Abstract
Research on postprocessing of glass fiber–reinforced anionic polyamide-6 (APA-6) composites was performed in order to optimize the processing time. The two main points of investigation were the cure time and the annealing cycle. It was shown that the optimal mechanical properties (short-beam strength) coincided with the maximum degree of crystallinity for a cure time of 90 min at a cure temperature of 180°C. Annealing close to the crystallization temperature of APA-6 showed the largest increase in degree of crystallinity. For enhanced mechanical properties, however, the processing history is of importance. It was seen that for short cure times, sufficient annealing time at elevated temperatures is beneficial for the polymer chains to branch, cross-link, or adhere to the fibers, hence, significantly improving the mechanical properties.
Keywords
Introduction
A thermoplastic resin system was investigated for the production of large, fiber-reinforced structures, such as wind turbine blades, by means of a vacuum infusion process. Thermoplastic composites exhibit several advantages over their thermoset counterparts: lower processing times, easy assembly of parts through welding, recyclability, and high toughness. However, as a result of high melt viscosity, high processing temperatures and pressures are needed for good impregnation and consolidation of the composites. Moreover, vacuum infusion is only viable with resin viscosities below 1 Pa.s. To overcome the high viscosity of thermoplastic resins and make them suitable for liquid moulding, reactive resin systems are investigated. In addition, such systems generally show significantly reduced processing temperatures (as compared to the corresponding melt processable systems) as a result of both processing from the monomer and the simultaneous occurrence of polymerization and crystallization. 1
A wide interest in liquid moulding of thermoplastic resins, such as polybutylene terephthalate (PBT), 2 – 5 polyamide 12 (PA-12), 6 – 9 and polyamide-6 (PA-6) 10 – 15 has been shown recently. These resin systems exhibit much lower processing times than the thermoset resins currently used for the production of wind turbine blades, like polyester, vinylester, and epoxies.16,17 For example, reactive PA-12 has a cure time of 1 h 9 and PA-6 12 and PBT3,5 of 30 min, whereas wind turbine blade grade polyester requires a cure cycle of 4–6 h with a postcure of 2 h. 18 Based on the processing time and various factors like availability, costs, and suitability for the vacuum infusion process, reactive or anionic PA-6 (APA-6) is considered one of the most suitable candidates for the manufacturing of wind turbine blades. 19
Extensive research has already been performed on melt processing of pure hydrolytic PA-6 resin, 20 – 29 showing an important effect of the annealing cycle on its physical and mechanical properties. It is known that after consolidation, PA-6 can undergo additional crystallization when held at an annealing temperature between its glass and melting temperature and, thus, improve its mechanical properties. 30 Normally, annealing is done at or above the crystallization temperature, leading to increased crystallinity and stiffness and reduced toughness. If a reduction in toughness is unwanted, a lower annealing temperature can be used, reducing the heat energy requirements at the same time.
These three temperatures, that is the glass transition temperature Tg (≈60–75°C), the cold crystallisation temperature Tcc (≈160–175°C), and the melting temperature Tm (≈210–220°C), are also of importance for annealing reactively processed APA-6. 29 However, due to the fact that crystallization is still in progress after the finalization of the polymerization stage, the cure cycle might also influence the final degree of crystallinity. Research on the effect of the cure time and the annealing cycle on the APA-6 system is mostly focused on the pure resin,12,31 while very little information is yet available on fiber-reinforced APA-6 composites.
Based on literature, the effect of annealing fiber-reinforced APA-6 for a period of time is expected to reorganize the crystals and remove imperfections in the primary crystal structure,23,32 to improve the crystallinity by inducing favorable crystal type transition (from γ-crystals to α-crystals), 33 to change the lamellar size and spherulite diameter of the crystals 31 and the amount of intercrystalline links, 34 to reduce the amount of tie-molecules upon lamellar thickening,35,36 and to decrease the amorphous phase mobility after annealing. 32
In this paper, a study is performed on the effect of cure and annealing on the physical and mechanical properties of glass fiber reinforced APA-6 composites, while optimizing the processing time. The properties that are investigated are the degree of conversion, the degree of crystallinity, the melting temperature, the density, the void content, and the short-beam strength. The research also focuses on identifying the most important postprocessing parameters and understanding the mechanisms related to the observed improvements. Different annealing temperatures and times are investigated as well as the effect of the cooling rate after annealing. The impact of the initial degree of crystallinity of the laminate prior to annealing is also analyzed through changes in the cure time and the cure temperature. It is expected that a shorter cure time in combination with an annealing cycle will result in similar properties as for the optimal cure time. Hence, it is thought that the mould time of the laminate can be reduced and the energy consumption during (post)processing can be decreased without compromising on the mechanical properties of the composite parts.
Experimentation
Materials
For the vacuum infusion of the APA-6 thermoplastic laminates, a reactive resin mixture was used, which consisted of three components: the anionic polymerisation grade ε-caprolactam monomer, activator C, and the initiator M.
E-glass fiber mats were used to reinforce the APA-6 composites. The 8-harness satin weave E-glass fabrics were supplied by Ten Cate Advanced Composites bv., Nijverdal, The Netherlands.
Processing
A specially designed mini-mixing unit was used to prepare the resin mixture for infusion under a nitrogen atmosphere and at a temperature of 110°C. After dispensing the volume needed for the infusion of a laminate into a buffer vessel, the resin was degassed for 5 min to remove N2 dissolved in the mixture during storage in the MMU tanks.
A balanced, symmetric 12-ply laminate (25 × 29 cm2) was prepared for vacuum infusion, after which it was placed between two infrared panels and heated to 180°C prior to infusion. After degassing, the resin was allowed to flow through the inlet line-injection into the laminate. The resin injection and fiber compaction were achieved under vacuum. The vacuum was maintained during the cure cycle.
Heat treatments
Four cure times were assessed: 30, 60, 90, or 120 min, at a cure temperature of 180°C. The laminates were then removed from underneath the infrared panels and naturally cooled to room temperature.
Test matrix for the annealing study with the different investigated cure and annealing parameters

Cooling rates within laminate after annealing for different annealing temperatures a

The cooling rates used are either naturally cooled in air (NC) or cooled inside the oven (SC).
It is known that low cure temperatures yield a higher initial degree of crystallinity. 12 To investigate the effect of annealing for low-temperature cured composites, laminates were cured at 160°C and 170°C for 30 min. All the samples for the short-beam strength were taken from the same panel. They were then subjected to an annealing cycle as shown in Table 1.
Test matrix for isothermal crystallisation measurements by means of differential scanning calorimetry (DSC) and sample nomenclature
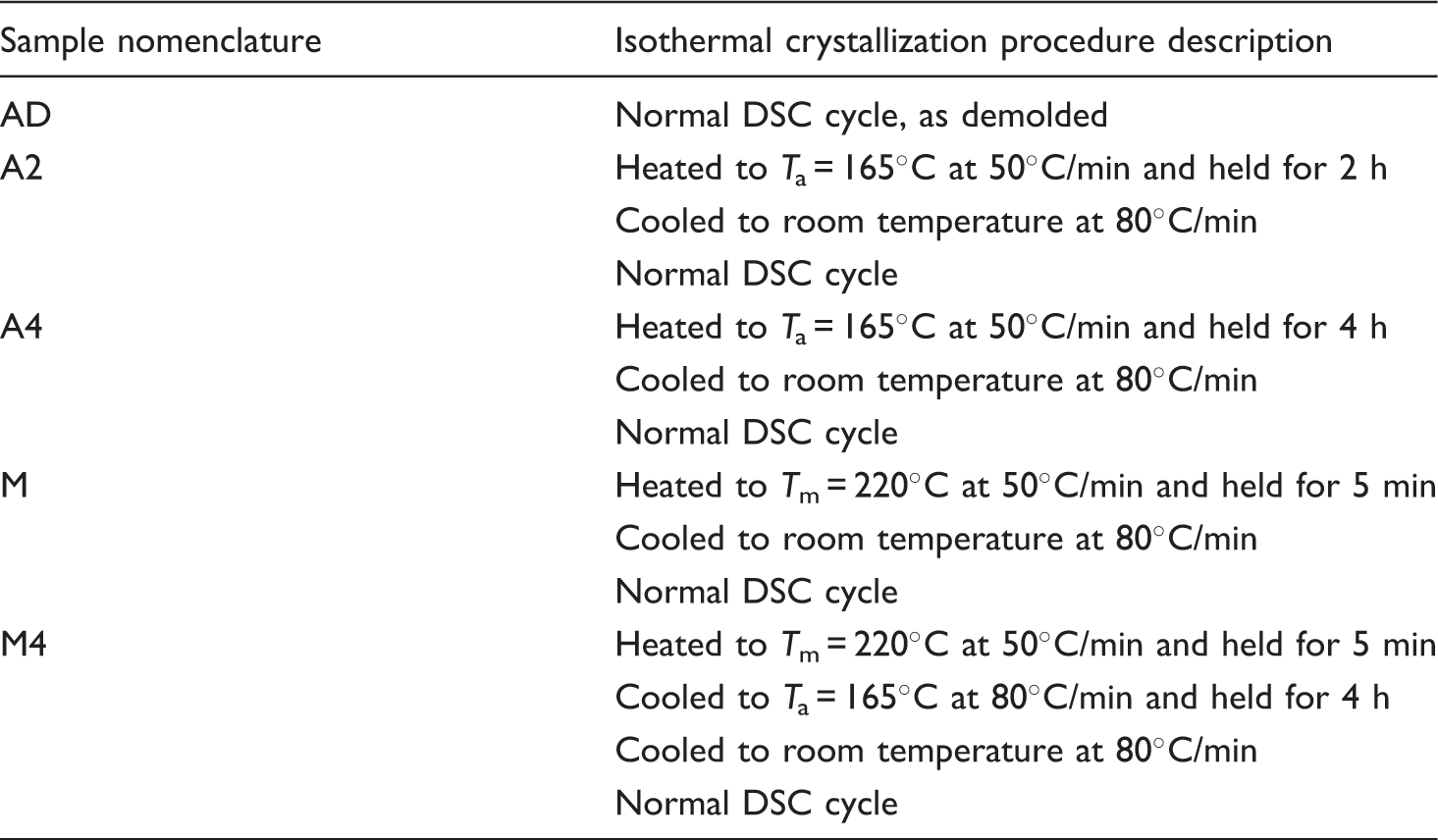
Analysis methods
Degree of conversion
To determine the degree of conversion (DOC) of the polymer, samples of the composite plate were taken near the resin outlet. After drying the samples at 50°C in a vacuum oven for at least 48 h, they were weighed (mtot) and refluxed overnight in demineralized water.
