Abstract
The Canadian Association of Radiologists (CAR) metabolic dysfunction-associated steatotic liver disease (MASLD) Working Group (WG) is a multidisciplinary working group composed of radiologists, hepatologists, and family physicians. In this 3-part series, we provide Delphi consensus-based guidance on clinical and imaging findings for patients with known or suspected MASLD (formerly termed nonalcoholic fatty liver disease or NAFLD). Part 1 focuses on the detection and grading of hepatic steatosis on imaging; Part 2 on risk-stratification of patients with MASLD, including a patient pathway using serological and imaging investigations; and Part 3 on the implementation of practice recommendations for quality assurance using shear wave elastography (SWE) and magnetic resonance elastography (MRE) programs. In Part 2 of these guidelines, the WG provides 14 recommendations for risk stratifying MASLD patients into low and high risk for advanced hepatic fibrosis (F ≥ 3) using a combination of serological and imaging investigations. Population screening and risk stratification of MASLD patients using a clinical and serological investigation (FIB-4) followed by imaging pathway is recommended. Clinical, technical, and grading recommendations for point shear wave elastography (pSWE), 2-dimensional SWE (2D-SWE), and MRE are discussed.
Background
The Canadian Association of Radiologists (CAR) metabolic dysfunction-associated steatotic liver disease (MASLD) Working Group (WG) was established in 2024 through the Canadian Society of Abdominal Radiologists (CSAR) and is composed of radiologists, hepatologists, family physicians, a radiology trainee, and an ultrasound technologist from across Canada. The WG was convened to provide unified practice recommendations in patients with MASLD. The WG developed a 3-part comprehensive set of guidance recommendations. Part 1 focuses on the detection and grading of hepatic steatosis on imaging; Part 2 on risk stratification of patients with MASLD, including a patient pathway using serological and imaging investigations; and Part 3 on the implementation of practice recommendations for quality assurance using shear wave elastography (SWE) and magnetic resonance elastography (MRE) programs. Background details and methodology for the CAR MASLD WG in addition to a list of 17 guidance statements for imaging detection and multi-modality grading of hepatic steatosis are described in Part 1. 1 Part 2 of this series offers recommendations for population level risk stratification of patients with MASLD.
Several non-invasive measures for screening and risk stratification of the MASLD population have been developed including serological (blood-based) and imaging (imaging-based) investigations. The Fibrosis-4 Index (FIB-4) has become the most widely accepted blood-based investigation calculated from patient age, aspartate aminotransferase level (AST), alanine aminotransferase level (ALT), and platelet count. Imaging-based investigations of liver stiffness, an indirect measure of liver fibrosis, include vibration-controlled transient elastography (TE) (FibroScan; Echosens), shear wave elastography (SWE), and MR elastography (MRE). Shear wave elastography is further subdivided into point SWE (pSWE) and 2 dimensional SWE (2D-SWE). Transient elastography is the most widely studied tool having been used in liver specialist clinics since the mid-2000s and uses an external mechanical vibration to generate a quantitative evaluation of liver stiffness without structural details of the liver. By contrast, SWE uses a focused short-duration acoustic impulse which enables a single measurement (pSWE) or an image (2D-SWE). Shear wave elastography software is typically integrated into ultrasound machines and available as an add-on vendor-specific proprietary technology which can be performed in conjunction with B-mode and color Doppler imaging. Finally, MRE measures shear stiffness by using an external driver to produce and transmit mechanical vibrations and phase-contrast sequences with motion-encoding gradients to image shear-wave propagation. Unlike ultrasound-based elastography techniques, MRE hardware and software are standardized across major MRI vendors.
Recent North American and European guidelines have offered recommendations for risk-stratifying patients with MASLD using non-invasive investigations.2-5 Most society and multi-society clinically-based guidelines have recommended a 2-step blood-based followed by imaging-based recommendation to risk-stratify patients into low versus high-risk for advanced hepatic fibrosis (F ≥ 3).2-4 One guideline recommends further risk stratification by disease severity. 3 All guidelines use modality and vendor agnostic thresholds for their recommendations. By contrast, the Society of Radiologists in Ultrasound offers an image-based multi-threshold risk stratification approach using either pSWE or 2D-SWE. 5
Part 2: Liver Stiffness Recommendations
A multi-round Delphi methodology using a 5-point Likert scale with consensus threshold established as a score of ≥4.0 by ≥60% of the WG was used and is explained in further detail in Part 1. 1 Through this process, a list of 14 recommendations for risk stratifying patients with MASLD were developed and are shown in Table 1. The CAR MASLD WG strategy for population screening and risk stratification of MASLD patients is shown in Figure 1.
