Abstract
Circadian disruption is pervasive in modern society and associated with increased risk of disease. Chronic jet lag paradigms are popular experimental tools aiming to emulate human circadian disruption experienced during rotating and night shift work. Chronic jet lag induces metabolic phenotypes tied to liver and systemic functions, yet lack of a clear definition for how rhythmic physiology is impaired under these conditions hinders the ability to identify the underlying molecular mechanisms. Here, we compared 2 common chronic jet lag paradigms and found that neither induced arrythmicity of the liver and each had distinct effects on rhythmicity. Instead, more frequent 8-h forward shifts of the light schedule induced more severe misalignment and non-fasted hyperglycemia. Every other day shifts eventually uncoupled behavioral and hepatic rhythms from the light cycle, reminiscent of free-running conditions. These results point to misalignment, not arrhythmicity, as the initial disturbance tied to metabolic dysfunction in environmental circadian disruption and highlight considerations for the interpretation and design of chronic jet lag studies.
Keywords
In modern society, maintaining a robust circadian rhythm is a challenge. Everyone experiences some form of circadian disturbance throughout life (e.g., rotating shift work, light pollution, mistimed meals, social jet lag, sleep deprivation), and often disturbances are unavoidable. Approximately 20% of workers in industrialized societies work in rotating, night, or alternative shifts (Sweileh, 2022). A major consequence of rotating shift work is misalignment of eating and sleeping patterns with the natural light-dark cycle, a biological conflict that increases risk for metabolic syndrome (Chaput et al., 2023; Mentzelou et al., 2023), cardiovascular disease (Morris et al., 2016), neurological disease (Cho, 2001), and certain cancers (Cordina-Duverger et al., 2018). Understanding the molecular underpinnings of circadian disturbances can facilitate the development of interventions to combat the associated health impairments.
Variable light schedule paradigms have been developed in mice to model human rotating shift work (reviewed in Opperhuizen et al., 2015). The paradigms consist of large, repetitive shifts of the light-dark cycle forward (and/or backward) in time, not unlike serial flights to distant time-zones, hence the term chronic jet lag (CJL). CJL is a popular tool in the chronobiology field and is being employed to better understand the relationship between the circadian clock system and metabolism (Kettner et al., 2015; Casiraghi et al., 2016; Desmet et al., 2021), metabolic disease (Kettner et al., 2016; Xie et al., 2017; Padilla et al., 2024), cancer (Filipski et al., 2004; Lee et al., 2010; Chun et al., 2022), cognition (Gao et al., 2020; Acosta et al., 2023; Kumari et al., 2024), neurological disease (Horsey et al., 2019; Otsuka et al., 2020; Siddique et al., 2022; Liu et al., 2024), the immune response (Castanon-Cervantes et al., 2010; Liu et al., 2018; Cheng et al., 2022), intestinal homeostasis (Li et al., 2021; Liu et al., 2021; Desmet et al., 2023), the microbiome (Thaiss et al., 2014; Voigt et al., 2014; Zheng et al., 2023), and aging (Davidson et al., 2006; Xu et al., 2009; Inokawa et al., 2020), among others. However, lack of a clear biological definition as to how CJL impacts rhythmic physiology is limiting the ability to glean deep insights. This is evident from the literature; CJL is described to induce circadian “disruption,” “dysfunction,” “dysregulation,” “disorganization,” “desynchronization,” “misalignment,” or “reprogramming” (Vetter, 2020).
By impinging on the clock system in one or more ways, different iterations of CJL are shown to induce metabolic impairments such as increased serum triglycerides (Casiraghi et al., 2016; Xie et al., 2017), decreased insulin sensitivity (Zhong et al., 2019; Trebucq et al., 2023), leptin resistance (Kettner et al., 2015), alterations to glucose tolerance and blood glucose level (Zhong et al., 2019; Trebucq et al., 2023), body weight gain (Casiraghi et al., 2016; Trebucq et al., 2023; Koike et al., 2024), and hepatic steatosis (Fleet et al., 2016; Koike et al., 2024; Padilla et al., 2024). These phenotypes are likely the result of multifaceted impairments to rhythmic physiology (central and peripheral clocks) and are likely to involve the liver, a central regulator of systemic metabolism that houses a clock controlling carbohydrate and lipid homeostasis (Daniels et al., 2023). It is difficult to interpret phenotypic outcomes without a more precise definition for how CJL affects behavioral and molecular rhythms.
In this study, we sought to define the state of the liver clock, central clock-driven behavior, and systemic metabolism, under 2 common CJL paradigms. Of the 2 CJL paradigms tested, neither induced arrhythmicity in the liver and each had distinct effects on rhythmicity. Every other day shifts for 2 months uncoupled rhythms from the light cycle not unlike free-running conditions. More frequent shifts of the light schedule induced more severe misalignment and stronger impairment of non-fasted glycemia. These results provide important considerations for the interpretation and design of CJL studies and may ultimately help pinpoint precisely the clock mechanisms underlying metabolic dysfunction.
Materials and Methods
Animals
Animal experiments were conducted in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals and all experiments were conducted with approval from UTHSCSA’s local Institutional Animal Care and Use Committee (IACUC). ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines were also followed wherever possible. Male C57BL/6J mice were purchased from The Jackson Laboratory (Strain #: 000664) and entered the study between the ages of 4 and 8 weeks so that all mice were ~16 weeks old at completion. Mice were fed ad libitum chow diet (7912 Teklad Irradiated LM-485 Mouse/Rat Diet) and were group housed except for locomotor activity experiments, which were performed on a separate cohort of single-housed mice to enable circadian parameters to be calculated for each individual animal.
