Abstract
A significant delay in the timing of endogenous circadian rhythms has been associated with delayed sleep phase disorder (DSPD). More recently, other mechanisms have also been proposed to account for this disorder. To further explore the etiology of DSPD, the present study compared nocturnal melatonin profiles of 26 DSPD patients (18 males, 8 females; age, 21.73 ± 4.98 years) and 17 normally timed good sleepers (10 males, 7 females; age, 23.82 ± 5.23 years) in a time-free, dim-light (<10 lux) laboratory environment. A 30-h modified constant routine with alternating 20-min sleep opportunities and 40 min of enforced wakefulness was used to measure the endogenous melatonin circadian rhythm. Salivary melatonin was sampled half-hourly from 1820 h to 0020 h and then hourly from 0120 h to 1620 h. DSPD patients had significantly later timed melatonin profiles that were delayed by approximately 3 h compared to normal sleepers, and there were no notable differences in the relative duration of secretion between groups. However, melatonin secretion between dim-light melatonin onset (DLMO) and acrophase was less prominent in DSPD patients compared to good sleepers, who showed a more acute initial surge of melatonin following the DLMO. Although the regulatory role of melatonin is unknown, abnormal melatonin profiles have been linked to psychiatric and neurological disorders (e.g., major depression, obsessive compulsive disorder, Parkinson disease). These results therefore suggest that in addition to a delayed endogenous circadian rhythm, a diminished initial surge of melatonin secretion following DLMO may contribute to the etiology of DSPD.
Keywords
A delay in circadian rhythm timing is currently assumed to be a significant contributing factor for the etiology of delayed sleep phase disorder (DSPD). By measuring physiological phase markers, such as core body temperature and melatonin, past empirical studies have identified a 2- to 6-h delay in the timing of circadian rhythms of patients with DSPD compared to those who habitually sleep at conventional times (Oren et al., 1995; Ozaki et al., 1996; Saxvig et al., 2013; Shibui et al., 1999; Uchiyama et al., 2000a). These phase markers have been shown to correspond with the sleep/wake pattern, where early sleepers have early timed phase markers and late sleepers have later timed circadian phases (Duffy et al., 2001; Mongrain et al., 2004). Other pathophysiological changes observed in DSPD include an abnormally slower circadian oscillator, as indicated by a longer period length (Campbell and Murphy, 2007; Micic et al., 2013; Regestein and Monk, 1995; Weitzman et al., 1981), and an abnormal phase-response curve to light, including a decreased ability to phase advance from light following the sleep period (Czeisler et al., 1981; Ozaki et al., 1996) and/or an increased phase-delay effect to evening light (Aoki et al., 2001).
Although animal and a few human studies indicate an influence of melatonin secretion on physiological, immunological, medical, and psychiatric disorders, the regulatory role of this hormone is not conclusively established (Bolitho et al., 2014; Karasek and Winczyk, 2006). Delays in melatonin secretion are associated with delays in sleep patterns of DSPD patients, and exogenous melatonin can adjust the timing of circadian clocks (Burgess et al., 2010; Lewy et al., 1992; Lewy et al., 2001). In terms of sleep disorders, effects of overall amounts of nocturnal melatonin secretion and sleep have been researched in sleep disturbances such as insomnia but not in circadian rhythm sleep disorders (CRSDs) (Mahlberg et al., 2006; Myers and Badia, 1995; Zeitzer et al., 1999). Sleep disturbances and low levels of melatonin are highly prevalent in the elderly (Haimov et al., 1994; Zisapel, 2005). However, it is unclear if lower melatonin levels in elderly patients with sleep disorders are observed simply due to a spurious relationship with age (i.e., melatonin decreases with age, and sleep disturbance is more prevalent in elderly) (Mahlberg et al., 2006; Zeitzer et al., 1999). Nevertheless, reductions in melatonin secretion have also been found in young adulthood (i.e., 20-30 years of age), during a period in the life span where DSPD is most prevalent (Kennaway et al., 1999). These findings suggest melatonin secretion factors other than just the timing may affect sleep parameters and lead to sleep disorders. For example, absolute melatonin secretion and the nocturnal profile of secretion may also be linked to sleep. However, this has not been comprehensively researched in DSPD patients and was the aim of the present study.
Melatonin has been investigated in DSPD patients across the sleep period, and significant delays in melatonin phase timing were correlated with the sleep/wake patterns of DSPD patients compared to controls (Shibui et al., 1999). The midpoint of melatonin secretion and dim-light melatonin onset (DLMO) and dim-light melatonin offset (DLMOff) times were delayed in DSPD patients, but the duration of secretion did not differ compared to controls. In a more recent finding, DSPD patients appear to have a slower rate of increase in saliva melatonin levels compared to controls, in addition to the later DLMO (Rahman et al., 2009). However, since the objective in this study was investigated from partial DLMO sampling (i.e., 1900-0300 h), data were not available to determine whether there was an extended period of melatonin secretion following acrophase of melatonin concentration. Furthermore, the apparent slower rate of increase in mean melatonin curves could have arisen by greater variance of DLMO times in the DSPD sample. Therefore, it would be important to further investigate the rate of increase to acrophase, timing of acrophase, and rate of decrease from acrophase. The present study examined differences in overall melatonin secretion profiles between adults diagnosed with DSPD and conventionally timed good sleepers. This should contribute to the limited knowledge currently available about the etiology of the disorders and, in turn, may aid in developing a diagnostic profile for the circadian rhythm disorders and improve treatment effectiveness.
Materials and Methods
Participants
Forty-three participants were selected for a laboratory-based study at the Flinders University Sleep Laboratory. Samples comprised 26 patients diagnosed with DSPD (18 males, 8 females; age, 21.73 ± 4.98 years) and 17 normally timed good sleepers (10 males, 7 females; age, 23.82 ± 5.23 years). There were no significant differences in age, t(41) = 1.32, p = 0.194, or sex, χ2 (1, N = 43) = 1.167, p = 0.280, between the groups. The selection process occurred in 2 phases. Phase 1 screening consisted of a questionnaire battery assessing lifestyle, current sleeping patterns, and sleeping preferences. Phase 2 screening confirmed sleeping patterns using a prospective 7-day sleep diary and actigraphy monitoring. These measures were used to assess DSPD criteria according to the third edition of the International Classification of Sleep Disorders (ICSD-3) (American Academy of Sleep Medicine [AASM], 2014). To confirm eligibility, responses and logs were further analyzed by the senior author (LL), who has more than 20 years of clinical and research experience with CRSDs.
