Abstract
Lung cancer is the second-most diagnosed cancer and the leading cause of cancer-related death in Canada. The updated CAR/CSTR Practice Guideline on CT Screening for Lung Cancer reflects advancements in evidence since the 2016 guideline, including findings from the NELSON trial and preliminary data from multiple provincial lung cancer screening programs, and aims to support Canadian diagnostic imaging departments in implementing organized lung cancer screening programs. The guideline emphasizes screening with the use of low-dose CT (LDCT) to reduce lung cancer mortality in appropriately selected individuals with increased risk of lung cancer, using eligibility criteria based on risk prediction models such as the PLCOm2012. It outlines training requirements for radiologists, standardized CT and reporting protocols, quality assurance measures, and the integration of AI tools for nodule risk stratification. The document also highlights emerging areas for investigation, including the potential for biennial screening and equitable access to programs across Canada.
Introduction
Lung cancer is the second-most diagnosed cancer and is the leading cause of cancer related death in Canada. 1 In 2011, the National Lung Cancer Screening Trial (NLST) 2 demonstrated a significant decrease in lung cancer mortality in the population screened with low-dose CT (LDCT). Since the initial publication of the Canadian Association of Radiologists (CAR) Guide on CT Screening for Lung Cancer in 2016, 3 the Dutch-Belgium Lung Cancer Screening Trial (NELSON) has also been published, 4 providing additional data in support of lung cancer screening.
This updated guide produced by the CAR and the Canadian Society of Thoracic Radiologists (CSTR) will address the advancement in knowledge since the publication of our 2016 guide. It is intended for radiologists, medical radiation technologists, and administrators working in diagnostic imaging departments in Canada. This guide is not intended to replace evidence-based recommendations or influence referring providers. Instead, it aims to assist diagnostic imaging departments and clinics in implementing organized lung cancer screening programs and addressing individual requests for screening. It is important to note that provinces and regions may have varying resources for lung cancer screening and may need to adjust their practices accordingly. The management of patients once a diagnosis of lung cancer has been established is outside the scope of this document.
Lung cancer screening is a rapidly evolving area of medicine, and this guide will be revised as needed. There remain many unanswered questions; while large-scale randomized controlled trials offer essential data on patient outcomes including risks of screening, the comparison of some variables including comparative durations of screening, definitions of high-risk population and definitions of positive screen may never be assessed in sufficiently powered randomized trials. Rather than avoid assessing practices for which there is no or limited RCT evidence, we have reviewed the relevant literature to provide rationale for decisions.
Since 2016, several provinces have launched organized lung cancer screening programs, with others implementing pilot projects (Figure 1). Many Canadians at high-risk for lung cancer have been enrolled, leading to early-stage diagnoses and reduced morbidity and mortality. 5 Additionally, Canadian programs demonstrate higher cancer detection rates compared to U.S. data, as their inclusion criteria are better tailored to identify the highest-risk individuals (Table 1).
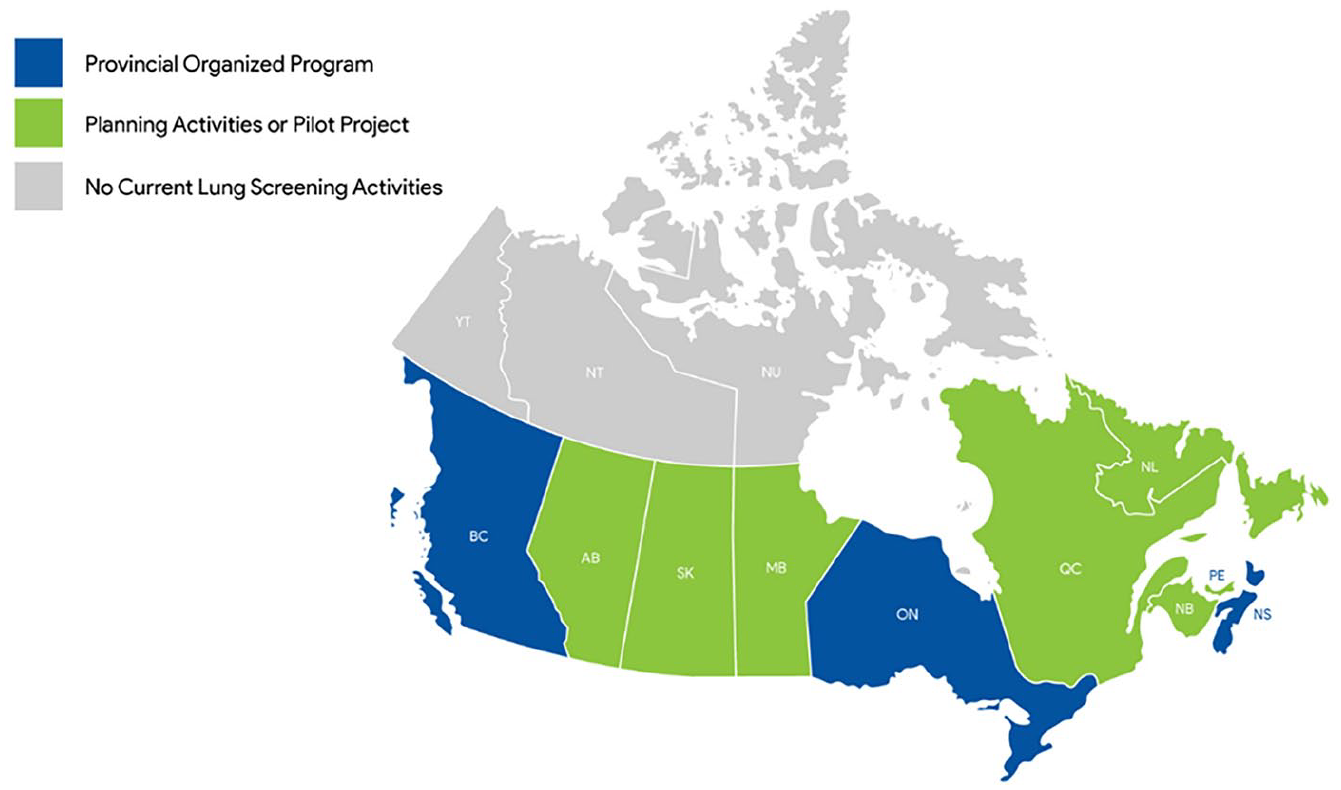
Status of lung cancer screening programs in Canada (as of November 2024).
