Abstract
Acute liver failure (ALF) and acute-on-chronic liver failure (ACLF) both have high mortality rates without liver transplantation. Artificial liver support systems may benefit patients with liver failure, serving as a bridge to transplantation or as a destination therapy allowing recovery. There are currently two types of artificial liver support systems: non-biological and biological. Non-biological artificial liver (NBAL) support systems primarily focus on detoxification by removing toxins through selective membranes and adsorbent materials. Well-known NBAL systems are plasma exchange, Molecular Adsorbent Recirculating System (MARS), Single-Pass Albumin Dialysis (SPAD), and the Fractionated Plasma Separation and Adsorption System (Prometheus). NBAL therapies consistently reduce bilirubin and improve encephalopathy; however, pivotal randomized controlled trials such as RELIEF (MARS) and HELIOS (Prometheus) did not confirm a survival advantage, although plasma exchange improved transplant-free survival in acute liver failure. Biological artificial liver (BAL) support systems use human or animal-derived hepatocytes to temporarily replace liver function, including the Extracorporeal Liver Assist Device (ELAD), HepatAssist, and stem-cell-based systems. Early BAL studies showed biochemical and neurological improvements, but large trials such as VTL-308 failed to demonstrate a significant survival benefit over standard medical therapy. Overall, while NBAL and BAL therapies can improve encephalopathy, renal function, and cholestasis, current evidence does not show a clear mortality benefit, and artificial liver support systems remain supportive rather than curative.
Introduction
Acute liver failure (ALF) is a life-threatening condition characterized by a rapid onset of severe liver dysfunction, coagulopathy, and hepatic encephalopathy in individuals without pre-existing liver disease. 1 Approximately 2000 to 3000 cases of ALF occur each year in the United States, accounting for 7% of annual liver transplants. 2 If liver transplantation is not performed, ALF is associated with a high mortality rate.3–5 In contrast, acute-on-chronic liver failure (ACLF) is a clinical syndrome that results from the acute deterioration of liver function in patients who have underlying chronic liver disease. ACLF is marked by an intense systemic inflammatory response triggered by precipitating events, multiple organ failures, and a high short-term mortality rate.6–10 Both ALF and ACLF have significant mortality rates, and liver transplantation remains the only definitive treatment. However, the limited availability of organs is a major barrier. In 2023, only 58.2% of patients eligible for liver transplantation received deceased donor liver transplants within one year. Additionally, the pre-transplant mortality rate among adult candidates on the waiting list was 12.9 deaths per 100 patient-years. 11
Artificial liver support systems can be utilized for patients with ALF or ACLF to supplement liver function as a bridge to definitive liver transplantation or recovery of the native liver. This article provides an updated summary of the current evidence regarding artificial liver support systems for patients with ALF and ACLF.
Pathophysiology of Acute Liver Failure
Acute liver failure (ALF) is a rapid decline in liver function in patients without pre-existing liver disease, characterized by coagulopathy and hepatic encephalopathy. The most common causes worldwide are acetaminophen toxicity and viral hepatitis. 12
ALF results from extensive hepatocyte necrosis leading to loss of the liver's detoxification, metabolic, and synthetic functions. Injury may occur from direct hepatotoxic insults (eg, drugs or toxins) or viral-mediated damage. 13 Impaired detoxification results in the accumulation of neurotoxins, particularly Ammonia, due to the disruption of the urea cycle. 14 Hyperammonemia contributes to astrocyte swelling, increased blood–brain barrier permeability, and cerebral edema, which is a major cause of morbidity in ALF. 15
Coagulopathy arises from reduced hepatic synthesis of clotting factors and regulatory proteins. Despite bleeding risk, patients may also develop microthrombosis due to an imbalance in coagulation pathways. Systemic inflammation and cytokine release can lead to multiorgan dysfunction. 16 Cerebral edema and sepsis remain the leading causes of death in patients with ALF. 13
Pathophysiology of Acute-on-Chronic Liver Failure
Acutely decompensated cirrhosis refers to the hospitalization of patients with cirrhosis due to sudden clinical deterioration, commonly presenting with ascites, gastrointestinal bleeding, hepatic encephalopathy, or bacterial infection. 6
Acute-on-chronic liver failure (ACLF) is a severe syndrome that may develop in this setting and is associated with high short-term mortality. 17 It occurs when an acute hepatic or extrahepatic insult triggers systemic inflammation and rapid clinical deterioration in patients with chronic liver disease.