Phase 1 Screening
An initial sample of 497 interested candidates responded to poster advertisements posted at major university campuses in Adelaide, as well as online websites and through word of mouth. Informed consent was obtained from participants prior to their involvement in both the screening and laboratory phases. Ethics approval for the experiment was granted by the Southern Adelaide Clinical Human Research Ethics Committee. All respondents completed screening phase 1 to determine their eligibility as either a normal sleeper or having DSPD. The test battery was administered as an online survey (SurveyMonkey.com) and consisted of a semistructured General Health and Medical Questionnaire interview, Munich Chronotypes Questionanaire (MCTQ) (Roenneberg et al., 2003, 2004; Zavada et al., 2005), Horne and Östberg’s Morningness-Eveningness Questionnaire (MEQ) (Horne and Ostberg, 1976; Zavada et al., 2005), Pittsburgh Sleep Quality Index (PSQI) (Buysee et al., 1989), DSPD–Sleep Timing Questionnaire (DSPD-STQ) developed by the authors, Sheehan Disability Scale (SDS) (Arbuckle et al., 2009; Leon et al., 1997), and Depression Anxiety Stress Scale21 (DASS21) (Antony et al., 1998; Brown et al., 1997; Lovibond and Lovibond, 1995a, 1995b).
Inclusion/exclusion criteria
To meet criteria for DSPD, respondents had to score <42 on the MEQ (i.e., be evening types) (Rahman et al., 2009) and report daytime dysfunction and lifestyle impairments as a result of their sleeping pattern on the SDS. DSPD patients were required to report scores ≥4 (range 0-10) on any given SDS subscale, as this is the cutoff indicative of significant impairment and dysfunction. The overall score and individual responses from the PSQI were used to screen for other sleep disturbances. Otherwise, in line with ICSD-3 criteria, those with DSPD were healthy good sleepers when sleeping at their habitual delayed time (AASM, 2014). Participants had to indicate delayed sleep patterns of at least 2 h in contrast to “conventional” sleep times (i.e., sleep onset later than 0200 h) and who were unable to fall asleep at a preferred earlier time according to the DSPD-STQ (AASM, 2014).
The SDS was used to select normally timed good sleepers and identify the absence of any impairment in functioning at work/school, social life, and family life as a result of their sleep pattern. Normal sleepers indicated little to no sign of distress or daytime dysfunction associated with their sleep patterns (scores ≤2 on all 3 SDS items), a PSQI score <6 (Buysse et al., 1989), conventional sleep times (i.e., sleep onset = 2200-2400 h), and neither morning nor evening type (MEQ score from 42-58). A qualified clinician (LL) confirmed the suitability of good sleepers.
DSPD-STQ
The DSPD-STQ was developed to assess subjective feelings regarding symptoms of DSPD and evaluate circadian misalignment. The authors aimed to tap into patients’ subjective impressions of their current sleep pattern by asking potential patients the following: “Please indicate the time when your ‘body clock’ currently allows you to fall asleep most easily.” “How long do you think it would take you to fall asleep at this time?” “How difficult would you find it to fall asleep at this time?” “Please indicate the time when your ‘body clock’ currently allows you to wake up most easily?” “How difficult do you find it to wake up at this time?” To obtain an impression of circadian misalignment, respondents were then asked to disregard their “body clock” and only consider daytime obligations and social commitments; questions included the following: “Please indicate the time when you would PREFER to fall asleep most easily.” “With your CURRENT sleeping pattern in mind, how long do you think it would take you to fall asleep at this PREFERRED time?” “With your CURRENT sleeping pattern in mind, how difficult do you think it would be to fall asleep at this PREFERRED time?” A single measure of motivation was also assessed by asking patients to “please indicate how motivated you are to change the timing of your CURRENT sleeping pattern to your PREFERRED.” Questions regarding difficulty to fall asleep/wake up were measured on a 5-point Likert-type scale ranging from 0 = not at all to 4 = extremely.
Participants were not on medication (i.e., sleep/over-the-counter [OTC] medication; herbal remedies likely to affect sleep, alertness, and melatonin and circadian rhythms). Body mass index (BMI) was not outside normal range of <18 and >32 kg/m2. Participants smoked <1 cigarette/day, consumed an average of <250 mg caffeine/day, and averaged <14 standard drinks/week of alcohol consumption. Exclusion criteria also included history of substance abuse in the past 6 months, pregnancy or lactation, and transmeridian travel (≥2 time zones) in the past 2 months. Suitable candidates indicated no psychiatric disorders in the past 12 months and night shift work in the past 2 months. Female participants were either in the follicular phase of their menstrual cycle during in-laboratory data collection or used a form of hormonal control (i.e., Etonogestrel implant or the contraceptive pill). Upon completion of the study, participants received monetary compensation proportional to the time spent in the study and AUD$500 for full involvement.
Phase 2 Screening
During a week of screening and an additional week before the laboratory phase, participants kept a sleep/wake diary and wore a Mini Mitter Actiwatch, AW64 (Philips Respironics, Pensacola, FL) activity motion monitor. Actiware 5 software was used to download and process recorded data that were used to assess diligence in keeping the sleep diary. Recorded actigraphy data were collected in 30-sec epochs and analyzed both manually, to insert the “rest” interval, and then with the Actiware sleep scoring algorithm. The starting point of the “rest” interval was set at the time of reduced activity by visually inspecting actigraphy data in conjunction with the sleep diary. The ending point was set at the time of increased activity where it coincided with reports on the subjective sleep diary. Measures obtained from actigraphy analyses included bedtime, get-up time, time in bed, total sleep time, onset latency, sleep efficiency, and wake after sleep onset. These measurements were further used to confirm the sleep pattern of potential candidates and monitor compliance with a regular sleep/wake schedule before participation in the laboratory session.
Outcome Measures
Melatonin radioimmunoassay
Saliva was collected using Salivettes (cat. 51.1534; Sarstedt Australia Pty. Ltd., Mawson Lakes, South Australia) while the subjects lay in a supine position in bed, in dim light (<10 lux), to minimize the masking effects of physical movement and light on endogenous melatonin production. Food and water were consumed only after saliva collection to reduce contamination or dilution of the sample. Participants were instructed to place the swab in their mouth and accumulate saliva for 2 min. After collection, samples were stored frozen at −20 °C. For analysis, samples were thawed and centrifuged for 10 min at 2500 rpm, the swabs removed from the casing, and the supernatant retained. A sensitive (4.3 pM) direct radioimmunoassay (RIA) using reagents from Buhlmann Laboratories AG (Allschwil, Switzerland) (Voultsios et al., 1997) was used to measure melatonin in the saliva. Intra-assay coefficient of variation was always <10%. Interassay coefficient of variation was 15% at 100 pM.
