Abstract
Biological rhythms that are mediated by exogenous factors, such as light and temperature, drive the physiology of organisms and affect processes ranging from cellular to population levels. For elasmobranchs (i.e. sharks, rays, and skates), studies documenting diel activity and movement patterns indicate that many species are crepuscular or nocturnal in nature. However, few studies have investigated the rhythmicity of elasmobranch physiology to understand the mechanisms underpinning these distinct patterns. Here, we assess diel patterns of metabolic rates in a small meso-predator, the epaulette shark (Hemiscyllium ocellatum), across ecologically relevant temperatures and upon acutely removing photoperiod cues. This species possibly demonstrates behavioral sleep during daytime hours, which is supported herein by low metabolic rates during the day and a 1.7-fold increase in metabolic rates at night. From spring to summer seasons, where average average water temperature temperatures for this species range 24.5 to 28.5 °C, time of day, and not temperature, had the strongest influence on metabolic rate. These results indicate that this species, and perhaps other similar species from tropical and coastal environments, may have physiological mechanisms in place to maintain metabolic rate on a seasonal time scale regardless of temperature fluctuations that are relevant to their native habitats.
The biological clock regulation of endogenous circadian rhythms is fundamental to physiological systems across eukaryotes (Dunlap and Loros, 2016). It is well established that exogenous light plays a large role in regulating these rhythms, which affect all levels of biological organization from cellular processes to whole-organism and population behaviors (Reebs, 2002; Foulkes et al., 2016). However, the environment also provides other exogenous abiotic signals, which can also be cyclical. These “zeitgebers” or time givers may also play a role in regulating endogenous rhythms (Foulkes et al., 2016). For aquatic organisms such as fishes, cyclical patterns in water temperatures are often related to light, where water heats and cools across the day and night hours, respectively, offering another influence on endogenous rhythms (López-Olmeda et al., 2009; Sánchez-Vázquez et al., 2019). Indeed, for various physiological traits such as metabolic rate (i.e. the sum of all reactions occurring in the body), both light and water temperature are known to be influential (López-Olmeda et al., 2009). For example, the metabolic rate of diurnal animals is typically higher during the day than at night, and the opposite is true for nocturnal animals (Kim et al., 1997; Nixon and Gruber, 1988). Furthermore, there is a direct relationship between water temperature and physiological rate functions in nearly all aquatic ectotherms (Angilletta, 2009); however, our understanding of these 2 exogenous cues and their relative influence at the whole-organism physiological level is understudied for nocturnal-pattern fishes.
Of the aquatic ectotherms, the elasmobranchs (i.e. sharks, rays, and skates) host an array of species that are crepuscular or nocturnal with diel periods of swimming activity (Metcalfe and Butler, 1984; Kneebone et al., 2018; Kadar et al., 2019; Kelly et al., 2020, 2021; Byrnes et al., 2021). However, the ways in which cyclical biotic (e.g. predator-prey interactions) and abiotic (e.g. temperature, dissolved oxygen, salinity, light, tidal stage) factors are reflected in their physiology are unclear and likely complex. For example, research on 3 species of rays (Glaucostegus typus, Himantura australis, and Pateobatis fai) indicated that these meso-predators remain in very shallow areas with sub-optimally warm temperatures to reduce interactions with potential predators (Vaudo and Heithaus, 2013). Lemon shark (Negaprion acutidens) diel activity patterns have been found more strongly influenced by light patterns than temperature or body size (Byrnes et al., 2021). Indeed, temperature may be the master factor governing most physiological pathways, and this may be via its influence on metabolism (Angilletta, 2009). But in an ecological context, it may be challenging to tease apart other factors that interact with temperature, such as time of day, availability of a heterogeneous environment to facilitate thermoregulatory behaviors (Nay et al., 2020), and predation risk (Vaudo and Heithaus, 2013).
Within elasmobranchs, there are 2 main respiratory modes, ram-ventilating, where sharks must continuously swim to pass water over their gills, and buccal pumping, in which demersal sharks, skates, and rays use buccal movements to suction water into their mouths and across their gills for oxygenation (Butler and Metcalfe, 1988). The type of respiratory mode used is innately linked to activity patterns, where many ram-ventilating species change the magnitude of their activity in relation to the time of day but always maintain some level of activity for respiratory purposes (Papastamatiou et al., 2018; Shipley et al., 2018). In contrast, buccal pumping species can remain motionless and potentially use a form of sleep during the day and increase activity at night (Kadar et al., 2019; Kelly et al., 2020, 2021). Theoretically, if activity levels increase at night, then metabolic rate should also increase due to the increase in oxygen demand (Whitney et al., 2007). However, only a few studies on elasmobranchs have reported oxygen uptake rates over a 24-h period or between day and night (Nixon and Gruber, 1988; Hove and Moss, 1997; Whitney et al., 2007; Kelly et al., 2022) to clearly demonstrate a relationship between time of day and metabolic rate.
The first aim of this study was to characterize diel patterns in metabolism in a tropical benthic elasmobranch species. The second aim was to determine how temperature and photoperiod influence these diel patterns in metabolism. We used the well-studied epaulette shark (Hemiscyllium ocellatum), which is a species known for its physiological tolerance to challenging environmental conditions and one that is amenable to captivity (Wise et al., 1998; Heinrich et al., 2014, 2016). Moreover, this species can be found on tidally-isolated, shallow reef flats where a 3-4 °C temperature cycle – influenced by tides and time of day – regularly occurs over a 24-h period (Nay et al., 2020). We used intermittent-flow respirometry chambers to measure oxygen uptake rates as a proxy for metabolism for each shark. Furthermore, we implemented constant and diel temperature cycles like those experienced by this species during spring and summer. Finally, we acutely removed photoperiod to examine the master factor influencing changes in metabolic rates in this species.
Materials and Methods
Ethics
All experimental protocols in this study were assessed and approved by the James Cook University Animal Ethics Committee (protocol A2655). Collections were conducted under the appropriate Great Barrier Reef Marine Park Authority (GBRMPA #G19/43380.1) and Queensland Fisheries (#200891) permits.
