Abstract
Individuals vary in how their circadian system synchronizes with the cyclic environment (zeitgeber). Assessing these differences in “phase of entrainment”—often referred to as chronotype—is an important procedure in laboratory experiments and epidemiological studies but is also increasingly applied in circadian medicine, both in diagnosis and therapy. While biochemical measurements (e.g., dim-light melatonin onset [DLMO]) of internal time are still the gold standard, they are laborious, expensive, and mostly rely on special conditions (e.g., dim light). Chronotype estimation in the form of questionnaires is useful in approximating the timing of an individual’s circadian clock. They are simple, inexpensive, and location independent (e.g., administrable on- and offline) and can therefore be easily administered to many individuals. The Munich ChronoType Questionnaire (MCTQ) is an established instrument to assess chronotype by asking subjects about their sleep-wake-behavior. Here we present a shortened version of the MCTQ, the µMCTQ, for use in situations in which instrument length is critical, such as in large cohort studies. The µMCTQ contains only the core chronotype module of the standard MCTQ (stdMCTQ), which was shortened and adapted from 17 to 6 essential questions, allowing for a quick assessment of chronotype and other related parameters such as social jetlag and sleep duration. µMCTQ results correspond well to the ones collected by the stdMCTQ and are externally validated by actimetry and DLMO, assessed at home (no measure of compliance). Sleep onset, midpoint of sleep, and the µMCTQ-derived marker of chronotype showed slight deviations toward earlier times in the µMCTQ when compared with the stdMCTQ (<35 min). The µMCTQ assessment of chronotype showed good test-retest reliability and correlated significantly with phase markers from actimetry and melatonin (DLMO), especially with measurements taken on work-free days. Because of its brevity, the µMCTQ represents an ideal tool to estimate individual internal time in time-critical contexts, from large cohort studies to individualized medicine.
Our daily lives are controlled by at least 2 “clocks,” which historically used to be in phase: (1) the sun clock that defines day and night, dawn and dusk, or photoperiod that—depending on latitude—changes over the year and (2) social clocks that represent “local time” and allow us to interact with others (school, work, business hours). Over the course of history, we introduced time zones and Daylight Saving Time (DST), thereby separating local time and sun time. Circadian clocks synchronize to the 24-h day predominately through light and darkness, but the strength of this zeitgeber has greatly decreased during industrialization, as humans live predominantly in buildings throughout the day and artificially illuminate the nights. Everyone’s circadian system establishes its own specific phase relationship with the zeitgeber cycle (phase of entrainment [PoE]), evidenced by the difference in timing of biological rhythms in reference to the light-dark cycle between individuals, for example, melatonin, temperature, peak of activity/activity onset, peak of cortisol, or behavioral timing like that of sleep and wake (Roenneberg et al., 2003a). These differences in PoE are commonly called chronotype, ranging from extreme early chronotypes (larks) to extreme late chronotypes (owls). As a consequence of the changed light-dark cycles, the PoE of extreme early types has become even earlier and that of all the other chronotypes has become delayed, greatly widening the difference between early and late chronotypes, especially within urban populations (Roenneberg et al., 2007b; Stothard et al., 2017; Swaminathan et al., 2017; Wright et al., 2013).
Since practically all functions in our body are directly or indirectly organized by the circadian clock, temporal inconsistencies between biological and social timing become problematic, increasing the need to estimate individual internal time (PoE, chronotype, circadian phase) in research or medicine (from diagnosis to treatment). The gold standard for assessing circadian phase is measuring dim-light melatonin onset (DLMO) in samples collected in highly controlled settings, in blood, urine, or saliva (Benloucif et al., 2008; Klerman et al., 2002; Lewy and Sack, 1989). However, these measurements are expensive and cumbersome, involving multiple, well-timed samplings. Although circadian researchers are currently developing methods to assess circadian state with 1 to 2 measurements, these so far still require sampling blood, involving many known complications that limit their application in large-scale studies (Braun et al., 2018; Laing et al., 2017; Wittenbrink et al., 2018). A cost-effective, scalable, and noninvasive solution to this challenge is the use of questionnaires.
The first questionnaire developed to detect individual differences in circadian rhythms, the Morningness-Eveningness Questionnaire, uses temporal preferences to compute a score and classify individuals into chronotypes accordingly (Horne and Ostberg, 1976). The Munich ChronoType Questionnaire (MCTQ), on the other hand, was introduced in 2003 (Roenneberg et al., 2003b) and considers chronotype as the phase relationship between the circadian system of an individual and the zeitgeber cycle: their circadian state. Since it is virtually impossible to assess the phase of all rhythmic processes in humans, the MCTQ uses sleep timing as a phase marker to estimate chronotype. The standard core questions inquire about sleep times separately for work and work-free days. Considering that sleep on work-free days is not as restricted by social constraints, chronotype can be estimated using the midpoint between sleep onset and sleep end on free days (MSF), which is corrected for potential oversleep on free days (to compensate for sleep debt accumulated over the workweek, MSFsc), therefore accounting for the homeostatic process influencing sleep. Other modules assess the use of stimulants or biographic information, for example.
The stdMCTQ-MSFsc has already been shown to correlate well with data from sleep logs, wrist actimetry, and DLMO (Kantermann et al., 2015; Kitamura et al., 2014; Roenneberg et al., 2007a; Wright et al., 2013). It has been used for 15 years to assess thousands of peoples’ sleep behavior (the MCTQ database associated with its online version alone contains ≈300,000 entries).
Thus, the µMCTQ was developed as an ultra-short version of the stdMCTQ. This questionnaire makes use of the same principles as the stdMCTQ but contains only the essential questions of the MCTQ’s core chronotype module. As with the stdMCTQ, MSFsc serves as a marker for chronotype and an approximation for PoE. The µMCTQ therefore allows for a swift assessment of the timing of an individuals’ clock, which can be especially useful in larger cohort studies, long durations of investigation, or for efforts of personalized medical practice.
Here we present the validation of the circadian phase assessment by the µMCTQ against assessments by the stdMCTQ (study 1) as well as DLMO and actimetry (study 2). In the supplementary material, we provide additional data supporting the validity of the stdMCTQ for assessing PoE (study 3). Our results show that both the µMCTQ and the stdMCTQ have good validity against commonly used physiological and behavioral markers of PoE.