Experimental Procedure
This study used the first 30 h of a larger 80-h modified-constant routine study and involved saliva collection between 1820 h and 1620 h the following day. Samples were collected half-hourly between 1800 h and 0000 h and hourly thereafter. Laboratory sessions occurred between April and August 2012 to 2014 (i.e., Southern Hemisphere winter time). The entire 80-h experiment commenced at 1800 h on Thursday afternoons and ceased at 0200 h on Monday mornings. This schedule was standardized across every experimental session. All participants attended the Flinders University Sleep Laboratory for a 1-h meeting 1 week prior to their scheduled laboratory session to familiarize themselves with the environment they would occupy during the experimental session and the procedures of the protocol (e.g., their rooms, snacking, salivettes, and saliva collection).
Participants were instructed to maintain their typical sleep pattern during the 7 days prior to their assigned laboratory session. Twenty-four hours ahead of the study, participants were asked to refrain from strenuous exercise, napping, or consumption of alcoholic or caffeinated beverages. Compliance to these instructions was monitored by a 7-day sleep/wake diary and actigraphy, provided at the familiarization meeting. On the day of admission to the laboratory, participants were instructed to have their last meal at 1300 h and attend before 1600 h. Consumption of cheese, bananas, chocolate, and tomatoes prior to commencement of and during the laboratory session was not allowed since their intake has been said to perturb spontaneous release of salivary melatonin (Peuhkuri et al., 2012; Voultsios et al., 1997).
Upon arrival, participants were familiarized and set up for the experimental procedures, shown to their separate bedrooms at 1730 h, and permitted to habituate to their new environment but were instructed to remain awake. Two or 3 participants typically resided in the sleep laboratory for any one laboratory session. The room remained in almost complete darkness (i.e., <1 lux) during sleep opportunities and was dimly lit at a maximum of 10 lux at the participant’s head position during enforced wakefulness. A constant room temperature of 20 °C was maintained across the protocol.
Laboratory sessions
The protocol formally commenced with a 20-min sleep opportunity at 1800 h. Each hour, these 20-min sleep opportunities alternated with 40 min of enforced wakefulness until the conclusion of the experiment, to simulate sequential 1-h “days.” During their laboratory stay, participants followed a 1-h ultradian routine while residing in a sound-attenuated room, free from zeitgebers (i.e., no time indicators such as windows, mobile phones, radios, TV, and clocks). While in their room, participants were required to remain in bed and maintain a near-supine position, with head and shoulders sufficiently elevated to enable ease of reading, writing, and so on. This postural position was also maintained during sleep opportunities. Toilet breaks were the only exception when participants were allowed to leave the bed and use normal toilet facilities. Basic hygiene activities such as toilet opportunities and brushing teeth (with water only) were kept to a minimum and permitted only when necessary. These occurred near the end of enforced wakefulness intervals, once saliva samples had been collected. During the 40-min wakeful intervals between sleep opportunities, participants were allowed to engage in sedentary activities such as reading, studying, listening to music, watching DVDs, and having casual conversations with researchers to remain awake and occupy free time.
This environment eliminated the influence of time cues on feelings of sleepiness and alertness. Participants were blind to the duration of sleep and wakefulness periods to further ensure their states of sleepiness remained unaffected by external factors or estimates of clock time. Saliva samples were collected immediately after sleep opportunities (i.e., at 20 min past the hour) and again 50 min past the hour, between 1620 h and 0020 h.
Every second hour, participants were given an opportunity to drink 200 mL of water and consumed a small equi-caloric meal. Snack opportunities occurred 30 min past the hour and 10 min after initial saliva sample collection.
Statistical Analysis
Since salivary samples were assayed half-hourly between 1820 h and 0020 h and hourly thereafter, intermediate half-hourly data points were missing in the latter part of the melatonin curve. As such, individual missing values were added by mathematical linear interpolation between the previous and subsequent hourly values. The same method was then implemented to identify individual DLMO and DLMOff times. DLMO/DLMOff was determined by linear interpolation, where the corresponding x-coordinate (“time”) matched a y-coordinate of 10 pM (Voultsios et al., 1997) or 25% of maximum overall concentration for those individuals (n = 4) with low absolute levels (e.g., <40 pM for peak melatonin value). Timings of overall melatonin profiles were then adjusted and aligned to onset times for both normal sleepers and those with DSPD. Data are presented as the mean and standard error in all figures. In tables, means and standard deviations, as well as mean differences with complementary standard errors between groups, are reported. Where appropriate, a repeated-measures analysis of variance (ANOVA) was used and between-group differences compared using t tests. Cohen’s d values (Cohen, 1988) are also provided to indicate size of effects.
Results
Table 1 shows the sleep characteristics, including subjective and objective habitual sleep parameters, sleep quality, sleep preferences, and daytime impairments, related to the sleeping pattern of the DSPD subjects and normal sleepers. Scores on these variables were obtained during phase 1 screening of the study. This information confirms the validity of the selection process with significant sleep pattern delays observed in the DSPD patients. The table also illustrates a significant lifestyle disruption in the DSPD patients as well as an earlier desired sleep period.
Group Differences between DSPD Patients and Normal Sleepers in Current Estimated Sleep Timing, Sleep Quality, and Associated Impairments Derived from Prescreening Measures.
