Abstract
The rising global burden of cancer drives increased demands for medical imaging, which is essential throughout cancer care. However, delivering medical imaging presents significant environmental challenges including high energy use, reliance on single-use supplies, and the production of environmental pollutants. Environmental factors, such as ultraviolet radiation, wildfire smoke, and carcinogenic pollutants contribute to rising cancer rates, while extreme weather events driven by climate change disrupt cancer care delivery—highlighting the close connection between patient and planetary health. This review explores opportunities to improve the environmental sustainability of oncologic imaging, emphasizing the importance of patient-relevant outcomes—such as quality of life and overall survival—as a guiding principle in cancer care. Key strategies include optimizing imaging schedules to reduce low-value imaging, selecting modalities with lower environmental impact where clinically appropriate, minimizing waste streams, and adopting energy-efficient practices. Artificial intelligence offers the potential to personalize imaging schedules and improve efficiency, though its benefits must be weighed against energy use. Mobile imaging programs and integrated scheduling reduce patient travel-related emissions while promoting health equity, particularly in underserved communities. Future research should focus on optimizing imaging intervals to address patient-relevant outcomes better, expanding the use of abbreviated imaging protocols, and the judicious deployment of artificial intelligence, ensuring its benefits justify energy use.
Introduction
The global burden of cancer continues to rise, with a prevalence of 84.6 million cases in 2021—a 31.4% increase since 2010. 1 This increase is driven by environmental factors, aging populations, improved detection, and advancements in treatment that lead to prolonged survival. As a result, healthcare systems face increasing demand for medical imaging, which is essential throughout cancer care—from screening and early detection to diagnosis, treatment, response assessment, and long-term surveillance.
Imaging is essential to modern cancer care but comes with environmental challenges, including high energy use, reliance on single-use materials, and a significant carbon footprint. 2 At the same time, environmental factors such as climate change and pollution contribute to increased cancer risk and negatively affect cancer outcomes. Particulate air pollution from fossil fuel combustion and wildfires is a recognized carcinogen, implicated in increased lung cancer incidence and mortality in population studies.3,4 Ultraviolet radiation exposure—exacerbated by ozone depletion—increases skin cancer risk. 5 Extreme weather events disrupt healthcare delivery, delaying cancer diagnosis and treatment, while hindering health promotion and cancer prevention efforts. 6
These interconnected issues demonstrate the need to address both the environmental determinants of cancer and the sustainability of cancer care systems. The triple bottom line framework—integrating economic, social, and environmental considerations to define value—offers a useful approach to assessing sustainability in cancer imaging. 7 This review explores opportunities to improve the sustainability of medical imaging across the cancer care continuum, guided by a focus on patient-relevant outcomes—such as relief of symptoms, quality of life (QoL), and overall survival (OS).