39
After this process, the residue was dried and weighed again. As the monomer dissolved in the water, the mass loss could be attributed to the monomer (mmon). Additionally, the weight of the fibers (mfib) was determined by burning off these residues in a carbolite oven at 565°C for 1h; according to ASTM D 2584–02. The degree of conversion could then be calculated as follows:
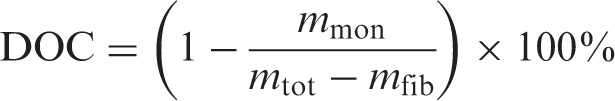
It should be taken into account that low-molecular oligomers susceptible to be formed during polymerization also dissolve during the reflux process. 40 As these oligomers, as well as the monomer, do not have any load-carrying capabilities, Equation (1) quantifies the amount of nonload-carrying substances in the composite but does not provide the exact degree of conversion.
Degree of crystallinity
The degree of crystallinity (Xc) and the melting temperature (Tm) of the polymer were determined by means of a Perkin Elmer differential scanning calorimeter (DSC). Disc-shaped samples of approximately 5 mg (msp) were taken from the laminate near the resin outlet. They were dried at 50°C in a vacuum oven for at least 48 h prior to testing. In the DSC, the test sample was held at 25°C for 2 min and then heated up to 250°C at a rate of 10°C per min. The results had to be corrected in account of the fiber content of the sample (mfib), which was determined by burning off the resin after the DSC test. The degree of crystallinity was calculated as follows:
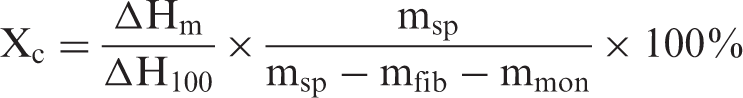
In the study on annealing, crystallinity samples were taken before and after annealing for every laminate. Since there is some variation in the initial values of the crystallinity for the different laminates, the mean change in crystallinity compared to its initial state (%) is being studied.
Short-beam strength
The short-beam strength tests were conducted on a Zwick Roell 20 kN testing machine according to the ASTM D2344 standard. The test specimens for the short-beam strength test were rectangular shaped and with the following nominal dimensions: 16.2 × 5.4 × 2.7 mm3. From the maximum force (Pmax), the interlaminar shear strength (ILSS) was calculated as follows:
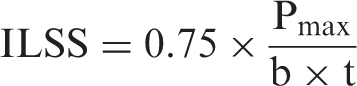
Constituent content
ASTM Standard D792-00, Test Method A was used to calculate the practical density (ρp) of the composite. The samples (two per plate) for the determination of the density were taken close to the outlet where the samples for DOC and Xc were taken. The test specimens were dried at 50°C in a vacuum oven for at least 72 h prior to testing. Both specimens were tested three times for accurate results.
To determine the fiber volume percentage, ASTM Standard D3171-99, test method I was used. The specimens used for the density tests were put in a crucible, burned off in a carbolite oven at 565°C for 1 h. The residue was cooled in a desiccator and weighed. From the ignition loss of the specimen, the fiber weight percentage (FWF) could be calculated:
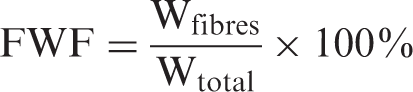
The void content (Vv) can be calculated according to ASTM Standard D2734-92:
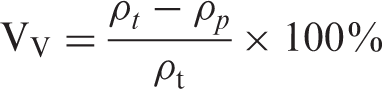
In which ρp and ρt are the practical and theoretical densities, respectively. The theoretical density (ρt) is calculated as follows:
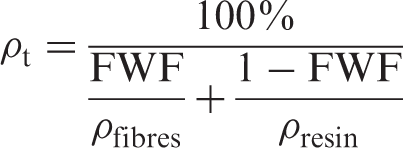
Results and discussion
Cure time
For the effect of cure time on the degree of crystallinity, Figure 1 shows a linear increase to a peak value of 37% at 90 min after which it levels off to a constant crystallinity at 120 min. It can also be seen that the practical density of the composite follows the same behavior as the degree of crystallinity, which implies a linear relationship. Figure 2 shows that the maximum short-beam strength, 77 MPa, coincides with the cure time of 90 min for peak crystallinity. The effect of cure time on the degree of conversion is not significant; the degree of conversion was approximately constant, around a value of 95%.
