Abstract
Ketones are known to constitute an important fraction of fuel for consumption by the brain, with brain ketone content generally thought to be low. However, the recent observation of 1-mmol/L levels of brain β-hydroxybutyrate (BHB) in children on the ketogenic diet suggests otherwise. The authors report the measurement of brain BHB and lactate in the occipital lobe of healthy adults using high field (4-T) magnetic resonance spectroscopy, measured in the nonfasted state and after 2-and 3-day fasting-induced ketosis. A 9-mL voxel located in the calcarine fissure was studied, detecting the BHB and lactate upfield resonances using a 1H homonuclear editing sequence. Plasma BHB levels also were measured. The mean brain BHB concentration increased from a nonfasted level of 0.05 ± 0.05 to 0.60 ± 0.26 mmol/L (after second day of fasting), increasing further to 0.98 ± 0.16 mmol/L (after the third day of fasting). The mean nonfasted brain lactate was 0.69 ± 0.17 mmol/L, increasing to 1.47 ± 0.22 mmol/L after the third day. The plasma and brain BHB levels correlated well (r = 0.86) with a brain–plasma slope of 0.26. These data show that brain BHB rises significantly with 2-and 3-day fasting-induced ketosis. The lactate increase likely results from ketones displacing lactate oxidation without altering glucose phosphorylation and glycolysis.
As an alternative to glucose, ketones are known to constitute an important fraction of fuel for consumption by the brain under the conditions of certain diets, fasting, or vigorous exercise (Balasse et al., 1978; Robinson et al., 1980). Much of the understanding of human brain ketone use (Hasselbalch et al., 1995; Blomqvist et al., 1996) is based on models developed from rodent data (Gjedde and Crone, 1975; Hawkins and Biebuyck, 1978), possibly because of a lack of stable radiotracer analogue for use in humans. However, significant differences exist between the rodent and human brain, particularly with regard to brain metabolism in fasting. For example, Hasselbalch et al. (1994) showed that in 3.5-day fasted humans, total brain glucose consumption decreases by 25% with more than a 13-fold increase in total brain ketone use. Calculating the fractional ATP production from the two fuels in the brain shows a 35% contribution from ketones under the fasting condition. This rate of ketone body consumption is considerably higher than that seen in lightly anesthetized 2-day fasted rats, reported at 3% to 5% (Mans et al., 1981; Hawkins et al., 1986).
Further studies focus on the ketone transport parameters in rodent and human brain. In rodents, most studies report that the concentrations of brain ketones are low (Hawkins et al., 1971, 1986; Gjedde and Crone, 1975). Although data from brain uptake studies of labeled ketones show variable values for the Michaelis-Menten and diffusion parameters, these data, together with the ready availability of oxidative enzymes, suggest that transport across the blood–brain barrier dominates in the control of ketone body consumption (Gjedde and Crone, 1975; Cremer et al., 1982; Hawkins et al., 1986). In humans, Blomqvist et al. (1996) also found supportive data for this view using 11C-β-hydroxybutyrate positron emission tomography. However, the complexity of acquiring human data have made conclusive measurements difficult. Thus, additional work is ongoing to better define the parameters of ketone transport in human brain, particularly given the difference relative to rodents in the fractional contribution of ketones to brain fuel use in fasting.
Given such a model where the ketone consumption is transport limited and brain oxidation rates are high, we would predict brain content of ketones to be low relative to plasma levels. As reported by Hawkins et al. (1978) and Ruderman et al. (1974), this is clearly the case in rodents. However, the recent magnetic resonance (MR) spectroscopic observation of 1-mmol/L levels of brain β-hydroxybutyrate (BHB) in children on the ketogenic diet suggests that brain BHB can become relatively high (Novotny and Rothman, 1996). Whether such accumulation of brain BHB could occur in healthy ketotic adults is the focus of this report. We used high-field (4-T) MR spectroscopy to measure brain BHB levels in adults, before and after 2 and 3 days of fasting. In the occipital lobe, brain BHB increases from levels at or below the noise threshold of the data (< 0.1 mmol/L) to 0.98 ± 0.16 mmol/L after a 3-day fast. Furthermore, an increase in brain lactate also occurs during this same period, increasing from a nonfasted level of 0.69 ± 0.17 to 1.47 ± 0.22 mmol/L.