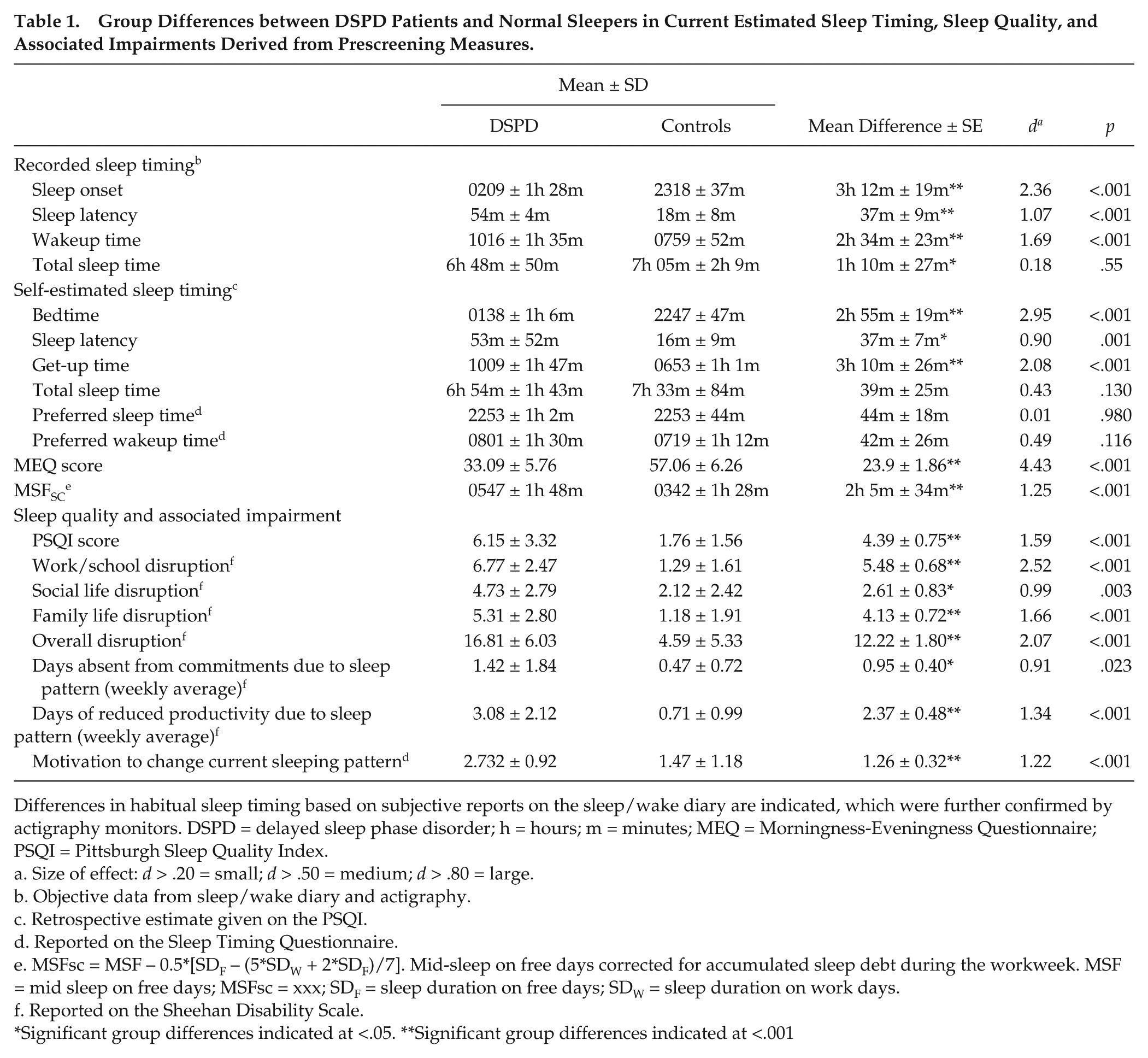
Differences in habitual sleep timing based on subjective reports on the sleep/wake diary are indicated, which were further confirmed by actigraphy monitors. DSPD = delayed sleep phase disorder; h = hours; m = minutes; MEQ = Morningness-Eveningness Questionnaire; PSQI = Pittsburgh Sleep Quality Index.
Size of effect: d > .20 = small; d > .50 = medium; d > .80 = large.
Objective data from sleep/wake diary and actigraphy.
Retrospective estimate given on the PSQI.
Reported on the Sleep Timing Questionnaire.
MSFsc = MSF − 0.5*[SDF – (5*SDW + 2*SDF)/7]. Mid-sleep on free days corrected for accumulated sleep debt during the workweek. MSF = mid sleep on free days; MSFsc = xxx; SDF = sleep duration on free days; SDW = sleep duration on work days.
Reported on the Sheehan Disability Scale.
Significant group differences indicated at <.05. **Significant group differences indicated at <.001
Melatonin Profiles
Melatonin levels were less than the detection threshold (4.3 pM) in DSPD patients and controls prior to 1820 h and after 1520 h the next day. The mean onset of melatonin secretion (DLMO) in the normal sleepers occurred at 2054 h ± 1h 27m, while for DSPD patients, it was delayed by approximately 3 h (2359 h ± 2h 3m; p < 0.001; Figure 1). In line with previous studies using healthy sleeping participants (Burgess et al., 2003; Burgess and Eastman, 2005), DLMO preceded sleep onset by 2h 38m ± 1h 39m in controls and 3h 26m ± 1h 45m in DSPD patients. Worthy of note, shorter sleep onset–DLMO intervals were typically observed in controls than in patients, but this was not a significant difference (p = 0.149).