Light Schedules, Entrainment, and Precise Timing of Measurements
Upon arrival, mice were allowed a period of 2 weeks to acclimate to the animal facility and circadian entrainment room. Mice were housed in standard home cage environments inside circadian cabinets (Actimetrics) at room temperature (23.9 °C ± 1.5 °C) and humidity monitoring (~30%). Light intensity, measured with a luminometer, inside the cabinet at cage level was ~100 lux. All lights schedules and shifts were fully automated using the programmable lighting feature of the cabinets. Thus, mice were only disturbed for standard husbandry activities (e.g., change bedding, supply food and water). Bedding was changed every 2 weeks. All light schedules consisted of daily periods of 12 h of light and 12 h of dark. For CJL paradigms, on shift days, mice experienced longer or shorter exposures to light and dark to accommodate the shifts as shown in the schematics in Figure 1. Mice between 4 to 5 weeks-of-age were randomly assigned to the following paradigms: (1) control 12 h light:12 h dark (LD); (2) 8-h advance twice per week for 3 months (8-h adv 2×/wk-CJL); (3) 8-h advance every other day for 2 months (8-h adv e.o.d.-CJL). Constant light was employed as a control known to disrupt rhythmicity (Hamaguchi et al., 2015; Yamamuro et al., 2020). The precise timing of each measurement as it relates to the CJL schedule is shown in Supplemental Tables S1 and S2. Tissue was collected during CJL on what we reasoned to be a representative day that takes into account the effect of the shift but also allows the mice a re-entrainment opportunity before the next shift. Accordingly, 8-h adv 2×/wk-CJL mice were sampled in between shifts (2 days post shift) and 8-h adv e.o.d.-CJL mice were sampled on the day of the shift, which is 2 days after the previous shift, and which is the longest period that the mice have a chance, if any, to re-entrain before the next shift occurring later that day. Note that tissue harvest was performed when light schedules aligned with working hours, which meant that tissue harvest occurred on day 61 for 8-h adv e.o.d.-CJL and day 85 for 8-h adv 2×/wk-CJL (chosen instead of day 95, which was the next opportunity to harvest tissue). Food intake was measured on the day of the shift. Body weight and body composition measurements were taken during light hours, assuming that these parameters exhibit negligible diurnal rhythmicity.
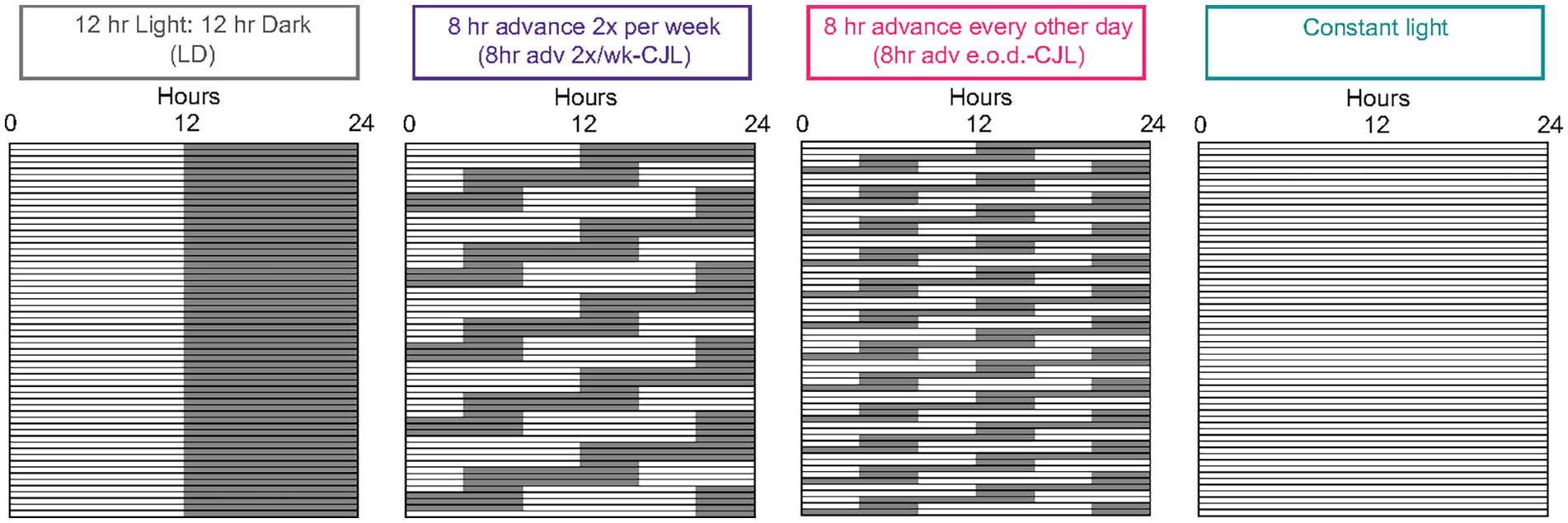
Light schedule paradigms of the study. Male C57BL/6J mice starting at 4-5 weeks-of-age were exposed to chronic jet lag (CJL) schedules, which were based on commonly published paradigms: (1) control 12 h light:12 h dark (LD); (2) 8-h advance twice per week for 3 months (8-h adv 2×/wk-CJL); (3) 8-h advance every other day for 2 months (8-h adv e.o.d.-CJL). Constant light was included as control known to disrupt rhythmicity. An example portion of each paradigm is illustrated so that the light schedule shifts can be appreciated. See each figure legend for sample sizes.
Blood Glucose Measurement
Non-fasted blood glucose was measured at the time of tissue harvest using a hand-held blood glucose meter (Nova Max). LD—
Body Composition
Body composition was determined using a QMR machine (Echo Medical System), which utilizes nuclear magnetic resonance (NMR) to reliably and accurately scan (<2 min) the physical state of tissue of a mouse in a plastic restrainer tube. Measurements were taken when light cycles aligned with human light hours.
Western Blot
Western blot was performed as described previously (Smith et al., 2023b). Briefly, livers were homogenized in RIPA buffer supplemented with protease inhibitors, incubated on ice for 10 min and then sonicated. Samples were then centrifuged at max speed at 4 °C for 10 min and protein concentration was determined in the supernatant by BCA assay. Protein (20-40 ug) was separated on an SDS-PAGE gel, transferred to a PVDF membrane, and blocked with 5% milk in TBST at room temperature for 1 h. Primary antibodies were diluted in 5% milk TBST and incubated with blots overnight at 4 °C (PER2—Cat# PER21-A, Alpha Diagnostic; REV-ERBα-Cat# 13418 [E1Y6D], Cell Signaling). Total protein was visualized with TGX Stain-Free gel technology (Bio-Rad). Blots were thoroughly washed then incubated with secondary antibodies conjugated to HRP (Millipore Sigma, AP160P and 12-348) for 1 h at room temperature. Blots were again thoroughly washed, incubated with HRP substrate, imaged with a ChemiDoc Imaging System (Bio-Rad) and quantified using ImageJ software.
