Abstract
This is a visual representation of the abstract.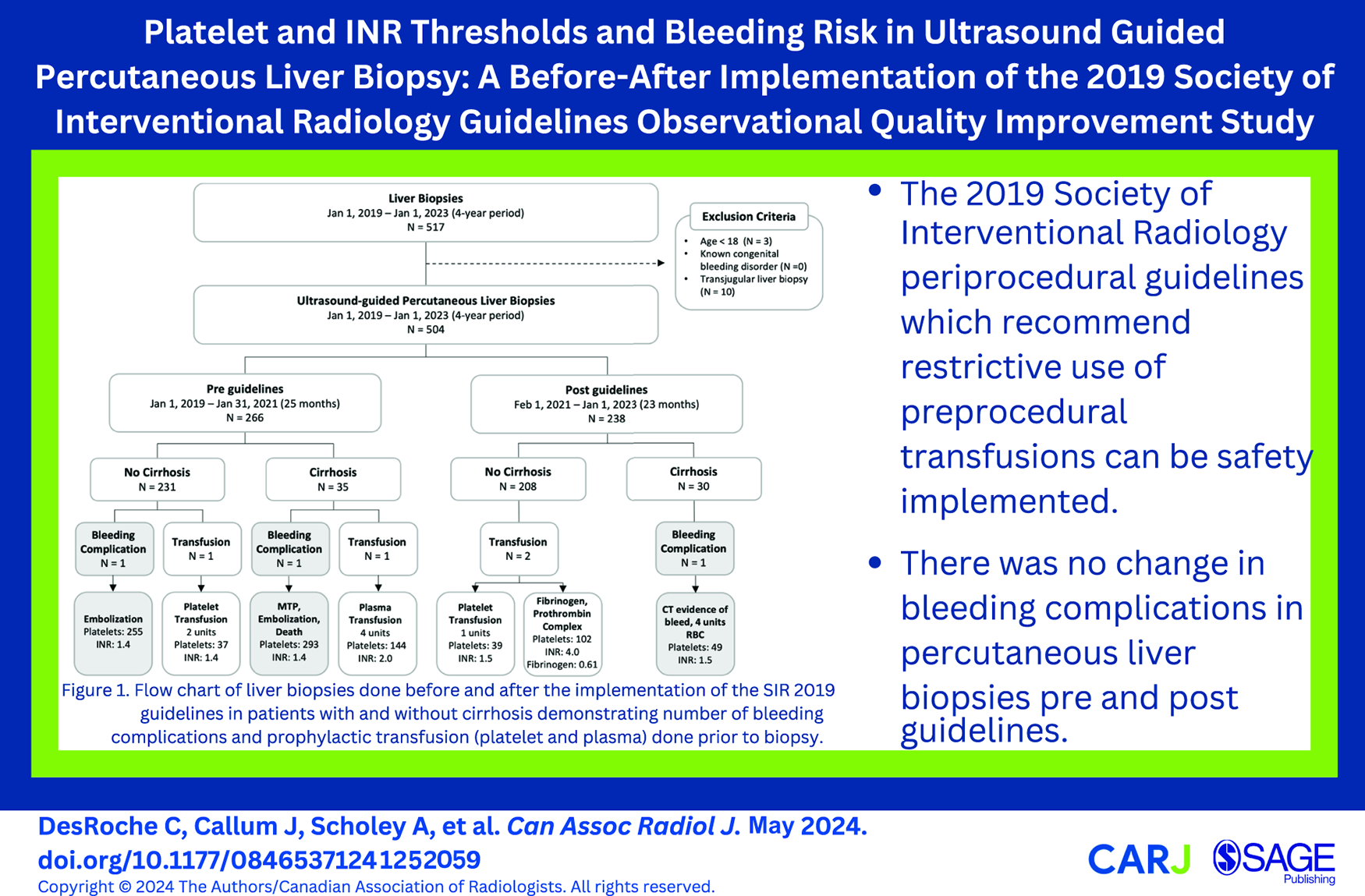
Introduction
Ultrasound guided percutaneous liver biopsy remains the gold standard for establishing a diagnosis and directing the management of acute and chronic liver disease. 1 The procedure is generally well-tolerated with subclinical bleeding occurring in up to 23% of procedures and severe haemorrhage requiring transfusion or other intervention in 0.35% to 0.5% of procedures. 2
Laboratory values such as low platelet counts or high International Normalized Ratio (INR) are thought to increase the risk of procedural bleeding,3,4 however the American Society for the Study of Liver Disease position paper on liver biopsy concluded that there is “no specific PT-INR and/or platelet count cut-off at or above which potentially adverse bleeding can be reliably predicted.” 5 In the absence of clear thresholds, prophylactic transfusion strategies in the context of abnormal platelet counts and INR vary between institutions.