Summary Recommendations for Ultrasound and MR Liver Stiffness Measurements.

Note. TE = transient elastography; pSWE = point shear wave elastography; 2D-SWE = 2D shear wave elastography; MRE = magnetic resonance elastography; CAR MASLD WG = Canadian Association of Radiologists Metabolic Dysfunction-Associated Steatotic Liver Disease Working Group; IQR/M = interquartile range/median.
Appropriate patients include: (1) 2 or more metabolic risk factors, (2) type 2 diabetes, (3) steatosis on any imaging modality or elevated aminotransferases.
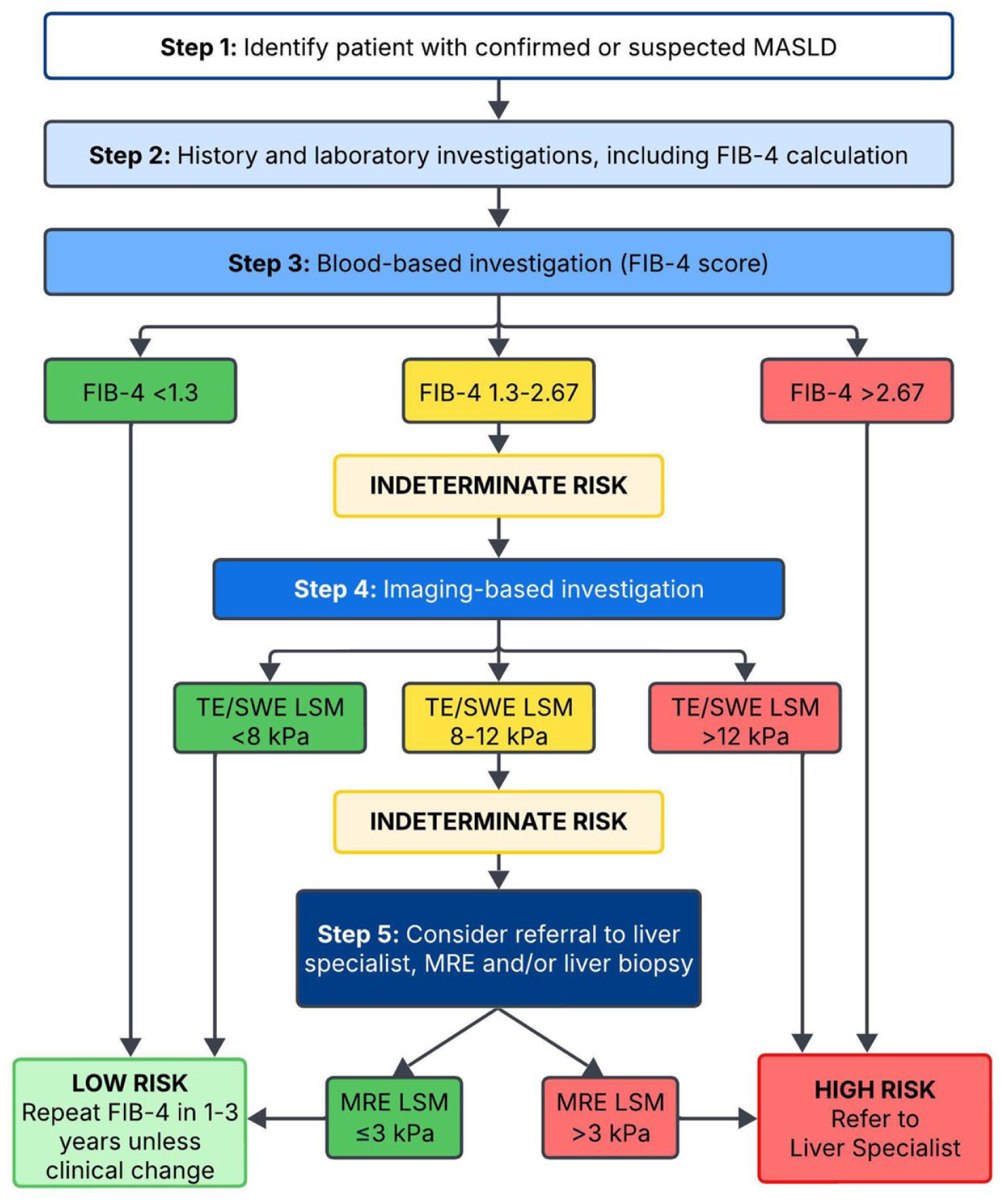
CAR MASLD Working Group strategy for population screening and risk stratification of MASLD patients.