Degree of crystallinity for different cure times. Short-beam strength for different cure times.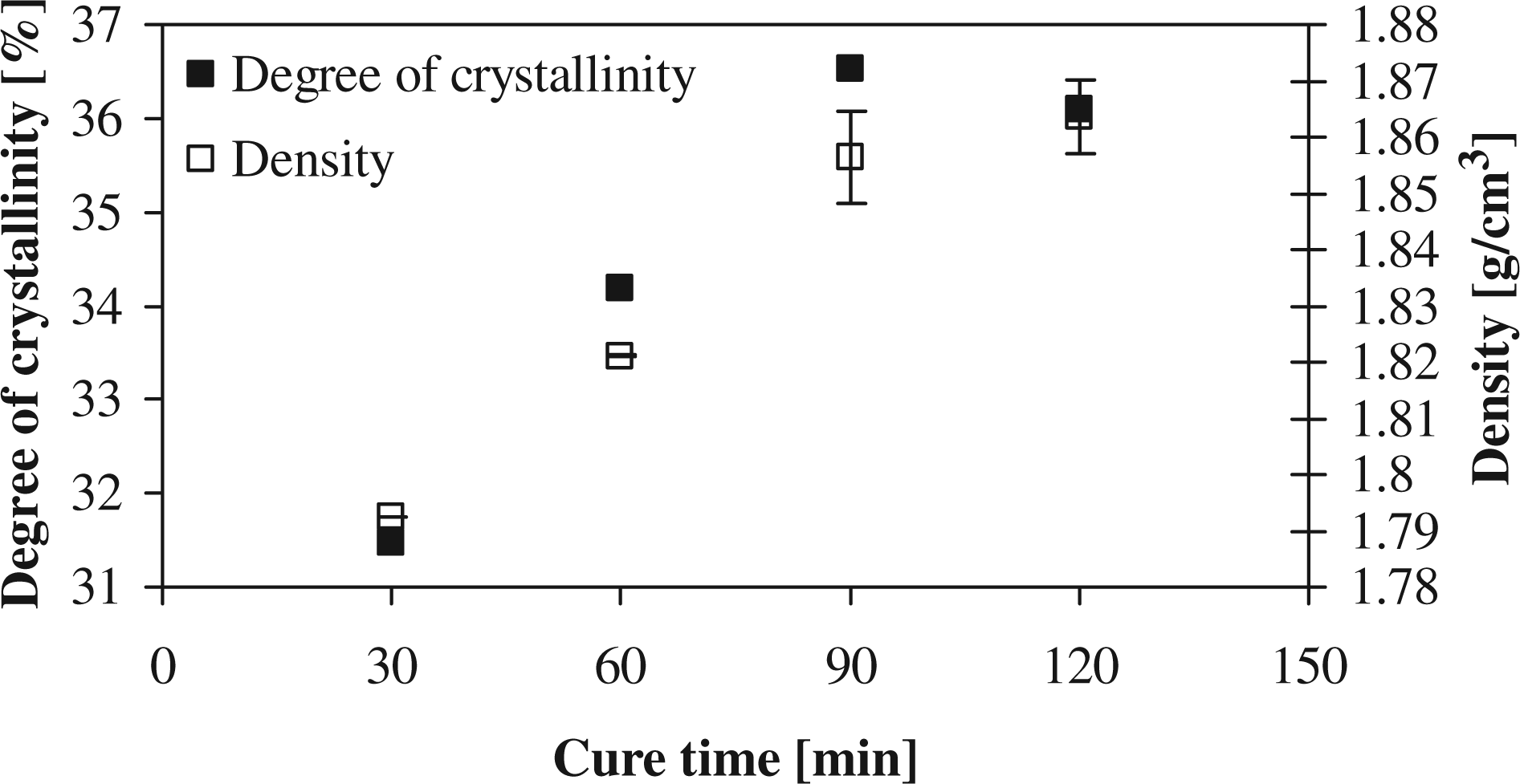
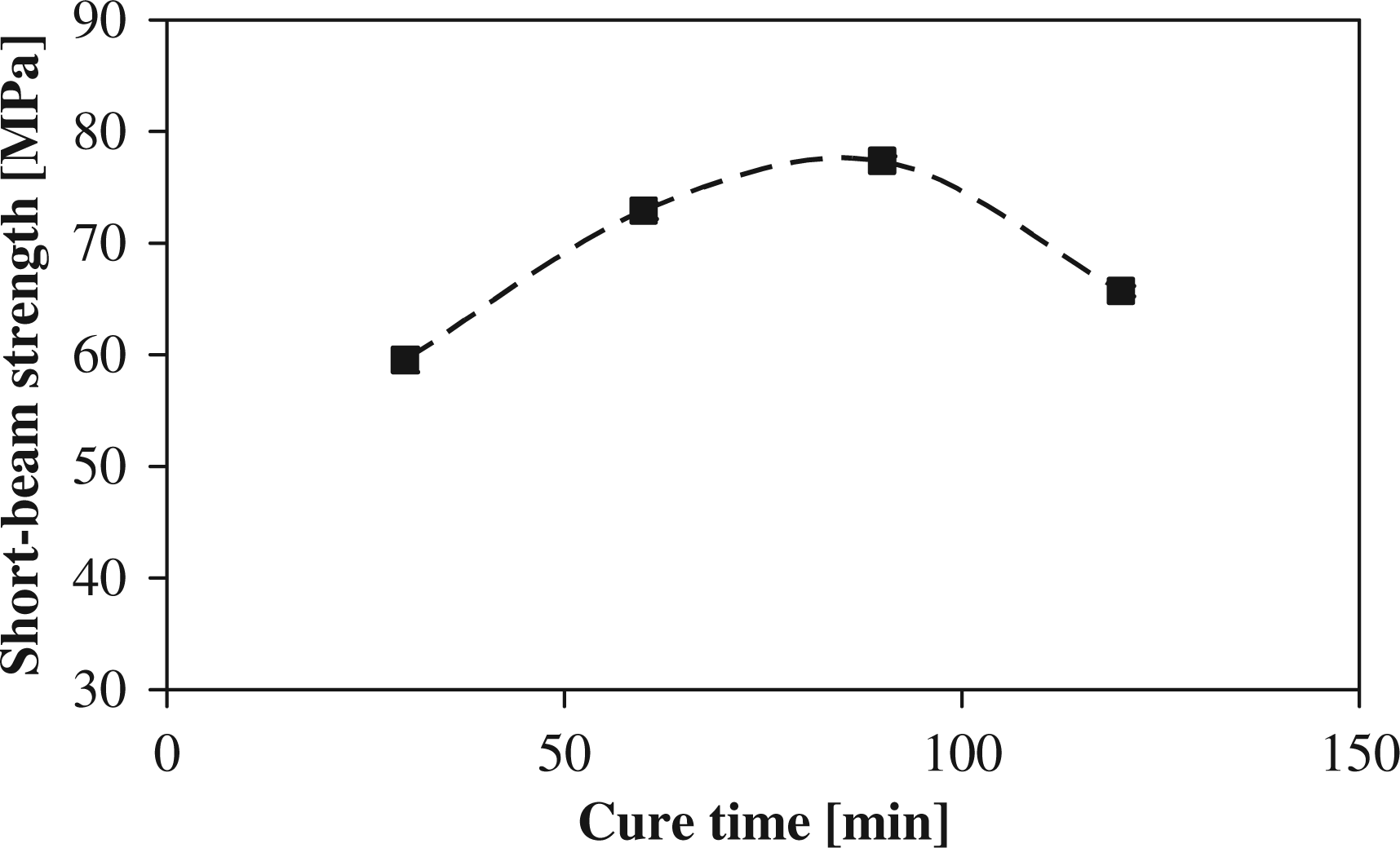
The increased crystallinity with cure time can be explained by the greater extent of primary crystallization that occurs during curing over the secondary crystallization that only initiates from subsequent cooling. With an increased primary crystallization, a greater amount of the more perfect α crystals are formed, while γ or the less perfect α crystals formed during secondary crystallization make up a lower composition of the overall crystal structure. An inherently greater extent and perfection of the crystal structure in the polymer clearly would increase the strength of the polymer due to increased covalent bonds present. This also explains the increase in the mechanical properties with cure time, confirming the direct relationship between the short-beam strength and the degree of crystallinity found by van Rijswijk et al. 15 The observed increase in density with cure time confirms the increase in crystalline order in the polymer, as was observed for PA-12. 43
To sum up, a linear relationship between the short-beam strength and the degree of crystallinity was observed. It was seen that the degree of crystallinity increased with cure time, as did the mechanical properties. The optimal degree of crystallinity and mechanical properties were obtained after 90 min of cure.
Annealing results
As seen in the last section, full crystallization and optimal mechanical properties have not been reached yet for the selected cure times in the annealing study. In this section on annealing, the following parameters were studied:
Annealing temperature Annealing time Cooling rate after the annealing cycle Initial degree of crystallinity of the composites
Annealing temperature
The effect of the annealing temperature on the change in crystallinity compared to its initial state is presented in Figure 3. It can be seen that for both curing times, the crystallinity increases significantly with increasing annealing temperatures, whereby the largest increase is found at 175°C–180°C, which is close to the recrystallization temperature of the laminate (165 ± 2°C), as was determined in the DSC measurements.
Relative change in crystallinity (compared to value before annealing) at different annealing temperatures.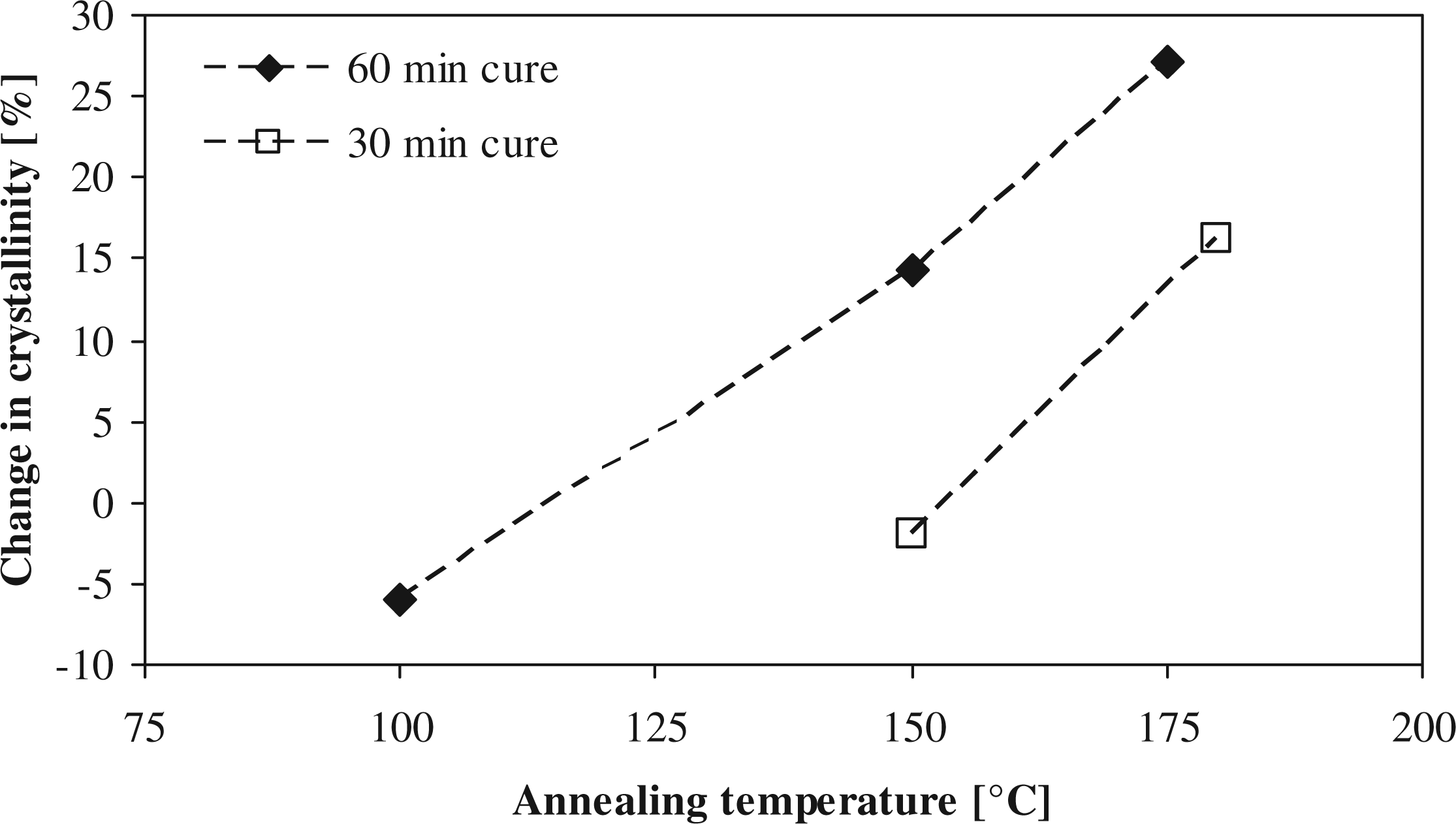
When crystalline polymers are heated to temperatures slightly under melting temperature, their lamellar crystal thickness may increase due to sufficient thermal energy given to the polymer that enables molecular motion.30,44 Gogolewski 21 outlines the fact that recrystallization of the polymer proceeds at a faster rate at higher temperatures due to the increase in the number of dislocations in the crystal structure, which forms very favorable nucleation sites. Furthermore, the release of topological constraint and stresses in the amorphous phase at higher temperature promotes crystallization. 33
The thermal energy seems crucial in the annealing process, as temperatures close to the Tg do not show an increase in crystallinity. It can be concluded that having a lower annealing temperature reduces the postprocessing effectiveness on the nylon-6. In Figure 3, even a decrease in crystallinity was seen for the annealing at 100°C. Shan et al. 44 researched annealing of PA-6 at an annealing temperature of 100°C. In that study, a decrease in crystallinity was observed with increasing time. It was concluded that not enough thermal energy was available to move the molecule chain and arrange it in a crystal lattice. Babatope et al. 45 stated that rupture of hydrogen bonds can occur around 60°C for PA-6.6, while still having low chain mobility. Therefore, the degree of crystallinity might decrease while the mechanical properties did not decrease, as was observed in this study.
