Abstract
Thoracic interventions are frequently performed by radiologists, but guidelines on appropriateness criteria and technical considerations to ensure patient safety regarding such interventions is lacking. These guidelines, developed by the Canadian Association of Radiologists, Canadian Association for Interventional Radiology and Canadian Society of Thoracic Radiology focus on the interventions commonly performed by thoracic radiologists. They provide evidence-based recommendations and expert consensus informed best practices for patient preparation; biopsies of the lung, mediastinum, pleura and chest wall; thoracentesis; pre-operative lung nodule localization; and potential complications and their management.
Introduction
The Canadian Association of Radiologists (CAR), Canadian Association for Interventional Radiology (CAIR) and Canadian Society of Thoracic Radiology (CSTR) Working Group on Thoracic Interventions has compiled the following best practice guidelines focused on the interventions commonly performed by thoracic radiologists, rather than those more commonly performed by interventional radiologists. As such, ablation treatment of lung cancer and endovascular interventions are not discussed. Recommendations are based on a comprehensive literature review and are relevant to most practice settings in Canada. In cases where there is a paucity of data or low-quality evidence to support certain practices, the Working Group has provided recommendations based on available evidence and expert consensus.
Patient Preparation
Thoracic interventions can be safely performed on an outpatient basis.1-3 Pre-emptive post-procedure admission can be considered for patients on home oxygen, those who live alone or in remote locations with limited at-home support or access to health care. In general, patients should be accompanied after discharge and have a reliable adult to stay with overnight in case of a delayed complication.
Biopsy of chest wall lesions and thoracentesis are low bleeding risk procedures and pre-procedure haematological testing is not required. If on anticoagulation medication, the International Normalized Ratio (INR) should be corrected to ≤ 2–3 and platelet transfusion is recommended if the count is below 20 × 109/L. All other thoracic interventions are high bleeding risk procedures. Therefore, pre-procedure haematological testing (INR, platelet count and haemoglobin) is recommended. 4 If elevated, the INR should be corrected to ≤1.5–1.8 and platelet transfusion is recommended if the count is below 50 × 109/L. In the absence of risk factors, laboratory results within 3 months of biopsy are acceptable. Patients who are on anticoagulation and antiplatelet medications require pre-procedure thrombotic risk assessment and management of medications (Supplementary Appendix A). This should be done in consultation with the primary care provider, the physician who prescribed the medication, or a thrombosis or haematology service. 4 Uncorrectable coagulopathy is an absolute contraindication for high bleeding risk procedures.
Pulmonary function tests (PFTs) are not routinely performed but can be considered in patients with severe COPD and interstitial lung disease (ILD). 3 Thoracic interventions carry a very low risk of procedure-related infection. Routine antibiotic prophylaxis is not indicated. 5 Patients undergoing procedures to diagnose infection (empyema, lung biopsy for atypical infection) are often already on antibiotics.
On the day of the procedure, patients should be assessed by the radiologist performing the procedure to obtain informed consent, record pre-procedural vitals (heart rate, blood pressure, respiratory rate and oxygen saturation) and review recent laboratory values and medications (Supplementary Appendix B). Placement of an intravenous catheter is suggested to enable rapid administration of medication and fluid to treat complications such as pain, vasovagal reaction, or bleeding. Marking the side of the procedure in the supraclavicular fossa facilitates rapid identification of the side of the procedure for chest tube placement to treat a pneumothorax. Unless sedation is used, fasting before thoracic interventions is not required.
Biopsies
Lung
Image-guided percutaneous transthoracic needle biopsy (PTNB) is a well-known minimally invasive procedure in the diagnostic workup of thoracic lesions, performed by chest, general or interventional radiologists. 6 The choice of imaging guidance modality, target lesion and approach is at the radiologist’s discretion, taking into consideration patient safety, equipment availability, cytology/pathology and post-biopsy support. In general, targeting enlarging, peripheral and larger lesions, those in the upper lung zones and the non-necrotic hypermetabolic part of a lesion without crossing a fissure, vessel or bulla is preferred. Bronchoscopic biopsy is recommended for central lesions abutting or involving the central airways, endobronchial lesions and paratracheal and subcarinal lymph nodes.7,8 PTNB is not routinely recommended for patients with ILD due to lower diagnostic yield 9 ; surgical lung biopsy has the highest diagnostic yield for ILD. 10
Indications
1. Initial diagnosis of lesions suspicious for malignancy (imaging characteristics, patient age, smoking and family history of lung cancer, +/− Brock or Herder model).
11
Preoperative histologic confirmation of malignancy has been shown to significantly reduce the rate of unnecessary resection.
12
2. Molecular analysis for initial assessment and re-biopsy for recurrent or progressive disease: the genetic profile of tumors provides options for management with targeted chemotherapy or immunotherapy and can change after initial treatment. For re-biopsy, either viable tissue of progressive disease from the primary lesion or a metastasis can be targeted.
13
3. Multiple nodules in patients without known primary or single or multiple nodules in patients with more than one primary malignancy or when there was a long remission after treatment of primary cancer and tissue confirmation of metastases is required. 4. Research: to assess the efficacy of new drugs. 5. Persistent, non-resolving consolidation not diagnosed by clinical and laboratory investigations: diagnosis of non-infectious granulomatous disease or unusual fungal infections not responding to empiric antibiotic therapy in immunosuppressed patients.
14
Mycobacterial infection can also be diagnosed in 80% of patients.
15
Specific Situations
Cavitary Lesions
When feasible, the most hypermetabolic and thickest portion of the wall of the cavity should be targeted. Samples should be sent for microbiological evaluation when no malignant cells are identified on rapid on-site evaluation (ROSE). 16 Core needle biopsy (CNB) is highly accurate for diagnosis of benign lesions. 17
Small Nodules
While reasonable sensitivity and accuracy can be achieved for biopsy of small lung nodules (<10 mm), 18 CNB or combination of Fine Needle Aspiration (FNA) + CNB appear to yield higher diagnostic accuracy than FNA alone.18,19 Several studies have shown reduced diagnostic accuracy for biopsy of small lung nodules <10 mm20,21 or <15 mm.22,23 Ability to perform PTNB on small lung nodules while maximizing diagnostic yield and minimizing complications varies across institutions according to local expertise. 24
Sub-Solid Lesions
The solid component of sub-solid nodules is an independent predictor of malignancy (Figure 1).