Part 2A: Image-Based Liver Stiffness Measurement Indications
Rationale: In North America, an estimated 30% of the general population have some form of MASLD, with approximately 6% demonstrating features of metabolic dysfunction-associated steatohepatitis (MASH) and 3% of all patients demonstrating features of MASH and significant hepatic fibrosis (F≥2).6-9 The latter group is deemed to be at-risk for liver-related events such as liver failure, portal hypertension complications, and/or hepatocellular carcinoma and are known to portend a worse prognosis.10,11 Non-invasive risk stratification into low and high risk groups for identifying these at-risk patients without liver biopsy has been endorsed by multiple American and European liver societies.2-4 This practice satisfies the Canadian disease and test principles for screening programs including epidemiology, natural history, and target population criteria. 12 To further achieve both intervention and system principles necessary for an ethical screening program including program infrastructure, system integration, and cost-effectiveness, an accurate but affordable and widely available pathway for risk stratification is necessary. A 2-step, blood-based followed by imaging-based, non-invasive approach acknowledges the potential for higher-accuracy with imaging-based investigations while also balancing the need for a widely available and cost-effective initial approach.2-4 The preferential initial use of a blood-based investigation is based on low cost and wide availability. 13 The supplemental utility of imaging-based investigations in intermediate-risk patients as identified by a blood-based investigation has been suggested to improve the overall accuracy of risk stratification of these patients in differentiating low-risk community surveillance patients and patients requiring additional, potentially more costly or invasive investigations and/or specialist referrals for management. 3 For these reasons, this approach is supported by the CAR MASLD WG for population level screening and referral pathways in primary care. The American Gastroenterological Society recommends screening and risk stratification for MASLD patients with type 2 diabetes mellitus, 2 or more metabolic risk factors (including central obesity, high triglycerides, low HDL cholesterol, hypertension, prediabetes, or insulin resistance), elevated aminotransferases, and/or incidental imaging features suggestive of hepatic steatosis. 4
a. FIB-4 score <1.3 = Low risk for advanced liver disease. Repeat FIB-4 examination in 1-3 years.
b. FIB-4 score 1.3-2.67 = Indeterminate risk for advanced liver disease. Proceed with imaging-based investigation for liver stiffness measurement.
c. FIB-4 score >2.67 = High risk for advanced liver disease. Refer to a liver specialist.
