Abstract
Immune-mediated inflammatory diseases (IMIDs) such as rheumatoid arthritis, inflammatory bowel disease, and asthma share common pathophysiological pathways characterized by chronic inflammation and subsequent tissue damage involving multiple body sites. Circadian rhythms are 24-h body cycles that regulate immune activity and control the magnitude of immune response based on time of day. Chronotype is a person’s individual circadian phase preference, ranging from morningness to eveningness, which is known to influence the risk of cardiometabolic and mental health disease. We systematically reviewed the literature to assess the association of questionnaire-based chronotype and patients with IMID. A comprehensive search of MEDLINE and Embase identified 12 studies meeting the inclusion criteria, conducted in 7 countries and covering 4 IMIDs to include 15,625 IMID patients and 410,783 healthy controls. Results showed that later chronotype may be a risk factor for worse quality of life and increased symptom burden in patients with IMIDs. In addition, chronotype may be a risk factor for IMID incidence, but the direction and magnitude of this effect were not consistent across individual IMIDs. Chronotype assessment could contribute to risk stratification in patients with IMIDs. Cross-disciplinary collaboration to understand the role of circadian rhythms and chronotype in driving common inflammatory pathways could help to improve outcomes for patients with IMIDs.
Immune-mediated inflammatory diseases (IMIDs) encompass a broad range of conditions characterized by chronic inflammation leading to tissue damage. Examples of IMIDs include rheumatoid arthritis (RA), inflammatory bowel disease (IBD), psoriasis, asthma, and systemic lupus erythematosus (SLE), which all share common features that facilitate consideration under the umbrella term IMID.
IMIDs significantly cluster, for example, patients with IBD have an increased risk of IMIDs including RA and psoriasis (Bernstein et al., 2005; Burisch et al., 2019). This may be in part due to multiple shared genetic susceptibility loci (Parkes et al., 2013). IMIDs can also involve multiple tissues, for example, patients with psoriasis can develop psoriatic arthritis, patients with IBD can develop extra-intestinal manifestations such as joint or skin inflammation, and patients with SLE can acquire multiple organ inflammation involving the skin, joints, and kidneys, suggesting inflammatory pathways can drive similar damage across multiple organs (Antonelli et al., 2021; Figus et al., 2021; Korman, 2020).
Indeed, the paradigm for grouping IMID pathogenesis by organ is evolving to align with shared molecular inflammatory pathways. Many IMIDs cluster around signature cytokines including interleukin (IL)-23 in IBD and psoriasis, IL-6 in RA, and tumor necrosis factor-α (TNFα) as a common downstream effector in inflammatory arthritis and IBD (Schett et al., 2021). TNFα inhibition with monoclonal antibodies has been a revolutionary treatment for patients with some, but not all, IMIDs, highlighting the importance of cross-specialty mechanistic research. A growing number of cytokines are now therapeutic targets across the IMIDs.
Circadian Rhythms
Circadian rhythms are repeating 24-h cycles that organize physiological processes to maximize host efficiency and survival. Nearly every cell contains the molecular clockwork machinery required to generate feedback loops that run the biological clock and regulate a multitude of clock-controlled outputs. Internal clock time is set by external stimuli such as light:dark cycles or meal timing. Circadian rhythms regulate immune function including lymphocyte trafficking and a magnitude of inflammatory response (Scheiermann et al., 2018). Experimentally, disruption of circadian rhythms often leads to more severe inflammatory phenotypes, as demonstrated in IMID murine models including RA (Gibbs and Ray, 2013), asthma (Durrington et al., 2020), IBD (Codoñer-Franch and Gombert, 2018), and psoriasis (Ando et al., 2015). Humans with circadian disruption as a result of shift work are at higher risk of IMIDs such as asthma and COVID-19 (Maidstone et al., 2021c, 2021a).
