Abstract
The identification and characterization of rhythmically expressed mRNAs have been an active area of research over the past 20 years, as these mRNAs are believed to produce the daily rhythms in a wide range of biological processes. Circadian transcriptome studies have used mature mRNA as a primary readout and focused largely on rhythmic RNA synthesis as a regulatory mechanism underlying rhythmic mRNA expression. However, RNA synthesis, RNA degradation, or a combination of both must be rhythmic to drive rhythmic RNA profiles, and it is still unclear to what extent rhythmic synthesis leads to rhythmic RNA profiles. In addition, circadian RNA expression is also often tissue specific. Although a handful of genes cycle in all or most tissues, others are rhythmic only in certain tissues, even though the same core clock mechanism is believed to control the rhythmic RNA profiles in all tissues. This review focuses on the dynamics of rhythmic RNA synthesis and degradation and discusses how these steps collectively determine the rhythmicity, phase, and amplitude of RNA accumulation. In particular, we highlight a possible role of RNA degradation in driving tissue-specific RNA rhythms. By unifying findings from experimental and theoretical studies, we will provide a comprehensive overview of how rhythmic gene expression can be achieved and how each regulatory step contributes to tissue-specific circadian transcriptome output in mammals.
Circadian rhythms can be observed in almost all organisms, from cyanobacteria to humans (Liu, 1995; Claridge-Chang, 2001; Bell-Pedersen et al., 2005; Kaneko and Cahill, 2005; Maniscalco et al., 2014; Schmelling et al., 2017). Circadian rhythms allow organisms to anticipate and respond to environmental changes caused by the earth’s rotation, such as temperature, light, or predation, and they provide advantages for survival and fitness. These rhythms range from nitrogen fixation and photosynthesis in cyanobacteria, conidiation in Neurospora, leaf movement and stomata opening in plants, parasite–host interaction in malaria, mating behavior in flies, and the sleep cycle in animals (Sargent et al., 1966; Sakai and Ishida, 2001; McClung, 2006; Robertson, 2008; Morris et al., 2012; Cohen and Golden, 2015; Cabral et al., 2019).
Circadian transcriptome studies during the past 2 decades have demonstrated that rhythmic RNA expression is pervasive in various organisms. In cyanobacteria, almost all genes are rhythmically expressed (Liu, 1995; Nakahira et al., 2004; Ito et al., 2009), whereas in Neurospora, approximately 20% of genes are rhythmically expressed (Hurley et al., 2014; Sancar et al., 2015). In multicellular organisms, such Arabidopsis, Drosophila, and Mus musculus, approximately 5% to 40% of mRNAs were found to be rhythmic, depending on the tissue (Doherty and Kay, 2010; Rodriguez et al., 2013; Kojima and Green, 2015; Abruzzi et al., 2017; Mateos et al., 2018; Yeung and Naef, 2018; Chen and Mas, 2019; Wang et al., 2020). Recent studies also revealed that more than 50% of transcripts are rhythmic in at least 1 tissue. Interestingly, rhythmicity for approximately 10% to 30% of RNAs are conserved between organisms (Zhang et al., 2014; Mavroudis et al., 2018; Mure et al., 2018).
In this review, we will discuss the molecular mechanisms that drive and sustain rhythmic gene expression and how they collectively determine the rhythmicity, phase, and amplitude of RNA expression. We integrate findings from both mathematical and experimental studies, as each regulatory process (e.g., transcription, degradation, translation, localization) is intimately linked to other processes, in contradiction to the widespread belief that each process is independently regulated (Garneau et al., 2007; Labno et al., 2016), and our biological intuitions can be easily misled by this complexity. We will primarily focus on mouse studies, with an emphasis on the liver because of its robust circadian transcriptome profile and availability of various data sets. However, the lessons learned from these studies are broadly applicable to other organisms and tissues, as the processes that control rhythmic RNA expression are fundamental and common to all tissues and organisms.
Rhythmic RNA Expression: Why do we Care and What do we Know?