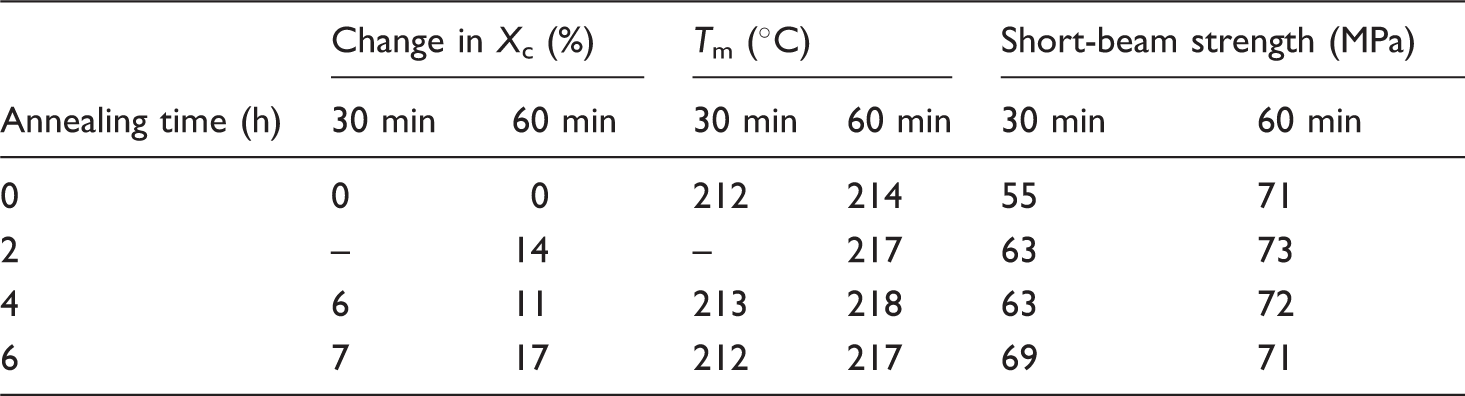
Cooling rate and degree of crystallinity after cure, change in crystallinity, and final degree of crystallinity after annealing for different cure times
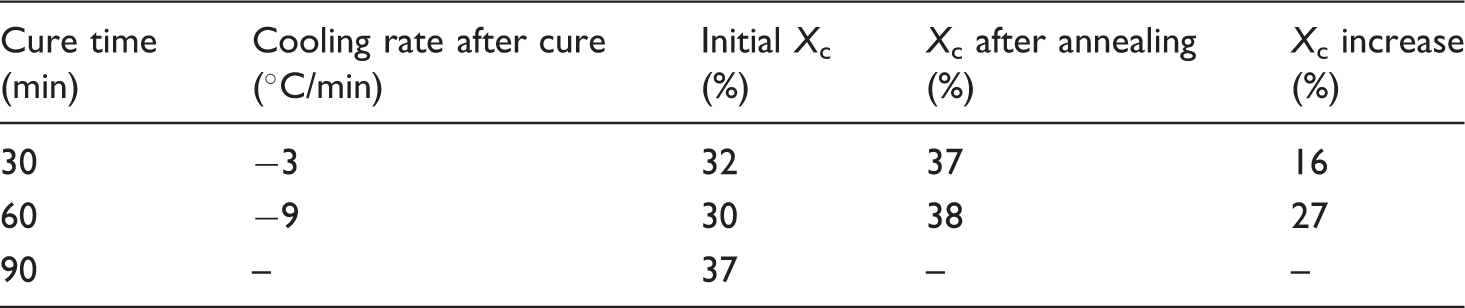
In Table 4, it can be seen that, for both cure times, a maximum degree of crystallinity of around 37% is obtained after annealing. This value also corresponds to the maximum degree of crystallinity found for a cure time of 90 min. To investigate the effect of the thermal history on the obtainable Xc during annealing, isothermal crystallization DSC measurements were performed, see Figure 4. It can be seen that the samples annealed with thermal history yield a higher Xc compared to the samples without thermal history. It can be concluded that when the samples are annealed as demolded, the highest degree of crystallinity can be obtained. It must, however, be mentioned that the crystal perfection and thus the melting temperature were better if annealed from the molten state. A melting temperature of 221°C was found compared to 218°C for the sample annealed from the demolded state.
Degree of crystallinity isothermally crystallized by differential scanning calorimetry (DSC) for different annealing conditions.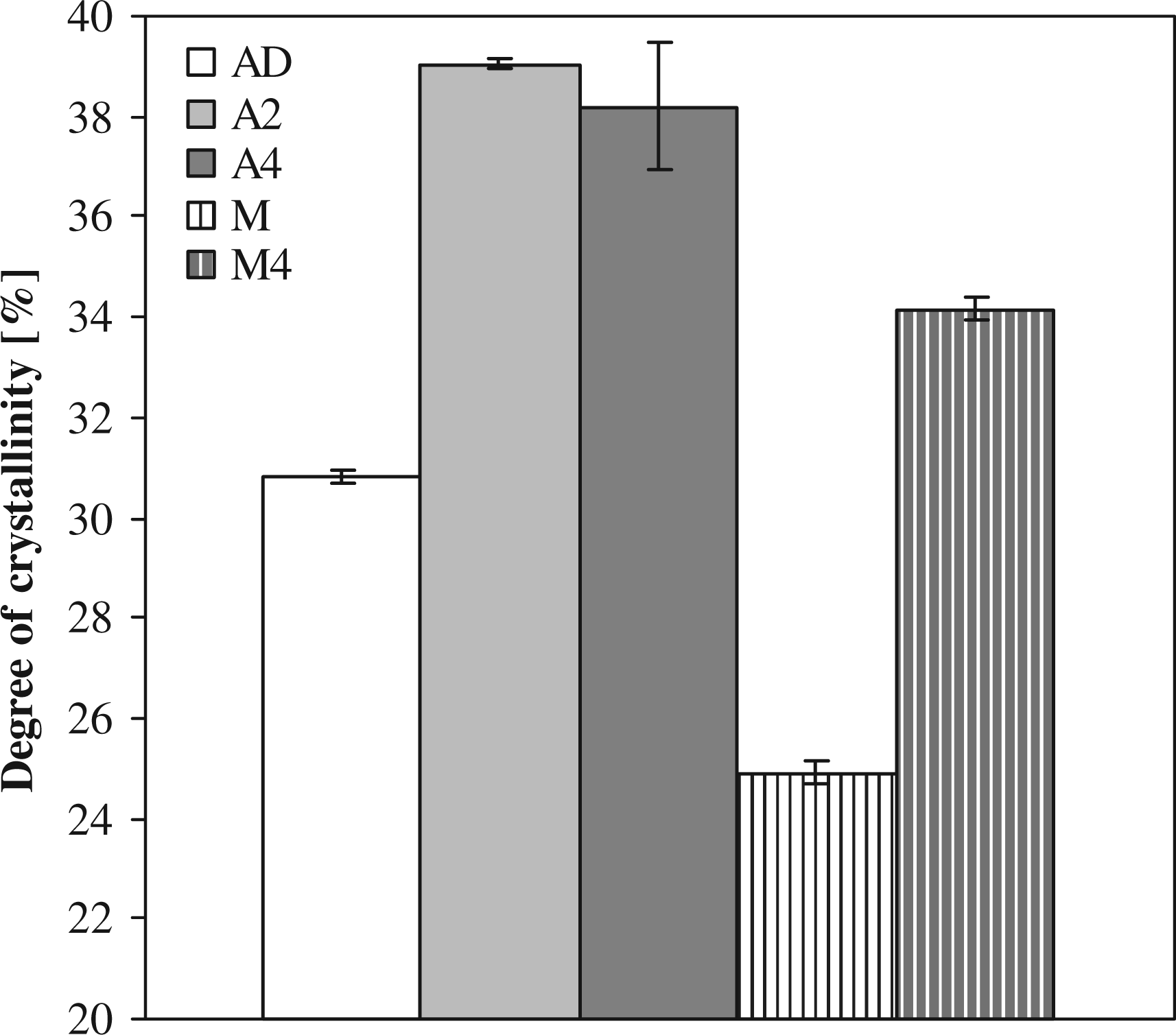
A double melting peak is observed for the annealed samples, as can be seen in Figure 5. This phenomenon was discussed by Shan et al. for annealed PA-6
26
in which multiple melting peaks were observed, when the initial crystallinity was low. The high-temperature melting peak was found to be a result of the melting of thick lamellae that where formed during primary crystallization (processing), while the low-temperature melting peak was a result of the melting of thin lamellae that where formed during the secondary annealing phase. It can be seen in Figure 5 that the high-temperature melting peaks do not change with annealing time validating that the second low-temperature melting peak can be attributed to the annealing. This difference in the distribution of lamellar thickness in the annealed samples is dependent on the annealing temperature. The double melting peaks are also seen for an annealing temperature of 150°C, while for 180°C, a single melting peak is observed. For a high annealing temperature, an increase in the lamellar thickness of the crystals and partial melting of the primary crystals takes place resulting in a single melting peak.