25
In one study, the diagnostic accuracy of computed tomography (CT) analysis without PTNB was equivalent at predicting malignancy to preoperative PTNB of sub-solid nodules with a solid component measuring >5 mm.
26
In another study, of 356 sub-solid lung nodules, 330 (92.7%) were resected without preoperative PTNB and the rate of malignancy was 95.2% (314/330), for 242 part-solid and 72 pure ground-glass nodules.
27
Hence, some persistent sub-solid nodules with characteristic imaging features of primary lung adenocarcinoma, demonstrating growth and hypermetabolism on FDG-PET are surgically resected without PTNB.
28
72-year-old woman with incidental part-solid nodule in the right upper lobe suspicious for invasive adenocarcinoma targeted for tissue diagnosis with computed tomography-guided percutaneous transthoracic needle biopsy. (A) Radio-opaque grid over anterior chest wall in supine position for localization. Freezing needle should then be introduced to the subpleural space and local anaesthetic injected while patient suspends breathing (not shown). (B) Introducer placed just at the edge of the solid part of the nodule. (C) Fine needle aspiration or core biopsy needle is then inserted through the introducer, and biopsies are obtained from the solid component. Note the tip of the biopsy needle within the solid component of the nodule.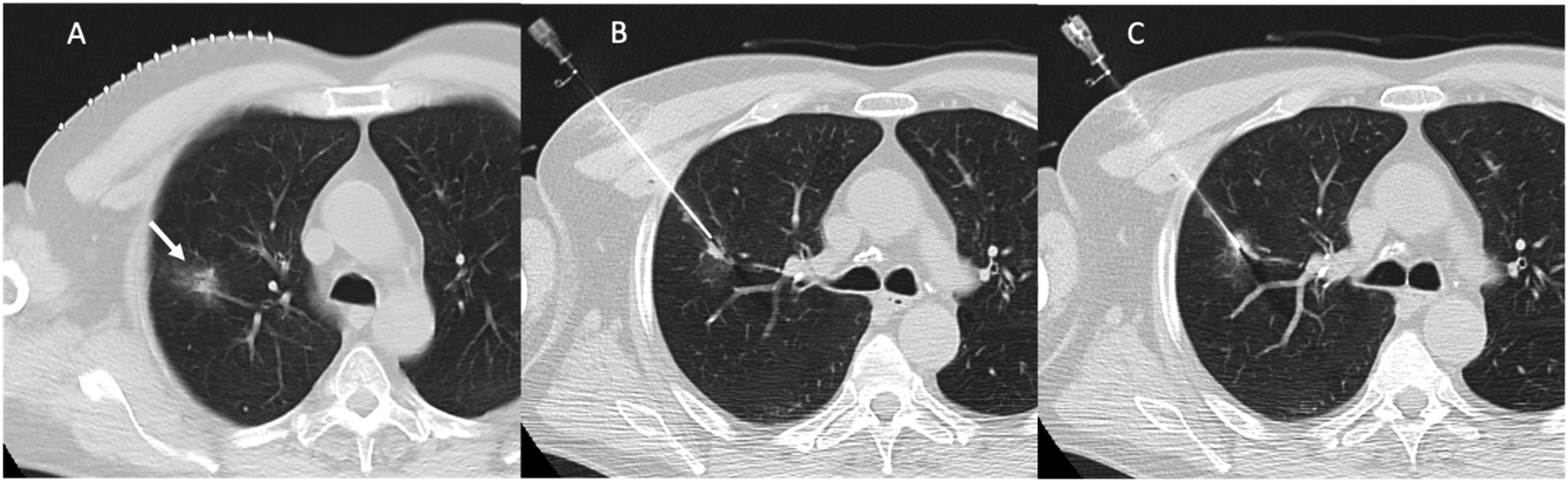
Relative Contraindications to PTNB:
1. Patient Limitations: Unable to follow breath-hold instructions and maintain position. 2. Lack of impact on patient management: Biopsy results should guide management. 3. FEV1 ≤35% predicted: PFTs are rarely performed pre-biopsy and following this recommendation would eliminate many patients who could benefit from tissue diagnosis. In addition, hypoxaemia is not necessarily associated with a specific FEV1.29,30 and there is paucity of data to suggest a clear FEV1 threshold, below which biopsy should not be performed. No biopsy request should routinely be declined based on the severity of emphysema. Decision to proceed with biopsy should be decided after evaluation of the risks vs benefits. 4. Single functional lung: PTNB should only be considered when tissue diagnosis is absolutely necessary, ICU and surgical assistance are available and the biopsy can be performed by an experienced radiologist. In a study of 14 patients with prior pneumonectomy who underwent PTNB, Cronin reported a pneumothorax rate of 25%, but all were small and asymptomatic, and none required a chest drain.
31
5. Positive pressure ventilation: Patients on positive pressure ventilation may have increased risk of pneumothorax
32
and air embolism.33-35 6. Pulmonary hypertension: There is insufficient data to support an increased risk of haemorrhage.3,36,37 In a study of 74 patients with mild to moderate pulmonary hypertension, there was no difference in the rate of pulmonary haemorrhage, haemoptysis or haemothorax compared to a control group.
38
Another study showed no increased risk of pulmonary haemorrhage in patients with a dilated main pulmonary artery on CT.
39
Nevertheless, PTNB in patients with severe pulmonary hypertension should be considered with caution. 7. Recent myocardial infarction: There is insufficient data regarding a safe time interval to PTNB. If possible, a delay of 4–6 weeks is reasonable3,40,41
When there are safety concerns for PTNB, the risks and benefits of the procedure should be discussed at a multidisciplinary conference. Complete staging (CT abdomen, PET/CT) is suggested to identify safer biopsy sites, allow preliminary treatment planning and prognostication, and establish goals of care. Select patients with suspicious nodules who are at high risk of complications may be offered treatment without histologic confirmation. 12
Imaging Modalities for Biopsy Guidance
Computed Tomography
This is the most commonly used modality.42-44 Advantages include the ability to target smaller, more central lesions, avoid fissures, bullae, vessels and the ability to target a specific part of a lesion, for example, the wall of a necrotic lesion or the solid component of a part-solid nodule and select the shortest biopsy trajectory.