MATERIALS AND METHODS
A 4-T Varian Inova whole-body imaging spectrometer and a 7-cm 1H surface coil (Varian Instruments, Palo Alto, CA, U.S.A.) were used for all studies. The surface coil was placed immediately posterior to the occipital ridge. Scout imaging was performed using an inversion recovery gradient echo with 128 × 128 resolution to locate the volume of interest. A 3 × 3 × 1 cm (9-mL) tissue voxel was selected through the calcarine fissure for all spectroscopic data. The detection of both the BHB (1.20 ppm) and lactate (1.33 ppm) upfield methyl resonances was achieved using a homonuclear editing sequence (Hetherington et al., 1985) optimized at 1.20 ppm. In this sequence, a 1-3-3-1 refocusing pulse is used to eliminate J-modulation of the coupled methylene resonances. On alternate scans, J-modulation is induced by application of a shaped DANTE (delays alternating with nutations for tailored excitation, Morris and Freeman, 1978) 180° pulse centered at 4.15 ppm, inverting both the methylene resonances of BHB and lactate. On the non–J-modulated scans, off resonance effects are minimized by applying the DANTE symmetrically about the BHB 1.20-ppm position (i.e., at −1.75 ppm). Subtraction of these two scans results in the “edited” spectrum, detecting the methyl-coupled resonances of BHB and lactate. Spatial localization was achieved through two dimensions of ISIS (image selected in vivo spectroscopy, Ordidge et al., 1986) (3 × 3 cm) and slice-selective (1-cm thick) excitation. Water suppression was achieved by applying a binomial refocusing (1-3-3-1) pulse optimized for 1.20 ppm. Repetition time was 2 seconds; echo time was 136 milliseconds. For each measurement, 17 minutes of signal averaging (512 acquisitions) were used. The total duration of the study was less than 60 minutes.
Data were processed using a 50-Hz convolution difference with a 2-to 7-Hz Lorentz-to-Gauss conversion for an optimal signal-to-noise ratio. Phasing was performed using the non– J-modulated spectrum as a reference. Quantification of the BHB and lactate was performed using an internal reference of N-acetyl aspartate (NAA) at 10 mmol/L (Pan et al., 1998). The NAA was selected as the reference given its minimal concentration difference between gray and white matter.
Five healthy adult volunteers were recruited from the laboratory community under Institutional Review Board guidelines and were screened for abnormalities of glucose, liver, and hematologic functions. Of these five, four completed the fasting study. Volunteers were admitted to the General Clinical Research Center of the Albert Einstein College of Medicine during the final 2 days of the fast. Fasting was initiated after the evening meal in all cases, with the data collected at 40 to 48 hours (2 days) and at 64 to 72 hours (3 days) after beginning the fast. All volunteers were required to drink at least 2 L of water each day of the fast and to take vitamin and mineral supplements. Plasma BHB levels were measured 1 hour before each of the two MR studies using an Analox GM7 analyzer (Analox Instruments, U.S.A.). Three of the four fasted volunteers participated in a second fast at least 90 days after the initial study. The data from all seven of the fasting studies (and five nonfasted) were used to generate the plots in Figs. 3 and 4.
