Abstract
The Canadian Association of Radiologists (CAR) Endometriosis Working Group was tasked with providing guidance and benchmarks to ensure the quality of technique and interpretation for advanced imaging modalities associated with diagnosing endometriosis. Advanced pelvic ultrasound is essential in diagnosing and mapping pelvic endometriosis, but pelvic MRI serves as an excellent imaging tool in instances where access to advanced ultrasound is limited, or an alternative imaging modality is required. Despite the known utility of MRI for endometriosis, there is no consensus on imaging protocol and patient preparation in Canada. To improve patient care and support excellence in imaging, the Working Group has developed recommendations for the use of pelvic MRI to assess for endometriosis with an aim to standardize MRI technique for use in both community and academic practices across Canada. The guidelines provide recommendations regarding imaging technique and patient preparation for pelvic MRI, along with suggestions for structured reporting of pelvic MRI for endometriosis.
Introduction and Background
Endometriosis is a common condition, affecting approximately 10% of reproductive-age individuals assigned female at birth in Canada, with symptoms such as chronic pelvic pain and infertility negatively impacting the quality of life.1,2 Diagnosing endometriosis can be challenging as the disease often presents with non-specific or variable symptoms, can involve multiple organ systems, and conventional physical examination may be of limited diagnostic value. Although laparoscopy is considered the gold standard for diagnosis, it is invasive and is limited in the assessment of subperitoneal structures as well as obliterated peritoneal spaces. Therefore, there has been a growing movement toward non-invasive imaging assessment of pelvic endometriosis to evaluate the extent of the disease, to help optimize operative/management planning, and improve patient outcomes.3,4
Advanced pelvic ultrasound (US) plays an essential role in diagnosing and mapping pelvic endometriosis. 5 In regions where the availability of specially-trained professionals to perform advanced pelvic ultrasound is lacking, in cases where the use of ultrasound is limited (e.g. inability to perform transvaginal ultrasounds), the area being investigated is outside the field of view of ultrasound, or an alternative modality is required to work up an ultrasound finding (e.g., there is negative/abnormal sliding sign but a full advanced pelvic ultrasound could not be performed, et cetera), pelvic MRI serves as an excellent alternative imaging tool for diagnosis and mapping of pelvic endometriosis. 6 It can address the varying clinical presentations, as well as provide the benefits of reproducibility, non-invasiveness, and observer independence.
In the last few years, many societies have developed recommendations and guidelines relevant to the role of imaging in endometriosis.
7
For example, the
Despite the known utility of MRI for endometriosis, there is no consensus on imaging protocol and patient preparation across Canada. This lack of uniform technique can lead to suboptimal evaluation and reduced inter-reader agreement. To improve patient care and support excellence in imaging, the Canadian Association of Radiologists (CAR) Endometriosis Working Group has developed recommendations for the use of pelvic MRI to assess for endometriosis with an aim to standardize MRI technique for use in both community and academic practices across Canada. The guidelines were developed by the Working Group based on an extensive scoping review of existing literature, followed by a Delphi process to achieve consensus on imaging technique and patient preparation recommendations for pelvic MRI. The final practice guidelines detail these recommendations and include suggestions for endometriosis MRI structured reporting.
Methods
A comprehensive scoping review was conducted by 2 of the authors (EP and AS) to determine the current state of evidence for pelvic MRI protocolling and patient preparation in cases of endometriosis. A literature search was conducted in Pubmed using the search strategy “((endometriosis[MeSH Terms]) OR (endometriosis)) AND (mr imaging[MeSH Terms] OR (MRI))” to October 2023. Two reviewers (EP and AS) independently screened the title/abstracts and any conflicts were resolved by consensus opinion. The full text of records identified as potentially relevant were further reviewed by the 2 reviewers with data extracted for the purposes of the scoping review. Studies that described an MRI protocol for endometriosis in detail were included in the review. Results from the literature search are summarized in the PRISMA diagram (Figure 1).
