Abstract
The immune system exhibits circadian rhythms, and its response to viral infection is influenced by the circadian clock system. Previous studies have reported associations between the time of day of vaccination against COVID-19 and production of anti-SARS-CoV-2 antibody titer. We examined the effect of vaccination time of day on anti-SARS-CoV-2 antibody titer after the first dose of vaccination with the mRNA-1273 (Moderna) COVID-19 vaccine in an adult population. A total of 332 Japanese adults participated in the present study. All participants were not infected with SARS-CoV-2 and had already received the first dose of mRNA-1273 2 to 4 weeks prior to participating in the study. The participants were asked to provide basic demographic characteristics (age, sex, medical history, allergy, medication, and mean sleep duration), the number of days after the first dose of vaccination, and the time of day of vaccination. Blood was collected from the participants, and SARS-CoV-2 antibody titers were measured. Ordinary least square regression was used for assessing the relationship between basic demographic characteristics, number of days after vaccination, time of day of vaccination, and the log10-transformed normalized antibody titer. The least square mean of antibody titers was not associated with the vaccination time and sleep durations. The least square means of antibody titers was associated with age; the antibody titers decreased in people aged 50 to 59 years and 60 to 64 years. The present findings demonstrate that the vaccination time with mRNA-1273 was not associated with the SARS-CoV-2 antibody titer in an adult population, suggesting that these results do not support restricting vaccination to a particular time of day. The present findings may be useful in optimizing SARS-CoV-2 vaccination strategies.
The circadian clock system generates and integrates daily rhythms in many physiological and behavioral functions. Immune function is also influenced by the circadian clock system (Scheiermann et al., 2013; Wyse et al., 2021). The primary outcome of vaccine administration is the production of an antibody for the disease targeted by the vaccine. Regarding the effect of vaccination on antibody production, an increase in antibody titer due to inactivated influenza vaccine was higher in older individuals but not tested in younger individuals receiving morning vaccination than in those receiving afternoon vaccination (Kurupati et al., 2017; Long et al., 2016a, 2016b; Phillips et al., 2008). Adjusting the time of day of vaccine administration could be a simple yet effective strategy to enhance antibody production, which helps protect against viral infection. Circadian rhythms have been increasingly accepted as an important determinant of immune response. Many of immune parameters such as cytokine levels, antigen presentation, and lymphocyte proliferation show circadian variation both in mice and humans (Arjona and Sarkar, 2006; Hornung et al., 2002; Petrovsky and Harrison, 1998). In addition, the severity of viral infection is dependent on the time of day of encountering the pathogen (Borrmann et al., 2021; Sengupta et al., 2019). The rhythm of T-cell counts and its functions are dependent on glucocorticoid rhythm which is under the control of circadian pacemaker in the brain (Shimba et al., 2018; Dimitrov et al., 2009). The circadian clock controls the magnitude of response of T cell to antigen presentation, and the circadian clock within T cell is required for the circadian regulation (Nobis et al., 2019). At the molecular clock level, mice loss of Bmal1 led to be elevated viral burden and more severe lung pathology in mice infected at specific time of day compared with the wild-type mice (Ehlers et al., 2018). Bmal1 deletion in either club cells or alveolar cells resulted in increased morbidity and inflammation as well as mortality after H1N1 influenza type A virus, respectively (Zhuang et al., 2019; Issah et al., 2021). Thus, circadian clock system and circadian variation of various immune functions may contribute to time of day modulation of vaccine’s efficacy.