RESULTS
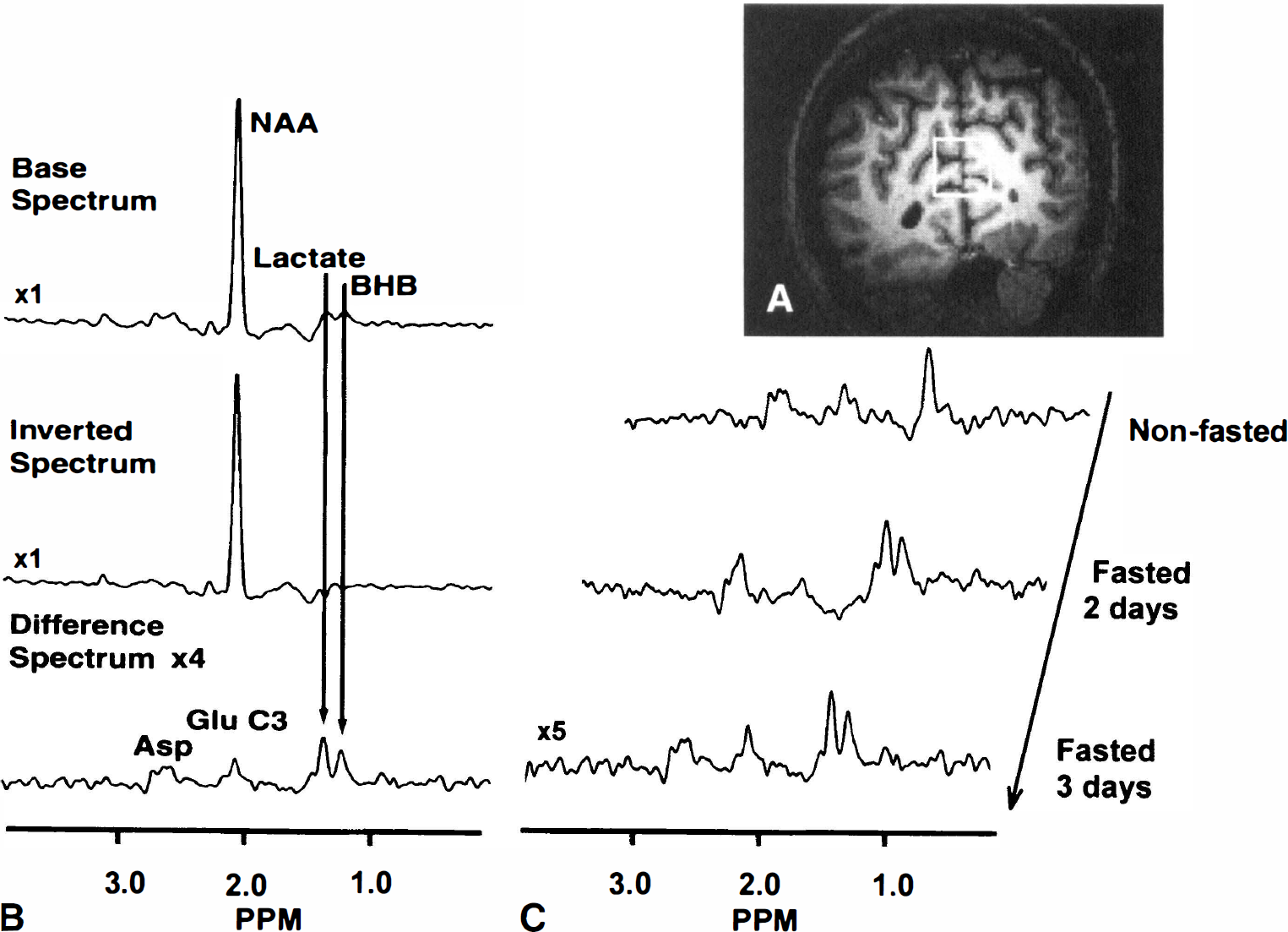
All of the fasted volunteers became ketotic, as evaluated by urine and blood BHB measurements. The development of the ketosis as manifest in plasma BHB levels is shown in Fig. 1 for all four volunteers. Fig. 2A shows the anatomic image with the selected 9-mL voxel of sampling. Fig. 2B shows the spectra displaying the editing process with the non–J-modulated (top), J-modulated (middle), and “edited” difference (bottom) spectra, indicating the lactate and BHB resonances. Three edited spectra from the same volunteer under normal dietary conditions, and after 2 and 3 days of fasting, showing the increases in both the BHB and lactate resonances are shown in Fig. 2C. Clearly, there is an increase in the intensity of BHB relative to lactate as the fast progresses. Fig. 3 shows the brain BHB concentrations from the nonfasted and fasted subjects plotted against plasma BHB concentrations. Analysis of the brain-to-plasma BHB demonstrated an excellent correlation with r = 0.86, P < 0.0001.