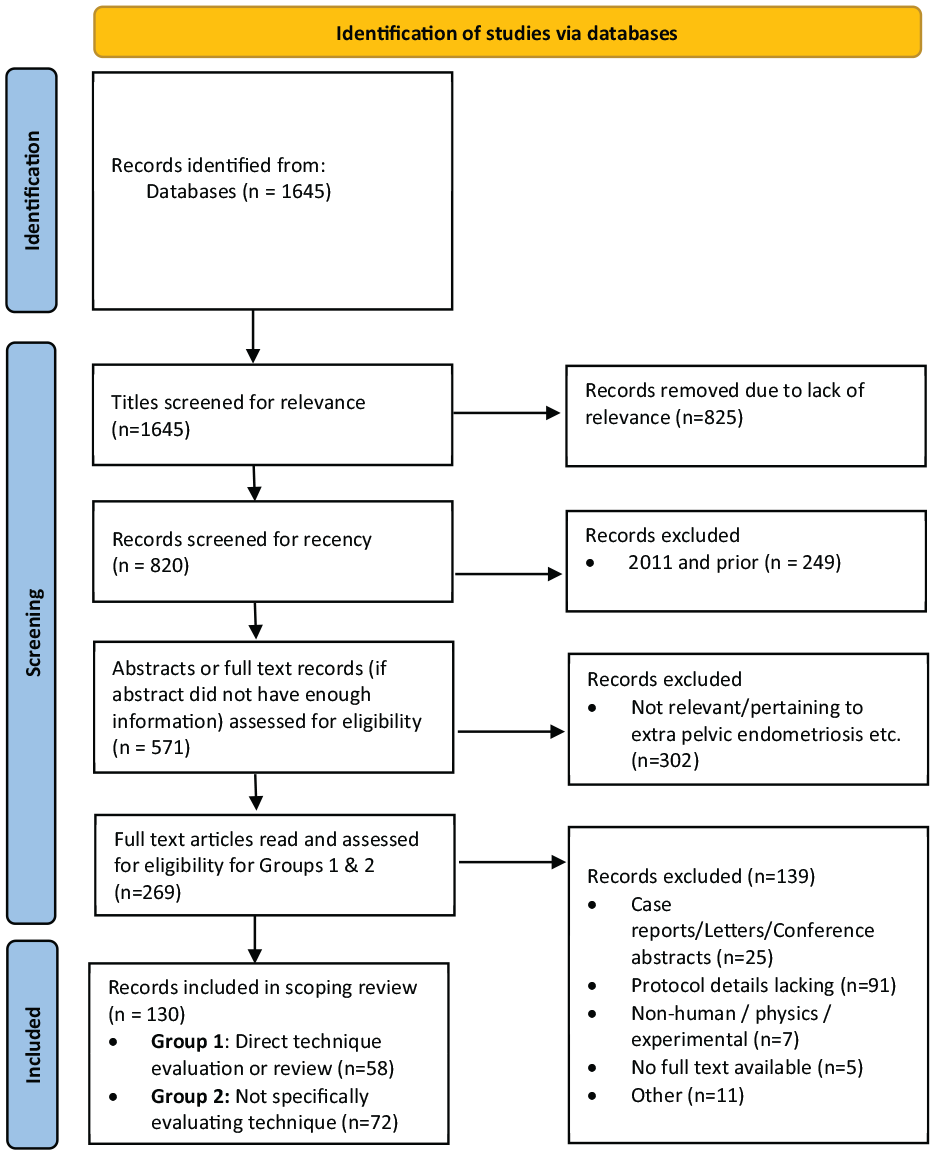
PRISMA flowchart—Literature search #1.
This evidence was used to inform the recommendations where possible, with expert consensus applied when the published evidence was insufficient. A cut-off of 70% agreement across studies was selected to mean strong evidence “for” or “against” a protocol/sequence or patient preparation, and anything <30% agreement across studies was deemed insufficient evidence to make a recommendation for or against. For recommendations with 30% to 69% agreement, a Delphi process was employed to achieve expert consensus for or against each scenario. An exception was made in a few cases where the level of agreement in the scoping review did not reach the 30% to 69% cut-off to gauge the working group opinion regarding the situational application of the following: the use of endoluminal gel, 3D T2 sequences, and diffusion weighted sequences.
The Working Group, composed of 10 abdominal radiologists who are experts in endometriosis imaging, were asked to complete 2 survey rounds to rate the proposed recommendations using the online survey tool SurveyMonkey®. Cut-offs for agreement and disagreement were previously established and communicated to all Working Group members before beginning the Delphi process. Recommendations were rated on a 7-point Likert scale, such that (1) Strongly Disagree; (2) Disagree; (3) Somewhat Disagree; (4) Neither Agree nor Disagree; (5) Somewhat Agree; (6) Agree; and (7) Strongly Agree.
Working Group members provided anonymous responses and feedback that was reviewed and evaluated at the end of Round 1. Statements rated 6.0 to 7.0 by ≥70% by the Working Group were determined to reach a consensus agreement in Round 1, and statements rated 1.0 to 2.9 by ≥70% by the Working Group members were reported as statements with insufficient evidence to make a recommendation for or against the proposed sequence or preparation. Statements rated 3.0 to 5.9 were included in Round 2 of the Delphi and incorporated suggestions for rewording or rephrasing, as indicated by the anonymous comments submitted by the Working Group members in Round 1. Working Group members were again asked to rate the revised statements in Round 2. The same cut-offs for agreement and disagreement were applied to Round 2 of the Delphi. The data was compiled, and the working group met to discuss the full set of recommendations to ensure broad consensus on the possible practical and policy-related implications. The final recommendations are presented in this practice guideline (Table 1).
Summary of the Recommendations Based on ≥70% Agreement Following a Scoping Review or Achieved Consensus Among Experts.

Recommendations based on scoping review. **Recommendations following Round 1 of the Delphi process; all other recommendations were determined following Round 2 of the Delphi process. †Recommendation based on both the scoping review and from the Delphi process.
Image Acquisition
For a summary of image acquisition parameters see Appendix A; for selected imaging examples, see Appendix B.
1.5 T Versus 3 T Scanners
MRI for the evaluation of pelvic endometriosis can be performed using either a 1.5 T or 3 T strength magnet. Most studies (54%) included in the scoping review used a 1.5 T scanner, 15% used 3 T scanner, 20% used both, and the remainder did not specify. This may be in part related to equipment availability rather than a specific preference for 1.5 T versus 3 T. While there are several studies that have demonstrated good performance of 3 T scanners for pre-operative deep endometriosis localization,13-15 there is a paucity of literature directly comparing the performance of 1.5 T versus 3 T.
3 T scanners have improved signal-to-noise (SNR) compared to 1.5 T, increasing contrast and spatial resolution, providing a more detailed depiction of pelvic anatomy and improve conspicuity of smaller deep endometriosis nodules. Increased field strength also allows for faster image acquisition, which can reduce patient discomfort and allow flexibility to include additional sequences if needed.
1.5 T scanners are preferred in cases where there is concern about susceptibility or motion artifacts (which are comparatively accentuated with 3 T). 16 Magnetic field inhomogeneity can also occur on 3 T scanners with larger fields of view (e.g., incomplete fat saturation in patients with larger body habitus). A 1.5 T scanner may be preferred for medical device compatibility.