Current Lung Cancer Screening Data.
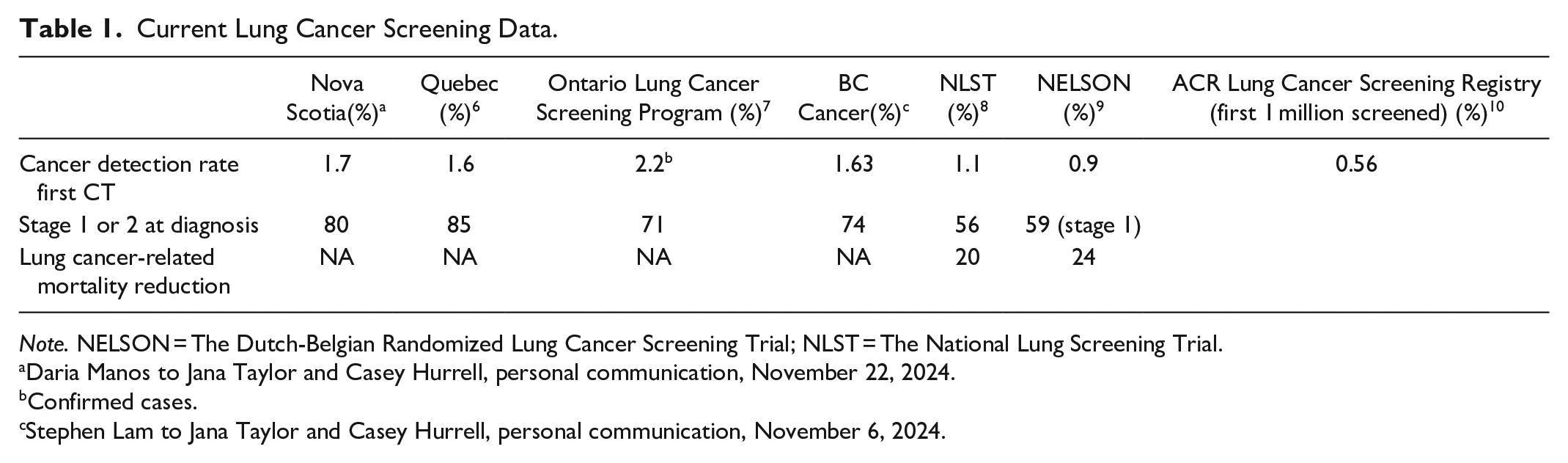
Note. NELSON = The Dutch-Belgian Randomized Lung Cancer Screening Trial; NLST = The National Lung Screening Trial.
Daria Manos to Jana Taylor and Casey Hurrell, personal communication, November 22, 2024.
Confirmed cases.
Stephen Lam to Jana Taylor and Casey Hurrell, personal communication, November 6, 2024.
The Canadian Task Force on Preventive Health Care (CTFPHC) and the Canadian Cancer Society both recommend lung cancer screening in high-risk individuals, 11 but there are variations in enrolment criteria and guidelines across Canada and in other countries. In the absence of any national or international standardized guidelines, the CAR and CSTR have provided recommendations based on the current literature and evidence-informed expert opinion.