General Considerations
Imaging modalities commonly used in cancer care include radiography (eg, mammography), ultrasound (US), computed tomography (CT), magnetic resonance imaging (MRI), nuclear medicine imaging (eg, single photon emission computed tomography [SPECT] and positron emission tomography [PET]), and interventional procedures. The applications and sustainability considerations for each modality are summarized in Table 1. The environmental impact of imaging varies significantly by modality, with radiography and US having lower impacts, while MRI, nuclear medicine, and interventional procedures are associated with higher impacts (Figure 1). Sustainability can be enhanced by prioritizing imaging modalities with lower impacts when multiple options are clinically appropriate.
Oncology Imaging Modalities, Applications, and Actions to Improve Environmental Sustainability.

Note. CO2e = carbon dioxide equivalent; CT = computed tomography; MRI = magnetic resonance imaging; SPECT = single photon emission computed tomography; PET = positron emission tomography; CEUS = contrast-enhanced ultrasound.
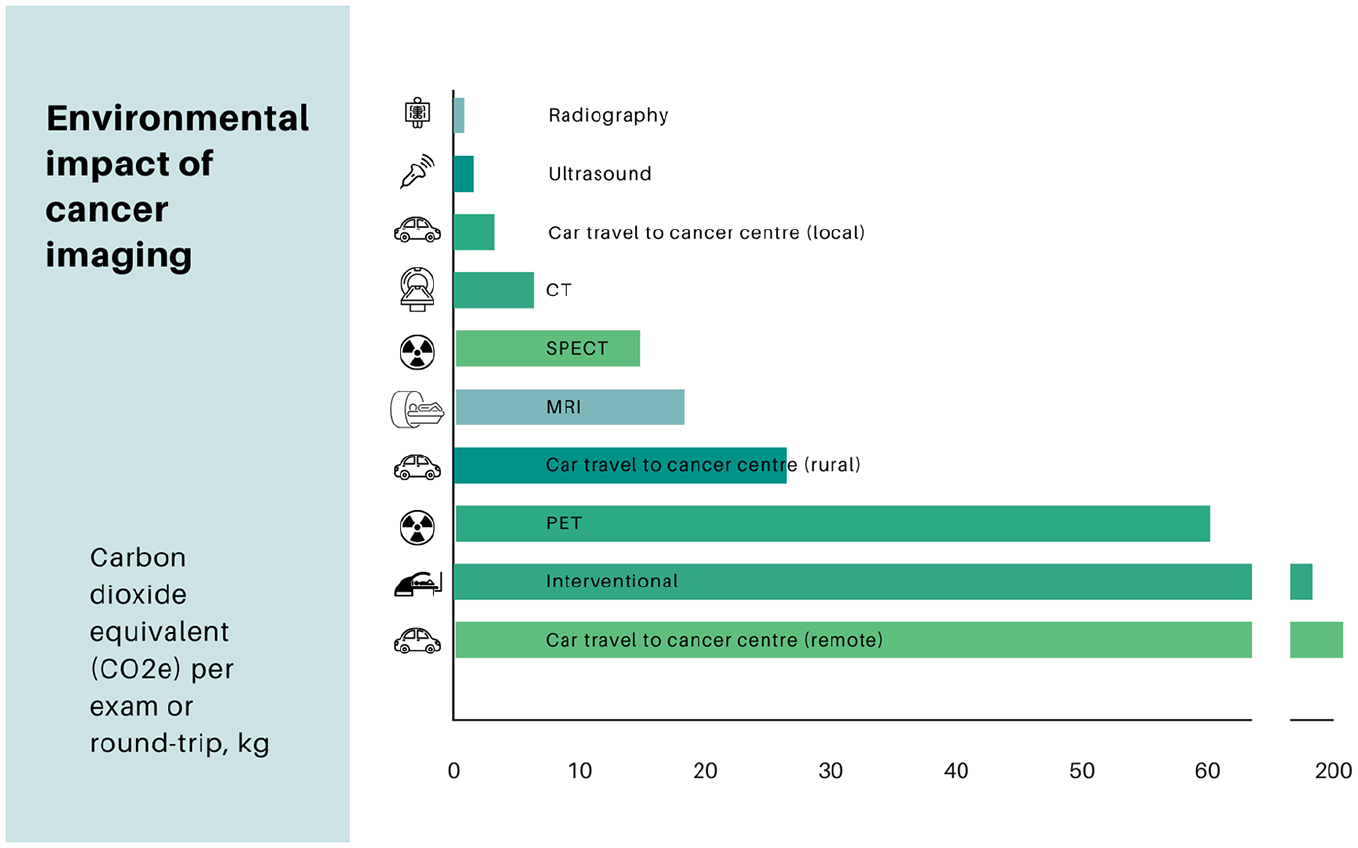
Estimated mean greenhouse gas (GHG) emissions per imaging test, by modality, and per roundtrip travel to a cancer care centre. Mean CO2 equivalent (CO2e) emissions for production and use phases were derived from previous reports.20,60 Emission estimates are as follows: 77 to 243 kg per interventional procedure, 54 to 67 kg per PET scan, 17.5 to 19.7 kg per MRI, 11.6 to 14.4 kg per SPECT, 6.6 to 9.2 kg per CT, 0.5 to 1.2 kg per ultrasound, and 0.5 to 0.8 kg per radiograph. The midpoint of each range was used for visualization. Mean CO2e emissions from patient travel were estimated at 3.0 kg for local residents, 27.1 kg for rural residents, and 205.3 kg for remote residents, based on median round-trip travel distances reported in a study of cancer patients from Manitoba, Canada: 18 km for urban patients, 164 km for rural patients, and 1244 km for remote regions. 10 Travel emissions were calculated assuming an average gasoline-powered vehicle. Estimates for SPECT and PET were based on 2000 to 2500 studies annually over a 10-year equipment lifespan.
Several overarching strategies can reduce the environmental impact of imaging throughout the cancer care continuum (Figure 2). Cancer imaging often involves serial imaging examinations for screening, treatment response assessment, or post-treatment surveillance. A key strategy to reduce low-value imaging is to optimize imaging intervals, guided by patient-relevant outcomes. Radiologists and healthcare leaders should acknowledge that many clinical trials informing current imaging schedules emphasize surrogate outcomes, such as cancer detection rate (CDR), or metrics like progression-free survival (PFS), which have shown limited correlation with clinical outcomes. 8 Further research is needed to refine imaging protocols to align more closely with patient-relevant endpoints, such as QoL and OS. Rigorous epidemiological methods are critical in addressing potential biases, such as lead-time and length biases, which can overstate the benefits of certain imaging strategies. 9
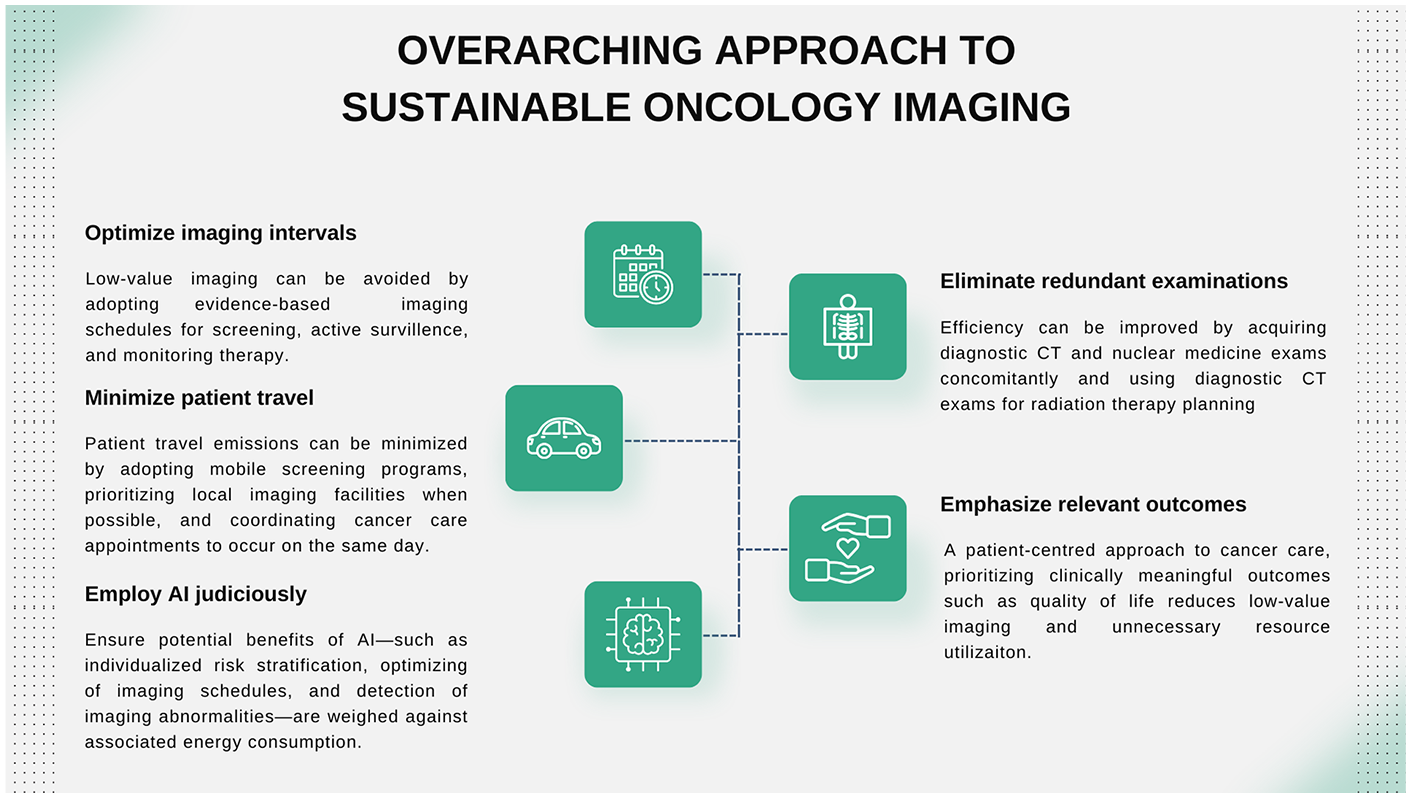