Quantitative PCR
The RNeasy Plus Mini Kit (Qiagen, 74136) was used to extract RNA from
Locomotor Activity and Circadian Analyses
Locomotor activity of single-housed mice (
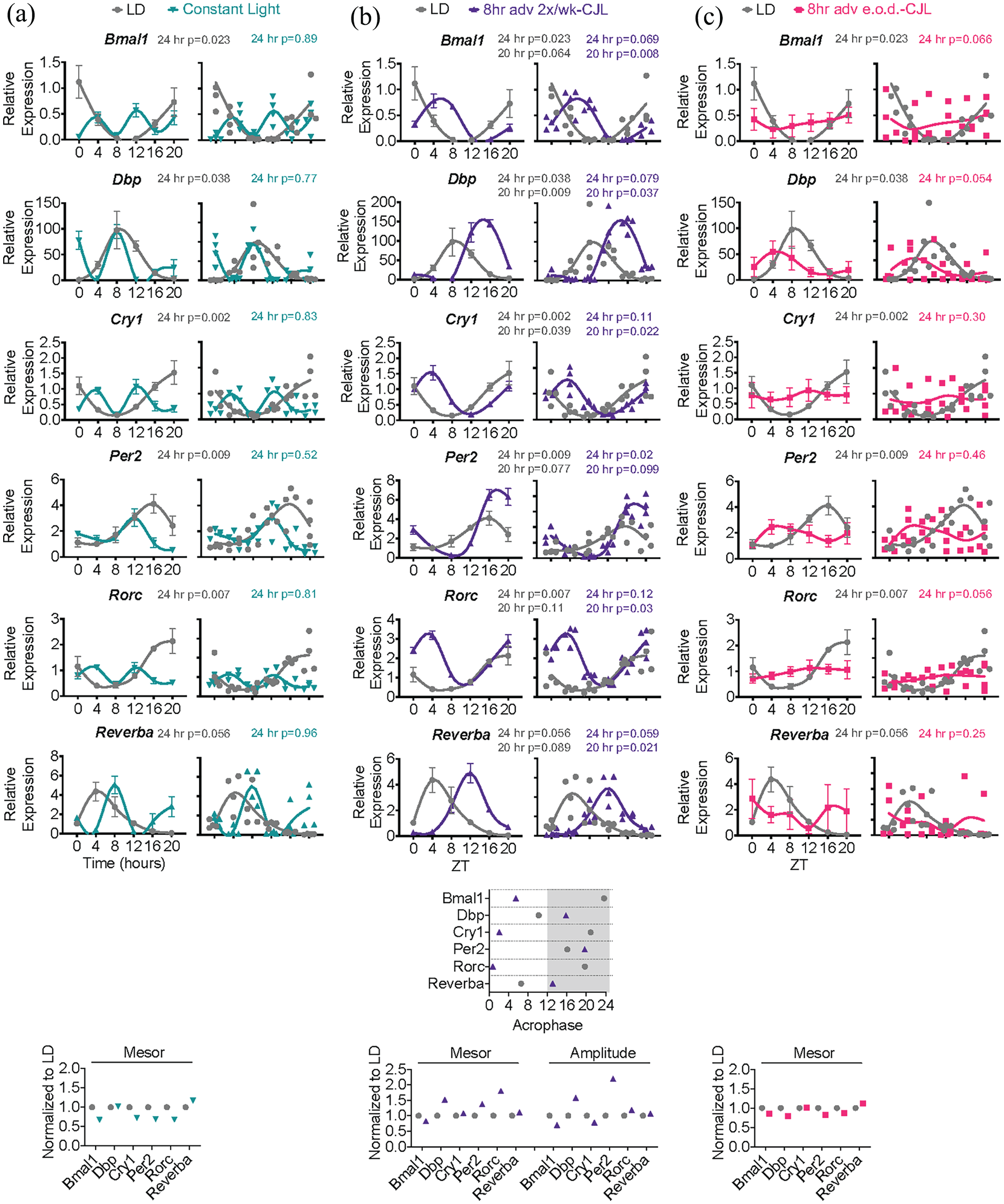
Chronic jet lag paradigms distinctly misalign the liver clock. (a-c) Mice were subjected to light schedules as in Figure 1. Top—expression of clock genes by qPCR in the liver at 6 timepoints over 24 h (
Statistical Analyses
All data are mean ± SEM unless otherwise indicated. For each experiment, sample size, statistical test, and significance threshold can be found in the figure legend and/or main text. Circadian statistical analyses are described within the corresponding methods section. Field standards were used to determine appropriate sample sizes for circadian experiments. Data were analyzed in Prism 6.0 software (GraphPad). Statistical assumptions were assessed for the appropriateness of ANOVA. Kolmogorov-Smirnov tests were used to assess Gaussian distribution (normality). Bartlett’s tests were used to assess homogeneity of variance. Observations within each group were independent. Mice were randomly assigned groups and sampled. All data passed these assumptions for ANOVA except for period data for locomotor activity. For this data, the non-parametric Kruskal-Wallis test was applied. Period data had a significant outlier by Grubb’s test that was noted in the figure legend and left in for transparency. F values and degrees of freedom are reported for each statistical test in the corresponding figure legend as F (DFn, DFd).
Data Availability
Data generated in this study has been deposited to the BioStudies database (Sarkans et al., 2018), accession number S-BSST1673.