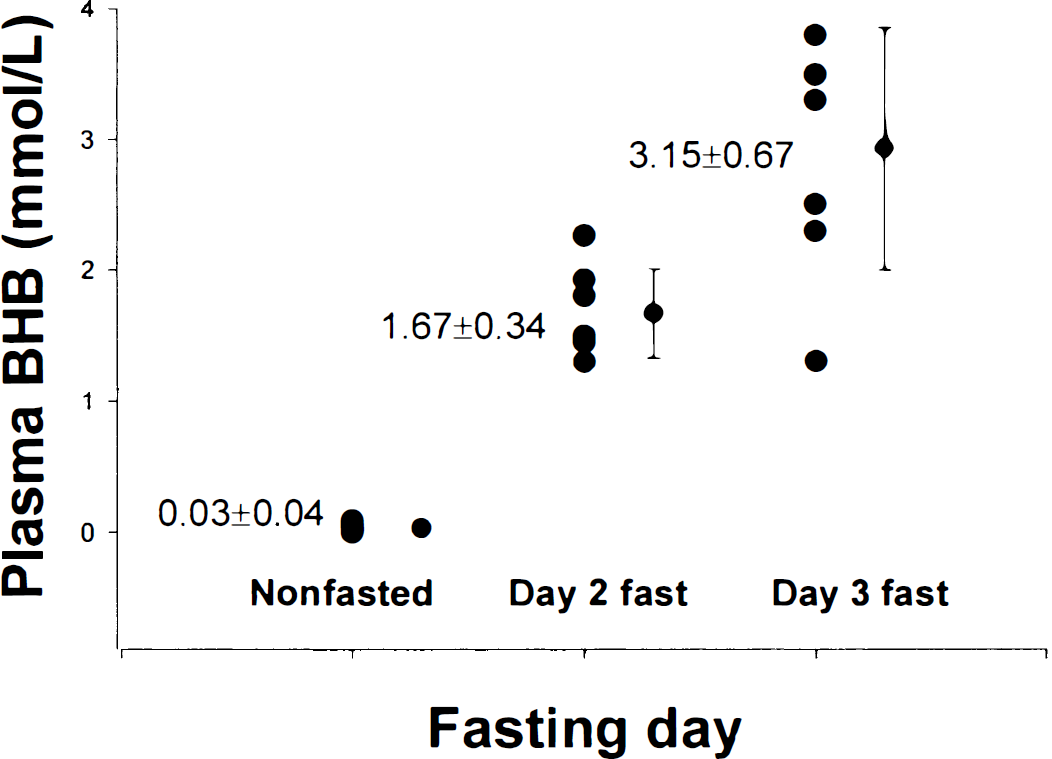
Plot showing the data, mean, and standard deviation from all seven data sets of plasma brain β-hydroxybutyrate (BHB) levels with days of fasting.