Fluoroscopy
Fluoroscopy has a lower radiation dose and comparable accuracy and complication rate to CT. 45 Low cost, and real-time visualization of needle advancement are among other advantages. It has largely been replaced by CT but is still used in some centres to target larger lesions. Newer fluoroscopy units can perform cone beam CT to supplement fluoroscopic guidance or be used as stand-alone to guide lung biopsies. 46 Effective dose largely depends on tube voltage and the length of the procedure.
Ultrasound
This modality should be reserved for lung lesions abutting the pleura, pleural or chest wall lesions. Advantages are the lack of ionizing radiation, low cost, real-time visualization of needle advancement and low complication rate. In one study, the diagnostic yield was 89.5%. 47
Role of Fine Needle Aspiration vs Core Needle Biopsy
FNA is sufficient for diagnosis and immunohistochemical analysis of most malignancies. Rapid on-site evaluation (ROSE) decreases the rate of non-diagnostic samples. 48 For lung cancer diagnosis, the sensitivity, specificity, accuracy and complication rate of FNA and CNB are comparable. 49 CNB should be considered at the time of initial biopsy when FNA does not yield adequate material after ROSE, or when ROSE is unavailable and when lymphoma or a benign diagnosis is suspected. Where ROSE is not available, CNB may be performed exclusively, without FNA.
For lymphoma, samples should be obtained for flow cytometry based on local laboratory protocols. In addition, CNB is recommended for sub-solid lesions with no or a very small solid component to improve diagnostic yield. 50 CNB may also be required to assess response to a new drug in a research study or when a specific molecular marker is required. The coaxial technique is recommended to allow multiple passes and reduce the number of pleural punctures. A 19 G introducer with a 21G needle for FNA and 20G for core is adequate in most cases. 18G core samples slightly increase the accuracy for the diagnosis of lymphoma. 51 When CNB is performed, it is good practice to confirm the location of the cutting needle within the lesion prior to obtaining samples.
Post-Procedure Care
Patients should be transferred to an observation area, and remain on a stretcher, with the biopsy side down, when possible. Supplemental oxygen may improve patient comfort and maximize resorption of pneumothorax when present. 52 A chest x-ray to exclude complications should be obtained within the range of 301–60 minutes post-biopsy, for stable, asymptomatic patients.
Complications and Management
Potential Common and Rare Complications From Thoracic Intervention Procedures.
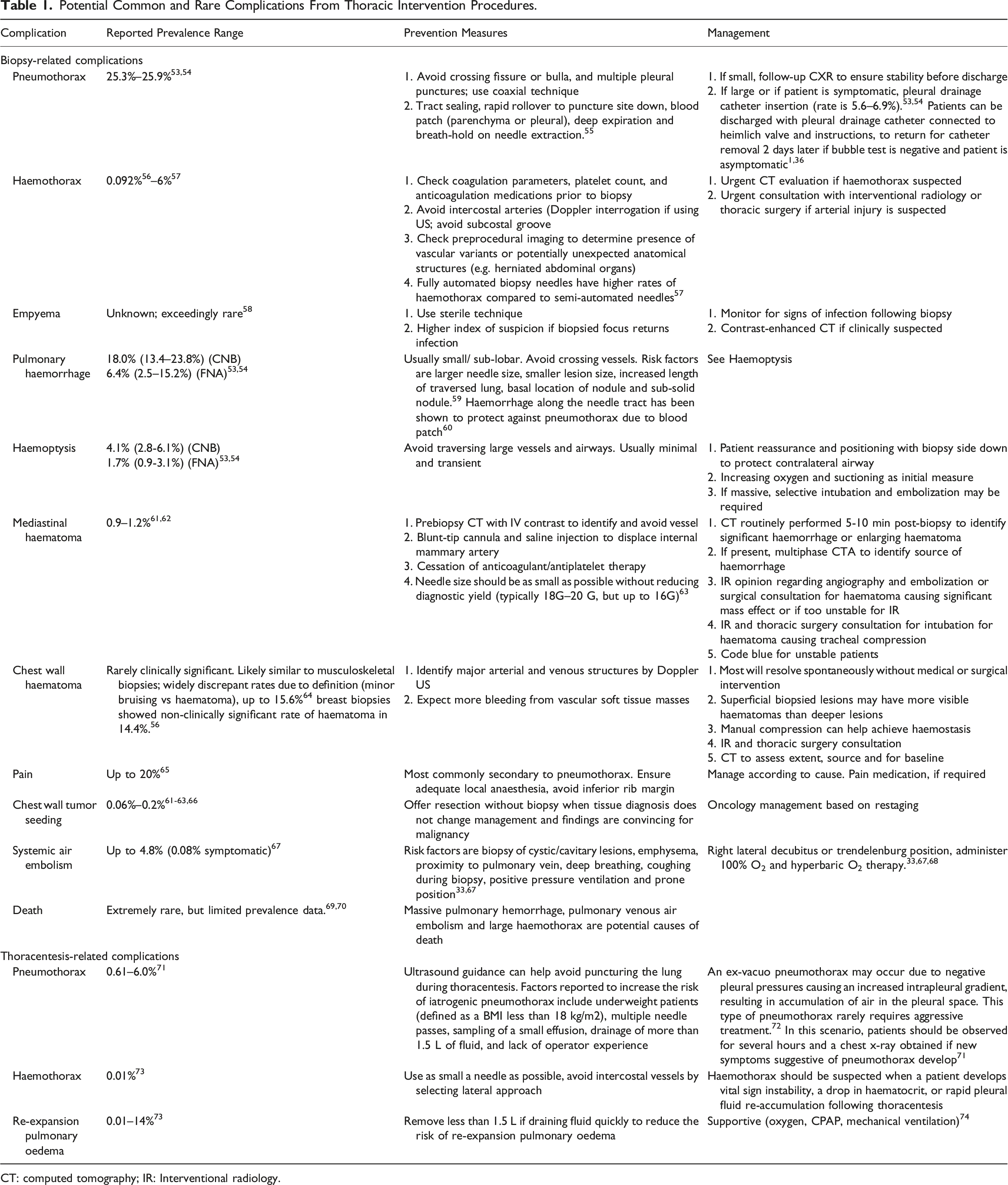
CT: computed tomography; IR: Interventional radiology.