Results
Frequent 8-h Forward Shifts Induce Free-Running-Like Conditions in the Liver
A literature search revealed more than 20 CJL paradigms with differences in the amount, direction, frequency, and duration of light schedule shifts, as well as differences in methodology for how shifts were imposed, for example, programmable light schedule shifts versus physically moving cages between rooms. Commonly used paradigms involved 8-h forward shifts (advances) of the light schedule for a period of 1 to 4 months. For this study, 3 paradigms were investigated in adult male C57BL/6J mice using programmable circadian cabinets: (1) control 12 h light:12 h dark (LD); (2) 8-h advance twice per week for 3 months (8-h adv 2×/wk-CJL); (3) 8-h advance every other day for 2 months (8-h adv e.o.d.-CJL) (Figure 1). Constant light was also employed as a paradigm known to disrupt rhythmicity (Hamaguchi et al., 2015; Yamamuro et al., 2020). Constant light actograms can be found in Supplemental Figure S1. The precise timing of each measurement as it relates to the CJL schedule is detailed in Supplemental Tables S1 and S2. Tissue was collected during CJL on a representative day that we reasoned takes into account the effect of the shift but also allows the mice a re-entrainment opportunity before the next shift.
To probe the impact of CJL on liver clock function, livers were harvested at 6 diurnal timepoints. Expression of
In contrast, an apparent loss of rhythmicity was observed under 8-h adv e.o.d.-CJL; however, large error bars were noticeable despite mesor values comparable to LD, and further inspection revealed that individual data points spanned the minimum and maximum of LD control values. Working under the assumption that liver transcripts reflect the phase of the animal, these data indicate that rhythmicity was not lost, but rather phase dispersion between animals was occurring (Figure 2c). Phase dispersion refers to the phenomenon observed in free-running conditions wherein mice retain rhythmicity yet lose phase coherence among one another (i.e., inter-individual desynchrony), due to differences in endogenous circadian period (Eckel-Mahan and Sassone-Corsi, 2015). Although phase dispersion between animals is a likely explanation for these data, it cannot be ruled out that the phase dispersion is occurring only at the level of the liver, and that central clocks are actually phase aligned between mice, as is a possibility if internal desynchrony between the central clock and liver clock occurs within one animal.
Whole cell extracts from 36 mice were used to test whether protein abundance mirrored the RNA findings (Figure 3). As expected, PER2 and REV-ERBα protein accumulated consistently at specific times of day in LD control livers. Protein accumulation from the 18 8-h adv e.o.d.-CJL samples followed no observable 24 h pattern even though individual data points were similar to the minimum and maximum expression detected in LD control and average daily expression was comparable with LD, indicating that livers of individual mice were still rhythmic, but animals were phase dispersed. Together, these data show that liver clock rhythmicity is retained yet misaligned in CJL, with consistent misalignment in 8-h adv 2×/wk-CJL and free-running-like conditions in 8-h adv e.o.d.-CJL.
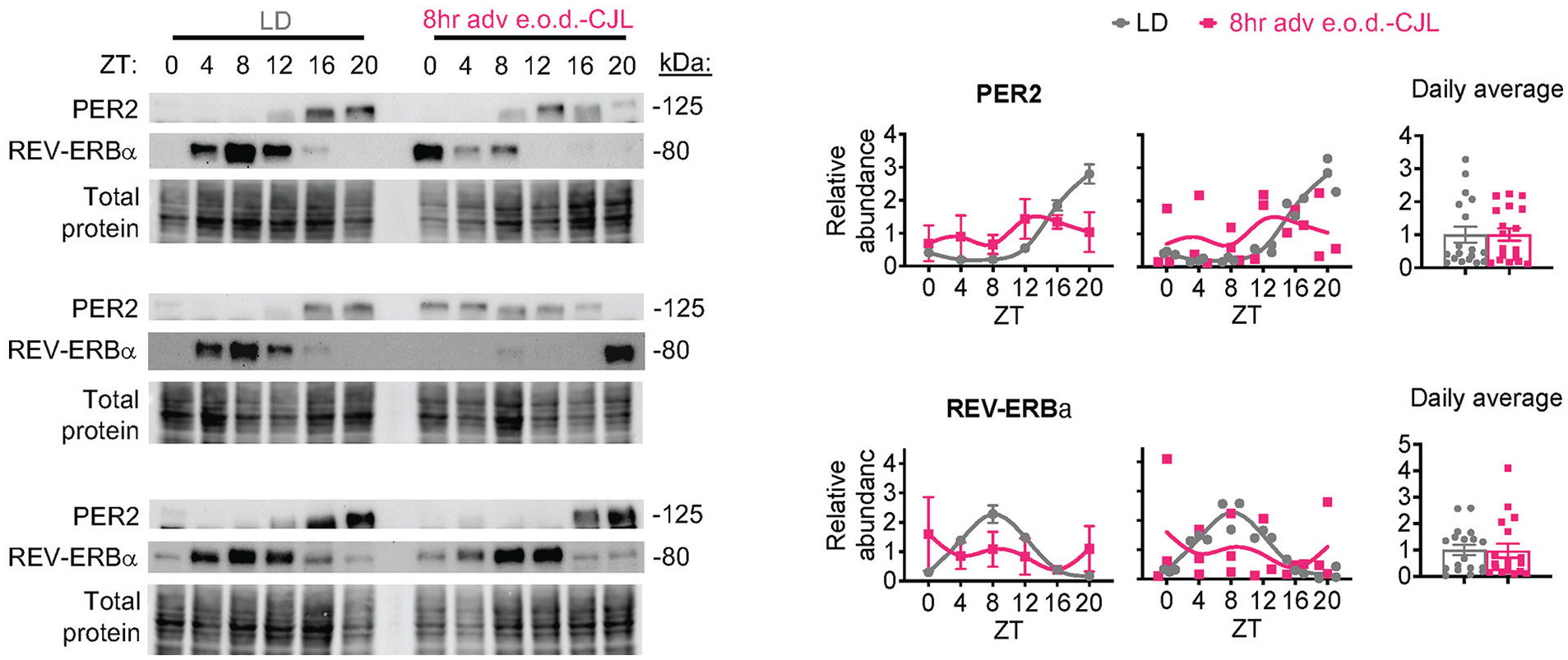
Frequent 8-h forward shifts induce a free-running-like liver clock. Mice were subjected to every other day chronic jet lag (8-h adv e.o.d.-CJL) as in Figure 1. Western blot analysis of liver whole cell lysates (
Frequent 8-h Forward Shifts Uncouple Behavioral Rhythms From the Light-Dark Cycle
To determine the effects of CJL on central clock-driven behavior, ambulatory locomotor activity was analyzed with chi-square and periodogram tests (Brown et al., 2019) (Figure 4a). All mice regardless of light schedule retained rhythmicity. The 2 CJL paradigms had overlapping and distinct effects on rhythmic parameters. CJL reduced periodogram amplitude by 68.8% and 49.7% in 8-h adv 2×/wk-CJL and 8-h adv e.o.d.-CJL, respectively (Figure 4b), indicating that the strength of rhythmicity was blunted in both paradigms. Both CJL paradigms induced misalignment of locomotor activity with the light-dark cycle, evidenced by a blunting of activity during lights-off and an increase in activity during lights-on (Figure 4c). Total activity tended to be lower in CJL but was not statistically significant (Supplemental Figure S1). Circadian period was markedly different between the 3 light schedules. 8-h adv 2×/wk-CJL shortened period (22.72 ± 1.26 SD) whereas 8-h adv e.o.d.-CJL lengthened period (25.33 ± 0.11 SD) (Figure 4d).