Methods
Development of the µMCTQ
The µMCTQ was developed starting from the original MCTQ (Roenneberg et al., 2003b), in the following referred to as standard MCTQ (stdMCTQ). The stdMCTQ asks simple questions about sleep-wake behavior, separately for work and work-free days. Its core module, which focuses on the estimation of chronotype, contains a total of 17 questions (other optional modules, e.g., regarding the use of stimulants and sociodemographics have varying lengths). The original idea of the stdMCTQ core questions was to accompany people into and out of bed (see Fig. 1 for the complete questionnaire). Since sleep onset (falling asleep) and bed time (going to bed) are often confused, we asked people questions about every step: (1) going to bed, (2) being busy in bed before deciding (3) to prepare for sleep (e.g., by switching off lights), (4) falling asleep (the last 2 indicate sleep latency), (5) waking up, and (6) finally getting up (the last 2 indicate sleep inertia). The stdMCTQ and more information on it can be found at http://thewep.org/documentations/mctq.

The core module of the standard Munich ChronoType Questionnaire (stdMCTQ) asks questions about sleep timing in relation to the weekly structure. It does so by leading the sleeper in and out of bed in 6 steps, both for workdays and work-free days. In total, participants need to answer 14 to 17 questions (depending on specific answers). On a separate page, participants are given instructions on how to fill in the stdMCTQ, for example, “take an example month” or “according to the past 4 weeks.”
Naturally, in aiming for brevity, the µMCTQ (Fig.2) contains only questions from the core chronotype module of the stdMCTQ and no questions from the optional additional stdMCTQ modules. The core module was then reduced to questions pertaining to the core variables, which were slightly modified but are content-wise identical. Since only sleep onset and sleep end as well as the weekly structure are used to calculate the most important stdMCTQ variables (MSF, MSFsc, sleep duration, and social jetlag), we reduced the 17 questions to 6 questions, probing only these events. We also tried to make participants aware that we do not mean bedtime or rise time but actual time of falling asleep or waking up.

The entire ultra-short version of the Munich ChronoType Questionnaire (µMCTQ) consists of a short explanatory introduction, 2 work-related questions, another short instruction, and 2 questions about sleep timing each for work and work-free days. Thus, participants have to answer 6 questions in total.
Both the stdMCTQ and the µMCTQ estimate chronotype in 2 steps. First, the midpoint of sleep on work-free days (MSF) is calculated based on sleep onset (SOf) and sleep end (SEf): MSF = SOf + (SEf – SOf)/2, whereby (SEf – SOf) provides the sleep duration on work-free days (SDf). The same variables can be assessed for workdays: midpoint of sleep on workdays (MSW = SOw + (SEw – SOw)/2) and sleep duration on workdays (SDw = SEw – SOw).
The second step to compute the chronotype indicator is to further correct MSF for the potential sleep debt accumulated during the workweek. This linear correction is based on the weighted average of sleep duration across the week (SDweek) and on the sleep duration on work-free days (SDf). The difference of the two is taken as an estimate for how much longer subjects slept on a work-free day (versus if they had no prior sleep debt, when SDf ≤ SDw): if SDf ≤ SDw, MSFsc = MSF; if SDf > SDw, MSFsc = MSF – (SDf – SDweek)/2. Free days are used for chronotyping, as those are the days assumed to be relatively free of constraints on sleep-wake behavior. In the stdMCTQ, participants are asked to specify the need of an alarm clock (“yes” or “no” answer): MSFsc calculations are considered only when the participant does not use/need an alarm clock on work-free days. In the µMCTQ, however, participants are asked to report their wake-up times only on work- free days on which they do not use an alarm clock. Therefore MSFsc, derived from the µMCTQ can be computed for all subjects. A detailed overview of the calculations of the mentioned variables can also be found in the supplementary material (Suppl. Table S2).
Validation of the µMCTQ
The validation of the µMCTQ against the stdMCTQ was carried out at the University of Padova, Italy, and is further referred to as study 1. The study for the validation of the µMCTQ against the phase markers from melatonin (DLMO) and actimetry data was carried out at the Ludwig Maximilian University in Munich, Germany, and is referred to as study 2. Data supporting the validity of the stdMCTQ against actimetry are included in the supplementary material and referred to as study 3.
The Italian version of the µMCTQ, used in study 1, was obtained by the Sackett procedure (i.e., forward translation, expert evaluation, independent back-translation, pretesting, and definition of the final version). The German µMCTQ version used in study 2 was a simple translation from the English µMCTQ version into German. Both translated versions (German and Italian) ask individuals to use the 24-h military time format (e.g., 23:00 h instead of 11:00 p.m.). Table 1 offers an overview of the different sample characteristics in the different studies. Detailed descriptions of the study designs can be found below.
Data set characteristics.
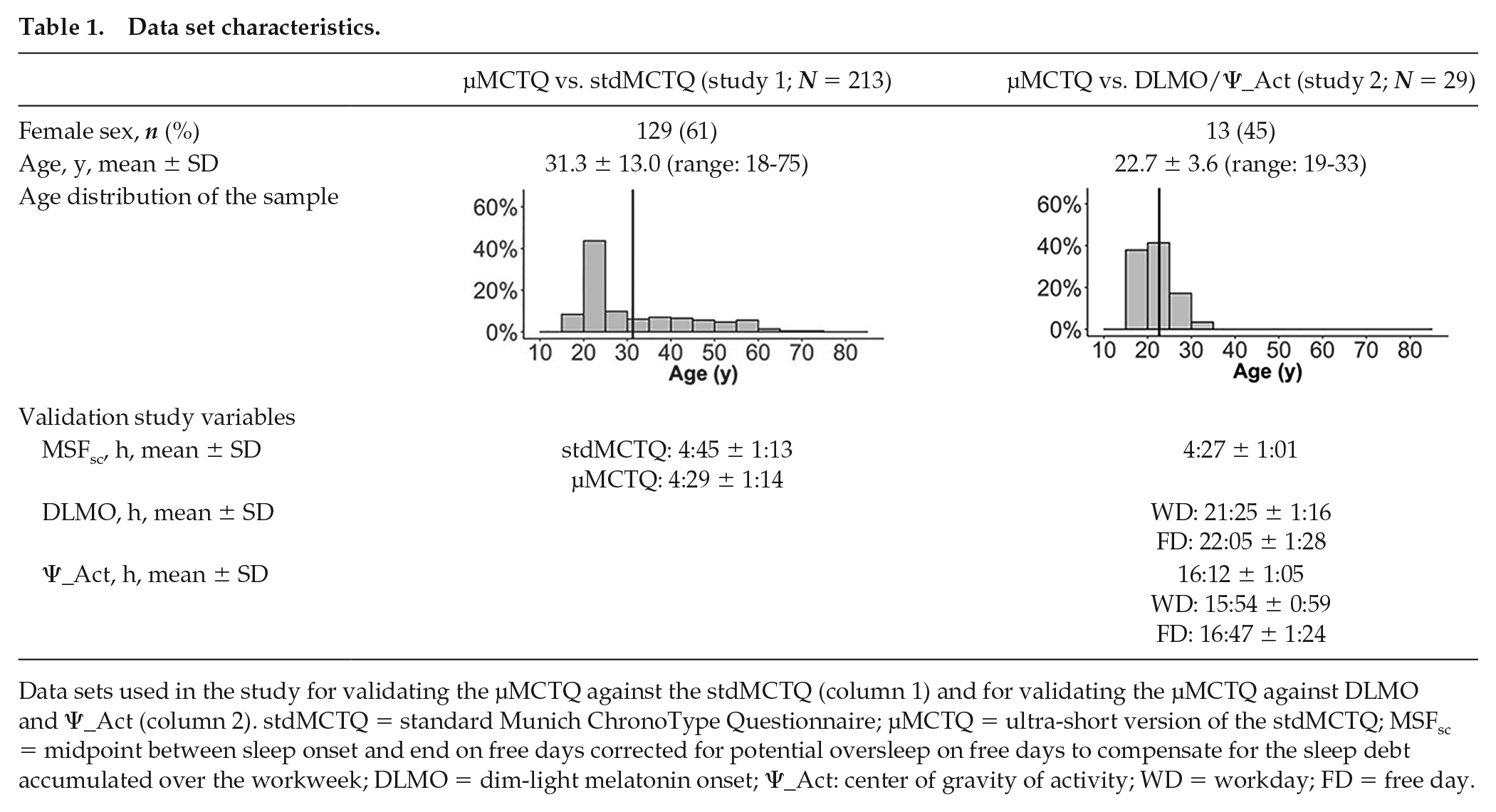
Data sets used in the study for validating the µMCTQ against the stdMCTQ (column 1) and for validating the µMCTQ against DLMO and Ψ_Act (column 2). stdMCTQ = standard Munich ChronoType Questionnaire; µMCTQ = ultra-short version of the stdMCTQ; MSFsc = midpoint between sleep onset and end on free days corrected for potential oversleep on free days to compensate for the sleep debt accumulated over the workweek; DLMO = dim-light melatonin onset; Ψ_Act: center of gravity of activity; WD = workday; FD = free day.