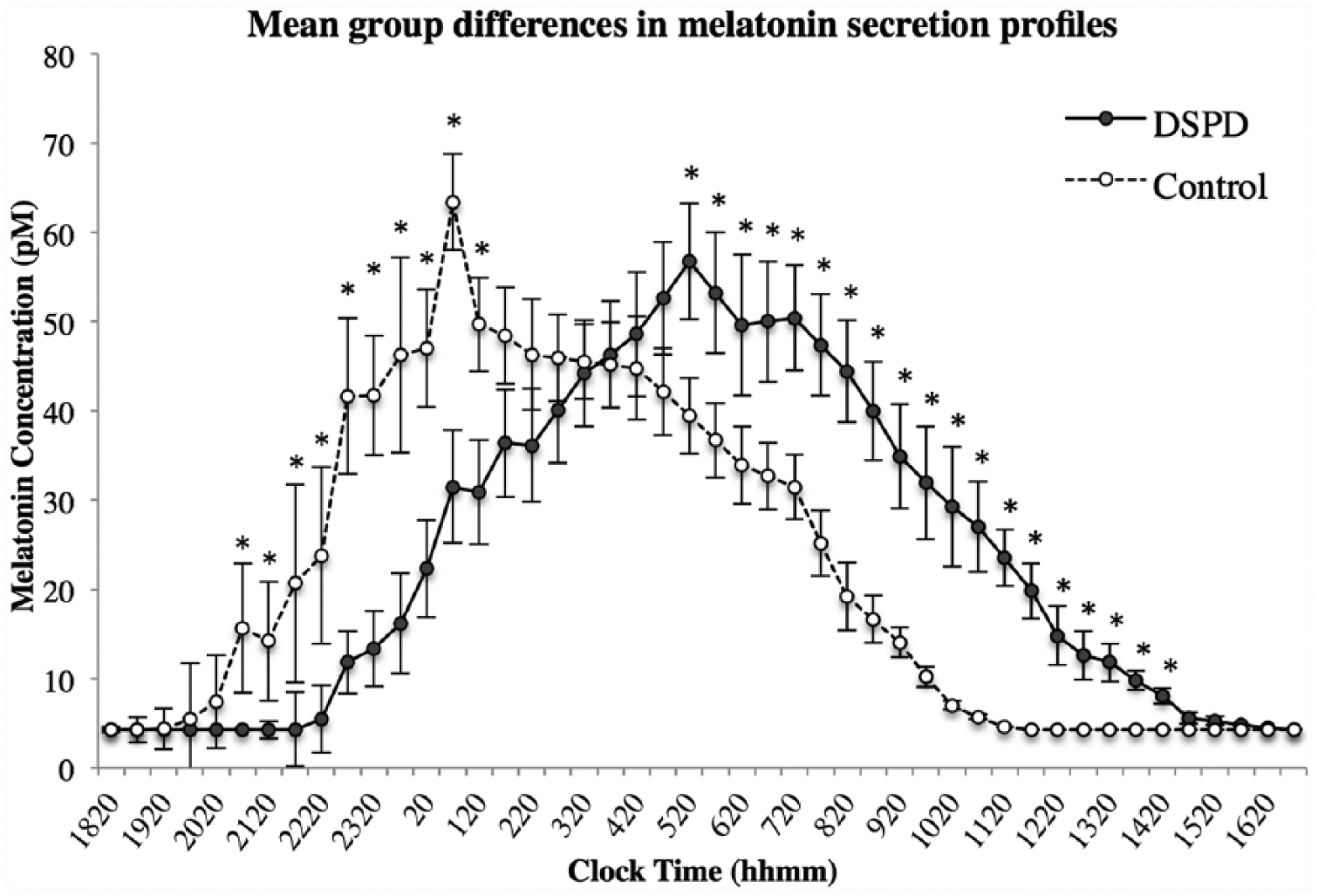
Delayed sleep phase disorder (DSPD) and control group differences in melatonin secretion at half-hourly intervals during the first 22 h of data collection. Significant differences (p < 0.05, 2-tailed) in melatonin secretion between groups are indicated by *, and error bars denote standard error values.
Differences of overall melatonin secretion at half-hourly clock time intervals were compared between groups using independent samples t tests. 1 Figure 1 illustrates significant differences in melatonin amounts during the rising phases and descending phases of the profiles. These differences between the groups are most likely due simply to the delay of the DSPD curve with respect to the normal sleepers. Visual inspection of the profiles suggested that the rate of melatonin increase in the DSPD subjects was less than in the controls. However, this reduced slope could be due to greater variability of the timing of the DLMOs in the DSPD group and thus the mean curve being less representative of individual profiles in the DSPD group.
To control for the circadian phase difference between groups and individuals, the profiles were therefore replotted relative to the DLMO for each group, as shown in Figure 2. Since the variability in total melatonin production may also affect results, these mean curves of absolute melatonin concentration were replotted, controlling for total melatonin production. The area under the curve (AUC) was calculated for each individual and the following formula used to establish percentages: (half-hourly concentration value/AUC) * 100. These percentages were then averaged for each group (see Figure 3). Differences between groups at each time point were again compared with independent samples t tests. After this correction for total melatonin production, the melatonin profiles of DSPD and control participants differed 1.5 to 5 h following DLMO. Bonferroni corrections were not used in this analysis since differences between profiles cluster only in a particular section of the nocturnal profile, with consecutive time points during this section serving effectively as replication studies.
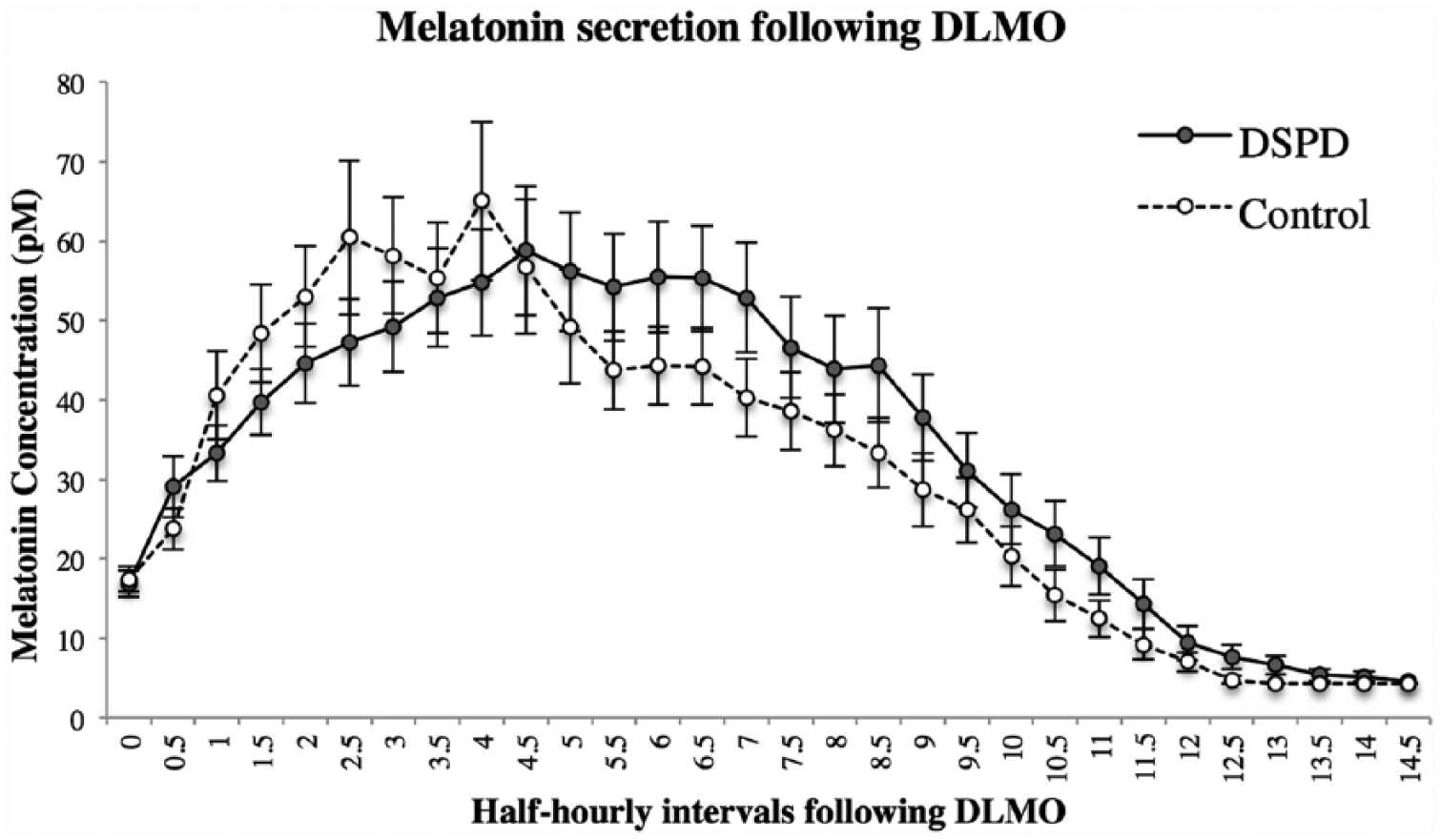
Mean melatonin secretion from dim-light melatonin onset (DLMO) between good sleepers and those with delayed sleep phase disorder (DSPD). Profiles are aligned relative to individual times of DLMO; error bars denote standard error values.