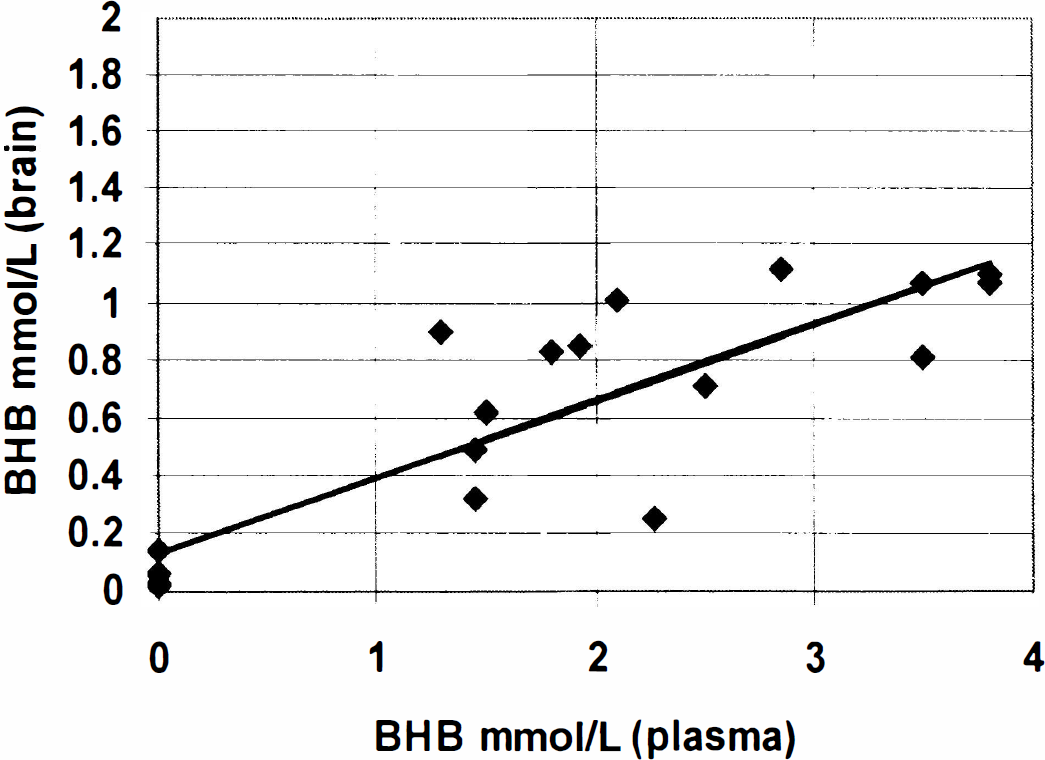
Regression data from all the data sets showing brain brain β-hydroxybutyrate (BHB) relative to plasma BHB. The regression for the brain BHB–plasma BHB correlation has an r value of 0.86 and slope of 0.26 (P < 0.0001).
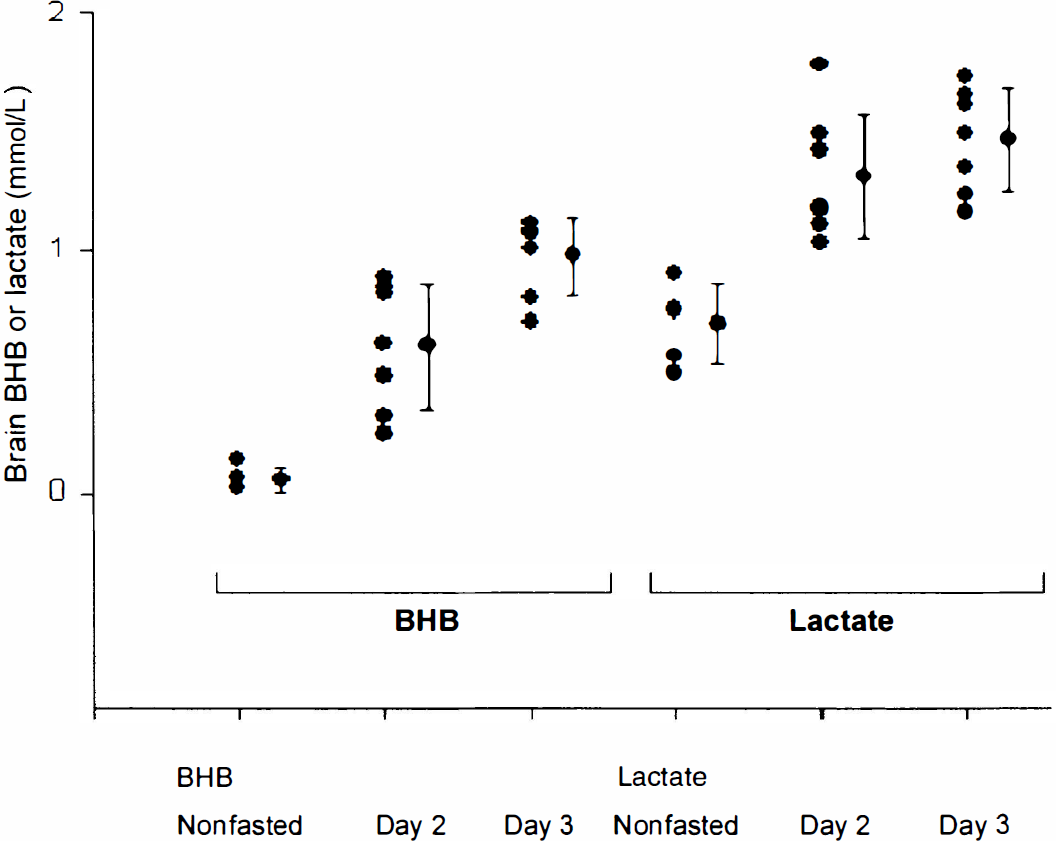
Plot showing the progression of the fast–brain brain β-hydroxybutyrate (BHB; left) and lactate (right) with days of fasting.
Figure 4 shows a compilation of the brain lactate and BHB data as a function of fasting state. Using the brain NAA level as an internal reference at 10 mmol/L (Pan et al., 1998), lactate increased from a nonfasting level of 0.69 ± 0.17 to 1.31 ± 0.26 mmol/L after 2 days of fasting, reaching 1.47 ± 0.22 mmol/L after the third fasting day. The low nonfasted lactate concentration is in good agreement with previous MR spectroscopic measurements in the occipital lobe (Hanstock et al., 1988; Prichard et al., 1991; Sappey Marinier et al., 1992), which range from 0.5 to 1 mmol/L. The mean BHB concentration after 2 days of fasting increased from a baseline of 0.05 ± 0.05 mmol/L (n = 5 different volunteers) to 0.60 ± 0.26 mmol/L (n = 7 data sets, 4 different volunteers),increasing further to 0.98 ± 0.16 mmol/L (n = 7 data sets, 4 different volunteers) after the third day.
DISCUSSION