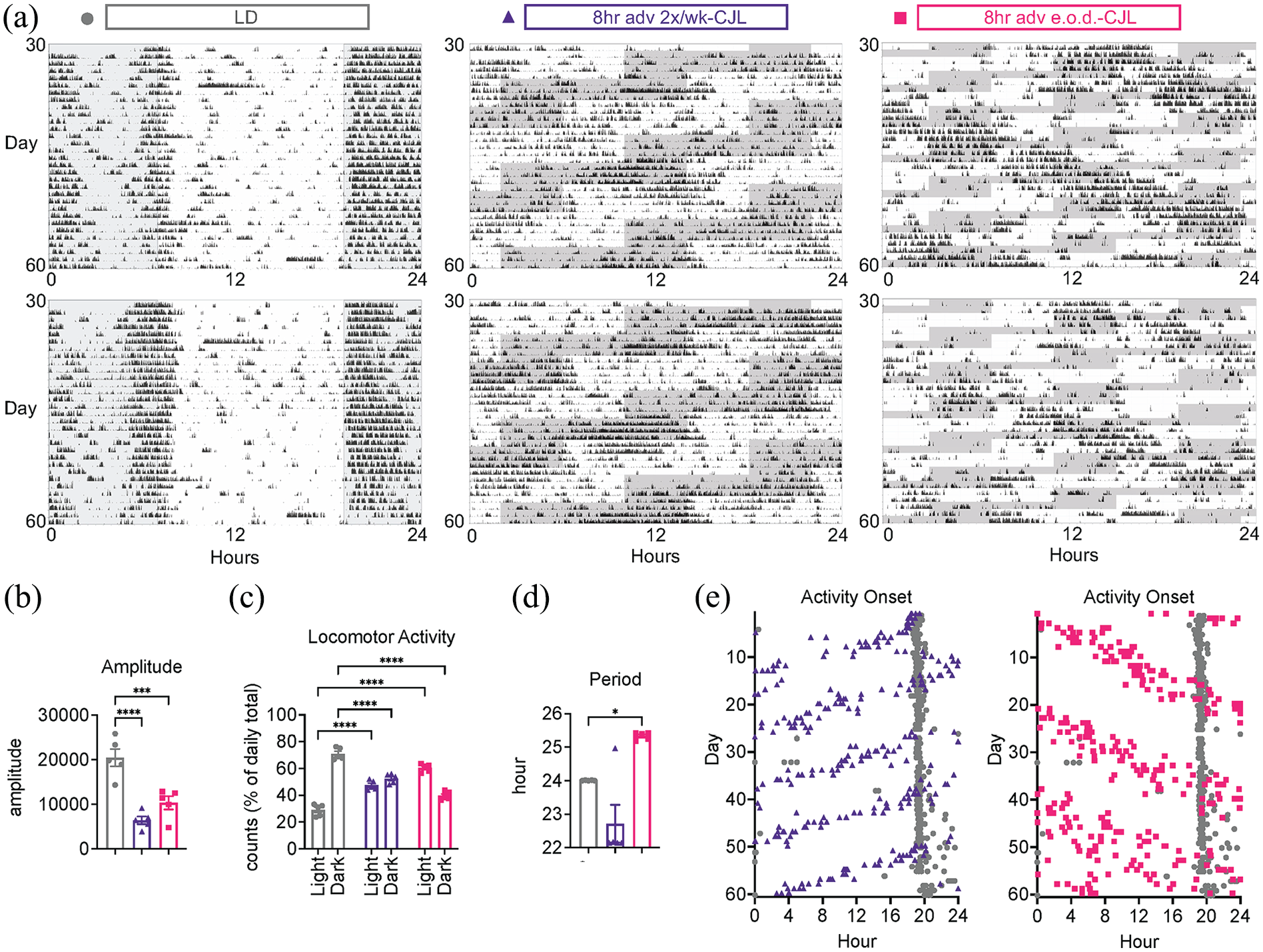
Frequent 8-h forward shifts uncouple behavioral rhythms from the light-dark cycle. Mice were subjected to light schedules as in Figure 1. (a) Two representative actograms from each paradigm. (b-d) Analysis of locomotor activity from day 30 to 60 as in (a) (
Activity onsets were plotted to assess the phase of each animal and interrogate how mice behaved under the light schedule shifts over 2 months (Figure 4e) (Brown et al., 2019). Activity onsets of mice in 8-h adv 2×/wk-CJL advanced with advancements of the light schedule, fitting with a shortened period. Conversely, activity onsets of 8-h adv e.o.d.-CJL mice became delayed and did not follow advancements of the light schedule, fitting with a lengthened period. Eventually, mice in 8-h adv e.o.d.-CJL became uncoupled from the light cycle, evidenced by highly variable activity onsets reminiscent of free-running conditions wherein mice exhibit phase dispersion (i.e., inter-individual desynchrony). This effect was not observed with 8-h adv 2×/wk-CJL. These data indicate that several aspects of central clock-driven behavior are uniquely modified according to CJL paradigm.
