Abstract
Patients who have undergone hematopoietic stem cell transplant (HSCT) may experience cognitive impairment that can persist after treatment. Several studies have shown that bright light therapy may improve cognition, potentially due to its effects on the circadian system via brain regions that respond preferentially to light. In this double-blind randomized controlled trial, the efficacy of bright light therapy on cognition was examined in HSCT survivors. Forty-seven HSCT survivors at an urban hospital in the United States were screened for mild cognitive impairment, randomized to either bright white light (BWL) or comparison dim red light (DRL) conditions using a block randomization approach, and instructed to use their assigned light box every morning upon awakening for 30 min for 4 weeks. Assessments occurred at baseline, the end of the second week of the intervention, the end of the intervention, and at follow-up (8 weeks later). The primary outcome was objective cognitive function as measured by a global composite score on neuropsychological tests. Secondary outcomes included cognitive performance in individual domains, self-reported cognitive function, fatigue, sleep and sleep quality, and circadian rhythm robustness. Repeated-measures linear mixed models for both objective and self-reported cognitive function indicated significant main effects for time (ps < 0.05) suggesting significant improvements in both conditions over time. Time by light condition interaction effects were not significant. Models focused on secondary outcomes yielded no significant effects. Both BWL and DRL groups demonstrated significant improvements in objective cognitive and self-reported cognitive function over time, but there was no hypothesized effect of BWL over DRL nor associations with circadian rhythm robustness. Therapeutic effects of both light conditions, practice effects, and/or placebo effects may account for the findings.
Keywords
As survival rates for patients with hematological malignancies have improved, cancer- and treatment-related morbidities have become more salient to survivors (Copelan, 2006). Among hematopoietic stem cell transplant (HSCT) survivors, cancer-related cognitive impairment is an established late effect that can persist after treatment (Buchbinder et al., 2018) and impair quality of life (Wu et al., 2012; Wu et al., 2019). Close to 50% of HSCT patients show vulnerability to cognitive impairment 3 months after transplant (Jones et al., 2013), and 18% to 36% will continue to experience persistent symptoms even 3 years post-transplant (Sharafeldin et al., 2018). Domains of cognitive functioning that can be affected vary depending on the study, but can include learning and memory, executive function, motor function, and psychomotor speed (Jones et al., 2013; Sharafeldin et al., 2018).
Circadian disruption may be a risk factor for cognitive impairment (Musiek et al., 2018; Reid et al., 2011) due to its significant role in the regulation of physiological and behavioral systems important for healthy cognition (Reid et al., 2011). Indeed, circadian disruption has been implicated both as a precursor and consequence of cancer and its treatment (Ancoli-Israel et al., 2014; Roenneberg and Merrow, 2016; Savard et al., 2009). For HSCT patients, this is of particular importance, as hematopoietic stem cell proliferation and differentiation appear to be under circadian control (Méndez-Ferrer et al., 2009) and HSCT requires exposure to intensive chemotherapy regimens that could lead to circadian disruption (Ancoli-Israel et al., 2014).
Light is the strongest zeitgeber of the circadian system due to its effects on the brain, including the suprachiasmatic nucleus, via a non-image forming photoreceptor system (Vandewalle et al., 2009). Brain regions involved in alertness respond preferentially to light in the blue light range (~480 nm; LeGates et al., 2014). Hence, light influences alertness, sleepiness, and arousal—all important drivers of cognition—through the circadian system (Cajochen et al., 2014; Smolders and de Kort, 2014). Indeed, recent evidence indicates that disrupted circadian rhythms may increase the risk of cognitive problems in cancer survivors (Ancoli-Israel et al., 2022). Studies have already demonstrated preliminary efficacy of bright light therapy for improving cognitive performance in healthy and neurological populations (Bersani et al., 2008; Killgore et al., 2020; Yamadera et al., 2000). The dose of light delivered in these studies has ranged from exposures of 30 min to 1 h each morning, with lengths of treatment ranging from 5 days to 6 weeks. Studies in cancer populations have tended to adhere to a more standardized protocol based on the early work of Ancoli-Israel and colleagues, in which they compared morning bright white light (BWL) with a comparison dim red light (DRL) as a treatment to prevent fatigue in breast cancer patients undergoing chemotherapy (Ancoli-Israel et al., 2012). Across numerous studies, morning BWL has shown promise in improving or preventing cancer symptoms that may also be mediated by circadian disruption, such as fatigue (Ancoli-Israel et al., 2012; Johnson et al., 2018; Redd et al., 2014), depressed mood (Valdimarsdottir et al., 2018), and sleep disturbance (Wu et al., 2018), and these effects have, in some cases, been sustained even 3 weeks after light therapy had been terminated (Redd et al., 2014; Wu et al., 2018). It is also important to highlight that BWL may also directly modulate cortical and subcortical brain structures responsible for cognitive skills (Vandewalle et al., 2009). In past studies with cancer patients and survivors, researchers have typically used a dose of 30 min of BWL (compared with a comparison DRL) each morning for 4 weeks.