Differential scanning calorimetry (DSC) melting curves of anionic polyamide-6 (APA-6) samples annealed at an annealing temperature of 165°C, for 0 (AD), 2 (A2), or 4 (A4) h (curves shifted for clarity).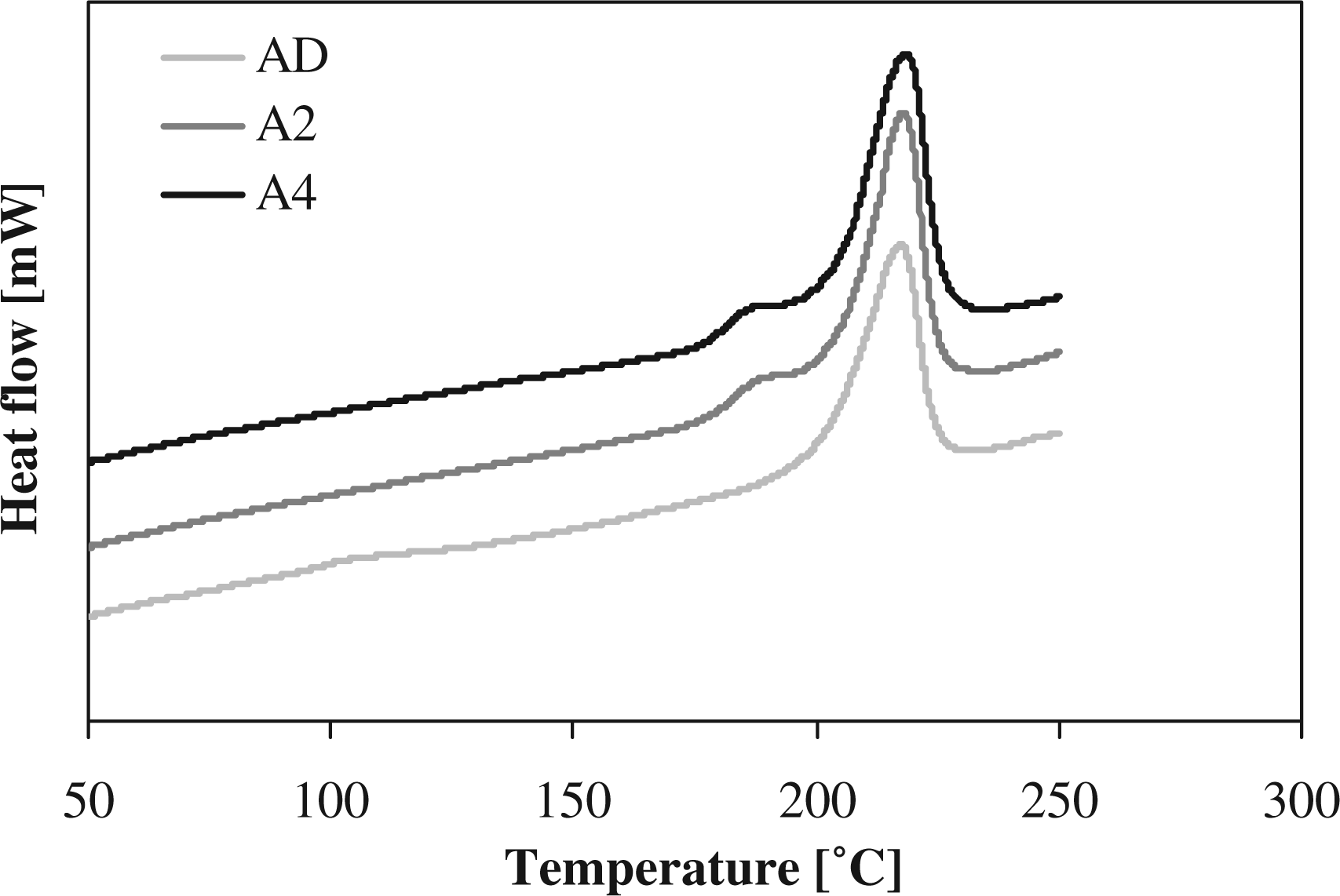
It can be assumed that an increase in melting temperature and degree of crystallinity is a measure for increase in the crystalline order, crystal size, and perfection in the polymer.25,43 The results for the melting temperature with annealing temperature are shown in Figure 6. It can be seen that the melting temperature does not increase for the annealing temperature of 100–120°C while a small increase in melting temperature is observed for the higher annealing temperatures. This complies with the results found for the degree of crystallinity. When the melting temperatures for both cure times are compared, it can be seen that the Tm for the longer cure times is higher to start with. It can be concluded that the short cure cycle, although with a higher degree of crystallinity, yielded less-perfect crystals. Therefore, more improvement in the perfection of the crystals and thus of the Tm during annealing can be expected.
Melting temperatures for different annealing temperatures.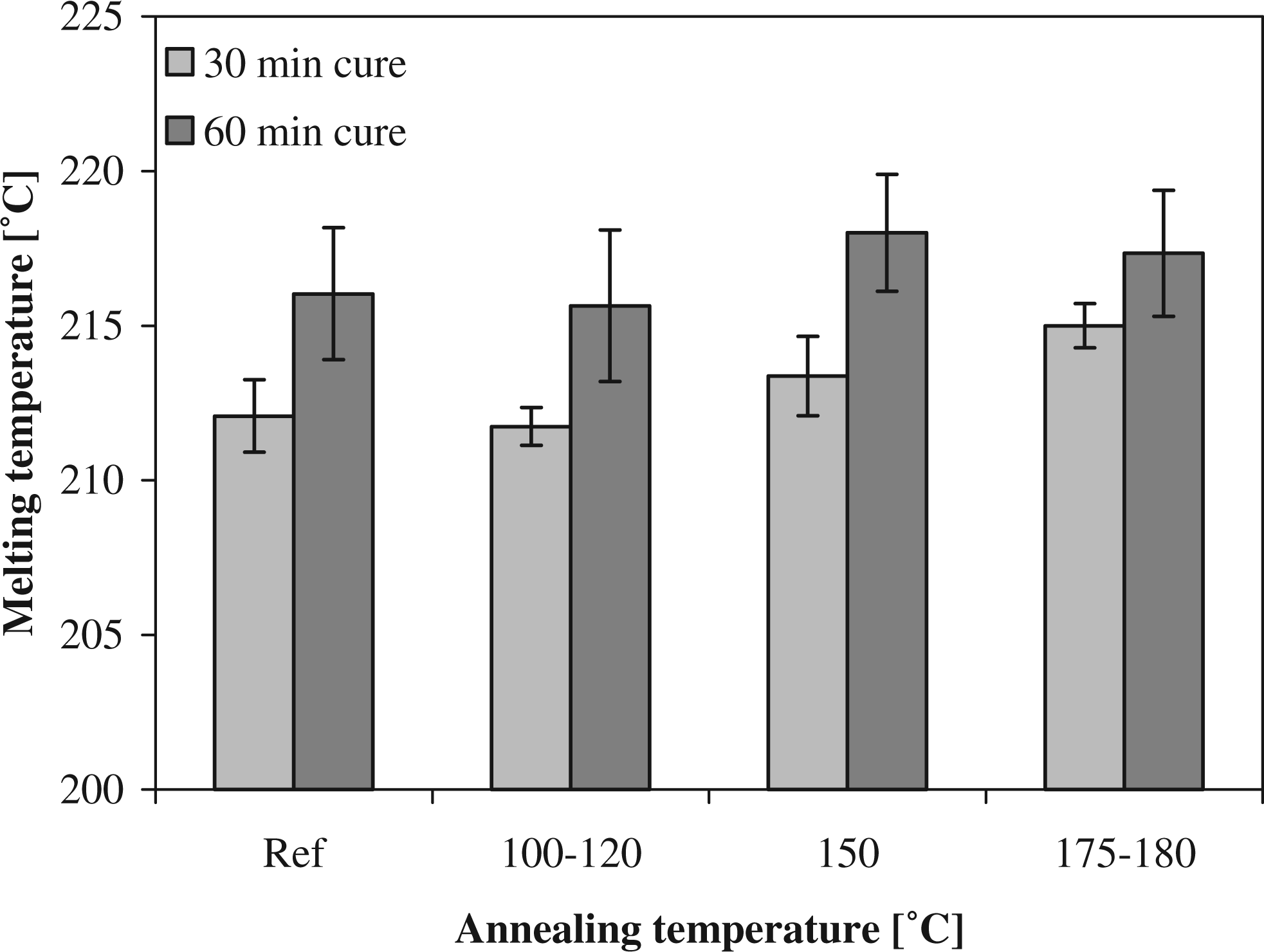
For laminates with a cure time of 60 min, the results for the short-beam strength with respect to the annealing temperature (see Figure 7) do not show the same trend as for the degree of crystallinity, as the values remain almost constant. A peak value of 73 MPa was reached for the 60-min cured laminate, compared with the 71 MPa obtained with an un-annealed sample. However, for the 30-min cured laminate, the same trend is seen as for the change in crystallinity, going from a baseline of 60 MPa to a maximum of 69 MPa. The largest difference between the two cure times is their fiber-to-matrix bond, its intertwining with the bulk matrix, and the degree of branching in the bulk, which is better developed in the 60-min cured laminates. It can thus be concluded that the degree of crystallinity is not the only parameter determining the mechanical properties. Gogolewski et al.
