Abstract
The circadian system regulates daily rhythms of physiology and behavior. Although extraordinary advances have been made to elucidate the brain mechanisms underlying the circadian system in nocturnal species, less is known in diurnal species. Recent studies have shown that retinorecipient brain areas such as the intergeniculate leaflet (IGL) and olivary pretectal nucleus (OPT) are critical for the display of normal patterns of daily activity in diurnal grass rats (Arvicanthis niloticus). Specifically, grass rats with IGL and OPT lesions respond to light in similar ways to intact nocturnal animals. Importantly, both the IGL and OPT project to one another in nocturnal species, and there is evidence that these 2 brain regions also project to the superior colliculus (SC). The SC receives direct retinal input, is involved in the triggering of rapid eye movement sleep in nocturnal rats, and is disproportionately large in the diurnal grass rat. The objective of the current study was to use diurnal grass rats to test the hypothesis that the SC is critical for the expression of diurnal behavior and physiology. We performed bilateral electrolytic lesions of the SC in female grass rats to examine behavioral patterns and acute responses to light. Most grass rats with SC lesions expressed significantly reduced activity in the presence of light. Exposing these grass rats to constant darkness reinstated activity levels during the subjective day, suggesting that light masks their ability to display a diurnal activity profile in 12:12 LD. Altogether, our data suggest that the SC is critical for maintaining normal responses to light in female grass rats.
Light affects behavior and overall health, including academic performance, eating habits, stress levels, sleep behaviors, and quality of life (Wright et al., 2013). It also plays a role in the entrainment of circadian rhythms and masking, by stimulating activity in diurnal (day-active) animals and inducing sleep in nocturnal (night-active) animals (Langel et al., 2018; Yan et al., 2018). Light affects activity levels, including sleep and wakefulness, via retinofugal pathways, which include major projections from the retina to the suprachiasmatic nucleus (SCN), geniculate complex, pretectum, and the superior colliculus (SC) in both nocturnal and diurnal species (Morin and Studholme, 2014; Gaillard et al., 2013). The SCN, also referred to as the master clock in mammals (Stephan and Zucker, 1972), is critical for maintaining endogenous circadian rhythms (Hastings et al., 2014; Gall et al., 2016). In contrast, the geniculate complex, which includes the intergeniculate leaflet (IGL), is part of the pathway for conscious visual processing. The IGL modulates entrainment to light and is involved in maintaining acute responses to light, also known as masking (Harrington and Rusak, 1986; Johnson et al., 1989; Pickard, 1989; Edelstein and Amir, 1999; Pickard, 1994; Gall et al., 2013, 2014). The pretectum, which includes the olivary pretectal nucleus (OPT), is involved in maintaining the pupillary light reflex and is critical for masking (Trejo and Cicerone, 1984; Miller et al., 1999; Gall et al., 2017). The SC has been less studied for its role in circadian rhythms and masking behavior, especially in a diurnal species.
The SC receives visual, auditory, and somatosensory input and organizes appropriate motor responses to these stimuli in nocturnal animals (King, 2004). Intrinsically photosensitive retinal ganglion cells (ipRGCs) along with direction- and motion-sensitive retinal ganglion cells transmit visual information to the SC (Langel et al., 2015; Fu et al., 2005; Hong et al., 2011; Huberman et al., 2008; Huberman et al., 2009; Kim et al., 2008; Zhang et al., 2012). The superficial layers of the SC process visual stimuli, whereas deeper layers also process somatosensory and auditory stimuli (Cang and Feldheim, 2013; Dhande and Huberman, 2014; Bednárová et al., 2018; Ito and Feldheim, 2018; Cohen et al., 2008; Castro-Alamancos and Favero, 2016; Bezdudnaya and Castro-Alamancos, 2014). In nocturnal animals, the SC has been extensively explored for its role in initiating saccades or rapid eye movements from one target to the other (Wang et al., 2015). In addition to its role in integrating stimuli of various modalities (Ursino et al., 2009), the SC also plays an important role in spatial navigation, visual guidance of locomotion, and maintaining nocturnal activity patterns (Lines and Milner, 1985; Altman, 1962). Interestingly, the SC has been shown to mediate the effects of light on sleep in nocturnal albino rats (Miller et al., 1998), and GABAergic neurons in the SC are essential for acute dark induction of wakefulness in nocturnal mice (Zhang et al., 2019), suggesting that the SC may be critical for maintaining masking responses to light and darkness. Although these characteristics have been established in nocturnal animals, little is known about the role of the SC in diurnal animals, especially how the SC is involved in responding to acute presentations of light.