Rhythmic mRNA expression patterns must be tightly regulated to exhibit the correct period, phase, and amplitude, as they ultimately drive the diverse range of rhythmic biological processes. Rhythmic mRNA expression is also dynamic, as its profile changes in response to aging, feeding pattern, type of diet, or other stimuli (Kuintzle et al., 2017; Sato et al., 2017; Chaix et al., 2018; Greenwell et al., 2019). For example, the number of rhythmically expressed RNAs is 23% fewer in old mice compared with young mice, and the identity of about half of the rhythmic RNAs is different between young and old (Sato et al., 2017). Therefore, deciphering the regulatory mechanisms of rhythmic RNA expression not only deepens our understandings of how circadian processes are driven and sustained but also provides important insights as to how environmental factors disrupt rhythmic mRNA expression.
The rhythmicity of mRNAs is sometimes a poor predictor of the rhythmicity of cognate proteins; in fact, 50% of rhythmically expressed proteins do not exhibit rhythmic mRNA expression patterns (Reddy et al., 2006; Koike et al., 2012; Menet et al., 2012; Mauvoisin et al., 2014; Robles et al., 2014; Wang et al., 2018b; Mauvoisin and Gachon, 2020). However, RNA rhythms are considered important for rhythmic protein synthesis, as approximately 65% to 70% of rhythmic mRNA abundance correlates with rhythmic ribosome footprints (i.e., an indicator of protein synthesis; Atger et al., 2015; Janich et al., 2015). Because rhythmicity of proteins can be determined not only by rhythmic protein synthesis but also by rhythmic protein degradation as well as mean protein half-lives, it will be of great interest in the future to measure globally the protein half-lives and/or protein degradation kinetics to fully understand how rhythmic protein levels are regulated. In addition, the biological significance of the RNA rhythms of the remaining 30% that do not lead to rhythmic protein synthesis remains unclear. Do these RNAs have functions without producing a protein? Are these RNA rhythms merely a noise and biologically irrelevant? These questions await further investigation.
There are several key similarities and differences in the rhythmic mRNA expression patterns between tissues. One common feature is that the mRNA expression of the core clock genes is rhythmic in most tissues, and their peak phase is very similar (Zhang et al., 2014; Mavroudis et al., 2018; Mure et al., 2018). However, the number, identity, and phase of noncore clock genes are considerably different (Panda et al., 2002; Storch et al., 2002; Ueda et al., 2002; Hughes et al., 2009; Harbour et al., 2014; Yoshitane et al., 2014; Zhang et al., 2014; Mavroudis et al., 2018; Mure et al., 2018). In mice, the liver is the most rhythmic, with 16% of total expressed RNAs cycling, followed by the kidney (13%), lung (12%), brown fat (8%), and heart (6%; Zhang et al., 2014). These tissues are metabolically active (Yeung et al., 2018) and can presumably afford the high metabolic cost of sustaining rhythmic RNA expression (Wang et al., 2015). On the other hand, only 3% to 5% of expressed RNAs are rhythmic in other tissues, such as the adrenal gland, aorta, and hypothalamus (Zhang et al., 2014). The rhythmicity of each gene is also tissue specific, and less than 1% are rhythmic in all tissues examined (Zhang et al., 2014; Mavroudis et al., 2018; Mure et al., 2018). Most tissues display a bimodal distribution of peak phase of RNA expression in both nocturnal and diurnal animals. Their peak phases are largely unique to each tissue, although many have at least 1 peak at around ZT18 to ZT24 (where ZT0 is defined as time [hours] of lights-on and ZT12 is defined as time of lights-off). These data raise an important question: How is the rhythmicity, phase, and amplitude of each gene regulated, while still exhibiting the aforementioned similarities and differences between tissues and organisms?
Who is in the Driver’s Seat in Regulating Rhythmic RNA Expression? Synthesis or Degradation?