The first official case of coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was reported in December 2019. It has since spread rapidly and evolved into a pandemic. To protect against COVID-19, the COVID-19 vaccines have been quickly developed in a short period of time. Regarding the time of day of vaccination on the antibody response to the COVID-19 vaccine, 2 recent reports (Wang et al., 2022; Zhang et al., 2021) have found that the time of day of administration differentially affects the SARS-CoV-2 antibody titer after the first- and second-dose vaccination in health care workers. The first study demonstrated a stronger immune response to morning vaccination with an inactivated COVID-19 vaccine (Sinovac Covid-19 vaccine), which resulted in a higher antibody level (Zhang et al., 2021). Another study demonstrated the opposite conclusion, with higher antibody levels detected when the SARS-CoV-2 mRNA vaccine BNT162b2 (Pfizer-BioNTech) was administered in the afternoon rather than in the morning (Wang et al., 2022). From these 2 reports in health care workers, it has been revealed that the antibody titer response following the COVID-19 vaccine was also influenced by vaccine type, participant age, sex, and number of days post vaccination. However, there are currently no reports investigating the effect of time of day of administration on the immune response to mRNA-1273 (Moderna) in adult population with no experience of night and rotating shift work. In the present study, we examined whether the time of day of vaccination affects the SARS-CoV-2 antibody titer after the first dose of mRNA-1273 in the Japanese adult population.
Materials and Methods
Ethics Statement
This study was approved by the Ethical Review Board for Life Science and Medical Research, Hokkaido University Hospital (approval no. 021-0012) and was performed in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants after they received an explanation of the study.
Study Design and Recruitment
This study was performed between 11 and 27 August 2021. Participants who received the first dose of mRNA-1273 2 to 4 weeks prior were recruited using a mailing list at Hokkaido University. In the present study, the participants received the COVID-19 vaccine at the Hokkaido University as the COVID-19 in-house vaccination program. In the University’s in-house vaccination program, a total of 22,000 university members (18 years of age or older) including students, faculty, staff members, and other approved personnel of Hokkaido University and the Otaru University of Commerce were available to receive the COVID-19 vaccine at the Hokkaido University Gym from 17 July to 12 September 2021. The vaccination program was only conducted on weekends, and the participants were required to make a reservation when they prefer to get the vaccine. All health care workers had already received the COVID-19 vaccine at the start of the in-house vaccination program. Therefore, all the participants were considered neither shift workers nor health care workers. Considering the feasibility of this study, we set the sample size to 500. We did not conduct power analysis before collecting samples. A total of 419 participants were enrolled in the study, and 332 of the 419 participants completed demographic questions, including age, sex, date and time of vaccination, past medical history, medication use (antiplatelet drugs, anticoagulant drugs, antihypertensive drugs, diabetes treatment drugs, and hyperlipidemia drugs), allergic history, and mean sleep duration. To measure the level of antibody titers of the SARS-CoV-2 antibody, blood samples were collected at the Hokkaido University Hospital. Blood samples were separated into serum and stored at −80 °C until use. Serum antibody titers against the SARS-CoV-2 neutralizing antibody were assayed using a chemiluminescence enzyme immunoassay and HISCL SARS-CoV-2 S-IgG reagent (Sysmex Corporation, Kobe, Japan). The titer range of detection was 200 to 20,000 BAU/mL; titers under the lower limit of detection were considered 20 BAU/mL. We also measured antibody to SAR-CoV-2 N-IgG (Sysmex Corporation, Kobe, Japan) to identify individuals who have previously been infected by the SARS-CoV-2 virus. Individuals previously infected with COVID-19 were excluded from the analysis. The cut off value of the antibody titer of N-IgG to identify the individuals previously infected was set at 10 BAU/mL.
Statistical Analysis
A general linear regression model was used for assessing the relationship of common logarithm (log10) transformed SARS-CoV-2 antibody titers with days after vaccination and time of day of vaccination adjusting sex, age, allergy, medication, and sleep duration (5-6 h, 7 h, and 8-9 h). The time of day of vaccination were categorized into 2 groups of morning and afternoon group. JMP Pro 16 software (SAS Institute Japan) was used for all the statistical analyses. A confidence level was set to 95%.