Frequent 8-h Forward Shifts Induce Distinct Metabolic Impairments
Metabolic impairments are associated with CJL in mice (Kettner et al., 2015; Casiraghi et al., 2016; Kettner et al., 2016; Zhong et al., 2019; Padilla et al., 2024) and humans exposed to acute CJL-like conditions in the laboratory setting (Scheer et al., 2009; Morris et al., 2016; Wefers et al., 2018; Qian et al., 2019). Light versus dark phase food intake was measured over 24 h on the day of the shift during the last week of the paradigm. As expected, LD mice consumed ~80% of food during the measured dark phase (Supplemental Figure S2A). 8-h adv 2×/wk-CJL mice consumed more food during the measured light phase and less food during the measured dark phase, compared to LD mice. 8-h adv e.o.d.-CJL mice consumed ~75% of food during the measured dark phase, similar to LD mice (Supplemental Figure S2A). Note the limitation that food intake on one day (or average food intake across all days) does not necessarily reflect food intake on any given day, and in contrast to LD mice that consistently eat mostly during the dark phase, CJL mice may eat more, less, or the same amount in dark compared to light depending upon endogenous rhythms and light masking on any given day. Total food intake was similar between LD, 8-h adv 2×/wk-CJL, and 8-h adv e.o.d.-CJL mice. Non-fasted resting blood glucose was measured at the time of tissue harvest 2 days post shift, reasoning that this would be a representative day that takes into account the effect of the shift as well as allows mice an opportunity for re-entrainment, if any. Sampled over the circadian cycle, non-fasted blood glucose reflects the ability to maintain whole-body glucose homeostasis in the face of natural feeding and fasting periods, whenever they may occur. Blood glucose was markedly different between the 3 light schedule paradigms (Figure 5a). As expected, blood glucose was relatively stable across light and dark in LD control mice. The 2 CJL paradigms increased blood glucose to varying degrees; 8-h adv 2×/wk-CJL increased blood glucose by 17.5% during the measured dark phase (ZT16-ZT20) while 8-h adv e.o.d.-CJL increased blood glucose by 38.9% during the measured light period (ZT4-ZT8). Blood glucose was also variable over 24 h in constant light (Supplemental Figure S2B). Similar to food intake, these measurements likely only reflect certain days, and not all days, of CJL. Nonetheless, these data show that unlike LD mice, CJL mice experience days of unstable, elevated non-fasted blood glucose. Additional systemic metabolic readouts were assessed at the 2 month mark of each paradigm to enable direct comparison between the 3 groups. Body weight gain was similar among the 3 groups (Figure 5b and 5c), yet body composition differed, with only 8-h adv e.o.d.-CJL lowering fat-to-lean mass ratio by 32.76% (Figure 5d).
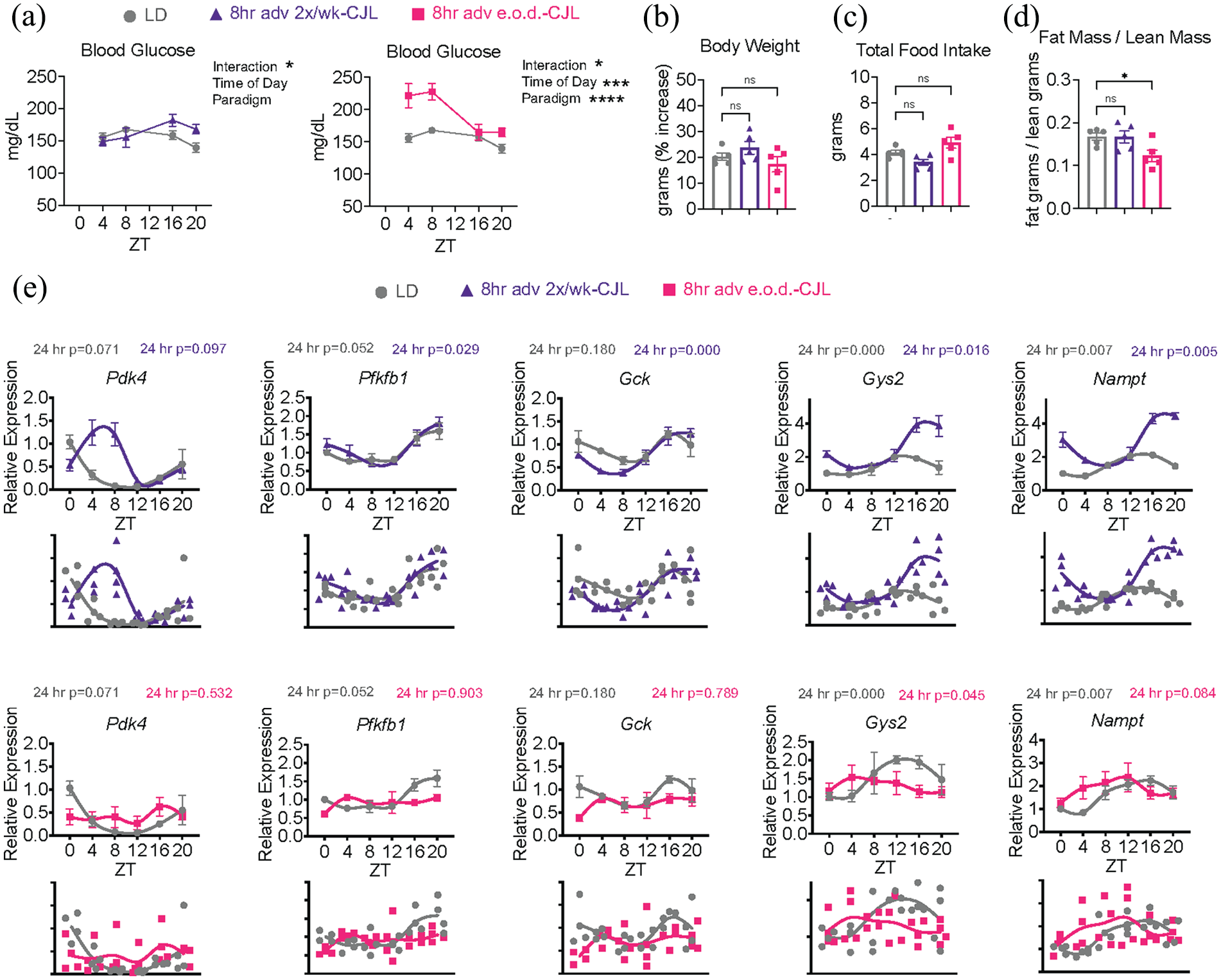
Frequent 8-h forward shifts induce distinct metabolic impairments. Mice were subjected to light schedules as in Figure 1. (a) Blood glucose at completion of the paradigm (LD—
To ask whether changes in clock-controlled metabolic output genes coincided with the observed phenotypes, a list of the most consistently rhythmic genes across published liver circadian transcriptome datasets (Brooks et al., 2023) was used to identify output genes related to glucose homeostasis (
Discussion
Here, we sought to more precisely define the impact of CJL on liver and central clock function, with a focus on associated systemic metabolic impairments. We tested 2 common CJL paradigms differing in the frequency of light schedule shifts and observed distinct effects on rhythmicity. The results show that more frequent shifts under 8-h adv e.o.d.-CJL induce more severe misalignment. Two months of every other day 8-h forward shifts induces a free-running-like state of the liver clock and central clock-driven behavior, such that mice are uncoupled from the light-dark cycle and inter-individual desynchrony (i.e., phase dispersion) occurs. This severe misalignment is uniquely associated with loss of fat mass and greater impairment of non-fasted blood glucose compared to less frequent shifts under 8-h adv 2×/wk-CJL. These results show that misalignment of the liver clock, rather than arrhythmicity, is associated with impaired systemic metabolism under environmental circadian disruption and that choice of CJL paradigm can significantly impact study outcomes and interpretation.
