Abstract
The COVID-19 pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a global crisis with unprecedented challenges for public health. Vaccinations against SARS-CoV-2 have slowed the incidence of new infections and reduced disease severity. As the time of day of vaccination has been reported to influence host immune responses to multiple pathogens, we quantified the influence of SARS-CoV-2 vaccination time, vaccine type, participant age, sex, and days post-vaccination on anti-Spike antibody responses in health care workers. The magnitude of the anti-Spike antibody response is associated with the time of day of vaccination, vaccine type, participant age, sex, and days post-vaccination. These results may be relevant for optimising SARS-CoV-2 vaccine efficacy.
The circadian clock is an endogenous 24-h clock that regulates many aspects of physiology, including the response to infectious disease and vaccination (Allada and Bass, 2021). A recent report demonstrated significant daytime variation in multiple immune parameters in >300,000 participants in the UK Biobank, highlighting the diurnal nature of innate and adaptive immune responses (Wyse et al., 2021). Human lung diseases frequently show time-of-day variation in symptom severity and respiratory function and the circadian transcriptional activator BMAL1 has been shown to regulate respiratory inflammation (Ehlers et al., 2018; Ince et al., 2019). Influenza A virus infection of circadian-arrhythmic mice is associated with elevated inflammatory responses and a higher viral burden (Edgar et al., 2016; Sengupta et al., 2019). The time of day of influenza vaccination in elderly men affected antibody responses with higher titres noted in the morning (Phillips et al., 2008; Long et al., 2016). An additional influenza vaccination study reported that the time of sample collection rather than vaccination had a more significant effect on antibody responses (Kurupati et al., 2017). We and others have proposed a role for circadian signalling in regulating SARS-CoV-2 host immune responses and COVID-19 severity (Ray and Reddy, 2020; Maidstone et al., 2021; Sengupta et al., 2021). Clearly, it is important to assess whether the time of SARS-CoV-2 vaccination impacts host antibody responses.
In the UK, health care workers were identified as a priority group to receive SARS-CoV-2 vaccine starting in December 2020. At this time, the Alpha B.1.1.7 variant was the dominant circulating strain. As part of this initiative, data were collected on all asymptomatic staff members (Eyre et al., 2021; Lumley et al., 2021) in keeping with enhanced hospital infection prevention and control guidelines issued by the UK Department of Health and Social Care. Anonymised data were obtained from the Infections in Oxfordshire Research Database with Research Ethics Committee approvals (19/SC/0403, ECC5-017 (A)/2009). Peripheral blood samples were collected during December 2020-February 2021 and were tested for anti-Spike (Abbott IgG assay) (Ainsworth et al., 2020) and anti-nucleocapsid (Abbott SARS-CoV-2 IgG anti-nucleocapsid assay) antibody levels. We analysed anti-Spike responses during the 2-10 weeks after vaccination. In this data set, 2190 people contributed one blood sample, 549 contributed two samples, and 45 three or more samples (total of 3425 samples). Participants with evidence of prior SARS-CoV-2 infection (PCR for viral RNA or anti-nucleocapsid antibody), samples with anti-Spike responses < 50 AU, and samples obtained after second vaccination were excluded.
Data from 2784 participants (Table 1) were analysed using linear mixed modelling to investigate the effects of time of vaccination on anti-Spike antibody levels. Variation between participants was modelled with fixed factors of time of day of vaccination (Time 1, 0700-1059 h; Time 2, 1100-1459 h; Time 3, 1500-2159 h) (Suppl. Fig. S1), vaccine type (Pfizer, mRNA bnt162b2 or AstraZeneca, Adenoviral AZD1222), age group (16-29, 30-39, 40-49, or 50-74 years), sex, and the number of days post-vaccination. A B-spline transformation of days post-vaccination was used to model the non-linear pattern of anti-Spike responses (log10 transformed) (Suppl. Fig. S2). This analysis allowed us to estimate the average anti-Spike levels in each participant group at 2 and 6 weeks post-vaccination (Figure 1).
Participant numbers.