Results
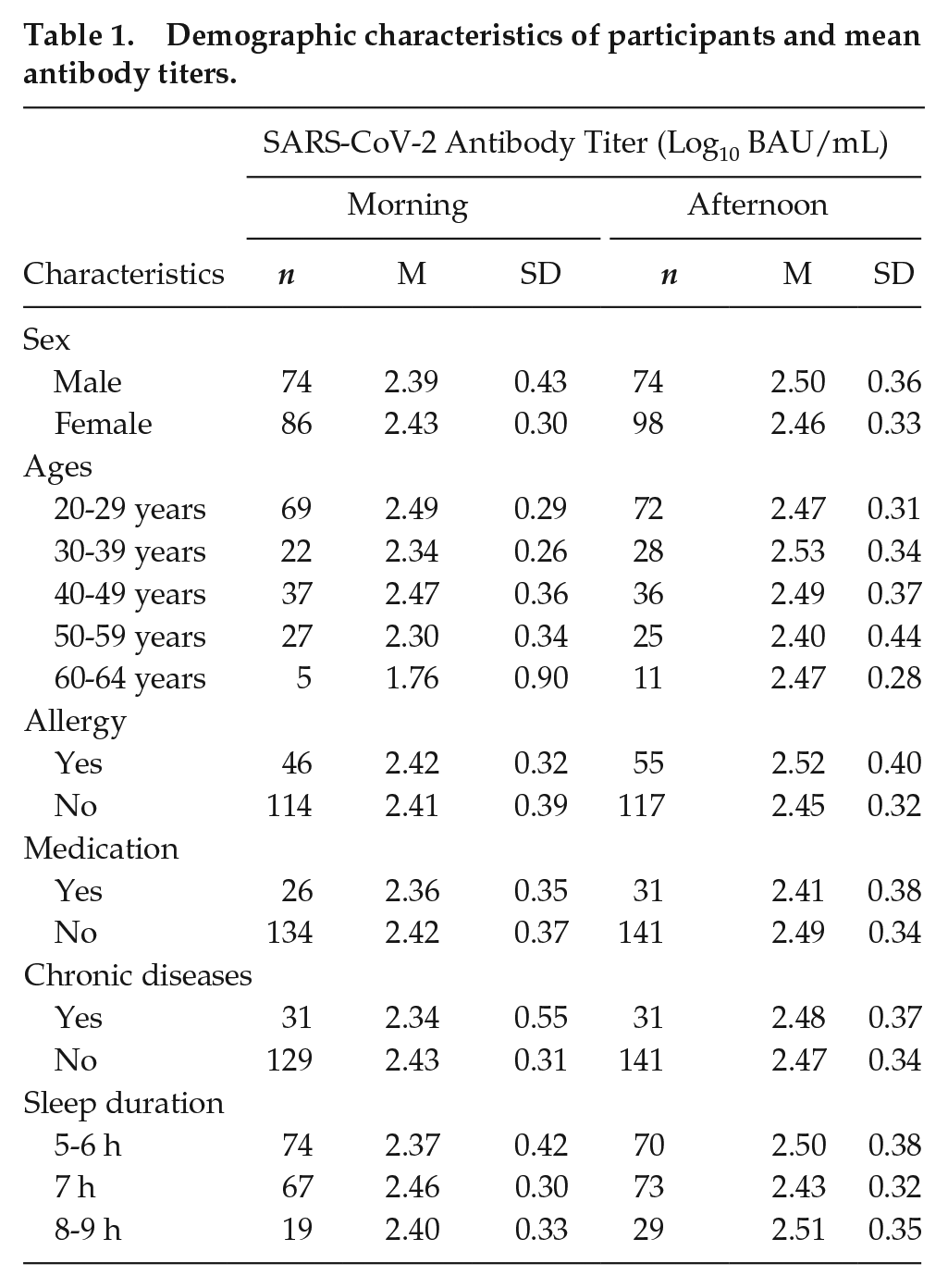
Table 1 summarizes the association between the demographic characteristics (sex, ages, allergy, medication, chronic diseases, sleep duration, and time of day of vaccination) and log10 transformed antibody titers in the study participants (n = 332).
Demographic characteristics of participants and mean antibody titers.