Rationale: Direct and indirect blood biomarkers of liver disease lack sufficient accuracy for risk stratifying MASLD patients when used alone.3,14 Several combination values of blood tests have shown improved accuracy for identifying patients with advanced hepatic fibrosis (F ≥ 3) including: (1) Fibrosis-4 Index (FIB-4), a calculation using age, AST, ALT, and platelet count; (2) an AST to platelet ratio index (APRI); and (3) an NAFLD fibrosis score (NFS), a calculation using age, BMI, impaired fasting glucose, AST, ALT, and platelet count. Based on a combination of cost, availability, and evidence to date, FIB-4 has since become the most widely recognized calculation for risk stratifying MASLD patients with blood-based investigation.13,15 FIB-4 demonstrates an area under the receiver operating characteristic curve (AUC) of around 0.7 for detecting MASH and clinically significant fibrosis (F ≥ 2) and around 0.75 to 0.8 for detecting advanced hepatic fibrosis (F ≥ 3).14,16 An original study of 541 patients risk stratified MASLD patients using a low risk threshold of <1.3 with a sensitivity of 74% and negative predictive value (NPV) of 90% and a high risk threshold of >2.67 with a specificity of 98% and positive predictive value of 83% for detecting advanced hepatic fibrosis in patients withMASLD. 17 More recent meta-analyses have approximately supported these performance values with one individual patient data meta-analysis including over 5000 patients identifying a lower FIB-4 threshold of 0.88 for achieving a sensitivity of 90%.18,19 Based on these findings, multiple recent clinical guidelines have now adopted FIB-4 thresholds of <1.3 to define low risk populations and >2.67 to define high risk populations in MASLD.2-4 FIB-4 scores between 1.3 and 2.67 represent an indeterminate risk category and warrant further investigation for appropriate risk stratification. Both American and European guidelines have endorsed an imaging-based liver stiffness measurement (LSM) to further risk stratify MASLD patients in this setting to reduce the number of liver biopsies needed. 18 Of note, a recognized limitation of FIB-4 is decreased accuracy at extremes of age and some authors have recommended an age stratified threshold where patients ≤35 years of age are not stratified with FIB-4, patients aged 36 to 65 use above noted thresholds, and patients aged ≥65 use a low risk threshold of 2.0 rather than 1.3. 16 Additionally, alterations of indirect biomarkers underpinning the FIB-4 calculation for reasons other than hepatic fibrosis such as thrombocytopenia, active alcohol use, acute liver inflammation, and chronic renal disease can falsely alter the FIB-4 score. 3 The appropriate use of FIB-4 in these settings should be determined based on approved regional practices by liver specialists.
Part 2B: Comparative Utility of Modalities for Liver Stiffness Measurement
Rationale: Imaging-based investigations for risk stratifying patients with MASLD quantifies the elastic/shear modulus properties of liver parenchyma to generate a Liver Stiffness Measurement (LSM). This LSM serves as a surrogate marker for hepatic fibrosis. The specific techniques for acquisition of a LSM fundamentally differs between each of the modalities with TE and MRE using a form of external mechanical vibrations and SWE using an acoustic vibration known as an acoustic radiation force impulse (ARFI) to generate a single shear wave measurement (pSWE) or a 2-dimensional shear wave image (2D-SWE).20-23 Further, these techniques are often proprietary to the vendor and may differ between vendors within a single modality such as SWE. Given these fundamentally different techniques and proprietary technologies underpinning the underlying acquisition of each LSM, variable performance may exist between modalities or even between vendors within a single modality. To address this question, the American Association for the Study of Liver Disease (AASLD) recently performed a meta-analysis evaluating the utility of each non-invasive imaging-based modality for detecting and grading hepatic steatosis in over 60,000 adult patients with chronic liver disease across multiple etiologies. 24 Within this analysis of 240 articles, 54 studies evaluated these tools in patients with MASLD. No significant differences in sensitivity between modalities were identified for detecting significant (F ≥ 2) and advanced (F ≥ 3) hepatic fibrosis when allowing for some differences in ranges and thresholds by modality (eg, pSWE was reported as velocity [m/s]). MRE appeared to be more specific than the other tools. No head-to-head studies were identified showing clear superiority of one modality. Similar results have been shown in other meta-analyses using different methodologies. 25 As such, multiple societies have now recommended the use of either TE, SWE, or MRE depending on availability.2-4 The CAR MASLD WG supports this approach. While MRE may be the most accurate, it is expensive, resource intensive and has limited availability, which likely precludes MRE from routine use as a risk stratification tool at most institutions.
Part 2C: SWE Liver Stiffness Measurement Threshold Recommendations
Rationale: Current evidence does not show clear superiority of any single image-based investigation for obtaining a LSM. Further, the WG is not aware of any currently available literature showing clear superiority or threshold discrepancies between SWE vendors at the present time. As such, the WG supports both a vendor and modality-agnostic image-based LSM threshold when using TE or SWE for patients deemed to be indeterminate risk on the initial blood-based investigation.