Although definitions vary among international consortia, ACLF is generally characterized by acute decompensation accompanied by organ failure and high short-term mortality.17–24 Table 1 summarizes the definitions proposed by the European Association for the Study of the Liver–Chronic Liver Failure Consortium, the North American Consortium for the Study of End-Stage Liver Disease, and the Asian Pacific Association for the Study of the Liver.
Definitions of Acute-on-Chronic Liver Failure.
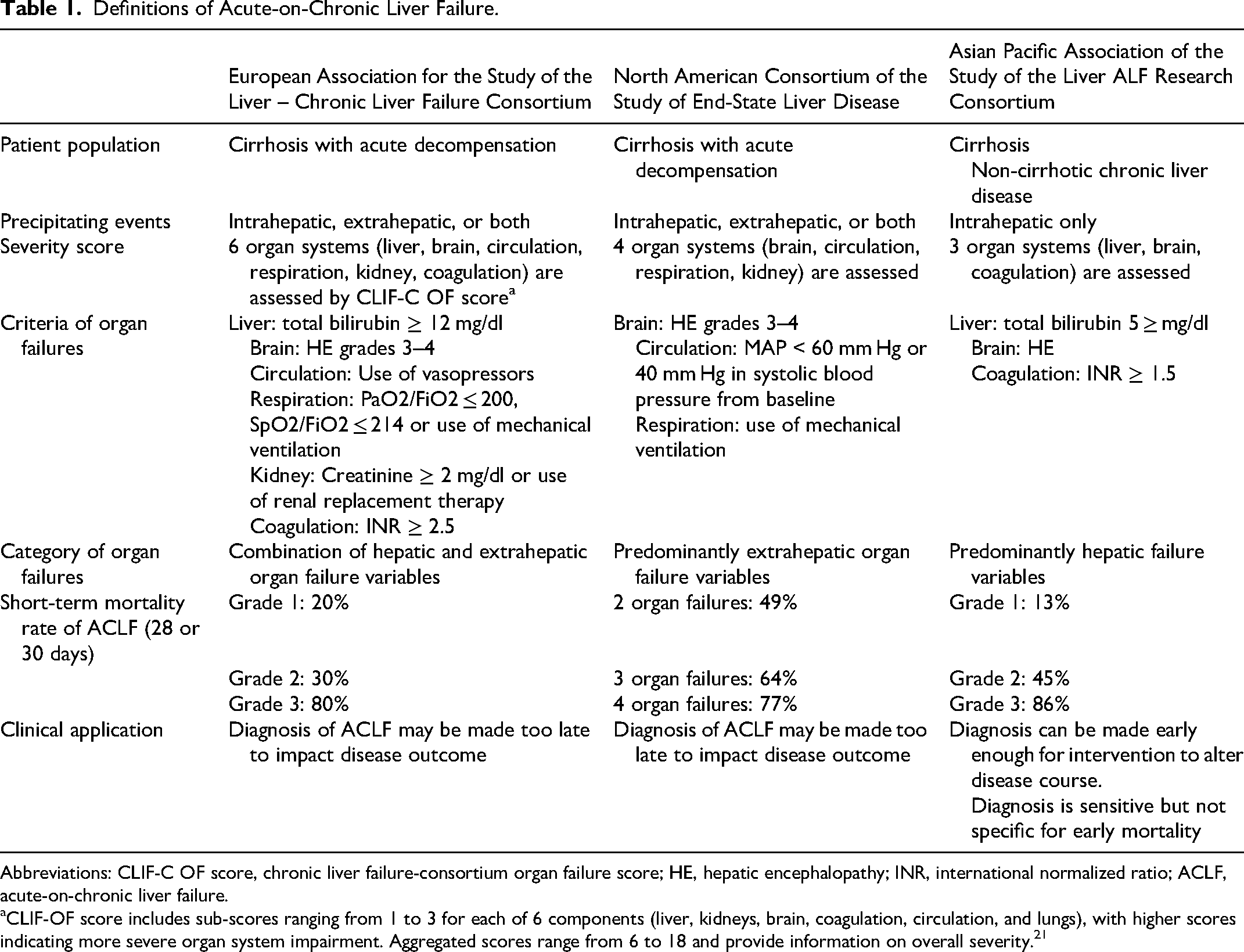
Abbreviations: CLIF-C OF score, chronic liver failure-consortium organ failure score; HE, hepatic encephalopathy; INR, international normalized ratio; ACLF, acute-on-chronic liver failure.