The response of SARS-CoV-2 antibody titers to mRNA-1273 has been reported to increase 2 to 4 weeks after the first dose of vaccination (Widge et al., 2021). In the present study, the effect of post vaccination days on the antibody titers was analyzed by conveniently dividing into 3 groups based on the days after vaccination (13-20, 21-27, and 28-45 days after the first dose of vaccination). Table 2 shows the mean and standard deviation of log10 transformed antibody titers in 13 to 20, 21 to 27, and 28 to 45 days. The antibody titers were similar between the 3 categories of the day after vaccination in 13 to 20, 21 to 27, and 28 to 45 days group, respectively.
Mean antibody titers association between the days after vaccinations and time of day of vaccination.
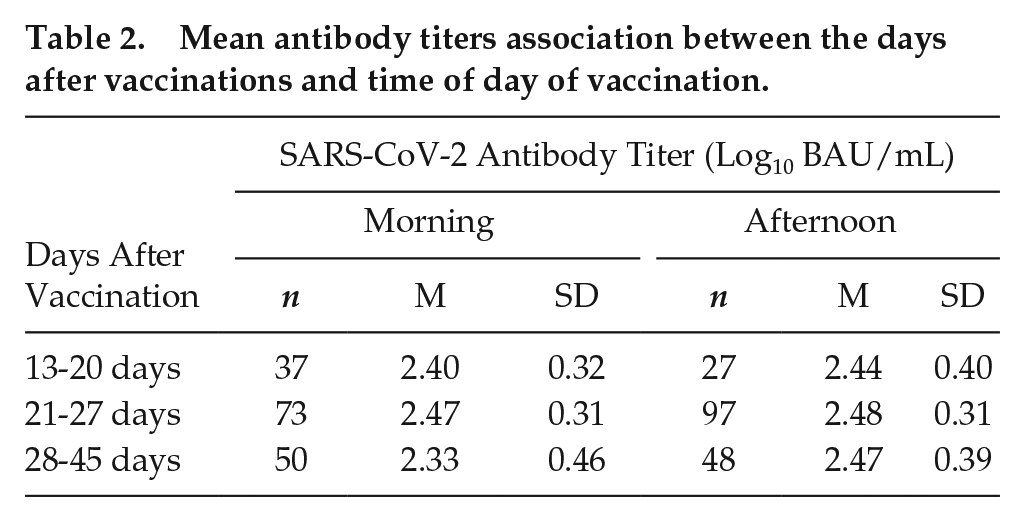
Table 3 summarizes the association between antibody titer and several factors using a general linear regression analysis. Antibody titers were decreased in participants aged 50 to 59 and 60 to 64 years independent of time of day of vaccination (Figure 1a, Table 3). There were no obvious differences for antibody titers between morning and afternoon groups. From a general linear regression model, a significant association was not found between the time of day of vaccination and antibody titers (Figure 1b, Table 3).
The association between antibody titer and several factors using a general linear regression analysis.