Overview of sustainable oncology imaging strategies. Key approaches to reducing the environmental footprint of cancer imaging include: (1) optimizing imaging intervals to minimize low-value imaging through evidence-based guidelines, (2) minimizing patient travel emissions by prioritizing local facilities and coordinating appointments, (3) eliminating redundant examinations by integrating diagnostic CT and nuclear medicine imaging where feasible, (4) employing artificial intelligence (AI) judiciously to balance the benefits of personalized care with energy consumption, and (5) emphasizing clinically relevant outcomes to reduce low-value imaging and prioritize quality of life. These measures align with a patientcentred approach to sustainability in oncology care.
Patient travel represents a significant source of emissions in cancer care, which, in many instances, is comparable to or exceeding those of imaging procedures (Figure 1).10,11 To mitigate this, healthcare systems can adopt strategies such as mobile screening programs, coordination of cancer care appointments—including imaging tests—to occur on the same day, and prioritizing virtual follow-up appointments when clinically appropriate. For patients living far from cancer centres, follow-up imaging can be conducted at local facilities, provided standardized imaging protocols and robust information technology systems are in place to ensure seamless comparison with prior studies from other institutions.
Artificial intelligence (AI) offers promising opportunities to improve sustainability in cancer imaging. Applications include individualized risk stratification to optimize screening and follow-up schedules, enhanced detection of imaging findings, and the potential for abbreviated imaging protocols. However, these benefits must be balanced against the substantial energy consumption of AI development and deployment. 12
Specific strategies to improve sustainability at each stage of the cancer care continuum are detailed below and summarized in Figure 3.
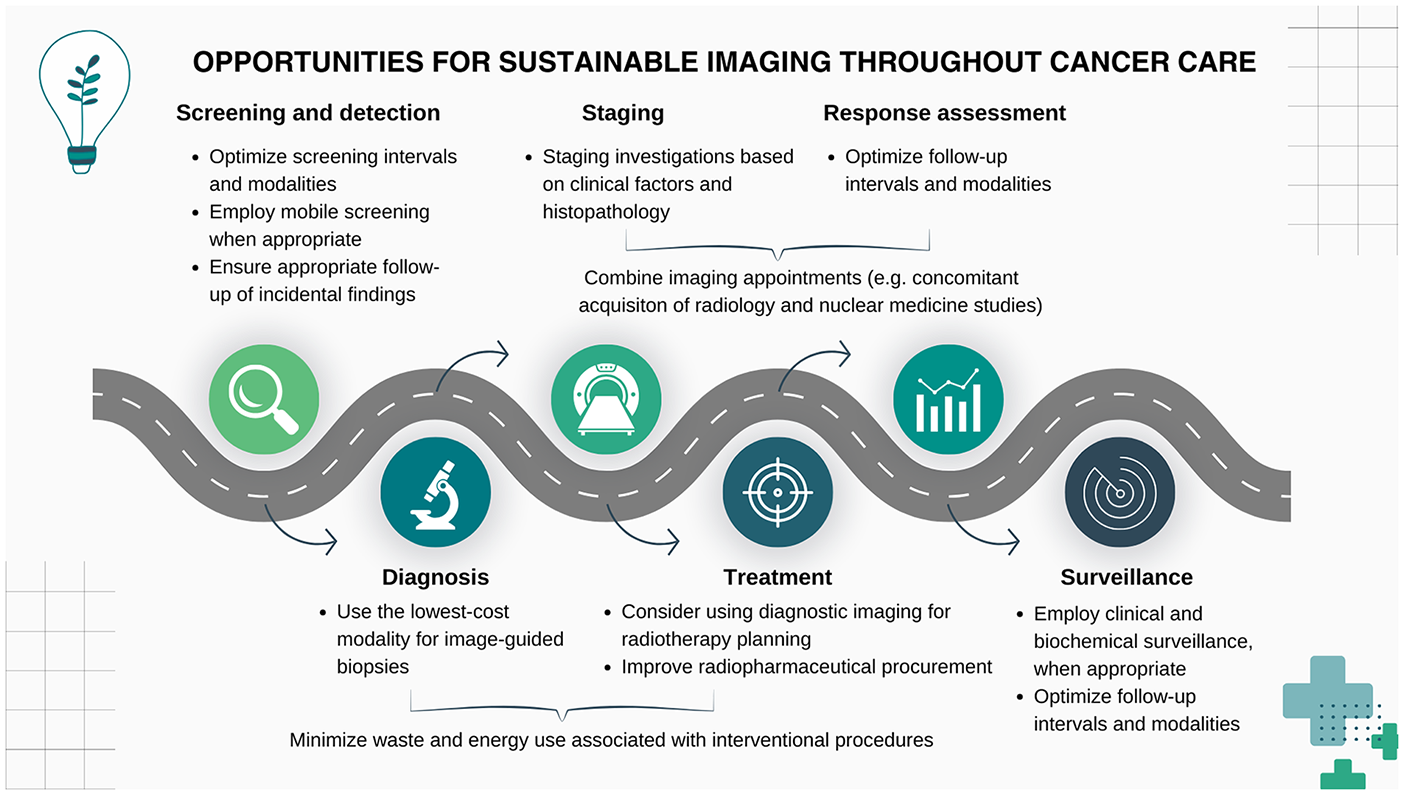
Summary of opportunities for more sustainable imaging throughout the cancer care continuum. Key strategies include optimizing screening and follow-up intervals, employing mobile screening, and combining imaging appointments during staging. For diagnosis, use cost-effective imaging modalities and minimize waste. Treatment strategies involve integrating diagnostic imaging into radiotherapy planning and optimizing radiopharmaceutical procurement. Surveillance emphasizes clinical and biochemical methods where appropriate, reducing unnecessary imaging while maintaining quality care.
Screening and Early Detection
Screening plays a vital role in early cancer detection, which not only improves patient outcomes but also has the potential to reduce the overall emissions associated with advanced-stage care. Various collaborators—including primary care physicians, cancer care specialists, public health, and epidemiological experts—must be engaged in developing sustainable screening strategies that balance the sometimes competing priorities of patient advocacy and resource stewardship. Additionally, health promotion should be emphasized as part of a comprehensive approach to care, ensuring that new cases are not only detected early, but prevented whenever possible.
