Abstract
The circadian system drives the temporal organization of body physiology in relation to the changing daily environment. Shift-work (SW) disrupts this temporal order and is associated with the loss of homeostasis and metabolic syndrome. In a rodent model of SW based on forced activity in the rest phase for 4 weeks, we describe the occurrence of circadian desynchrony, as well as metabolic and liver dysfunction. To provide better evidence for the impact of altered timing of activity, this study explored how long it takes to recover metabolic rhythms and behavior. Rats were submitted to experimental SW for 4 weeks and then were left to recover for one week. Daily locomotor activity, food intake patterns, serum glucose and triglycerides, and the expression levels of hepatic
Keywords
A large part of the worldwide active population is involved in shift-work (SW) (ILO, 2008). Epidemiological studies show that SW is a risk factor for disturbed circadian rhythms and enhances the risk of developing metabolic dysfunction, including obesity, type-2 diabetes and metabolic syndrome (Biggi et al., 2008; Gan et al., 2015; Knutsson et al., 2004; Proper et al., 2016).
The mammalian circadian system, including that of humans, consists of a master clock, the suprachiasmatic nucleus (SCN), which resides in the hypothalamus and synchronizes the organs and systems according to the environmental LD cycle by transmitting temporality to peripheral biological clocks (Kalsbeek et al., 2011). The expression of circadian rhythms in the SCN and in the periphery depends on a set of so-called clock genes. In the absence of external stimuli, these genes can maintain self-sustained oscillations with a period of about 24 h and are responsible for endogenous rhythms (Hogenesch and Herzog, 2011). Clock gene transcripts form complex positive (
The circadian system may become temporarily and severely disorganized under perturbing factors such as SW (Buijs et al., 2016), which represents a condition during which environmental signals contradict the internal timing driven by the circadian system. We have previously reported that, in a SW rat model, forced activity during the light phase does not affect SCN synchronization with the LD cycle. However, it triggers a condition of internal desynchronization, where peripheral oscillators are temporally uncoupled from the master clock (Salgado-Delgado et al., 2010b; Salgado-Delgado et al., 2013). This desynchronization results in altered daily patterns of locomotor activity, food ingestion, metabolites (i.e., glucose and triglycerides) and the hepatic expression of metabolism-related genes (e.g.,
It is still not clear whether this internal circadian desynchrony can be restored and, with it, the internal homeostasis. In the present study, we explored the speed for recovery of behavioral and metabolic rhythms in rats undergoing a 4-week experimental SW protocol. At the end of the protocol, animals could recover for one week. Daily patterns of locomotor activity, food intake, plasma glucose and triglycerides, hepatic metabolic (
Materials and Methods
Animals and Housing
Male Wistar rats, weighing 180 to 200 g at the beginning of the experiment, were housed in individual transparent acrylic cages (40 × 50 × 20 cm) and placed in soundproof lockers housing 8 animals each, in a soundproof monitoring room. Rats were maintained in a 12:12 h LD cycle with lights on at 0700 h, defined as zeitgeber time 0 (ZT0), constant temperature (22°C [1°C]), circulating air and free access to water and food (Rodent Laboratory Chow 5001; LabDiet, IL). Experiments were approved by the ethics committee of the School of Medicine at Universidad Nacional Autonoma de México, in accordance with the national guide of care and use of experimental animals (Norma Oficial Mexicana NOM-062-ZOO-1999).
Groups
Rats were randomly assigned to 1 of 2 groups: control (CTRL) or experimental SW. CTRL rats were housed in individual cages in the monitoring system and were left undisturbed for 4 weeks except during the implantation of the jugular cannula. Rats assigned to the experimental SW were submitted to a forced activity protocol during the light phase, which corresponds to the animal’s sleep/rest interval.
Experimental Design
Experiment 1. Recovery speed in general activity
All rats were monitored in their home cages for 8 to 10 days in LD conditions to establish a baseline. To determine the recovery in general activity, rats were exposed to a 4-week long protocol of forced activity. The evaluation of recovery began the first day the animals (
Experiment 2. Recovery of metabolic variables
At the end of the third week of the SW protocol a series of CTRL (
During the recovery week, blood samples were obtained on the third, fifth and seventh day of recovery (RD3, RD5, and RD7, respectively) and at 4 time points: ZT0, ZT6, ZT12 and ZT18 for both the CTRL and SW groups. Data at ZT0 are double-plotted as ZT24. Blood samples (240 µl) were collected in Eppendorf tubes (1.8 ml) containing a clot-activator gel (BD Vacutainer 367983, BD, Mexico City, Mexico) and were centrifuged at 2500 rpm for 10 min. Plasma was stored in 100-µl aliquots at −80°C until assay.
Experiment 3. Recovery of metabolic and clock genes in the liver
To determine metabolic and clock gene expression, another group of rats was exposed to the 4-week long SW protocol, after which tissue was collected on RD3, RD5, and RD7. We avoided an unnecessary use of animals for RT-PCR experiments by analyzing gene expression in the CTRL group only on RD7. These results were compared with those in the SW group. Tissue samples were obtained at ZT0, ZT6, ZT12 and ZT 18 (
Shift-work Protocol
To induce activity during the rest phase, SW rats were placed in slow rotating drums (designed by Omnialva SA de CV for our laboratory) for 8 h daily, as previously described (Salgado et al., 2008). The drums rotated with a speed of 1 revolution/3 min. Drums had ample enough space to allow rats to move, eat and drink freely (more details in Salgado et al., 2008).