The efficacy of BWL on cancer-related cognitive impairment is yet to be examined. Hence, in this study, we examined the efficacy of BWL therapy among HSCT patients screened for mild cognitive impairment. As mentioned earlier, a range of cognitive functions are likely to be impaired in HSCT patients. Furthermore, a pilot study by Ancoli-Israel and colleagues (unpublished), conducted as part of a larger scale study focused on testing the effect of BWL therapy on fatigue, sleep, and circadian rhythms (Ancoli-Israel et al., 2012), found that breast cancer patients undergoing chemotherapy experienced improvements in global cognition when they received BWL compared with DRL. Therefore, the primary outcome for our study was objective cognitive functioning (i.e., a global composite score of overall cognitive function). Effects on cognitive performance in individual domains, self-reported cognitive functioning, fatigue, sleep and sleep quality, and circadian activity rhythms were also explored.
Materials and Methods
Procedures
This was a double-blind randomized controlled trial (RCT) that compared BWL with a comparison DRL condition in HSCT survivors. Study approval was obtained from Northwestern University’s Institutional Review Board (#STU00201700-CR0003). Potential participants were approached either in-person at a pre-scheduled clinic appointment at Northwestern Memorial Hospital or (e)mailed a recruitment letter with a study brochure attached. Recruitment lasted from May 2016 to July 2019. Written informed consent was obtained from all individual participants included in the study. Screenings were conducted by telephone or in-person. Participants who met eligibility criteria were then randomized by the principal investigator to receive 1 of 2 types of light boxes. All boxes were labeled with 1 of 2 codes representing DRL or BWL and only known to a researcher outside the immediate lab team. Outcome measures were assessed at baseline (pre-intervention), the end of the second week of the intervention (for self-report measures only), the end of the 4-week intervention, and 8 weeks post-intervention. If the end-of-intervention assessment could not occur immediately after the intervention, the participant was encouraged to continue the intervention until their appointment so that there would be no time gap between intervention completion and follow-up assessment. Neuropsychological assessments were conducted in-person at Northwestern University’s Department of Medical Social Sciences. Questionnaires were administered via REDCap in-person or online at home if participants had time constraints (Harris et al., 2009). Participants wore an Actiwatch (Spectrum Plus, Philips Respironics, Bend, OR) and completed sleep logs for 72 consecutive hours at each time point.
Participants
To be included, participants needed to (1) be older than 18 at screening and at least 16 at the time of HSCT; (2) be 1 to 5 years post-HSCT for a hematological malignancy; (3) stable clinical status (i.e., complete, very good, or partial response); (4) mild (grades 1-2) or no graft-versus-host disease (GvHD); (5) have English language proficiency; (6) have Internet access; and (7) demonstrate scores greater than 3 (“quite a bit”) or 4 (“very much”) on one or both cognitive questions of the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ C-30; Aaronson et al., 1993). The inclusion criteria was expanded to include allogeneic HSCT recipients part way through the study due to the slow pace of recruitment.
Participants were excluded if they met any of the following criteria: (1) pregnancy; (2) confounding medical or psychological diagnosis; (3) visual, hearing, or physical impairment sufficient to interfere with participation; (4) history of brain surgery; (5) history of bipolar disease/mania; (6) history of seasonal affective disorder; (7) current sleep disorder; (8) prior use of light therapy; (9) use of photosensitizing medications; (10) plans to travel across meridians; or (11) employment in shift work.
Apparatus
Light Box
Litebook 1.2 (Litebook, Ltd. Medicine Hat, Canada) was used to deliver BWL and DRL. The BWL Litebook employs 60 premium white light emitting diode (LED) lights that contain emitters with a spectral emission peak at 464 nm and fluorescent phosphors which provide a secondary spectral peak at approximately 564 nm at a distance of 20 inches. The LEDs are contained in a 3.875 × 3 inch elliptical display and collectively emit light that appears white (Desan et al., 2007). The DRL Litebook used as a comparison light is identical in appearance and dimensions to the BWL Litebook, with the exception that it contains red LED lights emitting ~50 lux with peak wavelength at 680 nm (Wu et al., 2018). Circadian photoreceptors are less sensitive to the red light frequency (Lee et al., 1997). Participants were instructed to place the Litebook at a 45° angle 18 to 20 inches from the face for 30 min upon awakening every morning for 4 weeks, and to write down when they used it each day. A HOBO data logger (Onset, MA) tracked when the device was turned on and off.
Actiwatch
The Actiwatch Spectrum Plus (Philips Respironics, Bend, OR) was used for circadian activity rhythm assessment. The Actiwatch has a sampling rate of 32 Hz. Parameters were set to a 1 min epoch length, medium sensitivity, and activity and marker selections were on the default settings. Participants were instructed to wear the Actiwatch continuously on the non-dominant wrist for 72 h and to press the event marker at bedtime and wake time. Participants completed a sleep log (adapted from the consensus sleep diary; Carney et al., 2012) for each night of actigraphy. These data were used to edit actigraphy data.