Common imaging-based screening programs include breast, lung, hepatocellular, and colorectal cancers. Many screening studies can provide opportunistic screening of other health conditions, such as cardiovascular disease (CVD) and bone health, further enhancing the value of imaging. Little research directly quantifies the environmental costs and benefits of screening programs. However, we can infer significant potential cost-savings associated with early detection by using healthcare expenditures as a proxy of resource utilization and associated environmental costs. For example, a 2023 study demonstrates an exponential increase in expenditures with advancing breast cancer stage, with a greater than 10-fold increase in costs associated with the treatment of Stage IV cancer compared to Stage I. 13
It is essential to weigh the benefits of early detection against the risks of false positives and unnecessary follow-up. These carry tremendous financial and environmental costs, with, for example, the costs associated with false positive mammograms in the United States estimated at US$2.8 billion per year. 14 Optimizing screening intervals and selecting appropriate modalities are key strategies to mitigate these harms. Quality assurance systems that ensure positivity rates fall within expected ranges reduce unnecessary callback investigations.
Life expectancy and competing comorbidities must be considered in determining who is likely to benefit from screening, as the mortality benefits of screening generally emerge years after the screening test. 15 For example, most patients with known metastatic cancer should not undergo screening for other cancers, as the mortality risks of metastatic cancer generally outweigh the mortality benefits of screening.16,17 Forgoing unnecessary screening avoids financial costs, environmental impacts, and the potential complications and harms associated with downstream testing and interventions.
Breast Cancer Screening
Breast cancer screening guidelines in Canada and internationally vary in prescribed age of commencement, modalities used, and imaging frequency. These variations reflect differences in risk assessment strategies and interpretations of evidence. For example, the Canadian Association of Radiologists (CAR) recommends annual mammography screening for women at average risk starting at age 40, while also acknowledging in the guideline’s footnotes that screening for women aged 40 to 49 should involve shared decision-making based on individual risk factors and preferences. 18 The Canadian Task Force on Preventive Health Care (CTFPHC) advises routine screening beginning at age 50 in average-risk women but endorses an individualized approach for younger patients, through shared decision-making with a healthcare provider. 19
Mammography is the mainstay of breast cancer screening, using low-energy X-rays to image the breasts. Although the equipment used in mammography differs from that used for general radiography, both X-ray-based modalities have relatively low energy demands and comparable environmental impact per exam. 20 X-ray tubes used in radiography draw more power than those used in mammography, but this difference is roughly offset by the longer period of tube activation in mammography exams. 21 Beyond cancer screening, mammography provides an opportunity for early detection of CVD, with several studies demonstrating an association between the presence of breast arterial calcifications (BAC) and CVD. 22 Identifying BAC on routine mammography improves resource efficiency and may promote health equity by identifying a condition that is underrecognized in women.