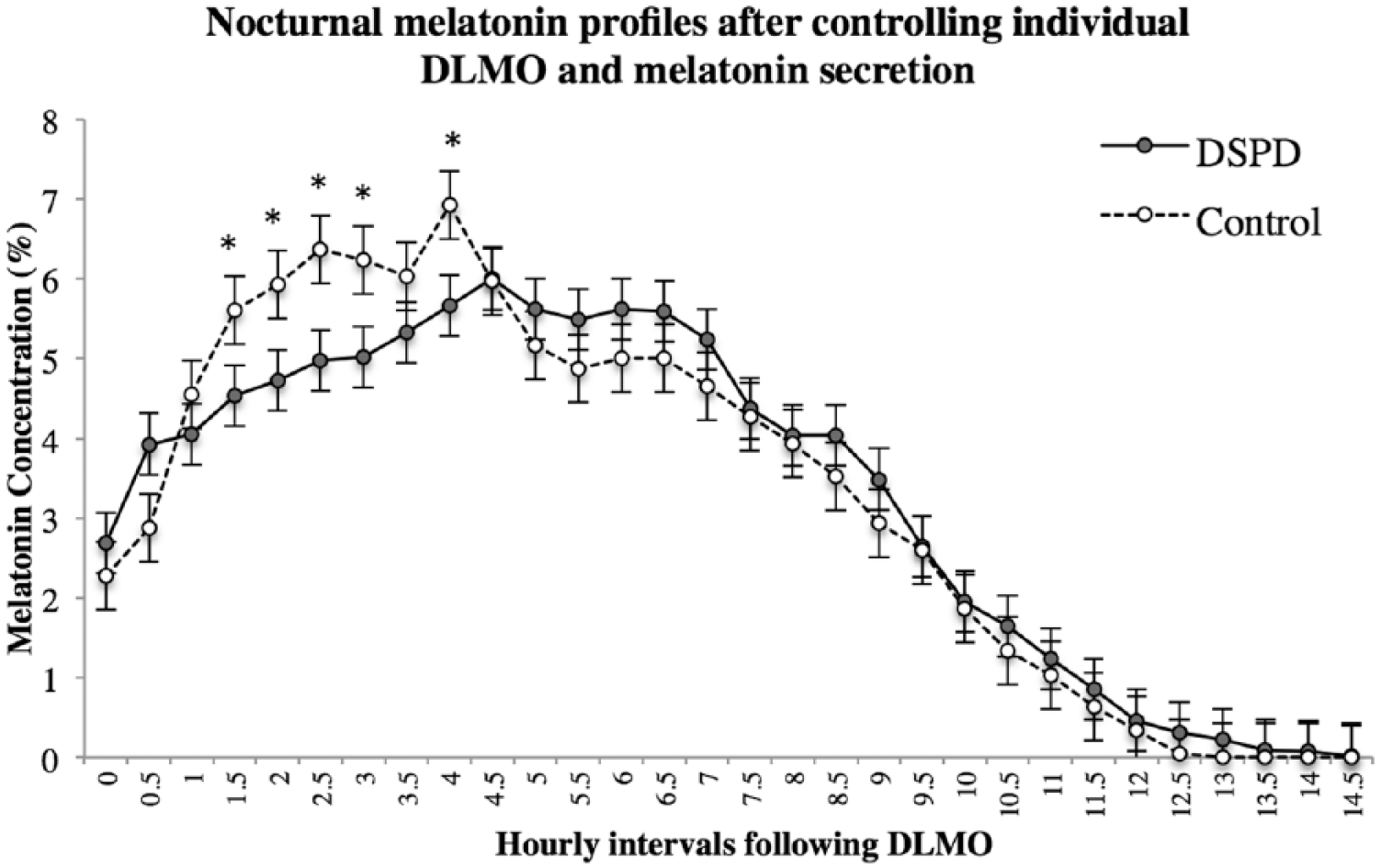
Melatonin secretion relative to overall amount of melatonin produced by individual participants. Percentages were calculated to control for variability in total melatonin secretion: (half-hourly concentration value/AUC) * 100. Profiles are aligned at half-hourly intervals from times of dim light melatonin onset (DLMO). Significant differences (p < 0.05, 2-tailed) in percentage of melatonin secretion between good sleepers and those with delayed sleep phase disorder (DSPD) are indicated by *, and error bars denote standard error values. The melatonin profiles differed between groups 1.5 to 5 h following DLMO.
More global differences in melatonin profiles between those with DSPD and normal sleepers were compared using a series of independent samples t tests. The profile characteristics that were compared are presented in Table 2. Duration was calculated between DLMO and DLMOff. Although there was a tendency for those with DSPD to show a relatively later peak of melatonin secretion, they secreted similar concentrations of melatonin before and after the acrophase of the profile, while normal sleepers secreted more melatonin in the first half of the night. Since the between-group differences occurred predominantly in the upslope of the melatonin profile, a measure of slope was also calculated by dividing the AUC during this time by the number of hours lapsed between individuals’ DLMO and peak (pM/h). Results suggest a trend for those with DSPD to have a slower rate of melatonin increase per hour, which, although statistically nonsignificant, showed a medium to large effect size according to Cohen’s d benchmarks. The AUC between DLMO and the fifth hour was also tested between groups since predominant differences are found during this period. This difference showed significantly less melatonin secretion of the DSPD group in the first 5 h following DLMO.