Mathematical studies have demonstrated that for an mRNA to be rhythmically expressed, RNA synthesis, degradation, or a combination of both must be rhythmic, and the average degradation rate must be short enough (<10 h; Wuarin et al., 1992; Luck et al., 2014; Wang et al., 2018a). Nevertheless, most circadian transcriptome studies measure the rhythmicity of the mature mRNA levels, due in part to the ease of measurements, particularly at the genome-wide level. This has made it challenging to understand how the dynamics of rhythmic synthesis and degradation contribute to the rhythmic RNA profiles, as these measurements do not always capture the kinetics of RNA synthesis and/or degradation. In contrast, theoretical studies can fill this gap and provide important insights into how the rhythmicity, phase, and amplitude of rhythmic RNA are regulated.
Theoretically, the balance between synthesis and degradation is important as it offers considerable flexibility in both the phase and amplitude of rhythmic RNA expression. Mathematical studies have made several interesting predictions about the phase and amplitude of rhythmic RNA expression. For example, the phase of RNA expression can only be within 6 h of the peak phase of RNA synthesis if the RNA synthesis is rhythmic and degradation is constant, whereas it can be at any time within the 24-h cycle if the amplitude of RNA degradation rhythms is greater than that of RNA synthesis rhythms (Luck et al., 2014). Unexpectedly, an increase in the amplitude of RNA degradation rhythms lead to higher mean RNA levels, and this effect is more profound when the phase difference between RNA synthesis and degradation is larger (up to 12 h) or when the RNA half-life becomes shorter. The amplitude of RNA rhythms can also increase as the amplitude of degradation rhythms increases, as long as RNA half-lives are less than 10 h (Luck et al., 2014).
Some of these predictions are intuitive, whereas others are not. Are these predictions supported by experimental observations? What do we know about the mechanisms that alter the rates of RNA synthesis and degradation? Can the tissue-specific rhythmicity be explained by differences in dynamics? Below is a summary of the current literature that focuses on the dynamics of RNA synthesis and degradation.
Mechanisms to Rhythmically Synthesize RNAs
The core clock mechanism is considered the major force in rhythmically synthesizing mRNAs and hence is the central focus of circadian transcriptome studies. In mammals, nearly every cell contains an endogenous clock mechanism, and the core clock genes form interlocking transcription–translation feedback loops to generate cell-autonomous circadian rhythms (Takahashi, 2017). Many of the core clock genes encode transcription factors and regulate rhythmic RNA synthesis of target genes by recognizing target DNA sequences (Ueda et al., 2005; Takahashi, 2017). For example, ARNTL (or BMAL1)–CLOCK heterodimer recognizes an E-box sequence and activates the rhythmic RNA synthesis of target genes, whereas the nuclear receptor proteins, ROR and NR1D (or REV-ERB) proteins, recognize and interact with RORE sequences to activate or repress target RNA synthesis, respectively. DBP and NFIL3 (or E4BP4) proteins bind to a D-box and activate or repress target RNA synthesis, respectively (Takahashi, 2017). Among these, ROREs are most abundant, followed by D-boxes then E-boxes, when noncoding regions that are conserved between human and mouse are scanned (Kumaki et al., 2008). The phase of target RNA expression is directed by the expression phase of circadian transcription factors: RNAs with E-boxes peak early in the morning, followed by those with D-boxes at dawn. RNAs with ROREs peak late at night, at least in peripheral tissues (Ueda et al., 2005; Koike et al., 2012; Fang et al., 2014; Zhang et al., 2015; Zhang et al., 2017; Yoshitane et al., 2019). The phase of RNA expression that contains more than 1 type of motif would ultimately be determined by the number of each motif (Ueda et al., 2005; Kumaki et al., 2008; Ukai-Tadenuma et al., 2008).
