Abstract
To examine whether immunization time affects the immune responses elicited by the BNT162b2 COVID-19 vaccine, we investigated the possible association between total SARS-CoV-2 spike protein receptor binding domain (TAbs-RBD) and neutralizing (NAbs-RBD) antibodies with vaccination time. A cohort of 468 healthcare workers (mean age [±SD]: 48 [±13] years), were included in the study. One month after the second dose, healthcare workers who were vaccinated between 1500-2200 h had higher TAbs-RBD compared to 0700-1100 h and 1100-1500 h (p = 0.006). One month after the third dose, healthcare workers who were vaccinated between 0700-1100 h and 1500-2200 h had significantly higher TAbs-RBD compared to 1100-1500 h (p = 0.034). However, no association of NAbs-RBD with vaccination time was detected after each of the 3 doses (p > 0.4). Despite the possible effect of BNT162b2 vaccination time in TAbs-RBD levels, possibly due to rhythmic expression of clock genes, neutralizing activity was not associated with vaccination time and, therefore, further investigation is required.
COVID-19 vaccines have been demonstrated to protect against critical COVID-19 infection and substantially contribute to reduction of hospitalization rates (Dagan et al., 2021). In addition to various clinical and epidemiological parameters, the time of day of immunization administration may impact the magnitude of the immune response induced by vaccination (Borrmann et al., 2021).
Immune response after vaccination is commonly evaluated by measurement of antibodies. Total (TAbs-RBD) antibodies bound to receptor binding domain (RBD) antigen and neutralizing (NAbs-RBD) antibodies directly inhibit the SARS-CoV-2 spike protein via the RBD region. NAbs-RBD inhibit binding of the spike protein to ACE2 receptor on the surface of the human cells (Dogan et al., 2021), which prevents viral entry and is therefore more effective against SARS-CoV-2 infection.
Recently published studies by Wang et al. (2022) and Zhang et al. (2021) examined the effect of time of day of SARS-CoV-2 vaccination on antibody levels. Both studies reported a diurnal effect in the immune responses after SARS-CoV-2 vaccines, nevertheless in different directions. Wang et al. reported that BNT162b2 and Adenoviral AZD1222 COVID-19 vaccine recipients had significantly higher Total Receptor Binding Domain antibodies (TAbs-RBD) when receiving first dose of BNT162b2 vaccine between 1500-2159 h probably due to circadian rhythm effects (Wang et al., 2022). The Zhang et al. (2021) study evaluated the Neutralizing Receptor Binding Domain antibodies (NAbs-RBD), as well as B and T cell responses after first and second dose of inactivated vaccine (Sinopharm). They found higher NAbs-RBD responses as well as higher activation of B and T cells in participants who were vaccinated in the morning (0900-1100 h) after both doses (Zhang et al., 2021).
The aim of this study was to investigate the association of TAbs-RBD and NAbs-RBD levels with the time of day of BNT162b2 COVID-19 immunization at different time points after the first, second, and third vaccine doses.
Materials and Methods
We prospectively measured TAbs-RBD (U/ml) and NAbs-RBD levels (%) of the SARS-CoV-2 spike protein in a cohort of 468 healthcare workers who received the first 2 doses of the BNT162b2 COVID-19 vaccine in January 2021 and the third dose within the period October-December 2021.
Each participant completed a form containing demographic, epidemiological data and different information regarding the COVID-19 vaccination, like the time of vaccination. Hours (h) are expressed in military time. The immunization daytime was divided for analysis into 3 time periods: 0700-1100 h, 1100-1500 h and 1500-2200 h.
Serum samples were collected between 0800-1600 h at 5 different time points: 21 days after the first dose; 1, 4, 8 months after the second dose; and 1 month after the third dose of the BNT162b2 COVID-19 vaccine. For the detection of TAbs-RBD, the Elecsys Anti-SARS-CoV-2 S reagent (Roche Diagnostics, Basel, Switzerland) was used and values ≥0.8 U/ml were classified as positive. Anti-RBD neutralization titers (NAbs-RBD) were measured with the ELISA cPass SARS-CoV-2 neutralization antibody detection kit (GenScript Biotech Corporation, Piscataway, New Jersey, USA; FDA-approved) and, according to the manufacturer, ≥30% were regarded positive. Participants who were initially tested positive for the SARS-CoV-2 nucleocapsid antigen or who were tested positive in the subsequent sampling (Elecsys Anti-SARS-CoV-2; Roche Diagnostics Basel, Switzerland) method were excluded from the analysis because they were positive for natural COVID-19 infection.
The study protocol was approved by the Scientific and Bioethics Committee of “Aghia Sophia” Children’s Hospital (No. 2794) and was in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants.