In 2019, the Society for Interventional Radiology (SIR) published consensus-based guidelines recommending restrictive use of pre-procedure transfusion. 6 The guidelines recommend platelet transfusion if pre-procedure platelet count is <50 × 109/L and plasma transfusion if INR is >1.5 to 1.8 for high-risk procedures, of which solid organ biopsies are classified. When compared to the 2012 guidelines, 7 the 2019 SIR periprocedural guidelines were more specific regarding patients with chronic liver disease, permitting a more liberal INR threshold (up to 2.5) and lower platelet threshold (down to 30 × 109/L) for this patient population. Table 1 provides a detailed comparison of the recommended thresholds for transfusion in percutaneous liver biopsies as outlined in the 2012 versus 2019 SIR periprocedural guidelines.6,7
Comparison of the 2012 and 2019 SIR Periprocedural Guidelines Laboratory Thresholds for Percutaneous Liver Biopsy.
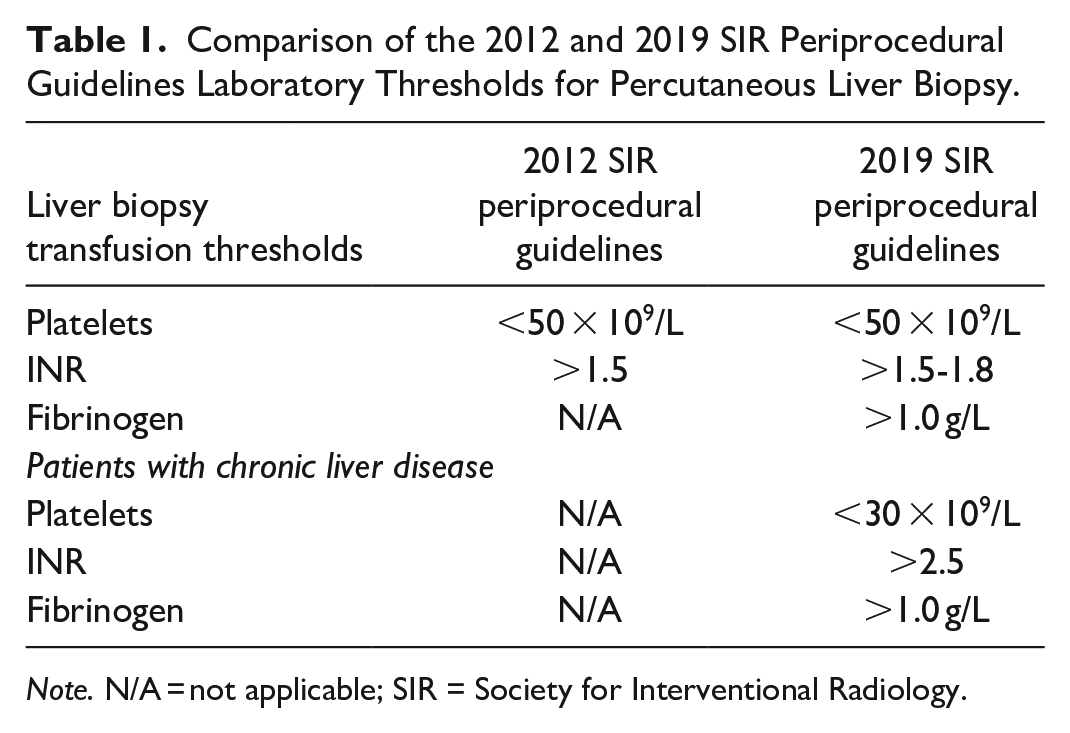
Note. N/A = not applicable; SIR = Society for Interventional Radiology.
The purpose of the present observational study is to describe our institutional experience in the adoption of the 2019 SIR guidelines for periprocedural management of bleeding risk in patients undergoing percutaneous ultrasound guided liver biopsy, comparing major bleeding adverse events and pre-procedural blood product utilization before and after the implementation of the 2019 SIR guidelines.
Methods
A single academic institution retrospective review of all ultrasound-guided percutaneous liver biopsies from January 1, 2019 to January 1, 2023 was conducted. Institutional research ethics board exemption was received for this quality improvement study.
Patient Selection and Characteristics
The study population consisted of patients referred to our tertiary centre for liver biopsy identified by search of the radiology information system (Nuance mPower, MA, USA). Exclusion criteria included patients less than 18 years of age, known congenital bleeding disorder, and transjugular liver biopsies. All other ultrasound-guided percutaneous liver biopsies were included. The identified studies were categorized into 2 groups based on date: those before our institution implemented the 2019 SIR guidelines (Jan 1, 2018-Jan 31, 2021), and those after (Feb 1, 2021-Jan 1, 2023). The date range was selected to allow for consistency in operator, and comparable number of biopsies in each group. Demographic information (age and sex), inpatient versus outpatient status, and indication for liver biopsy were recorded for each case.