For RNAs to be actively synthesized, the chromatin structure must be open for the transcription initiation complex to be recruited to a transcription start site and to initiate transcription (Haberle and Stark, 2018). One way to assess the openness of chromatin is to detect DNase I–hypersensitive sites (Boyle et al., 2008). Interestingly, approximately 8% of genomic loci that are sensitive to DNase I undergo rhythmic changes in their DNase I sensitivity, and their phase coincides with rhythmic recruitment of RNA polymerase II (RNAPII) and H3K27ac modification, indicating rhythmic nucleosome displacement at these sites (Sobel et al., 2017). ARNTL-CLOCK also promotes rhythmic nucleosome removal, creating a permissive chromatin structure (Menet et al., 2014). Although ARNTL-CLOCK or other transcription factor binding is required to initiate RNA synthesis, this binding alone is not sufficient to drive rhythmic RNA synthesis, and the recruitment of both ARNTL-CLOCK and other transcription factors appears to initiate RNA synthesis (Trott and Menet, 2018). ARNTL as well as NR1D2 regulate the rhythmic formation of the chromatin loop structure between enhancers and promoters, which ultimately determines the accessibility of the transcription initiation complex required for RNA synthesis (Aguilar-Arnal et al., 2013; Kim et al., 2018). Rhythmic changes in epigenetic markers, such as histone modifications and DNA methylation, also result in changes in chromatin structure (Lawrence et al., 2016), although DNA methylation appears to have little or no role in regulating rhythmic RNA expression (Vollmers et al., 2012). Some histone modifications, such as H3K9ac, H3K4me3, and H3K27ac, show robust rhythmic patterns at promoter regions near transcription start sites, and their modification patterns coincide with the rhythmic recruitment of RNAPII to these sites. However, most expressed RNAs, including those that are not expressed rhythmically, also show rhythmic histone modification as well as RNAPII recruitment (Koike et al., 2012). It is therefore not clear whether these characteristics are bona fide indicators for active and rhythmic RNA synthesis.
Until recently, rhythmic RNA synthesis has not been directly measured. Initially, the kinetics of RNA synthesis were inferred from the pre-mRNA level (i.e., the intron expression level), as intronic changes reflect changes in transcriptional activity (Koike et al., 2012; Atger et al., 2015; Gaidatzis et al., 2015). More recent techniques, such as GRO-seq, PRO-seq, and Nascent-seq, directly detect nascent and actively transcribed RNAs and can be used for measuring RNA synthesis (Menet et al., 2012; Fang et al., 2014; Stark et al., 2019). By comparing the rhythmicity of pre-mRNA/nascent RNA and mature RNA, these studies concluded that rhythmic RNA synthesis can be observed for 16% to 66% of rhythmically expressed RNA, despite the use of the same algorithm and statistical threshold to detect the rhythmicity of both pre-mRNA/nascent RNA and mature mRNA within each study (Koike et al., 2012; Le Martelot et al., 2012; Menet et al., 2012; Atger et al., 2015). It is unclear, however, why there is this significant variation between studies. It could be due to either experimental conditions (i.e., feeding paradigm, sampling intervals, light conditions, etc.) and/or statistical methods (i.e., rhythmicity detection algorithm, rhythmicity cutoff, etc.; Luck and Westermark, 2016; Hughes et al., 2017). Regardless, these studies clearly indicate that rhythmic RNA synthesis contributes to sustaining rhythmic RNA expression, at least for some rhythmic mRNAs. At the same time, these studies also indicate that rhythmic RNA synthesis alone does not account for all rhythmic RNAs. So, how is the rest of the rhythmic RNA regulated if not synthesized rhythmic? Do these data indicate that rhythmic degradation plays a significant role?
Is Rhythmic RNA Degradation Important for Driving and Sustaining Rhythmic RNA Expression?
RNA degradation has been seen as a passive mechanism to simply degrade old, aberrant, and unwanted RNAs to maintain cellular homeostasis (Garneau et al., 2007; Labno et al., 2016). However, turnover of mRNAs is actually a highly regulated process and is an important regulatory mechanism affecting gene expression that is critical for the cell cycle, cell proliferation and survival, stress response, and development of neurodegenerative diseases, among others (Graham et al., 2010; Eser et al., 2014; Alkallas et al., 2017; Zheng et al., 2018). Mature mRNAs are degraded in both the 5′ to 3′ and 3′ to 5′ directions and can be preceded by deadenylation (i.e., shortenings of poly[A] tails) and/or decapping in the cytoplasm (Garneau et al., 2007; Labno et al., 2016; Fig. 1). Although only 1 enzyme, RNAPII, synthesizes all mRNAs in the nucleus, more than 15 enzymes are involved in degrading mRNAs (Garneau et al., 2007; Ibrahim et al., 2008; Nagarajan et al., 2013; Labno et al., 2016; Silva et al., 2017).