However, while ultrasound-based elastography methods (TE, pSWE, and 2D-SWE) measure shear wave speed (m/s), the accepted practice is to report the computed Young’s modulus in kilopascals (kPa), and so our recommendation is to adopt this convention irrespective of modality. Further, while TE and MRE both use external mechanical vibration and provide results in kPa, TE reports the Young’s modulus while MRE reports the shear stiffness (understood as the magnitude of the complex shear modulus) and these 2 parameters should not be interpreted on the same scale.
a. <8 kPa – Low risk for advanced liver disease. Return to routine surveillance with FIB-4 in 1-3 years recommended.
b. 8-12 kPa – Indeterminate risk for advanced liver disease. MR elastography, liver biopsy, and/or liver specialist referral may be appropriate.
c. >12 kPa – Rules in advanced liver disease. Liver specialist referral is recommended.
Rationale: For MASLD patients, multiple American and European societies have endorsed a modality (TE or SWE) and vendor agnostic LSM of <8 kPa in patients with indeterminate risk stratification when using FIB-4 to define a low-risk patient population.2-4 This is supported by AASLD’s recent meta-analysis demonstrating sensitivities of over 80% and likely closer to 90% for both TE and SWE for excluding advanced hepatic fibrosis (F ≥ 3) in MASLD patients at this threshold. 24 Taken on a large patient population screening level, a high negative predictive value (>85%) would be achieved at this threshold. 18 By contrast, thresholds with sufficient specificity to warrant a liver specialist referral differ between guidelines, with some recommending referral at the 8 kPa threshold and others defining high risk patients warranting definitive referral above a 12 kPa threshold.2-4 Given current specialist demands and limited availability in Canada, the CAR MASLD WG supports an indeterminate range of 8 to 12 kPa for which further investigations such as MRE and/or liver biopsy may be appropriate to further assist in risk stratifying these patients. These investigations may be performed in conjunction with and/or following a liver specialist referral, depending on regional practice patterns.
a. <5 kPa – Low risk for advanced liver disease. Return to routine surveillance with FIB-4 in 1-3 years recommended in appropriate patients.*
b. 5-13 kPa – Indeterminate risk for advanced liver disease. Further testing with FIB-4 is recommended. MR elastography, liver biopsy, and/or liver specialist referral may be appropriate.
c. >13 kPa – Rules in advanced liver disease. Liver specialist referral is recommended.
*Appropriate patients include: (1) 2 or more metabolic risk factors, (2) type 2 diabetes, (3) steatosis on any imaging modality or elevated aminotransferases.
Rationale: Contrary to recent clinical guidelines noted above, the Society of Radiologists in Ultrasound (SRU) has offered recommendations for risk stratification of MASLD patients using SWE without the pre-existing requirement of a blood-based investigation. 5 As a result, it may be regional practice to offer SWE for some patients without first utilizing a blood-based investigation. In this case, the SRU has adopted a conservative “rule of four” guideline in which a 5 kPa threshold “rules out” disease while a 13 kPa threshold “rules in” disease. The SRU further risk stratifies patients as unlikely to have advanced hepatic fibrosis without other known clinical signs (5-<9 kPa) and suggestive of advanced hepatic fibrosis but requiring further testing confirmation (9-13 kPa). However, they explicitly recognize that MASLD patients may have lower thresholds for advanced hepatic fibrosis and also suggest further testing in some patients with thresholds between 7 and 9 kPa. These values are somewhat supported by results of AASLD’s recent meta-analysis. 24 As a measure of precaution and recognizing that some variability likely exists between vendors and SWE modalities (pSWE vs 2D-SWE), the CAR MASLD WG supports a more broadly encompassing indeterminate range for which further investigation with both FIB-4 and possibly additional investigations (such as MRE and/or liver biopsy) and/or liver specialist referral may be appropriate, determined on an individualized basis and regional practice patterns.