Chronotype
Chronotype relates to a person’s individual circadian phase and ranges on a scale from morningness to eveningness to give an indication of when key homeostatic processes are most active. Chronotype is shaped by environmental and genetic influences and can be interpreted qualitatively as a personality trait or quantitatively as a phase of entrainment (Jones et al., 2019; Roenneberg, 2015). Phase of entrainment can be calculated with invasive sampling, such as oral swabs for dim light melatonin onset (Lewy et al., 1999) or blood tests for biomarker expression panels (Wittenbrink et al., 2018). However, these costly profiles are harder to perform on large populations. The use of questionnaires such as the Morningness-Eveningness Questionnaire (MEQ) (Horne and Ostberg, 1976) and the Munich Chronotype Questionnaire (MCTQ) (Roenneberg et al., 2003) correlates well with invasive tests and can be completed quickly using either paper or electronic versions to maximize participant recruitment (Kantermann et al., 2015).
The MEQ consists of 19 questions, with higher scores indicating more morningness. The MCTQ collects data on sleep timing during work and work-free days to calculate a midpoint of sleep on free days, corrected for oversleep (MSFsc) (Roenneberg et al., 2012), with earlier midpoint of sleep suggesting a morning preference with earlier phase of entrainment.
Chronotype is a risk factor for cardiometabolic and mental health, with evening types being more susceptible to conditions such as obesity and bipolar disease (Hulsegge et al., 2018; Walsh et al., 2022). Here, we aimed to systematically review the evidence of an association between chronotype and patients with IMIDs.
Materials and Methods
Search Terms and Strategy
This protocol was registered with PROSPERO (CRD42021240942). A comprehensive search was conducted in MEDLINE and Embase, via OVID, to include human studies in English language published between 1 January 2000 and 16 April 2021. The search strategy is included in the appendix and the search process is described as a PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flowchart (Figure 1).
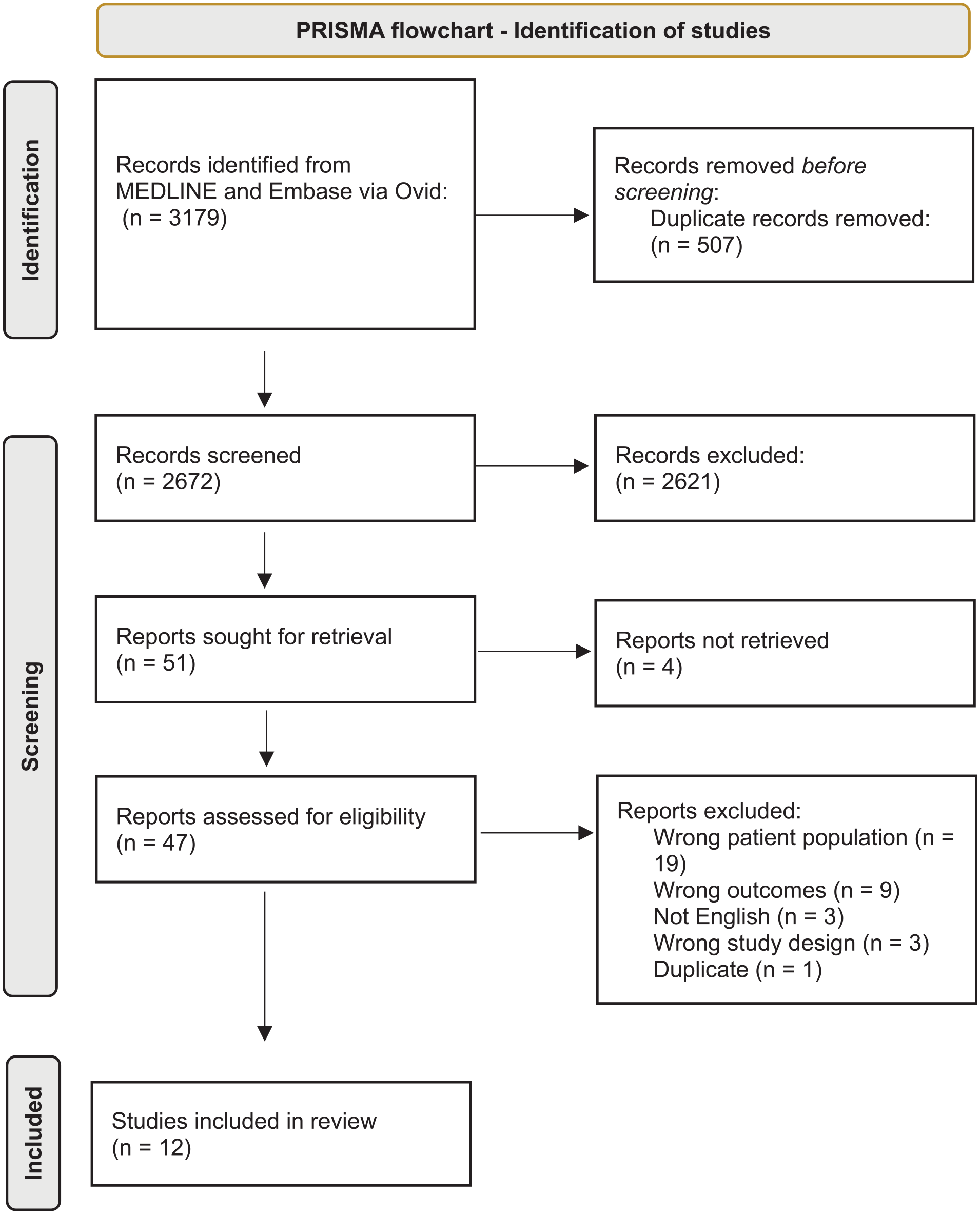
PRISMA flowchart for identification of studies.