Measures
Objective Cognitive Function
Objective cognitive function was assessed with paper-and-pencil neuropsychological tests of the domains found to be impaired in HSCT patients (Jones et al., 2013; Sharafeldin et al., 2018) as well as tests recommended by the International Cognition and Cancer Task Force (Wefel et al., 2011): reaction time (Psychomotor Vigilance Task [Khitrov et al., 2013]); simple attention (Conners Continuous Performance Task 3rd Edition [Conners and Sitarenios, 2011]); processing speed (Trail-making Test A [Reitan, 1958], Wechsler Adult Intelligence Scale–Fourth Edition [WAIS-IV] Coding [Wechsler, 2008], Delis-Kaplan Executive Function System [D-KEFS]—Color-word Inhibition: Color Name and Word Reading Time [Delis et al., 2004]); working memory (WAIS-IV Digit Span [Wechsler, 2008]); verbal fluency (D-KEFS: Verbal Fluency [Delis et al., 2004]); visuospatial construction (WAIS-IV Block Design [Wechsler, 2008]); verbal memory (Hopkins Verbal Learning Test–Revised [HVLT-R; Benedict et al., 1998]); visual memory (Brief Visuospatial Memory Test–Revised [BVMT-R; Benedict et al., 1996]); and executive functioning (Trail-making Test B [Reitan, 1958]), D-KEFS Color-word Inhibition: Combined [Delis et al., 2004]). Alternate forms were used at each time point for HVLT-R and BVMT-R. Global composite and individual domain z-scores were calculated as described in Supplementary Materials 1. Premorbid function (Wechsler Test of Adult Reading [WTAR; Wechsler, 2001]) was used to characterize the sample. See Supplementary Table S1 for additional neuropsychological test scoring and domain calculation information.
Self-reported Cognitive Function
The 33-item Patient Assessment of Own Functioning Inventory (PAOFI) is a reliable and valid measure of perceptions of cognitive functioning (Bell et al., 2013; Chelune et al., 1986). Using a Likert-type scale from 1 (almost always) to 6 (almost never), ratings of 1 to 3 were scored “1” indicating impairment, and ratings from 4 to 6 were scored “0” indicating no impairment. Total impairment was calculated by summing the number of impaired items on the Memory, Language and Communication, and Higher Cognitive Functions subscales (Chelune et al., 1986). There are no established clinical cutoffs for this measure, but one study that compared a clinical group with a non-clinical group of 150 participants deemed a score of ≥8 to be clinically significant (i.e., 1 standard deviation above the mean; Richardson-Vejlgaard et al., 2009).
Fatigue
The FACIT-Fatigue scale (Smith et al., 2010) consists of 13 items scored on a Likert-type scale from 0 to 4. Higher scores indicate less fatigue. Scores of <34 indicate clinically significant fatigue (Van Belle et al., 2005).
Sleep Quality
The Pittsburgh Sleep Quality Index (PSQI) is a 19-item self-report measure that is used to assess sleep quality (Buysse et al., 1989). A global score >5 indicates poor sleep quality. Internal consistency for the PSQI is generally good, ranging between 0.70 and 0.83 (Mollayeva et al., 2016).
Actigraphy Sleep Outcomes
The following sleep outcomes were computed at each time point based on actigraphy: total sleep time (in hours), sleep efficiency (the percentage of time in bed when the person is sleeping), and wake time after sleep onset (the amount of nocturnal sleep time when the person is awake).
Circadian Activity Rhythms
Activity was measured with the Actiwatch Spectrum Plus (Philips Respironics, Bend, OR) and analyzed with Philips Actiware software, V9.4 SAS software (SAS Institute Inc., Chicago, IL, USA). Each participant’s activity data were fit to a 5-parameter extended cosine model (Marler et al., 2006) which yielded the pseudo F-statistic, which indicates “goodness of fit” of the fitted function. A larger value indicated more robust rhythms (Liu et al., 2013).
Treatment Credibility and Expectancy
Treatment credibility and outcome expectancy were assessed at baseline with the mean of the first 3 items of the Credibility/Expectancy Questionnaire. Items are scored on a Likert-type scale from 1 to 9, and the term “symptoms” was replaced with “thinking, memory and concentration problems.” Higher scores represent greater credibility and outcome expectancy (Devilly and Borkovec, 2000).
Treatment Satisfaction
The item “How do you rate this treatment overall?” from the Functional Assessment of Chronic Illness Therapy—Treatment Satisfaction–General (FACIT-TS-G) scale was used to measure treatment satisfaction (Peipert et al., 2014). Participants responded on a 5-point Likert-type scale from “poor” to “excellent.”
Power Analysis
As part of a larger study examining the effect of light therapy on fatigue, sleep, and circadian rhythms (Ancoli-Israel et al., 2012), Ancoli-Israel and colleagues administered a battery of tests of objective cognitive function before and after light therapy in 10 participants (BWL n = 5; DRL n = 5). The observed effect size in the pilot data was 0.33, indicating that a sample size of at least 19 (i.e., 10 participants in each group) would be required to achieve power at 0.80 at a significance level of 0.05 (unpublished data).
Data Analyses
Descriptive statistics were used to characterize sociodemographic, clinical, and outcome variables. Group differences were analyzed with independent t-tests (i.e., age, time since diagnosis, time since most recent transplant, WTAR, PAOFI, fatigue, sleep outcomes, pseudo F-statistic, and light box use), chi-square tests (i.e., gender, race, marital status, educational level, employment status, annual household income, diagnosis, and type of transplant), and Fisher’s exact tests (i.e., conditioning regimen and receipt of total body irradiation) using SPSS Statistics 27 (SPSS Inc., Chicago, IL, US). Analyses of outcomes between groups over time were undertaken using repeated-measures linear mixed models (LMMs) using SAS procedure MIXED. A dummy-coded group variable (DRL vs. BWL) was entered as the independent variable to test main effects and a time by group variable to test interaction effects. Effect sizes were calculated using
Results
Participant Characteristics
A total of 47 patients participated in the study (see Figure 1). Participants ranged in age from 28 to 78 years old, time since diagnosis ranged from 1.94 to 19.79 years, and time since most recent HSCT was 1.29 to 5.29 years. Table 1 summarizes other core participant characteristics. Groups did not differ significantly in age, gender, race, relationship status, educational level, employment status, annual household income, diagnosis, time since diagnosis, nor time since most recent transplant.