Statistical analysis
For descriptive statistics, mean and standard deviation (SD) were used for parametric variables or median and interquartile range (IQR) for variables with non-normal distribution. One-way analysis of variance (ANOVA) two-way ANOVA and 3 × 2 mixed ANOVA or Kruskal-Wallis test due to normality violation were used. Statistical significance level was set at p-value less than 0.05. Statistical analysis was performed with SPSS version 26.0 (IBM Corp. Armonk, NY, USA).
Results
A total of 468 healthcare workers with a mean age (±SD) of 48.33 (±12.98) years, 361 (77.14%) women, met the inclusion criteria. Differences in TAbs-RBD antibodies after the first, second, and third dose of BNT162b2 vaccine with respect to the time of day of vaccination are presented in Table 1. One month after the second dose, healthcare workers who were vaccinated between 1500-2200 h had significantly higher TAbs-RBD compared to 0700-1100 h and 1100-1500 h (median TAbs-RBD [IQR]: 1,744 U/ml [1,152] vs 1,216 U/ml [1,193] vs 1,287 U/ml [1,559], respectively, p = 0.006). One month after the third dose, healthcare workers vaccinated between 1100-1500 h had significantly lower TAbs-RBD compared to 0700-1100 h and 1500-2200 h (median TAbs-RBD [IQR]: 13,585 U/ml [13,267] vs 17,885 U/ml [13,769] vs 20,387 U/ml [14,155], respectively, [p = 0.048]). No significant differences were detected regarding association of TAbs-RBD with time of day of vaccination after the first dose (p = 0.718), 4 months (p = 0.382), and 8 months after the second dose (p = 0.709).
Differences in levels of total SARS-CoV-2 spike protein receptor binding domain (TAbs-RBD) antibodies (U/ml) after the 3 doses of BNT162b2 vaccine regarding the time of day of immunization.
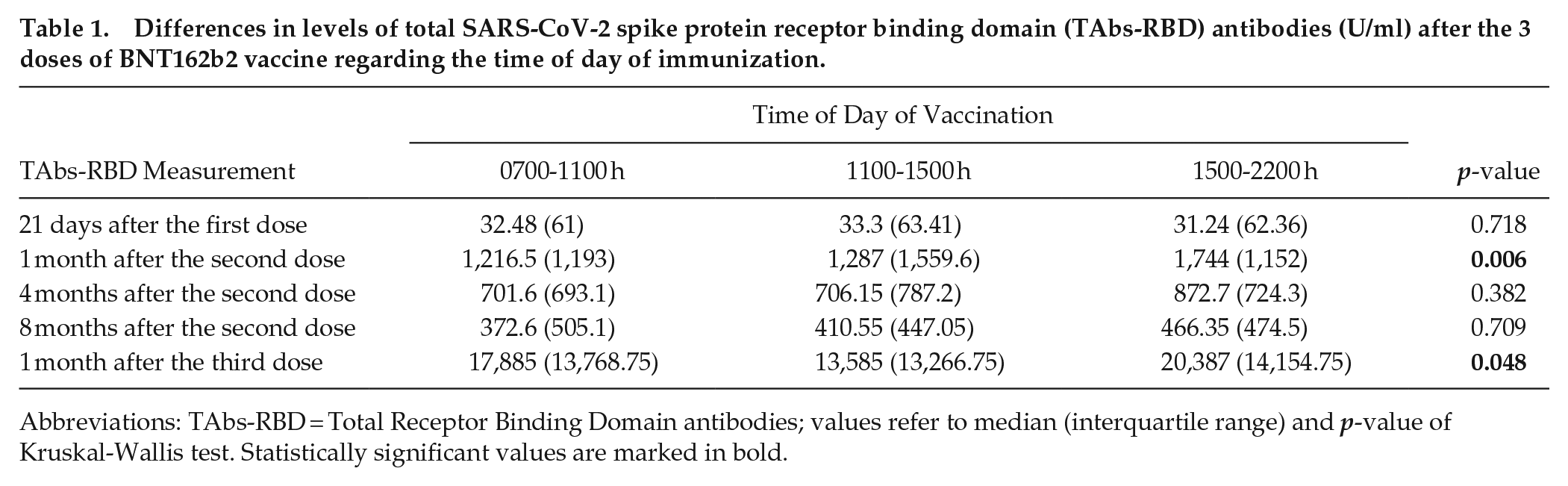
Abbreviations: TAbs-RBD = Total Receptor Binding Domain antibodies; values refer to median (interquartile range) and p-value of Kruskal-Wallis test. Statistically significant values are marked in bold.