Lung Cancer Screening Population Statistics
In 2024, it was estimated that there were 32 100 new cases of lung cancer and 20 700 Canadians died from lung cancer. 12 It is estimated that 1 in 15 Canadians are expected to be diagnosed with lung cancer in their lifetime. 1 The high rate of lung cancer mortality is due to the majority of lung cancers being diagnosed at an advanced stage (stage 3/4 = 70%) 1 that have a poor 5-year survival rate (5-year survival rate for stage 3 and stage 4 lung cancer of 17% and 4%, respectively). 1 By comparison, currently only 20% of lung cancers are diagnosed at stage 1, with an improved 5-year survival rate of 60%. 1 The absolute number of lung cancer deaths in Canada continues to increase as the population ages and increases in size, however lung cancer mortality has been decreasing since the 1990s, primarily attributed to a reduction in tobacco smoking in the Canadian population. 1
There are multiple risk factors for developing lung cancer, the greatest of which is tobacco smoking. 11 For this reason, incorporation of tobacco smoking cessation programs is recommended as part of all lung cancer screening programs. 11 Although smoking rates in Canada have decreased over the past 3 decades, smoking rates in some populations remain high. A high lung cancer incidence amongst some equity-deserving groups in Canada is multifactorial, and partially related to cigarette smoking use, and low smoking cessation rates. 13 In addition, social determinants of health, including inequitable access to health care services and health care bias is associated with diagnosis at more advanced cancer stages and poorer survival rates.13,14
Environmental exposures include radon gas, asbestos, air pollution, and exposure to second hand smoke. Radon gas is associated with lung cancer when high levels of radon are trapped in enclosed spaces (such as poorly ventilated homes or basements). Health Canada estimates 16% of all lung cancers are a result of radon gas exposure 15 and when a cigarette smoker is exposed to high levels of radon gas their lung cancer risk increases by 20%. 16 There is increasing evidence showing that air pollution is now the second largest risk factor for lung cancer. 17 Currently, there is no strong evidence to support cannabis smoking as a risk factor for lung cancer, with limited studies confounded by multiple factors. 18 E-cigarettes and vaping are relatively new products that contain numerous known carcinogenic substances. In part due to the lag time between exposure and development of lung cancer, their use as a risk factor for lung cancer is currently equivocal. 19
Summary of Current Evidence
The 2 largest and most robust lung cancer screening randomized controlled trials to date are the National Lung Screening Trial (NLST) 2 that enrolled ≥50 000 persons, aged between 55 and 74 with a ≥30 pack year history of smoking and the Dutch-Belgium Lung Cancer Screening Trial (NELSON) 4 that enrolled ≥15 000 persons, aged between 50 and 74 with a >15 cigarettes smoked daily for >25 years or >10 cigarettes smoked daily for >30 years. The NLST showed a 20% reduction in lung cancer mortality and a 6.7% reduction in all-cause mortality for 3 rounds of annual LDCT screening versus annual chest X-rays. The number needed to screen to prevent one lung cancer-related death was 320 in the NLST. The NELSON trial showed an overall 24% reduction in lung cancer mortality after 10 years of follow-up (Table 1). The NELSON data also showed even greater reduction in lung cancer mortality in women than in men, although the study was limited by a relatively low number of women enrolled in the trial.
A meta-analysis including 94 921 participants from 9 smaller randomized controlled trials other than the NLST and NELSON showed a 16% relative reduction in lung cancer mortality and a 3% relative reduction in all-cause mortality with low-dose CT screening. 20
Opportunistic Lung Cancer Screening
Opportunistic lung cancer screening involves performing CT on asymptomatic individuals on an ad hoc basis outside of organized lung cancer screening programs. These CT scans are often performed with variable technical parameters and lack systematic follow-up necessary to effectively manage detected lung nodules. 21 They may be performed on individuals who are not at sufficiently high risk of lung cancer and for whom the benefits may not outweigh the risks associated with screening. One Canadian study found that only 28% of individuals undergoing opportunistic lung cancer screening met Canadian Task Force on Preventive Health Care criteria for screening. 21 The NLST and NELSON trials provide strong evidence that lung cancer screening with specific selection criteria and standardized protocols reduces lung cancer related mortality,2,4 but there are potential harms and uncertain mortality benefit for opportunistic lung cancer screening.
Imaging centres that perform lung cancer screening must avoid screening patients who do not meet screening eligibility criteria. Screening and diagnostic pathways differ in triage levels, wait time targets, and image interpretation. Radiologists take responsibility for appropriate use of health care resources. For these reasons, appropriate patient selection is facilitated by screening within the context of an organized program.
Frequency and Duration of Screening
The frequency of lung cancer screening should minimize the number of CT scans to which participants are exposed, while optimizing the detection of interval lung cancers (lung cancers that present after a negative screening exam but before the next scheduled test). The NLST performed LDCT annually whereas the NELSON study performed LDCT at variable intervals and the Multicentric Italian Lung Detection (MILD) trial randomized patients to annual or biennial LDCT. MILD demonstrated a similar performance of annual versus biennial screening but was likely underpowered for a definitive conclusion as there were only around 1150 patients in each arm. 22 The NELSON study suggested that participants with a baseline scan demonstrating no nodules or tiny nodules (<100 mm3) were at very low risk of lung cancer at 2 years. 9 Real world evidence will be forthcoming as the Australian lung cancer screening program will include a component of biennial screening for those low-risk participants. 23 Currently, most programs operate with annual screening but that is likely to change as risk prediction modelling improves. 24 In Nova Scotia, patients who receive 2 negative screens are then screened on biennial basis, while in British Columbia, patients identified as having low risk nodules will be screened on a biennial basis.