Group Differences in Melatonin Profiles of Those with DSPD and Good Sleepers.
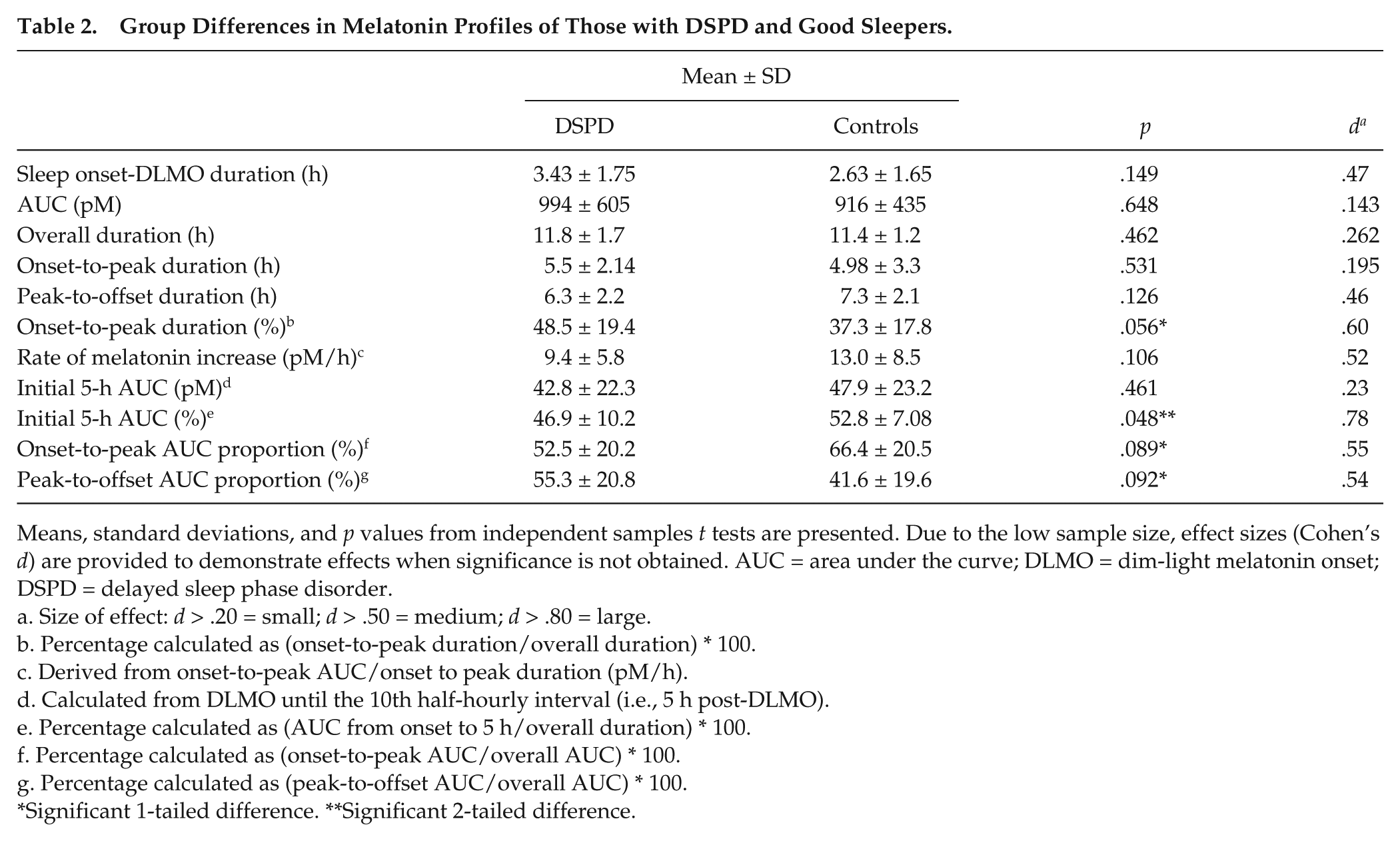
Means, standard deviations, and p values from independent samples t tests are presented. Due to the low sample size, effect sizes (Cohen’s d) are provided to demonstrate effects when significance is not obtained. AUC = area under the curve; DLMO = dim-light melatonin onset; DSPD = delayed sleep phase disorder.
Size of effect: d > .20 = small; d > .50 = medium; d > .80 = large.
Percentage calculated as (onset-to-peak duration/overall duration) * 100.
Derived from onset-to-peak AUC/onset to peak duration (pM/h).
Calculated from DLMO until the 10th half-hourly interval (i.e., 5 h post-DLMO).
Percentage calculated as (AUC from onset to 5 h/overall duration) * 100.
Percentage calculated as (onset-to-peak AUC/overall AUC) * 100.
Percentage calculated as (peak-to-offset AUC/overall AUC) * 100.
Significant 1-tailed difference. **Significant 2-tailed difference.
Discussion
This investigation explored differences in overnight melatonin profiles between adults with DSPD and normal sleepers to contribute further to the currently limited understanding of the DSPD etiology. Apparent in a previous study (Rahman et al., 2009), we expected to find a slower increase of melatonin to acrophase and an overall longer duration of melatonin secretion in those with DSPD. In our study, there was a significant delay in the melatonin secretion profiles of the DSPD patients compared to normal sleepers. Patients also showed a slower increase of melatonin from DLMO to acrophase, as evidenced by less absolute and proportional melatonin in the early part of the melatonin profile (1-5 h following DLMO). Derived profiles depict the robust nature of melatonin production in the early stages of the evening for normal sleepers compared to those with DSPD.
It has been established that the normal circadian period length (tau) is approximately 24.2 h long (i.e., 0.2 h longer than a 24-h solar day) (Burgess and Eastman, 2008; Czeisler et al., 1999; Micic et al., 2014). Therefore, most individuals would have a mild tendency to phase delay if entraining cues are absent. The melatonin profile we have observed in the normal sleepers may favor the stability of their circadian rhythm. Previous empirical studies suggest that doses of exogenous melatonin have phase-shifting effects when administration is timed appropriately relative to the melatonin phase-response curve (Burgess et al., 2010; Lewy et al., 1992). When administered 1 to 9 h before DLMO or at approximately from 1200 h to 2000 h, exogenous melatonin can have phase-advancing effects. There is also recent evidence that the peak phase advance in the phase-response curve is later for lower doses of melatonin and approaches the timing of the DLMO (Burgess et al., 2010). An extrapolation of this trend would suggest that even increases in physiologic concentrations of melatonin immediately following DLMO may have a small phase-advancing effect. Therefore, an early robust endogenous melatonin surge may facilitate a small daily advance in the circadian timing system and help to counter an average tendency to phase delay by 0.2 h each day. Patients with DSPD with a diminished initial surge of melatonin may not have this “protective mechanism,” leading to a tendency toward phase delay. This could also increase the difficulty with phase advancing and remaining stabilized to an earlier sleep time, even after treatment using evidence-based therapies (i.e., chronotherapy, melatonin, and bright-light therapy) (Alvarez et al., 1992).