Tumor Implantation Along Needle Tract and Pleural Recurrence
Following the report of a higher rate of pleural recurrence and one case of seeding along the needle biopsy tract during long-term follow-up of patients with Stage 1 lung cancer who had PTNB, 54 three systematic reviews and a meta-analysis were published with conflicting results. Wang et al 55 and Li et al 59 concluded that PTNB is not associated with an increase in total or pleural recurrence rate. However, for patients with subpleural lesions, PTNB did increase the risk of ipsilateral pleural recurrence. In Hong et al.’s systematic review and patient-level meta-analysis, the incidence of ipsilateral pleural recurrence in the PTNB group was higher than those in the other diagnostic procedures group for cases of isolated malignancy as well as concomitant metastases. 75 There are substantial limitations in the studies included in these reviews, such as small sample size, lack of consideration of type of cancer, tumor size, needle type and biopsy technique. Many studies also did not adjust for tumor location and distance from the pleura. 12
Discharge Post PTNB
Early discharge post-PTNB is safe when the patient has someone to stay with them at home overnight and they live reasonably close to a hospital (e.g. within 1 hour to closest hospital). A study of 506 patients undergoing PTNB confirmed the safety of discharge 30 minutes post-biopsy for patients with no pneumothorax or 60 minutes post-biopsy if asymptomatic with a stable small pneumothorax. 2 The rate of delayed symptomatic pneumothorax was 1.4% and there were no deaths or other major complications. 1 Patients should be provided clear instructions regarding returning to hospital if new symptoms develop such as shortness of breath, chest pain or haemoptysis. It is important to explain the low risk of delayed pneumothorax. In patients discharged with a pleural drainage catheter in place, additional instructions regarding catheter care and when to return to hospital for catheter removal should also be provided. High risk patients, those with borderline lung function and significant co-morbidity or inadequate home support may be better served with overnight hospital admission for observation. Community hospitals in remote areas require surgical support from the closest tertiary care centres, in the event of severe post-biopsy complications in order to ensure patient safety. Please refer to Supplementary Appendix C for a sample post-biopsy and discharge instruction pamphlet.
Air Travel Post PTNB
Patients from remote areas may require air transportation to return to their community and a prolonged post-PTNB hospital stay is inconvenient and incurs additional costs. Overall, there is paucity of data to guide recommendations regarding air travel post-biopsy. According to Federal Aviation Administration regulation, cabin pressure in commercial aircrafts is maintained to 5000–8000 feet above sea level. At this altitude, a pneumothorax only expands by 30%. The British Thoracic Society Standards of Care Committee recommends a minimum delay of one week after full resolution of pneumothorax on chest x-ray and two weeks after a traumatic pneumothorax or uncomplicated thoracic surgery. 76 Tam et al 77 reported that 14 of 179 patients who underwent PTNB and traveled within 14 days of biopsy experienced respiratory symptoms. However, there were no adverse events even when there was small stable pneumothorax prior to air travel. The Alaska Native Medical Center (ANMC), 78 reviewed the air travel records of 80 patients with traumatic pneumothorax, and concluded air travel was safe for patients with no or a stable small pneumothorax as early as 48 hours post-trauma. After considering all the evidence, the working group recommends avoiding non-essential air travel for 5–7 days post-biopsy. For patients who must fly home after PTNB, the working group recommends a minimum delay of 48 hours for those without or with a small stable pneumothorax. Modifications to these recommendations can be made for patients at particularly high risk for pneumothorax and who have significant co-morbidities or limited access to health care.
What to Do with a Non-diagnostic or Negative Biopsy Result
When FNA is performed, to minimize the rate of non-diagnostic biopsies, samples should be obtained with ROSE (where available); CNB can be performed during the same session if FNA is inconclusive. Samples should also be sent for microbiological analysis when infection is suspected. The rate of insufficient biopsies should be <10%. 28 The sensitivity for malignancy should range from 85 to 90% for lesions >2 cm. 3 If a suspicious lesion is negative for malignancy with no definite benign diagnosis, a repeat biopsy is indicated. When the cytology result is ‘non-diagnostic’, the description of the findings in the report should be carefully reviewed and incorporated into the decision to proceed with a repeat biopsy. In one study, the overall rate of malignancy when the report included non-specific benign findings was only 20.6% while 91.1% of lesions were eventually shown to be malignant when the report quoted the presence of atypical cells. 79
Mediastinum
Imaging-guided transthoracic mediastinal biopsies are safe and effective minimally invasive procedures. The most common indications include suspected thymic neoplasm, lymphoma, germ cell tumor, metastases or lung cancer invading the mediastinum. 80 Benign lesions may also mimic malignancy such as infection and granulomatous disease. 80 CT guidance is the most common modality due to its efficacy, safety and wide availability.61,80,81 The diagnostic yield for mediastinal PTNB is 92% and accuracy is 94%. 63 Diagnostic yield is lower for residual lymphoma (57%) than for initial diagnosis or recurrence (90%). 80 CT also allows for identification of post-procedure complications (Table 1).
Initial preparation begins with reviewing relevant prior imaging including CT, MRI and PET CT to identify FDG avid solid areas to target. Cystic lesions such as typical pericardial cysts do not require biopsy while potential hydatid cysts carry risk of anaphylaxis if ruptured during biopsy. 82
Biopsy approach planning should include a contrast-enhanced CT to delineate anatomy, rule out vascular lesions and ensure the trajectory avoids vascular structures while targeting non-necrotic areas. An anterior parasternal extrapleural approach can target the anterior (Figure 2) or middle mediastinum.61,83 Occasionally, the only viable access is by a transpulmonary approach which carries risk of pneumothorax.
61
A transsternal approach can avoid traversing the lung.
84
A perivertebral approach can target posterior mediastinal or subcarinal lesions.
83
A suprasternal or subxiphoid approach with needle and gantry angulation can be used to target superior mediastinal or more caudal lesions, respectively.