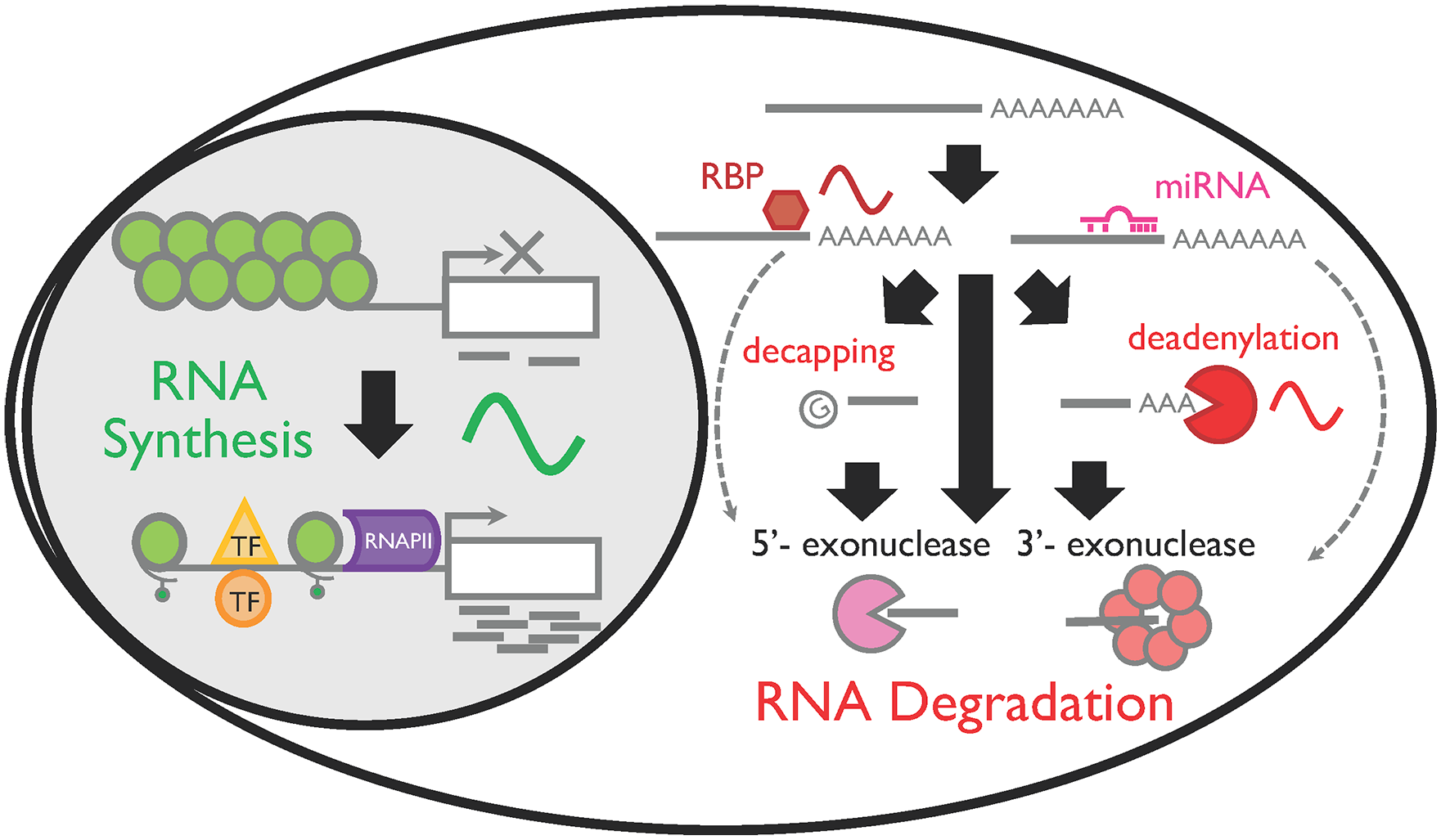
RNA life cycle and its rhythmic regulation in mammalian cells. RNA is synthesized by RNA polymerase II (RNAPII) in the nucleus. Rhythmic mRNA synthesis is achieved when the chromatin structure is open and RNAPII can be recruited to the transcription start site. Recruitment of transcription factors (TFs) is also necessary, but not sufficient, to rhythmically synthesize RNA. Once mRNAs are synthesized, they are transported in the cytoplasm. Mature mRNAs are degraded in both 5′ to 3′ and 3′ to 5′ directions and can be preceded by deadenylation (i.e., shortenings of poly[A] tails) and/or decapping. Interaction with RNA binding proteins (RBPs) or miRNA can also facilitate RNA degradation.