Rationale: As detailed in Recommendation #3, physical principles for LSM acquisition differ fundamentally between modalities. Further, unlike TE, which is a single-vendor product in North America, technical implementations of pSWE and 2D-SWE differ between vendors. MRE uses a standardized commercially available product across all major vendors. As such, it is possible that important differences between and within modalities are not clearly defined in the literature. The CAR MASLD WG recognizes the practical utility of a single unified threshold-based recommendation and endorses recent guidelines that have done the same. However, we also recognize the evolving nature of the SWE and MRE literature, and as adoption broadens, we acknowledge that modality and/or vendor specific recommendations may be necessary with future revisions of these guidelines.
Part 2D: pSWE and 2D-SWE Technical Recommendations
a. Patient should be fasting for a minimum of 4 h prior to the examination.
b. Measurements should be taken in an intercostal space with the patient in supine or slight lateral decubitus (30°) position with right arm in extension.
c. Measurement should be taken during breath hold at neutral breathing.
d. The 2D SWE region of interest can be positioned closer to the liver capsule, if reverberation artifacts are avoided; however, the measurement box should be positioned at least 15-20 mm below the liver capsule.
e. The CAR MASLD WG recommends reporting in kilopascals regardless of modality or vendor. Shear wave speed may be optionally reported as a supplement.
f. In most systems, the maximum ARFI push pulse is at 4-4.5 cm from the transducer, which is the optimal location for obtaining measurements.
g. In most systems, the ARFI push pulse is attenuated by 6-7 cm, limiting adequate shear wave generation.
h. Major potential confounding factors include severe liver inflammation indicated by AST and/or ALT elevation greater than 5 times upper normal limits, obstructive cholestasis, liver congestion, acute hepatitis, and infiltrative liver disease (these all lead to overestimation of the stage of fibrosis).
i. Ten measurements should be obtained with pSWE, and the final result should be expressed as the median together with the IQR/M.
j. Fewer than 10 measurements with pSWE can be obtained (at least 5); however, the IQR/M should be within the recommended range.
k. For 2D SWE, 5 measurements should be obtained when the manufacturer’s quality criteria are available, and the final result should be expressed as the median together with the IQR/M.
l. The most important reliability criterion is an IQR/M of ≤30% of the 10 measurements (pSWE) or 5 measurements (2D SWE) for kilopascals and ≤15% for measurements in velocity (in meters per second).
m. Adequate B-mode liver imaging is a prerequisite for point and 2D SWE as shear waves are tracked with B-mode.
Rationale: The reliability of SWE LSM is highly dependent on operator technique. To unify and standardize acquisitions across practices and technicians, the SRU has adopted similar standards to those recommended by the European Federation of Societies for Ultrasound in Medicine and Biology and the World Federation for Ultrasound in Medicine and Biology. 5 The CAR MASLD WG also endorses these technical recommendations but further suggests preferential unified reporting in kPa across modalities, including with pSWE which has traditionally reported in velocity rather than Young’s Modulus. Shear wave speed may be optionally reported as a supplement. Specific attention should be made to potential confounders of elevated LSM including recent postprandial status and other causes of liver inflammation noted in Recommendation 8h.
The CAR MASLD WG also recognizes that recent literature has proposed the evaluation of SWE LSM reliability on an individualized measurement basis, and this approach has been endorsed by the French Hepatology and Abdominal Radiology Societies.26,27 The WG consensus is that this technique remains investigational at the present time and may be cumbersome in clinical practice, requiring a conversion chart for the coefficient of variation. 26 We currently support the SRU recommendation for a combined IQR/median of ≤30% as the reliability measure of choice (recommendation 8i).
Part 2E: SWE Surveillance Threshold Recommendations
Rationale: Given potential variability between vendors and modalities described in recommendations 3 and 7 and even potential variability between machines, the CAR MASLD WG supports SRU recommendations for using the same machines to reduce the risk of technical variability for the same patient on follow-up, when possible. We further support the SRU suggested delta change ≥10% as being clinically significant to account for variability that may occur within and/or between vendors for SWE.5,28
Part 2F: MRE Indications
Rationale: While MRE may be the most accurate modality for LSM, MRE availability, resource demands and cost, limit its use as the first-line elastographic technique for screening at the population level screening. The WG agrees that this should not be the routinely preferred modality for image-based investigation in Canada.
a. When FIB-4 score ranges between 1.3 and 2.67 and liver stiffness measurement ranges between 8 and 12 kPa.
b. Failed US elastography (such as obese patients and patients with a large volume of ascites).
c. Where US elastography techniques are unreliable due to technical issues or show a wide variability (IQR/median ≥30%).
d. Where US elastography findings are discrepant or contradictory with clinical and laboratory findings.