We have used high-field MR spectroscopy to measure brain BHB concentrations in 2-and 3-day fasted normal adult volunteers, detecting an increase from less than 0.1 mmol/L in the normal dietary state to approximately 1 mmol/L by the third day of fasting. The observed BHB and lactate resonances represent contributions from brain tissue, CSF, and blood compartments. However, we believe that the measurements result largely from the tissue compartment because of the following constraints. First, imaging data from primate and human brain show that the fractional contributions from blood and CSF in this region is less than 10% (Eichling et al., 1975; Kohn et al., 1991). Using a CSF-to-blood ratio for BHB of 0.15 (Lamers et al., 1987) and a plasma concentration of 3.5 mmol/L, the contribution from the nonbrain compartments is expected to be 0.17 mmol/L, thus giving a lower limit for the tissue BHB concentration of 0.9 mmol/L. Second, the line width of the BHB is consistent with that seen for NAA, which we know is located only within brain tissue (Urenjak et al., 1993). Thus, the contribution of BHB from nontissue compartments to the apparent BHB signal is likely to be small.
The elevated lactate also likely represents primarily the brain tissue compartment. Although the CSF-to-blood ratio of lactate is high (approximately 1.0;Lamers et al., 1987)), the blood concentration of lactate does not appear to change with fasting (Hasselbalch et al., 1994). Thus, the increase in the lactate resonance is unlikely to arise from nontissue compartments.