In addition to mammography, supplemental screening modalities have been proposed, though they remain controversial. Digital breast tomosynthesis (DBT) involves acquiring multiple low-dose X-ray images of the breast while a detector moves in an arc; the images are subsequently reconstructed into tomographic slices for review. Adding DBT to traditional two-dimensional (2D) mammography has shown promise in reducing false positive rates and unnecessary follow-up exams in observational studies,23,24 with a multicentre randomized trial currently underway. 25 There is little published data on the energy requirements for DBT, but it is likely higher than for 2D mammography due to the multiple image exposures, motorized gantry, and postprocessing involved. However, the potential reduction in callbacks and follow-ups with DBT may offset its higher energy use, which warrants validation in future studies. Traditionally DBT is combined with 2D mammography, with both being interpreted together. Resource and workflow efficiency can be improved by forgoing the 2D acquisition and reconstructing synthetic 2D mammogram images from the data acquired during DBT. Many observational studies have demonstrated equivalent accuracy of this approach compared to the traditional approach DBT combined with 2D mammograms,23,24,26 while reducing radiation dose and potentially energy consumption.
The effects of supplemental US screening on resource utilization and patient outcomes are also under investigation, particularly in patients with dense breast parenchyma, which decreases the sensitivity of mammography. Some studies demonstrate an increase in cancer detection; however, whether this translates into improved patient outcomes requires further investigation.27,28 Given the additional imaging and follow-ups often triggered by supplemental US, its environmental implications warrant careful consideration.
MRI screening is generally reserved for high-risk patients, such as those with a strong family history or carriers of cancer-predisposing genetic mutations. Given the substantial associated energy demands associated with MRI, screening requests should be vetted using a validated clinical risk assessment tool to ensure that only truly high-risk patients undergo screening. When MRI screening is indicated, abbreviated breast MRI protocols have demonstrated promising results in early non-inferiority studies, significantly reducing scan times and energy consumption compared to standard MRI protocols.29-32
A recent UK-based study highlighted the potential benefits of adopting an AI-driven risk stratification model for breast cancer screening. 33 The AI model accurately predicted future breast cancer risk by integrating mammographic images with demographic and health history data, enabling personalized screening intervals. The findings suggest that AI-assisted individualization of screening schedules could optimize resource use and promote environmental sustainability.
Finally, mobile mammography programs deliver screening services directly to underserved rural and remote communities, significantly reducing the need for patient travel. By minimizing travel-related emissions, these initiatives lessen environmental impact and promote greater health equity. 34
Lung Cancer Screening
Annual low-dose CT (LDCT) screening for lung cancer in individuals with a history of smoking is a cost-effective approach that improves patient outcomes. 35 Additionally, it can reduce the environmental burden associated with managing advanced-stage cancer.
The utility of LDCT can be enhanced by incorporating simultaneous screening for coronary artery disease (CAD) and reduced bone mineral density (BMD)—two conditions strongly associated with smoking. Semi-quantitative visual assessment of coronary artery calcification (CAC) on non-contrast thoracic CT scans, including LDCT, has been shown to correlate with CAD and is recommended as part of routine interpretation.36,37 CT attenuation values of L1 vertebral trabecular bone on LDCT scans can be a proxy for estimating BMD. 38 These practices may identify at-risk patients sooner, prompting earlier intervention, and potentially avoiding the morbidity and resources associated with adverse cardiac events and insufficiency fractures.
As with mammography, mobile LDCT units have proven effective in reaching underserved populations—including low-income, rural, and Indigenous communities—who face disproportionately high risks of lung cancer, 39 thereby improving both health equity and environmental sustainability.
Hepatocellular Carcinoma Screening
Screening for hepatocellular carcinoma (HCC) in patients with risk factors such as liver cirrhosis or chronic hepatitis B infection reduces mortality.40,41 Screening programs most commonly combine semi-annual US examinations with biochemical surveillance (e.g., serum alpha-fetoprotein). In patients with poor sonographic liver visualization due to body habitus or diffuse liver disease, MRI has been proposed as an alternative screening modality, but it carries higher costs and energy use. Retrospective studies using simulated abbreviated MRI protocols have shown promising early results, with prospective validation of less energy-intensive abbreviated MR protocols currently underway.42,43
Colorectal Cancer Screening
In Canada, average-risk colorectal cancer screening typically involves fecal occult blood testing (FOBT) or fecal immunochemical testing (FIT), with positive screens referred for optical colonoscopy (OC). 44 In certain clinical scenarios and jurisdictions, patients may be referred for primary OC screening. CT colonography (CTC) presents a promising non-invasive alternative to OC, potentially avoiding the resources associated with OC, including equipment sterilization, sedation, and single-use supplies. A systematic review and meta-analysis of 49 studies demonstrated a similar sensitivity of CTC compared to OC for detecting colorectal cancer. 45 However, further research is needed to establish the mortality benefit of primary CTC screening and to determine the potential financial and environmental costs of incidentally detected extracolonic findings associated with downstream healthcare utilization. 46
Managing Incidental Findings
Managing incidental findings is a critical component of sustainable radiology practices. Studies indicate that 8% to 37% of radiology reports include follow-up recommendations, many of which are inconsistent with guidelines, such as those from the American College of Radiology (ACR).47,48 The absence of follow-up recommendations, or unclear or inappropriate recommendations, can lead to unnecessary investigation and follow-up.