Study 1: Validation of the µMCTQ against the stdMCTQ
Participants
For the validation of the µMCTQ against the stdMCTQ, we recruited 361 healthy volunteers as part of a series of popular science initiatives (open Padova University event “Veneto Research Night” 2015) during which Padova University scientists provided the general public with information on their ongoing research. Forty-eight subjects were excluded because of significant medical history, shift work, and/or incomplete questionnaire responses. Because of alarm clock use on free days, 87 participants were excluded from all analyses. Thus, the final population included 213 individuals (129 women; age [mean ± standard deviation]: 31.3 ± 13.0 years). See Table 1 for further details.
Study Design
Subjects completed a personal datasheet to include basic demographic and medical information, height, and weight before they filled out the Italian translations of the µMCTQ followed by the stdMCTQ (always in the same sequence). The researchers G.F., M.S., and S.M. provided assistance and instructions on completion of the questionnaires. Participants provided written, informed consent.
Measurements
Participants filled in the µMCTQ (time span referring to the past 6 weeks) and stdMCTQ (referring to a “regular week”).
Statistical Analyses
Bland-Altman plots
The relationship between the µMCTQ and the stdMCTQ was studied using Bland-Altman plots (Bland and Altman, 1986, 1999). The Bland-Altman plot is a graphical method to compare 2 measurements techniques: the differences between the measurements obtained by the 2 techniques are plotted against the average of the measurements. Horizontal lines are drawn at the mean difference and at the 95% limits of agreement, which are defined as the mean difference ±1.96 times the standard deviation of the differences. If these limits do not exceed the maximum “allowable” difference between the methods (i.e., the differences are not yet physiologically or clinically relevant), the 2 measurement methods are considered to be in agreement and can be used interchangeably. Finally, if some degree of correlation exists between the differences and the averages on the Bland-Altman plot, the over- or underestimation of one method versus the other increases/decreases depending on the absolute value or size of the measurement. Since the differences between measurements were not normally distributed, correlations were tested using Spearman’s rho.
Correlations
Correlations between variables from the stdMCTQ and the µMCTQ were tested using Pearson’s
Study 2: Validation of the µMCTQ against Activity and Melatonin Phase (DLMO)
Participants
Thirty participants (15 women, age [mean ± standard deviation]: 22.7 ± 3.6 years) were recruited by convenience sampling (mostly students recruited by flyers on campus). Exclusion criteria were irregular work schedules or shift work or a transmeridian flight during a 3-month period prior to study participation. All participants provided informed consent; the study was approved by the Ethics Committee of Ludwig Maximilian University (approval 517-15) and conducted in accordance with the Declaration of Helsinki.
Study Design
Because of reasons of feasibility, the study spanned the autumn DST change. The cohort was divided into 2 groups. Group 1 (G1; 9 women and 6 men) started the study shortly before the DST change in October 2015 and was monitored for 6 weeks. Group 2 (G2; 6 women and 9 men) started a week after the time change and was monitored for 4 weeks. One subject (from G1) did not fill in the questionnaire correctly and had to be excluded.
Participants filled out the µMCTQ once at study onset. Throughout the course of the study, they filled out an online version of the µMCTQ on a daily basis (similar to a sleep-log), but the daily assessments were used only to generate date-type data (work- or work-free day).
Measurements
In addition to the µMCTQ, actimetry and home DLMO data were collected for circadian phase determination. In G1, saliva samples were collected during the first week before the time change, and the µMCTQ was filled in 8 days before the time change. In G2, saliva samples were taken in the second week after the time change, and the µMCTQ was answered 5 days after the time change.
Activity phase assessment
Actimeters are wrist-worn devices that measure locomotor activity by accelerometry. Devices (Daqtometers by Daqtix GmbH) were worn throughout the entire study period (G1: 6 weeks, G2: 4 weeks). Recordings from G1 before the DST change and the first week after the time change were excluded from the analyses. Activity was recorded at 1 Hz, and the average activity counts were stored every 30 s. For analyses, data were averaged into 10-min bins. Participants kept a diary about day types (work or work-free days). We used the software ChronoSapiens (Chronsulting UG; Roenneberg et al., 2015) to assess the daily phase of general locomotor activity measured by the wrist-worn actimeters. For every day, we calculated the acrophase of the best 1-harmonic, 24-h cosine fit (Ψ_Act). The advantage of this phase marker is that it does not rely on any other computations of the actimetry signal such as algorithms identifying sleep. Ψ_Act for every subject was calculated using daily averages across all days. Individual averages were also calculated separately for workdays (Ψ_Actw) and work-free days (Ψ_Actf). For the calculation of the general Ψ_Act results, we also included days not specified as work or work free. Data are expressed as local time.