It is possible that the T2 relaxation rate of NAA differs from both that of BHB and lactate, resulting in concentrations that differ from these reported values. However, based on studies in animal brain comparing MR spectroscopic studies and brain extract lactate measurements, we do not anticipate such an effect to be large (Schupp et al., 1993). Furthermore, because no evidence suggests a change in relaxation times with fasting, we believe that the changes in concentration are a true physiologic effect.
Consideration of acetoacetate
The other major ketone body is acetoacetate, which has its magnetic resonance signal at 2.27 ppm), the redox (oxidized) couple to BHB. Under normal conditions, the ratio of acetoacetate to BHB is a measure of the redox potential. Their interconversion is believed to be rapid (Hawkins et al., 1986), such that the concentration of acetoacetate would be directly determined by a scaling factor from BHB. The ratio of acetoacetate to BHB in plasma after a 3-day period of fasting in humans is approximately 1:3 (Balasse et al., 1978). Studies of chronically ketotic rats (Devivo et al., 1978) show no significant change in the brain redox potential. Thus, these measurements of BHB are expected to linearly reflect total ketone concentrations.
Relation between plasma and brain β-hydroxybutyrate during fasting
Based on the regression analysis between brain and plasma BHB, there appears to be a strong correlation between them with r = 0.86 and slope = 0.26, as shown in Fig. 3. We anticipate that two factors affect this correlation: net ketone transport and oxidative flux. A wide range of Michaelis–Menten Km values has been reported for BHB transport (2.5 to 13.9 mmol/L) in rodents (Gjedde and Crone, 1975; Pollay et al., 1980). Given the current data, with either a symmetric Michaelis Menten model (where Km is much larger than BHB) or a diffusion-dominated model, we expect the efflux of BHB to be approximately 0.26 that of the influx (although if the Km is on the order of the extracellular BHB, the percentage of efflux would be greater than this minimum estimate). Because fasting has been shown in rodents to result in changes in transport parameters (Gjedde and Crone, 1975), it is possible that such adaptive changes also occur in humans. However, independent of whether or not transport parameters are increased with fasting, it is nonetheless clear that given the substantial increase in brain BHB, the activity of the ketone body oxidation pathway is not large compared with its net transport activity.
Increased lactate with fasting
An increased brain lactate with 2-and 3-day fasting is consistent with the data of Owen et al. (1967). Owen et al. used whole-brain arteriovenous difference measurements to observe an increased lactate efflux with prolonged (3-week) fasting. This may be compared with Hasselbalch et al. (1994), who studied 3.5-day fasted nonobese subjects, reporting no significant change in jugular venous lactate efflux as determined by the Fick's principle. In the current study, we believe that the in vivo MR measurement of tissue lactate is a more direct measure of the metabolic state of the brain tissue, thus allowing the detection of the increased lactate even during this relatively brief period of fasting.
It is possible that the increase in lactate results from a pH change with fasting. For example, if the brain tissue were to become alkalotic, this could contribute to an increase in the concentration of brain lactate, as well as the other ketoacids. However, as far as we are aware, no studies have specifically examined this issue in fasting, although a model of chronic dietary ketosis in rats showed no pH changes (Al-Mudallal et al., 1996).
Physiologically, the increased lactate necessarily reflects the balance between production and clearance. On the production side, lactate is known to increase during functional stimulation. However, the rate of neuronal activity and glucose consumption is unlikely to rise with ketosis. Autoradiographic studies in conscious rats show no change in glucose use with ketosis resulting from either fasting or ketone infusion (Corddry et al., 1982). Instead, ketosis and fasting are known to ameliorate seizure activity (Huttenlocher, 1976), which thus may decrease functional activity and lactate production.