Uchiyama et al. (2000b) challenged the notion that DSPD was merely a circadian pacemaker timing disorder. Similar to the present study, they employed a 10/20-min sleep/wake ultradian protocol in an attempt to explain patients’ inability to reset the sleep phase. Following 24 h of sleep deprivation, they determined sleep phase by sampling hourly plasma melatonin and measured relative half-hourly sleep propensity via polysomnograph recordings during 10-min sleep opportunities. While sleep-deprived controls were able to compensate for sleep loss at most phases of the circadian pacemaker, DSPD patients were unable to sleep until a significantly later time in their melatonin profiles. Uchiyama et al. (2000b) concluded that DSPD patients had a poor ability to compensate for prior sleep loss, which could imply that the delay in DSPD sleeping patterns is due to a deficit in the accumulation of homeostatic sleep drive across time, thus contributing to delays of bedtimes, later light exposure, and stronger tendency to phase delay. A blunted initial melatonin secretion profile may also be contributing to the apparent inability to initiate sleep early.
The findings of the present study give further support to mechanisms other than circadian rhythm delays for the etiology of DSPD. To the authors’ knowledge, 3 separate studies have thus far assessed melatonin between DSPD patients and normal controls to enable profile comparisons (Rahman et al., 2009; Shibui et al., 1999; Uchiyama et al., 2000b). However, none have directly assessed relative amounts of melatonin secretion between groups at circadian phase–aligned time points. By visual examination, it appears that the presented figures illustrating melatonin curve differences across groups, in all 3 studies, show a similar blunted initial melatonin release for DSPD patients in comparison with the profiles of good sleepers. Nonetheless, since their aims were different from those of the present study, none ran specific analyses to assess whether these differences in profile shapes were statistically significant. If these observations are consistent with the significant results of the present investigation, it may explain the poor compensatory mechanism in sleep propensity of DSPD suggested by Uchiyama et al. (2000b). Hypothetically, the lack of robustness in the DSPD melatonin profiles during the first half of the curve may reduce feelings of sleepiness and sleep propensity in DSPD. This, in turn, may delay bedtime and artifactually increase phase delaying light before bedtime and delay exposure to light in the morning. Therefore, the less robust early melatonin secretion pattern in DSPD may not only reduce protection against the tendency to phase delay but also lead to greater phase delay from the phase-delaying effects of later evening and early morning light exposure.
Likewise, previous studies have alluded to discrepancies in behavioral tendencies that may be delaying sleeping patterns of those with DSPD. Evening types have been found to have a significantly delayed circadian rhythm of subjective sleepiness compared to morning-type sleepers. A study by Lack et al. (2009) reported that evening types had a delayed endogenous circadian rhythm occurring 2 to 3 h later compared to morning types. However, the subjective sleepiness rhythm was delayed by 7 h in evening types as opposed to morning types. Lack and associates reasoned that, since subjective sleepiness rhythms were more delayed than circadian rhythm differences in people with DSPD, circadian rhythms simply follow lifestyle choices driven by subjective sleepiness and subsequent differential exposure to time cues such as dark-light cycles. Taking into account the results of the present study, it would be interesting to see if evening types (considered susceptible to the development of DSPD) also have a diminished secretion of melatonin in the hours immediately following melatonin onset.
There is compelling empirical evidence to support the efficacy of exogenous melatonin in successfully advancing sleep time in patients with DSPD (Lewy et al., 1999). The results of the present study may further advocate this form of intervention to correct patients’ melatonin profiles and sleep pattern by augmenting melatonin amounts in the early part of the evening and sleep period. Melatonin amounts of approximately 1 to 5 mg have been found most effective for the treatment of DSPD, and it is recommended that doses should be kept as low as possible and as early as the patient can tolerate (van Geijlswijk et al., 2010). Furthermore, these authors encourage clinicians to measure circadian timing to determine the most effective phase for administration of exogenous melatonin. Although the current diagnosis of DSPD relies mostly on self-report sleep diaries, this study further highlights the need for objective measures (i.e., actigraphy and/or salivary DLMO) (Rahman et al., 2009; Zee, 2010) to diagnose DSPD and facilitate the most efficacious treatment.
Outcomes of the present study suggest that the mechanism behind DSPD may be multifaceted, and an anomaly in patients’ melatonin profiles may also be contributing to a delay in their circadian system and sleeping patterns. Results suggest that DSPD patients, in comparison with normal controls, show a significant depression in melatonin secretion during the first half of the profile, followed by a melatonin decline not unlike that of the good sleepers after the acrophase. It would seem that a lack of this initial surge of melatonin secretion in the early part of the DSPD night could be making patients vulnerable to circadian delay. These results therefore provide further support for the treatment of DSPD with orally administered melatonin.
Footnotes
Acknowledgements
This project is funded by the Australian Research Council. We thank Flinders University Honours and third-year placement students for their assistance with data collection. Further thank you to Mark Salkeld and the Adelaide Research Assay Facility in the Robinson Research Institute for performing the melatonin assays.
Author Contributions
GM organized and carried out experiments, contributed to data analysis, wrote the manuscript, and assisted with the primary study design. LL and NL contributed to recruitment, organization of all experiments, and data analysis and drafted the manuscript. SAF and HJB also drafted the final paper and, along with LL, designed the overall investigation. MG contributed to data analysis and assisted with the primary study design and drafting the manuscript. DJK contributed to assaying melatonin samples and assisted with data analysis and drafting the manuscript.
Conflict of Interest Statement
LL is a shareholder in Re-time Pty Ltd. MG has performed paid speaking engagements for the Australian Psychological Society and is a shareholder in ResMed. HJB was paid speaker for: SLEEP meeting CME course (2010, 2012); Society of Behavioural Sleep Medicine CME (2011); Ohio State University CME (2011); and University of Chicago CME (2012). SF was supported to give a keynote at Working Time Society, Denmark (June, 2015). DJK has license and royalty agreements with Buhlmann Laboratories for the G280 melatonin antibody.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