CLIF-OF score includes sub-scores ranging from 1 to 3 for each of 6 components (liver, kidneys, brain, coagulation, circulation, and lungs), with higher scores indicating more severe organ system impairment. Aggregated scores range from 6 to 18 and provide information on overall severity. 21
Bacterial infections and alcohol consumption are the predominant triggers in Europe and North America, while hepatitis B virus flares and hepatitis E virus superinfection are more common precipitants in Asia.
Systemic inflammation plays a central role in ACLF pathogenesis and is commonly triggered by infections, alcoholic hepatitis, or gastrointestinal bleeding.25–27 The severity of systemic inflammation correlates with the number of organ failures and short-term mortality. Inflammatory responses related to infection, liver injury, or bacterial translocation contribute to metabolic dysfunction and multiorgan failure.26,28–30
History of Artificial Liver Support Systems
The concept of artificial liver support emerged from renal dialysis in the 1950s, with early systems using hemoperfusion and plasma exchange to remove circulating toxins.
In the 1990s, extracorporeal albumin dialysis systems were developed to enhance toxin removal in patients with ALF and ACLF. These non-biological artificial liver (NBAL) devices remove both water-soluble and albumin-bound toxins using plasma separation and adsorption technologies. 31 Major systems include the Molecular Adsorbent Recirculating System (MARS), Single-Pass Albumin Dialysis (SPAD), and Fractionated Plasma Separation and Adsorption (Prometheus). Among these, MARS is the only device approved by the FDA for drug-induced ALF and hepatic encephalopathy.
Biological artificial liver (BAL) systems have been developed to provide metabolic and synthetic liver functions using hepatocytes within bioreactors.32,33 Current systems under clinical evaluation include HepatAssist, the Extracorporeal Liver Assist Device (ELAD), and stem cell–based liver support systems. A summary of artificial liver support systems is presented in Table 2.
Artificial Liver Support Systems in ALF and ACLF.
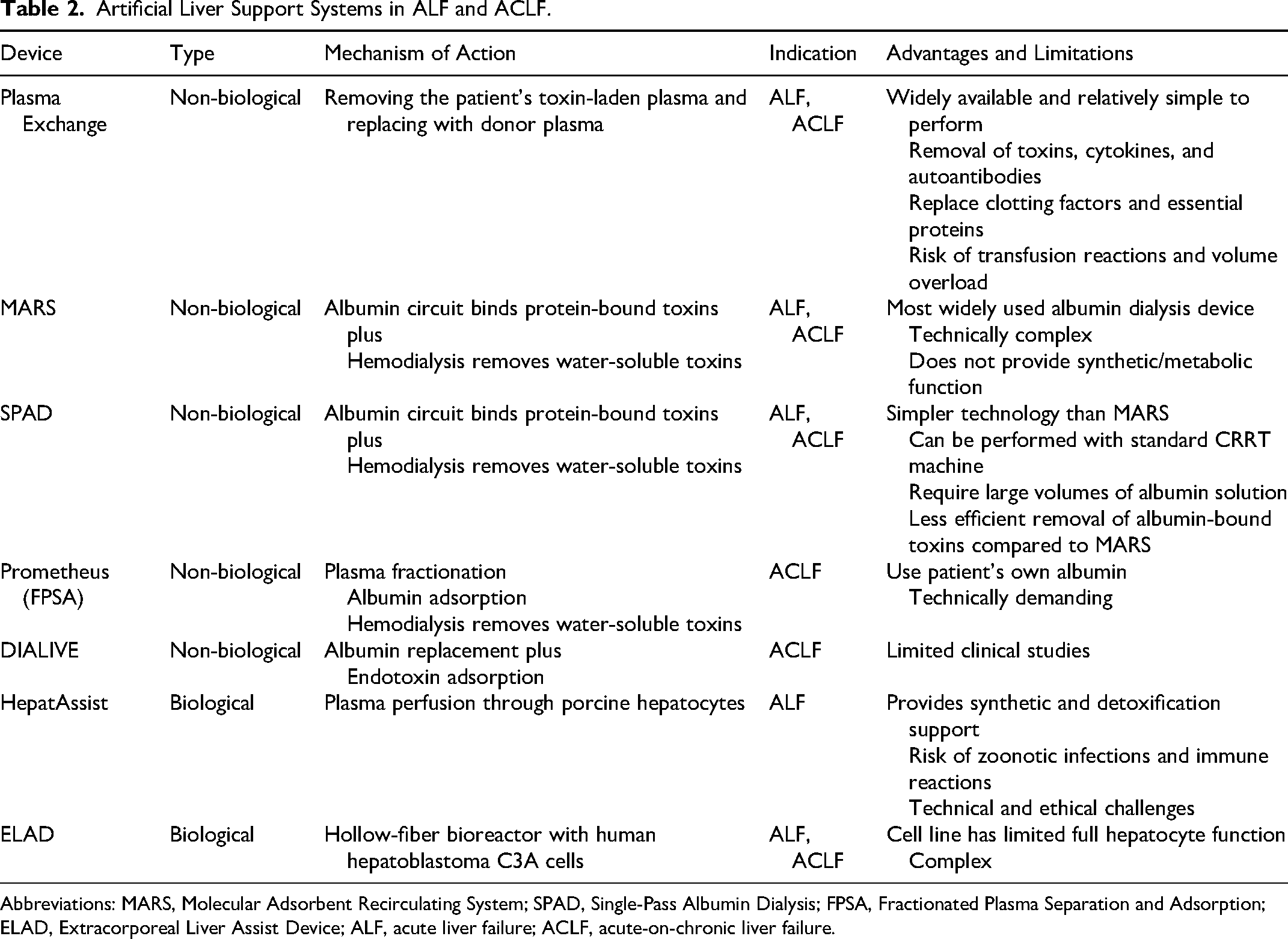
Abbreviations: MARS, Molecular Adsorbent Recirculating System; SPAD, Single-Pass Albumin Dialysis; FPSA, Fractionated Plasma Separation and Adsorption; ELAD, Extracorporeal Liver Assist Device; ALF, acute liver failure; ACLF, acute-on-chronic liver failure.
Non-Biological Artificial Liver Support Systems