DLMO assessment
DLMO was assessed at home in this study for reasons of feasibility. Home DLMOs have previously been reported to show a good correspondence to lab DLMOs (Burgess et al., 2015; Pullman et al., 2012), especially when participants’ compliance is being objectively monitored (which, however, was not the case in our study). Participants had an appointment with study team members, during which they were instructed on how to collect saliva samples. The volunteers were asked to collect 7 saliva samples hourly once on a workday and once on a work-free day starting 6 h before their usual sleep timing. Subjects were told to close the blinds and turn off as many lights as possible 1 h before saliva collection started and were given blue light–blocking sunglasses to wear during the period of collection. They were also instructed to dim screen lights of electronic devices, not to change their position at least 5 min before taking the sample, and to rinse their mouth with clear water before collection. They were also told not to eat chocolate or bananas or to drink coffee or alcohol during the period of saliva collection. A written version of the instructions was provided as well. Samples were collected using Salivette cotton swabs (Sarstedt AG & Co.). The participants wrote down the times of collection into a log provided by the study team, without further measures of compliance, and they kept the samples in the fridge (~4°C) until bringing them to the laboratory (storage duration <7 days), where they were processed and kept at −20°C until further use (storage duration <3 months). The duration of sample storage was in accordance with manufacturer guidelines.
Chrono@work (Groningen, the Netherlands) analyzed the samples. Melatonin concentrations were assessed using direct saliva melatonin radioimmunoassay test kits (RK-DSM; Bühlmann Laboratories, Alere Health, Tilburg, the Netherlands). DLMO was calculated by linear interpolation between the time points before and after melatonin concentrations crossed and stayed above the threshold of 3 pg/mL. We opted for the fixed threshold method for sample size reasons (insufficient points to calculate baseline for some individuals), but similar results were seen using the threshold of 2 standard deviations above the baseline (see Suppl Fig. S8). The lower limit detection of the kit was 0.3 pg/mL. The intra-assay variability was 15.9% at low melatonin concentration (mean = 2.0 pg/mL,
Statistical Analyses
Shapiro-Wilk test and inspection of histograms were used to test the variables for normality, and all variables showed a normal distribution. Correlations between MSFsc (as assessed by the µMCTQ) and DLMO as well as the different Ψ_Act values were tested using Pearson’s correlation. An alpha level of 0.05 was chosen. SPSS 24 and GraphPad Prism 6 were used for statistical analyses. Graphs were plotted using the R package ggplot2 (Wickham, 2016).
Test-Retest Reliability
We performed test-retest measurements in 37 subjects to test chronotype reliability (µMCTQ-MSFsc) over 2 different time frames. We correlated assessments taken 56 to 63 or 14 to 18 days apart. Twenty students filled in the English version of the µMCTQ (age [mean ± SD]: 23.8 ± 3.3 years; 40% female; interval between assessments: 56-63 days). Eighteen subjects recruited through snowball sampling filled in the German or English online versions of the µMCTQ (age [mean ± SD]: 33.5 ± 7.8 years; 39% female; interval between assessments: 14-18 days).
Results
Study 1: Validation of the µMCTQ against the stdMCTQ
Generally, sleep timing and MSFsc corresponded well between the µMCTQ and stdMCTQ but showed systematic deviations toward earlier times in the µMCTQ in most of the assessed variables.
The µMCTQ estimated sleep onset on workdays and work-free days (SOw and SOf) earlier than the stdMCTQ (mean difference ± SD: SOw 24.7 ± 28.4 min and SOf 30.8 ± 37.7 min; Fig. 3), with limits of agreement of less than 120 min: −30.9 to 80.3 min for workdays and −42.1 to 104.7 min for work-free days.
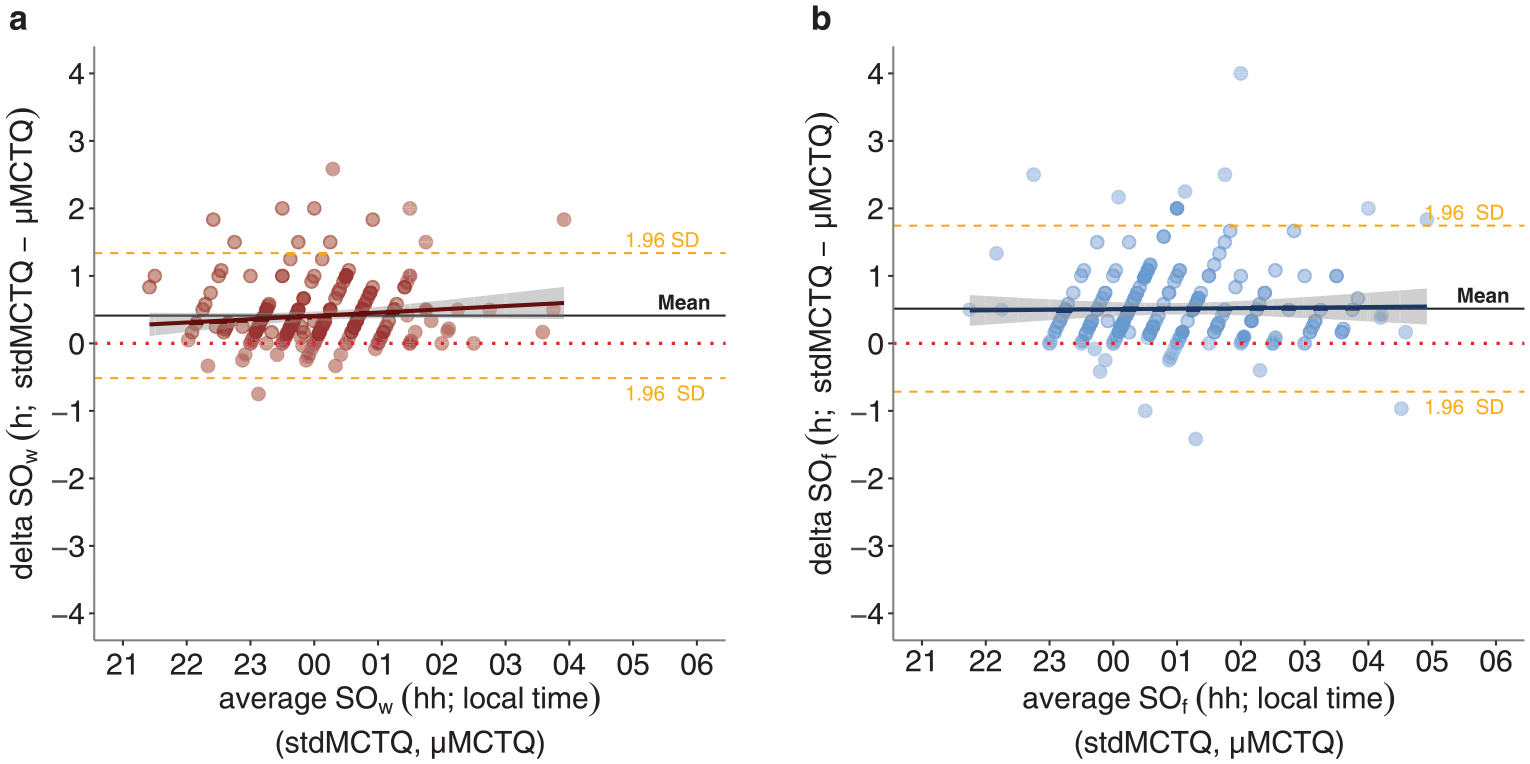
Bland-Altman plots of sleep onset on workdays (a) and work-free days (b) (method- differences on the
In contrast, the 2 tools produced similar estimates for sleep end on both workdays and work-free days (mean difference ± SD: SEw −2.7 ± 19.0 min, SEf 2.0 ± 28.7 min; Fig. 4), with a limit of agreement of less than 60 min: SEw −39.9 to 34.6 min, SEf −54.3 to 58.3 min.