Patients were classified into 2 groups depending on the presence or absence of cirrhosis at or before the time of biopsy. Patients with cirrhosis were identified based on (1) Liver biopsy showing F4 fibrosis, (2) Fibroscan kPa value >12; FIB-score >3.25, Non-alcoholic fatty liver disease fibrosis score of >0.676, (3) Clinically diagnosed cirrhosis from liver clinic or GI consult notes, (4) Imaging evidence of portal hypertension in a patient with known chronic liver disease, and (5) History of a liver decompensation (ascites, variceal bleeding, encephalopathy, hepatocellular carcinoma [HCC]) event in a patient with known chronic liver disease. For patients with cirrhosis, the Model for End-stage Liver Disease Sodium (MELD-Na) scores were calculated as a predictor of hepatic dysfunction. 8
Additional clinical information was detailed including use of medications that influence coagulation such as anticoagulants (eg, Warfarin or Non-Vitamin K antagonist oral anticoagulant within 49 hours, antiplatelet agents, non-steroidal anti-inflammatory drugs [NSAID], or selective serotonin reuptake inhibitors [SSRIs]). The Charlson Comorbidity Index 9 —a composite score of age and multiple comorbid conditions was calculated for each patient. Additional clinical features related to increased procedural bleeding risk such as ascites, portal hypertension, and renal dysfunction were recorded. Renal dysfunction was defined as any patient requiring dialysis, or where serum creatinine (Cr) >200 µmol/L.
Preprocedural Workup and Technique
Laboratory parameters obtained within 7-days pre-procedure including serum creatinine, bilirubin, sodium, albumin, aspartate aminotransferase, alanine transaminase, alkaline phosphatase, haemoglobin, ABO-blood group, INR, fibrinogen, and platelet count were recorded. Nadir haemoglobin within 7 days post procedure were also recorded.
If a frozen plasma, platelet, fibrinogen concentrate, or cryoprecipitate transfusion was received 48 hours prior to the procedure, the number of units or grams/international units (g/IU) transfused was recorded. If additional blood work was done post transfusion and prior to biopsy, the platelet count, INR, and fibrinogen were recorded to analyze if guideline parameters were reached.
Liver biopsies were performed by 5 interventional radiologists using real-time ultrasound guidance and an 18-gauge spring-loaded core-biopsy device (Max-Core, Becton Dickinson, Franklin Lakes, NJ). The type of biopsy (targeted vs non-targeted), gauge of needle, number of passes, biopsy location, and technique (coaxial vs non-coaxial), and use of biopsy tract plugging with gelatin sponge slurry (Gelfoam) were recorded.
Chart review was conducted to note any post procedural adverse events within 7 days of the procedure. Major haemorrhagic adverse events were defined as those that required interventional measures, such as transfusion of blood products, embolization, surgery, or resulted in death from haemorrhage, and minor adverse events as those that resolved spontaneously with conservative measures, such as fluid therapy and analgesia. It has been noted that most post biopsy haemorrhages occur within the first 2 to 4 hours post procedure, 10 however a 7-day period was selected to assess for haemorrhagic adverse events to ensure that all events were captured. Data on haemorrhagic event rates was collected from (1) Imaging evidence of subcapsular/perihepatic/intraperitoneal haematoma on subsequent imaging up to 7 days after the biopsy, (2) Follow-up notes describing a haemorrhagic adverse event within 7 days of the procedure, (3) The requirement of interventional measures such as transfusion of blood products, embolization, or surgery within 7 days of the procedure, or (4) Death from haemorrhagic adverse events.
Statistical Analysis
Descriptive statistics were calculated for continuous variables, whereas percentages and relative percentages were calculated for categorical and ordinal variables. A P value of <.05 was considered statistically significant for all analyses. Exploratory and diagnostic plots were used to assess potential violations of statistical assumptions. For comparison of liver biopsies pre versus post implementation of the 2019 SIR guidelines, binary variables were assessed using Fisher’s exact test and Wilcox rank sum test was used for continuous variables. All analyses were performed in R (v4.2.2, R Core Team 2022).
Results
Pre Versus Post Guideline Comparison
A total of 504 percutaneous ultrasound guided liver biopsies were performed on 498 patients during the study time period. Of these, 266 (53%) liver biopsies were performed prior to the implementation of the 2019 SIR guidelines (January 1, 2018-January 31, 2021), and 238 (47%) were performed after (February 1, 2021-January 1, 2023). There were no statistically significant differences in the demographics (Age, Sex, Side of Liver, Type of Biopsy, Blood Group O vs Non-O) between the 2 groups (Table 2).
Demographic Characteristics, Biopsy Information, and Laboratory Values of Patients Receiving Liver Biopsy Pre Versus Post SIR Guidelines Implementation.