Animal Collection and Husbandry
Nine adult epaulette sharks, including 4 females (69-83 cm total length [TL]) and 5 males (83-98 cm TL) were collected and used for experimentation. Maturity was assessed based on the 55 cm TL size at maturity for both sexes reported by Heupel et al. (1999). Three females were hand-collected with dip nets in shallow water from Magnetic Island (n = 1, -19.129041 S, 146.877586 E) and Balgal Beach, QLD, Australia (n = 2, -19.021387 S, 146.418124 E) in February and March 2020, respectively. Sharks were transported back to the Marine and Aquaculture Research Facility Unit at James Cook University (Douglas, QLD, Australia) within 2 h of capture in 50 L of clean, continuously aerated seawater. An additional 5 mature males and 1 female were sourced from Cairns Marine in September 2020 (Cairns, QLD, Australia).
Sharks were habituated to captivity for at least 3 months prior to experimentation. During habituation, sharks were maintained in same-sex pairs (with 1 group of 3 males) in 5 indoor 1000 L round aquaria filled to ~850 L, but connected to a shared 5500 L-reservoir with a heater, protein skimmer, bio-filtration, and UV-sterilization. Water quality parameters (pH, nitrites, nitrates, ammonia) were monitored daily for the first 3 months of introduction to the system and were subsequently recorded biweekly, given that water quality was stable (Supplement Table S1). To ensure reservoir and aquaria temperatures were comparable, temperature was monitored within the external reservoir using a sensor that controlled the heater/chiller system and individual HOBO pendant loggers (Onset, USA) attached to the standpipe of each aquarium. Ambient day time lighting in the laboratory consisted of 4 ceiling mounted light-emitting diode (LED) (35 watt, 3600 lumens, 1210 x 115 x 75 mm LWH) panels evenly distributed across the room, and the light: dark (L:D) cycle was 12:12 h during the 3-month habituation and subsequent experimentation. Each aquarium had one large air stone, a lid constructed of 30% light blocking shade cloth to prevent jumping, and a 30 cm diameter by 70 cm long PVC tube was provided as shelter for each shark. All sharks in the study were fed 2% of their body mass,
Respirometry
To estimate the routine metabolic rate (RMR) of sharks, oxygen uptake rates (ṀO2) of each shark were measured using intermittent-flow, static respirometry over a 24-h period. Each respirometry set-up was comprised of 3 opaque PVC chambers with baffled ends to allow even water flow and a 15 cm by 8 cm viewing window allowing ambient light into the chamber. Two were small chambers (15 cm in diameter and 82 cm long, 15.5 L total volume of the chamber, recirculating pump, and external loop; 10.1-16.3 respirometry setup: organism volume), and 1 was a larger chamber (23.5 cm in diameter and 101.5 cm long, 44.6 L total volume of the chamber, recirculating pump, and external loop; 22.2-27.9 respirometry setup: organism volume) to accommodate the range of body sizes. Preliminary trials indicated that the chamber size precluded significant movement of the animals, and no differences in metabolic rate estimates were detected due to chamber size. For each trial, the appropriate respirometry chamber was selected and submerged in 850 L aquaria within the study system to ensure identical water quality between holding conditions and trial conditions. Each chamber consisted of a flush pump (small chambers: 1200 L h-1; large chamber: 2400 L h-1) that pumped water from the water bath and one recirculating pump (small chambers: 1200 L h-1; large chamber: 2400 L h-1) that circulated water through an external loop for proper homogeneous mixing within the chamber. Oxygen levels were measured every 2 sec using an OXROB3 fiber optic probe inserted approximately 5 cm into the chamber proper via the overflow outlet (Bouyoucos et al., 2020a) connected to a Firesting Optical Oxygen Meter (Pyroscience GmbH, Aachen, Germany).
Sharks were fasted 48 h prior to and throughout trials to ensure metabolic rates were estimated while the sharks were in a post-absorptive state (Heinrich et al., 2014). No other sharks in the system were fed during respirometry to eliminate cueing on food scents from other aquaria through shared circulating water. Upon commencing each trial, individual sharks were carefully introduced into the respirometry chamber prior to the lights turning on (i.e. at 0600 h); the system was continuously flushed for the following 6 h. Preliminary trials indicated that chamber O2 levels decreasing immediately upon introducing the shark into the system but then slowly increased to near 100% air saturation over the first 4 h and remained constant thereafter. As in other benthic shark respirometry studies (e.g. Heinrich et al., 2014; Tunnah et al., 2016), a 6-h habituation period was deemed sufficient.
After the 6-h habituation period, at 1200 h, a relay timer was used to intermittently turn off the flush pump (i.e. for 5 min in the small chambers, 7.5 min in the larger chamber), which was deemed the measurement period. This time interval was long enough to ensure that the decline in O2 could be detected but short enough such that O2 levels within the chambers did not decrease below 80% air saturation (Svendsen et al., 2016). Following each of the O2 uptake measurement periods, the flush pump was turned on for 10 min, thus returning O2 levels in the chamber water back to 100% air saturation. These measurement and flush cycles were repeated for 24 h until 1200 h on the following day, totalling 82 or 96 measurement periods (in the large and small chambers, respectively), which ensured a sufficient number of data points for each individual shark. Initially, during the first several respirometry trials, sharks were observed in the chamber every hour over the 24-h period to confirm they were at rest during the overnight hours when they are known to be more active (Heupel and Bennett, 1998).
At the end of each respirometry trial, each shark was removed from its chamber, weighed (to the nearest 5 g), and then returned to their respective holding aquarium. Sharks consumed food within an hour after removal from the chamber. Empty respirometry chambers were also cycled for 30 min before and after each trial to account for microbial accumulation within each chamber, which was later subtracted from the total oxygen uptake rates. To reduce microbial build-up, each chamber was cleaned with a 10% bleach solution, thoroughly rinsed with freshwater, and allowed to air-dry after each trial.
Experimental Treatments
Throughout their native distribution range, epaulette sharks experience annual temperatures that average daily between 20 and 28 °C (Heupel et al., 1999). To capture physiological changes in the upper end of this temperature range (i.e. from spring to summer), we used treatments progressing from low to high temperatures between 24.5 and 28.5 and acutely removed photoperiod during the 24-h trials, as detailed below, to determine how daily or seasonal cycles influence metabolic rates (Table 1). The experiments began with a constant 25 °C treatment, followed by the introduction of diel cycles where water temperatures were the lowest at 0600 h and highest at 18:00 PM to reflect warming and cooling of a reef flat across the daytime hours (Table 1). For example, in the 25-28 °C diel treatment, water temperature was set to 25 °C at 0600 h, warming across the day to 28 °C at 1800 h, and then cooling overnight back down to 25 °C (Table 1). All water temperature changes between treatments occurred slowly at 0.5 °C every other day and sharks were allowed to acclimate for at least 5 weeks before respirometry ensued (Johansen et al., 2021).