Eligibility Criteria
Study eligibility was assessed by 2 independent reviewers (T.B. and A.M.A), according to the inclusion and exclusion criteria below:
Inclusion criteria:
(1) Case-control or cross-sectional studies; (2) included patients with RA, IBD, psoriasis, asthma, or SLE; (3) included chronotype data from either MEQ or MCTQ; (4) all clinical settings.
Exclusion criteria:
(1) Study participants <18 years old; (2) review articles.
Data Extraction and Quality Assessment
Two reviewers (T.B. and A.M.A.) independently examined and extracted the key data using Covidence software. Study quality and risk of bias were assessed by the Newcastle-Ottawa Cohort Scale (NOS) (Wells et al., 2000) and the modified NOS for cross-sectional studies (Herzog et al., 2013). Broadly, the NOS considers studies across 3 domains: study group selection, comparability between groups, and exposure, with the modified NOS considering outcome rather than exposure.
Results
Twelve studies met the inclusion criteria (Basnet et al., 2018; Chakradeo et al., 2018; Chrobak et al., 2018; Ferraz et al., 2008; Habers et al., 2021; Henry et al., 2017; Keskin and Sahbaz, 2020; Maidstone et al., 2021b; Merikanto et al., 2014a, 2014b; Singh et al., 2016; Wohlfahrt et al., 2014). The included study characteristics are detailed in Table 1 and the main findings are described in Table 2 and Supplementary Table 1. Studies were conducted across 7 countries: the United States (n = 3 studies), Finland (n = 3), Poland, the Netherlands, Turkey, the United Kingdom, and Brazil (all n = 1), with 1 study collecting data internationally. Five studies were classified as case-control and 7 studies were cross-sectional in design. There were no randomized controlled studies or cohort studies. Participants with IMIDs totaled 15,625 with 410,783 healthy controls across all studies (Table 3). Two studies did not have a healthy control (HC) population (Henry et al., 2017; Wohlfahrt et al., 2014) and 1 study did not report numbers of participants with IMID and controls (Basnet et al., 2018). IMIDs included IBD (n = 4 studies), RA (n = 3), asthma (n = 3), both RA and asthma (n = 1), and psoriasis (n = 1). No relevant papers examining SLE were included.
Characteristics of studies included in systematic review.