Coil
The use of a phased array coil placed over the pelvis with the patient supine is the current standard for pelvic MRI and is also specifically recommended for endometriosis evaluation to increase the SNR and improve spatial resolution. Flexible coils are preferred to rigid coils for their improved SNR and adaptability to a variety of body habitus types. 17 In the scoping review, most studies (70%) indicated using a phased array coil, while the remaining studies did not specify. Previous studies have investigated the use of endorectal coils18-20; however, they are cumbersome in practice, have an increased cost, and limited patient tolerance. In the opinion of the Working Group, the benefits do not outweigh the downsides; thus, the use of an endorectal coil is not recommended.
Standard Orthogonal T2 Sequences
Orthogonal sagittal and axial T2 sequences without fat saturation are essential for evaluating pelvic endometriosis. Most deep endometriosis nodules are characterized by their typical T2 hypointense fibrotic signal and are best identified when contrasted against the hyperintense signal of visceral fat. Previous consensus guidelines have also recommended at least 2 T2-weighted orthogonal planes, including axial and sagittal, with mixed opinions on inclusion of a coronal T2 plane.21,22 In the scoping review, axial T2-weighted images were included in 94% of studies, sagittal in 92%, and coronal in 54%.
The sagittal T2-weighted sequence is the optimal plane for visualizing the pelvis as it is divided into the 3 compartments (anterior, middle, posterior) used for systematically localizing sites of endometriosis involvement, as recommended by the Society of Abdominal Radiology and based on the 2016 International Deep Endometriosis Analysis consensus guidelines.23,24 It is also essential for assessing uterine position, endometrial and junctional zone thickness, the anterior and posterior cul-de-sacs, and rectosigmoid and vaginal nodules due to the craniocaudal orientation of these structures. The axial plane is useful for evaluating the ureters, round ligaments, and often the uterosacral ligaments, as they are usually oriented perpendicular to the transverse plane.
A coronal T2-weighted sequence is not essential but is recommended. It provides a useful cross reference when evaluating the uterine junctional zone (particularly if the uterine long axis is horizontal), sciatic nerves, pelvic sidewall, uterosacral ligaments, pelvic small bowel/appendix, and depth of invasion of high rectosigmoid nodules. Kissing ovaries—in which both ovaries are medialized by adhesions—are a characteristic feature of advanced endometriosis and are also more readily appreciated in the coronal plane. It is also helpful when assessing the tubular configuration of hemato/hydrosalpinges to distinguish them from endometriomas.
Oblique T2
The uterosacral ligaments support the uterus in the pelvis, extending from the cervix to the anterior sacrum, and are commonly involved in deep endometriosis. The European Society of Urogenital Radiology guidelines recommend a thin slice axial oblique T2 sequence oriented in a cross-sectional plane through the uterosacral ligaments (USL) that was based on a 2011 study by Bazot et al.23,25 In practice, the quality of the images acquired usually allows identification of the USLs on T2 axial sequences (approximating the mesorectal fascia) and acquiring a separate oblique-axial plane is generally not required). However, it could be optionally included if the USLs are difficult to visualize, such as in patients with reduced visceral fat, obscuration by distended bowel, or significant distortion from fibrosis. Rectosigmoid/mid-rectal deep endometriosis nodules may also be better assessed in cross-section in this plane.
The standard coronal plane can be sufficient to assess the uterine body in cross-section (for junctional zone evaluation or for distal sigmoid nodules which are often oriented parallel to the uterine body) in cases where the uterus has a predominantly transverse lie. However, if the uterine body position is more axially oriented, there may be value in obtaining an oblique coronal plane oriented perpendicular to the long axis of the uterus. The SAR guidelines consider this as optional but could potentially replace the conventional coronal plane. 22 As it may be difficult to know the uterine orientation in advance and whether an oblique plane would simply just replicate an existing axial or coronal sequence, we share the opinion that this is an optional sequence, and its inclusion would depend on local practice.
In the scoping review, an oblique T2-weighted (oriented either to the USLs or uterus) was acquired in only 23% of studies, considered optional in 3%, and either not acquired or not specified in the remaining 74%.
3D T2
Given the number of T2 planes that are potentially acquired in most endometriosis protocols, it has been proposed that a single volumetric 3D T2 sequence (such as CUBE, VISTA, SPACE) could be acquired and reformatted in multiple planes, replacing conventional T2 sequences. In theory, this could reduce overall scan time and allow for thinner slice reconstructions. However, optimizing the 3D sequence to have comparable image quality and spatial resolution to conventional T2 sequences, without an overly lengthy acquisition time, remains a challenge in clinical practice. 26
Bazot et al found that 3D CUBE resulted in significantly reduced image quality and worsened motion artifact compared to conventional T2 sequences, although diagnostic accuracy for deep endometriosis was similar. 25 Other studies have found comparable image quality between 3D and 2D T2 for deep endometriosis or female pelvic imaging in general.26,27 In our scoping review, 3D T2 was utilized in 15% of studies.
In the consensus group’s experience, the image quality of 3D T2 is highly variable and scanner-dependent; in many cases not comparable to conventional T2. We thus consider this optional and should not replace conventional T2 sequences if the image quality is not comparable.
T1/T1 Fat-Saturated Sequences
The use of a T1 fat-saturated/suppressed sequence is essential for endometriosis assessment. This sequence is used to increase the conspicuity of T1 hyperintense haemorrhagic foci that are often seen with deep endometriosis lesions, adenomyosis, and to identify hematosalpinx.28-31 The sequence can also differentiate T1 hyperintense ovarian endometriomas from macroscopic fat within dermoid cysts. An axial T1 FS plane is recommended as it is included in most standard pelvic protocols and is sufficient for the abovementioned indications.