From Monday to Friday, SW rats were taken out of their home cages at ZT2 and placed in the rotating drums for 8 h; at ZT10 rats were returned to their home cages, where they rested until the next day. On Saturday and Sunday, rats were left undisturbed in their home cages. This procedure was carried out for 4 weeks.
Monitoring of Behavioral Rhythms
General activity in the home cage was continuously monitored using pressure sensors placed under individual cages. Behavioral events were collected with a digitized system and automatically stored every minute in a PC for further analysis with the SPAD9 (Sistema de Procesamiento y Adquisición de Datos, Version 1.1.1) program designed for this system. For each group, a mean activity wave was constructed for the baseline and for the fourth weekend of the protocol. For both waveforms, the percentage of diurnal and nocturnal activity was calculated. The acrophase and the mean daily total activity counts were calculated for the baseline, and then for each day of the recovery period.
Monitoring of Food Intake
The amount of consumed food was weighed twice a day (at ZT0 and ZT12) at baseline, on the last day of the SW protocol, and on the subsequent 7 days of recovery. The amount of ingested food was compared between the respective light (rest) and dark (active) phases.
Jugular Catheter Surgery
At the end of the third week of the SW protocol, a series of CTRL (
Serum Metabolite Determination
Blood samples for both the CTRL and SW groups were obtained on RD3, RD5, and RD7 at 4 time points: ZT0, ZT6, ZT12 and ZT18. Glucose levels were estimated using a commercial colorimetric kit (Hycel de México; Jalisco, México), which is based on the reaction between glucose and fenol-4-aminofenazone as a chromogen. Serum triacylglycerides (TAG) were assessed with a commercial diagnostic kit (SERA-PAK plus, Bayer; Sees, France) based on the reaction with 4-amino-antipiridin. Both glucose and TAG reactions were measured at 500 nm with a spectrophotometer (Novaspec II Visible, Amersham Pharmacia Biotech, Cambridge, UK).
Semi-quantitative qRT-PCR
Total RNA was isolated from liver with Trizol (Invitrogen, Thermo Fisher Scientific Inc., Waltham, MA, USA) according to the manufacturer’s recommendations. Equal quantities of total RNA were used for complementary DNA generated by SuperScript III (Invitrogen). Metabolic genes
Primer sequences used for semi-quantitative PCR.
Statistical Analysis
The acrophase, amplitude, mesor, and rhythmicity significance were calculated for samples taken on RD3, RD5, and RD7 for locomotor activity, serum metabolites and gene expression by performing a cosinor analysis test. Only the acrophase of locomotor activity was evaluated daily. In addition, total activity counts per day during the recovery week were compared with the baseline activity. Recovery was achieved when the acrophase and activity counts were not statistically different from baseline values.
The single cosinor analysis was performed as previously described (Nelson et al., 1979), using the formula: Y(t) = M+ACos[(2π/T)*(t-φ)], where Y = collected data; M = mesor; A= amplitude; φ = acrophase; T = period, t = sampling time. Double-plotted data (ZT24) were not included in the cosinor analysis. Circadian rhythmicity significance (24-h period) was assumed when the amplitude of the fitted curve was statistically >0.
Data were classified by groups (CTRL vs. SW) and time (day) and are represented as mean ± standard error of the mean (SEM). Food intake data were analyzed with a 2-way ANOVA for 2 factors: days (9) and phase (light and dark). Serum concentration of glucose, TAG and daily profiles of clock and metabolic genes were evaluated also by a 2-way ANOVA for the factors group (2 levels) and time (4 levels).