43
showed that in the presence of oxygen, polyamide chains can cross-link and/or branch above a temperature of 140°C. In APA-6 composites, this mechanism can also induce a better fiber-to-matrix bond. By annealing, the structure of the polymer chains is improved, the amount of dislocations reduced, and a more entangled network formed, leading to a more distinct increase in mechanical properties after annealing a 30-min cured laminate, when compared to a more perfect laminate (60 min of cure).
Short-beam strength for different annealing temperatures.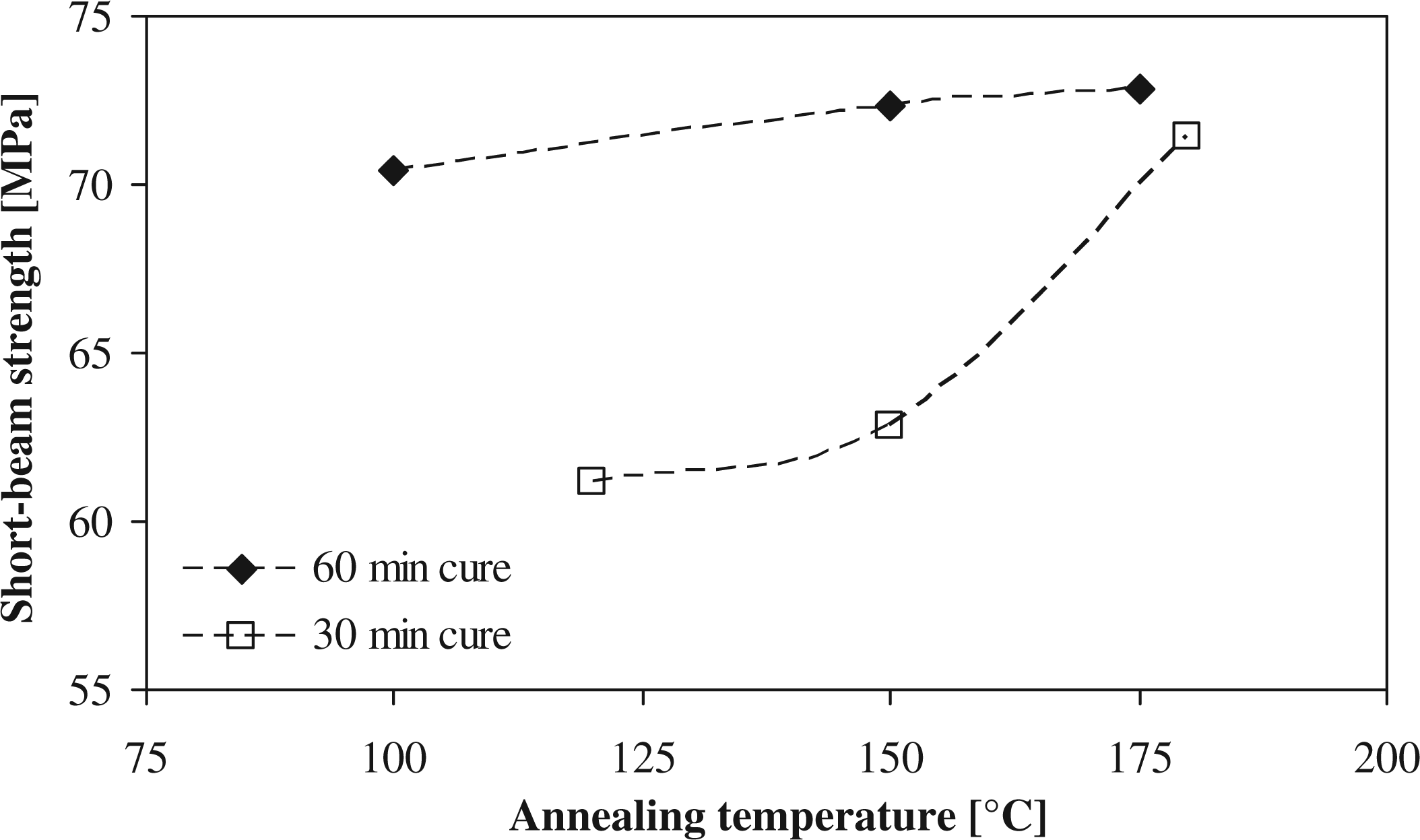
Annealing time
Effect of annealing time on degree of crystallinity, melting temperature, and short-beam strength, for different cure times
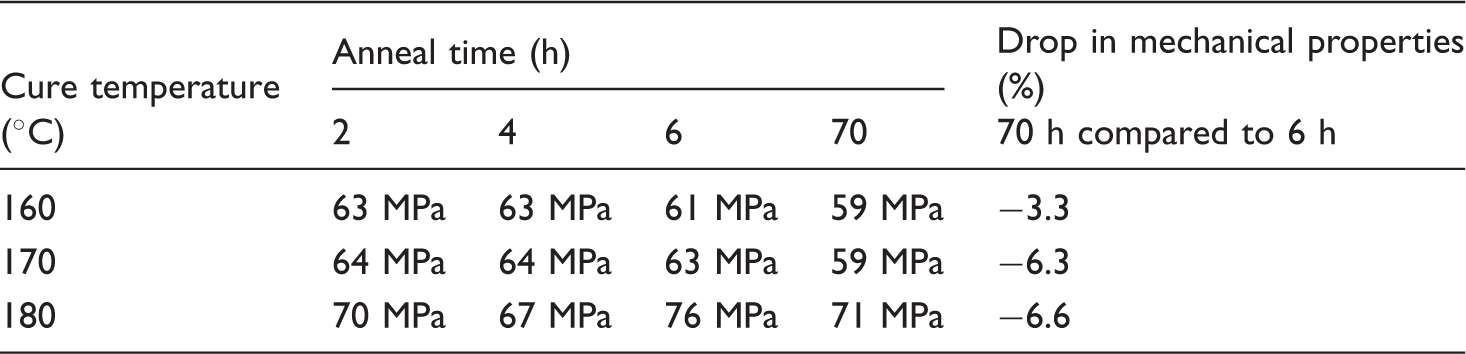
Short-beam strength for laminates annealing at 180°C, for different annealing times and cure temperatures; drop in mechanical properties between a short (6 h) and long (70 h) annealing times

Samples processed at a cure temperature of 180°C for 30 min after which they are subjected to an annealing cycle: (A) 120°C, (B) 150°C, (C) 180°C for 2, 4, 6, and 24 h, from left to right. The last sample in (C) was annealed for 70 h.

Cross-section of laminate annealed at 180°C for 70 h. Through-the-thickness oxidation (same laminate). Top: unoxidized sample (only top and bottom layer oxidised), bottom: oxidized sample through the thickness.