83
(A) Planning CT with laser light guidance and skin surface grid to identify skin entry location. (B) Under CT guidance, the coaxial introducer needle is advanced in a stepwise manner. (C) Saline can be injected (arrow) through the introducer to widen the mediastinal path or displace the mammary vessels. (D) Once the introducer is within the mass, core biopsies and fine needle aspiration may be performed. CT: computed tomography.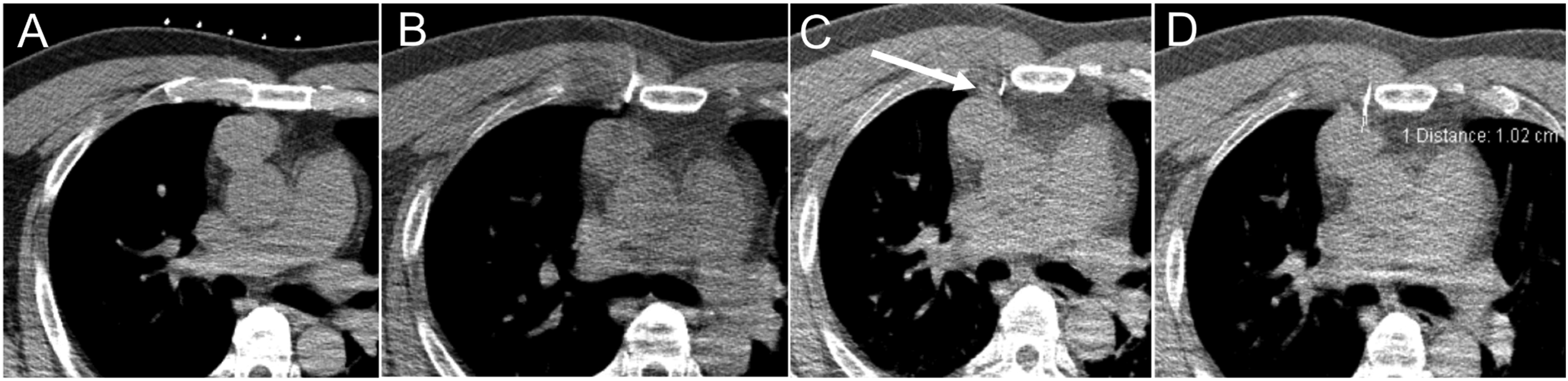
Needle length and size is selected based on lesion characteristics and is most commonly a coaxial technique. There is no difference in diagnostic yield between 18 and 20G needles. 63 For possible lymphoma, samples should be sent for flow cytometry, per local institutional protocols.
A post-procedure scan is performed to assess for complications such as haemothorax, mediastinal haemorrhage, pneumomediastinum, pulmonary haemorrhage or pneumothorax where transpulmonary approach was employed.
After biopsy, the patient is discharged to monitored nursing care for up to 2 hours and a chest x-ray is performed 30 minutes to 1 and 2-hours post-biopsy depending on radiologist preference. The patient is discharged home after radiologist reassessment, provided the post-biopsy chest x-ray shows no complication. Complication risks and management are outlined in Table 1.
Pleura
Pleural biopsies can be undertaken for pleural thickening, focal pleural mass, or for microbiological analysis of suspected empyema. In contrast to lung biopsies, many pleural biopsies can be performed with ultrasound guidance rather than CT.
Advantages of ultrasound guidance include flexibility with patient positioning (sitting, prone, supine and lateral decubitus), the ability to compensate for respiratory and patient movement in real-time and the lack of radiation. In particular, lesions in the inferior chest that are more subject to movement with diaphragmatic excursion may benefit from ultrasound guidance due to real-time visualization. 85 A study performed at a Canadian institution also showed shorter procedure times and reduced wait times for ultrasound-guided biopsy as compared to CT, while showing similar diagnostic yield and pneumothorax rates. 86
Ultrasound-guided biopsy performs better when pleural thickening is larger and nodular in morphology. 87 In one study, a nearly 100% yield was achieved with pleural thickening >20 mm 88 and a recent study suggested that optimal cut-off value for pleural thickening could be as little as 4.5 mm. 87 A smaller angle of incidence in relation to the pleura increases the length of fragment obtained, an important factor for smaller pleural lesions. 89 Depending on the patient’s body habitus and lesion location, low-frequency (2–5 MHz) or high-frequency (5–10 MHz) probes can be utilized. Where safe to do so, a 16G rather than an 18G needle increases diagnostic yield particularly for mesothelioma. 89
Lesions without adequate peripheral pleural contact or without a transcutaneous ultrasound window (e.g. mediastinal pleural lesions) and those that may be obscured by ribs or other osseous structures are more amenable to biopsy under CT guidance. The technique for CT-guided biopsy is similar to lung biopsy. When pleural biopsy is unsuccessful or cannot be performed, surgical thoracoscopy may be considered.
Risks for pleural biopsy are similar to those of lung biopsy, including pneumothorax, haemothorax, infection and haematoma along the needle tract being the most common. Seeding of tumor along the needle tract, particularly in the setting of mesothelioma is a consideration. 89 Where there is clinical concern for this, consultation with surgical colleagues prior to biopsy is recommended. Preprocedural considerations and postprocedural monitoring are similar to those for lung biopsy, as described earlier.
Chest Wall
Neoplasms of the chest wall may arise from osseous structures or soft tissues and are a heterogeneous group of tumors that may be benign or malignant. Osseous malignancies include osteosarcoma, chondrosarcoma, Ewing sarcoma and myeloma, while soft tissue malignancies include undifferentiated pleomorphic sarcoma, liposarcoma, angiosarcoma and tumors of nerve sheath and cutaneous origins. 90 Chest wall invasion from intrathoracic malignancy, metastatic disease, melanoma and lymphoma would be other malignant considerations. 90