The treatment acclimation temperatures and photoperiods (NP = no photoperiod) used during respirometry across the study from August 2020-June 2021.
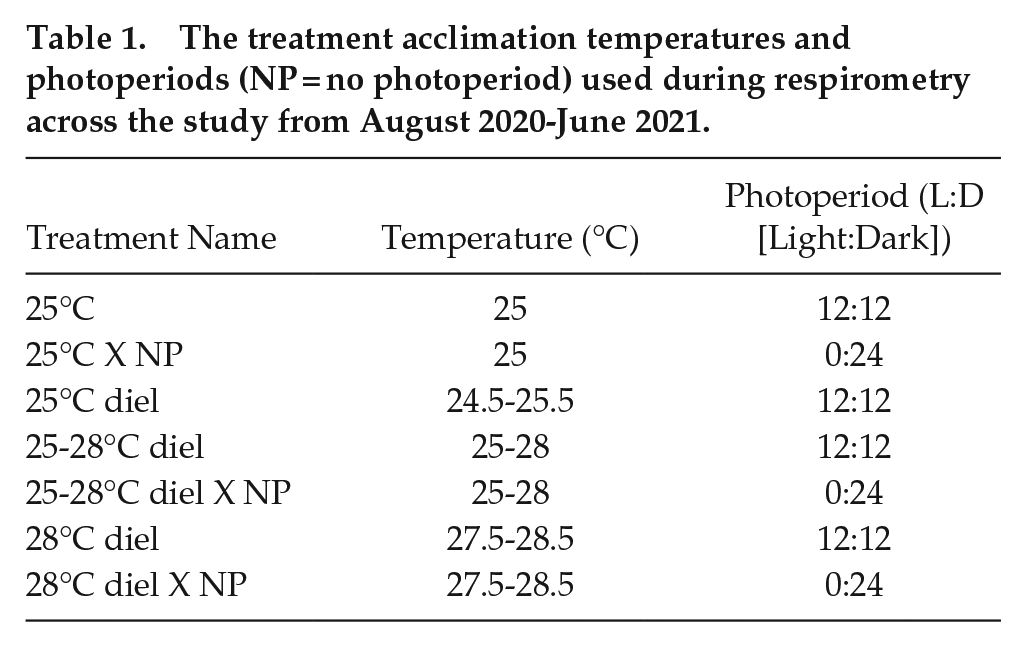
The previously described respirometry method was implemented during each experimental treatment, where trials were repeated at least twice for each temperature treatment, once under the ambient photoperiod (12:12 L:D), and once in complete darkness (i.e. using 80% light-blocking shade cloth and an opaque black tarp) (Table 1). The dark photoperiod trials totaled 42 h of dark exposure (12 h the night prior to the trial, 6 h of habituation in the chamber, followed by the 24-h trial). Sharks were allowed at least 1 week of recovery time between trials. Sharks were maintained under a 12:12 h (L:D) photoperiod when not in respirometry.
Ṁ O2 Calculations
Oxygen uptake rates (ṀO2 in mg O2 h-1) were calculated as the absolute value of the slope of the linear decrease of oxygen during each measurement period using the RespiroRS package in R (Merciere and Norin, 2021; version 4.1.1, R Core Team, 2021). Background respiration was less than 5% of shark respiration and was modeled linearly across each trial and subtracted from ṀO2 estimates (Rummer et al., 2016). Oxygen uptake slopes were linear (mean R2 = 0.96), and any uptake rates with R2 < 0.90 were removed from the dataset. ṀO2 values were mass adjusted to a 1 kg animal to account for the wide range of masses between sharks (0.91-2.00 kg) using the equation described in Bouyoucos et al. (2020a) and a scaling coefficient of 0.89 (Jerde et al., 2019). Therefore, because we standardized for mass, ṀO2 units are reported as mg O2 h-1.
Statistical Analyses
Ten generalized additive mixed model structures (gamm, mgcv package; Wood, 2021; Table 2) were performed to assess patterns in RMR over the 24-h respirometry trial between temperature and photoperiod treatments. Random effects, including sex, shark ID, and trial number for each shark were tested to account for conditioning effects to repeated respirometry (Table 2). The first order autocorrelation function (CorAR1) was included in all models to account for temporal autocorrelation from serial ṀO2 sampling. The model with the lowest Akaike information criterion (AIC) value was selected as the best fitting model. For each trial, diel scope (the change in ṀO2Rest between day [minimum] and night [maximum]; Figure 1) was calculated using the equation:
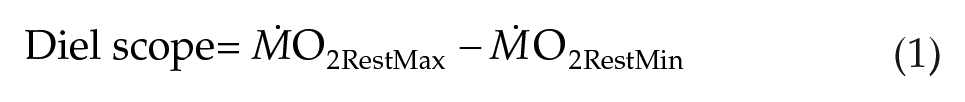
Ten generalized additive mixed models (GAMM) assessing change in metabolic rate estimates (as ṀO2) across a 24-h trial with a smoother (s[time]) and AR1 function for temporal autocorrelation applied. The fixed effects of the first model and the random effects of the third model were chosen, as it had the lowest AIC value.