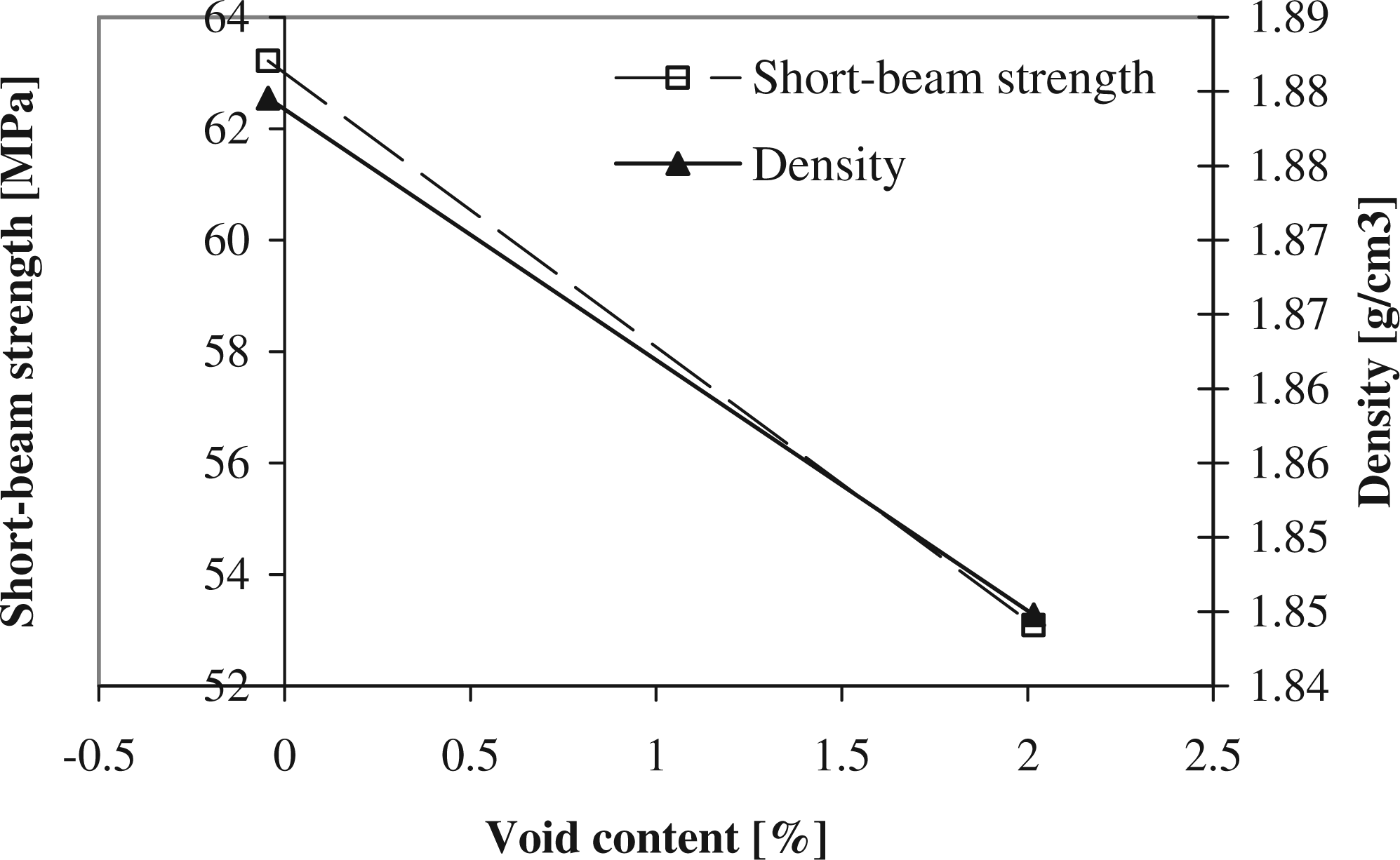
Short-beam strength and density of samples that were nonoxidized (0% voids) and oxidized (2% voids) through the thickness against their void content.

Cross-section of laminate annealed at 180°C for 70 h: unoxidized sample (only exposed layers oxidized; magnification of × 2.5).

Cross-section of laminate annealed at 180°C for 70 h: oxidized sample through the thickness (magnification of × 2.5).
Cooling rate after annealing
A reduction in the cooling rate after annealing keeps the polymer chains mobile for a longer duration, which allows more time to form crystals and thus increase the crystallinity of the polymer
46
or increase the level of perfection of the crystals. The laminates obtained after cooling inside the oven yield an increase in crystallinity of 16% compared to 11% obtained with natural cooling. The cooling rate appears to be less significant than the annealing temperature with respect to degree of crystallinity, as can be seen in Figure 13. As the polymer chains have sufficient thermal energy for crystal formation during the annealing phase, less can be gained from a slow cooling rate. The effect of the cooling rate also proves to be insignificant for the mechanical properties.
Change in crystallinity at different cooling rates.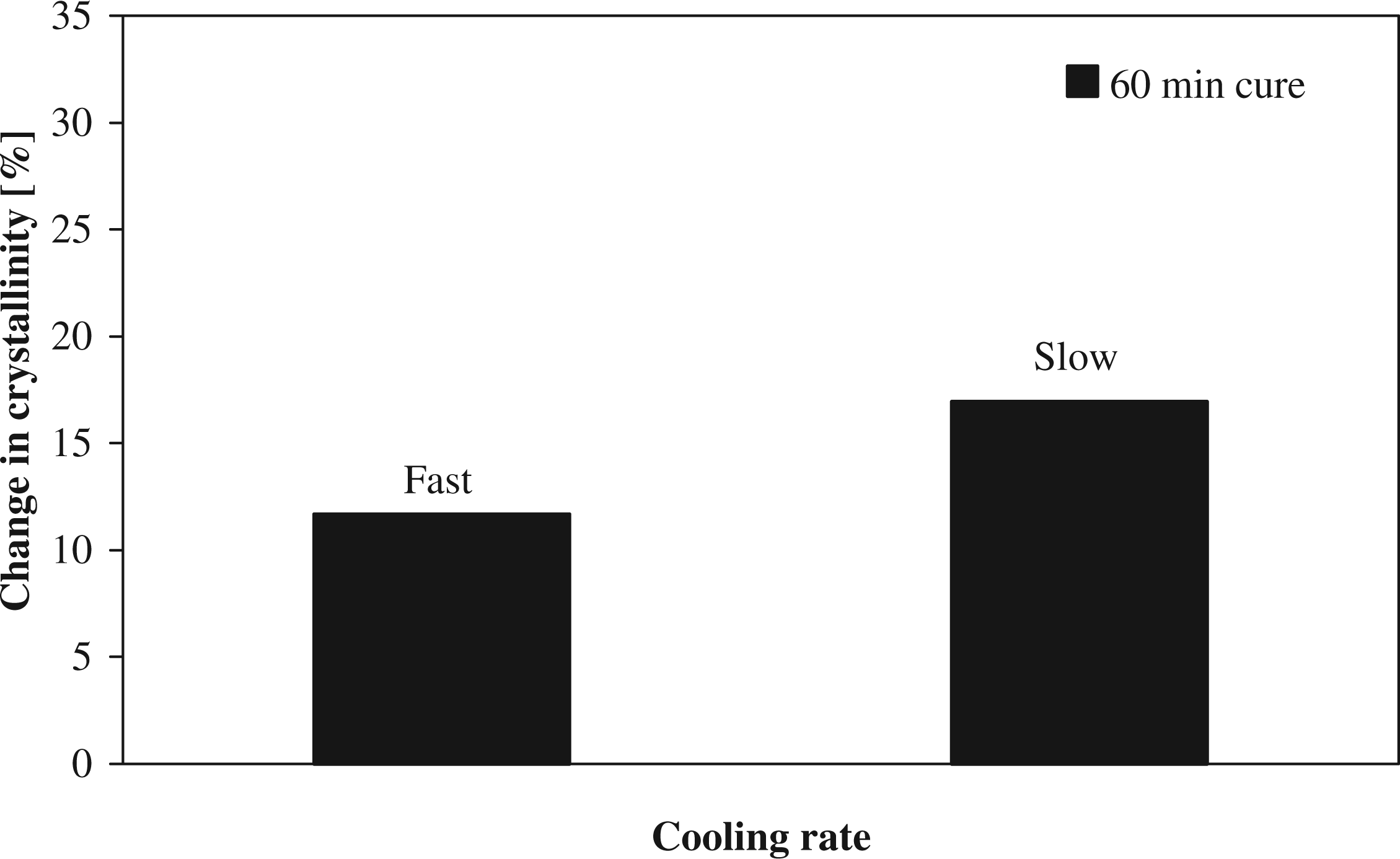
Effect of initial degree of crystallinity
In the previous sections, it was seen that the cure time is an important parameter in the annealing study. For short cure times, the mechanical properties could be improved with increasing annealing temperatures and times. These results were obtained with a high cure temperature and a low initial degree of crystallinity. In this section, the effect of annealing for lower temperature cured laminates and thus higher initial degree of crystallinity is discussed. In Figure 14 and 15, it can be seen that the mechanical properties’ increase for annealing temperatures of 150 and 180°C is used, when it is assumed that the short-beam strength without annealing is close to the value obtained after 2 h annealing at 120°C. This is consistent with the results found in section on annealing temperature. For an annealing temperature of 120°C, it can be seen that more time is needed to improve the mechanical properties as less thermal energy is available for the movement of the polymer chains. The maximum mechanical properties are obtained for the cure temperature of 180°C with a large difference when compared to the lower cure temperatures, as can be seen in Table 6. As the amorphous phase is larger in these polymer chains, the effect of branching and cross-linking on the mechanical properties is more significant. It can also be seen that the drop in mechanical properties with long annealing time is the highest at these high cure temperatures, as was discussed in the section on annealing time.