Independent data from separate cohorts of mice indicated that (1) behavioral rhythms were weaker (periodogram amplitude) under 8-h adv 2×/wk-CJL, and (2) the robustness (i.e., amplitude) of liver clock gene expression rhythms was comparable to LD control in 8-h adv 2×/wk-CJL. Assuming the behavior of the qPCR cohort was similar to the locomotor activity cohort, this unexpectedly suggests that the robustness of liver rhythmicity was maintained despite weaker behavioral rhythms under 8-h adv 2×/wk-CJL. Synchronizing cues for the liver, including feeding, body temperature, and glucocorticoids, are set by central clocks in the brain and intertwined with the sleep-wake cycle and rest-activity cycle (Koronowski and Sassone-Corsi, 2021). There is evidence that central clocks also provide a driving input to rhythmicity in the liver (Husse et al., 2014; Greco et al., 2021; Petrus et al., 2022). However, rhythmic amplitudes of clock genes in the liver under 8-h adv 2×/wk-CJL were similar, or even increased (
As a system, the circadian clock has 2 main tasks, drive endogenous rhythms and align them with the light-dark cycle. In terms of metabolism, this means anabolic and catabolic pathways need to be temporally segregated within cells or tissues and aligned with periods of resting-fasting and activity-feeding that follow the light-dark cycle (Panda, 2016). The present study points to alignment as the initial factor dictating the relationship between circadian control and metabolic dysfunction. The notion that loss of rhythmicity impairs metabolism comes from clock mutant mice, which have overlapping and distinct metabolic phenotypes (Guan and Lazar, 2021) known to stem in part from developmental abnormalities (Yang et al., 2016) and loss of clock-independent functions of clock genes (i.e., moonlighting Lipton et al., 2015; Michael et al., 2015), in addition to loss of rhythmicity. Still, the phenotypes of clock mutant mice can be recapitulated in part by environmental circadian disruption paradigms such as CJL. For example, clock mutations and CJL both induce fatty liver (Shimba et al., 2011; Kettner et al., 2016), desensitize responses to insulin (Dyar et al., 2014; Zhong et al., 2019), and impair blood glucose homeostasis (Turek et al., 2005; Harfmann et al., 2016; Kettner et al., 2016). Well-controlled laboratory studies with human participants support the notion that misalignment contributes to phenotypes of energy balance (Scheer et al., 2009; Qian et al., 2019), fatty acid metabolism (Wefers et al., 2018; Harmsen et al., 2021), insulin sensitivity (Wefers et al., 2018), and relevant disease biomarkers (Morris et al., 2016). The liver clock, specifically, contributes to systemic energy balance and glucose homeostasis by regulating glycogen storage (Doi et al., 2010; Koronowski et al., 2019) and gluconeogenesis (Zhang et al., 2010; Smith et al., 2023a). Here, we found that misalignment, not loss of rhythmicity, of the liver clock and rhythmic output genes related to glucose metabolism was associated with impairment of glycemic control and body composition.
In contrast to our results, studies implementing CJL over the entire lifespan have indicated loss of liver rhythmicity. A similar but slightly different CJL protocol (two 8-h shifts per week, one advance and one delay) administered from 4- to 90-weeks-of-age induced metabolic dysfunction-associated steatotic liver disease (MASLD) in more than 95% of mice (vs. 20% of control mice) (Kettner et al., 2016) and had a similar effect on mice with humanized livers (Padilla et al., 2024). Clock gene expression indicated a substantial loss of amplitude for many clock genes from a 3-timepoint experiment, although specific rhythmic analyses were not performed. A possible explanation for this discrepancy is that metabolic impairment that persists chronically over a long period of time leads to loss of rhythmicity. The reciprocal regulatory relationship between the clock and metabolism supports this notion, wherein metabolism also regulates the clock (Reinke and Asher, 2019). Rhythmic parameters can be modified via nutrient-responsive promoter elements, metabolite-dependent post-translational modifications, clock cooperating co-regulators that respond to metabolic and redox flux, and diurnal ligands that regulate PAS domain containing and nuclear receptor type clock proteins (Koronowski and Sassone-Corsi, 2021). Through these mechanisms, dietary interventions are known to rewire or reprogram circadian function in the liver. This can be beneficial as in time-restricted feeding interventions (Vollmers et al., 2009; Hatori et al., 2012; Greco et al., 2021) or lead to deleterious effects such as loss of circadian robustness in the context of high fat diet-induced obesity (Kohsaka et al., 2007; Eckel-Mahan et al., 2013). Here, we observed the onset of metabolic phenotypes after 2 to 3 months of CJL without loss of rhythmicity in the liver. As a working conceptual model, the sequence of events may be that misalignment impairs metabolism, and if left unresolved, metabolic impairment eventually leads to loss of liver rhythmicity. Longer, more thorough studies are needed to test this notion.