All analyses were followed by a Tukey post hoc test with significant values set at
Results
Recovery of General Activity After 4 Weeks of Shift-work
At baseline, SW rats exhibited clear daily rhythms entrained to the LD cycle (Figure 1A and 1B), and nocturnal activity represented 74% of the total daily activity (Figure 1B). After 4 weeks, the proportion of nocturnal activity decreased to 52% (Figure 1A and 1B, lower panel). On the first day of recovery (RD1), the acrophase of general activity showed a phase advance of 1 h (
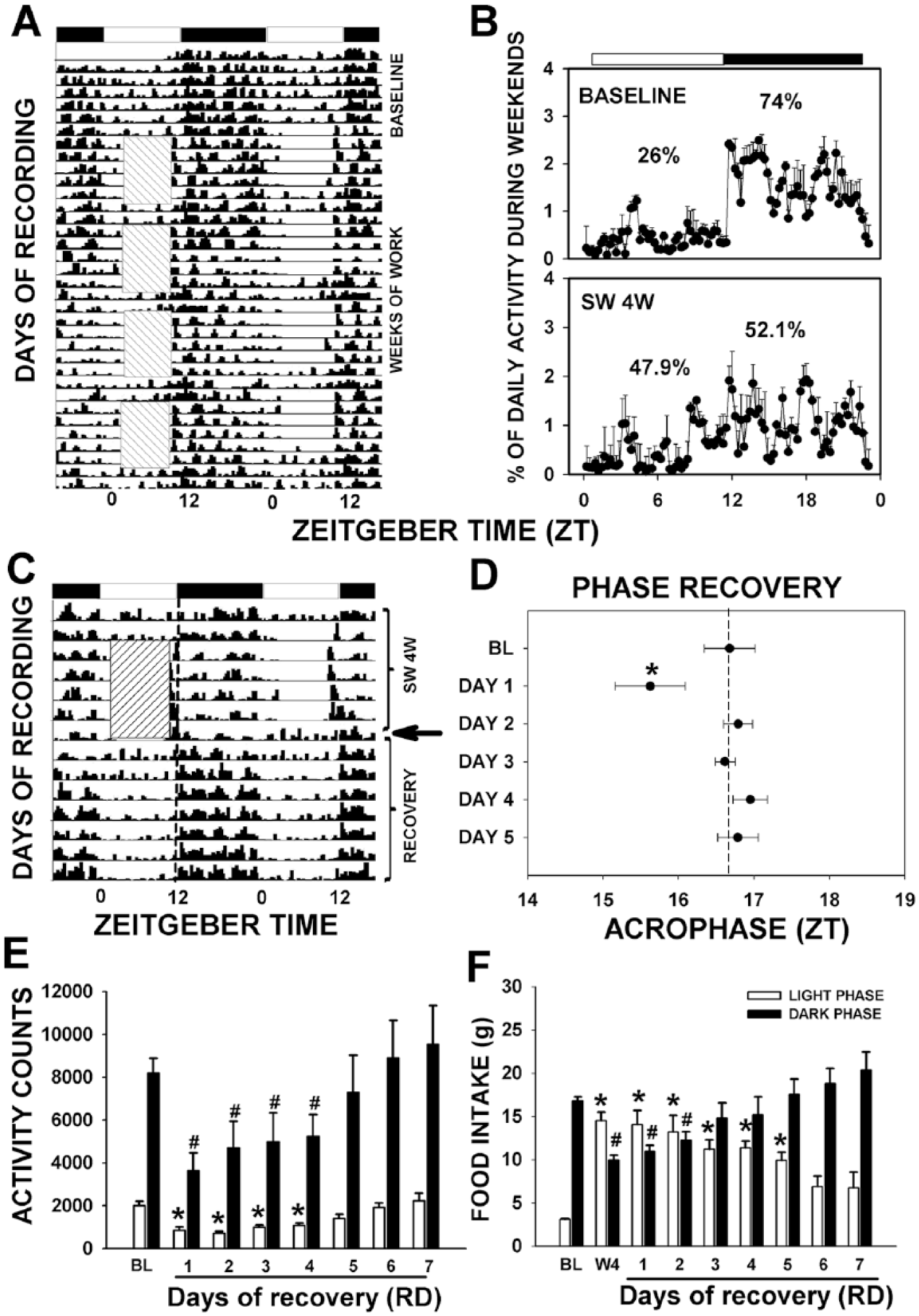
Shift-work (SW) alters daily locomotor activity and food intake pattern. (A) Representative double-plotted actograms of SW animals. Boxes with diagonal lines represent time in the activity wheel. Black and white bars represent the LD cycle. (B) Percentage of daily locomotor activity during the weekends in the baseline (BL) and fourth week of SW. (C) Days of recording during the fourth week of SW and the recovery week. (D) Acrophases of locomotor activity of SW animals during the recovery week. Dashed lines represent the expected acrophase time with respect to the BL. (E) Total daily locomotor activity counts during recovery time compared with BL. (F) Mean daily food intake during the BL, the fourth week (4W) of SW, and the following recovery days (RD). White bars represent mean food intake during light phase (ZT0 to ZT12) and black bars during the dark phase (ZT12 to ZT24). Data are represented as mean and SEM. *
Activity counts were significantly reduced during the first days of recovery and progressively reached similar activity levels on RD5 (Figure 1E).
Shift-work Affects Feeding Behavior and Recovery on RD6
At baseline in the SW group, 79% of total daily-ingested food was consumed during the active/dark phase (
TAG and Glucose Rhythms Were Restored Within One Week
Plasma TAG and glucose concentrations were measured at ZT0, ZT6, ZT12, and ZT18 on RD3, RD5, and RD7 and for the CTRL group on RD7.