Rationale: MRE has been validated against histopathology and has shown higher overall diagnostic accuracy compared to TE and SWE in MASLD patients based on multiple meta-analyses.3,24 Meta-analyses by Singh et al and Su et al investigated the accuracy of MRE in grading liver fibrosis using histopathology as the reference standard. Singh et al (697 patients from 12 studies) found AUCs of 0.88 (95% CI, 0.84-0.91) for F ≥ 2, 0.93(95% CI, 0.90-0.95) for F ≥ 3, and 0.92 (95% CI, 0.90-0.94) for F = 4. 29 Su et al (989 patients from 13 studies) found AUCs of 0.97 for F ≥ 2, 0.96 for F ≥ 3, and 0.98 for F = 4. 30 A systematic review and pooled analysis comparing MRE with transient elastography (TE) in 230 patients with MASLD found that MRE had superior accuracy to TE in determining fibrosis grade. AUCs for MRE versus TE were: 0.92 (95% CI, 0.88-0.96) versus 0.87 (95% CI, 0.82-0.91) for F ≥ 2 (P = .03), 0.93 (95% CI, 0.89-0.96) versus 0.84 (95% CI, 0.78-0.90) for F ≥ 3 (P = .001), and 0.94 (95% CI, 0.89-0.96) versus 0.84 (95% CI, 0.73-0.94) for F4 (P = .005). 31
Unlike ultrasound elastography techniques and liver biopsy, which can only sample small regions of the liver, and liver biopsy, which has inherent risks of an invasive procedure, MRE serves as a non-invasive tool which can evaluate a substantially larger liver volume. This allows MRE to estimate whole liver disease burden more accurately, a noteworthy consideration as fibrosis often affects the liver heterogeneously. As such, MRE is a preferred tool for risk stratifying patients with MASLD, particularly when other more accessible and cost-effective modalities fail or remain indeterminate. Additionally, MRE serves an increasingly important role in enabling longitudinal patient monitoring and treatment response evaluation and is a desirable tool for use in clinical trials.
Nevertheless, ultrasound elastography techniques remain more readily available and cost-effective than MRE, making TE and SWE more practical screening tools in large patient populations such as MASLD. As an adjunct to TE and SWE, MRE can serve a supplementary role in the scenarios detailed in recommendation #11.
Part 2G: MR Magnet Strength Recommendations
Rationale: MRE can be performed on both 1.5 and 3 T MR systems, the choice of which depends on local availability and resources. Due to higher gradient strength, 3 T MR systems are more susceptible to MR artifacts than 1.5 T systems. A study of 781 MRE examinations found a higher technical failure rate on 3 T (15.3%) compared to 1.5 T (3.5%) with the main causes of failure being iron deposition, high body mass index, and massive ascites. 32
Part 2H: MRE Liver Stiffness Threshold Recommendations
a. ≤3 kPa – Low risk for advanced liver disease. Return to routine surveillance with FIB-4 in 1-3 years recommended in appropriate patients.
b. >3 kPa – Advanced liver disease may be present. Liver specialist referral is recommended.
Rationale: A 2021 meta-analysis of LSMs in MASLD patients performed by the LITMUS investigators included 11 studies evaluating MRE. 33 Thresholds ranging from 2.86 to 4.14 identified significant hepatic fibrosis (F ≥ 2) with a sensitivity of 78%, specificity of 89% and AUC of 0.91 while thresholds of 2.99 to 4.80 identified advanced hepatic fibrosis (F ≥ 3) with a sensitivity of 83%, specificity of 89% and AUC of 0.92. By contrast, a 2024 meta-analysis performed by AASLD used thresholds between 3.2 and 3.6 kPa for identifying significant hepatic fibrosis with a sensitivity of 78% and specificity of 90% and thresholds of 3.6 to 3.9 kPa for detecting advanced hepatic fibrosis with a sensitivity of 82% to 93% and specificity of 90% to 95%. 24 Using these results, the AASLD has endorsed a MRE-LSM <3.1 kPa in MASLD patients with a FIB-4 score >1.3 to 2.67 as warranting further surveillance or possible biopsy. 3 A recent multi-society European guideline has also suggested surveillance in appropriate patients without advanced hepatic fibrosis in appropriate patients, with a referenced MRE-LSM threshold of 3.53 kPa in their guidelines. 2 For simplicity, we recommend a threshold of ≤3 kPa as warranting low risk stratification in patients with intermediate risk blood-based and US image-based scoring.