Non-biological artificial liver (NBAL) support systems primarily focus on detoxifying the blood. These systems employ methods such as filtration, adsorption, and dialysis membranes to remove toxins bound to albumin or dissolved in plasma selectively. Common approaches within NBAL support systems include plasma exchange and albumin dialysis devices, with commercially available devices such as MARS, SPAD, and Prometheus.
In the ALF population, a multicenter, randomized controlled trial compared high-volume plasma exchange with standard medical therapy in 182 adults with ALF, who were randomized to receive either standard medical therapy alone or standard medical therapy plus high-volume plasma exchange for three days. The transplant-free survival was 58.7% in patients treated with high-volume plasma exchange, compared with 47.8% in those treated with standard medical therapy. The incidence of severe adverse events was similar in the two groups. The study also found that systemic inflammatory response syndrome and Sequential Organ Failure Assessment scores decreased in the high-volume plasma exchange group compared with the standard medical therapy group, with similar adverse events observed across both groups. However, in patients who were transplanted, plasma exchange before transplantation did not improve survival compared with patients who received standard medical therapy alone. 34 Two small, single-center, randomized controlled trials showed mixed findings in investigating plasma exchange compared with standard medical therapy in patients with ALF. One trial compared plasma exchange with standard medical therapy in 40 adults with ALF. They found that standard-volume plasma exchange was associated with a higher 21-day transplant-free survival rate (75% vs 45%; HR 0.30, 95% CI 0.01-0.88, p = .04). 35 A second trial involving 40 patients with ALF found that plasma exchange resulted in transient biochemical improvements but did not improve 30-day transplant-free survival. 36
In the ACLF population, a meta-analysis of plasma exchange found that it was associated with a significant reduction in mortality at 30 days, 90 days, and 1 year compared with standard medical therapy. 37 This survival benefit with plasma exchange is observed across all definitions and etiologies of ACLF, regardless of the technique and volume of plasma exchange. However, most studies are from Asia, so they need to be confirmed in the European and the United States populations of patients with ACLF. The APACHE trial was a multicenter, randomized controlled trial that assessed whether adding plasma exchange with 5% albumin to standard medical therapy improves 90-day survival in patients with ACLF. The trial was terminated in April 2025 due to funding challenges.