The Dixon method is a technique of fat suppression using chemical shift and mathematically combines the acquired in and out of phase T1 sequences to obtain post-processed fat and water maps, the latter equivalent to a conventional T1 fat-saturated sequence with often more uniform fat suppression.32,33 All 4 sets of images (T1 in-phase, T1 out-of-phase, fat map, water map) can be generated with a single breath hold. The Dixon method has been gaining popularity in the past decade, with all the major MRI vendors now offering some variation of a Dixon sequence. If available, the Dixon water map can replace the conventional axial T1 FS sequence as alluded to above. This is also in agreement with the ESUR guidelines. 21
A sagittal T1 FS sequence is not essential but recommended, as it can be useful for cross-referencing the axial plane and more confidently localizing haemorrhagic foci in the anterior and posterior cul-de-sac. The SAR guidelines recommend T1 FS imaging in at least 2 planes, although they do not specify which planes. 22 This consensus group’s opinion is that a sagittal sequence can be omitted if reducing scan time and protocol complexity is a priority, as a single axial plane will suffice in most cases.
A T1 non fat-saturated axial sequence is included in many routine pelvic MRI protocols to characterize macroscopic fat in dermoid cysts. This sequence is also included as part of the Dixon set of sequences as the T1 in-phase component. If Dixon is unavailable, this can be acquired optionally as it is not specifically useful for endometriosis assessment but is beneficial for other gynaecologic indications such as the characterization of dermoid cysts.
In the scoping review, axial T1 FS was included in the protocol of 82% of studies, sagittal T1 FS in 37%, and axial T1 (non-fat saturated) in 71%.
Diffusion-Weighted Imaging
Diffusion-weighted imaging (DWI) sequences are also included in most routine pelvic protocols, typically acquired in the axial plane with 2 to 4 b-values and a generated apparent diffusion coefficient (ADC) map. In the scoping review, a DWI sequence was acquired in 22% of studies, optional in 5%, and either not specified or not acquired in the remaining 73% of studies.
It is well known that benign endometriomas can demonstrate diffusion restriction,31,34,35 while there is little literature on how deep endometriosis nodules behave on DWI. From what has been published, and from our own experience, deep endometriosis nodules will often demonstrate low ADC values due to the T2-“blackout” effect. This effect arises from an intrinsically low T2 signal due to the predominantly fibrotic composition and no significant hyperintensity on high b-value DWI (ie, only mild or no true restricted diffusion). An exception to this may be the presence of true diffusion restriction localized to intralesional haemorrhagic components.22,36,37
While DWI is not useful for the detection of deep endometriosis itself, it can be helpful for detecting diffusion restriction associated with malignant degeneration, a rare complication of both ovarian endometriomas and extra-ovarian endometriosis.38-41 While assessment may be confounded by benign endometriomas that can also restrict diffusion (as noted earlier), a localized DWI abnormality correlating to suspicious nodularity that is detected on other sequences should raise concern for malignant transformation. 42 DWI may also be useful for differentiating deep endometriosis from other malignancies or detecting incidental malignancy, for example, Busard et al found that bowel deep endometriosis nodules demonstrated lower signal intensity on high b-value DWI compared to colorectal carcinoma. 43
DWI is also helpful for lymph node detection and localizing the ovaries, which are usually hyperintense on high b-value DWI and can be challenging to find when they are smaller in volume (such as in patients on oral contraceptives) or obscured by the anatomic distortion that can co-occur with deep endometriosis.
The consensus group recommends the use of an axial DWI sequence, though not essential, as part of an endometriosis protocol, particularly when gadolinium is not used. The axial DWI sequence can compensate for the otherwise reduced sensitivity for alternative diagnoses or malignant degeneration on a non-contrast study.
Gadolinium
Like DWI, the primary reason for including gadolinium would be to detect malignant degeneration either in relation to or incidental to endometriosis. However, the population most impacted by endometriosis—namely premenopausal females—are, in general, at lower risk of malignancy compared to older demographic groups. 44 False positives can also occur with polypoid forms of endometriosis and endometrioma decidualization, both of which enhance and can mimic malignancy.31,45
Contrast has otherwise limited utility in endometriosis-specific imaging as most deep endometriosis nodules progressively enhance similarly to fibrotic tissue and it has not been shown to improve the detection of nodules compared to the routine non-contrast sequences.46,47 Occasionally, contrast may improve confidence when a rectosigmoid endometriosis nodule is difficult to distinguish from stool content.
The benefits of omitting gadolinium include reduced cost and scan time, no risk of allergic reaction or sensitivity, the ability to perform studies during “off” hours (i.e. does not require on-site radiologist supervision) and alleviates public concerns regarding long-term gadolinium deposition.
Given the risk-benefit considerations, the consensus group agrees that the use of contrast is optional. Contrast was included in the protocols of 41% of studies in the scoping review. Alternative strategies include using contrast in select patient populations, for example:
Patients over 40 years of age (as ovarian cancer risk increases more steeply in this demographic 48 ).
Patients with a known adnexal mass/endometrioma requiring characterization.
If a finding on a non-contrast study is felt to require contrast for characterization, the patient could be recalled for a limited follow-up study.
When contrast is used, pre and post gadolinium subtracted images are useful in detecting areas of true enhancement within endometriomas and in other areas of concern.
Susceptibility Weighted Imaging
Susceptibility-weighted imaging (SWI) increases the sensitivity for the detection of chronic haemorrhagic foci and can theoretically be used to improve the characterization of deep pelvic endometriosis. Most studies evaluating the use of SWI sequences have been in the context of differentiating endometriomas from other ovarian cysts.49-51 There is very limited evidence that SWI may improve sensitivity for extra-ovarian endometriosis.52,53 However, there can be several other causes of increased susceptibility in the pelvis such as bowel gas, foreign bodies (such as IUD), and calcifications that can confound interpretation. 22 Only 11% of studies in the scoping review used a SWI sequence, thus it fell below the threshold for recommendation; however, further study may change this in the future.
Patient Preparation
For a summary of patient preparation recommendations, see Appendix C.