In addition to the direct innervation of the SC by light, several other retinorecipient brain areas also project to the SC in nocturnal rodents, such as the IGL and the OPT, brain regions that are within the geniculate complex and pretectum, respectively (Taylor et al., 1986). The IGL and OPT also connect with one another (Morin and Blanchard, 1998). Both of these brain areas receive input from ipRGCs in diurnal grass rats as well and are involved in non–image-forming vision (Langel et al., 2015). We have recently shown that the IGL and OPT are critical for the normal expression of behavior in response to light in grass rats (Gall et al., 2013). Given that the SC receives direct retinal input in nocturnal and diurnal species (Ellis et al., 2016; Gaillard et al., 2013), is involved in the triggering of rapid eye movement sleep in nocturnal rats (Miller et al., 1998), and is disproportionately large in the diurnal grass rat compared with nocturnal species (Gaillard et al., 2013), we used diurnal grass rats to test the hypothesis that the SC contributes to a comprehensive day-active profile and plays an important role in the behavioral response to light. To test this hypothesis, we performed bilateral electrolytic lesions of the SC in female grass rats and examined how circadian rhythms and acute responses to light were affected by these lesions. Our results suggest that the SC is critical for maintaining normal behavioral responses to acute pulses of light and sustained periods of light. These results have important implications for the mechanisms underlying masking responses to light.
Materials and Methods
All experiments were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80-23) and were approved by the Institutional Animal Care and Use Committee of Hope College. All efforts were made to minimize the number of animals used.
Subjects
Eighteen adult female grass rats (Arvicanthis niloticus) were obtained from a breeding colony maintained at Hope College. Consistent with our previous reports on the effects of brain lesions in retinorecipient areas in grass rats (Gall et al., 2013, 2014, 2016, 2017), female grass rats were used in this study. Plexiglas cages (34 × 28 × 17 cm) were used to singly house each animal. Each cage contained a polyvinyl chloride pipe for enrichment purposes. Food (PMI Nutrition Prolab RMH 2000, Brentwood, MO) and water were provided ad libitum. Animals were singly housed for at least 4 weeks prior to assigning animals to an experimental condition while being exposed to 12:12 LD conditions at 300 lux during the light phase and complete darkness for the dark phase. Lux levels were sampled weekly over the course of the experiment.
Experimental Procedures
To measure general activity counts and patterns, infrared sensors were placed above the cages and connected to a computer located in an adjacent room. Data were collected in 1-min bins using VitalView 1.2 software (Oakmont, PA) in 12:12 LD conditions (lights-on at 0700 h; lights-off at 1900 h) for at least 2 weeks prior to surgery. Animals were maintained in 12:12 LD conditions for at least 2 weeks following surgery. Grass rats were then placed in constant darkness (DD) for 7 days. Finally, grass rats were reentrained to 12:12 LD conditions for 4 weeks. After 2 weeks in this condition, grass rats were given 2-h light pulses at zeitgeber time (ZT) 14, 18, and 22 and 2-h dark pulses at ZT 2, 6, and 10, as described previously (Shuboni et al., 2012). ZT0 was defined as the time when the lights turned on, and ZT12 was when the lights turned off. Light pulses were given first in 3-day cycles, with day 1 being the maintenance day in 12:12 LD, day 2 being the baseline day in 12:12 LD, and day 3 being the day of the light pulse. The same procedure was repeated for dark pulses.
