Abstract
Light is the major environmental time cue that synchronizes the endogenous central circadian pacemaker, located in the suprachiasmatic nuclei of the hypothalamus, and is detected exclusively by the eyes primarily via specialized non-rod, non-cone ganglion cell photoreceptors. Consequently, most blind people with no perception of light (NPL) have either nonentrained or abnormally phased circadian rhythms due to this inability to detect light. Conversely, most visually impaired participants with some degree of light perception (LP) exhibit normal entrainment, emphasizing the functional separation of visual and “nonvisual” photoreception. The aims of the study were to identify the prevalence of circadian disorders in blind women, with the further aim of examining how eye disease may relate to the type of circadian disorder. Participants (
Keywords
The human circadian system is entrained to the 24-h day through daily ocular exposure to the solar light-dark cycle (Czeisler and Gooley, 2007; Czeisler et al., 1995). The light signal is detected exclusively by the eyes and transmitted to the circadian pacemaker via a subset of intrinsically photosensitive melanopsin-containing retinal ganglion cells (ipRGCs; Berson et al., 2002; Gooley et al., 2003; Hattar et al., 2002; Provencio et al., 2002). These photoreceptor cells project primarily to the suprachiasmatic nucleus (SCN), the site of the master circadian pacemaker, via a dedicated retinohypothalamic tract (RHT). When the light signal is unable to transmit to the SCN, the circadian system cannot be reset to the 24-h solar day and usually reverts to its intrinsic non-24-h period (Lockley et al., 2007).
To assess circadian entrainment, a strongly endogenous circadian marker is required, such as the melatonin or cortisol rhythm, as opposed to markers that are easily confounded by external factors, such as sleep. The melatonin rhythm may be measured in 4 to 8 hourly urine samples collected over at least 24 h through the measurement of the major melatonin metabolite, 6-sulfatoxymelatonin (aMT6s), which faithfully reflects the production of melatonin by the pineal (Bojkowski et al., 1987). In previous studies, while most participants with light perception (LP) exhibited normal circadian entrainment (Lewy and Newsome, 1983; Lockley et al., 2007; Lockley et al., 1997; Sack et al., 1992; Skene et al., 1999a), at least half of blind people with no perception of light (NPL) had nonentrained rhythms (Lockley et al., 2007; Lockley et al., 1997; Sack et al., 1992; Skene et al., 1999a), demonstrating that the internal clock was not synchronized with the light-dark cycle, presumably due to disrupted retinal processing (Czeisler et al., 1995). The remaining NPL participants exhibited rhythms with a stable 24-h period with either a normal (~30%) or abnormal (~20%) phase relative to the light-dark and social cycle (Lockley et al., 2007; Lockley et al., 1997).
There are a number of explanations for the observation of apparently entrained circadian rhythms in totally blind patients. First, the duration of the study may be too short to detect significant changes in circadian phase in individuals with circadian periods close to, but not exactly, 24 h. Second, nonphotic times cues, although relatively weak in humans (Mistlberger and Skene, 2004), may also be able to entrain some individuals to a 24-h cycle, particularly those with intrinsic periods close to 24 h (Klerman et al., 1998). Third, a small number of individuals may have a circadian period of exactly 24 h, which would be indistinguishable from entrainment without specialized studies. Finally, a small proportion of patients retain circadian photoreception even in the absence of any visual responses (Czeisler et al., 1995; Zaidi et al., 2007; Gooley et al., 2012; Vandewalle et al., 2013). In these participants, light is able to entrain the circadian system even when the rod and cone photoreceptors of the outer retina are nonfunctional, likely via stimulation of the ipRGCs (Berson et al., 2002; Gooley et al., 2003; Hattar et al., 2002; Provencio et al., 2002).
The aims of the current study were to measure circadian rhythm entrainment status among blind women living in North America and assess the relationship between the etiology and severity of visual blindness and circadian rhythm disorders. We hypothesized that the risk of circadian rhythm disorders would be higher in NPL patients with damage to the ganglion cell layer or optic nerve as compared to patients with disorders of the outer retina.