Use of Antiperistaltic Agents
Small bowel peristalsis causes motion artifact which can degrade image quality and limit the detection of subtle endometriosis lesions. 22 It is also important to decrease uterine peristalsis, which can result in apparent thickening of the junctional zone. This can be falsely misinterpreted as adenomyosis, a common coexisting condition. 54 62% of studies in the scoping review used an antiperistaltic agent. Since the review agreement percentage was below 70%, this recommendation is based on the group consensus after the first Delphi round of. Hyoscine butylbromide (HBB, Buscopan, Sanofi-Aventis) is an anticholinergic agent that can be administered IM or IV (20 mg). It is administered shortly (i.e. within 15 minutes) before starting the MRI exam to maximize the effective duration. IV HBB acts rapidly and is more reliable than the IM route, which can take longer to act, with both routes comparable in duration of effect. 55 Alternatively, or in cases where HBB is contraindicated, 1 mg of glucagon can be administered IM, IV or subcutaneously, although IV administration is preferred.
Menstrual Cycle Timing
As endometriosis is hormonally sensitive, it is theoretically possible that it may change in conspicuity depending on the menstrual cycle timing. Previously published protocol recommendations have explicitly not timed the MRI study based on the menstrual cycle phase, as there is no strong evidence to support this practice and it would be logistically challenging as many patients may have irregular or unpredictable menstrual cycles.56,57 In a study by Botterill et al, no statistically significant difference in the imaging appearance of endometriosis in menstruating versus non-menstruating scans was found. 58 The majority (92%) of studies in the scoping review did not specify menstrual cycle timing as part of patient preparation for imaging of endometriosis. Out of the 8% that did specify menstrual cycle timing, most were performed in the first half of the menstrual cycle with one review article specifically recommending against MRI during menstruation. 59
Patient Position
Pelvic MRI is generally performed with patients in the supine position. Although only 15% of studies in the scoping review specified using “supine position” it is presumed that all used this position by convention. A few studies discussed the benefit of prone positioning for patients with claustrophobia.59,60 Regardless, supine position should be the default, though prone can be helpful if patients are claustrophobic. Alternatively, scanning feet first in supine position may also be helpful for claustrophobia. 61
Bladder Distension
In 31% of studies in the scoping review, bladder distention was utilized or recommended. An empty bladder can decrease the conspicuity of bladder nodules, though an overdistended bladder can distort pelvic anatomy by effacing the vesicouterine pouch and can be uncomfortable for the patient. Moderate bladder distention can be achieved by refraining from voiding for 1 hour prior to the scan. It is well tolerated by patients, can improve detection of endometriosis foci, improves visibility of the vesicouterine pouch 62 and can help displace bowel loops away thereby decreasing bowel related artifacts. 63 Adequate bladder distention also helps delineate the distance between endometriosis implants and ureteral orifices which can help with operative planning. 64 Providing a written explanation to patients about why it is helpful prior to pelvic MRI may improve compliance.
The Use of Endoluminal Gel
Opacification of the rectum and vaginal canal with ultrasound gel (sterile or non-sterile) has been recommended by some studies. Luminal distension with aqueous gel separates the normally closely opposed walls of these structures and its T2 hyperintensity contrasts with the low T2 signal of endometriosis nodules, improving conspicuity.65,66 In one study by Chassang et al, endoluminal gel improved the sensitivity (from 63% to 81.7%) for the detection of deep pelvic endometriosis. The use of vaginal gel was particularly useful for assessing deep endometriosis affecting the rectovaginal septum, uterosacral ligaments and posterior vaginal fornix. 67 Hughes et al described the protocol in detail for vaginal gel insertion by self-administration which was preferred by patients as it was relatively comfortable without needing sedation or analgesia. 60 cc of warmed aqueous ultrasound gel was used in a pre-filled bladder syringe with a narrow beak. Warming prior to insertion can be helpful as it thins the consistency of the gel and decreases artifact-causing air bubbles. 68 Gel viscosity can also be reduced by dilution with water; with a total volume of 50 to 100 cc for vaginal use and 100 to 150 cc for rectal use suggested. 69
Other studies did not find significant improvement in endometriosis detection with the use of endoluminal gel. 46 As per the scoping review, only 21% utilized rectal gel distention and 24% utilized vaginal gel distention. Due to the added time and cost, patient acceptance and logistical complexities, as well as the lack of conclusive evidence to confirm its benefit in all cases, the group consensus regarding the use of endoluminal gel (vaginal or rectal) is that it is optional and not essential. It can be considered if there is specific concern for a vaginal or rectal nodule. An example protocol for endoluminal gel distension is included in Appendix D.
Abdominal Strapping
Abdominal strapping uses a broad belt tied over the abdomen and pelvis to decrease the artifact caused by respiratory motion. 62 This can help in pelvic MRI evaluation of endometriosis. 21 However, only a minority of studies (7%) in the scoping review specified the use of abdominal strapping and the use of surface coils already provides some degree of strapping. Therefore, the use of abdominal strapping is considered optional.