Studies have investigated whether specific mechanisms that regulate RNA stability (i.e., degradation rate) are regulated rhythmically and contribute to rhythmic RNA expression. For example, miRNAs destabilize target RNAs and inhibit translation (Fabian and Sonenberg, 2012; Yates et al., 2013). Inactivating miRNA biogenesis by knockout of Dicer, which encodes the ribonuclease that converts pre-miRNAs to mature miRNAs, alters the expression of up to 30% of rhythmic RNAs in mice (Du et al., 2014). Rhythmic polyadenylation dynamics can also lead to rhythmic RNA degradation, because the length of poly(A) tails and the site of polyadenylation are important for RNA stability and translation efficiency (Nicholson and Pasquinelli, 2019). Interestingly, 2.9% of genes alternate their poly(A) site usage with a circadian period in the mouse liver (Gendreau et al., 2018). Furthermore, 2.3% of genes exhibited rhythmicity in the length of poly(A) tails, also in the mouse liver (Kojima et al., 2012). Because the expression of some deadenylases, including Nocturnin, is rhythmic in several tissues (Wang, 2001; Pizarro et al., 2013), the regulation of the rhythmic poly(A) tail length may not be limited to the liver. Interaction with RNA-binding proteins is another mechanism regulating rhythmic RNA degradation, as RNA-binding proteins recognize specific sequence elements in target RNAs and ultimately modulate its degradation rate (Garneau et al., 2007; Gerstberger et al., 2014). In fact, RNA degradation of Period2 and Cryptochrome 1, core clock genes, are rhythmic and regulated by RNA-binding proteins (Woo et al., 2009; Woo et al., 2010).
More recently, transcriptomic data were combined with mathematical modeling to assess the contribution of rhythmic degradation in rhythmic RNA expression. In this study, the degradation rate was first assumed to be either rhythmic or constant. The model that fits the RNA expression profile best, based on the synthesis rate inferred from the pre-mRNA expression levels (i.e., the intron level), was ultimately chosen to determine whether each RNA underwent rhythmic RNA degradation (Luck et al., 2014; Wang et al., 2018a). These analyses predicted that at least 35% of rhythmically expressed mRNA are rhythmically degraded (Luck et al., 2014; Wang et al., 2018a).
These findings clearly suggest that rhythmic degradation also plays an important role in rhythmic RNA expression. Rhythmic degradation also is energetically a cost-effective method to regulate rhythmic RNAs as compared with nonrhythmic degradation (Jo et al., 2018). Given that the regulatory mechanism for RNA degradation is more diverse and a higher number of enzymes are involved compared with RNA synthesis, rhythmic degradation may serve as an advantage to provide flexibility in setting the phase or amplitude of RNA expression. However, the circadian kinetics of RNA degradation have never been experimentally measured, partly due to a lack of methodology. The measurement is particularly challenging in vivo and/or on a genome-wide scale, and it is still unclear as to what extent rhythmic degradation contributes to rhythmic RNAs. Newer techniques, such as metabolic labeling (Wada and Becskei, 2017; Yamada and Akimitsu, 2019), may circumvent these challenges and clarify to what extent rhythmic degradation contributes to rhythmic RNAs.