The two-way ANOVA analysis revealed a nonsignificant contribution of age in association of time of vaccination with TAbs-RBD 1 month after the second (p = 0.875) or after the third dose (p = 0.410). In a mixed model analysis involving TAbs-RBD and time of vaccination with age, no significant differences were detected 1, 4, and 8 months after the second dose (p = 0.779).
Regarding the association of time of immunization with NAbs-RBD (%), no statistically significant differences were detected in each time point after the 3 vaccine doses (p > 0.11)
Differences in SARS-CoV-2 spike protein receptor binding domain (NAbs-RBD) neutralizing activity (%) antibodies after the 3 doses of BNT162b2 vaccine regarding the time of day of immunization.
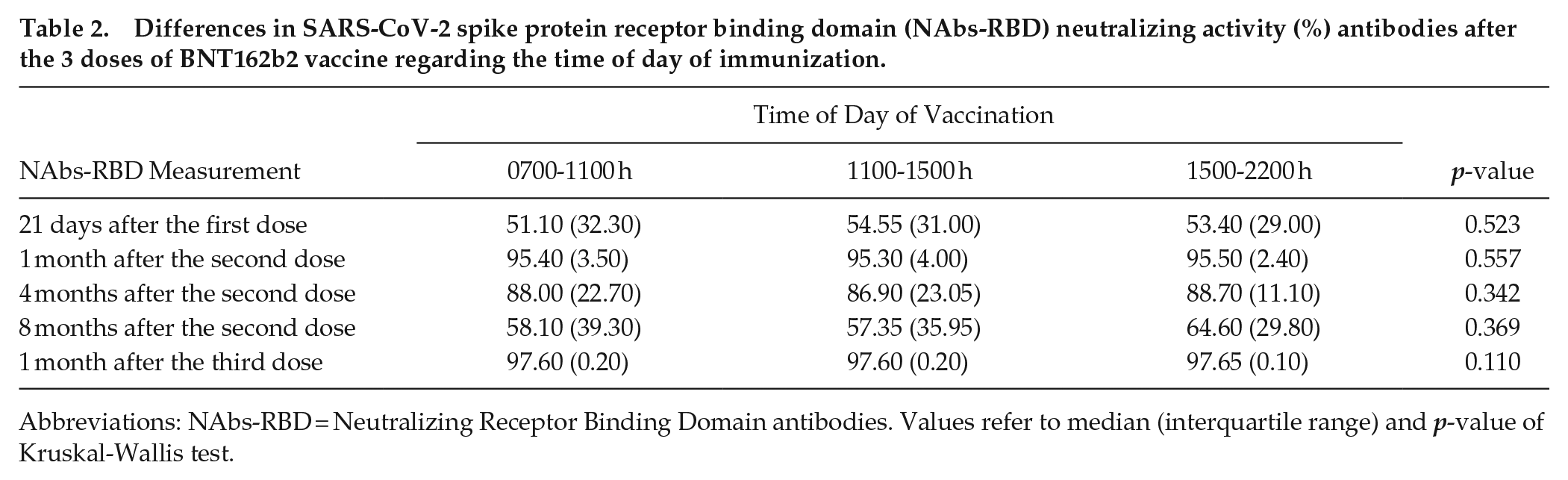
Abbreviations: NAbs-RBD = Neutralizing Receptor Binding Domain antibodies. Values refer to median (interquartile range) and p-value of Kruskal-Wallis test.
Discussion
Circadian rhythms affect various aspects of mammalian physiology, including the immune system and response to pathogens (Wyse et al., 2021). In this study, immunization with the BNT162b2 between 1500-2200 h elicited significantly higher TAbs-RBD levels 1 month after the second and third doses. However, no significant differences were detected after the first dose, or 4 and 8 months after the second dose. To the best of our knowledge, this is the first study investigating the role of time of immunization with mRNA vaccine with TAbs-RBD and NAbs-RBD 4 and 8 months after the second and 1 month after the third dose. Based on these results, we hypothesize that time-of-day effect could elicit higher TAbs-RBD levels, early after immunization, however, this effect could be less significant as antibodies levels decrease over time.
In a recently published study by Wang et al., an effect of the time of day of vaccination on the levels of TAbs-RBD levels after the first dose of COVID-19 vaccine (either BNT162b2 or Adenoviral AZD1222) was reported. More specifically participants who were vaccinated later in the day (after 1500 h) had higher immune responses compared with those who were vaccinated earlier. However, they do not measure NAbs-RBD and they did not report kinetics of antibodies after second and third dose of vaccine.