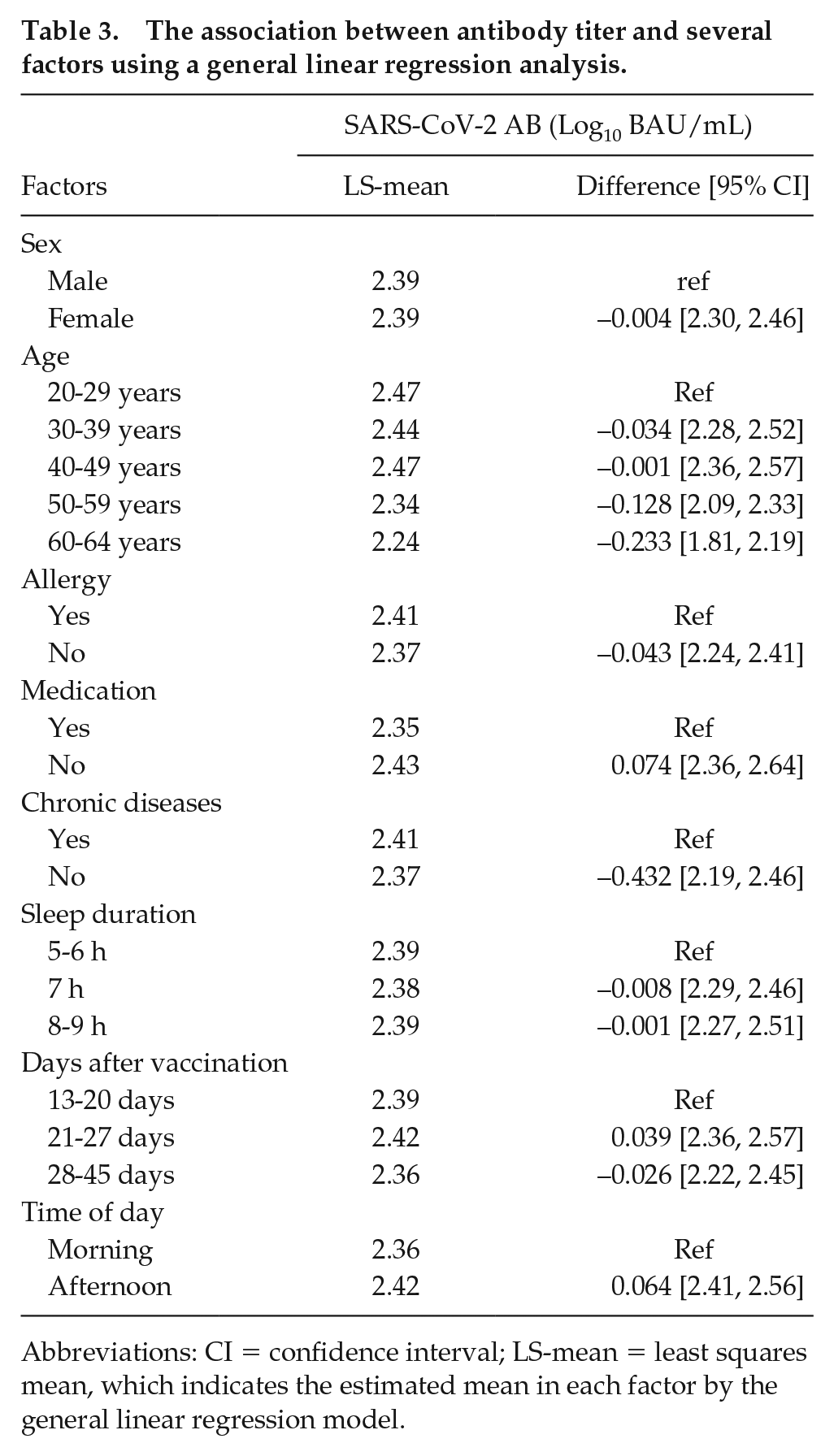
Abbreviations: CI = confidence interval; LS-mean = least squares mean, which indicates the estimated mean in each factor by the general linear regression model.