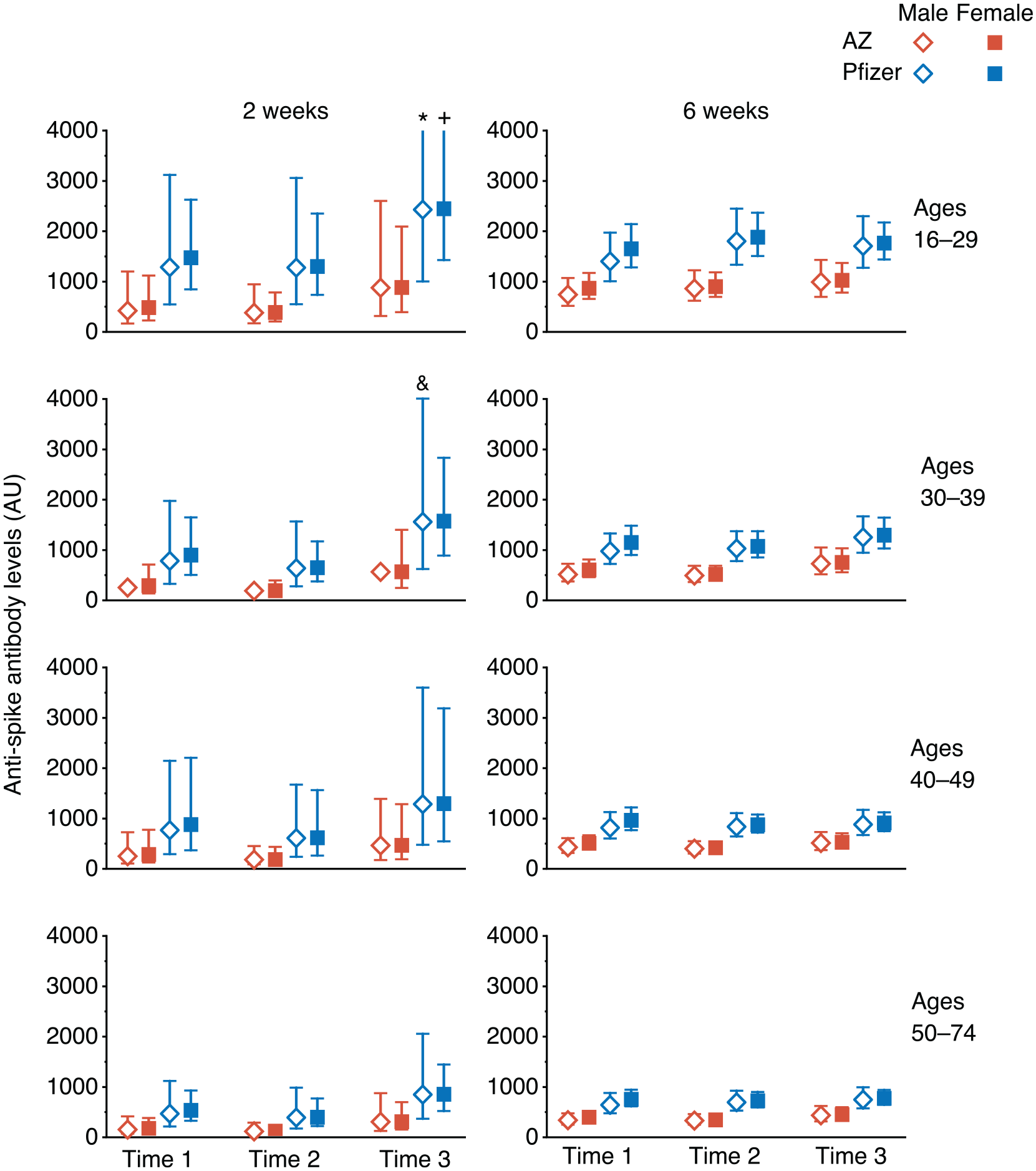
Estimated Anti-Spike antibody levels at 2 and 6 weeks after first SARS-CoV-2 vaccination, partitioned by age, sex, and time of day of vaccination (Time 1, 0700-1059 h; Time 2, 1100-1459 h; Time 3, 1500-2059 h). Mean value (symbol) with 95% confidence values (vertical line). Three confidence intervals extend beyond the Y-axis limits (* = 4275, + = 5996 and & = 4028). Abbreviation: SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2.