On RD3, SW displayed rhythmic levels of TAG but the acrophase presented at ZT7, in phase advance (~9 h,
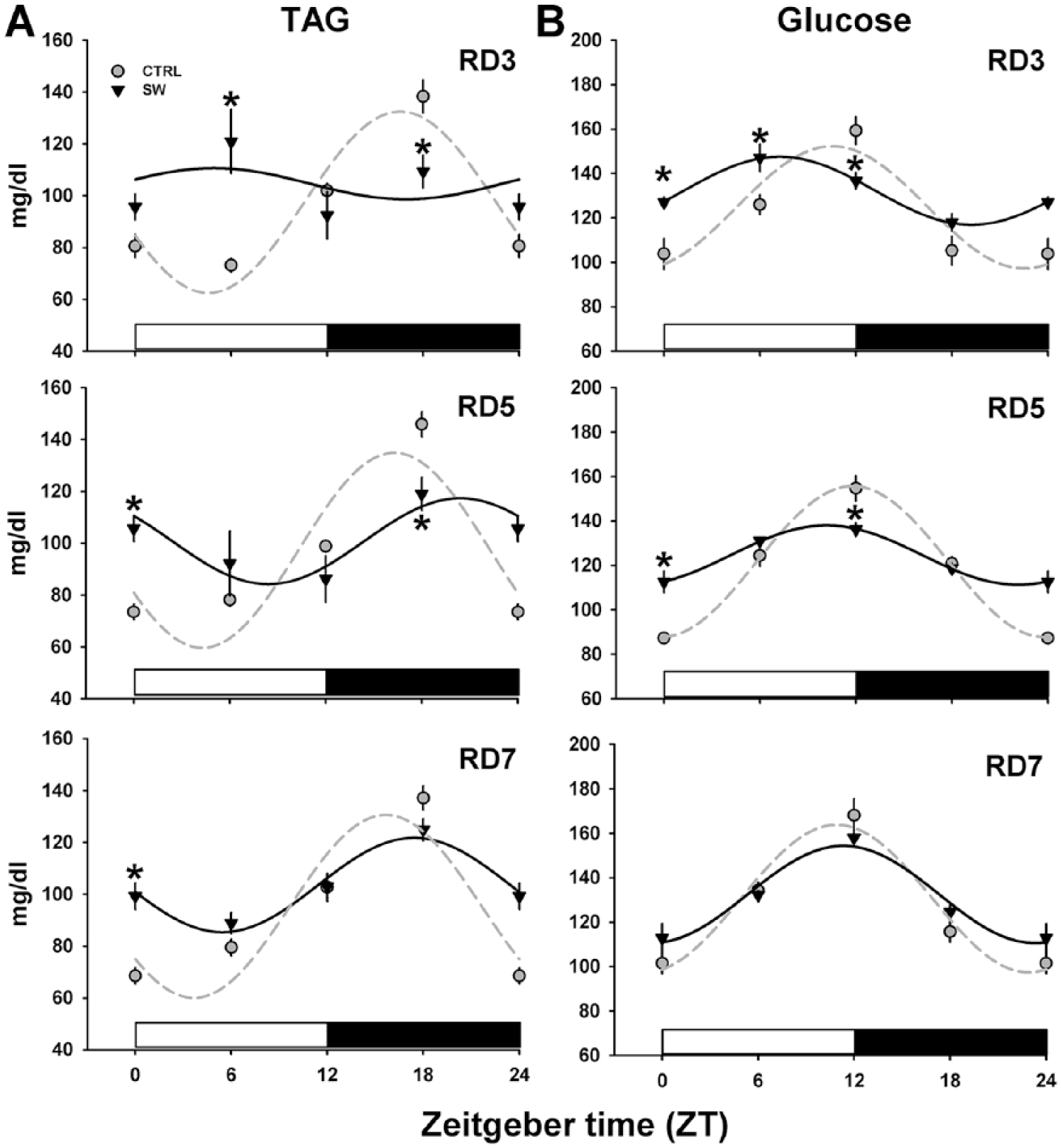
Recovery of plasma triglyceride and glucose daily patterns after shift-work (SW). Serum TAG (A) and glucose (B) fluctuations in rats in the SW group on the third, fifth and seventh day of the recovery week compared with the control (CTRL) animals. Black and white bars represent the LD cycle. Data are represented as mean (SEM).
Parameters of cosinor analysis (period = 24 h), including the mesor, amplitude, acrophase, and rhythm significance of locomotor activity, and clock- and metabolism-related genes for the entire week of recovery from shift work.