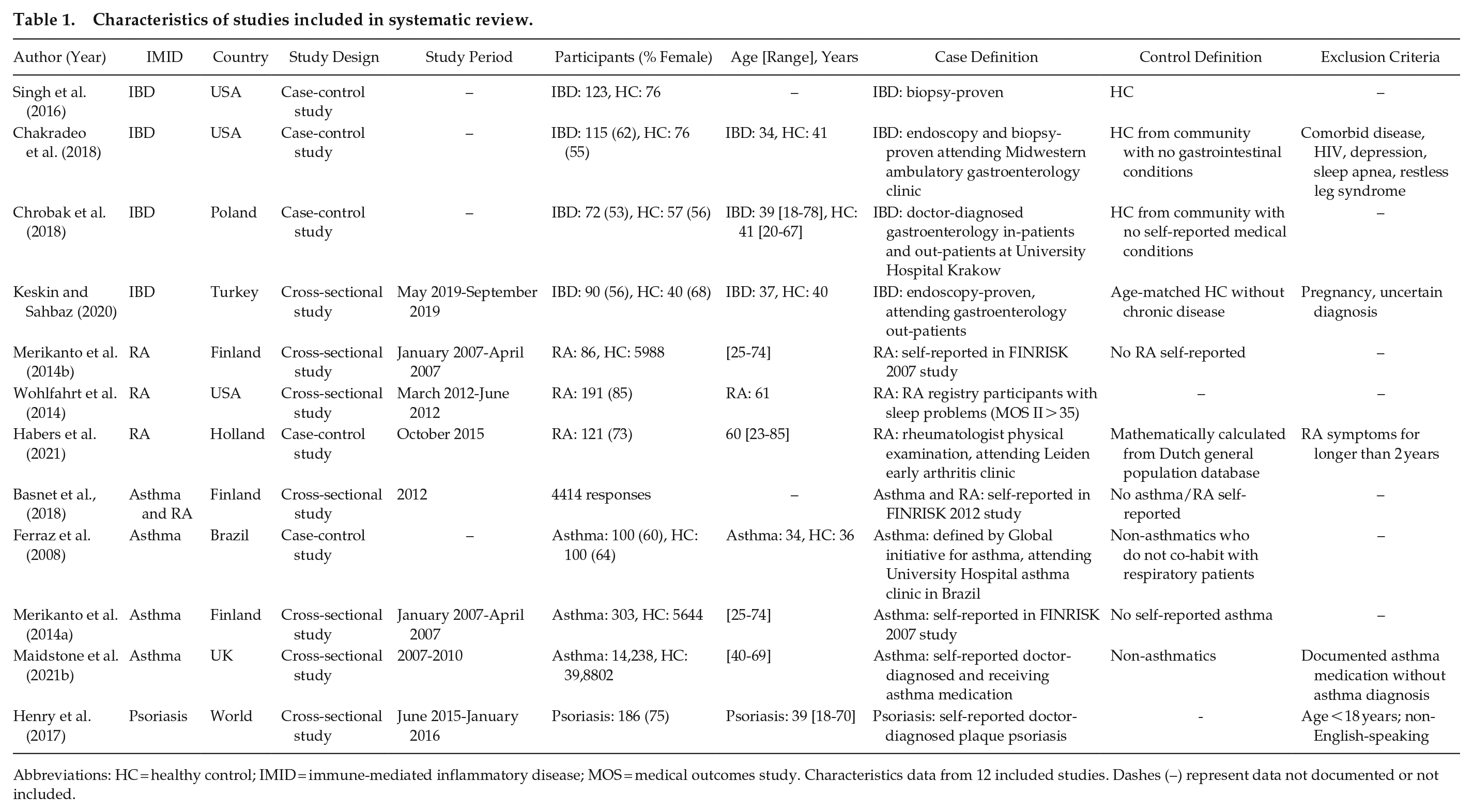
Abbreviations: HC = healthy control; IMID = immune-mediated inflammatory disease; MOS = medical outcomes study. Characteristics data from 12 included studies. Dashes (–) represent data not documented or not included.
Main comparisons, results, and risk of bias assessment for studies included in systematic review.
