triton X-100, were utilized to prepare a stable alumina suspension. The authors would like to emphasize that this work is part of a broader study aimed at fabrication of multilayered ceramic filter with graded porosity that can filter particles from micro- to nano-range size.
Materials and methods
Materials
Commercial α-alumina (α-Al2O3) powder (Buehler Co.) with average particle size of 0.3 µm was used for the preparation of the suspension. Figure 1 shows scanning electron microscope (SEM) microphotograph of as-received α-alumina powder. 15 Citric acid (99.5% purity), SDS and Triton X-100 were used as dispersants to study their effect on the stabilization and layer deposition characteristics of alumina powder suspension.

As-received α-alumina powder.
Preparation of suspension
For the preparation of the suspensions, a measured amount of α-alumina powder was slowly added to deionized water to make 1 wt% alumina suspension while stirring on magnetic stirring plate. The suspension was stirred for 30 min and sonicated for another 30 min using a probe ultrasonicator. The dispersants were then added to the previous suspension and further stirred for 30 min before starting the sedimentation tests. Table 1 shows the concentration of the used dispersants in 1 wt% alumina–water suspension. Zeta potential (ζ-potential) and pH of these suspensions were measured at room temperature. The zeta-potential measurements were performed using ZetaPALS zeta-potential analyzer (from Brookhaven), which uses Phase Analysis Light Scattering technique. Zeta potential was evaluated using Smoluchowski equation for electrophoretic mobility of charged, colloidal suspensions. Electrophoretic mobility is defined as the velocity of a particle in an electric field. The average pH and zeta-potential values of these suspensions are presented in Table 2.
Concentration of different dispersants in 1 wt% alumina–water suspension.