Another recent report studying a small cohort of young healthcare workers immunized with an inactivated SARS-CoV-2 vaccine (BBIBP-CorV, Sinopharm, Beijing) in the morning (0900-1100 h, n = 33) or afternoon (1500-1700 h, n = 30) showed increased B-cell responses and NAbs-RBD in participants vaccinated in the morning (Zhang et al., 2021). In our study we did not detect any association of time of vaccination with TAbs-RBD after the first dose but only 1 month after the second and third dose. However, the NAbs-RBD (%) was not statistically different after any of the 3 doses regardless of vaccination time.
These reported differences in immune responses among the previously reported studies could be possibly attributed to different types of SARS-CoV-2 vaccines and different laboratory methods for the detection of antibody responses. Zhang et al. (2021) studied a different inactivated SARS-CoV-2 vaccine which precludes direct comparisons. The study of Wang et al. (2022) uses 2 different vaccines (Pfizer mRNA or AstraZeneca adenoviral vaccine); however, they do not report NAbs-RBD or kinetics of antibodies after second and third vaccine dose. For these reasons the findings from the 3 studies are not directly comparable.
Limited data have been published on the association of immune responses with the time of day of vaccination. In a study involving preterm infants receiving a hexavalent vaccine (diphtheria, tetanus, pertussis, poliomyelitis, type B Haemophilus influenzae, and hepatitis B), it was found that children vaccinated between 1900-2200 h had better antibody responses compared to those vaccinated in the morning hours (Gottlob et al., 2019). On the contrary, BCG recipients vaccinated in the morning demonstrated stronger cellular immune responses and cytokine secretion than those in the evening (de Bree et al., 2020). These contradictory results could possibly be attributed to the different vaccine antigen types and response patterns.
The molecular mechanisms behind diurnal fluctuations in antibody levels are complex and many physiological processes, such as regulation by the endocrine system, are of importance (Borrmann et al., 2021). It is well established that circadian rhythms on a cellular level are regulated by the circadian transcription factors (such as BMAL1) which regulate rhythmic expression of clock controlled genes. The contribution of clock genes in T-cell diurnal proliferation as response to antigens has been demonstrated in mice with deletions in the Bmal1 gene of CD8T-cells and loss of circadian rhythm (Nobis et al., 2019). BMAL1 is a key clock gene with not only diurnal but also seasonal expression (Duguay and Cermakian, 2009).
Glucocorticoids are characterized by both suppressing and boosting effects on humoral and cellular immune responses by regulating certain lymphocyte subtypes redistribution and modifying cytokine expression pathways (Shimba et al., 2021). Cortisol could have a diurnal suppressing effect on T-cell expression, as reactive CD4+ and CD8+ T cell counts reach their peak levels at night possibly impacting adaptive immune responses, including antibodies (Kirsch et al., 2012). In addition to cortisol, increased catecholamine levels, such as epinephrine, are directly associated with enhanced CD8+ T cell levels via increased expression of beta-adrenergic and fractalkine receptors (CX3CR1) (Dimitrov et al., 2009). These physiologic mechanisms could possible explain higher immune response during late afternoon and night when cortisol levels are lower.
Other parameters that could affect hormonal and immune responses and have an impact in studies associating the time of immunization with the immune response are the age of the participants, the immune status, history of underlying diseases, or medications. In a previously published study from our group immune responses after BNT162b2 vaccine were negatively associated with older age, immunosuppression, or smoking (Michos et al., 2021).
Findings of this study indicate that, irrelevant of the circadian variation of TAbs-RBD antibodies, NAbs-RBD (%) are not associated with time of day of vaccination. Thus, at least in the laboratory functionality of antibodies, there is not a meaningful difference. Further studies associating clinical efficacy of immunization with diurnal variations of antibody responses after SARS-CoV-2 immunization will further elucidate these conflicting results from the previous studies. The examination of different vaccines using different laboratory methods makes it difficult to directly compare results and is something that should be considered in future study designs.
A limitation of this study was that the majority of the samples (95% of healthcare workers) were collected between 0800-1100 h and therefore the impact of sampling time in immune responses elicited by vaccination could not be evaluated. Based on the recently reported results by Wang et al. (2022), sample collection time does not significantly affect immune responses in seropositive and seronegative vaccines.
Optimization of COVID-19 vaccination is crucial for pandemic restriction. The hypothesis of a modified immune response after vaccination, depending on the time of day of vaccination, needs further investigation as the differences presented have not been associated with clinical outcomes. Future studies with large cohorts where time of day immunization can be associated with other epidemiological parameters are necessary to inform about the interplay of vaccination time and disease outcomes. Immunological factors which are associated with circadian rhythmicity like cellular immune responses and clinical outcomes such as symptomatic disease or severe clinical presentation shall be included in such analysis to provide solid evidence if this variability could have real clinical impact.