Options for tissue sampling include fine needle aspiration, core needle biopsy and surgical biopsy. 90 Consultation with surgical oncology prior to biopsy is imperative to avoid seeding of malignancy across tissue planes. Image guidance with ultrasound or CT can be used according to radiologist preference, with CT generally preferred for small masses near the lung and ultrasound for large masses well away from intrathoracic contents. 91
For chest wall biopsies well away from the pleural surface (Figure 3), immediate complications tend to be minor and include postprocedural pain, haematoma and possibly infection. For biopsy of lesions closer to the pleura and lung, complications are similar to pleural and lung biopsies. A right scapular biopsy in lung cancer metastasis. (A) Skin surface grid placed over the scapula following scout computed tomography image localization and laser light correlation. (B) After local anaesthetic, a 19G coaxial system is shown placed to the leading edge of the tumor deposit. Pathology demonstrated lung cancer metastasis (TTF-1 and CK-7 positive, p40 negative). Case courtesy of Dr Demetris Patsios.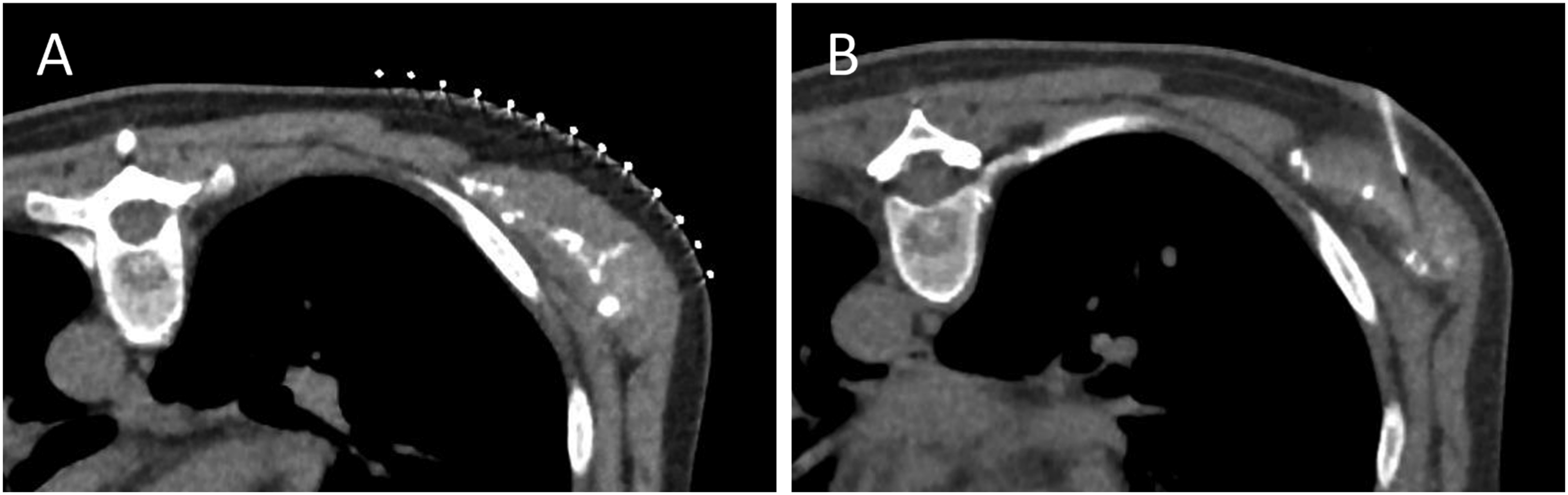
For lesions well away from the pleura and lung, there is a negligible risk of pneumothorax and a larger needle size should be considered to obtain larger cores for histopathologic analysis and avoid needle distortion or malfunction when targeting firm lesions. Biopsies can be performed with or without a coaxial needle system. For coaxial technique, the recommended needle size is up to 17G outer and 18G inner, but where safe to do so, up to 11G can be used. 92
Thoracentesis
Pleural effusions are a common manifestation of diseases localized to the pleura or underlying lung as well as multiple systemic conditions. Thoracentesis may be performed for either diagnostic or therapeutic purposes when respiratory function is impaired. Diagnostic thoracentesis is indicated when the underlying cause of the effusion is unclear or there is an unexplained lack of response to treatment. 93
Sampling of pleural fluid can allow for differentiation of a transudate from an exudate, as well as provide potential specific organisms in the case of empyema and identify the cell type of a malignant effusion. Common complications are listed in Table 1. Rare complications include injury to abdominal viscera and seeding along the needle tract in malignant effusions. There is a paucity of published data on recommended duration of outpatient monitoring post thoracentesis. Patient and institutional factors will likely influence appropriate monitoring intervals. However, the Working Group recommends a post-thoracentesis monitoring period in the range of 30 minutes–1 hour, similar to lung biopsies for uncomplicated thoracentesis.
Ultrasound guidance improves the rate of successful thoracentesis and decreases the risk of iatrogenic pneumothorax. It is particularly useful in cases of small or loculated effusions, or in non-mobile supine patients in an ICU setting. Ultrasound has a higher sensitivity than CT in the identification of complex pleural effusions with septations or echogenic fluid. 71
When possible, patients should be placed in an upright sitting position with arms elevated, or if unable, in a supine position with the ipsilateral arm raised over the head. This allows for maximal depth of the effusion in the lowest part of the chest. The procedure should be performed under site marking or direct visualization. If site marking is utilized, sampling must be performed immediately after, as any repositioning of the patient may result in fluid redistribution. Performing the procedure under direct visualization can be more technically challenging and has not been found to be associated with a lower rate of complications. 94 A lateral approach is preferred if there is sufficient fluid, as there is an increased risk of trauma to the intercostal vessels with a posterior or medial puncture. 40
For most diagnostic indications, a small gauge needle such as a 21 G needle connected to a 50 mL syringe is adequate to obtain a diagnostic sample. For therapeutic drainage of an uncomplicated effusion, a small size catheter of 5–6 F is usually adequate. Rapid removal of less than 1.5 L of fluid is recommended to avoid re-expansion pulmonary oedema. 94 For slow therapeutic drainage of complex effusions or intrapleural fibrinolytic therapy, a larger catheter size of up to 14 F can be used. 95
Pre-Operative Lung Nodule Localization
Pre-Operative Lung Nodule Localization Techniques and Common Advantages and Disadvantages.