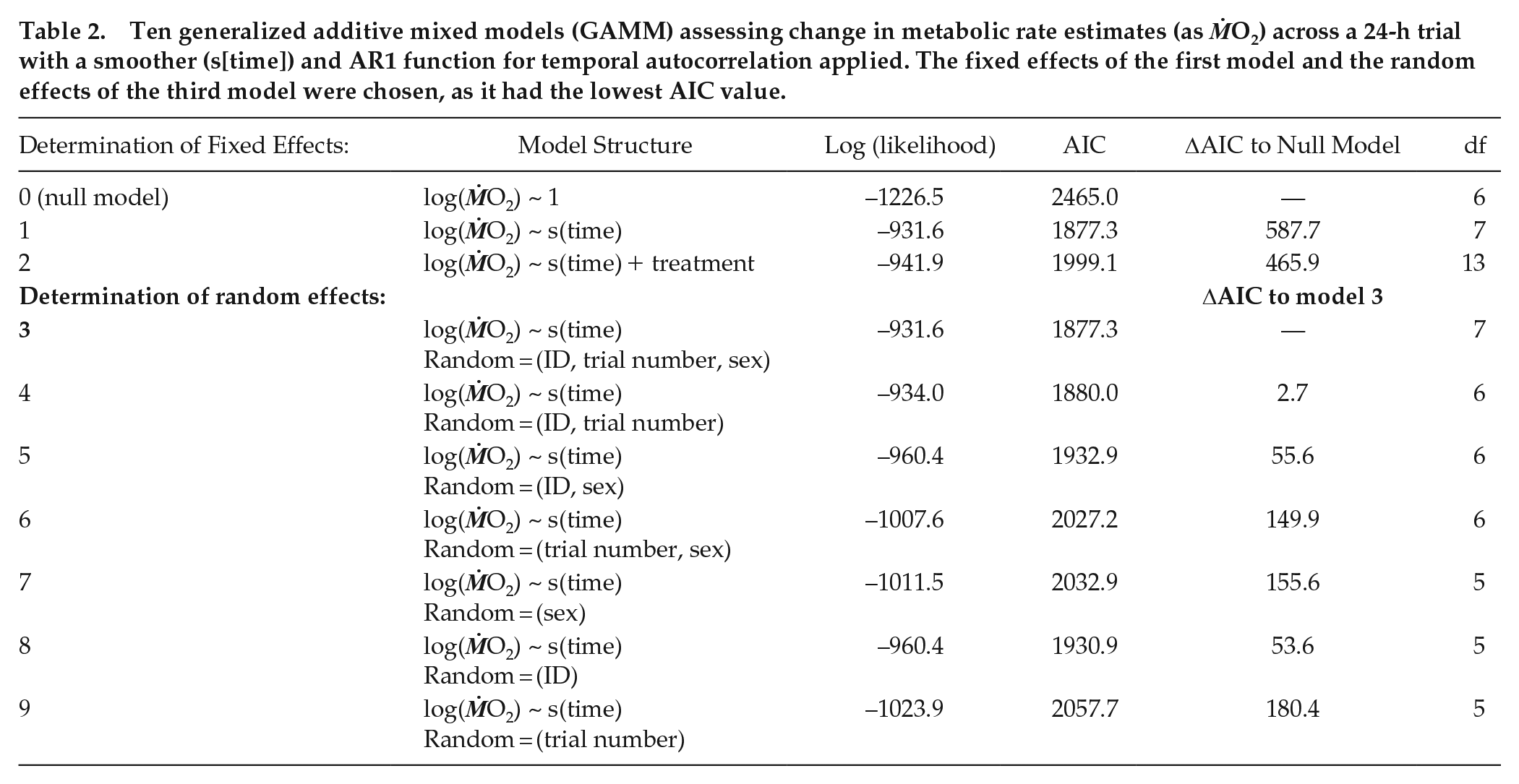
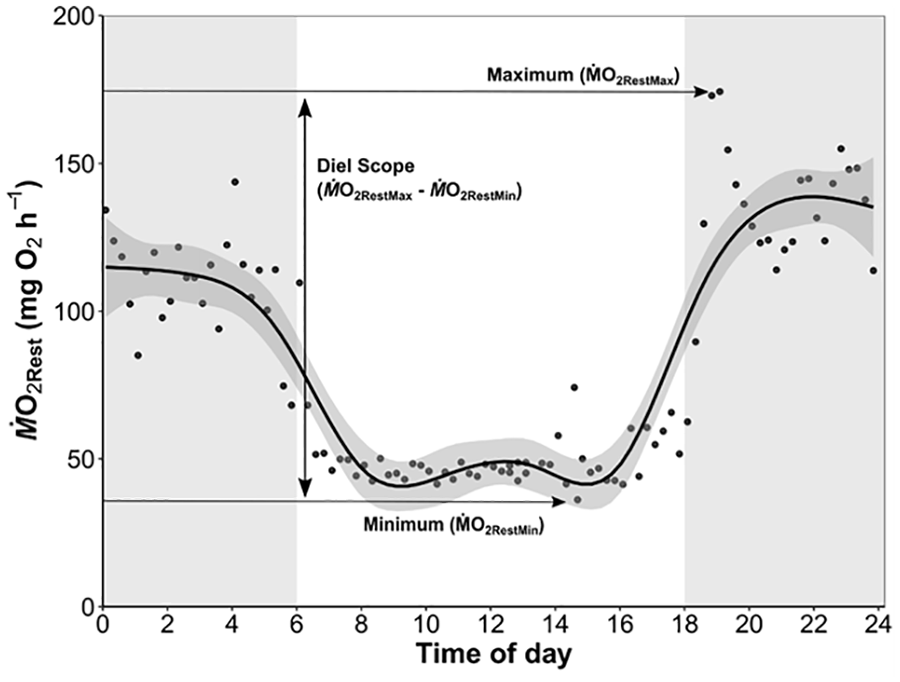
A representative ṀO2Rest trace from a shark in a 24-h respirometry trial. White and gray areas represent photoperiod of light and dark respectively. Horizontal arrows indicate the minimum and maximum ṀO2Rest GAMM (generalized additive mixed model) estimates from the trial, and the diel scope is the difference between these 2 extremes.
Finally, linear mixed-effects models (lme4 package, Bates et al., 2015) with combinations of shark ID, trial number, and sex as random effects were assessed to compare the mean, minimum, maximum, and diel scope of ṀO2 across treatments. Like the GAMM models, the linear mixed-effects model with the lowest AIC value was chosen for each ṀO2 type (Table S2345). All statistical analyses were conducted in R, where results were considered significant at α < 0.05.
Results
Of the 10 GAMM models (Table 2), model 3 without treatment as a fixed predictor and random effects of individual, trial number, and sex had the lowest AIC value and was therefore selected as the best fitting model (see Table 2). Overall, the diel pattern of increased ṀO2 at night and reduced ṀO2 during the day was unchanged despite changes in temperature and photoperiod cues (Figure 2). Moreover, neither temperature nor photoperiod affected minimum, maximum, or diel ṀO2Rest scope (Figure 3, see Supplement Table S2345). During the day, ṀO2RestMin was low (59.3 ± 10.2 mg O2 h-1, mean ± S.E.M) and increased 1.7-fold at night (101.4 ± 12.4 mg O2 h-1) (Figure 3b). Acutely removing photoperiod during respirometry did not change the diel pattern of ṀO2 under any temperature treatment (Figure 2). All results in this study were consistently observed in each individual, regardless of sex or potential conditioning from repeated experimentation across time (Supplemental Figure S1).