Recommendations on the duration of screening vary among medical societies. The Canadian Task Force stands out as the only organization recommending just 3 rounds of screening.11,24 Multiple studies demonstrate an ongoing high risk of lung cancer following negative screening CT. Data from screening programs, age-related changes in cancer rates, competing causes of death and growth rates of cancer conclude that screening beyond 3 years is appropriate.25,26 Many programs recommend continuing screening up to the age of 74 years and in some cases, up to 80 years. Once the patient is enrolled in a lung cancer screening program, it is up to the patient, their health care provider, and in some cases, the screening program to decide if screening should continue until the upper age limit. Screening may be stopped if the patient is found to have any medical condition with significant limitations of life span (eg, metastatic cancer), quality of life (eg, advanced dementia), or conditions that preclude investigation and curative treatment of detected lung cancer.
Impact of Lung Cancer Screening on Health Systems
There is legitimate concern over the implementation of lung cancer screening programs in the public health care system given the large number of current and former smokers and the impact this might have in terms of healthcare costs and wait times. In other jurisdictions, perceived impacts have generally not manifested, as real-world participation in lung cancer screening remains low with only a small proportion of eligible patients ultimately participating, even in the United States where organized lung cancer screening was first approved. 27 Cost effectiveness modeling data of 576 screening scenarios using a population-based cohort from Ontario, Canada found that lung cancer screening was cost effective and that annual screening was more cost-effective than biennial screening. 28 Results of the Ontario Lung Screening Pilot provide real-world data on the operation of a lung cancer screening program. 7 In metrics published by Tammemägi et al, there was no impact on CT wait times at sites where lung cancer screening was implemented and wait times across 7 metrics were comparable to provincial metrics. 7
Harms and Limitations of Screening for Individual Participants
The harms of lung cancer screening, including anxiety, harms related to investigation and treatment of CT findings, overdiagnosis, and radiation should be communicated to patients and health care providers. Patients and providers may overestimate the benefits of lung cancer screening and underestimate the harms. Overall, the harm benefit ratio of lung cancer screening is narrow and is optimized when screening is limited to those with a high risk of lung cancer. Participants who are not at high risk of lung cancer may experience the harms of screening but are less likely to benefit.
Although lung cancer screening CT is performed with a low-dose technique, all patients undergoing screening are exposed to radiation. It is very difficult to quantify an individual’s radiation harm related to participation in lung cancer screening, but some cancers are caused by medical imaging radiation. Younger patients and female patients have a higher rate of radiation-induced damage.29,30 A 10-year analysis of screening trial participants found that for every 108 lung cancers detected through a screening program, radiation from medical imaging caused one major cancer. 31 Older modeling studies suggest that widespread adoption of lung cancer screening could result in an overall 1.8% increase in lung cancer in the United States due solely to radiation exposure from participation in a screening program. 32
Patient anxiety related to CT results (lung nodules and incidental findings) is highest immediately following a scan 33 and can be mitigated with nodule management and reporting systems that appropriately communicate risk to the patient and referring clinician. A study on screening participants health-related quality of life (HRQoL) and anxiety levels from the Pan-Canadian Early Detection of Lung Cancer Study did not show any clinically significant difference in HRQoL between baseline and multiple survey points following initial screening, although a minority of participants demonstrated clinically-significant increased anxiety levels. 34
Physical harms associated with lung screening are predominantly related to investigation of CT findings, including complications from investigation and treatment of true positives, false positive, and incidental findings. In the NLST, there were 356 lung cancer deaths in the CT arm and 443 in the chest radiograph arm. 2 There were also higher rates of serious complications and deaths in the CT arm. A total of 16 people in the CT arm died within 60 days of an invasive procedure, meaning for every 4 lung cancer deaths prevented, one person died. Morbidity and mortality data for lung cancer treatment are evolving however, with the widespread adoption of less invasive surgical treatment, the development of non-operative curative intent lung cancer treatment, and the advent of more robust management protocols that limit invasive work up of CT findings, which may further reduce the potential harms of screening. 7
Overdiagnosis
Overdiagnosis results in harms arising from diagnostic procedures and treatments for lung cancer without prolonging life. The impact of overdiagnosis results from 3 issues: slow growing nonaggressive cancers (predominantly adenocarcinoma spectrum lesions); competing causes of death in the target population; and the relatively high rate of morbidity for the surgical treatment of early-stage lung cancer compared to early-stage colon, breast, or cervical cancer. When lung cancer screening trials were first published, there was significant concern about the potential for overdiagnosis. However, long term follow-up of NLST participants demonstrated no significant difference in rates of lung cancer diagnoses between the CT and CXR arms, 8 with persistent and significantly lower lung cancer mortality in the CT arm. This means that the cancers labelled initially as “overdiagnosed” in the CT arm may not have been overdiagnosed as they would have eventually become clinically apparent. New nodule management protocols not in use at the time of the NLST purposefully discourage invasive procedures and treatment for indolent neoplasms. For example, Lung-RADS categorizes a slowly growing ground glass opacity as a negative screen requiring surveillance rather than intervention. In this common situation, overdiagnosis is controlled by limiting overtreatment.