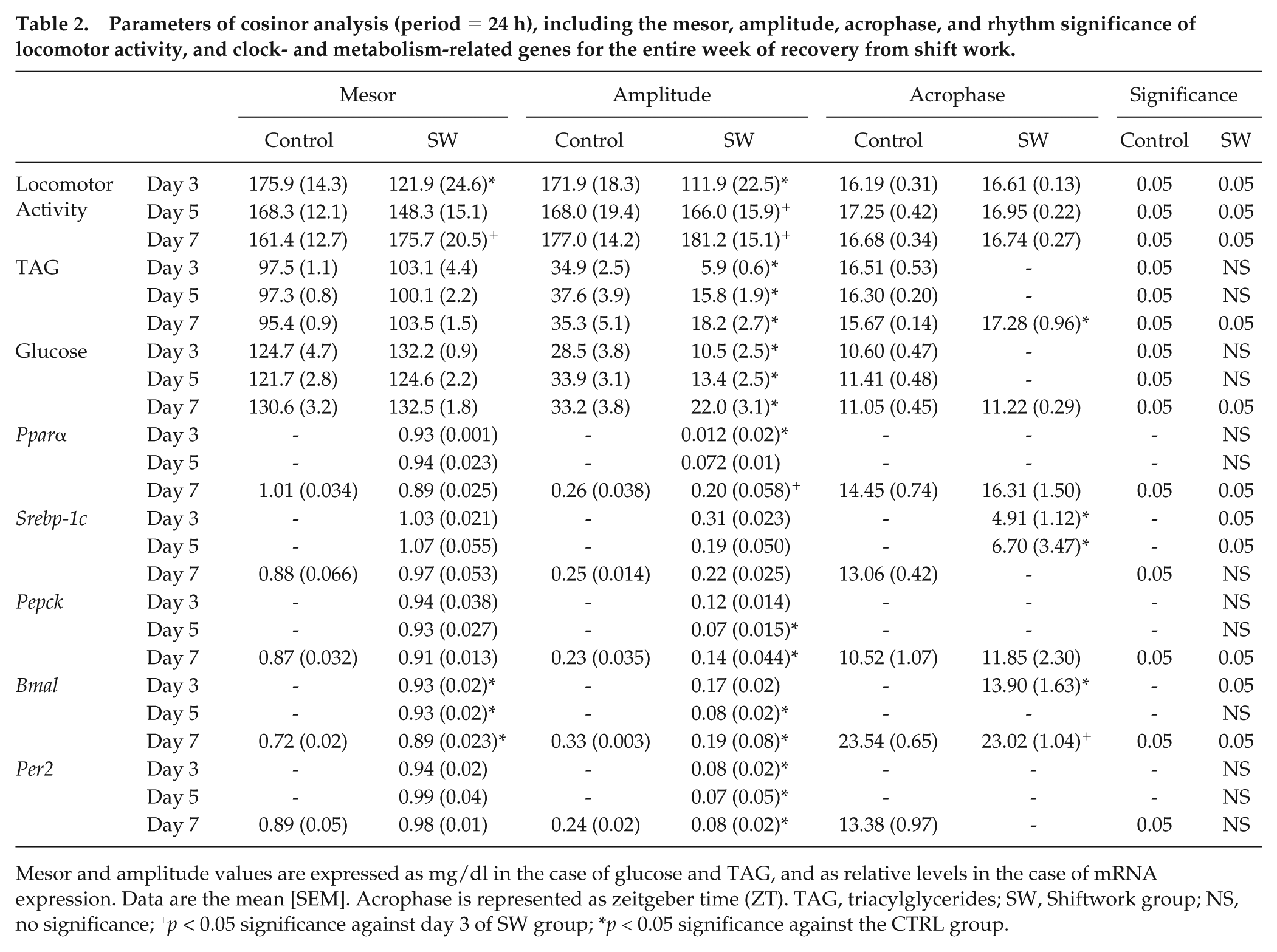
Mesor and amplitude values are expressed as mg/dl in the case of glucose and TAG, and as relative levels in the case of mRNA expression. Data are the mean [SEM]. Acrophase is represented as zeitgeber time (ZT). TAG, triacylglycerides; SW, Shiftwork group; NS, no significance; +
During recovery, plasma glucose of SW animals remained arrhythmic on days RD3 and RD5; however, on RD7, levels exhibited a rhythmic pattern in phase with the CTRL group (Figure 2B, Table 2).
Hepatic Metabolic Gene Rhythms Were Not Completely Restored during the Recovery Week
In the CTRL group,
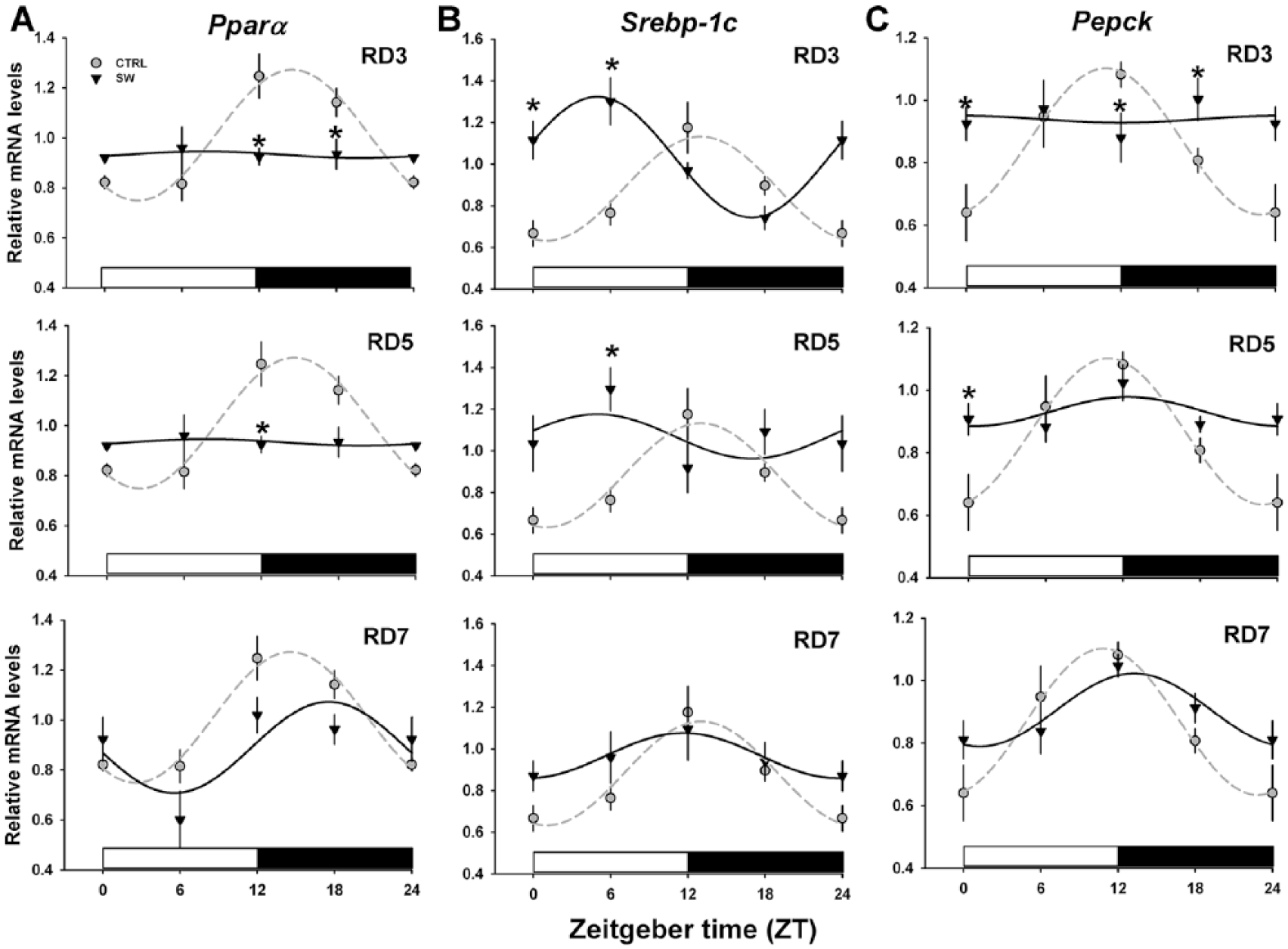
Recovery of metabolism-related gene expression in the liver after shift-work (SW). Lipid metabolism is represented by
Hepatic Clock Gene Rhythms Were Not Completely Restored during the Recovery Week
In the liver of CTRL rats,
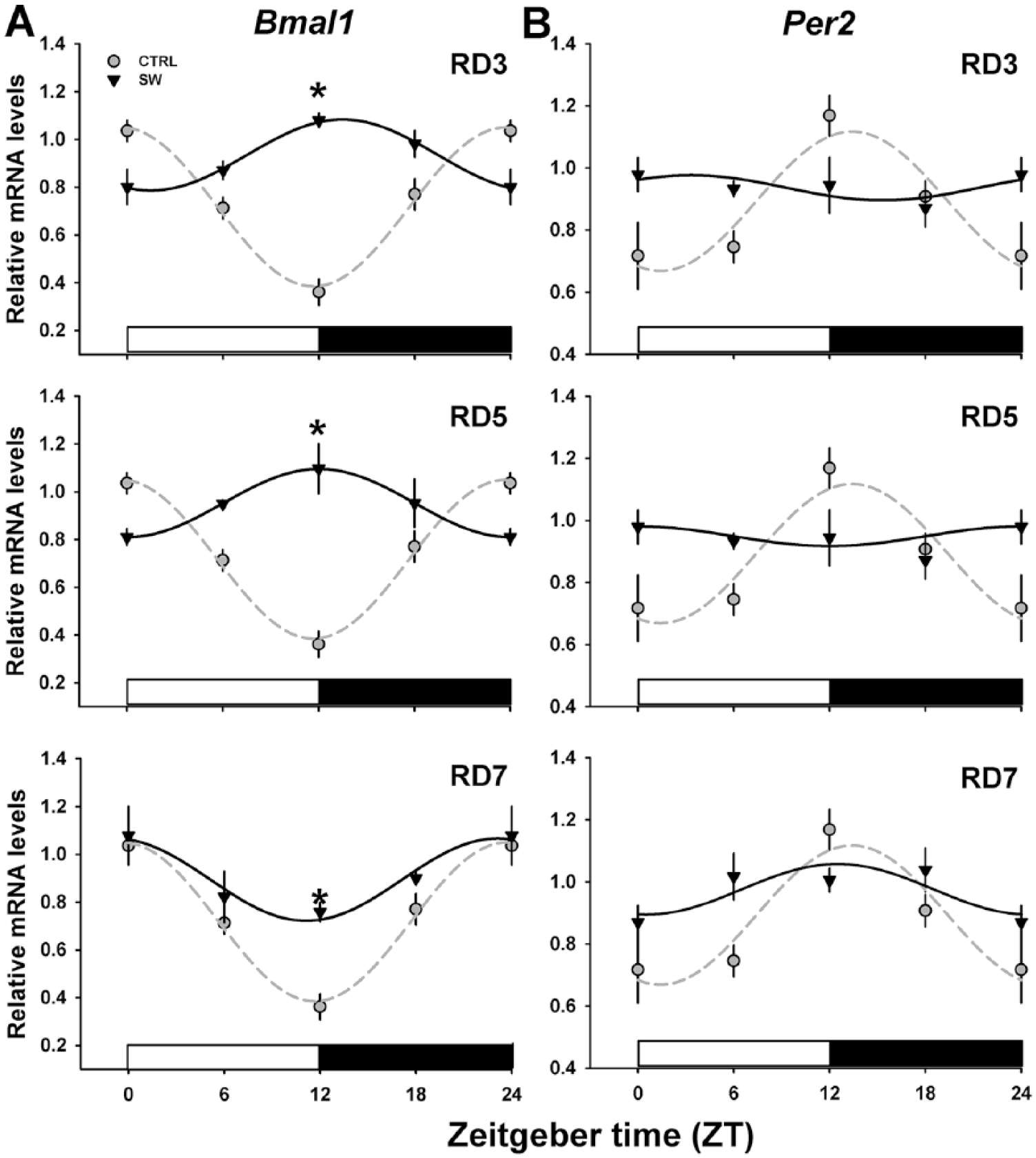
Recovery of clock gene expression in the liver after shift-work (SW). RT-PCR analysis of daily
Discussion
In this study, we used a SW rat model to induce internal desynchrony and to explore the kinetics of resynchronization (recovery) 1) at the behavioral level, by monitoring locomotor activity and food intake; 2) at the metabolic level, by quantifying the daily fluctuation of blood TAG and glucose; and 3) at the molecular level, by determining the temporal patterns of expression of