Radiology reports employing standardized templates and language, such as the ACR TI-RADS and O-RADS frameworks for incidental thyroid and ovarian findings, respectively, reduce variability and improve adherence to management guidelines.49,50 AI-powered tools, such as natural language processing, can analyze reports to identify and standardize follow-up recommendations for incidental findings. 51
Diagnosis
Biopsies remain essential for oncologic diagnosis, tumour grading, and, increasingly, molecular phenotyping used to inform management. Image-guided biopsy modalities include mammography, US, CT, and MRI. As with all interventional procedures, environmental impact can be reduced by choosing the lowest-cost modality to access a lesion, improving the energy efficiency of room climate control, and minimizing the use of disposable devices. 52 Consultation with oncology care teams to establish standardized protocols, including using an on-site cytopathology technologist when warranted, can reduce the number of repeat biopsies due to inadequate tissue sampling. 53
In certain cases, oncologic therapy can be initiated without a biopsy-proven diagnosis, particularly when the risks associated with biopsy are significant. A retrospective study employing a Markov model suggests that this approach, combined with stereotactic ablative radiotherapy, may be viable for pulmonary nodules with a high probability of malignancy. 54 Novel PET tracers, such as 68 Ga DOTA-TATE for neuroendocrine tumours and 18 F-oestradiol for breast cancer, offer the potential for non-invasive diagnosis and, in some instances, even insight into tumour grade and molecular phenotype.55,56 However, further research is needed to assess the trade-offs between the costs and morbidity avoided by forgoing biopsy and the substantial expenses and environmental impact of PET studies, while also considering the prognostic and therapeutic benefits that tissue sampling may offer. Additionally, emerging technologies such as “liquid biopsies,” which isolate circulating tumour nucleic acids or cancer cells from blood samples, represent a promising avenue for future investigation. 57
HCC is unique because it can be diagnosed by imaging alone, often alleviating the need for a biopsy. The ACR LI-RADS framework provides criteria for the non-invasive diagnosis of HCC using US, CT, and MRI. 58 Contrast-enhanced US (CEUS) provides a less energy-intensive alternative to MRI, with a systematic review suggesting similar diagnostic performance to MRI, although the authors note potential bias and significant heterogeneity in included studies, suggesting that further validation is needed. 59 Although US-enhancing contrast agents are potent greenhouse gases, owing to the small volumes used, this is likely outweighed by the benefits of avoiding energy-intensive MRI. 60 Developing more environmentally-friendly US-enhancing agents is a potential area of future research.
Staging, Treatment Planning, and Therapy
Staging and Treatment Planning
Initial staging investigations should be informed by clinical, biochemical, and histopathological assessment, weighing the costs of imaging against the potential harms of futile or suboptimal treatment of understaged cancer. For example, multiple consensus guidelines recommend against staging investigations in asymptomatic early breast cancer, as these are unlikely to alter clinical outcomes.61-63 Conversely, despite the considerable financial and environmental costs of PET/CT imaging, these are likely outweighed by the morbidity and costs of a futile thoracotomy performed for advanced-stage lung cancer. 64 Future studies on imaging strategies should incorporate not only clinically relevant outcomes but also economic costs and environmental impacts, such as greenhouse gas emissions—aligning with the triple bottom line framework for defining value.
Advanced imaging modalities like PET/CT have demonstrated superior sensitivity in detecting metastases that are occult on traditional imaging techniques such as CT and SPECT bone scans. For instance, a large randomized multicentre trial found that PET/CT imaging with a prostate-specific membrane antigen (PSMA) tracer offers significantly greater accuracy than conventional staging methods in high-risk prostate cancer. 65 This improved detection capability led to changes in management for a significantly higher proportion of patients compared to traditional staging approaches. However, PET/CT has a substantially higher carbon footprint than conventional imaging modalities, which must be considered when integrating it into routine practice. Further research is needed to evaluate the downstream costs, environmental impact, and clinical benefits of PSMA PET-directed care to ensure its effective and sustainable use in clinical practice.
Eliminating redundant imaging examinations can improve environmental sustainability. When both are indicated, nuclear medicine and diagnostic imaging studies can be acquired concomitantly, as most modern hybrid SPECT/CT and PET/CT scanners can acquire diagnostic-quality CT images. 66 Radiotherapy planning typically involves a dedicated simulation CT scan performed in the treatment suite. Several studies have examined using prior diagnostic CT imaging for planning purposes, thus eliminating this potentially redundant examination.67,68 Additionally, ensuring that existing imaging studies from outside centres are retrieved and reviewed, rather than repeated unnecessarily at the current centre, can further reduce the environmental impact of excess imaging.
Active Surveillance
As awareness increases about the risks of overdiagnosis and overtreatment, active surveillance is becoming a more common approach for managing certain cancers. This strategy is especially appropriate for cancers like prostate cancer, which tend to grow slowly. 69 Active surveillance involves regular monitoring through scheduled follow-ups, ensuring timely curative treatment if the cancer demonstrates signs of progression.
Protocols for active surveillance typically include serial imaging, and optimizing follow-up intervals, imaging protocols, and modalities can enhance both clinical outcomes and sustainability. For example, using biparametric MRI instead of multiparametric protocols for prostate cancer active surveillance can significantly reduce scan time, energy consumption, and the environmental impact associated with gadolinium-based contrast agents. 70 A study incorporating this approach, alongside a robust system for recalling patients if contrast is deemed necessary, showed promising results: only 5.7% of patients required follow-up with contrast administration. 71
Therapy
Oncologic therapies supported by medical imaging departments include interventional radiology placement of vascular access devices and the targeted intravascular delivery of therapeutic agents. Sustainable practices in these procedures include minimizing disposable device use, optimizing procedure suites’ energy efficiency, and adjusting climate control parameters during off-hours. 52 When clinically appropriate, travel-related emissions can be minimized by considering virtual post-procedure follow-up appointments.