Those at high risk for lung cancers are also at increased risk of death from heart disease, cerebrovascular disease, and COPD. 35 Overdiagnosis can be controlled by limiting overtreatment, committing resources to improving health in general (including smoking cessation), and limiting CT screening to those most likely to benefit.
Equity
Lung cancer disproportionately affects Canadians of lower socioeconomic status with younger age at time of diagnosis, higher stage at diagnosis, and higher mortality compared to Canadians of higher socioeconomic status.36,37 Although smoking rates are higher for those with lower income and education levels in Canada, smoking history alone does not fully account for the variability in lung cancer incidence. A similar relationship has also been demonstrated in other countries, including in the United Kingdom and the United States.38,39
A large Canadian census-based study found lower 5-year lung cancer survival rates among First Nations adults compared to non-Indigenous adults, with persistent disparity after controlling for income and rurality. 40 While US data demonstrate higher incidence of lung cancer, greater stage at diagnosis and worse outcomes for African Americans,38,41 similar results have not been shown in Canada. A Canadian census-based study did not demonstrate lung cancer disparities among visible and non-visible minorities 42 and a large analysis from Statistics Canada revealed relatively low rates of lung cancer among census-responders of Caribbean and African descent. 43
The PLCOm2012 risk model accounts for education level (as a marker of socioeconomic resources) and race when determining lung cancer risk. 44 In practice, use of the model means that those with higher education levels or people who describe themselves as “white” will have a lower calculated lung cancer risk than others with a similar age and smoking history. The use of education level to help determine CT eligibility has generally been accepted in Canada 5 ; however, the use of race in the model has been more controversial. Some studies have shown that, compared to age and pack-year assessment alone, the PLCOm2012 model allows more racially equitable identification of people at high risk for lung cancer.45-47 Criticisms of this approach include the fact that the PLCOm2012 study did not match the diversity of the US population it sampled, that the racial categories were oversimplistic, and the observed differences were unlikely to be related to race and more likely to reflect socioeconomic determinants of health. 48 Note that some provinces use the PLCOm2012 risk model without race. The lower age and pack-year requirements for CT screening eligibility in the 2021 USPSTF guidelines has been shown to improve racial disparities in eligibility.49-51
Regardless, if the mortality benefit of lung cancer screening is to be fully realized, the geographic, cultural, psychosocial, and systemic barriers to screening must be addressed. These barriers, documented in Canadian screening programs for other cancers52-54 have additional impact in lung cancer programs due to the underlying marked socioeconomic disparities in lung cancer incidence and outcomes. Understanding and reducing the causes of low lung cancer screening uptake in some populations may do more to improve equity than adjustments to LDCT eligibility. Failure to address these concerns may result in lung screening programs that exacerbate disparities in lung cancer outcomes.
Qualifications and Responsibilities of Personnel
Training Requirements for Radiologists Interpreting Screening Examinations
Since 2016 when our original guidelines were published, several organizations have established recommendations or requirements for radiologists reporting lung cancer screening studies. A dedicated fellowship in thoracic radiology is not required for reporting of these examinations, but cases should be read by radiologists with a regular practice of reporting thoracic CT scans. Minimum reporting volumes have been established by the Canadian Association of Radiologists Lung Screening Accreditation Program (300 chest CTs including 100 CT lung screens annually), 55 the United Kingdom National Health Service (500 chest CTs annually), 56 and the American College of Radiology (200 chest CTs in the prior 36 months). 57 Note that test banks or CME courses can be used to augment volumes.
Double reading and expert reading improve sensitivity and specificity for nodule detection and characterization, 58 suggesting that radiologists benefit from initial feedback from an experienced reader when beginning to interpret lung cancer screening studies. Both the Canadian Association of Radiologists Accreditation Standards and the UK National Health Service Quality Assurance Standards require completion of specific lung cancer screening training.
Even radiologists who are comfortable reporting chest CTs should engage in initial education specific to lung cancer screening, including but not limited to: the use of result categories and reporting standards used in their jurisdiction, harms of screening including overdiagnosis, and management of incidental findings. Most provincial screening programs in Canada use the American College of Radiology’s Lung-RADS® v2022 (Lung-RADS) results categories. In regions where there is no established provincial program, we recommend training in the use of Lung-RADS. Accredited training for lung cancer screening (the QUEST program) is available through the CAR Continuing Professional Development platform.
Radiologists involved in lung cancer screening should meet standards as outlined by the Canadian Association of Radiologists. These standards currently include minimum volumes as outlined previously, participation in a quality assurance program, and double reading of first 15 positive lung cancer screens.