Bowel Preparation
Fasting for 4 hours prior to the pelvic MRI scan can decrease bowel peristalsis which can theoretically improve visibility of endometriosis lesions. However, there is limited evidence for fasting leading to improved detection of endometriosis on pelvic MRI as antiperistaltic agents are usually sufficient for reducing bowel motility.22,70 Similarly, bowel preparation using enemas has limited evidence at this time. 57 Although 51% of studies in the scoping review recommended the use of bowel preparation, the working group consensus was that it is optional. Most Canadian academic institutions do not use bowel preparations due to patient discomfort, cost, and challenges with scheduling of a limited resource. 22
Structured Reporting
For complex diseases such as endometriosis, preoperative imaging plays a crucial role in presurgical planning and patient counselling.71,72 This requires effective communication between the radiologist and the clinical team, primarily via the radiology report. Traditional narrative reports can lack clarity and depend on the radiologist’s experience and training, while structured reports are proven to enhance accuracy and reduce errors. 73 This ensures comprehensive mapping of the disease and standardized description using a universally accepted lexicon.5,71
Various societies and working groups have published structured report templates for ultrasound and MRI reporting of pelvic endometriosis.71,72 Most published templates use a compartmental model to divide the pelvis, with some variations, such as keeping the uterus and ovary separate instead of grouping them under the “middle compartment” 71 or dividing the pelvis into more than 3 compartments. 74 Each variation has its merits, but the format and order of compartments are less important than ensuring a complete anatomical review and reporting findings that may change management using a standard lexicon.
To maintain consistency with the CAR ultrasound reporting template for endometriosis, the MRI template for standardized reporting also employs a 3-compartment model (anterior, middle, and posterior), grouping the ovary and uterus under the middle compartment (see Table 2). The compartments are listed in order of location of most frequent disease (middle, posterior, and anterior). A fourth subheading specifically for endometriosis is added, titled “Additional sites of endometriosis,” to accommodate other sites not covered by the 3-compartment model.
Example Structured Reporting Template.
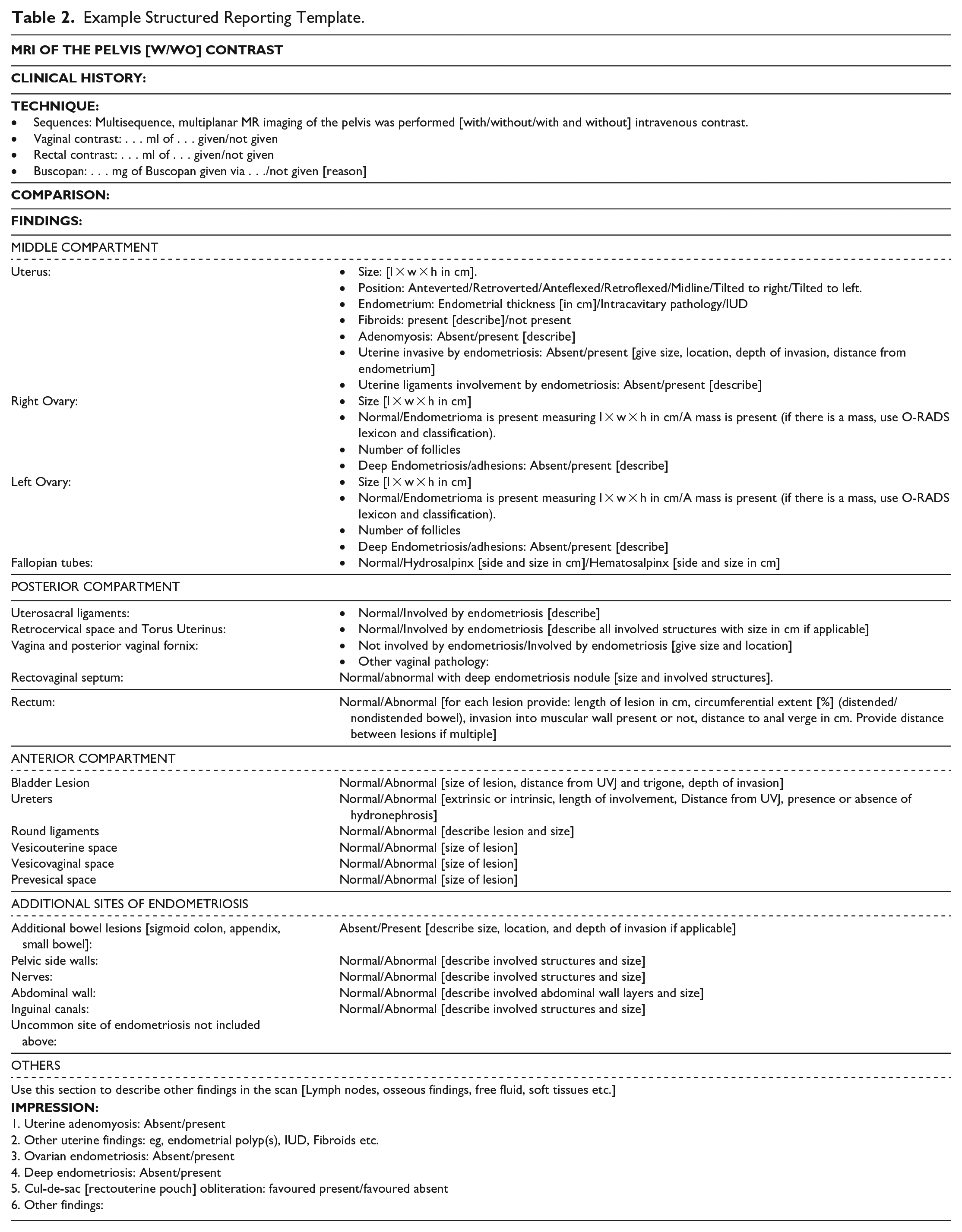
For each of these subheadings, the specific structures to evaluate and report on are listed. While the detailed imaging characteristics of deep endometriosis are not listed within the template as it is beyond the scope of this paper, a summary of key descriptors using a standard lexicon within the template is provided. For each rectal lesion, the template includes integral descriptors that should be reported by the radiologist and can modify the surgical approach or type of resection. For bowel lesions, the size and number of lesions, distance of the most caudal lesion from the anal verge, depth of invasion, and percent circumference of bowel involved (< or >50% circumferential involvement) must be described, as these factors can change the surgical approach which can range from shaving off a superficial lesion to a segmental bowel resection.71,72
When visualization of a structure is suboptimal, we suggest labelling it as “not visualized” or “suboptimally visualized” to ensure transparency and completeness. We also acknowledge that in instances of multiorgan involvement, radiologists may need to combine sections of the structured report. For instance, if a retrocervical lesion extends into the vaginal fornix/vagina, forming one lesion, combining these 2 sections may be appropriate or if the hematosalpinx is inseparable from the ovarian endometriomas the radiologist may choose to group both structures under “adnexa.” In the case of non-endometriosis related pathology, it is recommended to adhere to existing classification systems, such as ACR O-RADS™ for ovarian lesions75,76 and FIGO 77 classification for fibroids. Finally, it is recommended that this template is adjusted as per institutional preference and discussion between radiologists and clinical stakeholders.