The major alternate possibility for the increase in lactate is a decrease in clearance by metabolism or transport. Previously, Hasselbalch et al. (1994) demonstrated that in 3.5-day fasted volunteers, ketone oxidation could provide 35% of brain fuel needs. Thus, assuming that brain energy needs are unchanged, we expect an equivalent decrease in lactate oxidation, which in turn would result in an increase in lactate concentration (until lactate efflux sufficiently increases, accounting for the gap between production and metabolism). Alternatively, brain lactate could increase from an inhibition of lactate (brain to plasma) efflux, possibly through competitive inhibition by ketones of the monocarboxylic acid transporter. However, this is less likely for several reasons. First, as shown by Owen et al. (1967), net cerebral lactate efflux is increased with fasting, not decreased. Second, unidirectional lactate influx would be expected to either remain unchanged or decrease, since plasma lactate is relatively unchanged with fasting whereas plasma ketones are increased and could inhibit lactate influx (Hasselbalch et al., 1994, 1995). Thus, given an increase in net lactate efflux while unidirectional lactate influx most likely decreases, unidirectional lactate efflux is therefore also likely to be either unchanged or increased. An increase in unidirectional lactate efflux while the steady-state lactate concentration increases also most likely means a decrease in its metabolic clearance. We therefore favor a hypothesis in which lactate oxidation is displaced by ketones. In the future, it should be possible to directly test these possibilities using MR spectroscopy to measure the metabolic fate of labeled glucose and ketones (Rothman et al., 1992; Pan et al., 1997).
If the elevated lactate results from decreased metabolic clearance, this would provide evidence for the presence of continued obligate glucose use, as suggested by Pellerin and Magistretti (1995). In this hypothesis, the clearance of synaptic glutamate during functional activity requires ATP produced glycolytically by the astrocyte. This requirement for glycolytic ATP could not be replaced by ketone oxidation. Under nonfasting conditions, the lactate produced by this pathway would be oxidized in either the astrocyte or shuttled to the neuron. The effect of increasing ketone consumption would be to displace the lactate produced by this pathway away from oxidation, without affecting glucose phosphorylation and glycolysis (evidence for this has been observed by Corddry et al., 1982). Consequently, an increase in lactate concentration and clearance by transport also would be expected.
Comparisons between rodent and human brain
These human brain data showing an increase in BHB with fasting are in qualitative agreement with that from rodent brain. Quantitatively, however, the BHB increase in human brain is significantly larger than that reported by Hawkins et al. (1971) in rodent brain. This difference may result from lower transport relative to oxidation in the rodent. Consistent with this possibility is the known small contribution that ketones make to brain fuel consumption (Mans, 1981; Hawkins et al., 1986). Additionally, this difference also may result from the barbiturate anesthetic used in the rodent model (Hawkins et al., 1971), which may decrease steady-state brain ketone levels.
The human measurements of increased brain lactate with fasting also are qualitatively consistent with the increased lactate efflux reported by both Hawkins et al. (1971) and Ruderman et al. (1974). As described earlier, the increased lactate seen in these human data is most likely consistent with displacement of lactate oxidation by ketones without a change in glucose phosphorylation and glycolytic flux. Thus, flow through lactate efflux would be predicted to increase. We anticipate that demonstration of this shift in lactate metabolism toward ketone oxidation can be determined by using 13C-labeled ketones and glucose.
Conclusions
We have found that both brain BHB and lactate increase in fasting. The increased BHB suggests that ketone oxidation is not limited by its transport across the blood–brain barrier, contrary to what has been previously interpreted from rodent and human data. The apparent linear relationship between brain and plasma BHB suggests that exchange through the monocarboxylic acid transporter is rapid, allowing the concentration of plasma BHB to affect brain concentrations through mass action. This relationship also may reflect progressive activation of transport with fasting. The increase in lactate concentration with fasting may arise as a consequence of reduced metabolic lactate clearance caused by a shift from pyruvate to ketone oxidation. A continuous production of lactate through glycolysis has been suggested (Pellerin and Magistretti, 1994) to be necessary for clearance of synaptic glutamate under basal and activated levels of neuronal activity. Further studies using dynamic measurements of lactate, BHB, and metabolic rates should reveal the interdependency of ketones and glucose in fueling oxidative metabolism in the fasting human brain in basal and activated states.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Mr. Peter Brown for construction and design of the rf probe.