Methods
Participants
One hundred thirty women were recruited from a cohort of 1392 blind women who participated in a survey (Flynn-Evans et al., 2009). The survey was open to any woman residing in North America, who reported being diagnosed as legally blind, defined as best-corrected vision as 20/200 in the better eye using the Snellen Scale. Participants were eligible for the field study if they were not taking medication known to affect sleep, melatonin, or estrogen production (e.g., hormone replacement therapy, hypnotics, beta-blockers, antidepressants, benzodiazepines, etc.). The first 130 volunteers to meet these criteria were included in the study. Ethical permission for the study was granted by the Institutional Review Board at Partners Healthcare (2003-P-000263) and by the Department of Defense Human Subjects Research Review Board (HSRRB Log No. A-12744). Written informed consent was obtained from all participants. Self-reported light perception status (LP or NPL), visual acuity (able to see the top level of the vision chart, able to count fingers, able to see shadows and hand movements only and light perception only), visual field loss (central and/or peripheral loss), onset and rapidity of visual loss, and primary diagnosis of blindness were obtained from the survey.
Study Schedule and Sample Measurement
Participants were asked to complete daily sleep/nap diaries for 8 consecutive weeks. During this time, they were asked to collect 48-h sequential urine samples in 4-h episodes during the day and 8-h episode overnight on 2 (
Rhythm Analysis
The 6-sulfatoxymelatonin (aMT6s) values in ng/mL were converted to ng/h and subjected to best fit cosine analysis (SAS version 9.2; SAS Institute, Cary, NC), which is considered the standard method, to determine the aMT6s acrophase. Results were used in the present analysis if they showed a significant cosine fit (
In addition to cosine fitting, the mid-range crossing of the aMT6s rhythm was also calculated for all collections. The mid-range crossing of the aMT6s rhythm was determined by calculating the times when the rising and declining phases of the aMT6s rhythm reached half the peak value of the collection period. If the cosine results for both collections for a given participant were not significant, the rising phase of the midpoint crossing value plus the mean intercept between the cosinor acrophase and the rising phase of the midpoint crossing for all participants (2.77 h,
Circadian period (τ) was estimated as the rate of change per day in the timing of the peak aMT6s between the 2 urine collections (Arendt et al., 1988).
Assessment of Circadian Rhythm Disorders
Participants were classified using the clock time of aMT6s phase and were considered entrained at a normal phase (NE) if the mean of the aMT6s peak from collection 1 and collection 2 fell within the normal range for sighted participants (1.3-7.1 h clock time; Lockley et al., 1997) and the estimated circadian period fell within the normal variance for sighted participants entrained to a dim 24-h light-dark cycle (23.88-24.12 h; Wright et al., 2001). A participant was also classified as normally entrained if only 1 of the 2 cosine fits was significant and fell within the normal range for entrainment (
A participant was considered entrained at an abnormal phase (AE) if the estimated circadian period fell within the normal range described above, but the timing of the mean aMT6s peak fell outside of the normal range. Participants with mean acrophases between 7.2 and 16.2 h clock time were classified as entrained at a relatively delayed phase, and those with mean acrophases between 16.3 and 1.3 h clock time were classified as entrained at a relatively advanced phase. A participant was also classified as abnormally phased if only 1 cosine fit was significant and fell outside the normal range (
A participant was considered to have a nonentrained rhythm if the circadian period was outside the normal range (i.e., <23.88 or >24.12 h).
24-h aMT6s Production
The mean total 24-h aMT6s production in mg/24 h was calculated for each collection period by participant. Only participants who were not missing any voids during 2 of the 48-h collections were included in these calculations (
Results
Of the 130 participants, 127 had data sets available for analysis of circadian rhythms (1 night shift worker, 1 participant taking antidepressants, and 1 participant missing urine void times were excluded), and 114 had complete urine collection data sets available for analysis of 24-h aMT6s production. Of 254 × 48-h urine collection blocks assessed, 147 (58%) were significant at the
Among the 127 participants included in the analysis, 86 reported having LP or better (2 excluded participants had LP), and 41 reported having NPL (1 excluded participant had NPL). Table 1 describes the demographic characteristics of the participants. Demographic outcomes were similar among participants with LP and NPL, although those with NPL reported a significantly earlier onset of blindness compared to those with LP. Figures 1 and 2 show aMT6s peak timing and associated sleep data for representative participants with LP (Figure 1) and with NPL (Figure 2) for each circadian classification, NE, AE, and non-24-h, respectively. Figure 3 shows the circadian period distribution by light perception status among nonentrained participants. The period for nonentrained participants ranged from 24.13 to 25.65 h for those with τ > 24.12 h (
Demographic characteristics of female blind subjects with NPL and LP.