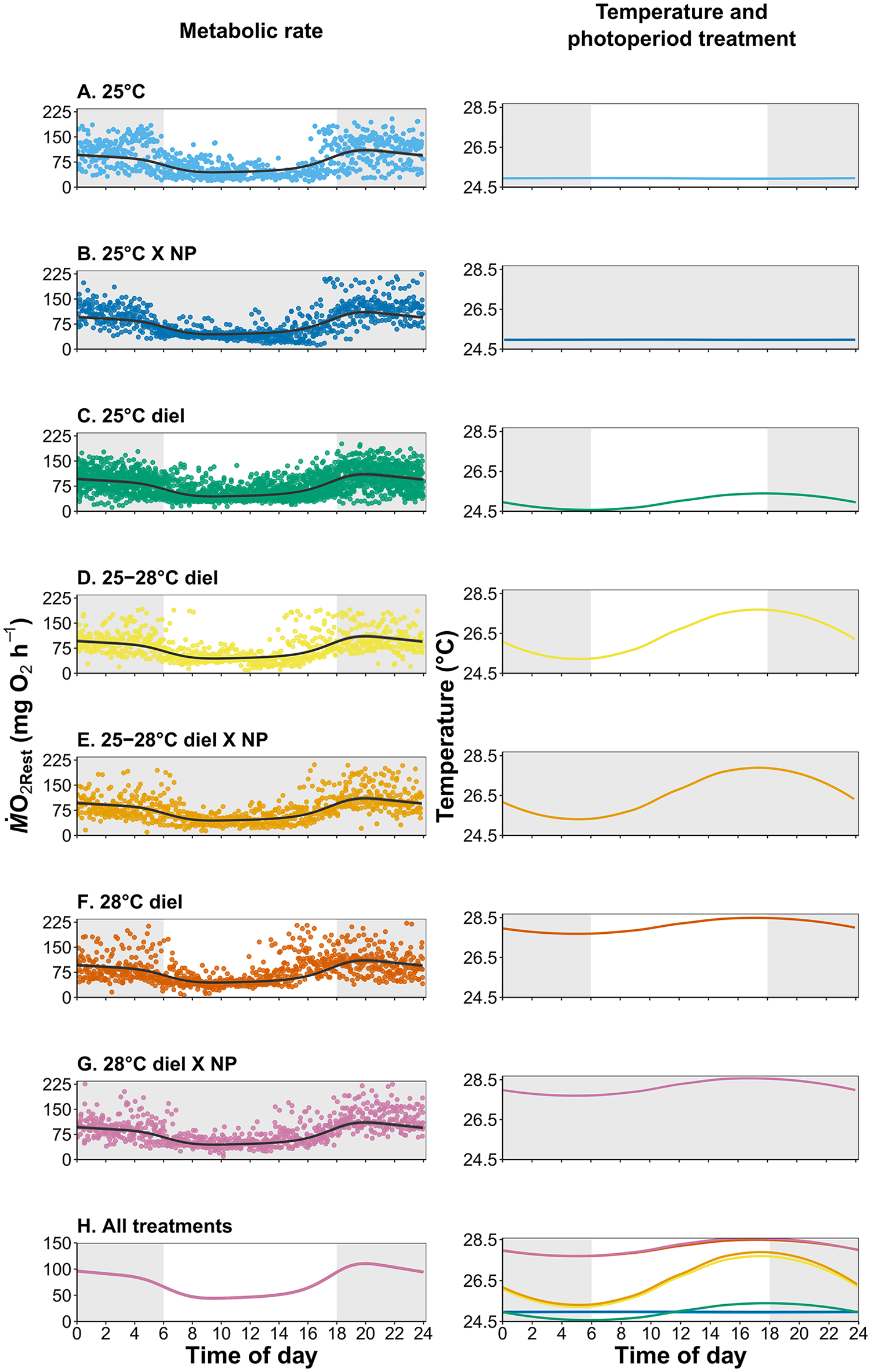
Resting metabolic rate estimates (as ṀO2Rest as data points) and GAMM (generalized additive mixed model 1) predicted fits over 24-h trial periods (left column) and the respective temperature and photoperiod treatments (NP = no photoperiod; right column) where white represents light periods and gray represents dark periods. No significance was found at α = 0.05 in the GAMM model fit between any of the treatment groups.
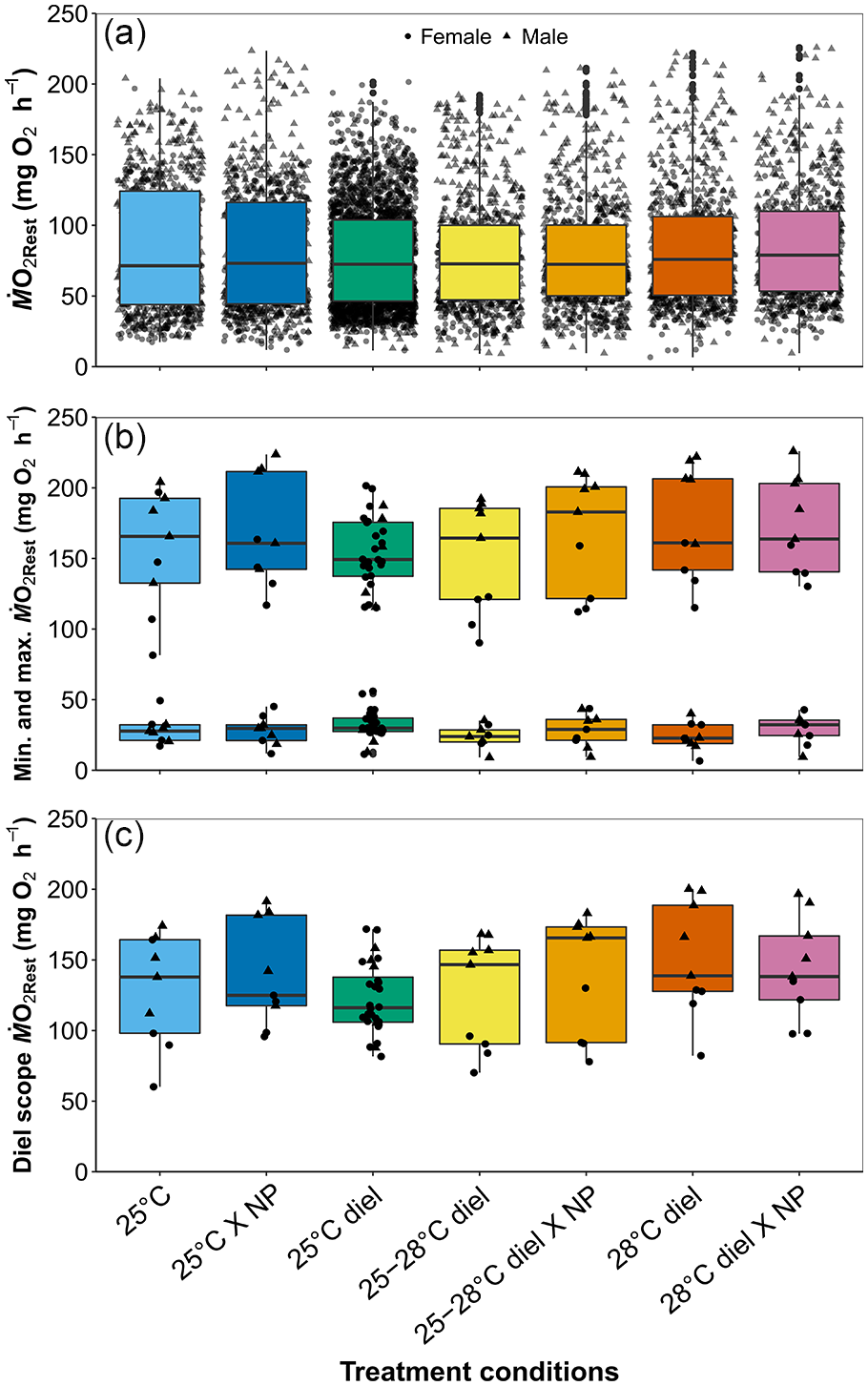
Boxplots of mean (a), minimum (b), maximum (b), and diel scope (c) of ṀO2Rest across treatments. Circular points represent females, and triangle points represent males. Colors correspond to treatment conditions in Figure 2. NP represents no photoperiod, and there was no significant difference between treatments at α = 0.05.