Using a linear mixed-model approach, we found that anti-Spike responses were higher in those who were vaccinated later in the day (p = 0.013), in those who received the Pfizer mRNA vaccine (p < 0.0001), in women (p = 0.013), and in younger participants (p < 0.0001) (Table 2). We observed significant interactions between days post-vaccination and vaccine type (p < 0.0001) and age (p = 0.032), but not with vaccine time (p = 0.238). Analysing the data using two time intervals (before or after 1 pm) gave similar results. We did not observe a significant effect of time of day of sample collection (using the same time intervals as for vaccination times) (p = 0.097), and this parameter was not included in the final model; results from the model including sample times are shown in Supplementary Table S1. Sixty-seven samples gave values beneath the cutoff (<50) in the anti-Spike assay and were classified as ‘non-responders’; we found no significant association with the time of day of vaccination for these samples (linear mixed-effects logistic regression, p = .23).
Type III tests of fixed effects from mixed-effects model.
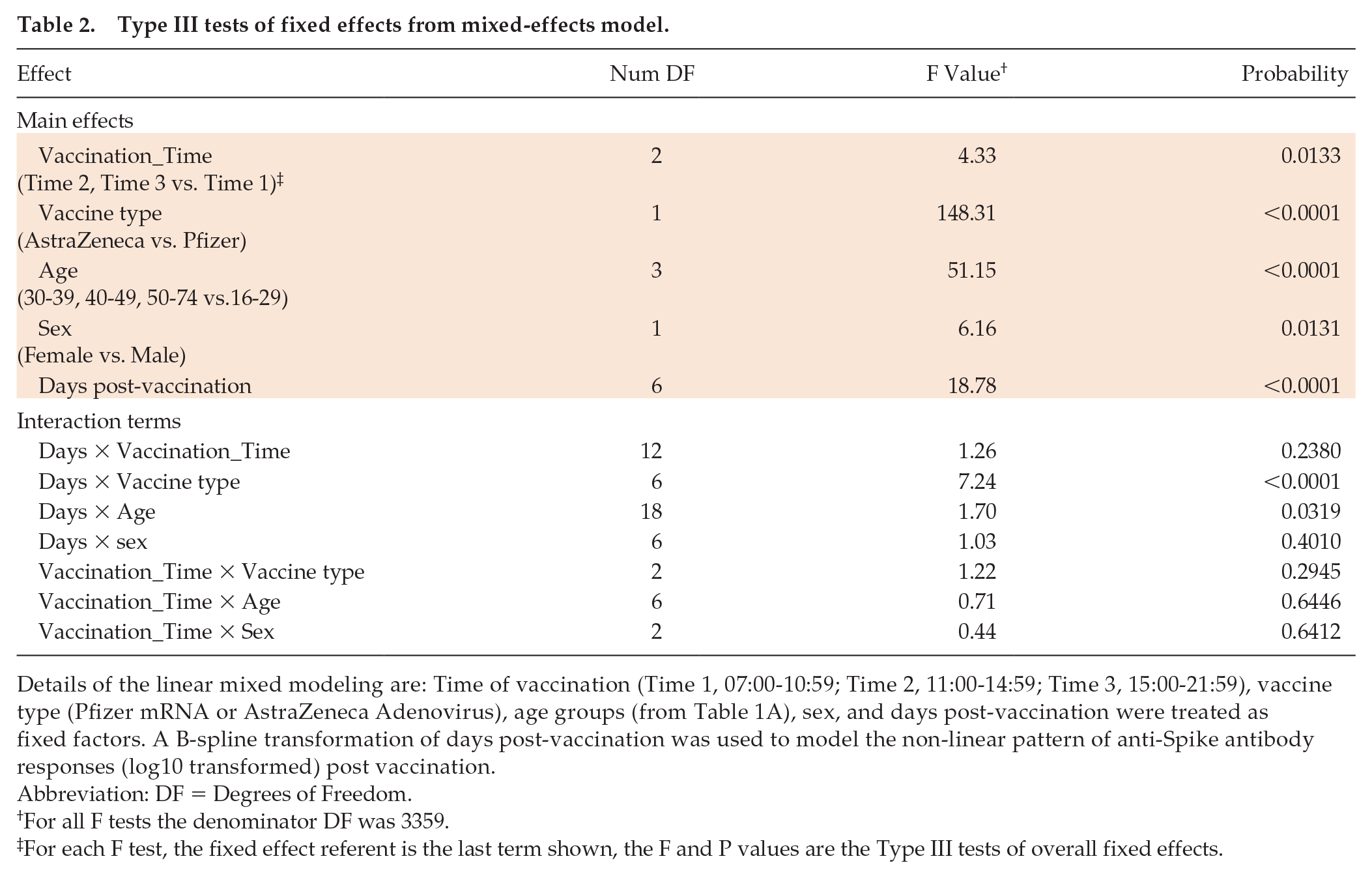
Details of the linear mixed modeling are: Time of vaccination (Time 1, 07:00-10:59; Time 2, 11:00-14:59; Time 3, 15:00-21:59), vaccine type (Pfizer mRNA or AstraZeneca Adenovirus), age groups (from Table 1A), sex, and days post-vaccination were treated as fixed factors. A B-spline transformation of days post-vaccination was used to model the non-linear pattern of anti-Spike antibody responses (log10 transformed) post vaccination.