BMI = body mass index; NPL = no perception of light; LP = light perception; Exercise = light to vigorous activities.
Among parous women only.
Circadian rhythms of sleep and melatonin among visually impaired participants with light perception. Each panel shows subjective sleep times (■) over 8 weeks double-plotted according to time of day (abscissa) and study day (ordinate). Participants collected sequential 4 to 8 hourly urine samples for 48 hours per week, and the peak time of urinary 6-sulphatoxymelatonin (aMT6s) production ( ) is plotted. Representative plots of each circadian rhythm type are presented: (a) normal entrainment, both the sleep-wake cycle and aMT6s rhythm remain entrained to 24 h with normal nighttime production of melatonin; (b) nonentrained, sleep-wake cycle and melatonin rhythm exhibit a non-24-h period, with recurrent episodes of disturbed nighttime sleep and many daytime naps when melatonin (representing the biological night) peaks during the day; (c) abnormal entrainment advanced phase, sleep-wake cycle and melatonin rhythm are entrained to the 24-h day, but aMT6s peak occurs earlier than the normal range (normal range = 1.3-7.1 h; note: acrophase for participant occurs on day 72 at 21.29 h); and (d) abnormal entrainment delayed phase, sleep-wake cycle and melatonin rhythm are entrained to the 24-h day, but aMT6s peak occurs later than the normal range.
) is plotted. Representative plots of each circadian rhythm type are presented: (a) normal entrainment, both the sleep-wake cycle and aMT6s rhythm remain entrained to 24 h with normal nighttime production of melatonin; (b) nonentrained, sleep-wake cycle and melatonin rhythm exhibit a non-24-h period, with recurrent episodes of disturbed nighttime sleep and many daytime naps when melatonin (representing the biological night) peaks during the day; (c) abnormal entrainment advanced phase, sleep-wake cycle and melatonin rhythm are entrained to the 24-h day, but aMT6s peak occurs earlier than the normal range (normal range = 1.3-7.1 h; note: acrophase for participant occurs on day 72 at 21.29 h); and (d) abnormal entrainment delayed phase, sleep-wake cycle and melatonin rhythm are entrained to the 24-h day, but aMT6s peak occurs later than the normal range.
Circadian rhythms of sleep and melatonin among participants with no light perception. Each panel shows subjective sleep times (■) over 8 weeks double-plotted according to time of day (abscissa) and study day (ordinate). Participants collected sequential 4 to 8 hourly urine samples for 48 h spaced by 2 to 4 weeks. The peak time of urinary 6-sulphatoxymelatonin (aMT6s) production ( ) is plotted. Representative plots of each circadian rhythm type are shown as described in Figure 1.
) is plotted. Representative plots of each circadian rhythm type are shown as described in Figure 1.