MARS is an extracorporeal liver support system designed to remove both water-soluble and albumin-bound toxins in patients with ALF or ACLF. Many toxins associated with liver failure, such as bilirubin, bile acids, aromatic amino acids, free fatty acids, and certain drugs, are protein-bound, primarily to albumin, making them difficult to clear using standard dialysis. MARS employs albumin-enriched dialysate on the opposite side of a semipermeable membrane. SPAD functions similarly to MARS by removing albumin-bound and water-soluble toxins but offers a simpler alternative. It operates on a single-pass dialysate that is discarded after use and requires large volumes of albumin solution. A retrospective cohort study indicated that prolonged MARS therapy (three or more sessions) in patients with ALF was associated with improved transplant-free survival. 38 Similarly, SPAD may also offer benefits in liver failure treatment. A randomized controlled trial compared 6-month survival treated with MARS versus standard medical therapy in 102 patients with acute liver failure. The study did not demonstrate a statistically significant improvement in 6-month survival among ALF patients treated with MARS compared with standard medical therapy. 39 In the RELIEF trial, a randomized controlled trial compared the 28-day survival rates of MARS with standard medical therapy, enrolling 189 patients with ACLF. The results showed no statistically significant survival benefit at 28 days. 40 However, MARS demonstrated improvement in hepatic encephalopathy and a greater decrease in serum creatinine and bilirubin. 40
Although no large randomized controlled trial specifically compared SPAD with standard medical therapy in ALF or ACLF, some case-control studies have suggested that SPAD is effective in removing protein-bound toxins in patients with ALF. 41
Unlike MARS and SPAD, the Prometheus system utilizes the patient's own albumin, which is filtered and regenerated through adsorption columns. In the HELIOS trial, the patients with ACLF were randomized to receive standard medical therapy alone or standard medical therapy plus the Prometheus. A total of 145 patients were enrolled in the trial. The HELIOS trial showed that although Prometheus improves biochemical markers of liver failure and is reasonably safe, it did not significantly improve transplant-free survival at 28 or 90 days in the ACLF population. 42 However, there is evidence that specific higher-risk subgroups (eg, MELD > 30, or those with hepatorenal syndrome) may benefit in terms of survival. Therefore, the treatment may be more effective when targeted.42–45
DIALIVE is a novel NBAL support device that combines to exchange dysfunctional albumin and remove endotoxin (damage and pathogen-associated molecular patterns) using endotoxin adsorbers (polymyxin-B-based cartridges). Unlike MARS or Prometheus, the dual actions of DIALIVE target the immune-inflammatory pathogenesis of ACLF, aiming to reverse the syndrome. In 2023, the DIALIVE trial, the first controlled randomized trial in 32 patients with alcohol-related ACLF treated with the DIALIVE device compared with standard medical therapy, showed that it was safe and reduced endotoxin and improved albumin function, which positively impact organ function. 46 Larger outcome trials are being planned to test for survival benefit. The summary of clinical trials of artificial liver support systems is in Table 3.
Clinical Trials of Artificial Liver Support in ALF and ACLF.