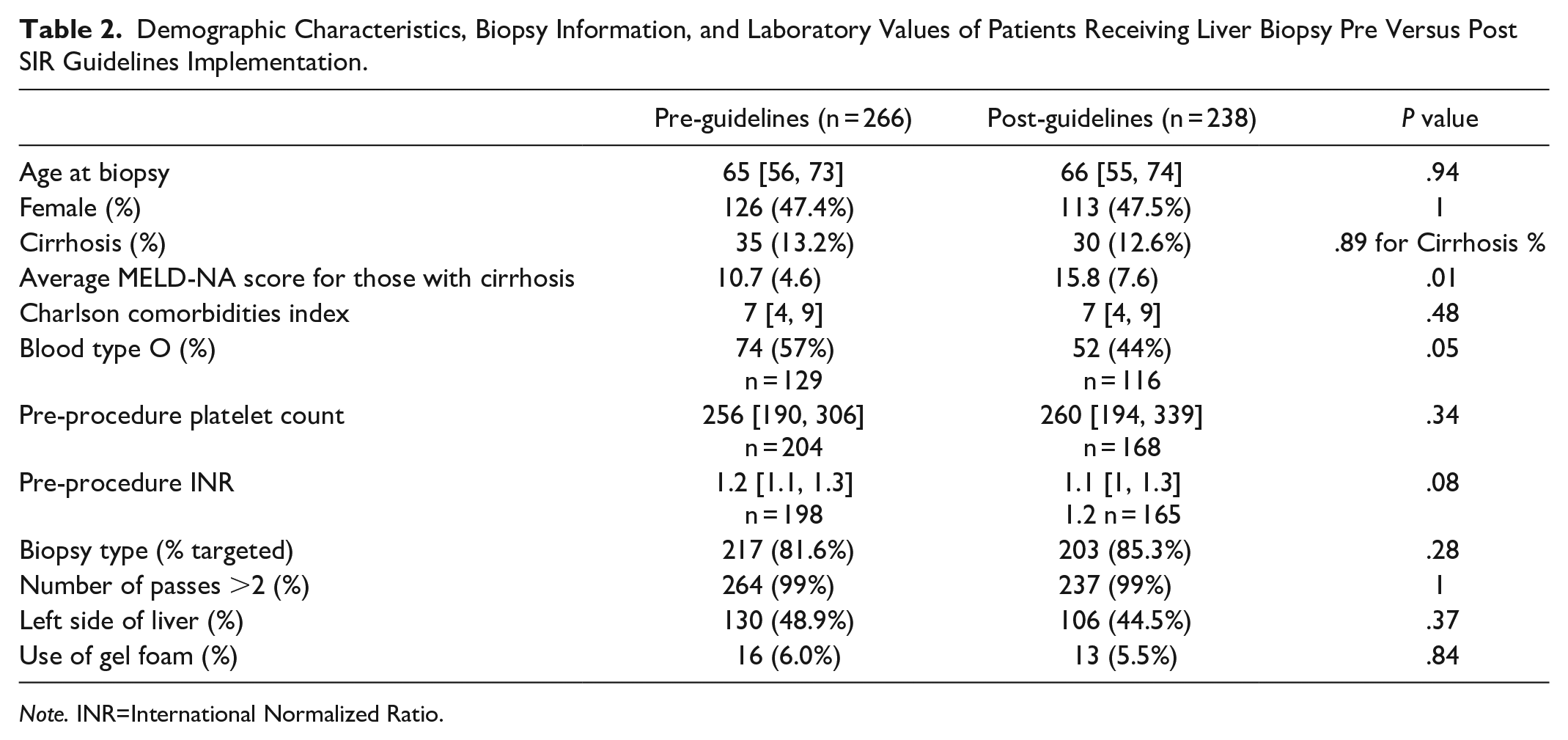
Note. INR=International Normalized Ratio.
A total of 65 patients (12.9%) were classified as having cirrhosis at or before the time of biopsy, of these 35 (54%) occurred prior to the implementation of the SIR guidelines and 30 (46%) after (P = .89). MELD-Na scores were significantly high in patients with cirrhosis in the post guideline period (mean scores 10.7 ± 4.6 pre-guidelines vs 15.8 ± 7.6 post-guidelines; P = .01).
Overall, the frequency of preprocedural blood work within 7 days prior to liver biopsy was 50.4% for platelet count, 48.2% for INR, and 2.2% for fibrinogen (Table 3). There were no statistical differences in the frequency of preprocedural blood work pre-guidelines versus post-guidelines for INR, platelet count, and fibrinogen (P = .19, .13, .28) respectively. Pre-procedural platelet counts were not statistically different (261 ± 112 × 109/L pre-guidelines and 275 ± 127 × 109/L post-guidelines, P = .34) (Table 2). INR also was not statistically different between the 2 groups (1.21 ± 0.20 pre and 1.23 ± 0.36 post, P = .08) (Table 2). Of patients with cirrhosis, 95.4% did not have fibrinogen included in their preprocedural bloodwork.
Frequency of Pre-Biopsy Blood Work Within 7 Days of the Procedure.