SDS: sodium dodecyl sulfate.
Average pH and zeta-potential values of different suspensions.
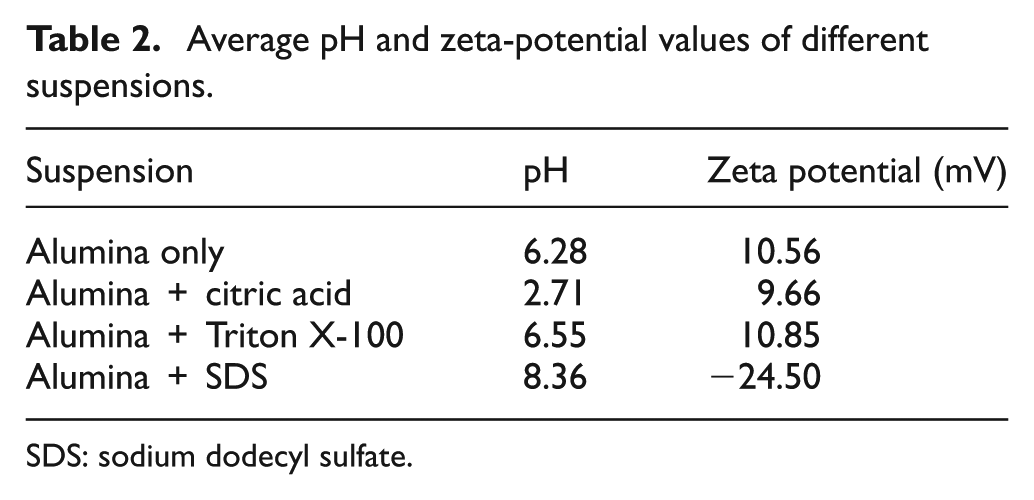
SDS: sodium dodecyl sulfate.
Sedimentation tests
For sedimentation tests, the suspension was slowly poured into 50-mL graduated vertical glass cylinders. The cylinders were covered with stoppers to avoid the effect of atmosphere. For settling rate calculations, ratio of column height in suspension (H) to the original suspension height (Ho) was recorded at specific intervals up to 90 min.
Deposition of microporous layers
Porous zirconia cylindrical substrates of 25 mm diameter and 4 mm thickness were first polished with 600 grit SiC grinding paper and then washed, ultrasonicated and dried to remove contamination and moisture. These substrates were placed in plastic containers. The prepared suspension, mentioned above, was poured over the substrates. The thickness of the deposited layers was controlled by varying the height of suspension above the substrate. The suspension was allowed to settle, and the clear suspension was gently sucked off using vacuum suction tube. The samples were then first dried in air for 12 h and then in oven at 120 °C for 2 h to remove the remaining moisture from the deposited layer.
Sintering of microporous layers
Based on the SEM analysis of layers deposited with the three dispersants, only layers deposited with SDS dispersant were sintered in a box furnace at 1400 °C for 2 h in air. Heating was kept constant at 4 °C/min, and cooling was done by switching off the furnace.
Results and discussion
Sedimentation behavior
Sedimentation behavior of the different suspensions was studied. The settling rate for the first 90 min of sedimentation is shown in Figure 2. It can be observed that suspension with citric acid was unstable and underwent quick sedimentation with fastest settling rate among the selected dispersants. It was reported that citric acid as a dispersant for alumina suspension is not suitable at pH > 4; the suspension would have poor dispersion and could result in flocculation. 9 The suspension with citric acid as a dispersant had a pH of 2.71 and zeta potential of 10.26 mV for the concentration used in this study. The pH is well below the limiting pH value of 4, as noted by Lina Wu et al. 9 but the ζ-potential was observed to be in similar range of alumina-only suspension, and hence, no improvement in suspension stability was observed. Addition of Triton X showed better stability than citric acid, although the ζ-potential value was in similar range. Triton X is a nonionic surfactant with large molecular size and long chain that produces steric hindrance between alumina particles thus improving the dispersability. 14 Conversely, it was observed that suspension with SDS as dispersant had the highest suspension stability even after 12 h. The ζ-potential of alumina suspension with SDS dispersant was measured to be −24.50 mV, which is much higher than that of alumina-only suspension. This high suspension stability can be attributed to the increase in absolute value of ζ-potential of the suspension with SDS, since SDS interacts with ceramic particles both electrostatically (through its sulfate group) and hydrophobically (through its alkane group) as observed in previous studies on the effect of SDS on the stability of ZrO2 12 and CaCO3 suspension 16 in water. The improvement in suspension stability of nano-alumina suspension with the increase in absolute ζ-potential values due to SDS was explained in a recent study. 17 It was reported that SDS can partially ionize in water and give anionic species, while alumina particles produce positive charges in water and have a strong attraction for anionic groups. At lower concentration of SDS, the anionic species dissociate from SDS and gets adsorbed on the positively charged alumina surface, which becomes negatively charged; thus, the effect of electrostatic stabilization of the suspension is obtained. With further increase of SDS concentration, the anion groups further add into the adsorbed layer making the absolute value of zeta potential increase, resulting in the increasing repulsive forces between the particles, thereby increasing the suspension stability. At the same time, large numbers of anion groups in the aqueous suspension hinder the particles from colliding and then reduce the collision probability between the particles. This leads to the suspensions showing significantly reduced agglomeration and enhanced mobility, ultimately resulting in more stable dispersion system. 17 The study, however, was done with lower concentrations of nano-alumina of 0.01 wt%, whereas a higher concentration of 1 wt% micro-alumina was utilized in this study. The selection of higher concentration of alumina was essential to deposit microporous layers with required thickness in reasonable amount of time. Most of the previous studies involving the use of SDS as dispersant involved only stability investigations; however, our objective was to deposit microporous layers with required pore size and integrity.
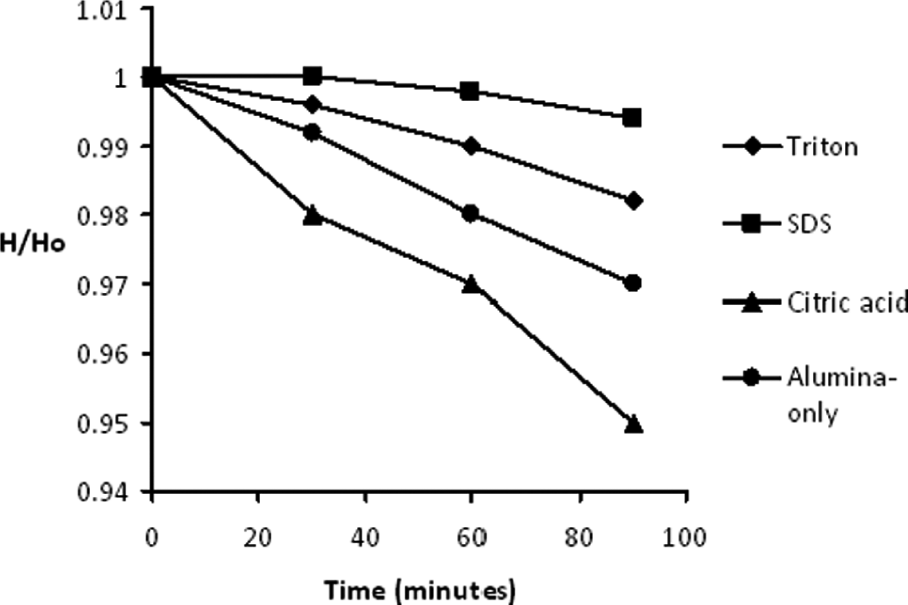
Settling rate of different dispersants in 1 wt% alumina–water suspension.
Deposited microporous layers
The SEM microphotographs of deposited layers (prior to sintering) with different dispersants are shown in Figures 3–6. It can be observed that the layer deposited with α-alumina suspension without any dispersant, Figure 3, was very poor exhibiting agglomerates of particles forming flakes. The layer deposited with citric acid, Figure 4, was also observed to be poor and showed agglomeration of particles. The layer deposited with Triton X, Figure 5, showed less agglomeration of particles but exhibited holes on the surface. On the other hand, the layers deposited with SDS dispersant, Figure 6, showed better surface characteristics with very few and smaller agglomerates compared to other suspensions. It is concluded that SDS is a good candidate for the preparation of α-alumina colloidal suspension.