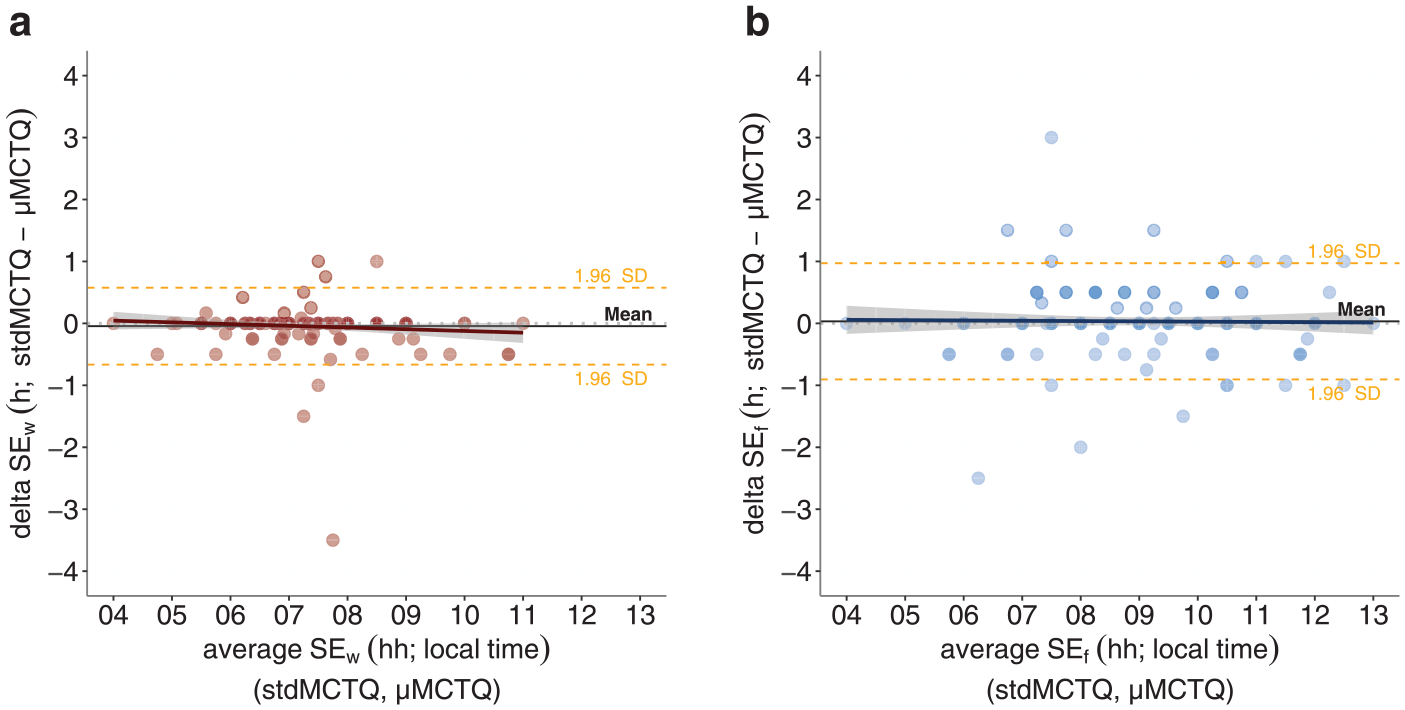
Bland-Altman plots of SEw (a) and SEf (b) (method differences on the
The average estimate of mid-sleep on workdays (MSW) and on work-free days (MSF) produced by the µMCTQ was less than 20 min earlier than that of the stdMCTQ (MSW mean difference ± SD: 11.0 ± 17.1 min; MSF mean difference ± SD: 16.4 ± 24.7 min; Fig. 5). MSFsc was estimated as less than 20 min earlier by the µMCTQ than by the stdMCTQ (MSFsc mean difference ± SD: 16.3 ± 27.1 min; Fig. 5). Their limits of agreement ranged from about 30 to 70 min: MSW −22.5 to 44.5 min, MSF −32.0 to 64.9 min; MSFsc −36.8 to 69.4 min.