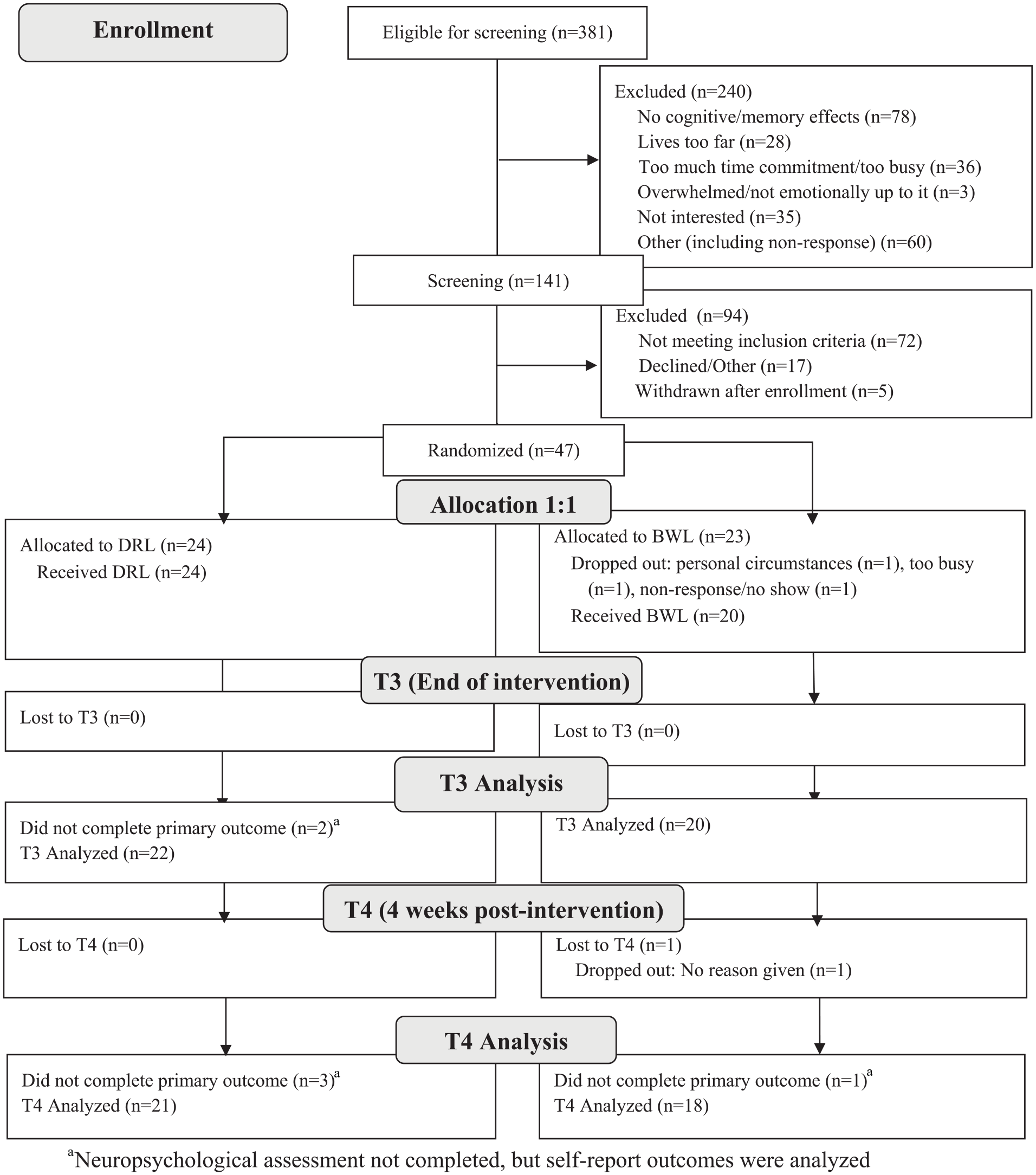
Consort flow diagram. Abbreviations: DRL = dim red light; BWL = bright white light.
Participant characteristics.