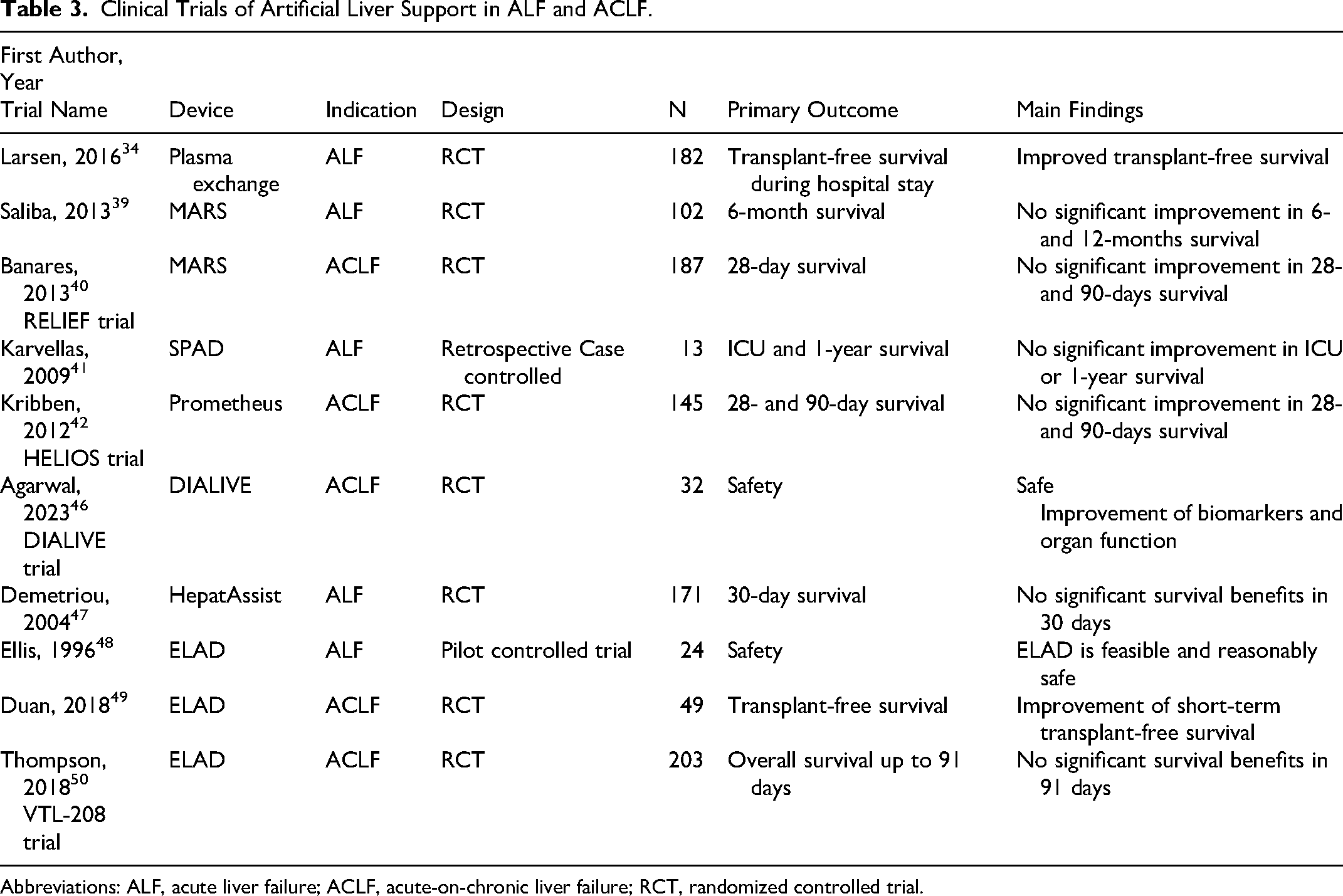
Abbreviations: ALF, acute liver failure; ACLF, acute-on-chronic liver failure; RCT, randomized controlled trial.
Biological Artificial Liver Support Systems
Biological artificial liver (BAL) support systems are cell-based devices that replicate the liver's metabolic and synthetic functions using hepatocytes in a bioreactor.
HepatAssist is one of the earliest BAL support systems. It incorporates porcine hepatocytes within hollow-fiber bioreactors and requires extracorporeal circulation. In this process, patient plasma is separated, detoxified, perfused through the hepatocyte bioreactors, and then returned to the patient. Phase I and II studies demonstrated feasibility, with improved biochemical parameters and possible short-term clinical benefits. However, the pivotal Phase III randomized controlled trial (the HepatAssist-12 trial) did not show a statistically significant overall survival benefit when compared to standard medical therapy. 47 The development of HepatAssist stalled due to the lack of a clear survival benefit in this pivotal trial, as well as the regulatory and ethical challenges associated with using xenogeneic hepatocytes, particularly the risk of porcine endogenous retroviruses. This outcome underscores the importance of conducting rigorous clinical trials to evaluate the efficacy of BAL systems and highlights the need for further research to address ethical and regulatory challenges.