What Underlies the Mechanisms of Regulating Tissue-Specific Rhythmic RNA Expression?
Although nearly every tissue and cell in the body contains the same core clock mechanism and is under the same systemic regulation, every tissue has a unique circadian profile in number, identity, phase, and amplitude of rhythmic RNA expression (Meireles-Filho et al., 2014; Zhang et al., 2014; Mavroudis et al., 2018; Mure et al., 2018). Tissue-specific rhythmic RNA expression may be derived from the unique rhythmic RNA profile at the single-cell level from various cell types and interindividual variability (Chakrabarti et al., 2018; Wen et al., 2020). The core clock machinery may also be slightly different between tissues, and each tissue has a unique network with a different hierarchy between each feedback loop, as suggested by a recent theoretical study (Pett et al., 2018). Nevertheless, little is known how the same core clock mechanism can drive such diverse rhythmicity in different tissues. Can differences in rhythmic synthesis or degradation machinery help explain the tissue-specific outputs of rhythmic RNA expression (Fig. 2)?
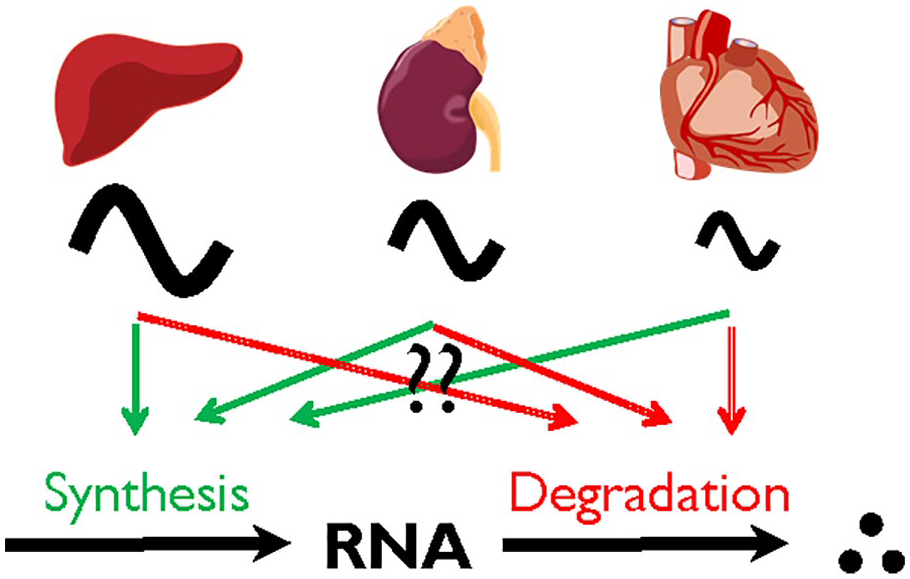
Rhythmic RNA expression is tissue specific. The number, identity, and phase of rhythmic RNA expressions are considerably different between tissues; however, it is currently unclear whether the dynamics of RNA synthesis or degradation contribute to the difference.
Recent ARNTL cistrome analysis revealed that ARNTL recognizes and interacts with its target in a tissue-specific manner (Trott and Menet, 2018), and a combined action between ARNTL-CLOCK and tissue-specific transcription factors is critical to drive tissue-specific rhythmic RNA synthesis (Yeung et al., 2018; Beytebiere et al., 2019). The chromatin loop structure is also regulated rhythmically in a tissue-specific manner, serving as another mechanism to regulate tissue-specific chromatin accessibility and RNA synthesis (Yeung et al., 2018).