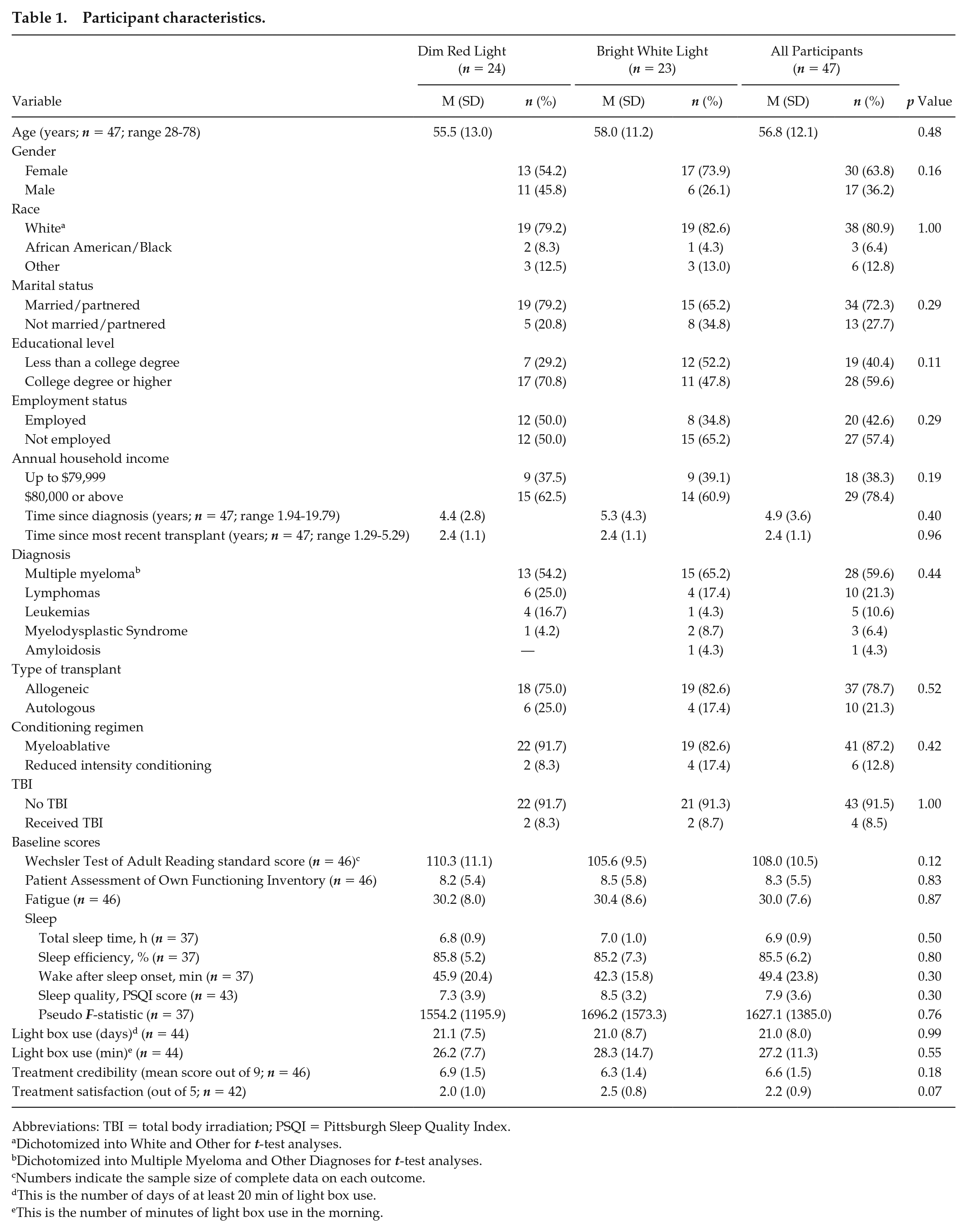
Abbreviations: TBI = total body irradiation; PSQI = Pittsburgh Sleep Quality Index.
Dichotomized into White and Other for t-test analyses.
Dichotomized into Multiple Myeloma and Other Diagnoses for t-test analyses.
Numbers indicate the sample size of complete data on each outcome.
This is the number of days of at least 20 min of light box use.
This is the number of minutes of light box use in the morning.
Objective Cognitive Function
There were no significant differences between groups at baseline in premorbid functioning (WTAR; Table 1). The repeated-measures LMM showed a significant main effect for time, F(2, 77) = 47.43, p < 0.0001, indicating improvement in the global composite score from baseline to follow-up (Figure 2a). This effect was large (
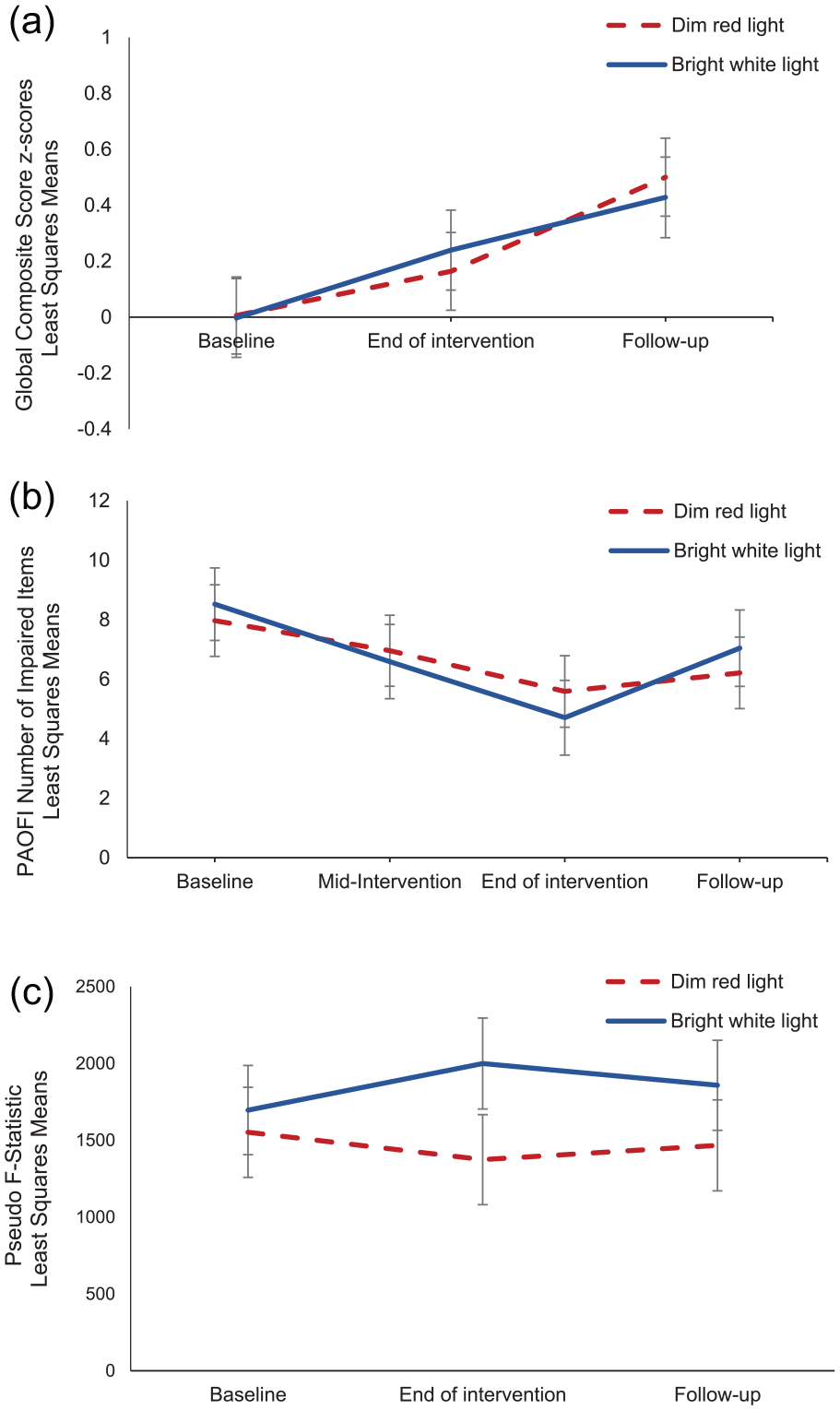
Repeated-measures linear mixed models were undertaken to examine mean changes in primary and secondary outcomes between groups over time. Least squares means and standard error bars are shown for (a) the Global Composite Score (z-scores) for objective cognitive function based on neuropsychological test performance, (b) the number of impaired items on the PAOFI, and (c) the pseudo F-statistic, a measure of circadian activity rhythm robustness, by group over time. Abbreviation: PAOFI = Patient Assessment of Own Functioning Inventory.
Self-reported Cognitive Function