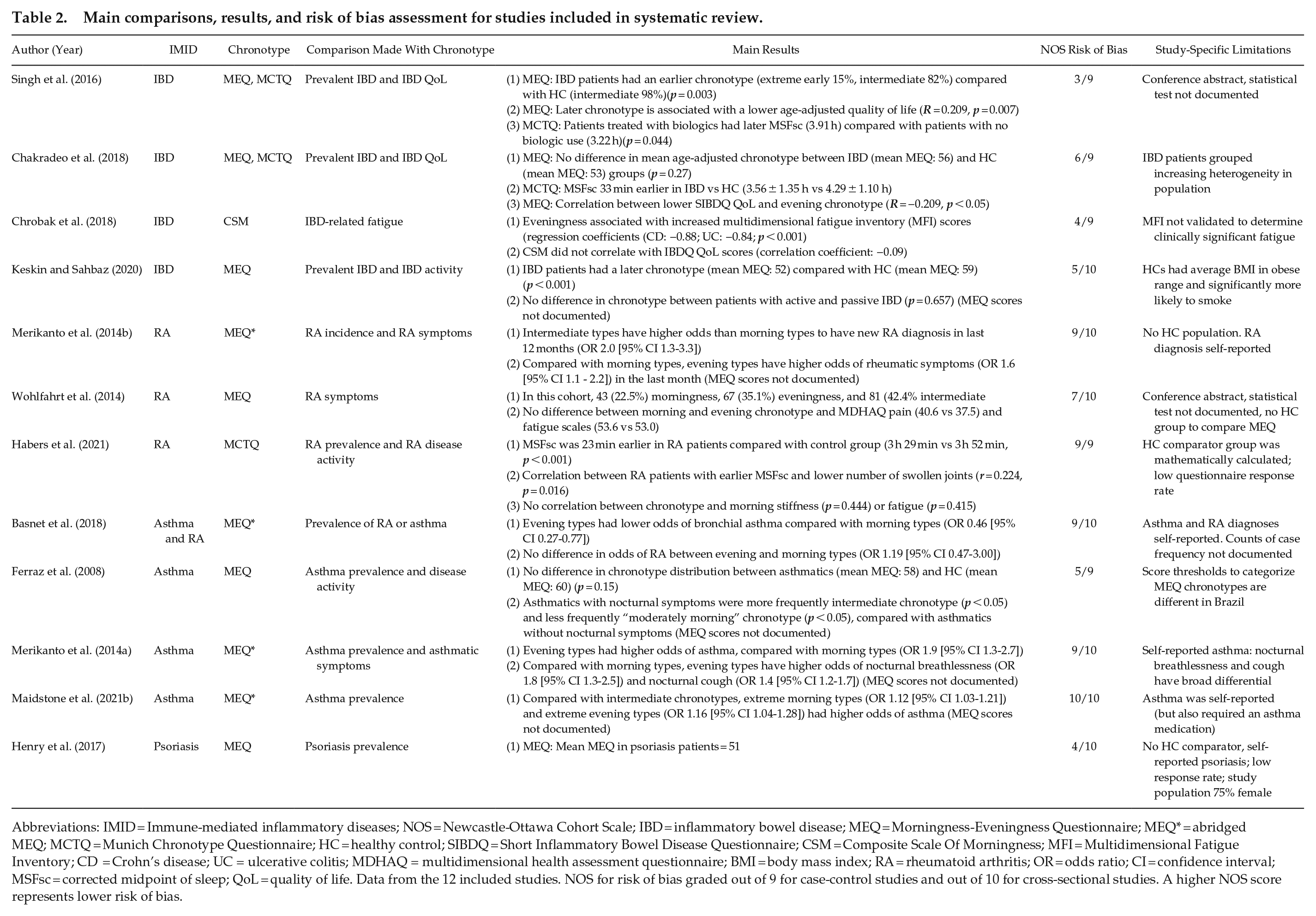
Abbreviations: IMID = Immune-mediated inflammatory diseases; NOS = Newcastle-Ottawa Cohort Scale; IBD = inflammatory bowel disease; MEQ = Morningness-Eveningness Questionnaire; MEQ* = abridged MEQ; MCTQ = Munich Chronotype Questionnaire; HC = healthy control; SIBDQ = Short Inflammatory Bowel Disease Questionnaire; CSM = Composite Scale Of Morningness; MFI = Multidimensional Fatigue Inventory; CD = Crohn’s disease; UC = ulcerative colitis; MDHAQ = multidimensional health assessment questionnaire; BMI = body mass index; RA = rheumatoid arthritis; OR = odds ratio; CI = confidence interval; MSFsc = corrected midpoint of sleep; QoL = quality of life. Data from the 12 included studies. NOS for risk of bias graded out of 9 for case-control studies and out of 10 for cross-sectional studies. A higher NOS score represents lower risk of bias.
The number of participants included in studies.