Quality Standards
Although LDCT screening exams are similar in technique to routine non-contrast CT scans of the chest, specific screening protocols have been designed to limit radiation, accurately characterize nodule density, and detect subtle growth. This CT protocol requires, at minimum, 16 detectors. Details on the protocol endorsed by the CAR and the CSTR are included in Appendix A. Note that intravenous contrast should not be used and that the field of view should be limited to the lungs. Due to the low-dose technique, screening CTs should appropriately appear noisy with limited resolution of the mediastinum and upper abdomen.
Audits of CT radiation exposure have found a wide range of radiation dose, even for studies labelled as “low-dose.”59,60 Centres performing screening CT that use the term “low-dose,” should perform appropriate quality assurance measures to ensure the CT dose index (CTDI) for standard-sized patients does not exceed 3.0 mGy.
Quality Indicators
Monitoring of quality indicators to measure the performance of lung cancer screening programs can help programs evaluate their practice and identify potential areas for improvement to optimize the benefits and minimize the harms of screening. In the United States, the National Lung Cancer Roundtable identified 6 key quality indicators. 61 In 2018, the Canadian Partnership Against Cancer convened the Lung Cancer Screening Quality Indicators Working Group which reached consensus on ten quality indicators. 62 These are listed in Appendix B.
Documentation
Minimum Elements of the Report
Effective communication in lung cancer screening is critical for optimal clinical management. 63 Structured reporting with the use of reporting templates is recommended to ensure consistent communication with referring clinicians, specialists, and patients, and to ensure that all pertinent information is included to guide clinical management. 64 Reports for LDCT lung cancer screening should include the reason for the exam (baseline, annual follow-up, or interval follow-up), technique, date of the most recent comparator exam, findings, and impression.
Nodule information which should be reported includes nodule location, attenuation, and other associated features which convey risk of malignancy, such as calcification, cavitation, presence of intralesional fat, and appearance of nodule margins. The number and minimum size of nodules to be reported may vary between institutions or screening programs. Based on Lung-RADS, the mean diameter of the nodule should be reported to one decimal point, obtained by averaging the long and short axis measurements from any plane to reflect the true size of the nodule. 65 The nodule volume may also be determined and reported to the nearest whole number in mm3. 65 Measurements can be performed using manual or automated/semi-automated techniques depending on available software. For part-solid nodules, both the size of the solid component and the overall size of the nodule should be reported. Image and series numbers of actionable nodules should be provided, as well as an indication of changes over time. Clinically significant incidental findings should also be noted.
The impression should include a summary of the most suspicious pulmonary nodule(s), classification (eg, Lung-RADS category), and a specific management recommendation. Any actionable incidental findings and their recommended follow-up should also be included. Using established guidelines for the management of incidental findings,66-68 is important to minimize the risks associated with their workup and maximize the benefits of early detection. A sample structured lung cancer screening report is available from Ontario Health (Cancer Care Ontario). 69
Organized screening programs generally communicate LDCT results directly to participants. Referring clinicians may be responsible for coordinating lung nodule management based on protocols established by the local screening program. Alternatively, programs may be responsible for this. In most organized screening programs, referring clinicians are responsible for managing all incidental findings.
Standardized Reporting Systems and Nodule Management Protocols
Use of a standardized reporting system and lung nodule management protocol 69 is recommended to ensure consistency, clarity, and comprehensiveness in communications related to screening exam results and to standardize management recommendations. Lung-RADS is the most widely used reporting system for lung cancer screening in North America. Other frameworks include LU-RADS, 70 a Canadian-designed classification system for CT lung cancer screening, and the PanCan nodule management protocol.71-73
Lung-RADS classification is primarily based on nodule size, attenuation (eg, solid, part-solid, non-solid), morphology, location, and change over time. 65 Each category corresponds to a specific management recommendation, ranging from interval LDCT, diagnostic testing (including diagnostic chest CT, PET/CT, or tissue sampling), or return to annual screening. 65 The most recent version, Lung-RADS v. 2022, provides additional guidance on management of atypical pulmonary cysts, juxtapleural nodules, airway nodules, and infectious or inflammatory findings. It also clarifies stepped management and use of the S modifier for incidental findings. 65
As an alternative to Lung-RADS, the PanCan nodule management protocol 71 is based on the risk of malignancy as determined using the PanCan lung nodule risk prediction model. 74 The PanCan model was developed based on the Pan-Canadian Early Detection of Lung Cancer Study. The model includes nodule size, type, upper lobe location, spiculation, nodule count, and the participant’s age, sex, family history of lung cancer, and presence of emphysema. 74 Some studies have shown that the PanCan model has superior accuracy compared to Lung-RADS in predicting malignancy. 75 While Lung-RADS requires only a standard PACS system, the use of the PanCan model requires either specialized software or data to be manually entered into a spreadsheet or dedicated application. The PanCan nodule management protocol allows for the first follow-up to occur 2 years after the baseline LDCT for individuals with low malignancy risk. 71
Artificial Intelligence
Artificial intelligence (AI) tools for lung nodule detection and determining nodule malignancy risk76,77 are now commercially available. 78 The British Columbia lung screening cancer program is currently using AI tools, with radiologist oversight, routinely in their lung cancer screening program. 72 AI tools for lung nodule risk stratification may reduce the number of interval follow-up LDCT exams and result in earlier diagnoses of lung cancer 79 and may minimize costs associated with follow-up of screen-detected lung nodules 80 Compared to models such as the PanCan model that require clinical variables such as family history of lung cancer, AI models can achieve similar or higher levels of performance based on imaging alone.76,77 It may be reasonable to include AI outputs from appropriately validated AI tools, which have received regulatory approval, in the radiology report. Further work should explore the optimal role of AI risk stratification tools and optimal risk thresholds to guide follow-up intervals and referrals for diagnostic testing. Implementation will require broad collaboration across health system partners to invest in these innovative tools.