At baseline, participants in both groups on average reported clinically significant cognitive impairment based on the number of impaired items on the PAOFI, but there were no significant differences between groups (Table 1). A repeated-measures LMM showed a significant main effect for time, F(3, 122) = 7.55, p = 0.0001, demonstrating a reduction in the number of impaired items on the PAOFI over time (Figure 2b). This was a large effect (
Fatigue
All participants met the cutoff for clinically significant fatigue (i.e., a score < 34; Van Belle et al., 2005) at baseline and there was no significant difference between groups (Table 1). A repeated-measures LMM indicated no significant main effects for time, F(3, 122) = 2.42, p = 0.07; condition, F(1, 45) = 0.97, p = 0.33; nor time by condition, F(3, 122) = 2.10, p = 0.10.
Sleep and Sleep Quality
There were no significant differences between groups in total sleep time, sleep efficiency, wake after sleep onset, nor sleep quality at baseline (Table 1). Repeated-measures LMM indicated no significant main effects for time, condition, nor time by condition on any of these sleep variables (all ps > 0.50).
Circadian Activity Rhythm Robustness
There was no significant difference between groups on the pseudo F-statistic at baseline (Table 1). A repeated-measures LMM indicated no significant main effect for time, F(2, 65) = 0.10, p = 0.90; light condition, F(1, 36) = 1.03, p = 0.32; nor time by light condition interaction, F(2, 65) = 1.44, p = 0.24 (Figure 2c). Figure 3 shows examples of participant actograms ranging from high to low levels of circadian activity rhythm robustness.