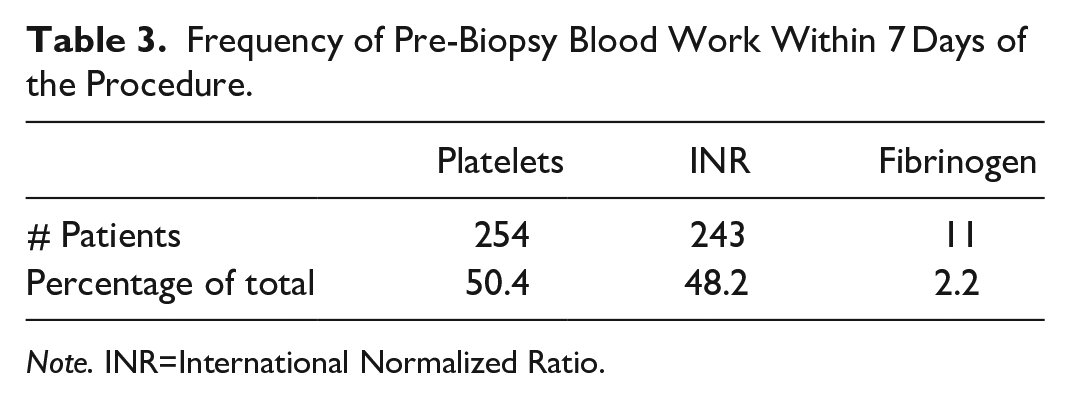
Note. INR=International Normalized Ratio.
Preprocedural Transfusions
Four patients (0.8%) received a preprocedural transfusion of blood products prior to liver biopsy, 2 of which occurred prior to the implementation of the SIR 2019 guidelines and 2 after (Figure 1). All 4 of the preprocedural transfusions were in keeping with the guidelines at the time of the biopsy. No adverse transfusion reactions were reported. None of the patients who received a preprocedural transfusion experienced a bleeding adverse event.
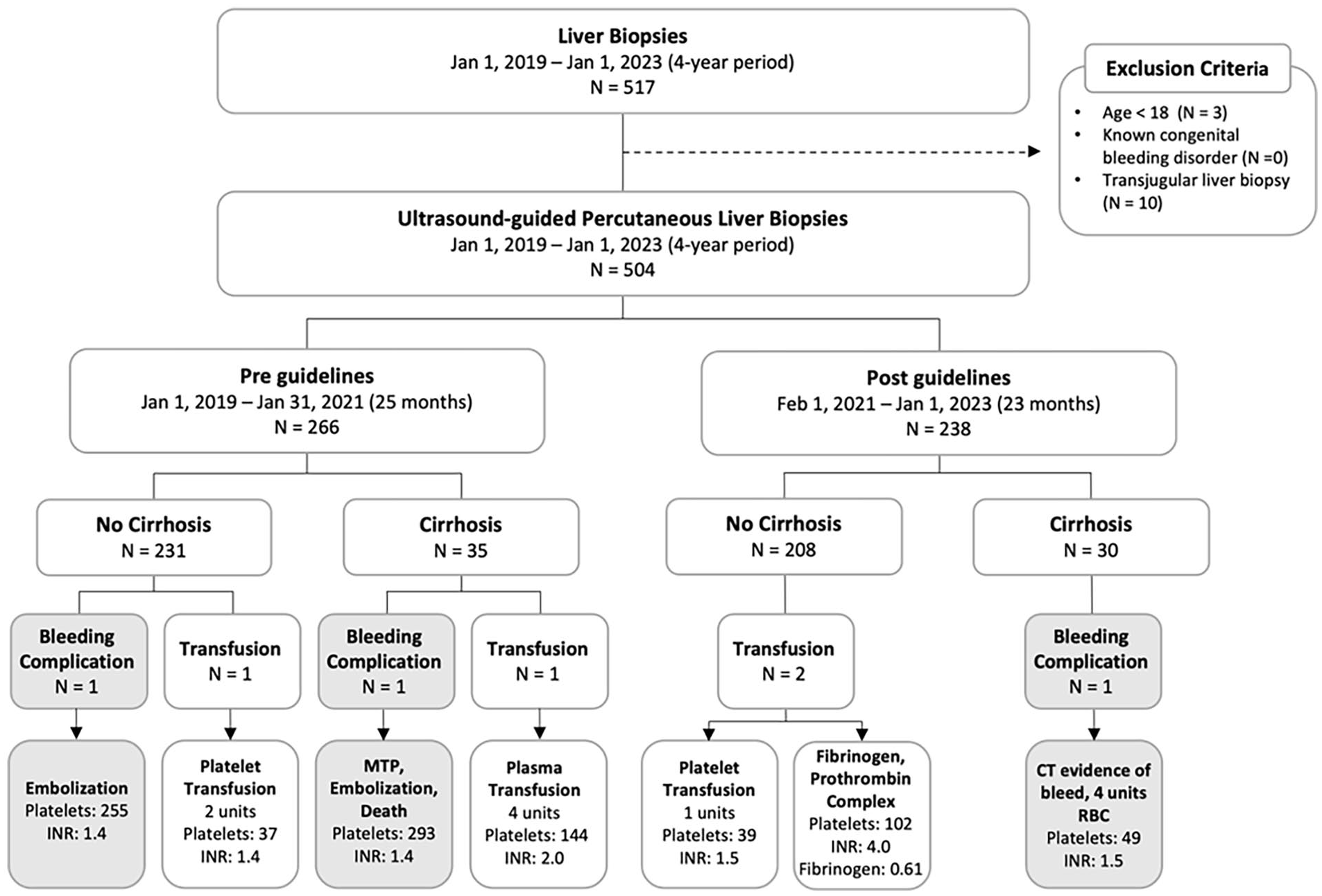
Flow chart of liver biopsies done before and after the implementation of the SIR 2019 guidelines in patients with and without cirrhosis. The number of prophylactic transfusions (platelet and plasma) done prior to biopsy and number of bleeding complications is demonstrated. Platelet count and INR values prior to the transfusion are listed for patients receiving a transfusion prior to biopsy. Platelet count (×109/L) and INR at the time of biopsy are listed for patients who experienced a bleeding complication.