Inclusion/Exclusion Criteria for Screening
The use of risk prediction models such as the PLCOm2012 in combination with age criteria instead of the NLST criteria of smoking and age has been shown to improve overall detection of cancers with a lower number of persons screened. The exact threshold of risk screening may vary among screening programs and may in part be dependent on the CT scanning capacity of the province. Ranges of 1.5% to 2% have been implemented in Canada, although modelling data suggests risk thresholds as low as 1.2% may be cost effective. Data modelling has suggested that a PLCOm2012 risk of 2% or higher has a cost effectiveness ratio of just under $50 000, which has been a threshold selected by several Canadian provinces as a balance of efficient detection of cancer and affordability to the health care system. 81
Recently, the US Preventive Services Task Force (USPSTF) recommended expanding eligibility from a 30-pack year smoking history to a 20-pack year smoking history, based on data from modelling studies predicting a greater reduction in lung cancer mortality, a reduction in the number of lung cancer deaths, and an increase in life-years gained. 82
The provinces with current active screening programs all have similar eligibility criteria. In general, individuals with a minimum age cut-off of 50 to 55 years to a maximum of 74 to 80 years with a smoking history of 20 years or more of daily cigarette smoking, who are active smokers or former smokers are eligible for risk assessment. If they have a threshold (currently 1.5% or 2%) 6-year lung cancer risk using the PLCOm2012 risk model, they are considered eligible for enrolment in the lung screening program. Exclusion criteria vary slightly between provinces but in general include individuals currently undergoing evaluation for suspicious pulmonary nodules, symptoms potentially suspicious of lung cancer, any serious comorbidity limiting survival, and inability to physically undergo CT scanning (Table 2).83,84
Summary of CAR/CSTR Recommendations.

Conclusion
Since the initial publication of these guidelines, there has been significant progress in lung cancer screening. The results of the NELSON trial provide additional data to support the efficacy of organized screening to decrease lung cancer mortality. Several Canadian provinces have fully implemented screening programs, and most others have implemented or are in the planning stages for pilot projects. Early results from provincial screening programs are encouraging, with rates of detection of early-stage lung cancers surpassing that of both the NELSON and NLST studies. New data from screening programs and risk modelling studies have improved identification of participants that would benefit from screening.
There remain several areas of investigation for best practices in lung cancer screening, which include potentially identifying a lower risk screening population that could undergo biennial rather than annual screening and the use of AI/CAD software to streamline reporting screening examinations. The need for equitable selection of participants for screening and access to screening programs across the country are also important considerations that need to be evaluated and addressed.