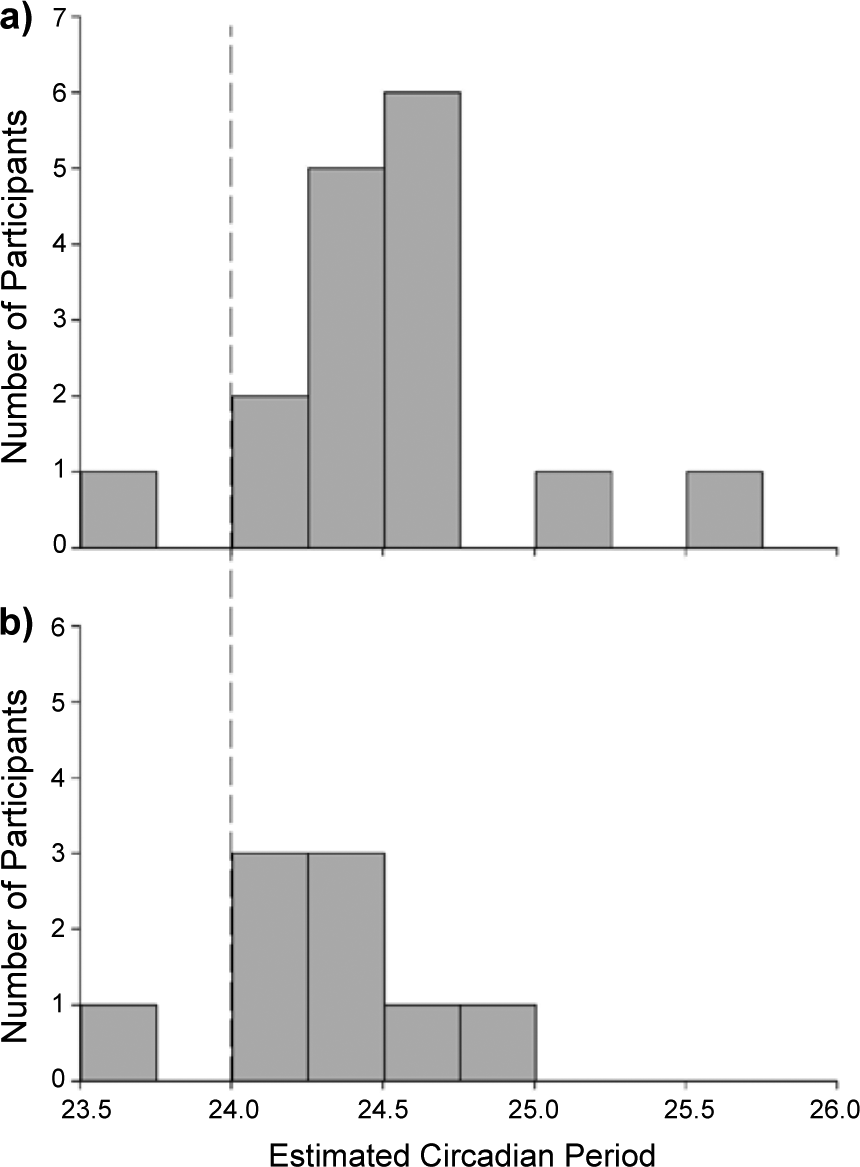
Distribution of calculated circadian period among participants classified as having non-24-hour circadian rhythms. NPL = no perception of light (a;
There were 127 participants with data sets available for circadian classification. Table 2 shows the distribution of circadian type by light perception status.
Distribution of circadian type by light perception status.
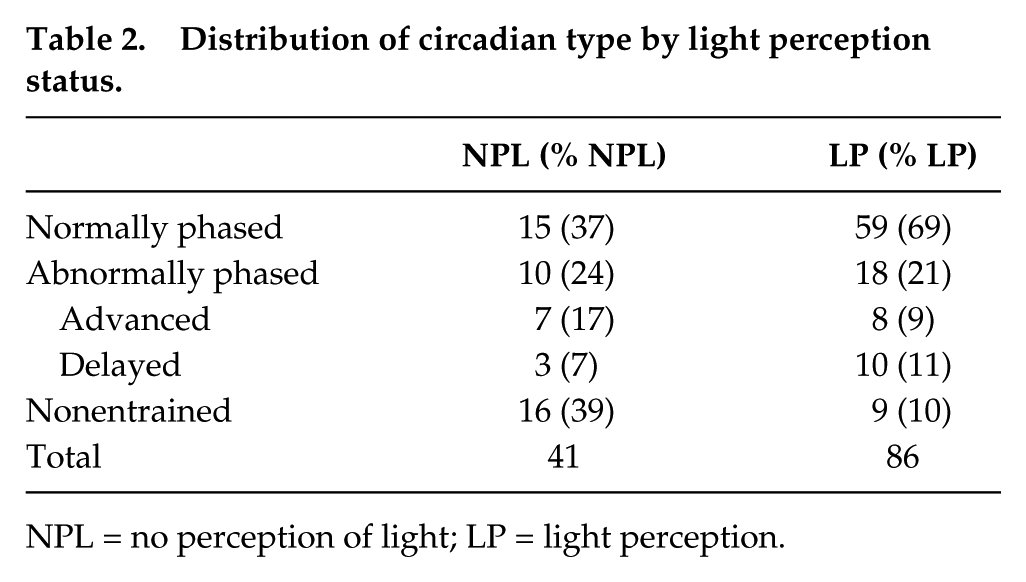
NPL = no perception of light; LP = light perception.