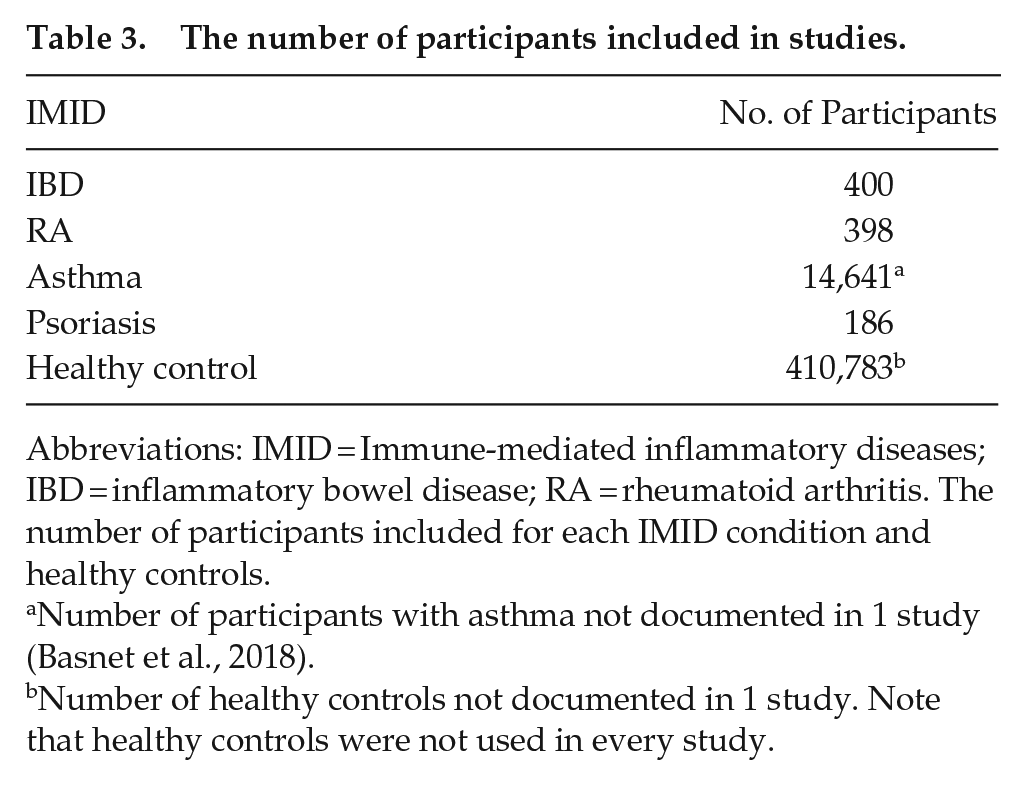
Abbreviations: IMID = Immune-mediated inflammatory diseases; IBD = inflammatory bowel disease; RA = rheumatoid arthritis. The number of participants included for each IMID condition and healthy controls.
Number of participants with asthma not documented in 1 study (Basnet et al., 2018).
Number of healthy controls not documented in 1 study. Note that healthy controls were not used in every study.
Case definition of included IMID patients ranged from self-report of doctor-diagnosed condition to confirmed clinical diagnosis by specialist doctor at secondary care clinics. Four studies independently verified case definitions with data from primary records (Chakradeo et al., 2018; Ferraz et al., 2008; Habers et al., 2021; Singh et al., 2016). Where HCs were used in case-control studies, individuals were identified in the local community with no self-reported IMIDs. In the majority of cross-sectional studies, HCs were identified by the absence of self-reported IMID being studied. One study mathematically calculated HC data by taking the MSFsc values from a large Dutch database and constructing a best fit line used to extrapolate age- and gender-matched data (Habers et al., 2021).
Comparability of cases and controls was considered to varying degrees across studies (Table 4). Maidstone et al. employed 3 detailed models (Maidstone et al., 2021b), whereas 4 studies did not document any methods for matching or adjusting data (Chrobak et al., 2018; Ferraz et al., 2008; Habers et al., 2021; Henry et al., 2017). The heterogeneity of reported data only permitted descriptive statistics. Data to perform a meta-analysis of effect estimates were unavailable.
List of covariates used by included studies.

Abbreviations: DAS = Disease activity score; BMI = body mass index; HC = healthy controls; TDI = Townsend Deprivation Index.
Four studies compared MEQ chronotype in IMID versus HC (Chakradeo et al., 2018; Ferraz et al., 2008; Keskin and Sahbaz, 2020; Singh et al., 2016). In 1 study, an earlier chronotype was more likely in IMID patients compared with HC (IBD), 2 studies found no difference in chronotype (IBD and asthma), and 1 study identified a later chronotype in IMID patients (IBD). Two of 2 (100%) studies with MCTQ data reported an earlier MSFsc in IMID patients compared with HC (Chakradeo et al., 2018; Habers et al., 2021).