Footnotes
Appendix A
Recommended CT Lung Screening Protocol.
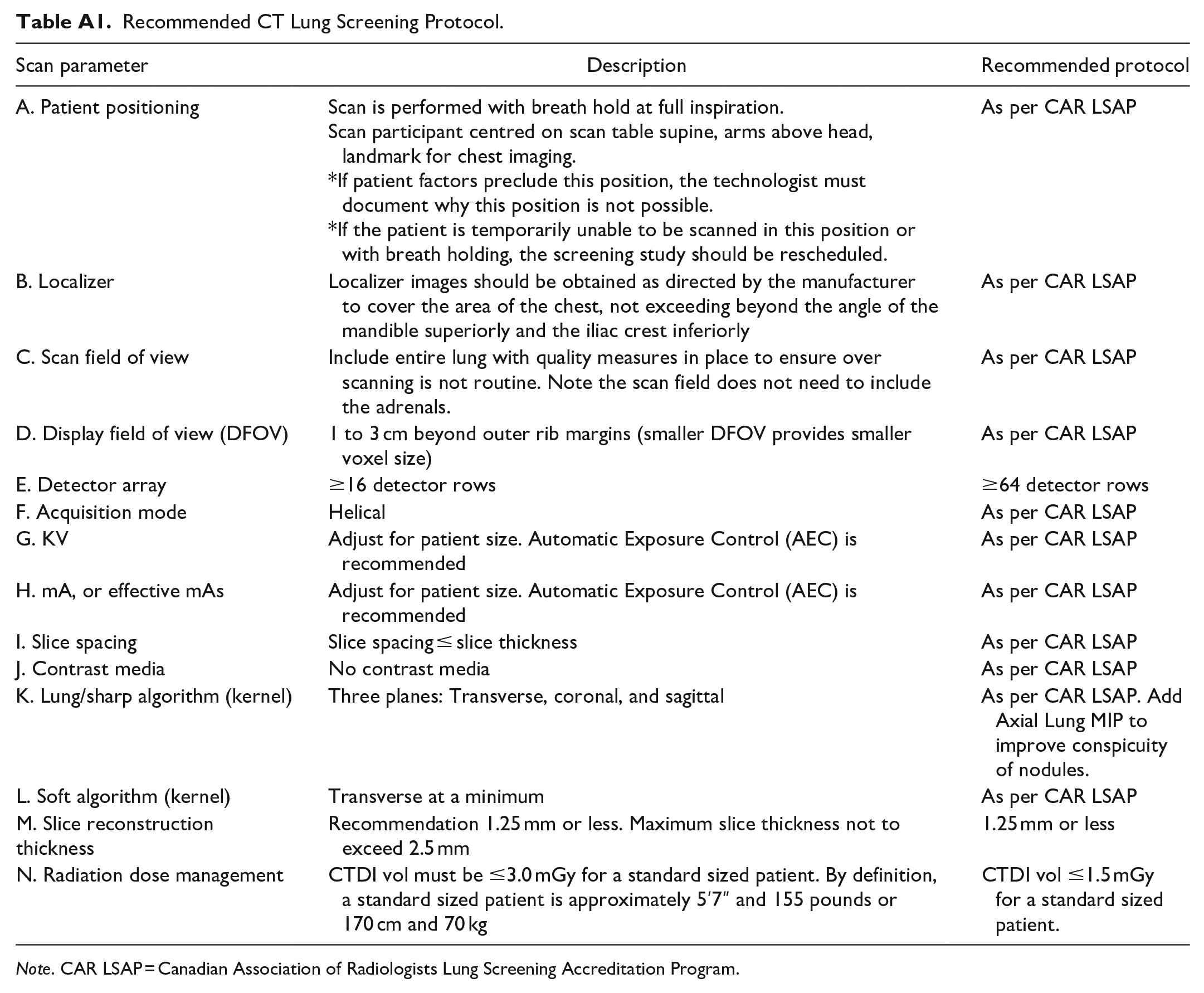
| Scan parameter | Description | Recommended protocol |
|---|---|---|
| A. Patient positioning | Scan is performed with breath hold at full inspiration. Scan participant centred on scan table supine, arms above head, landmark for chest imaging. *If patient factors preclude this position, the technologist must document why this position is not possible. *If the patient is temporarily unable to be scanned in this position or with breath holding, the screening study should be rescheduled. |
As per CAR LSAP |
| B. Localizer | Localizer images should be obtained as directed by the manufacturer to cover the area of the chest, not exceeding beyond the angle of the mandible superiorly and the iliac crest inferiorly | As per CAR LSAP |
| C. Scan field of view | Include entire lung with quality measures in place to ensure over scanning is not routine. Note the scan field does not need to include the adrenals. | As per CAR LSAP |
| D. Display field of view (DFOV) | 1 to 3 cm beyond outer rib margins (smaller DFOV provides smaller voxel size) | As per CAR LSAP |
| E. Detector array | ≥16 detector rows | ≥64 detector rows |
| F. Acquisition mode | Helical | As per CAR LSAP |
| G. KV | Adjust for patient size. Automatic Exposure Control (AEC) is recommended | As per CAR LSAP |
| H. mA, or effective mAs | Adjust for patient size. Automatic Exposure Control (AEC) is recommended | As per CAR LSAP |
| I. Slice spacing | Slice spacing ≤ slice thickness | As per CAR LSAP |
| J. Contrast media | No contrast media | As per CAR LSAP |
| K. Lung/sharp algorithm (kernel) | Three planes: Transverse, coronal, and sagittal | As per CAR LSAP. Add Axial Lung MIP to improve conspicuity of nodules. |
| L. Soft algorithm (kernel) | Transverse at a minimum | As per CAR LSAP |
| M. Slice reconstruction thickness | Recommendation 1.25 mm or less. Maximum slice thickness not to exceed 2.5 mm | 1.25 mm or less |
| N. Radiation dose management | CTDI vol must be ≤3.0 mGy for a standard sized patient. By definition, a standard sized patient is approximately 5′7″ and 155 pounds or 170 cm and 70 kg | CTDI vol ≤1.5 mGy for a standard sized patient. |
Note. CAR LSAP = Canadian Association of Radiologists Lung Screening Accreditation Program.
Appendix B
Acknowledgements
The Working Group would like to express its gratitude to the CAR members who took the time to review and provide their feedback on earlier drafts of the document. The Working Group would also like to express its thanks to the following organizations for their review and feedback: (i) Canadian Association of Medical Radiation Technologists (CAMRT), (ii) Canadian Task Force on Preventative Health Care (CTFPHC), and (iii) Canadian Partnership Against Cancer (CPAC).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