The current study was performed in male mice on a control chow diet and focused on the liver clock. Recent studies show that responses to CJL can vary by tissue (Pariollaud et al., 2022) and sex (Anderson et al., 2023) and that factors such as diet (Anderson et al., 2023) or level of regulation (Duong et al., 2024) (i.e., post-transcriptional, post-translational) may modulate the outcomes further. Although liver may be a decent readout of metabolic peripheral clocks, peripheral clocks respond to different entrainment cues and can respond differently to the same cue (Guo et al., 2005; Wolff et al., 2013). One study using a different CJL paradigm of 8-h advances every 2 or 3 days found that, in the lung, rhythmic expression for some clock genes, but not others, was lost after 8 weeks (Pariollaud et al., 2022). In this case, the possibility of phase dispersion was noted but ultimately argued against because certain genes remained rhythmic, suggesting that synchrony between animals was maintained. A comprehensive study is needed to assess the response of each peripheral clock to the exact same CJL paradigm(s).
Studies in mice (Anderson et al., 2023) and humans (Qian et al., 2019) show sexual dimorphism in the response to circadian misalignment. In mice on high fat diet, 3 months of a once weekly 8-h advance CJL resulted in highly variable clock gene expression (reminiscent of phase dispersion between animals) in the liver of males but not females (Anderson et al., 2023). Females were protected from cardiometabolic consequences of CJL and did not exhibit the microbiome dysbiosis involving diabetogenic branched chain amino acid production that was observed in males. In line with this, the liver circadian transcriptome is substantially sex-dependent (Talamanca et al., 2023; Astafev et al., 2024) and liver rhythmicity is influenced by the microbiome (Weger et al., 2019).
Another point of consideration is that mRNA may not reflect the final protein-level output. Here, we confirmed severe misalignment in 8-h adv e.o.d.-CJL at the level of clock genes and proteins. A comprehensive analysis of the liver after 4 cycles of 6-h advances CJL (according to Davidson et al., 2006) followed by 1 week of a standard light schedule and 2 days of constant darkness revealed that post-transcriptional and post-translational mechanisms accounted for most of the rhythmic changes to output genes under CJL (Duong et al., 2024). In addition to marked loss of rhythmicity, a substantial gain of rhythmicity was also observed for hundreds of output genes and proteins under CJL. Rhythmic core clock gene expression was maintained despite a “re-writing” of clock-controlled output genes, suggesting that circadian misalignment may, through yet-to-be-identified mechanisms, uncouple the clock from its physiological output.
One limitation of this study is that synchrony between organs within the same mouse could not be assessed with the resources at hand. A prevailing view is that central clock-driven cues provide inter-organ synchrony (Yoo et al., 2004; Tahara et al., 2012), allowing daily functions to be coordinated throughout the body. Systemic metabolism also relies on inter-organ coordination and communication (Priest and Tontonoz, 2019). We have shown previously that behavioral rhythms are necessary for liver and muscle clocks to synergistically provide glycemic control (Smith et al., 2023a), and inter-organ desynchrony is shown to occur with intermittent hypoxia in a mouse model of sleep apnea (Manella et al., 2020). Thus, future studies aimed at understanding how CJL impacts inter-organ synchrony within one animal can shed light on this aspect of the clock system.
Supplemental Material
sj-tif-1-jbr-10.1177_07487304241311328 – Supplemental material for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice
Supplemental material, sj-tif-1-jbr-10.1177_07487304241311328 for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice by Qing Zhang, Christopher Litwin, Kristi Dietert, Ioannis Tsialtas, Wan Hsi Chen, Zhihong Li and Kevin B. Koronowski in Journal of Biological Rhythms
Supplemental Material
sj-tif-2-jbr-10.1177_07487304241311328 – Supplemental material for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice
Supplemental material, sj-tif-2-jbr-10.1177_07487304241311328 for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice by Qing Zhang, Christopher Litwin, Kristi Dietert, Ioannis Tsialtas, Wan Hsi Chen, Zhihong Li and Kevin B. Koronowski in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-3-jbr-10.1177_07487304241311328 – Supplemental material for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice
Supplemental material, sj-xlsx-3-jbr-10.1177_07487304241311328 for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice by Qing Zhang, Christopher Litwin, Kristi Dietert, Ioannis Tsialtas, Wan Hsi Chen, Zhihong Li and Kevin B. Koronowski in Journal of Biological Rhythms
Supplemental Material
sj-xlsx-4-jbr-10.1177_07487304241311328 – Supplemental material for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice
Supplemental material, sj-xlsx-4-jbr-10.1177_07487304241311328 for Frequent Shifts During Chronic Jet Lag Uncouple Liver Rhythms From the Light Cycle in Male Mice by Qing Zhang, Christopher Litwin, Kristi Dietert, Ioannis Tsialtas, Wan Hsi Chen, Zhihong Li and Kevin B. Koronowski in Journal of Biological Rhythms
Footnotes
Acknowledgements
The Koronowski lab was supported by the National Institute of General Medical Sciences of the National Institutes of Health under award number R35GM150618 and a Young Investigator Award from the Max and Minnie Tomerlin Voelcker Fund.
Conflict Of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
NOTE
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