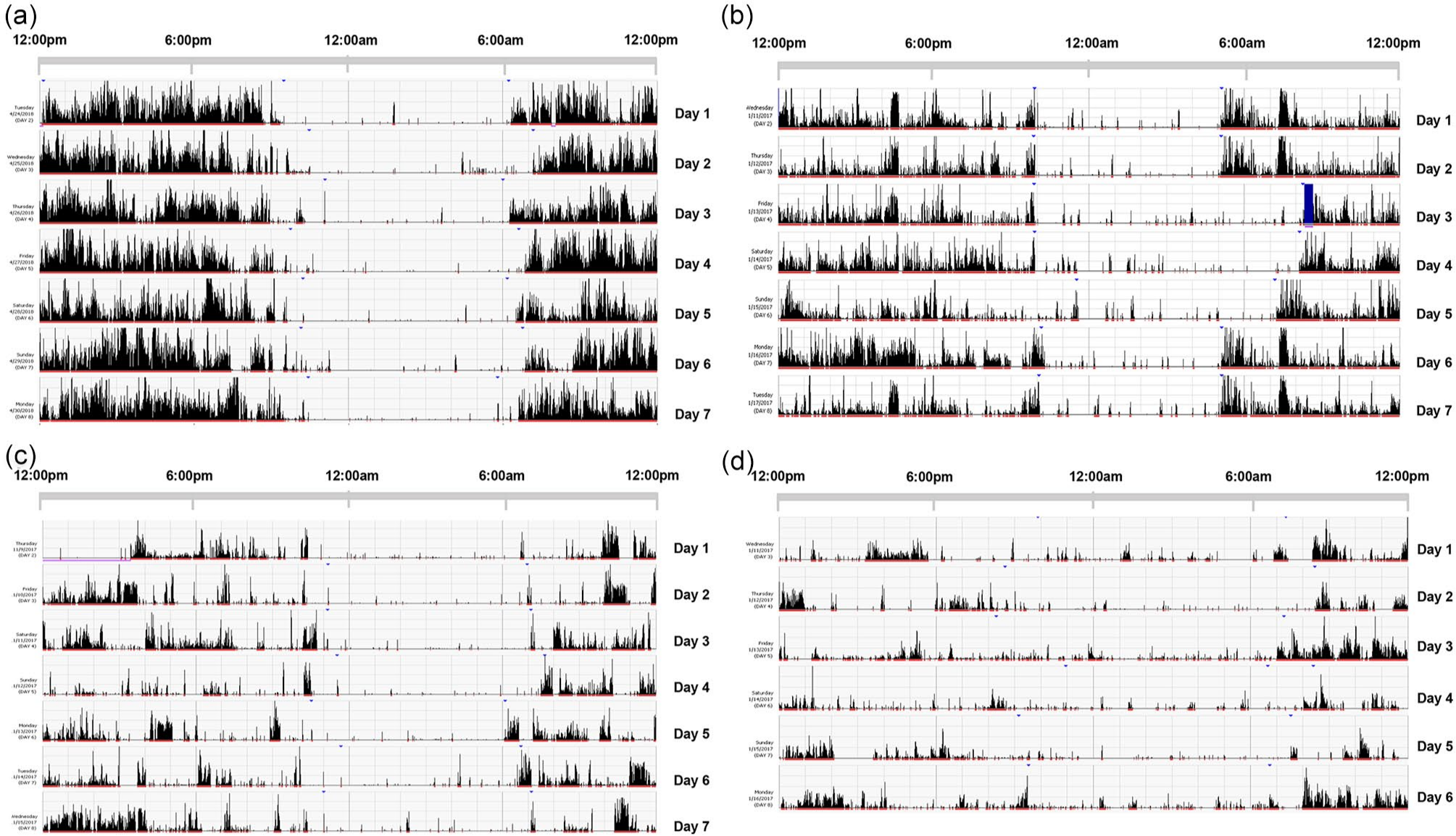
These figures show examples of participant actograms ranging from strong to weak circadian activity rhythms. The pseudo F-statistics are as follows: (a) = 6888, (b) = 1572, (c) = 734, and (d) = 217.
Treatment Credibility, Expectancy, and Satisfaction
Treatment credibility and expectancy did not differ significantly between DRL and BWL participants, but BWL participants were marginally more satisfied with their light box at the end of treatment (p = 0.07; Table 1).
Litebook Use
DRL and BWL participants both used their light box on average 75% of the prescribed 28 days for at least 20 min in the morning hours with no significant difference between groups (p = 0.99). There was also no significant difference between groups in the average number of minutes of morning light box use (p = 0.55; Table 1).
Discussion
To the best of our knowledge, this is the first study to test the efficacy of a 4-week BWL intervention to improve cognitive function in HSCT survivors. HSCT survivors demonstrated significant improvement on tests of cognitive performance and in self-reported cognitive function both during and after the intervention. However, there was no hypothesized effect of BWL over DRL in overall cognitive performance, nor in any individual domain. There are a number of possible explanations for these negative findings. First, the improvements in cognition may have represented real therapeutic effects of both conditions with neither wavelength of light demonstrating superiority over the other. Second, practice effects may have been responsible for the improved performance scores on the neuropsychological tests over time. Practice effects are score increases associated with repeated administration of cognitive tests. Although the use of alternate forms may reduce the magnitude of practice effects, they do not always eliminate them (Calamia et al., 2012). Third, participants may have shown improvement due to placebo effects—the expectancy effects that can increase perceived benefits of a treatment. However, this explanation is more applicable to the self-reported improvements in cognition than improvements on the neuropsychological tests, as “objective” performance on neuropsychological tasks tend to be less malleable to expectancy effects (Schwarz and Büchel, 2015). Fourth, DRL (long wavelength light) has frequently been used in light therapy studies as a standard comparison condition to BWL (short wavelength light) but is by no means a pure control condition. There is evidence to suggest that exposure to longer wavelengths of light can enhance subsequent impact of shorter wavelengths of light on brain responses involved in cognitive functions (Chellappa et al., 2014). Thus, it is possible that exposure to long wavelength light in the morning via DRL could even enhance the impact of any subsequent exposure to long wavelength light, such as from daylight.
When individual cognitive domains were examined, there were significant improvements on verbal memory, processing speed, simple attention, working memory, and visuospatial functioning over time. There was one inexplicable group effect favoring the DRL group in the area of visual memory, although due to the small sample size, further work is needed to determine whether this reflects an actual benefit of DRL on visual memory. In addition, unlike earlier studies (Johnson et al., 2018; Redd et al., 2014), there was no differential effect of BWL on the secondary outcome of fatigue, even though all participants exceeded the clinical cutoff for cancer-related fatigue at baseline. Contrary to earlier work (Wu et al., 2018), there were also no differential effects of BWL on objective sleep outcomes (i.e., total sleep time, sleep efficiency, and wake after sleep onset) and sleep quality, although participants were not screened for sleep problems and, therefore, did not always exceed clinically significant cutoffs (e.g., sleep efficiency was, on average, over 85%).