Circadian Type and Eye Condition
There were 19 different primary eye conditions reported among the 127 participants investigated in the circadian rhythm type analysis (Table 3). When eye conditions were collapsed into categories based on the area of the eye affected, 56% of the participants with eye conditions suspected to damage the ganglion cell photoreceptor layer (i.e., retinopathy of prematurity [Yang et al., 2009], diabetic retinopathy [Martin et al., 2004]) were classified as either nonentrained or abnormal phase entrained regardless of LP status. Conversely, participants who had eye conditions associated with damage to the retina outside the RGC photoreceptor layer (i.e., retinitis pigmentosa, macular degeneration) were classified as normally entrained 85% of the time regardless of LP status. Among those classified with eye conditions affecting the optic nerve (glaucoma and optic atrophy), 57% were classified as normally entrained. When this subgroup was restricted to only participants with NPL, 50% were classified as NE and 50% were classified as either AE or nonentrained.
Number of blind subjects of each circadian rhythm type by eye condition.
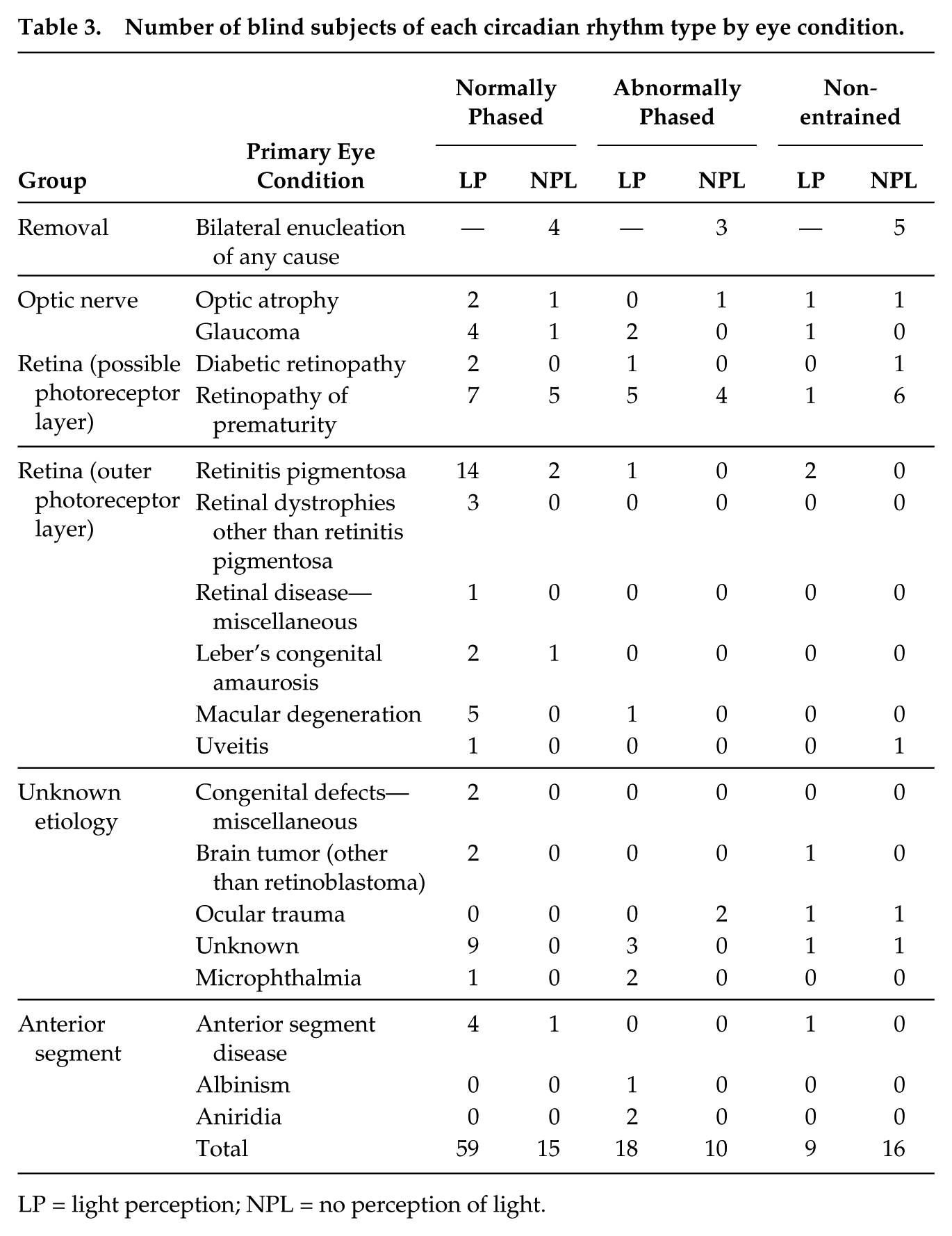
LP = light perception; NPL = no perception of light.