Surgery
Grass rats were anesthetized using isoflurane (2.5% initial rate; 1.0%-1.5% maintenance rate) and placed into a stereotaxic apparatus (Stoelting Co., Chicago, IL). A local anesthetic was injected into the top of the animal’s head (Lidocaine, Hospira Inc., Lake Forest, IL; 6 mg/kg, subcutaneously [s.c.]), and artificial tears (Butler Company, Columbus, OH) were administered on the surface of the eyes. An incision was made on the top of the scalp, and 4 small holes were drilled into the skull. Bilateral electrolytic lesions were formed with an insulated tungsten microelectrode (A-M Systems, model 5770, 500-µm diameter, 12-MOhm impedance; Sequim, WA, USA). The electrode was placed in 4 locations using the coordinates AP −0.22 and −0.34 cm from bregma, ML ±0.08 cm, and DV −0.25 cm, which were determined based on pilot surgeries on 10 grass rats. The Stoelting lesioning device was used to make lesions by producing 3.0 mA of DC current for 30 sec in the experimental group (n = 12). The incision was then closed using autoclips, and antiseptic ointment was applied (Nolvasan Antiseptic Ointment, Fort Dodge, IA; 1% chlorhexidine acetate in 10% sterile alcohol base) to prevent infection. The sham control group (n = 6) underwent the same procedure without applying any current. All animals received sterile saline (1.0 mL s.c.; Abbott Laboratories, Chicago, IL) to prevent dehydration. Directly following the surgical procedures, all animals were given an analgesic of ketoprofen (5 mg/kg body weight s.c.; Fort Dodge Animal Health). Each animal was placed on a warming blanket underneath the cage for at least 1 h to recover before they were moved back to the recording room under standard 12:12 LD conditions to continuously monitor activity.
Open-Field Test
We found that SC lesions in female grass rats significantly reduced activity levels during the lights-on phase, raising the possibility that heightened anxiety-like behavior in response to light was exhibited by these animals. To test this hypothesis, anxiety-like behavior was assessed using the open-field test during the lights-on phase at the end of the experiment following light and dark pulses in 12:12 LD conditions. Animals were placed in the open-field test (100 × 100 × 40 cm) for 5 min during the middle of the subjective day (1100-1500 h) following habituation to the experimental room at least 1 h prior to testing. A camera was used to record behavior during the open-field test (item 60528, Stoelting Co.), and ANY-maze behavioral tracking software (version 4.99, Stoelting Co.) was used to quantify time spent in the perimeter versus the center of the open-field test.
Histology