Short-beam strength at different annealing temperatures (Ta) and times for a cure temperature of 160°C and a cure time of 30 min. Short-beam strength at different annealing temperatures (Ta) and times for a cure temperature of 170°C and a cure time of 30 min.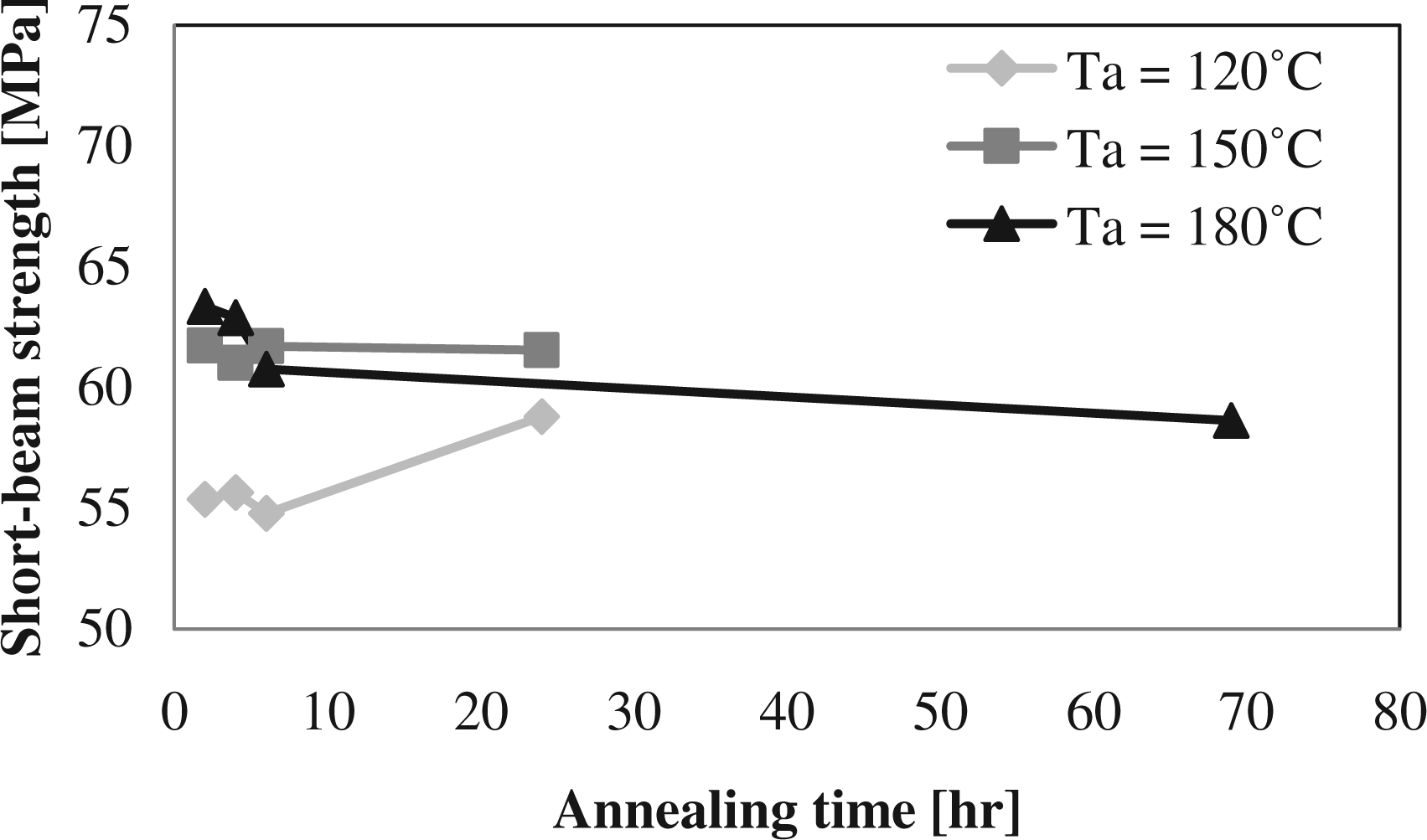
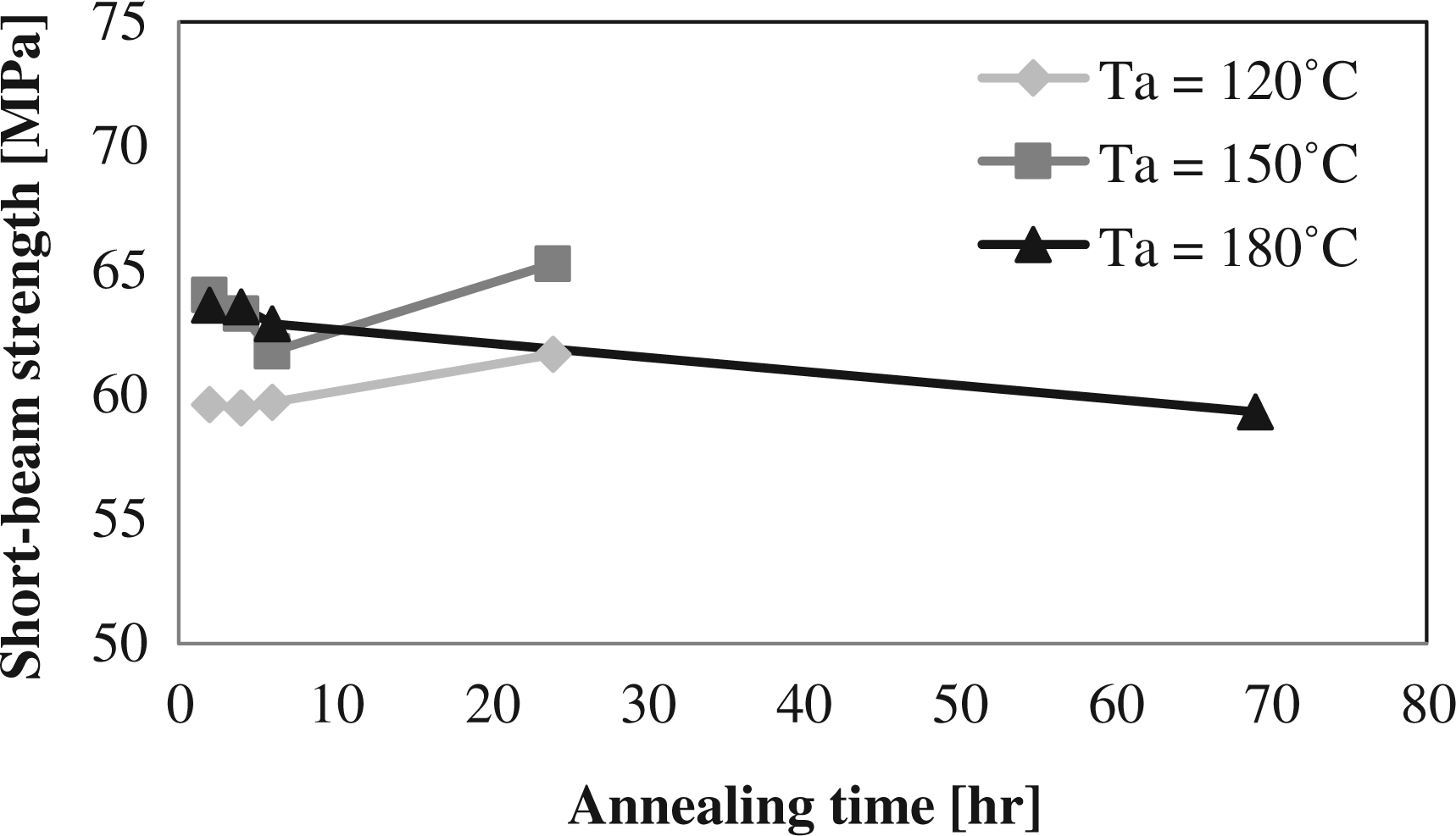
It can thus be concluded that the improvements in mechanical properties after annealing are related to the amorphous phase of the polymer. The largest improvements will be obtained in laminates with a low initial degree of crystallinity.
Conclusions
In this paper, the effect of cure time and annealing on the physical and mechanical properties of APA-6 composites was investigated. It was shown that the maximum short-beam strength (77 MPa) coincided with the maximum degree of crystallinity (37%) at a cure time of 90 min for a cure temperature of 180°C. The same properties could be obtained with a high-temperature annealing cycle, although requiring more postprocessing time. It was shown that annealing close to the crystallization temperature (175°C) of APA-6 showed the largest increase in degree of crystallinity and also mechanical properties, due to an increase in the fiber-to-matrix bond and branching/cross-linking of the matrix in the amorphous phase. As the optimal annealing temperature is close to the cure temperature of the laminate, it could be concluded that the selection of a cure time of 90 min is most effective with respect to time and energy, to obtain sufficient mechanical properties in combination with a high degree of crystallinity.
Footnotes
Acknowledgments
The authors would like to thank Sebastiaan Lindstedt, Herng SA Ang, Peter Alfano, Nader Ayoub, and Philippe-Alexandre Bérubé for their assistance and support during this research. Furthermore, Ten Cate Advanced Composites is acknowledged for kindly supplying the glass fibers. This work is part of the project PhD@SEA, which is substantially funded under the BSIK-programme (BSIK03041) of the Dutch Government and supported by the consortium WE@SEA.