Several eye condition categories had few participants (
24-h aMT6s Production
The amount of aMT6s (µg/24 h) produced over 24 hours from sample collection block 1 compared to sample collection block 2 was well correlated (
Discussion
This cohort represents the largest study to date describing circadian rhythm disturbance confirmed objectively among blind humans. Our study confirms that total visual blindness is a risk factor for nonentrained or abnormally phased circadian rhythms. These data further confirm that there is a high prevalence of circadian rhythm disturbance among participants with no perception of light (NPL). Nearly two-thirds of blind women with NPL were classified either as nonentrained (39%) or abnormally entrained (24%), while just over two-thirds of blind women with LP were classified as normally entrained (69%). Only about a third (37%) of blind women with NPL appeared to have normally phased entrainment. Bilateral enucleation and eye conditions likely to damage the retinal ganglion cell layer, such as retinopathy of prematurity, were associated with nonentrained rhythms. In contrast, eye conditions affecting the outer retinal photoreceptors, such as retinitis pigmentosa or macular degeneration, were primarily associated with preserved circadian entrainment.
There have been only a few large cohorts assembled to assess the prevalence of circadian rhythm and sleep disorders longitudinally among blind people in detail, ranging from ~11 to 67 blind patients (Aldhous and Arendt, 1991; Lewy and Newsome, 1983; Lockley et al., 1997; Sack et al., 1992; Skene et al., 1999a), although earlier work showed broader abnormalities in the blind in larger groups (Lockley et al., 2007). In the largest of these studies (
The prevalence of nonentrained rhythms in study participants is somewhat lower among those with NPL in the present cohort (~40%) compared to that reported previously (~55%; Sack et al., 1992; Skene et al., 1999a). Only about a third (37%) of blind women with NPL appeared to have normal entrainment. The difference in the proportion of NPL patients with nonentrained rhythms between the current and previous studies may be due to differences in the recruitment approach. The current volunteers were selected from a cohort who completed a survey on “health and sleep,” which may have attracted more volunteers without a circadian rhythm disorder than previous studies, which recruited for circadian sleep disorders. We excluded participants who were taking sleep medication, the use of which might be expected to be higher in those with circadian rhythm disorders. We also sampled circadian phase less frequently than in previous studies, which might reduce the specificity of identifying circadian disorders. Collectively, these methodological considerations may account in part for the lower prevalence of nonentrained rhythms found in this population compared to previous studies. Notwithstanding these issues, the prevalence of nonentrained circadian rhythms in blind people remains high and represents a major burden to a substantial proportion of the totally blind population.
The prevalence of nonentrained rhythms among those with LP was higher in the present cohort than in previous studies, when only 1 participant (3% of the total) was found to have a nonentrained rhythm (Lockley et al., 2007; Skene et al., 1999a). There are several possible explanations for this finding, the most likely being exposure to inappropriately timed or low-intensity light that is insufficient to entrain the circadian clock (Uchiyama and Lockley, 2009). Non-24-h disorder has been observed in other patient groups with a high likelihood of unusual light exposure patterns, including patients with delayed sleep phase disorder and psychiatric patients (Uchiyama and Lockley, 2009; Wulff et al., 2010), who may inadvertently self-select a light exposure pattern that results in a systematic daily phase delay in their rhythms, resulting in a circadian period longer than 24 h (Czeisler et al., 1999). As we did not collect light exposure information throughout the study, we are unable to determine whether self-selected light exposure is responsible for nonentrained rhythms in those with LP. It is also theoretically possible, but not yet demonstrated in humans, that LP participants could have visual photoreception in the absence of circadian photoreception. Such a situation would involve selective dysfunction of the ipRGC cells while maintaining normal rod, cone, and nonphotosensitive retinal ganglion cell function (Chen et al., 2011), although such a case has not yet been found. Further work in this population (e.g., demonstration of a failure to entrain to a light-dark cycle or to exhibit light-induced melatonin suppression under controlled laboratory conditions), however, is required to rule out the risk of false-positive results.