Discussion
The 24-h periodicity of resting metabolic rate (RMR) in epaulette sharks is biologically significant, where RMR nearly doubles at night when compared to daytime hours. Yet, this pattern was unaffected when sharks were acclimated to either of 2 ecologically relevant temperatures or upon acute photoperiod removal. There are a few examples in the literature documenting similar nocturnal metabolic patterns in sharks. For example, Nixon and Gruber (1988) reported diel metabolic patterns in lemon sharks (Negaprion brevistostris). However, this study used a large, static respirometry chamber, where sharks were able to increase their swimming speed/activity, which explained increased metabolic rates at night. Similarly, whitetip reef sharks (Triaenodon obesus) exhibit threefold increases in activity and metabolic rates at night (Whitney et al., 2007). In the little skate (Leucoraja erinacea), standard metabolic rate (SMR) follows a nocturnal rhythm as well, with nighttime rates estimated to be 3-4 times higher than daytime rates (Hove and Moss, 1997). In the current study, despite the confines of the respirometry chambers and resulting inactivity, epaulette sharks still exhibited marked differences (i.e. nearly double) in RMR between night and daytime hours. Although logistics precluded continuous monitoring of ventilation rates, it is likely that sharks increased ventilation/respiration rates at night despite inactivity, thereby accounting for the increased oxygen uptake rates. We suggest that the increase in RMR at night is related to when this species is actively foraging for food (Heupel and Bennett, 1998). Future studies where sharks are allowed to move freely, such as when using a whole-tank annular respirometry setup (e.g. Lear et al., 2017), would help strengthen the connections between activity type (e.g. hiding, walking, burst swimming, etc.), diel metabolic scope, and abiotic factors.
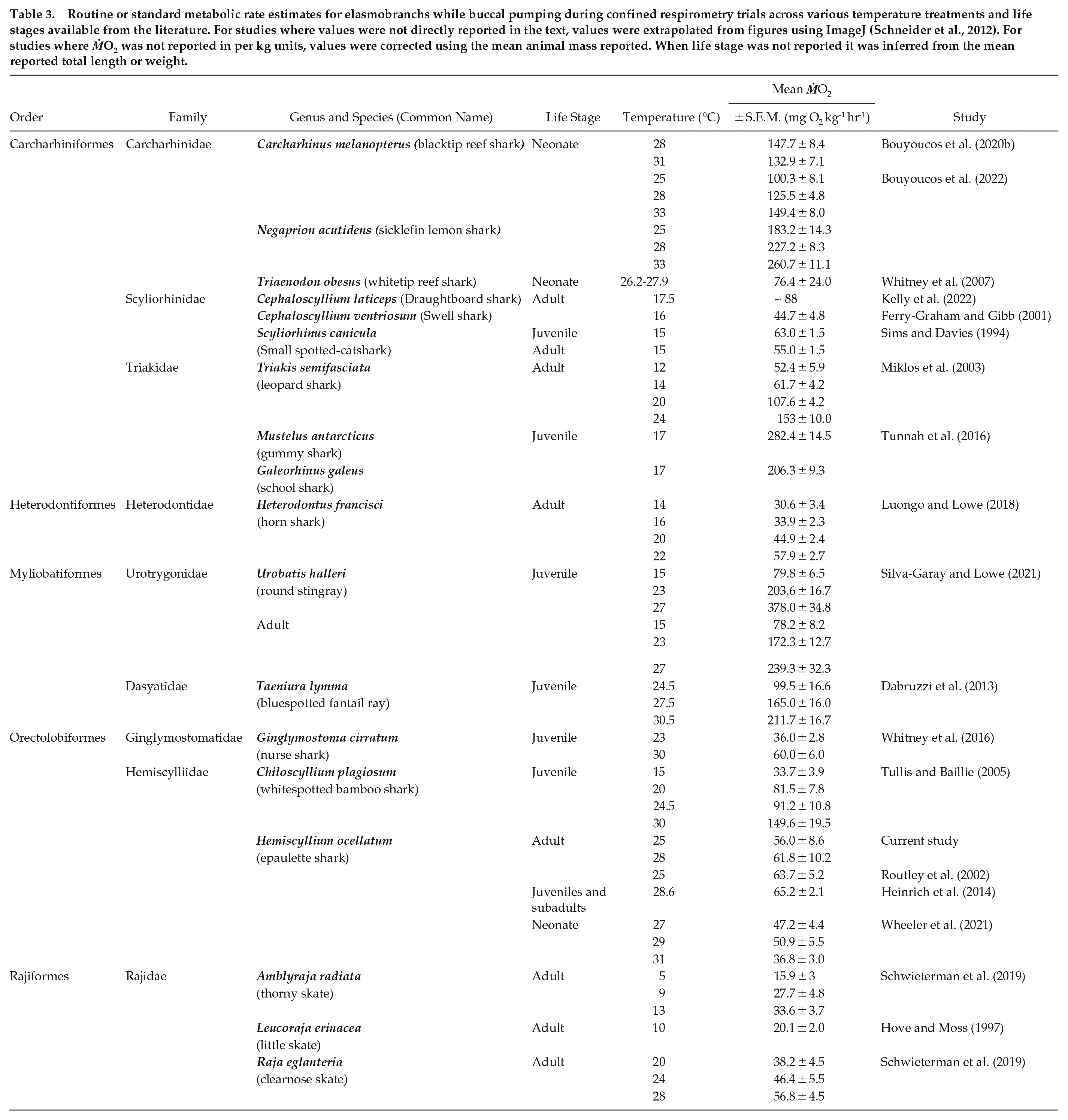
The oxygen uptake rate (ṀO2) patterns of epaulette sharks over a 24-h period are remarkably similar to distinct diel ventilation and activity patterns reported for other benthic sharks, including swell (Cephaloscyllium ventriosum), draughtboard (Cephaloscyllium isabellum), Port Jackson (Heterodontus portusjacksoni), and horn (Heterodontus francisci) sharks (Nelson and Johnson, 1970; Bass et al., 2021; Kadar et al., 2019; Kelly et al., 2020, 2021). Given the significantly lower metabolic rate estimates during the day, our findings also strongly support the behavioral sleep hypothesis proposed by Kelly et al. (2019, 2020, 2021). Indeed, we observed similarly sluggish behavior, as described by Kelly et al. (2019), where individuals were slow to react after being tipped out of shelters during the day, but typically came out of shelters on their own accord once the lights were turned off at night. Furthermore, we measured repeatedly low oxygen uptake rates, particularly between 8:00 and 15:00 h (Figure 2), thus indicating a period of rest. Whether epaulette sharks in this study were, from a behavioral and physiological perspective, truly sleeping or in a state of torpor during the day is unknown. This topic will require further investigation in the future. The mean ṀO2RestMin value across all treatments (58.9 ± 1.2 mg O2 kg-1 h-1, reported as per kg animal ṀO2 here for direct comparison to other studies) is remarkably similar to ṀO2 measured during the day in adult epaulette sharks by Routley et al. (2002) (63.7 ± 5.2 mg O2 kg-1 h-1; Table 3) and Heinrich et al. (2014) (65.2 ± 2.1 mg O2 kg-1 h-1; Table 3). Furthermore, for other elasmobranchs that exhibit buccal pumping during standard metabolic rate (SMR) estimates, reported values range 15.9 to 378.0 mg O2 kg-1 h-1 (Table 3). While partially dependent on temperature, values average 78.5 ± 17.6 mg O2 kg-1 h-1 for temperate species and 112.2 ± 13.2 mg O2 kg-1 h-1 for sub-tropical and tropical species (Table 3). Interestingly, Carcharhiniformes that buccal pump while confined in respirometry chambers, even though they naturally also ram ventilate, generally had higher SMR estimates, despite their inactivity during experiments (Table 2; Miklos et al., 2003; Whitney et al., 2007; Tunnah et al., 2016; Bouyoucos et al., 2020b, 2022). Given that ṀO2 values associated with daytime and/or inactivity are similar between tropical and temperate species, perhaps the metabolic savings of daytime behavioral sleep is similar across benthic buccal/spiracle pumping sharks and skates. More research is needed in this area.