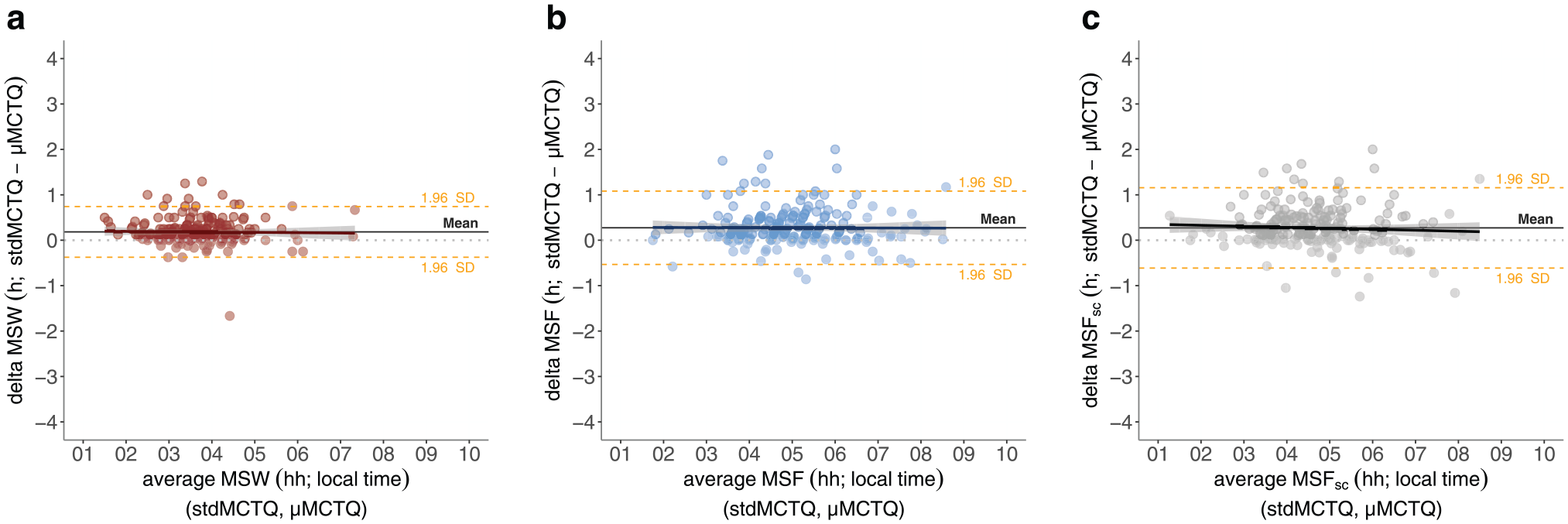
Bland-Altman plots of (a) MSW, (b) MSF, and (c) MSFsc (method differences on the
Correlations between the µMCTQ and stdMCTQ in terms of sleep onset (workdays and work-free days), sleep end (workdays and work-free days), MSF, MSW, and MSFsc were all statistically significant and produced coefficients ranging from 0.89 to 0.95 (Suppl. Fig. S1). The same correlations are shown in Supplementary Figures S2 to S4 differentiated into age categories.
Study 2: Validation of the µMCTQ against Activity (Ψ_Act) and Melatonin Phase (DLMO)
MSFsc, as an indicator of chronotype, showed a moderately positive correlation with Ψ_Act and Ψ_Actf and a tendency to correlate with Ψ_Actw (Fig. 6). Correlations of MSW and Ψ_Actw as well as MSF and Ψ_Actf are provided in the supplementary material (Suppl. Fig. S6).
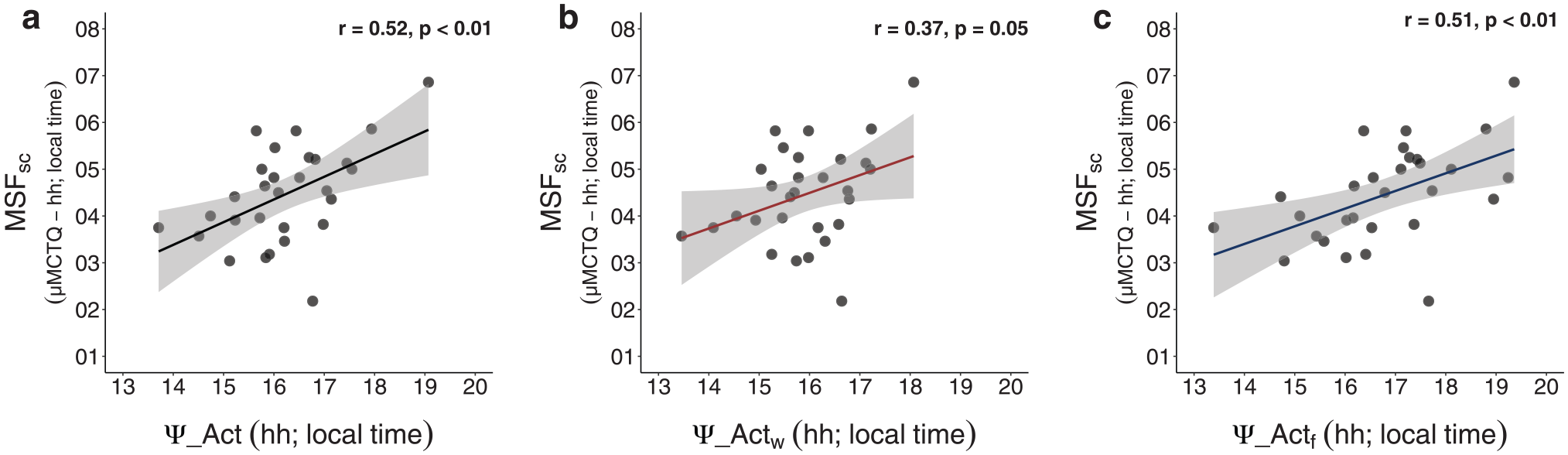
Associations between MSFsc from the ultra-short version of the Munich ChronoType Questionnaire (µMCTQ) and actimetry phase (Ψ_Act). MSFsc correlates significantly with Ψ_Act (a) as well as with Ψ_Act on work-free days (Ψ_Actf, c), but tended to be associated with Ψ_Act only measured on workdays (Ψ_Actw; b). The gray-shaded area around the regression line represents the 95% confidence interval. Results of Pearson correlations are provided in each graph. Data are expressed in local time for both variables.
MSFsc was positively associated with DLMOf but not with DLMOw (Fig. 7). Correlations of MSW and DLMOw as well as of MSF and DLMOf are provided in the supplementary material (Suppl. Fig. S7).