Based on the guidelines at the time of biopsy and the platelet count and/or INR, there were 18 patients (3.6%) who should have received a preprocedural transfusion but did not (Figure 2). Prior to the implementation of the 2019 SIR guidelines, 3 patients should have received a platelet transfusion based on a platelet count of <50 × 109/L, and 11 patients should have received a plasma transfusion based on INR values of >1.5. After the implementation of the 2019 SIR guidelines, there were 3 patients without cirrhosis who should have received a plasma transfusion as their INR was >1.8. No patients received a transfusion in either time period if their platelet count was greater than the SIR guideline recommendations or if their INR was less than the SIR guideline recommendations. Overall non-compliance prior to implementation was 5.3% and post implementation was 1.7% (P = .01).
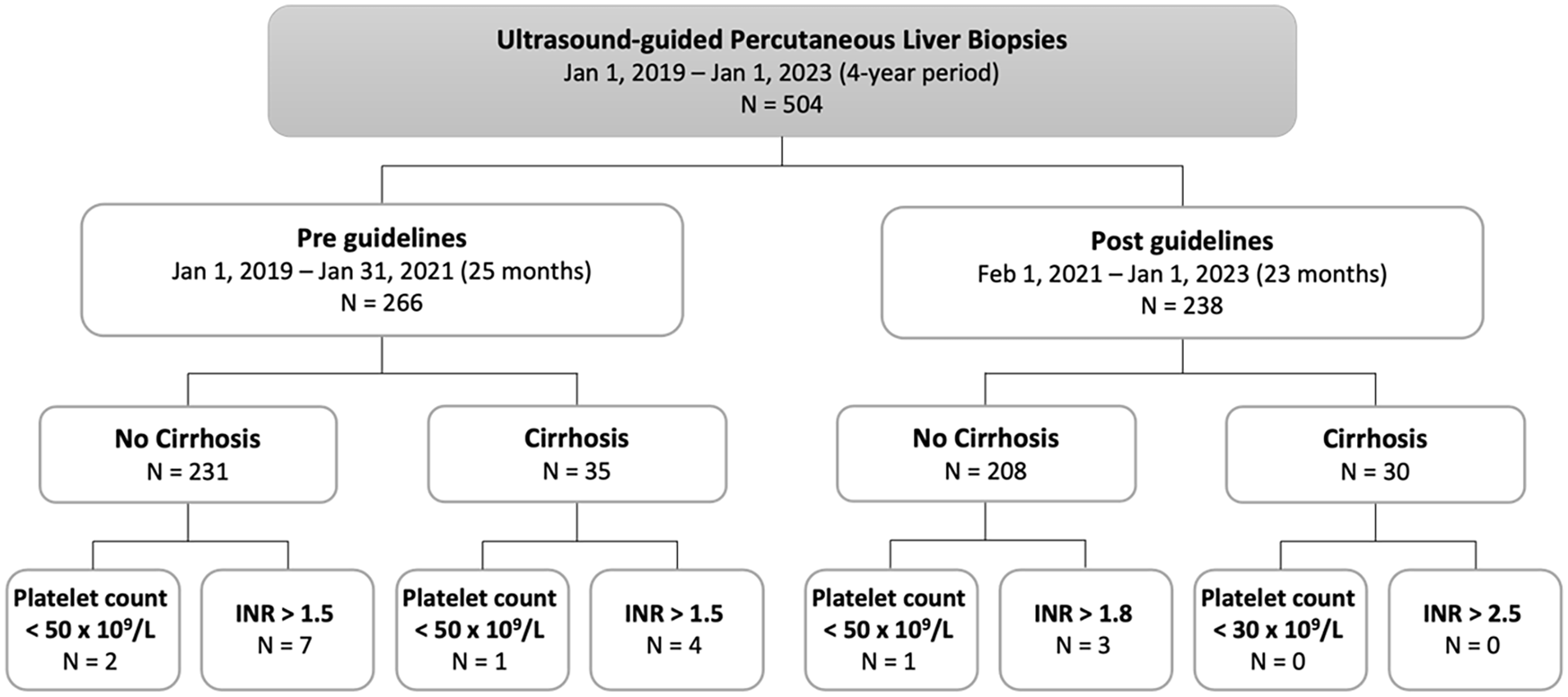
Flow chart of liver biopsies done before and after the implementation of the SIR 2019 guidelines in patients with and without cirrhosis. Guideline non-compliance is demonstrated by the number of patients who should have received a transfusion based on their platelet count or INR for each category. Pre guidelines transfusion for all patients, both those with and without cirrhosis with platelets <50 × 109/L or INR >1.5. Post guidelines platelet transfusions for patients without cirrhosis and platelets <50 × 109/L or INR >1.8 and for patients with cirrhosis platelets <30 × 109/L and INR >2.5.