Four studies examined the association of chronotype with odds of disease (Basnet et al., 2018; Maidstone et al., 2021b; Merikanto et al., 2014a, 2014b). No trends were seen across the studies, which used different comparators. For example, the three asthma studies demonstrated that evening types had higher odds of asthma compared with morning types (Merikanto et al., 2014a), lower odds of asthma compared with morning types (Basnet et al., 2018), and that extremes of chronotype have more asthma than intermediate types (Maidstone et al., 2021b).
Seven studies investigated the impact of chronotype on IMID quality of life and symptom burden (Chakradeo et al., 2018; Chrobak et al., 2018; Habers et al., 2021; Merikanto et al., 2014a, 2014b; Singh et al., 2016; Wohlfahrt et al., 2014). Five of 7 (71%) studies reported significantly worse morbidity in patients with later chronotypes, using validated metrics such as the Short IBD Questionnaire (SIBDQ), as well as patient-reported symptom burden. Two studies reported no difference in disease activity (IBD) or fatigue and pain (RA).
Discussion
This systematic review of 12 papers relating to the role of chronotype in IMID has revealed that a later chronotype may be a risk factor for worse quality of life and increased symptom burden across patients with IMIDs. The review also suggests that chronotype may be a risk factor for IMID, but the direction and magnitude of this effect vary between studies and IMID conditions.
The presence of a later chronotype associating with morbidity was interestingly observed in patients with asthma, RA, and IBD. Asthma and RA have clear diurnal variation in symptoms, with asthma exacerbations worse at night and RA joint stiffness and pain worse in the morning, correlating with peak rhythmic proinflammatory cytokine expression (Gibbs and Ray, 2013). In addition, IBD does not have a clear diurnal variation in symptoms and can present with nocturnal diarrhea, which may represent a loss of rhythmicity in homeostatic defecation. Together, this suggests that later chronotypes may be less able to effectively gate immune responses and suppress inflammatory cascades, regardless of the IMID’s peak inflammatory period. This encourages further investigation of circadian influence on common IMID inflammatory pathways.
Studies of MEQ chronotype did not demonstrate a predominant trend for chronotype in IMID. Where the mean MEQ was available, this fell within the intermediate category for IMIDs and HC, suggesting that statistically significant differences may only have limited biological significance. Two studies of MCTQ chronotype demonstrated an earlier MSFsc by 23-33 min in RA and IBD, respectively. This could suggest that phase of entrainment measured by MCTQ is more relevant than chronotype as a personality trait measured by MEQ. Regardless, further study is required to robustly characterize chronotype across a multitude of IMIDs, with reproducible and consistent metrics.
This study contributes to the growing evidence that chronotype is a relevant factor for disease development and progression. As evidence for the association of chronotype with inflammatory disease grows and the molecular drivers are investigated, the use of objective chronotype measures becomes more relevant as a method of potentially reducing bias that arises from self-reported questionnaire-based chronotype assessments. For example, blood tests such as BodyTime use biomarkers to calculate phase, with similar success in skin biopsies (Wittenbrink et al., 2018; Anafi et al., 2017). These more accurate measures of phase of entrainment could influence personalized chronotherapy by aligning medication administration to chronotype for improved efficacy.
The requirement for research into patients with IMIDs and novel insights into shared pathogenesis have a good prospect of progression with the help of IMID databases and biobanks, such as the National Institute for Health and Care Research (NIHR) IMID BioResource and IMID-Bio-UK biobanks; however, it is unclear whether chronotype is an included assessment. It is important to continue to investigate and understand common as well as unique pathways across IMID conditions with the aim of improving patient care through identification of diagnostic biomarkers and therapeutic advances such as prediction of response to therapy. Investigation of genetics across IMIDs provides an example of the rewards and challenges of identifying commonality across inflammatory diseases. Immunochip single nucleotide polymorphism (SNP) microarray technology has revealed 71 loci that significantly associate with 2 or more IMIDs; however, 14% of hits were discordant, meaning their modification of risk was increased in some IMIDs and down in others (Parkes et al., 2013). Of interest, genetic studies have highlighted components of the IL-23R response pathway significantly associated with a broad range of IMIDs, leading to novel understanding of the role of IL-23 in IMID pathogenesis, and contributed to the discovery of therapies targeting IL-23, currently licensed for use in psoriasis and IBD in the United Kingdom (Bianchi and Rogge, 2019). The circadian clock regulates components of this pathway, including Th17 effector cells and their innate counterparts, type 3 innate lymphoid cells (Yu et al., 2013; Butler and Gibbs, 2020; Godinho-Silva et al., 2019). Further work is required to explore the regulatory role of circadian rhythms in the IL-23 pathway across different inflammatory disease models.