Routine or standard metabolic rate estimates for elasmobranchs while buccal pumping during confined respirometry trials across various temperature treatments and life stages available from the literature. For studies where values were not directly reported in the text, values were extrapolated from figures using ImageJ (Schneider et al., 2012). For studies where ṀO2 was not reported in per kg units, values were corrected using the mean animal mass reported. When life stage was not reported it was inferred from the mean reported total length or weight.
It is unclear how long photoperiod cues need to be removed to interrupt the observed rhythmic pattern of metabolic rate in epaulette sharks, as many fish species will continue circadian rhythms under constant conditions (i.e. no photoperiod) for days to weeks (Reebs, 2002; de Almeida Moura et al., 2017). Some of the earliest chronobiology studies in elasmobranchs noted that horn and swell sharks exhibit varying degrees of endogenous control of activity levels after 15 or 18 days of constant dark or light respectively (Nelson and Johnson, 1970; Finstad and Nelson, 1975). Diel activity patterns can be interrupted by changes in photoperiod in Port Jackson sharks, but not as easily in draughtboard sharks (Kelly et al., 2020). Constant light photoperiod or anesthetization by tricaine methanesulfonate (MS-222) results in diminished diel SMR patterns in the little skate (Hove and Moss, 1997). In the current study, a 42-h dark exposure during respirometry trials did not affect mean RMR or diel scope. However, the purpose of dark photoperiod trials during this study was to assess whether light cues themselves were causing the diel pattern of metabolic rate, which we found was not the case (Figure 2). To tease apart how to remove or shift diel patterns seen here with light cues, the photoperiod should be removed for a longer period, at least on the order of several days to weeks (Nelson and Johnson, 1970; Reebs, 2002). More research in this area coupled with exploration of clock genes/cells may help further elucidate the role of the biological clock and circadian rhythms in elasmobranch physiology, which is essentially an unstudied field for this taxon.
Our findings also contribute to best practice respirometry methods for elasmobranchs. The strong diel ṀO2 pattern seen in our data is an important factor to consider when assessing metabolic rate as part of a study. Indeed, a mean value alone will not reflect this trend, and this 24-h pattern is likely often overlooked. Moreover, resting or standard metabolic rate estimates are not necessarily reflected by a single mean value, but rather a pattern of oxygen uptake rates over a 24-h period (e.g. see Speers-Roesch et al., 2018). Assessment over a full 24-h cycle allows for diel scope to be calculated, and the shifts in ṀO2 peaks and troughs can indicate if a species is crepuscular or nocturnal or perhaps show treatment effects (e.g. hypoxia, pCO2, temperature) that may otherwise be masked in using a mean ṀO2. Furthermore, if ṀO2 for a buccal pumping elasmobranch does not exhibit a diel pattern, it may be that optimal unstressed conditions that are needed to estimate SMR may not be met. This means that behavioral sleep may not occur due to the stress of the experiment. In this case, an alternative respirometry chamber design and/or a longer habituation period should be considered. It may be more difficult to achieve such repeatable results—particularly for ṀO2RestMin—with recently caught, large, or mobile species. Finally, while it is likely that the minimum ṀO2Rest values reported here represent the true standard metabolic rate (SMR) for epaulette sharks, we conservatively identify all metabolic rates in this study as variations (i.e. minimum, maximum, and diel scope) of RMR, on the basis that sharks were sexually mature and reproducing (Chabot et al., 2016). Our findings add to the growing body of literature that is helping to fine tune methods and techniques that are critical in the fields of conservation and ecological physiology.