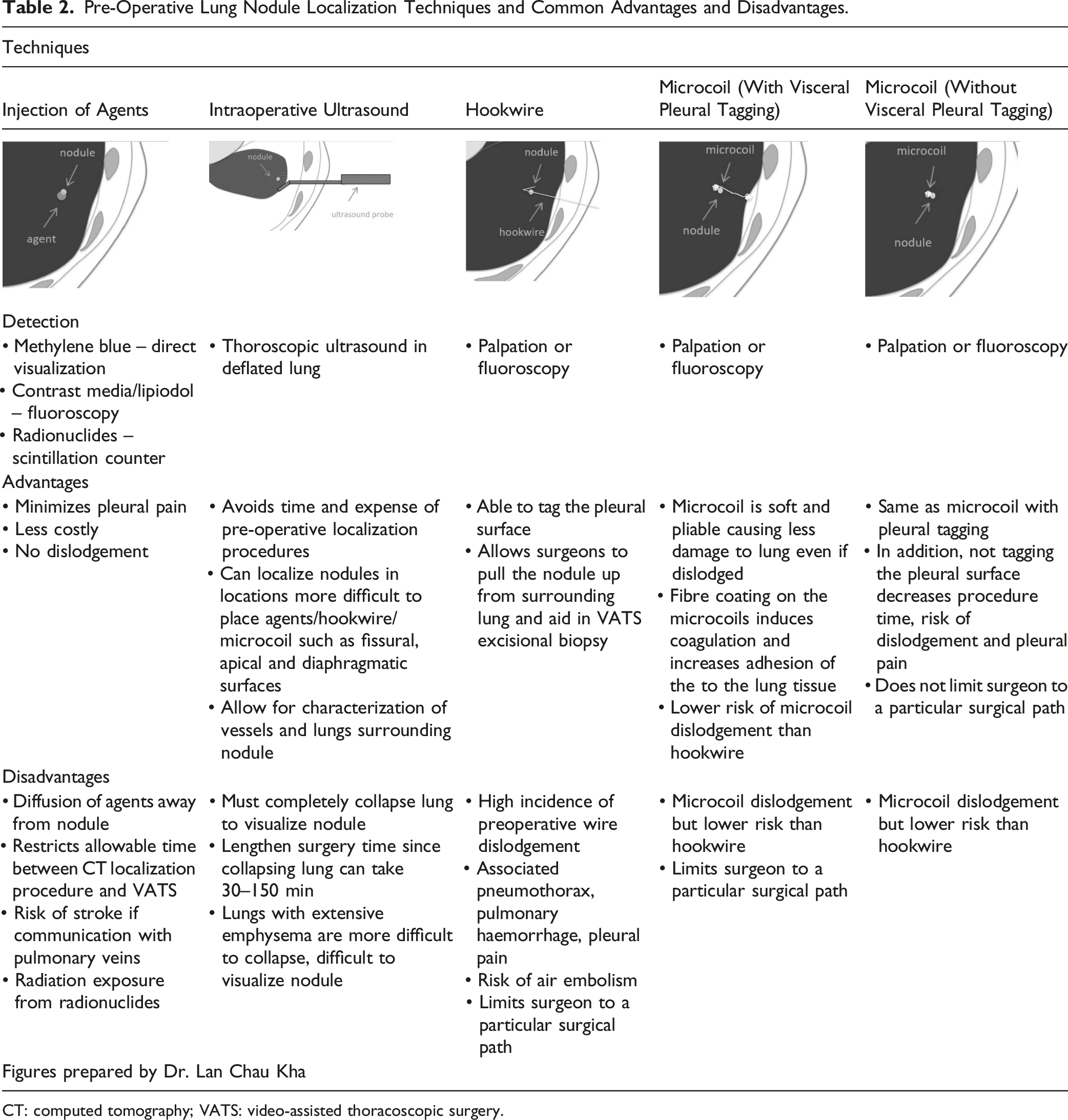
CT: computed tomography; VATS: video-assisted thoracoscopic surgery.
Mayo et al 97 first described the microcoil technique that involves percutaneous placement of one end of a microcoil adjacent to the target lung nodule and the other end marking the visceral pleural surface. 97 This allows the surgeon to localize the microcoil using fluoroscopy in addition to direct visualization of the visceral pleural marker during video-assisted thoracoscopic surgery. 98 A widely-used modification of this technique involves placement of the entire microcoil adjacent to the target nodule or alternatively, with the distal portion of the coil marking the deepest aspect of the nodule and the proximal portion marking the superficial aspect (Table 2). This shortens the CT procedure time, reduces patient radiation exposure while maintaining a similar safety profile. This modification also allows the surgeon to use a preferred surgical trajectory for resection rather than being constrained by the microcoil insertion trajectory. 99 Ideally, the microcoil insertion is followed by surgical resection on the same day. However, this can pose logistical challenges for coordination. In one Canadian centre, >200 microcoils were placed between 1 day and 2 weeks prior to surgery with a similar success rate and safety profile (unpublished data). A systematic review and meta-analysis of hookwire, microcoil and lipiodol nodule localization techniques found microcoil and lipiodol to have the highest (97–99%) successful localization rates with microcoils having the best safety profile. 100 Fiducial marker placement using gold seeds has similar success rates, but vascular embolization has been reported, 101 while microcoil shape and haemostatic fibre coating eliminate this possibility due to promotion of thrombosis. 97 Long-term outcomes and disease-free survival have also been excellent with the microcoil technique. 102
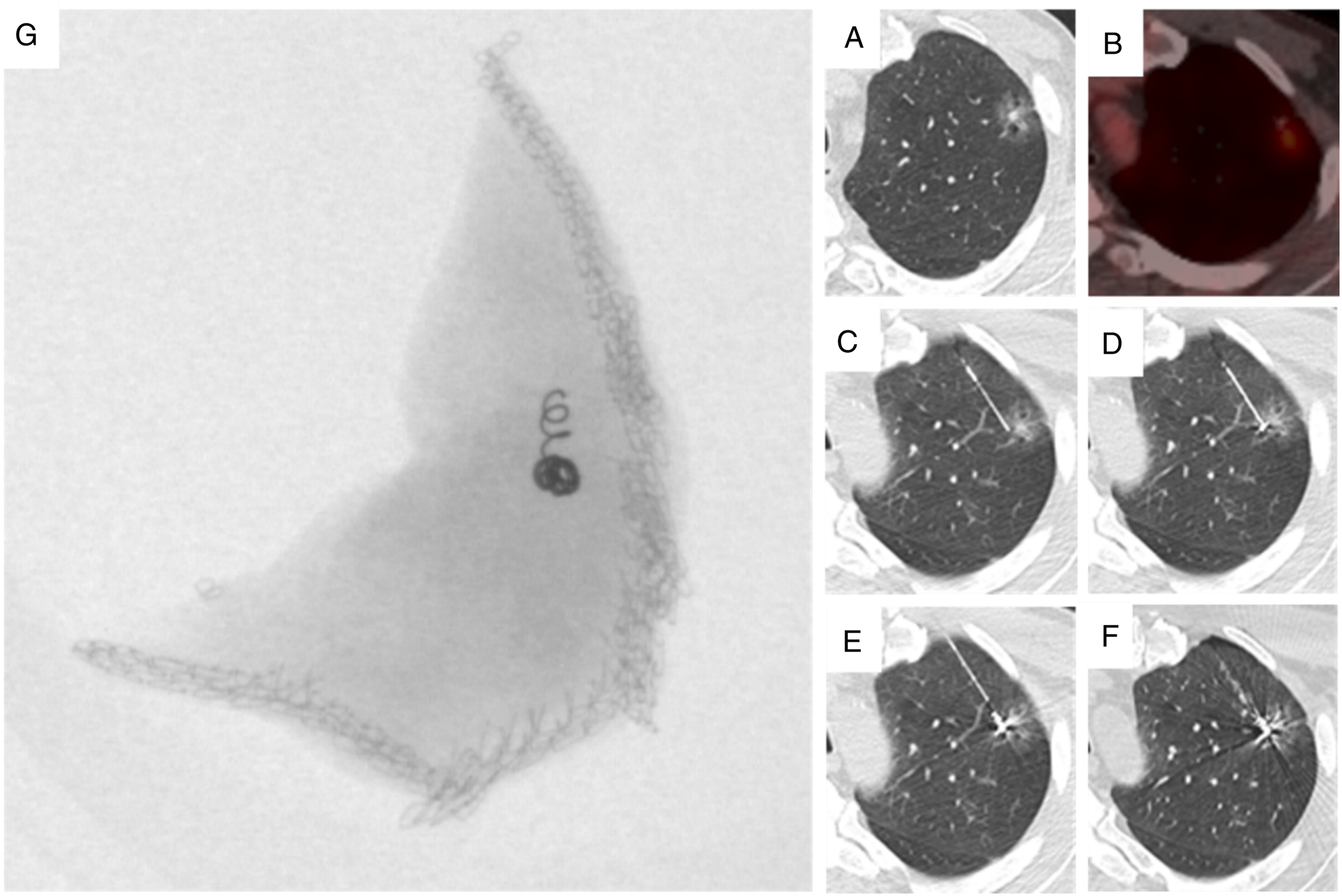
Refer to Figures 4 and 5 for more information about this technique; see Supplementary Appendix D for a video related to this technique. 44-year-old woman with treated breast cancer. (A and B) A new solid nodule was identified on surveillance CT as shown on axial lung windows and coronal MIP images. (C) After a non-diagnostic biopsy, a microcoil was deployed under CT guidance, posterior and medial to the nodule as shown on volume rendered image. (D) Repeat CT chest two weeks later, on the day of resection, demonstrates stable position of the microcoil, with no interval migration or dislodgement (D). The target nodule is shown in red (arrow) and the microcoil is shown in yellow (both C and D). Pathologic evaluation confirmed breast metastasis. CT: computed tomography. 