The robustness of circadian activity rhythms and associations with light condition and cognition were not significant, although it is possible that the sample sizes were too small to detect any effects on this secondary outcome as the study was only powered to detect effects on the primary outcome of cognition.
Strengths and Limitations
This study is characterized by a number of strengths. It is the first known study to examine the use of BWL for the treatment of cognitive impairment in cancer survivors. In addition, circadian activity rhythms were also assessed to determine whether BWL might be associated with this purported mechanism of cognitive change. The study also examined cognitive functioning through the gold standard method of assessment, that is, neuropsychological tests, in addition to a self-report measure of cognition, thus tapping into both important facets of cognition (Hutchinson et al., 2012).
However, there were also limitations. First, participants were not screened for circadian disruption at the outset. If circadian rhythm disturbances are purported to underlie cognitive impairment, then screening for such disturbances might enable identification of individuals most likely to benefit from light therapy. Unfortunately, no established demographically similar normative data nor clinical guidelines exist. We did, however, compare the circadian activity rhythms in our sample with community samples of men and women (Rogers-Soeder et al., 2018; Tranah et al., 2011). Our participants scored in the median range or higher compared with those samples, but were also, on average 19 to 26 years younger, which may account for their more robust circadian rhythms. Thus, we cannot conclude that our participants had circadian activity rhythm disruption from the outset. Second, the screening of participants may have been too lenient, and thus participants may have been insufficiently cognitively impaired to benefit from the intervention. Third, the study would have benefited from a no-light control condition, to better understand what may have led to improvements in cognition in both light conditions. Fourth, the study was not powered to examine the secondary outcomes including individual cognitive domains, fatigue, sleep and sleep quality, and circadian activity rhythms, nor to adjust for multiple comparisons. Fifth, although we excluded patients from the study who had received brain surgery or experienced brain cancer, we could not completely exclude HSCT patients who had received treatment without brain involvement; total body irradiation that also includes the brain, is a common conditioning regimen for some HSCT patients. Finally, there was heterogeneity in the participant sample with respect to clinical factors, such as type of transplant, diagnosis, and conditioning regimen. Having said that, there were no statistically significant differences between groups on sociodemographic or clinical factors.
In conclusion, although cognition improved in both treatment groups, we did not observe a hypothesized effect of bright light exposure on cognitive functions. Further research is necessary to address some of the aforementioned limitations to determine whether bright light therapy may ameliorate cognitive impairment in cancer survivors. Importantly, this study adds to the growing body of light therapy research among cancer survivors and highlights the need for further work in this area, including across different cancer types.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304221107833 – Supplemental material for Examining the Efficacy of Bright Light Therapy on Cognitive Function in Hematopoietic Stem Cell Transplant Survivors
Supplemental material, sj-pdf-1-jbr-10.1177_07487304221107833 for Examining the Efficacy of Bright Light Therapy on Cognitive Function in Hematopoietic Stem Cell Transplant Survivors by Lisa M. Wu, Heiddis B. Valdimarsdottir, Ali Amidi, Kathryn J. Reid, Sonia Ancoli-Israel, Katrin Bovbjerg, Rina S. Fox, Lauren Walker, Amreen Matharu, Erin T. Kaseda, John P. Galvin, Kehinde Adekola, Gary Winkel, Frank Penedo and William H. Redd in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank all of our participants and our research colleague, Diana Buitrago, at Northwestern University Feinberg School of Medicine, without whom this study would not have been possible. This work was supported by the National Cancer Institute (NCI) of the National Institutes of Health (Grant K07CA184145 [PI: L.M.W.]). L.M.W.’s effort was also supported by the European Union Horizon 2020 Research and Innovation Programme under the Marie Sklodowska-Curie grant agreement no. 754513 and the Aarhus University Research Foundation. R.S.F.’s effort was supported by the NCI of the National Institutes of Health (Grant K08CA247973 [PI: R.S.F.]). REDCap is supported at Northwestern University Feinberg School of Medicine by the Northwestern University Clinical and Translational Science (NUCATS) Institute and was supported, in part, by the National Institutes of Health’s National Center for Advancing Translational Sciences (Grant UL1TR001422).
Author Contributions
L.M.W., H.B.V., S.A.-I., G.W., and W.H.R. conceived and designed the study; L.M.W., H.B.V., K.B., L.W., A.M., J.P.G., K.A., and F.P. facilitated set up and data acquisition; data processing and analysis were performed by L.M.W., K.B., K.A., R.S.F., K.J.R., L.W., A.M., and G.W.; interpretation of data was performed by L.M.W., K.J.R., and A.A.; draft manuscript preparation was performed by L.M.W., A.A., K.J.R., S.A.-I., K.B., R.F., L.M.W., A.M., and E.T.K.; all authors revised it critically, reviewed the results, and approved the final version of the manuscript. Content is solely the responsibility of the authors and does not necessarily represent the official view of the National Institutes of Health.
Conflict of Interest Statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.A.-I. is a scientific consultant for Eisai, Idorsia, Merck, and Biogen. There are no other conflicts of interest to report.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