Life stage may also play an important role in the trends we observe in metabolic rates. Past studies have already indicated that epaulette embryos, neonates, and juveniles are negatively impacted by elevated temperatures (e.g. 27-32 °C; Gervais et al., 2016, 2018; Wheeler et al., 2021), those that represent current-day summer averages and ocean warming scenarios. For neonates, resting ṀO2 increased in sharks reared at 29 °C when compared to sharks reared at 27 °C but then decreased in sharks reared at 31 °C (Wheeler et al., 2021). The thermal effects observed by Wheeler et al. (2021) on embryos and neonates contrast the lack of thermal effects in the current study, which could be due to several factors. First, Wheeler et al. (2021) included warmer treatments than were used in the current study. Second, early life history stages (e.g. embryos and neonates) are known to have narrower thermal windows for an array of physiological traits, making them more vulnerable than later life stages (Angilletta, 2009). Third, studies on the effects of elevated temperatures on early life stages of epaulette sharks were conducted in the laboratory, where embryos did not experience diel fluctuations in abiotic conditions, thus perhaps not providing pre-conditioning, which may be important and is worthy of future investigations.
In contrast to early life stage epaulette sharks reared under controlled laboratory conditions, the adults investigated in this study presumably spent at least 2-6 years exposed to natural fluctuations in abiotic conditions in the wild before captivity (i.e. based on approximate age of maturation in captivity, Wheeler personal observation). This begs the question: how important are fluctuations in abiotic conditions in shaping physiological tolerance? Experiments that exposed epaulette sharks to repeated hypoxia (hypoxic-preconditioning) reported metabolic depression (Routley et al., 2002). Perhaps this species uses similar strategies for thermal compensation as well. Indeed, other sub-tropical and tropical elasmobranch species have been noted to have low metabolic temperature sensitivities as well, including blacktip reef sharks (Carcharhinus melanopterus), sicklefin lemon sharks (Negaprion acutidens), bull sharks (Carcharhinus leucas), and largetooth sawfish (Pristis pristis), (Bouyoucos et al., 2020a, 2022; Lear et al., 2020). This poses interesting questions revolving around mainly coastal, warm water species that experience large diel fluctuations in temperature and how this shapes their physiological responses. Future studies that assess other physiological mechanisms that may be more temperature sensitive, such as muscle dynamics or swimming speed, may help elucidate thermal effects not observed herein on metabolic rate (e.g. von Herbing, 2002; Lauder and Di Santo, 2015).
On Heron Island, the largest studied population of epaulette sharks (Heupel and Bennett, 2007), year-round average temperatures range 20 to 28 °C, with extremes of 16 and 34 °C (Nay et al., 2020). Water temperatures across a single day can be homogeneous across the partially isolated reef flat, and epaulette sharks do not behaviorally thermoregulate by moving to a preferred temperature (Nay et al., 2020). It is possible that, by not using behavioral thermoregulation, epaulette sharks must use other physiological mechanisms for thermal compensation. In the current study, sharks were given 5 weeks to acclimate to experimental temperatures, where shorter exposure times may have elicited greater changes in metabolic rates (Johansen et al., 2021). Studies where metabolic rate is assessed across a full annual temperature range while quantifying potential thermal compensation pathways (e.g. tissue citrate synthase and lactate dehydrogenase enzyme activities) may help discern why, in this study, temperature had no measurable impact on metabolic rate.
For epaulette sharks, time of day may have a stronger influence on metabolism than temperature. This may be reflective of their role in their natural environment where they dwell in the shallow reef flats that are subject to dramatic fluctuations in abiotic conditions. Within the context of predator-prey interactions, compensating for sub-optimal temperatures may represent a tradeoff to ensure survival (Vaudo and Heithaus, 2013), as this small, benthic, meso-predator hides during the day to avoid predators, even when water temperatures increase to potentially sub-optimal conditions. Our findings strongly support the notion that diel patterns in metabolic rate exist and are profound in this taxon, particularly for benthic buccal pumping elasmobranchs. This finding should be considered in future studies assessing metabolic rate in the laboratory. Finally, our findings contribute to the growing literature on this extremely tolerant shark species, where we continue to unveil different aspects of the epaulette shark’s ecological physiology to understand adaptations that may be key to how they survive and thrive under challenging conditions.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304221107843 – Supplemental material for Diel Rhythm and Thermal Independence of Metabolic Rate in a Benthic Shark
Supplemental material, sj-docx-1-jbr-10.1177_07487304221107843 for Diel Rhythm and Thermal Independence of Metabolic Rate in a Benthic Shark by Carolyn R. Wheeler, Jeff Kneebone, Dennis Heinrich, Jan M. Strugnell, John W. Mandelman and Jodie L. Rummer in Journal of Biological Rhythms
Footnotes
Acknowledgements
Support for this work was provided by an American Australian Association graduate scholarship (to C.R.W.), a JCU Postgraduate Research Scholarship (to C.R.W.), the Australian Research Council (ARC) Center of Excellence for Coral Reef Studies (to J.L.R.), the Australian Wildlife Society (to C.R.W.), the Australian Society for Fish Biology (to C.R.W.), and an anonymous donor to the New England Aquarium for funds contributed to equipment used in this study. The authors would like to thank A. Bacchar, M. Lonati, H. Nieves, S. Schlueter, and B. Warming for their assistance with husbandry during this project. Finally, we thank the staff of the Marine Aquaculture Research Facilities Unit at James Cook University for their invaluable technical support. Finally, we would like to thank the 2 anonymous reviewers whose comments and suggestions greatly improve this manuscript. ARC Center of Excellence for Coral Reef Studies; American Australian Association; Australian Wildlife Society, Australian Society for Fish Biology; New England Aquarium Corporation
Author Contributions
This project was conceived and designed by C.R.W., J.K., D.H., J.S., J.W.M., and J.L.R. Data were collected and analyzed by C.R.W. and J.K. All authors were involved in the preparation of the manuscript.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