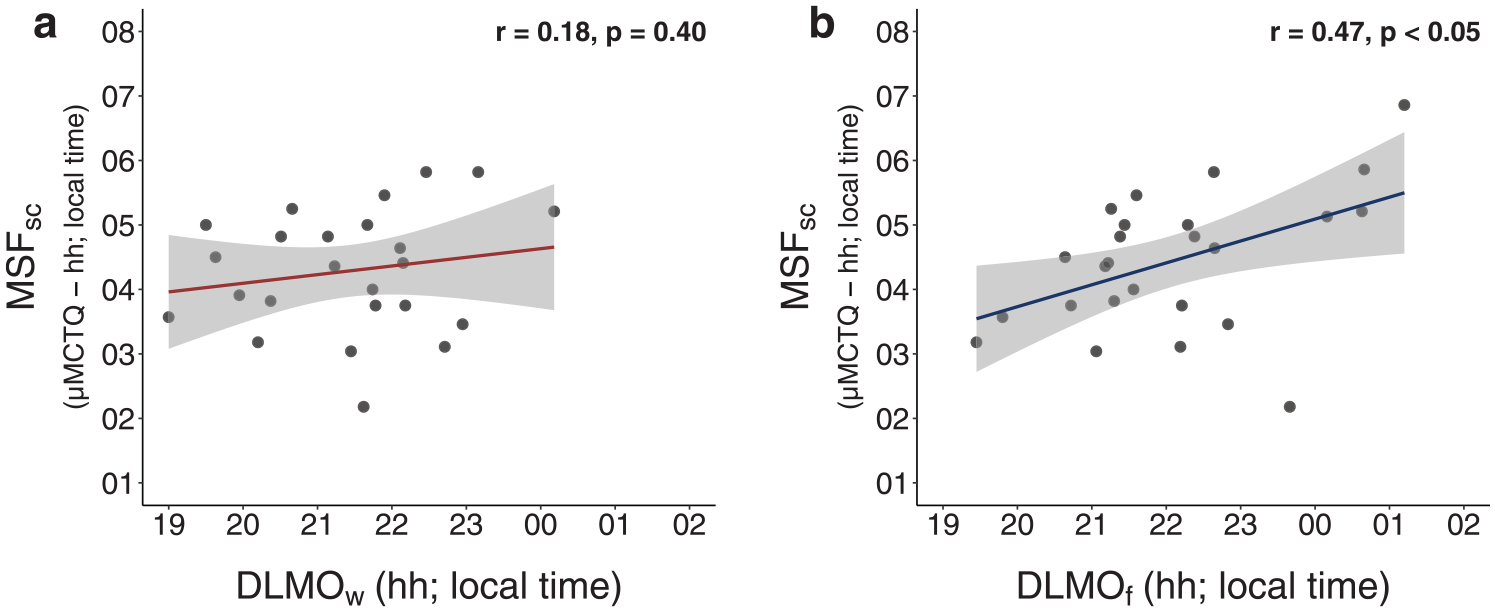
Association between MSFsc from the ultra-short version of the Munich ChronoType Questionnaire (µMCTQ) and dim-light melatonin onset (DLMO). MSFsc correlates significantly with DLMO measured on work-free days (DLMOf, b) but not with DLMO measured on workdays (DLMOw, a). The gray-shaded area around the regression line represents the 95% confidence interval. Results of Pearson correlations are provided in each graph. Data are expressed in local time for both variables.
The µMCTQ showed good test-retest reliability within different time frames (see the Methods section: ~60 days: Pearson’s
Discussion
Our results show that both the µMCTQ and the stdMCTQ are valid instruments to assess PoE and that the 2 questionnaires show good correspondence with each other. The indicator of chronotype (MSFsc), as measured by the µMCTQ, correlates with the timing of both melatonin (DLMO) and activity (Ψ_Act; center of gravity of best fit). We also replicate previous findings, showing the stdMCTQ to correspond with actimetry measures (see the supplementary material).
Correspondence between µMCTQ and stdMCTQ
Overall, all measurements derived from the µMCTQ and the stdMCTQ are in good agreement with each other. The µMCTQ yielded slightly earlier results for sleep onset and thus also for MSW, MSF, and MSFsc. A likely reason for this systematic difference in onset timing is that the µMCTQ lists and asks about less detail on the “going-to-bed-and-falling-asleep process.” The µMCTQ does not lead people in and out of bed but asks directly for the time that participants usually fall asleep. Some participants may interpret this as the time when they were prepared to sleep and would therefore indicate an earlier time than actual sleep onset depending on their sleep latency. Alternatively, some people may misinterpret the detailed descriptions in the stdMCTQ explaining the different stages from going to bed to getting up (Fig. 1). For example, different people may have different concepts of what “sleep preparation” means. Does sleep preparation include sleep latency or not? Since the stdMCTQ calculates sleep onset by adding sleep latency to the time people indicate for preparing for sleep, latency might occasionally be added twice. The average sleep latency in the MCTQ database is 15.4 ± 15.2 min for free days and 18.8 ± 17.5 minutes for workdays. Notably, sleep end was not different between µMCTQ and stdMCTQ. An earlier estimate of sleep onset also influences MSW, MSF, and MSFsc. A difference of less than 20 min for MSFsc is lower than the median intraindividual variance of Ψ_Actf observed in our study (MCTQ study median: 110 min, Q1-Q3: 40-250 min; µMCTQ study median: 126 min, Q1-Q3: 76-192 min) and is well within the variance of both questionnaires and other instruments.
Furthermore, correlations of the stdMCTQ with Ψ_Act (Suppl. Fig. S10) were not significantly stronger than those of the µMCTQ with the same measurement (Ψ_Actf vs. stdMCTQ-MSFsc and Ψ_Actf vs. µMCTQ-MSFsc: Fisher’s Z test, z = 0.26, ns; Ψ_Act vs. stdMCTQ-MSFsc and Ψ_Act vs. µMCTQ-MSFsc: Fisher’s Z test, z = −0.06, ns).
Both the µMCTQ and the stdMCTQ chronotype showed good test-retest reliability (see Suppl. Figs. S9 and S12). Regarding the stability of our chronotype estimation, we expect the indicator variable to vary across time. This is because we are estimating chronotype as circadian state. Transient state constructs, in contrast to more enduring trait dispositions, are expected to be susceptible to influences from the environment (Boyle et al., 2015). Yet, circadian phase (estimated both using MSF and DLMO) has been shown to be fairly reproducible over months (Kantermann and Eastman, 2018).
Validation of the µMCTQ against Actigraphy and DLMO
µMCTQ-MSFsc correlated with DLMO and Ψ_Act on work-free days but was not significantly correlated with either of these markers on workdays. Since it is assumed that MSFsc reflects an estimation of PoE less affected by social constraints, a weaker correlation with parameters gathered on workdays (DLMOw, Ψ_Actw) was expected. DST, which occurred during the participation of half the subjects, might have also contributed to the observed result. The µMCTQ assesses sleep behavior in the past 6 weeks, and the second group filled out the questionnaire and measured DLMO 1 week after the time change. The release from DST causes a delay in Ψ_Act and in sleep timing that is more gradual on workdays than on work-free days. It was also shown that the process of adjustment to the time change and how long this process takes is chronotype specific, with late types delaying more readily (Kantermann et al., 2007). The transition accentuates the misalignment between internal time and sleep on workdays, which may be reflected in our data.