Conclusion
MRI has become an integral modality in the non-invasive assessment of endometriosis, providing excellent soft tissue contrast for detection of endometriosis nodules and a comprehensive view of the pelvis to inform management decisions including surgical planning. The guidelines described in this paper were developed by a panel of expert radiologists with the aim of providing a standardized pelvic endometriosis MRI protocol which balances the need for optimal diagnostic image quality with efficient resource utilization and patient experience in the Canadian context.
Additionally, the group developed a suggested reporting template to facilitate radiologist evaluation, which emphasizes the common sites of involvement and aims to make communication between healthcare providers more uniform.
Footnotes
Appendix A
Summary of Recommendations for Image Acquisition Parameters.
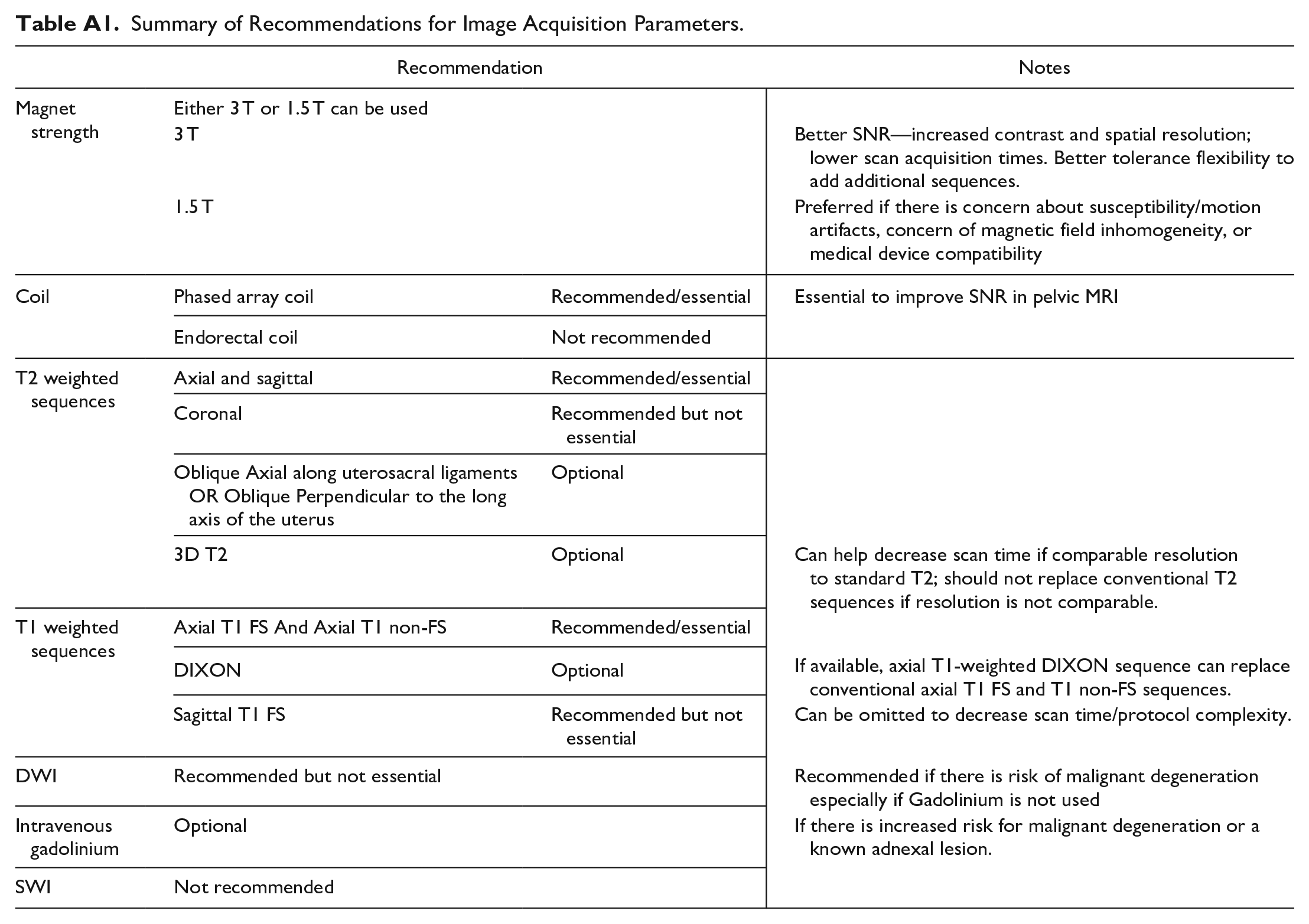
| Recommendation | Notes | ||
|---|---|---|---|
| Magnet strength | Either 3 T or 1.5 T can be used | ||
| 3 T | Better SNR—increased contrast and spatial resolution; lower scan acquisition times. Better tolerance flexibility to add additional sequences. | ||
| 1.5 T | Preferred if there is concern about susceptibility/motion artifacts, concern of magnetic field inhomogeneity, or medical device compatibility | ||
| Coil | Phased array coil | Recommended/essential | Essential to improve SNR in pelvic MRI |
| Endorectal coil | Not recommended | ||
| T2 weighted sequences | Axial and sagittal | Recommended/essential | |
| Coronal | Recommended but not essential | ||
| Oblique Axial along uterosacral ligaments OR Oblique Perpendicular to the long axis of the uterus | Optional | ||
| 3D T2 | Optional | Can help decrease scan time if comparable resolution to standard T2; should not replace conventional T2 sequences if resolution is not comparable. | |
| T1 weighted sequences | Axial T1 FS And Axial T1 non-FS | Recommended/essential | |
| DIXON | Optional | If available, axial T1-weighted DIXON sequence can replace conventional axial T1 FS and T1 non-FS sequences. | |
| Sagittal T1 FS | Recommended but not essential | Can be omitted to decrease scan time/protocol complexity. | |
| DWI | Recommended but not essential | Recommended if there is risk of malignant degeneration especially if Gadolinium is not used | |
| Intravenous gadolinium | Optional | If there is increased risk for malignant degeneration or a known adnexal lesion. | |
| SWI | Not recommended | ||
Appendix B
Appendix C
Summary of Recommendations for Patient Preparation for MRI for Endometriosis.