several hepatic metabolic and clock genes. We observed that recovery is faster at the behavioral and metabolic levels than for most of the molecular parameters investigated. During this recovery period, an internal desynchrony persisted among behavior, metabolism and hepatic gene expression.
Locomotor Activity
The SW protocol affected general locomotor activity in 2 ways. First, it shifted the daily acrophase by advancing the peak of activity by 1 h, leading to a desynchronized behavior (Figure 1). Second, it decreased the nocturnal activity counts as compared with the baseline (Figure 1E). Whereas resynchronization of the acrophase took only 2 days, the levels of nocturnal activity recovered within 5 days. According to previous reports, the SW protocol employed in the present experiment represents a model of internal desynchronization, in which the SCN activity and clock machinery oscillate in harmony with the LD cycle (Salgado-Delgado et al., 2010). Thus, the faster recovery of the activity rhythm, as compared with the peripheral oscillators, could indicate that light masks the actual effects of SW over endogenous circadian rhythmicity (Reddy et al., 2002; Yamazaki et al., 2000). Masking could involve retinorecipient centers that have been suggested to modulate the synchronizing effects of light, either in the thalamus or in hypothalamic areas, rather than the SCN (Gooley et al., 2003; Hannibal and Fahrenkrug, 2004). These thalamic and hypothalamic centers can respond directly to the light signals that drive behavior under a weak SCN, as we have previously shown (Salgado-Delgado et al., 2010a).
The delayed return of total locomotor activity to baseline levels might be related to the sleep disturbances induced by SW. As demonstrated by Grønli and colleagues (2017), SW degrades arousal and promotes micro-sleep episodes, thus reducing locomotion.
Metabolic Parameters
As our group already reported, SW abolished the daily rhythm of plasma glucose and shifted the TAG rhythm toward the light phase (Salgado-Delgado et al., 2010b).
It is interesting to note that TAG daily rhythms mirrored feeding behavior. They peaked during the light phase at the beginning of recovery week (RD3); were flattened at RD5 (when the amount of food consumed during the light phase was significantly higher than the baseline, and dark-phase food ingestion had already recovered); and peaked during the dark phase on RD7 (when 73% of the daily food ration was consumed). In this respect, there is evidence that, even in clock-disrupted mice, circadian regulation of lipid metabolism persists if meal access occurs in a shifted restricted time (Adamovich et al., 2014). These data are consistent with our results; daily TAG rhythms perfectly correlated with food intake patterns observed during the recovery week after SW.