Although DST might have influenced our results, µMCTQ-MSFsc was still significantly correlated with DLMOf, similarly to what has been shown in other publications using the stdMCTQ. Even stronger correlations between MSFsc (as per the stdMCTQ) and DLMO (home or lab) have been previously reported (Facer-Childs et al., 2019; Kantermann et al., 2015; Wright et al., 2013). However, when comparing the strength of correlations found in our study (µMCTQ validation) to the ones cited, a significant difference is detectable only between our data and data from Facer-Childs et al. (Fisher’s z = −2.1,
Study Limitations
The different age ranges in the cohorts should be noted, as study 2 was conducted in young students with a narrow age range. Nevertheless, correspondence between the questionnaires was good across different age categories (as shown in Suppl. Figs. S2 to S4). As the stdMCTQ was shown to be valid against Ψ_Act in a sample with a wide age range (18-81 years old; see supplementary material), and the µMCTQ corresponds well with the stdMCTQ in all aspects, we do anticipate similar validations at higher ages. Furthermore, the Korean stdMCTQ has already been shown to be valid against actimetry and sleep logs in a large sample of older adults (≥65 years,
In study 2, we acquired a small, homogeneous sample consisting of mostly young university students, which could potentially affect generalizability, and a release from DST occurred in half the subjects while recording actimetry data. Still, MSFsc was significantly correlated to both DLMO and Ψ_Act when they were taken on work-free days, despite the small sample size.
The µMCTQ was administered at the beginning of the study and refers to the previous 6 weeks, not corresponding with the time monitored by actimetry. The questionnaire was administered at the beginning to avoid a bias in subjects’ responses, since daily assessments of sleep timing were also implemented.
The µMCTQ in study 2 was administered in a translated German version without back-translation into English (original language).
The µMCTQ was reduced from the stdMCTQ in an intuitive manner, rather than using more objective dimension reduction techniques.
DLMO assessment in study 2 was done at home with no measures of compliance, and self- reported collection times in DLMOs assessed at home can be rather inaccurate (Kudielka et al., 2003). However, studies have compared lab-based and home-measured DLMOs and found significant correlation between the 2 conditions regardless of measuring compliance (Burgess et al., 2015; Pullman et al., 2012). Furthermore, Pullman et al. (2012) considered the at-home assessments of DLMO to be satisfactorily accurate (compared with the corresponding lab measurements) in 62.5% to 75% of the cases.
Comparison between µMCTQ and stdMCTQ
As a shortened questionnaire, the µMCTQ naturally collects less information about peoples’ sleep behavior than the stdMCTQ. The µMCTQ does not enable the estimation of sleep latency or inertia. Furthermore, in contrast to the stdMCTQ, the µMCTQ does not inquire about reasons for externally induced waking on work-free days other than an alarm clock (e.g., children, hobbies). Nonetheless, the µMCTQ also offers an advantage when compared with the stdMCTQ: it allows for the assessment of the circadian phase of people who mostly use alarm clocks on work-free days by asking them to consider only those work-free days on which they do not use an alarm clock. We use sleep behavior on free days as a proxy for chronotype because it is less confounded by social constraints and therefore is a closer reflection of entrained phase. Only for this conceptual reason, we usually do not compute MSFsc when subjects report using alarm clocks on free days in the stdMCTQ (Pilz et al., 2018; Roenneberg et al., 2012; data from study 1). However, several studies have computed chronotype based on people who use an alarm clock on free days. While this is theoretically possible, we strongly recommend stating it clearly in the publication. Individuals who use alarm clocks on work-free days show slightly later mid-sleep and MSFsc than subjects who do not (Suppl. Fig. S5).
Time Frames of the Questionnaires
The stdMCTQ has been used for more than 15 years in studies with varying research questions and designs. Originally, it asked people about their sleep behavior “in a regular week.” Depending on the specific study design and question, the time frame assessed by the stdMCTQ has been modified to fit the demands of the investigation. Here, we suggest using 6 weeks with the µMCTQ to obtain a stable assessment that can still accommodate, for example, seasonal changes. stdMCTQ (“a regular week”) and µMCTQ (past 6 weeks) in study 1 used different time frames for assessment but still corresponded well. Only the µMCTQ was used in study 2.
Concluding Remarks
We have developed and validated an ultra-short version of an already well-established instrument, the stdMCTQ. Both the stdMCTQ and the µMCTQ can be used to estimate PoE and can serve as good alternatives to time-consuming and more expensive measurements such as sleep logs, actimetry, and DLMO. They allow the calculation of quantitative, not qualitative, variables and thereby permit a range of statistical operations impossible to conduct with categorical data.
The µMCTQ, in alignment with the stdMCTQ, represents a valuable tool to assess information about the human clock in a concise manner. It uses relevant questions established by the stdMCTQ but might offer a better alternative for large samples and longer study durations, which would benefit from shorter questionnaires that offer relevant information about sleep-wake behavior.
Supplemental Material
AMCTQ_Supplement_rev2 – Supplemental material for The µMCTQ: An Ultra-Short Version of the Munich ChronoType Questionnaire
Supplemental material, AMCTQ_Supplement_rev2 for The µMCTQ: An Ultra-Short Version of the Munich ChronoType Questionnaire by Neda Ghotbi, Luísa K. Pilz, Eva C. Winnebeck, Céline Vetter, Giulia Zerbini, David Lenssen, Giovanni Frighetto, Marco Salamanca, Rodolfo Costa, Sara Montagnese and Till Roenneberg in Journal of Biological Rhythms
Footnotes
Acknowledgements
We would like to thank Marijke Gordijn for technical support with the melatonin assays and the participants for their time and willingness to participate. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–Brasil (CAPES) Finance Code 001 (A046/2013–CAPES/PVE), the Deutscher Akademischer Austauschdienst (PROBRAL–CAPES/DAAD), as well as the FoeFoLe program at LMU (registration No. 37/2013).
Author Contributions
T.R. and E.W. developed the µMCTQ. N.G., L.K.P., S.M., and T.R. designed the studies. L.K.P., N.G., D.L., and S.M. collected and organized the data for the µMCTQ validation and C.V., T.R., and E.W. did the same for the stdMCTQ validation. N.G., L.K.P., S.M., G.F., M.S., R.C., E.W., G.Z., C.V., and T.R. analyzed the data. N.G., L.K.P., S.M., and T.R. wrote the first draft of the article. All authors read, extensively revised, and approved the final article.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