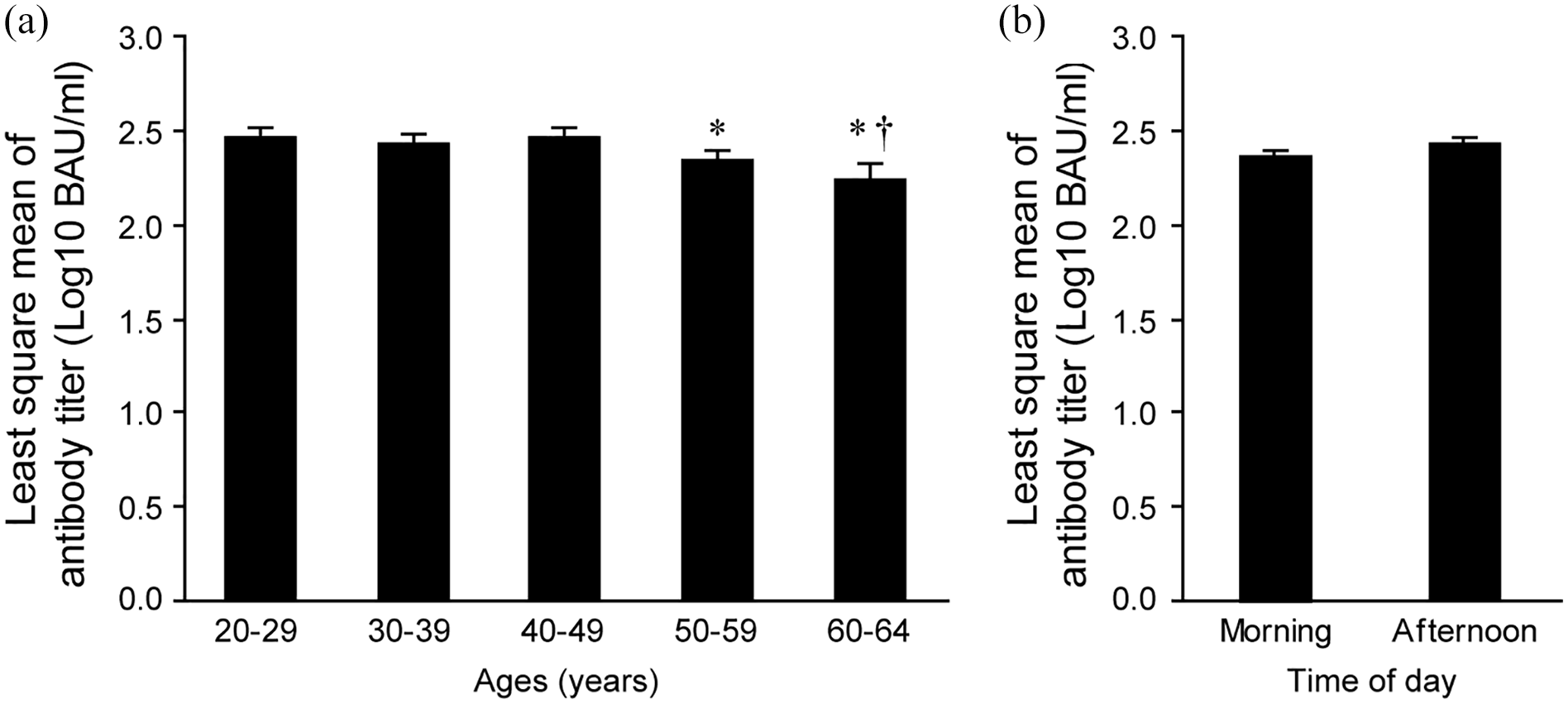
Associations between the least square means of SARS-CoV-2 antibody titers, participant’s ages, and time of day of vaccination. Association between the least square means of log10 transformed SARS-CoV-2 antibody titers between the participant’s ages (a). *p < 0.05 vs. 20 to 29 years. †p < 0.05 vs. 40 to 49 years. Student’s t-test. Association between the least square means of antibody titers and time of day of vaccination in the morning and afternoon (b).
Discussion
The present study examined whether the vaccination time of day alters the SARS-CoV-2 antibody titer after the first dose of the mRNA-1273 vaccine in a sample from the Japanese adult population. A general linear regression analysis revealed no significant association between the time of day of vaccination and the SARS-CoV-2 antibody titer (Figure 1b, Table 3). The antibody titers were decreased in participants aged 50 to 59 years and 60 to 64 years. The decrease in antibody titer in older age groups were consistent with the previous studies (Anastassopoulou et al., 2022; Grupel et al., 2021; Müller et al., 2022).