Abbreviation: DF = Degrees of Freedom.
For all F tests the denominator DF was 3359.
For each F test, the fixed effect referent is the last term shown, the F and P values are the Type III tests of overall fixed effects.
Our analysis of 2784 health care workers reveals a significant effect of the time of vaccination on anti-Spike antibody levels following the administration of two alternative SARS-CoV-2 vaccines (mRNA or Adenovirus based). A recent report studying a small cohort of health care workers immunised with an inactivated SARS-CoV-2 vaccine in the morning (0900-1100 h, n = 33) or afternoon (1500 1700 h, n = 30) showed increased B-cell responses and anti-Spike antibodies in participants vaccinated in the morning (Zhang et al., 2021). This contrasts with our observations and may reflect the use of an inactivated whole virus immunogen that will likely induce polytypic responses to a range of SARS-CoV-2 encoded proteins. Our observation contrasts with earlier studies in elderly men that reported higher anti-influenza titers in the morning (Phillips et al., 2008; Long et al., 2016). This may reflect differences between the cohorts studied, particularly with regard to immune status; we studied seronegative participants whereas responses to influenza vaccination will involve the stimulation of memory responses. Sample collection time in this study showed no significant association with anti-Spike levels, in contrast to previous reports (Kurupati et al., 2017; McNaughton et al., 2021). These data highlight the importance of recording the time of vaccination in clinical and research studies, and highlight the importance of considering time-of-day factors in future study designs that may reduce inter-individual variance and the number of participants needed to obtain statistical significance.
Additional studies are warranted to evaluate the circadian regulation of natural and vaccine-induced SARS-CoV-2 immunity. McNaughton and colleagues reported a diurnal variation in SARS-CoV-2 PCR test results, showing a 2-fold variation in Ct values implying higher viral RNA levels in the afternoon (McNaughton et al., 2021). These data are consistent with our recent study showing a role for the circadian component BMAL1 in regulating SARS-CoV-2 replication (Zhuang et al., 2021) that could influence the induction of host innate and adaptive responses.