Bleeding Adverse Events
A major bleeding adverse event resulting from liver biopsy occurred in 3 patients (0.6%), of these 2 occurred prior to the implementation of the SIR 2019 guidelines and 1 after (Figure 1). In the 2 bleeding adverse events occurring before the implementation of the 2019 SIR guidelines, both patients’ platelet count and INR were within the target level with a platelet count of 255 × 109/L and 293 × 109/L, INR 1.4 and 1.4, respectively. One patient underwent successful embolization of the right hepatic artery and was discharged home in stable condition after a 19-day hospital admission. The second patient required activation of a massive haemorrhage protocol, underwent embolization of the left hepatic artery, paracentesis draining 2 L frank blood and died 3 days post liver biopsy as a result of multiorgan failure secondary to the haemorrhagic adverse event.
After the implementation of the 2019 SIR guidelines, one bleeding adverse event occurred in a patient with cirrhosis and platelets of 49 × 109/L who did not receive a platelet transfusion in alignment with the threshold for transfusion post implementation of the 2019 SIR guidelines. The patient received 4 units of red blood cells as a result of the bleeding adverse event and were subsequently discharged home on day 16 post biopsy.
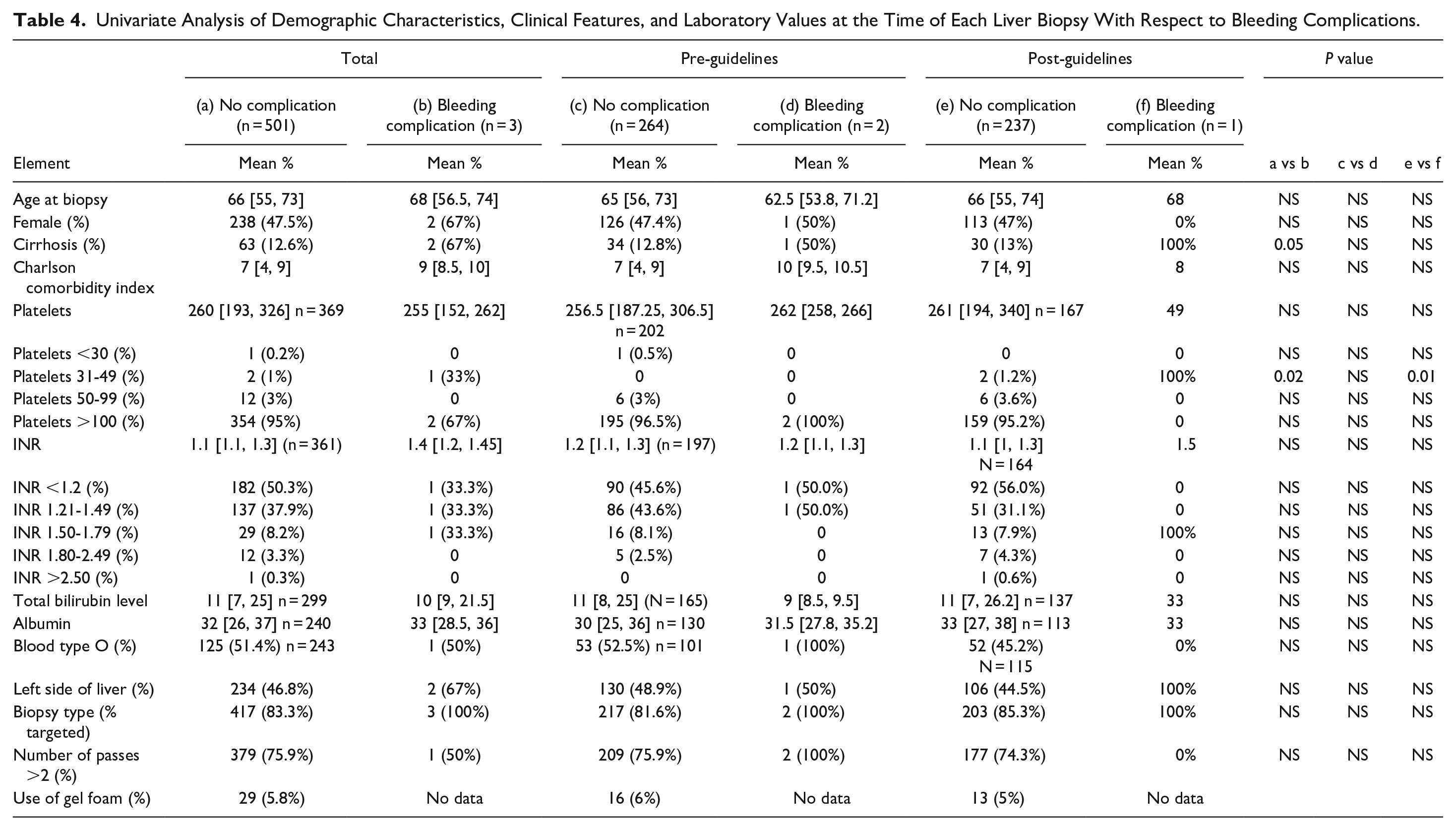
Univariate analysis of demographic characteristics, laboratory values, clinical and procedural features as relates to bleeding adverse events is demonstrated in Table 4. Presence of liver cirrhosis was associated with bleeding complications (12.6% in the no bleeding adverse event group vs 67% in the bleeding adverse event group, P = .047). There were only 3 patients with pre procedural platelets values of 31 to 49 × 109/L, 2 of which also had cirrhosis of which, 1 experienced a bleeding complication.