ELAD (Extracorporeal Liver Assist Device) is another BAL support system that utilizes a bioreactor containing immortalized human hepatoblastoma-derived C3A liver cells. In a small, randomized pilot trial conducted in the United Kingdom, patients with ALF were assigned to either the ELAD group or the standard medical therapy group. ELAD was well tolerated, with less frequent worsening of encephalopathy in the ELAD group compared with the standard therapy group (25% vs 58%). While survival rates were similar between the groups, the trial noted promising biocompatibility and stable hemodynamics. 48 Another randomized controlled trial in China, involving 49 patients predominantly experiencing ACLF due to chronic viral hepatitis, found that those receiving ELAD for 3 to 5 days in addition to standard medical therapy had significantly improved transplant-free survival compared to those receiving standard therapy alone at day 28. 49 These results suggest that ELAD could be a beneficial treatment for patients with ACLF, particularly those with chronic viral hepatitis.
In 2018, a large, multicenter, randomized controlled trial (VTL-208) across the United States, Europe, and Australia, 203 patients with severe alcoholic hepatitis (a form of ACLF) were randomized to receive either ELAD or standard medical therapy. Although ELAD showed improvements in biomarkers (eg, bilirubin, alkaline phosphatase), these did not translate into survival benefits. Subgroup analyses indicated that patients with MELD scores below 28 tended to have better survival rates, while those with MELD scores above 28 had worse outcomes with ELAD. 50 Based on insights from earlier studies, this Phase III trial focused on a more restricted population, excluding older patients, those with high MELD scores, kidney dysfunction, or coagulopathy, and enrolled 151 patients across the United States and Europe. However, this trial (VTL-308) failed to demonstrate significant survival benefits, even after selecting subpopulations that were theoretically more responsive. The development of stem cell-derived hepatocyte devices and hybrid systems that combine mechanical detoxification with biological metabolic support is still in the experimental phase.51–54
Bioartificial liver devices may have largely failed due to difficulty maintaining a sufficient mass of functional hepatocytes, rapid loss of cell activity outside the body, and engineering limitations in bioreactors that impair oxygen and nutrient delivery.
Emerging Technologies and Innovations
For patients who fail medical management, Liver transplantation remains the definitive treatment. Yet, it continues to face two major challenges: ischemia-reperfusion injury (IRI), which compromises graft function, and a critical shortage of suitable donor organs. To address organ scarcity, the use of marginal grafts from extended-criteria donors (ECD) and donation after circulatory death (DCD) has increased, though these organs are more susceptible to IRI.
Machine perfusion (MP) has become a pivotal advancement in liver transplantation, significantly enhancing the preservation and utilization of donor organs. By providing dynamic, physiologic support to livers ex vivo, MP not only improves graft function and survival particularly for marginal and DCD organs but also allows real-time assessment of organ viability. Hypothermic machine perfusion (HMP), especially when oxygenated (HOPE), protects the biliary system and may reduce ischemic injury. In contrast, subnormothermic perfusion promotes ATP restoration and limits cold-induced damage in preclinical models. Normothermic machine perfusion (NMP) maintains active metabolism, supports functional evaluation, and, in clinical trials, has been shown to reduce early allograft dysfunction and increase utilization of marginal grafts.
Beyond preservation, MP enables therapeutic interventions, such as defatting steatotic livers and delivering cell-based therapies, further expanding the pool of transplantable organs. Emerging innovations, including artificial intelligence–guided monitoring and standardized perfusion protocols, position MP as a transformative tool that addresses donor shortages, optimizes graft quality, and may redefine clinical outcomes in liver transplantation.55,56 Combining perfusion strategies for example, HOPE followed by NMP may offer synergistic benefits, although optimal protocols remain under investigation. While MP is not yet a complete replacement for static cold storage (SCS), its potential to improve outcomes and expand the donor pool is increasingly supported by clinical evidence. Future large-scale, randomized trials are essential to refine protocols, validate emerging technologies, and establish MP as a standard component of liver transplantation.55,56
Conclusion
Artificial liver support systems in ALF and ACLF provide short-term stabilization and detoxification, yet survival benefits remain inconsistent. Biological devices face logistical barriers and complexity, while non-biological systems offer symptomatic relief without survival benefits. Future research should explore combining biological and non-biological modalities, refine patient selection, and conduct large, multicenter, randomized controlled trials to establish the impact on survival.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