It is worth noting that despite the significant differences in anti-Spike levels detected in participants receiving Pfizer mRNA or AstraZeneca Adenoviral vaccines, both show comparable efficacies highlighting the robust nature of the host antibody response. Limitations of this retrospective observational study include: (a) relatively few participants had more than one anti-Spike antibody measurement, limiting our ability to study both longitudinal immune responses and the effect of time of day of sample collection; (b) the health profiles of our health care workers may differ from the general population and no information was available on their medical or medication history, except that they had no prior infection with SARS-CoV-2 and were seronegative; (c) there was limited serological sampling following second vaccination, precluding the analysis of time-of-day effects following a 2-dose schedule; (d) the extent to which anti-Spike levels are a correlate of clinical efficacy is not known; (e) the sleep and shift-work patterns of the participants, that are known to influence vaccine responses (Spiegel et al., 2002; Lange et al., 2003; Prather et al., 2021), were not available; and (e) our cohort does not include children or high-risk groups, such as the elderly or immunocompromised. We recommend future studies address these limitations when documenting natural and vaccine-induced SARS-CoV-2 immune responses.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304211059315 – Supplemental material for Time of Day of Vaccination Affects SARS-CoV-2 Antibody Responses in an Observational Study of Health Care Workers
Supplemental material, sj-docx-1-jbr-10.1177_07487304211059315 for Time of Day of Vaccination Affects SARS-CoV-2 Antibody Responses in an Observational Study of Health Care Workers by Wei Wang, Peter Balfe, David W. Eyre, Sheila F. Lumley, Denise O’Donnell, Fiona Warren, Derrick W. Crook, Katie Jeffery, Philippa C. Matthews, Elizabeth B. Klerman and Jane A. McKeating in Journal of Biological Rhythms
Supplemental Material
sj-docx-2-jbr-10.1177_07487304211059315 – Supplemental material for Time of Day of Vaccination Affects SARS-CoV-2 Antibody Responses in an Observational Study of Health Care Workers
Supplemental material, sj-docx-2-jbr-10.1177_07487304211059315 for Time of Day of Vaccination Affects SARS-CoV-2 Antibody Responses in an Observational Study of Health Care Workers by Wei Wang, Peter Balfe, David W. Eyre, Sheila F. Lumley, Denise O’Donnell, Fiona Warren, Derrick W. Crook, Katie Jeffery, Philippa C. Matthews, Elizabeth B. Klerman and Jane A. McKeating in Journal of Biological Rhythms
Supplemental Material
sj-pdf-3-jbr-10.1177_07487304211059315 – Supplemental material for Time of Day of Vaccination Affects SARS-CoV-2 Antibody Responses in an Observational Study of Health Care Workers
Supplemental material, sj-pdf-3-jbr-10.1177_07487304211059315 for Time of Day of Vaccination Affects SARS-CoV-2 Antibody Responses in an Observational Study of Health Care Workers by Wei Wang, Peter Balfe, David W. Eyre, Sheila F. Lumley, Denise O’Donnell, Fiona Warren, Derrick W. Crook, Katie Jeffery, Philippa C. Matthews, Elizabeth B. Klerman and Jane A. McKeating in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank Helene Borrmann, Xiaodong Zhuang, and Tanya Wilson for early discussions on this project, and Tess Lambe and Merryn Voysey for their constructive comments on the article. We also thank all OUH staff who participated in the staff testing programme and the staff and medical students who ran the programme. This work uses data provided by health care workers and collected by the UK’s National Health Service as part of their care and support. We thank all the people of Oxfordshire who contributed to the Infections in Oxfordshire Research Database. Research Database Team: L Butcher, H Boseley, C Crichton, O Freeman, J Gearing (community), R Harrington, M Landray, A Pal, TEA Peto, TP Quan, J Robinson (community), J Sellors, B Shine, AS Walker, D Waller. Patient and Public Panel: G Blower, C Mancey, P McLoughlin, and B Nichols.
EBK is funded by NIH K24-HL105664, P01-AG009975, and R01-HL128538. JAM is funded by a Wellcome Investigator Award 200838/Z/16/Z, UK Medical Research Council project grant MR/R022011/1, and Chinese Academy of Medical Sciences Innovation Fund for Medical Science, China (grant number: 2018-I2M-2-002). WW is funded by NCATS Harvard Clinical and Translational Science Center grant 5UL1TR002541-02. DWE is a Robertson Foundation Fellow. DCA is funded by the NIHR Oxford Biomedical Research Centre. The report presents independent research. The views expressed in this publication are those of the authors and not necessarily those of the NHS, NIHR, or the UK Department of Health. PCM is funded by a Wellcome intermediate fellowship grant Ref 110110/Z/15/Z.
Conflict of Interest Statement
WW has a consultancy for the National Sleep Foundation. PB has no relevant disclosures. DWE declares lecture fees from Gilead, outside the submitted work. EBK declares travel support from Gordon Research Conference, Sleep Research Society, Santa Fe institute, DGSM (German Sleep Society); consultancy for Circadian Therapeutics, National Sleep Foundation, Puerto Rico Science Technology Trust, Sanofi-Genzyme; partner owns Chronsulting. JAM has no relevant disclosures.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