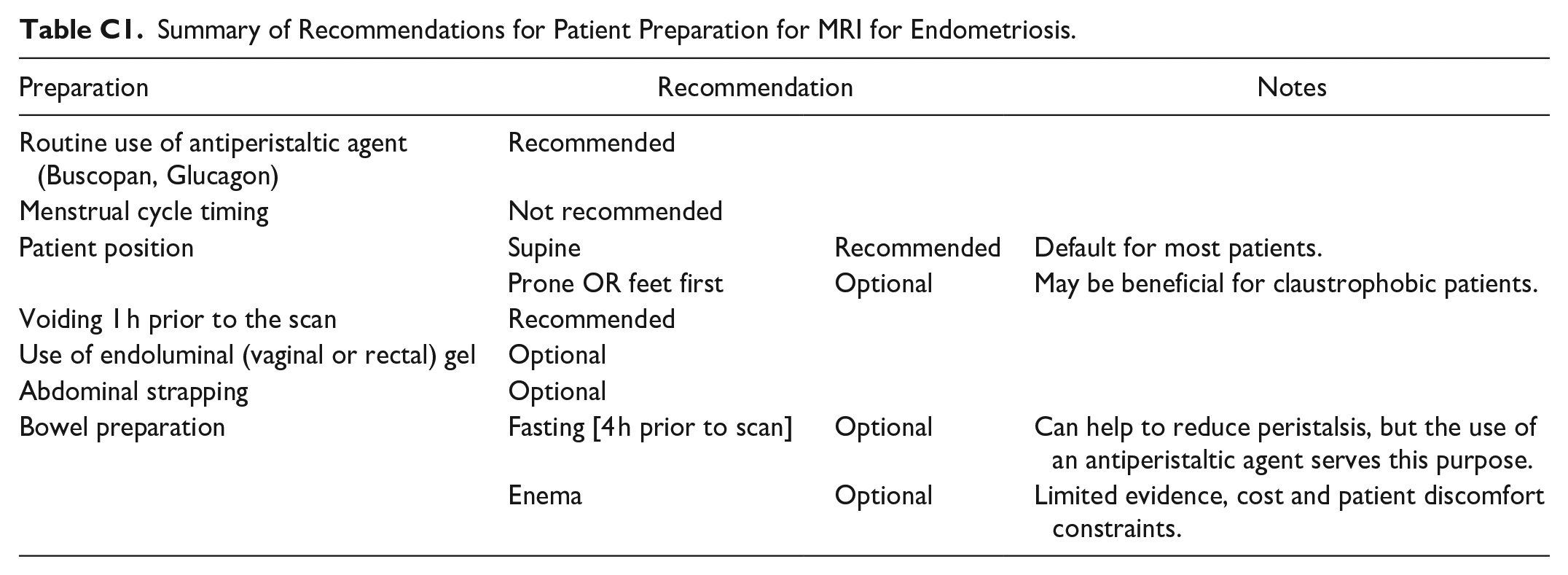
| Preparation | Recommendation | Notes | |
|---|---|---|---|
| Routine use of antiperistaltic agent (Buscopan, Glucagon) | Recommended | ||
| Menstrual cycle timing | Not recommended | ||
| Patient position | Supine | Recommended | Default for most patients. |
| Prone OR feet first | Optional | May be beneficial for claustrophobic patients. | |
| Voiding 1 h prior to the scan | Recommended | ||
| Use of endoluminal (vaginal or rectal) gel | Optional | ||
| Abdominal strapping | Optional | ||
| Bowel preparation | Fasting [4 h prior to scan] | Optional | Can help to reduce peristalsis, but the use of an antiperistaltic agent serves this purpose. |
| Enema | Optional | Limited evidence, cost and patient discomfort constraints. | |
Appendix D
Endoluminal Gel Administration.

| Vaginal gel | Rectal gel |
|---|---|
| 30-50 cc of ultrasound gel via 60 cc catheter tip syringe. To be self-administered by the patient. | Inform the patient that they may experience an urge to defecate. |
Acknowledgements
The authors acknowledge the assistance and contributions of the other members of the CAR Endometriosis Working Group: Dr. Mathew Leonardi. The authors would also like to thank the members of the Canadian Association of Radiologists who took the time to provide their feedback and peer review during the drafting of these guidelines.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Unrelated to the recommendations contained herein: Dr. Basma Al-Arnawoot has delivered talks for Bayer; Dr. Caroline Reinhold is the VP of Oncology for Spire Sciences Inc.; Dr. Ania Kielar is President of the Canadian Association of Radiologists.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