To our knowledge, this is the first systematic review of multiple IMIDs and the contribution of chronotype. A broad search strategy covering over 20 years of research identified a collection of papers that can be taken together to understand the role of chronotype preference in chronic inflammatory conditions. Collating these studies across different diseases will help to guide future work. This work does carry limitations, including the heterogeneity of data representation and comparator groups across studies. Raw chronotype data were not available from any included study, precluding quantitative meta-analysis. Many studies did not clearly define cases and lacked adjustment for common variables known to influence chronotype, such as age and sex, making it harder to draw comparative conclusions.
Conclusion
IMIDs are a collection of chronic, often debilitating inflammatory diseases managed by separate clinical specialties. A patient’s chronotype, easily calculated via questionnaire, may help to predict the severity of symptoms in IMIDs and could contribute to risk stratification for monitoring and treatment purposes. It must be recognized that IMID patient groups can vary by age, sex, and ethnicity, requiring a patient-centered multidisciplinary treatment approach rather than a one-size-fits-all strategy. However, there is growing evidence of commonality in pathology across IMIDs at a genetic, cellular, and environmental level. The trend for evening chronotypes to experience more serious symptoms across different IMIDs highlights the potential role of circadian rhythms in gating common inflammatory pathways in IMIDs. Further research can now focus on developing strategies for robust cross-disciplinary circadian research in the hope of furthering IMID management.
Supplemental Material
sj-xlsx-1-jbr-10.1177_07487304221131114 – Supplemental material for Chronotype in Patients With Immune-Mediated Inflammatory Disease: A Systematic Review
Supplemental material, sj-xlsx-1-jbr-10.1177_07487304221131114 for Chronotype in Patients With Immune-Mediated Inflammatory Disease: A Systematic Review by Thomas D. Butler, Aala Mohammed Ali, Julie E. Gibbs and John T. McLaughlin in Journal of Biological Rhythms
Footnotes
Appendix
Search strategy for Ovid MEDLINE and Epub Ahead of Print, In-Process, In-Data-Review & Other Non-Indexed Citations and Daily, 1946-present, Embase 1974-present, and EBM Reviews—Cochrane Database of Systematic Reviews 2005-present.

| Ovid MEDLINE(R) and Epub Ahead of Print, In-Process, In-Data-Review & Other Non-Indexed Citations, Daily and Versions <1946 to 6 May 2022> | |
| Embase <1974 to 6 May 2022> | |
| EBM Reviews—Cochrane Database of Systematic Reviews <2005 to 5 May 2022> | |
| 1. | chrono*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 2. | munich chrono* question*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 3. | morningness.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 4. | inflam*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 5. | autoimmun*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 6. | psoria*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 7. | asthm*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 8. | inflammatory bowel diseas*.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 9. | ulcerative colitis.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 10. | crohn* disease.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 11. | rheumatoid arthritis.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 12. | sle.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 13. | systemic lupus erythematosus.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 14. | 1 or 2 or 3 |
| 15. | 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 |
| 16. | 14 and 15 |
| 17. | limit 16 to humans |
| 18. | limit 17 to yr =“2000 -Current” |
| 19. | “review”.mp. [mp=ti, ot, ab, tx, kw, ct, sh, hw, tn, dm, mf, dv, kf, fx, dq, nm, ox, px, rx, an, ui, sy] |
| 20. | 18 not 19 |
Acknowledgements
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: TDB is a Medical Research Council Clinical Research Training Fellow (MR/S02199X/1). JEG is a Versus Arthritis Senior Research Fellow (22625), and research in the lab is additionally funded by the MRC (MR/S002715/1 and MR/P023576/1).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