64-year-old man with video-assisted thoracoscopic surgical resection of a left upper lobe pT2a, N0, M0 invasive adenocarcinoma (acinar predominant). (A). A sub-solid left upper lobe nodule (arrow) was detected on lung screening CT. (B). PET/CT revealed this nodule had an SUV of 2.7. The patient underwent pre-operative microcoil localization. (C). A 22-gauge Chiba Sugai needle with centimetre markers (Cook Canada, Stouffville, Ontario, Canada) was removed and placed on the sterile tray. An 80-mm-long 0.018-inch-diameter fibre-coated platinum microcoil (VortX 18, Diamond Shape; Boston Scientific, Cork, Ireland) was loaded into the needle and pushed using the 0.018-inch wire that comes with the microcoil package. A hemostat was then used to mark the pusher wire at two locations: (1) the length necessary to eject 30-mm of the microcoil and (2) the length necessary to eject the entire 80-mm microcoil from the introducer needle. (C) The introducer needle tip was then placed adjacent but deep to the nodule (arrow). (D) The tip of the stiff end of the pusher wire was then placed at the 5-cm mark from the tip of the introducer needle ensuring 3 cm of the microcoil which was ejected. E. As the microcoil position was correct, the remainder of the microcoil was deployed by inserting the stiff end of the guidewire to the second hemostat where the remainder of the coil was deployed in the lung parenchyma (arrow). The empty introducer needle and guidewire assembly were then withdrawn. It is also possible to deploy the entire length of the microcoil at the deep margin of the target nodule, for greater simplicity and faster procedural time. 3D reconstructions help the thoracic surgeons localize the microcoil in relation to the target nodule and plan the size of the resection volume and surgical approach. (F) A post-procedure thin section limited CT was performed to document the final position of the microcoil relative to the nodule and to assess for the presence of haemorrhage/pneumothorax. (G) A perioperative specimen radiograph was obtained confirming microcoil resection. Pathology confirmed negative surgical margins. CT: computed tomography.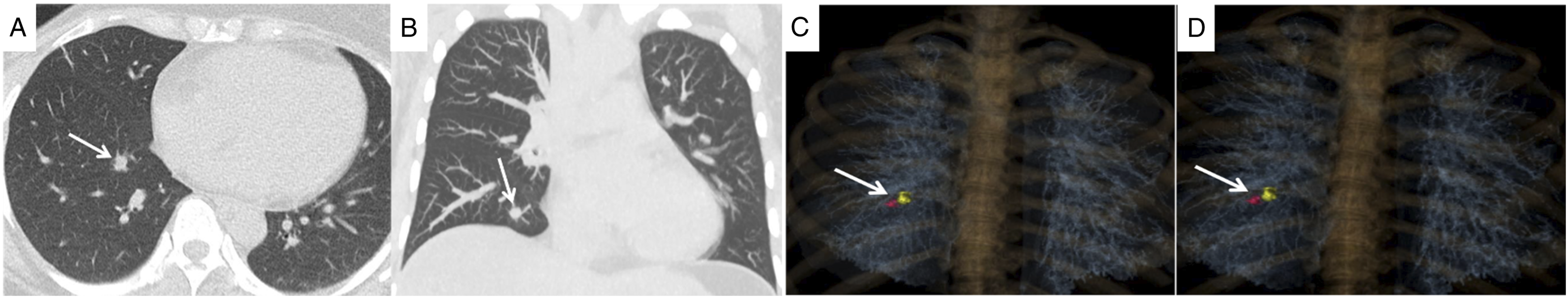
Conclusion
Summary of Key Recommendations.

Supplemental Material
Supplemental Material - Canadian Association of Radiologists/Canadian Association for Interventional Radiology/Canadian Society of Thoracic Radiology Guidelines on Thoracic Interventions
Supplementary Material for Canadian Association of Radiologists/Canadian Association for Interventional Radiology/Canadian Society of Thoracic Radiology Guidelines on Thoracic Interventions by Elsie T. Nguyen, Hamid Bayanati, Casey Hurrell, Matthew Aitken, Edward M. Cheung, Ashish Gupta, Scott Harris, Tony Sedlic, Jana Lyn Taylor, Gerald Gahide, and Carole Dennie in Canadian Association of Radiologists Journal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