Radionuclide theranostic procedures performed by nuclear medicine are also becoming increasingly prevalent. These treatments typically involve a pair of radionuclides—one primarily used for diagnostics and treatment planning, and the other for its cytotoxic therapeutic effects. Table 2 provides an overview of commonly used radionuclide pairs and the associated sustainability considerations.
Commonly Used Radionuclide Pairs in Theranostics and Sustainability Considerations Including the Environmental Impact of Radionuclide Production Methods (Nuclear Reactor vs Cyclotron), Risks Related to Long-Lived Radioactive Contaminants, Supply Chain Fragility, and Transportation Emissions. Specific Challenges and Benefits Are Noted for Each Production Method, Including Waste Generation, Energy Requirements, and Distribution Logistics.81,82

Note. MIBG = metaiodobenzylguanidine; PSMA = prostate-specific membrane antigen.
Specific considerations for reactor production include limited sources of feedstock materials, relatively large amounts of associated radioactive waste, and potential for co-generation of electricity.
Specific considerations for cyclotron production include large energy requirements and relatively lower amounts of radioactive waste.
The production and supply chains of radiopharmaceuticals are inherently complex, raising environmental, geopolitical, and ethical challenges. 72 Medical radionuclides are often short-lived and have limited sources of feedstock material, leading to high energy demands for transportation, vulnerable supply chains, and potential implications for global security. For instance, lutetium-177 (177Lu), which is widely used in theranostic applications, was until recently sourced exclusively from feedstock material mined in Russia. 73 Developing radionuclide production strategies that rely on regionally sourced materials would enhance sustainability while strengthening healthcare systems’ resilience.
Treatment Monitoring and Long-Term Follow-Up
Treatment Response Monitoring
A significant portion of imaging resources is dedicated to monitoring patients during and after cancer treatment. While minimizing unnecessary imaging is critical for sustainability, appropriate imaging for advanced cancer plays an essential role in identifying disease progression early, potentially preventing futile treatment with carbon-intensive therapies such as chemotherapy. Optimal follow-up strategies balance the goal of reducing low-value imaging with ensuring timely identification of patients who may not benefit from continued treatment.
Despite its importance, many current follow-up protocols are based on clinical trials that emphasize metrics with limited clinical relevance, such as PFS, or rely on expert consensus rather than robust research evidence. For example, the National Comprehensive Cancer Network acknowledges the absence of definitive data, offering broad and variable follow-up interval recommendations: every 2 to 6 months or 2 to 4 treatment cycles for metastatic breast cancer, every 6 to 12 weeks or 2 to 4 cycles for non-small cell lung cancer, and every 3 to 6 months for advanced-stage colon cancer. 74 Similarly, while there is significant interest in reducing radiation exposure in pediatric patients—a goal that aligns with sustainability efforts to minimize low-value imaging—a joint task force from the Society of Pediatric Radiology and the Children’s Oncology Group has highlighted the substantial lack of evidence to guide imaging strategies. In most cases, follow-up intervals and modalities are determined arbitrarily during trial design, demonstrating the need for evidence-based approaches. 75
Most studies on imaging follow-up intervals are small and retrospective in nature. For instance, a retrospective analysis of 80 patients who underwent whole-body FDG PET/CT after treatment for sinonasal malignancies found that follow-up imaging at 1 to 3 months and beyond 18 months post-treatment provided the highest predictive accuracy for recurrence. 76 Another study looking at a cohort of HCC patients found that follow-up imaging during the first year after therapy should be the most frequent. 77
The optimal imaging frequency during treatment can be estimated mathematically using PFS curves for various cancers and therapies. By calculating the PFS “half-life,” imaging intervals can be tailored to achieve a desired threshold of progression-free patients, with more frequent imaging for faster-progressing cancers. 78 However, it is important to recognize the limited clinical relevance of PFS as an endpoint, illustrating the urgent need for further randomized studies focused on evaluating outcomes that are truly meaningful to patient care such as OS and QoL. Clinical and biochemical assessments should take precedence whenever possible to reduce overreliance on imaging.
Radiologists and oncology care teams frequently face the challenge of differentiating disease progression from therapy-related changes (pseudoprogression). A “flare phenomenon” is a well-recognized occurrence in nuclear medicine bone scan follow-ups, where treated lesions may appear more conspicuous or new lesions seem to emerge, mimicking disease progression. To mitigate this issue, the Prostate Cancer Clinical Trial Working Group 3 recommends extending bone scan follow-up intervals to 16 weeks instead of the traditional 8 weeks. 79 Pseudoprogression is also a common challenge in brain tumour follow-up MRI examinations. A recent meta-analysis highlights the utility of amino acid PET tracers, which can effectively distinguish pseudoprogression from true disease progression, potentially reducing the need for costly follow-up imaging and invasive procedures. 80
Finally, some cancer therapies can cause cardiotoxicity, requiring ongoing monitoring of cardiac function during therapy. The commonly used for surveillance modalities and their associated sustainability considerations are summarized in Table 3.