Alumina layer deposited without dispersant (before sintering) at 500× and 5000× magnification.

Alumina layer deposited with citric acid (before sintering) at 500× and 5000× magnification.

Alumina layer deposited with Triton X-100 (before sintering) at 500× and 5000× magnification.

Alumina layer deposited with SDS (before sintering) at 500× and 5000× magnification.
Sintered microporous layer
Based on the above sedimentation results, only layer deposited with SDS dispersant was sintered at 1400 °C for 2 h under atmospheric condition. Figures 7 and 8 display SEM microphotographs of the sintered layer, cross section and top surface, respectively. It was observed that the sintered layer was free of cracks and exhibited good surface characteristics with good bonding between the alumina particles. The pore size as estimated from the SEM images was in the range 0.5–1.0 µm. It was noticed that the thickness of the deposited layer has a considerable effect on its integrity. Cracking of layers was observed during sintering for deposited alumina layers thicker than 100 µm as shown in Figure 9. However, thin layers (less than 100 µm) did not show any cracking after sintering.

Cross section of the deposited layer with SDS (sintered) on the zirconia support.

Surface structure of sintered alumina layer deposited with SDS dispersant.

Cracking of thick alumina layer after sintering.
Conclusion
Microporous α-alumina layer was successfully deposited on a porous zirconia substrate by simple and efficient suspension–sedimentation technique. It was observed that the use of a dispersant was critical in achieving a uniform deposition of alumina layer. SDS dispersant produced uniform microporous layer with minimal agglomeration of alumina particles as compared to citric acid and Triton X dispersants. The sintered alumina layer with SDS dispersant was found to be free of cracks as analyzed by SEM and exhibited good surface characteristics with good bonding between the alumina particles. The pore size of the sintered α-alumina microporous layer was in the range 0.5–1.0 µm. It is concluded that suspension–sedimentation and sintering technique is well suited to efficiently fabricate microporous alumina ceramic thin layers.
Footnotes
Funding
This study was supported by King Abdulaziz City for Science and Technology (KACST), Riyadh, Kingdom of Saudi Arabia (KSA), through the Science & Technology Unit at Deanship of Scientific Research, King Fahd University of Petroleum & Minerals (KFUPM), Dhahran, KSA, through Project # 08-ADV70-4 as part of the National Science, Technology and Innovation Plan.