In the case of BNT162b2, vaccination in the evening (1500-2059 h) was associated with a stronger immune response after vaccination, which resulted in an increase in antibody level in 16- to 29-year-old male and female patients and 30- to 39-year-old male health care workers (Wang et al., 2022). Furthermore, no influence of vaccination time on the antibody response in health care workers who received the ChAdOx1 adenovirus vaccine (AstraZeneca adenovirus vaccine) was demonstrated (Wang et al., 2022). In contrast to the immune response to BNT162b2, the first and second doses of inactivated COVID-19 vaccine in the morning (0900-1100 h) lead to a stronger immune response and increase in antibody titer as compared with the evening vaccination (1500-1700 h) at 28 and 56 days after the vaccination (Zhang et al., 2021). The present results showed no significant association between the time of vaccination and antibody titer (Table 3, Figure 1b), in contrast with the above recent reports (Wang et al., 2022; Zhang et al., 2021). Taken together, the present and recent findings suggest that the effect of the time of day of vaccination on the host immune response may depend on various factors, such as the type of vaccine (mRNA, inactivated, and adenoviral vaccines), sex, age, and number of days after vaccination (Wang et al., 2022). Furthermore, the differential effect of the vaccination time on the host immune response between BNT162b2 and mRNA-1273 might be explained by the large difference in the vaccine dose. BNT162b2 contained 30 μg (0.3 mL) of mRNA, whereas mRNA-1273 contained 100 μg (0.5 mL) of mRNA, which is more than 3 times the amount of mRNA compared with BNT162b2. A recent comparative study in health care workers revealed that higher antibody titers were observed in participants vaccinated with 2 doses of mRNA-1272 than in those vaccinated with BNT162b2 mRNA (Steensels et al., 2021; Khoury et al., 2021). Thus, the higher mRNA content in the mRNA-1273 vaccine compared with the BNT162b2 vaccine might explain the difference in effects regarding the time of day of vaccination between the mRNA-1273 and BNT162b2 mRNA vaccines. Another aspect to be considered is the association between participant race and antibody titer against the COVID-19 vaccine, which is not yet fully understood.
The present study is the first to demonstrate the influence of the time of day of vaccination on the antibody titer in Japanese adults who have received the first dose of mRNA-1273. However, our study has a few limitations which must be considered when interpreting these results in the broader context of translational opportunities within the circadian context. The time of day of vaccination was dependent on the time of day when the participants visited the vaccination site, so that the present study was not a randomized controlled trial. We did not examine other factors, such as participants’ physical activity (Chastin et al., 2021), circadian rhythm (Sengupta et al., 2021), and natural immune system (Netea et al., 2022), which have been associated with variations in the magnitude of response of the host immune system to viral infection. These limitations reduce our ability to conclusively rule out the possibility of circadian influence on antibody response humans. Future studies would also benefit from including other surrogates of immune response to vaccination, in addition to antibody titers. In addition, our data did not examine the effect of the vaccination time on the antibody titer after the second dose of vaccination, which induces a marked increase in the antibody titer (Steensels et al., 2021; Tré-Hardy et al., 2021). Furthermore, our data contained intra- and inter-individual differences in the host immune system. Further studies will be needed to carry out serial blood sample collections or cross-over studies that administer morning and evening vaccinations to the same individuals after performing the sample size estimation by a power analysis. It would be also needed to evaluate the effect of time of day of vaccination not only on the antibody titers but also other immune response markers (Zhang et al., 2021). While the current study does not support restricting vaccination to certain times of day, given the limitations associated with the study size and the non-randomized design and considering the large body of literature supporting clock-gated immune responses, we believe vaccination strategies should include time-of-day variable both in the vaccination and antibody testing steps. Larger randomized studies are needed to answer this question conclusively.
In conclusion, the present study provides evidence that the vaccination time with mRNA-1273 was not associated with the SARS-CoV-2 antibody titer in adult population, suggesting that these results do not support restricting vaccination to a particular time of day. The present findings may be useful in optimizing SARS-CoV-2 vaccination strategies.
Footnotes
Acknowledgements
This work was supported by the Agency for Medical Research and Development (AMED) under grant number JP20ek0210154.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