It is unclear how visual disorders may affect the nonvisual pathway from the eye to the SCN. The functional and anatomical separation of the visual and “nonvisual” photoreceptor systems, however, may make it possible to predict whether a blind person is predisposed to a circadian rhythm disorder by considering the etiology of his or her blindness and/or the severity of visual loss. In the current study, participants with eye conditions that did not affect the retinal ganglion cell layer of the eye containing the primary circadian photoreceptor melanopsin, such as retinitis pigmentosa (Mazzoni et al., 2008), macular degeneration (Medeiros and Curcio, 2001), or anterior segment disease (which, by definition, includes diseases only affecting the front third of the eye, cornea, iris, vitreous humor, ciliary body, and lens), were more likely to be classified as entrained. Those participants with potential damage to the ganglion cell layer or optic nerve, however, such as bilateral enucleation, malformed eyes, or retinopathy of prematurity, were more likely to be classified as having a circadian rhythm disorder. Despite the fact that this is the largest study of visual impairment and circadian rhythm assessment to date (
The range of circadian periods among nonentrained NPL participants in the present cohort extended from 24.12 to 25.65 h with a mean of 24.5 h (for nonentrained periods >24 h only). This range and mean are similar to the slightly longer periods that have been reported previously in the blind (Emens et al., 2010; Hack et al., 2003), although 2 of the nonentrained blind participants (1 NPL, 1 LP) had periods less than 24.13 h. It is possible that the 8-week study duration facilitated observation of nonentrained rhythms with a short circadian period: for example, a participant with an endogenous period of 24.2 h would only shift by 5.6 clock hours between the start and end of a 4-week study but by 11.2 h over an 8-week study, making it easier to detect a significant change across time. As mentioned above, it is also possible that our study was not long enough to detect nonentrained rhythms in those individuals with an intrinsic period of ~24.1 h or shorter who may not have shifted sufficiently to measure a change in phase. Patients with a period close to 24 h may also have been preferentially entrained by nonphotic time cues (Klerman et al., 1998).
We did not use sleep timing or duration information to classify circadian rhythm disorders, as is standard practice clinically, for several reasons. First, there is a high prevalence of sleep disturbance even among visually impaired and blind people who do not have misaligned circadian rhythms, and so sleep disruption is not an appropriate marker to define circadian disorders in this group (Lockley et al., 1997; Tabandeh et al., 1998; Leger et al., 1999). Second, while sleep behavior is correlated with circadian phase in entrained and nonentrained blind individuals, the circadian period estimated from sleep or activity in nonentrained blind individuals either greatly underestimates the endogenous period or indicates a 24-h sleep rhythm (Lockley et al., 1999). This discrepancy is likely due to the fact that most blind people try to sleep at night regardless of circadian phase (Lockley et al., 1999), in addition to the influence of the homeostatic regulation of sleep on total sleep time (Lockley et al., 1999), and as a result, sleep timing does not reflect the circadian sleep propensity rhythm accurately enough to use it as a reliable marker of circadian phase or period. By using aMT6s to define circadian phase, rather than sleep, the potential confounding effects of other sleep disorders, as well as the need to screen for them, were minimized. In those patients with circadian photoreception, the potential of light to confound phase assessments by suppressing melatonin should be considered but is likely to be minimal given the infrequency of urine sampling, the curve-fitting approach used, and the limited window when melatonin suppression can occur under normal conditions (typically up to 4 h from melatonin onset to bedtime; Wright et al., 2005; Gooley et al., 2011). Furthermore, the amount of melatonin measured per 24 h in LP and NPL blind patients does not differ on average, again suggesting that any differences in the melatonin profile due to light are subtle and unlikely to lead to false-positive detection of circadian phase or period disorders. Indeed, it has been hypothesized that blind people with NPL have higher levels of melatonin compared to those with LP due to a lack of melatonin suppression (Stevens and Davis, 1996), and this hypothesis has been central to the speculation that the lower risk of breast cancer reported among blind women (Flynn-Evans et al., 2009; Kliukiene et al., 2001; Verkasalo et al., 1999) might be due to elevated melatonin levels (Coleman and Reiter, 1992). Our current findings do not support this hypothesis. In the present study, we found no evidence to suggest that aMT6s production is affected by light perception or circadian rhythm type, nor did NPL participants have higher melatonin levels, consistent with smaller prior studies (Klerman et al., 2001; Lockley et al., 1997). Melatonin may still play a role in reducing cancer risk in the blind, but rather than the overall amount of melatonin, other factors (e.g., the duration or internal stability of the melatonin profile) may be important factors that remain to be studied in relation to cancer risk (Arendt, 1995).