Commonly Used Surveillance Modalities for Chemotherapy-Induced Cardiomyopathy (CIMP) With Specific Considerations for Environmental Sustainability. 83
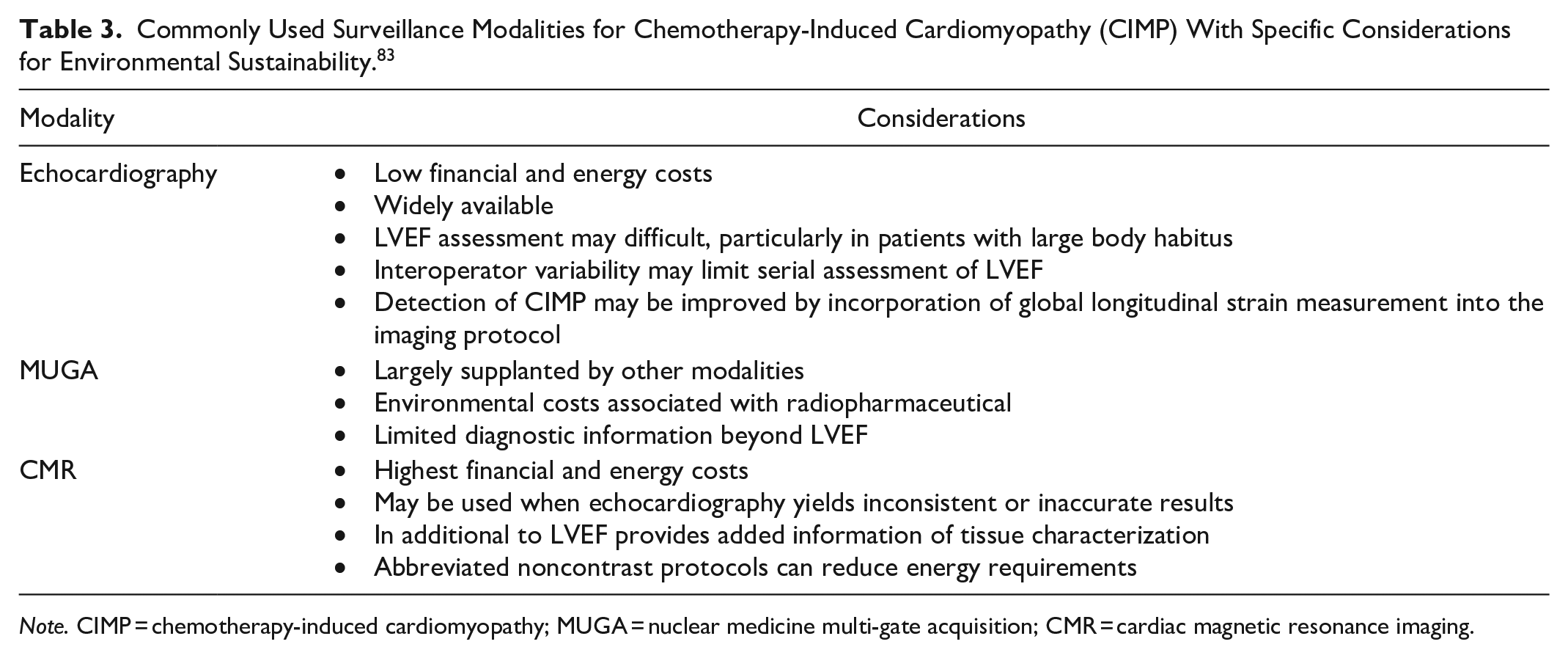
Note. CIMP = chemotherapy-induced cardiomyopathy; MUGA = nuclear medicine multi-gate acquisition; CMR = cardiac magnetic resonance imaging.
Post-Treatment Surveillance
Long-term surveillance in cancer survivors may involve clinical, biochemical, or imaging examinations. Because disease recurrence may occur years or even decades following treatment, a judicious follow-up approach should be adopted. Follow-up strategies should be tailored to the individual patient, considering functional status and competing mortality risks. Earlier knowledge of disease recurrence is unlikely to improve patient-relevant outcomes such as OS and QoL in many situations. For example, a large colorectal cancer study found no difference in OS between high-frequency (semi-annual) and low-frequency (12- and 36-month) CT follow-up groups. 81 Similarly, a study in ovarian cancer patients in remission demonstrated no survival benefit from routine CA-125 surveillance compared to clinical follow-up alone. 82 For most asymptomatic breast cancer survivors, follow-up imaging beyond annual mammography has not demonstrated improved OS or QoL. 83
Conclusion and Future Directions
Further research is needed to clarify the role of supplementary modalities in breast cancer screening, evaluate the role of AI in risk stratification and cancer detection, optimize imaging intervals in treatment monitoring and long-term surveillance (with a focus on patient-relevant outcomes), further validate the role of CEUS and abbreviated MRI protocols for liver imaging, determine the optimal clinical role of novel PET tracers (taking into account the substantial associated energy use), and develop more sustainable and resilient radiopharmaceutical supply chains.
In summary, radiologists and cancer care teams face the dual challenges of rising cancer prevalence, which drives increased demand for medical imaging, and the significant environmental impacts of imaging, which can adversely affect cancer care outcomes. Nevertheless, numerous opportunities exist to mitigate these impacts across the cancer care continuum.
Footnotes
Acknowledgements
We thank Dr. Caroline Mariano and Dr. Shilo Lefresne of BC Cancer for their valuable input from a clinical oncology perspective.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MJB: Co-Chair CAR Sustainable medical imaging committee; Co-Chair BC Cancer Planetary Health Unit; Doctors of BC grant for CT energy use study. The remaining authors above have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