In contrast, it remains unclear whether RNA degradation is regulated in a tissue-specific manner and whether its tissue-specific rhythmicity contributes to the tissue-specific RNA rhythms. Interestingly, a recent mathematical study predicted that the phase of deadenylation, a rate-limiting step for RNA degradation, directs the phase of RNA expression, regardless of the phase of RNA synthesis (Yao et al., 2020). Because some deadenylases are rhythmically expressed and their expression patterns are tissue specific (Pizarro et al., 2013), regulation of deadenylation is a potentially novel mechanism to regulate tissue-specific rhythmic RNA expression. In addition, some miRNAs and RNA-binding proteins also show clear tissue-specific expression patterns, and it is possible that they also contribute to the tissue-specific degradation of target RNAs (Gerstberger et al., 2014; Ludwig et al., 2016).
Recent studies also revealed that codon usage represents a universal and conserved mechanism to directly regulate mRNA stability from bacteria to humans (Hanson and Coller, 2018; Hia et al., 2019; Narula et al., 2019; Forrest et al., 2020). Codon usage itself would not directly explain the tissue specificity of rhythmic RNA expression per se, as the coding sequence is typically common in all tissues. However, the mechanism that links codon usage to mRNA stability may be dynamically regulated and exhibit tissue specificity and/or rhythmicity. Interestingly, codon usage of core clock genes is important for circadian function in cyanobacteria, Neurospora, and Drosophila (Xu et al., 2013; Zhou et al., 2013, Fu et al., 2016). It will be of future interest to clarify whether differences in codon usage alters the mRNA stability of these genes and, if so, whether the difference in mRNA stability directly affects the circadian clock function.
Understanding the mechanisms underlying tissue-specific rhythmic RNA expression will be the next big challenge. Over the past few years, we have started to gain some knowledge regarding how tissue-specific rhythmic RNA synthesis can be achieved (Yeung et al., 2018; Beytebiere et al., 2019). Mathematical studies have also predicted the importance of rhythmic RNA degradation in defining RNA rhythms. However, whether and how tissue-specific rhythmic degradation regulates rhythmic RNA expression is unclear and needs to be tested experimentally. To address this, it is crucial to develop a method that thoroughly monitors RNA degradation in vivo, regardless of the degradation pathway, as this will allow us to test many interesting predictions from mathematical studies and directly compare the results from different species that use distinct RNA degradation mechanisms.
Concluding Remarks and Future Perspectives
Over the past 20 years, significant progress has been made in identifying rhythmically expressed RNAs in various organisms and tissues, and we have obtained a comprehensive catalog of rhythmically expressed RNAs. This has helped identify pathways that are under circadian control and to determine how disruption of these patterns leads to abnormal physiology.
Both rhythmic synthesis and degradation are important for the regulation of rhythmic RNA expression. Theoretical studies have also suggested that rhythmic degradation offers more flexibility and versatility in determining the phase and amplitude of rhythmic RNA expression and therefore would be more advantageous in driving differential RNA expression between tissues. However, it is still unclear how the coordination between these 2 mechanisms ultimately determines the amplitude and phase of each cyclic RNA and how they drive tissue-specific rhythmicity. To fully understand how rhythmic RNA expression patterns are determined in each tissue, it is imperative to expand our analysis and focus on circadian dynamics of not only RNA synthesis and steady-state RNA levels but also RNA degradation. The comprehensive measurement of the 3 parameters (RNA synthesis, degradation, and its steady-state level) will also allow us to test provocative and sometimes unexpected predictions from theoretical studies and may help us uncover unexpected consequences of rhythmic RNA synthesis and/or degradation regulation: Is rhythmic synthesis or degradation important only for driving rhythmic RNA expression? If so, why do cells bother to rhythmically synthesize RNAs despite a high energetic cost, only to be expressed arrhythmically for RNAs with long half-lives? Does rhythmic synthesis or degradation play a role outside of rhythmic RNA expression? It is high time to appreciate this long-overlooked process of RNA regulation to fully understand how rhythmic RNA expression is achieved.
Footnotes
Acknowledgements
The authors thank Drs. Janet Webster and Jean Clarke (Fralin Life Sciences Institute, Virginia Tech) as well as the members of the Kojima Laboratory for their critical reading of the article. Work in the Kojima Laboratory was supported by R01GM126223 from the National Institutes of Health (to S.K.).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