Univariate Analysis of Demographic Characteristics, Clinical Features, and Laboratory Values at the Time of Each Liver Biopsy With Respect to Bleeding Complications.
Discussion
The rates of transfusion, bleeding adverse events, and non-compliance when applying the 2019 SIR guidelines for periprocedural management of bleeding risks to liver biopsies have not been previously published. We found that the implementation of the 2019 guidelines led to reduced non-compliance of 1.7% compared to the pre-implementation non-compliance rate of 5.3%. This is likely in part due to departmental education efforts for interventional radiologists, interventional radiology nurses, and a nursing patient navigator. Additionally, a standardized booking template according to risk pattern was developed in consultation with our local blood product transfusion expert. This speaks to the importance of local educational efforts in adapting new guidelines or practice changes. Additionally, we found that implementation of the guidelines did not significantly change the rates of pre-procedural transfusion, which were already low at 0.8%. Taken together, we demonstrate that with departmental education, the guidelines can be applied with a low non-compliance rate, while maintaining a low pre-procedural transfusion rate.
Our implementation of the guidelines did not alter the overall adverse event rate, with post implementation bleeding adverse event rate of 0.4%, compared to pre implementation rate of 0.75%. These adverse event rates are comparable to previously reported literature 2 and at the lower margin of the threshold rate suggested by the SIR quality improvement guidelines. 11 However, given the very low bleeding event rates limited comparisons can be made between pre and post guideline implementation representing a limitation to our study. Specifically, a small sample size limits comments on patients with platelets of 30-49 × 109/L.
Kitchin et al 2 previously reported their experience transitioning from the 2012 SIR pre-procedural guidelines to a less stringent threshold of INR <2.0, platelets >25 × 109/L, a threshold more liberal than the 2019 SIR guidelines. In their 9-year retrospective review, involving 1846 liver biopsies in 1740 patients, transitioning to the less stringent threshold resulted in reduction in platelet and plasma transfusion, but did not result in an increase of haemorrhagic adverse events. Their study’s stringent thresholds are not adjusted for chronic liver disease. INR is a poor predictor of bleeding risk in chronic liver disease, and platelet transfusions are less effective in this patient population. 12 There may be risk in plasma transfusion in the chronic liver disease population as these patients are in a prothrombotic state despite their increased INR. 13 Our study supports the separation of the chronic liver disease patients in the SIR 2019 guidelines, as being both practical and safe. We found that 95.4% of patients with chronic liver disease did not have fibrinogen levels included in their preprocedural bloodwork. Hepatic synthesis of fibrinogen is likely to be preserved when the coagulation factor levels are above 30% (INR below 1.5). The utility of including fibrinogen in periprocedural guidelines warrants further study.
This study provides a chronological delineation of pre and post 2019 SIR guidelines, as a point in time in the change of practice at our institution. The pre and post implementation patient populations were comparable in demographics, comorbidities, chronic liver disease, coagulation risk factors such as INR and platelets. Despite its retrospective nature, it is unlikely that significant adverse events would be missed, as transfusions, follow-up imaging, and mortality is easily identified in the medical and electronic laboratory records.
In conclusion, this single-centre data supports the use of the 2019 SIR guidelines for periprocedural management of bleeding risks as being applicable, safe, and requiring limited use of pre-procedural blood transfusions for liver biopsies. This study is limited by its single-centre retrospective nature. Further demonstration would require multi-centre evidence.
Footnotes
Abbreviation
SIR Society for Interventional Radiology
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author Alexandre Menard receives a speaker honorarium from Inari Medical and Philips Imaging and has received payment for expert testimony from the College of Physicians and Surgeons on Ontario. Author Jeannie Callum has received research grants from Canadian Blood Services and Octapharma and participates on a Drug Safety Monitoring Board for the FEISTY Trauma Trial. Author Emidio Tarulli receives consulting fees from the Medfall Group. The authors declare that they have no conflict of interest not listed above.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Queen’s University Department of Diagnostic Radiology/AIMS radiology group. The funding agencies had no role in the design of this study, collection, analysis, and interpretation of data or in writing the manuscript.