a. Normal: <2.5 kPa
b. Normal or inflammation: ≥2.5-2.9 kPa
c. Stage F1: ≥3.0-3.5 kPa
d. Stage F2: ≥3.5-4.0 kPa
e. Stage F3: ≥4.0-5.0 kPa
f. Stage F4: ≥5.0 kPa
Rationale: When performed outside of a risk stratification pathway, the CAR MASLD WG supports thresholds originally established by the Mayo Clinic and subsequently endorsed by the members of the Society of Abdominal Radiologists (SAR) Liver Fibrosis Disease-Focused Panel (DFP) and European Society of Radiology.34-39 These are independent of magnet strength, pulse sequences, or vendor platform. These values would align with meta-analyses indicating a high probability of being normal when <2.5 kPa and high probability of being abnormal when >5 kPa.24,33 We recognize that individualized defined thresholds endorsed in this approach between 3-5 kPa may lack nuance for potential threshold variability and disease severity overlap identified between studies in more recent pooled analyses outlined above.
Like other LSM modalities, other causes of increased liver stiffness including recent post-prandial status, hepatic inflammation, biliary obstruction, hepatic congestion, focal liver masses, and infiltrative liver processes should be recognized and avoided when possible. As such, it is important that MRE-LSMs are interpreted together with the clinical and laboratory data and risk stratification be determined on an individualized basis. Required fasting for 4 to 6 hours is also necessary before MRE examinations.
Conclusion
Part 2 of the CAR MASLD WG guidelines outlines 14 recommendations for best practice risk stratification of MASLD patients into low and high risk for advanced hepatic fibrosis using the FIB-4 score and image-based investigations. Indications, grading, and technical recommendations for performing SWE and MRE are provided based on existing literature. The CAR MASLD WG recognizes that the literature is rapidly evolving in this field as patient population risk stratification becomes more widely adopted. We anticipate periodic updates of these recommendations over the next 3 to 5-years. In addition to detecting and grading hepatic steatosis (Part 1) and risk stratifying MASLD patients (Part 2), Part 3 of the CAR MASLD WG recommendations will offer guidance for SWE and MRE program implementation and quality assurance. 40
Footnotes
Abbreviations
AASLD American Association for the Study of Liver Disease
ALT aminotransferase level
APRI AST to platelet ratio index
AST aspartate aminotransferase level
AUC area under the receiver operating characteristic curve
CAR Canadian Association of Radiologists
CLD chronic liver disease
CSAR Canadian Society of Abdominal Radiologists
CT computed tomography
FIB-4 Fibrosis-4 Index
HCC hepatocellular carcinoma
HRI hepatorenal index
HU Hounsfield units
HVPG hepatic venous pressure gradient
IP in-phase
LSM liver stiffness measurement
MASH metabolic dysfunction-associated steatohepatitis
MASLD metabolic dysfunction-associated steatotic liver disease
MRE magnetic resonance elastography
MRI magnetic resonance imaging
MRS magnetic resonance spectroscopy
NAFLD nonalcoholic fatty liver disease
NIMBLE Non-invasive Biomarkers of Metabolic Liver Disease
NPV negative predictive value
OP out-of-phase
PDFF proton density fat fraction
pSWE point shear wave elastography
PVT portal vein thrombosis
QUS quantitative ultrasound
SAR DFP Society of Abdominal Radiology Disease-Focused Panel
SRU Society of Radiologists in Ultrasound
SWE shear wave elastography
TE transient elastography
US ultrasound
WFUMB World Federation for Ultrasound in Medicine and BiologyWG working group
ORCID iDs
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