In summary, we have confirmed the strong association with total blindness and the risk of circadian rhythm disorders. The prevalence of circadian rhythm disorders in this large cohort was similar to previous reports for blind people with NPL, although we found a higher than expected proportion of women with LP classified as nonentrained. The distribution of circadian rhythm type by etiology of blindness shows that certain eye disorders may relate to an increased likelihood of developing a circadian rhythm disorder. Future studies should examine in more detail how certain eye conditions may predispose some blind people to circadian rhythm disorders and should further examine why some participants with light perception experience abnormal circadian rhythms.
Footnotes
Acknowledgements
The authors thank Dr. Elizabeth B. Klerman, Dr. Benita Middleton, Emeritus Professor Josephine Arendt, Dr. Richard G. Stevens, Dr. Eva S. Schernhammer, Eve Silver, Amy Ruell, and Dr. Joseph T. Hull for assistance with this project. The authors also wish to thank the students who assisted with data collection, including Kathleen Maguire, Emily McCoy, Rebecca Steinberg, Jennifer Markham, Kai Romero, Yunxue Xu, Michael Steinhaus, Inés Pacheco, Grace Kim, Folasade Odenyi, Naila Ramji, Anna Rosenblum, Emma Prokic, Erica Bloom, Natasha Makengo, Ashley Pawlisz, Nicholas Moser, Alana Vivolo, Jessica Giordano, Marissa Sheldon, Tomoko Okada, Liwei Fan, Kristoff Nelson, Kevin Sun, and Jane Flynn. The authors also wish to thank several organizations that donated time and effort to assist with this project, including the American Council of the Blind, the Bay State Council for the Blind, the Massachusetts Commission for the Blind, the Canadian National Institute of the Blind, the National Braille Press, the National Federation of the Blind, the Perkins Braille and Talking Book Library, radio reading services nationwide, and Ripco. Finally, the authors would like to thank Velir Studios for development of the study website. This work was supported by a US Department of Defense Breast Cancer Research Program Idea Award (BC030928, #W81XWH-04-1-0553 to SWL) and a predoctoral fellowship from the Harvard Medical School Division of Sleep Medicine Training Program in Sleep, Circadian and Respiratory Neurobiology (HL07901-11 to EEE).
Conflict of Interest Statement
Dr. Lockley is the principal investigator of 2 completed and 1 ongoing clinical trials of an investigational new drug, tasimelteon, a MT1/MT2 receptor agonist, to test its efficacy and safety for treating nonentrained circadian rhythms in totally blind patients, sponsored by Vanda Pharmaceuticals, Inc. The results were presented in support of Food and Drug Administration approval of tasimelteon in January 2014. Dr. Lockley also holds an investigator-initiated research grant and has completed 2 service agreement contracts from Vanda Pharmaceuticals, Inc. He has also participated in short-term (1-2 h) consulting with 14 financial firms on publicly available data regarding non-24-h sleep-wake disorder and the tasimelteon trials. The studies presented in the current paper were completed prior to the initiation of the sponsored clinical trials of tasimelteon. The other authors have nothing to disclose.