An additional contribution to the TAG phase advance might come from the changes observed in
The reason for the loss of rhythmicity in glucose daily patterns is unclear. Although plasma glucose rhythmicity is mainly regulated by the SCN (la Fleur et al., 2001), it is unlikely that the arrhythmicity observed in the SW rats during recovery reflects the central clock; we have shown that SW does not affect clock gene rhythmicity in the SCN (Salgado-Delgado et al., 2008). Thus, the breaking point for glucose homeostasis must be explored elsewhere, in the many conflicts triggered by the forced activity, the consequent sleep disturbances, the changes in feeding patterns, and all the circadian misalignments that affect metabolic oscillators downstream of the SCN and worsened by SW (Morris et al., 2015
Another consequence of SW was the loss of mRNA rhythm in the hepatic catabolic gene
Clock Genes
Most of the published literature reporting the ability of the circadian system to adapt to new time cues (e.g., light phase advance and/or restricted feeding schedules) indicates that there are significant differences among clock genes and tissues (Angeles-Castellanos et al., 2007; Davidson et al., 2009; Kiessling et al., 2010; Stokkan et al., 2001; Wu et al., 2010). In the present work, we did not change the photoperiod or restrict food access, and the changes that we observed at the behavioral and molecular levels were a spontaneous outcome of SW and the following recovery week. Our results show that SW induced a large phase advance of
Although
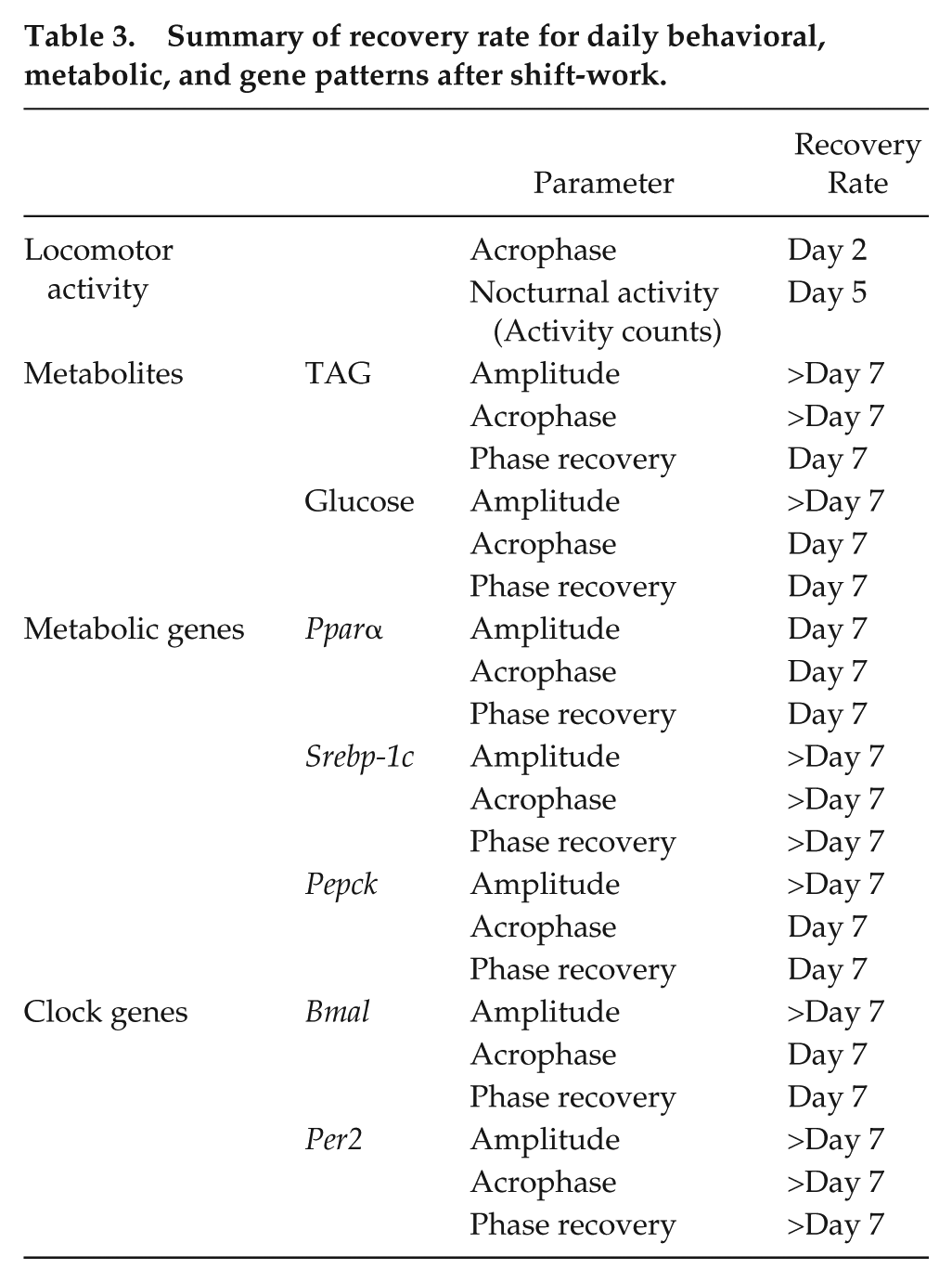
In conclusion, SW promotes a condition of internal desynchronization that results in either the alteration (TAG,
Summary of recovery rate for daily behavioral, metabolic, and gene patterns after shift-work.
Supplemental Material
TABLE_S1 – Supplemental material for Differential Recovery Speed of Activity and Metabolic Rhythms in Rats After an Experimental Protocol of Shift-Work
Supplemental material, TABLE_S1 for Differential Recovery Speed of Activity and Metabolic Rhythms in Rats After an Experimental Protocol of Shift-Work by Nadia Saderi, Adrián Báez-Ruiz, Lucia E. Azuara-Álvarez, Carolina Escobar and Roberto C. Salgado-Delgado in Journal of Biological Rhythms
Footnotes
Acknowledgements
This study was supported by Consejo Nacional de Ciencia y Tecnología (CONACyT), Mexico (grant number: 183078 to R.S.D., 243298 to N.S); UASLP-CA-254 and FAI 2017 to RSD.
Author Contributions
NS, CE, and ABR analyzed data and wrote the manuscript. RSD and CE conceived the project. RSD and NS designed and conducted the experiments. RSD is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