Animals were euthanized by intraperitoneal injections of sodium pentobarbital (Ovation Pharma-ceutical, Deerfield, IL) and immediately perfused transcardially with 0.01 M phosphate-buffered saline, pH 7.2, followed by 4% paraformaldehyde (Sigma-Aldrich, St. Louis, MO) with 75 mM lysine (Sigma-Aldrich) and 10 mM sodium periodate (Sigma-Aldrich) in 0.1 M PB (periodate-lysine-paraformaldehyde; PLP). Following perfusions, brains were immediately removed and postfixed in PLP for 4 h, then transferred to a 20% sucrose solution (J.T. Baker, Phillipsburg, NJ) and stored at 4°C for at least 48 h. Brains were sectioned in 50-µm coronal sections. Brain sections were stained using thionin, and raters blind to experimental condition (i.e., lesion vs. sham) and behavioral outcome (i.e., changes in activity levels) traced the lesioned areas of the brain. This was done by obtaining photographs of at least 6 brain sections of each subject and outlining the extent of the lesion using image software (Photoshop CS5, Adobe, San Jose, CA). Lesion sizes for the smallest lesions, largest lesions, and misses were overlaid on a trace of a grass rat brain section from the most rostral to most caudal extent of the lesion (Fig. 1).
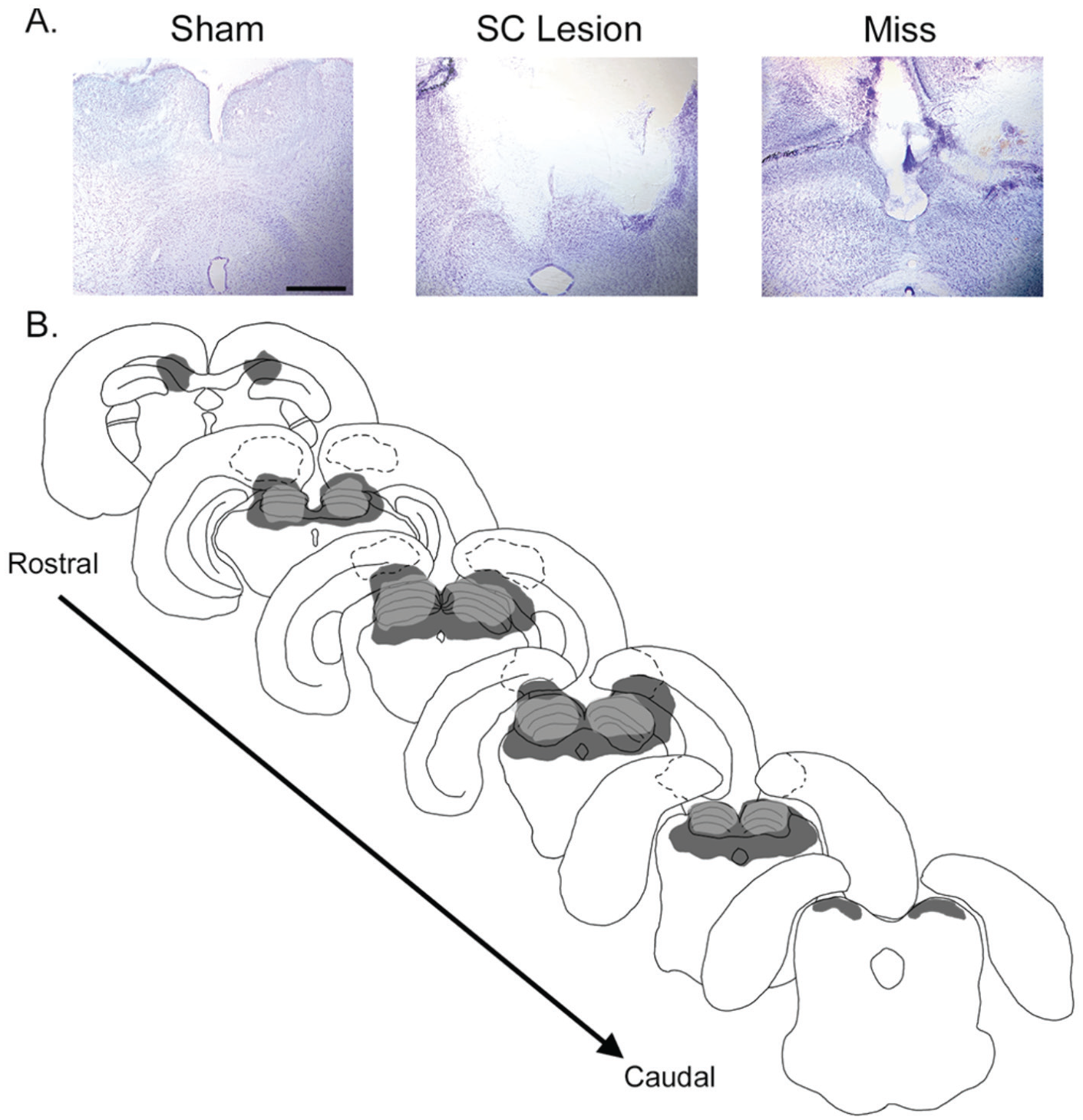
Histology from representative grass rats with lesions aimed for the superior colliculus (SC). (A) Thionin-stained brain sections from a representative sham grass rat with a complete, bilateral electrolytic lesion and a miss with damage to the cerebral cortex but a completely intact SC. (B) Drawings outlining the rostral-caudal extent of brain regions including the SC. The SC is indicated by the layered structure drawn at the dorsal portion of each section. Light-gray–filled regions indicate the smallest complete SC lesion, and dark-gray–filled regions indicate the largest complete SC lesion. Dashed regions indicate misses with damage to cortical structures. Scale bar represents 1 mm.
Data Analyses
Following histology, scorers, blind to experimental condition (i.e., lesion vs. sham) and behavioral outcome (i.e., changes in activity levels), identified 3 animals with either unilateral damage to the SC (n = 1) or damage to cortical structures, sparing the SC bilaterally (n = 3). These 4 animals were excluded from all statistical analyses, since in all 4 cases, the SC was at least unilaterally intact. Therefore, statistical analyses were based on 8 animals with complete SC lesions and 6 shams with an intact SC. Photomicrographs of at least 6 coronal sections were obtained for each animal.
In LD conditions, we analyzed the total activity during the day and night for 5 days for each animal immediately before surgery and 2 weeks after surgery. We obtained general activity data from infrared sensors (VitalView, STARR Life Sciences Corp., Oakmont, PA) and visualized these data using ActiView (STARR Life Science Corp.). A 2-way analysis of variance (ANOVA) was used to assess total activity counts during the day and night using a 2 × 2 design (time of surgery [presurgery vs. postsurgery] × time of day [day vs. night]) separately in shams and animals with complete SC lesions. In addition, a 2-way ANOVA using a 2 × 24 design (surgical condition [sham vs. SC lesion] × ZT [1-24]) was used to examine the time points at which differences in activity counts emerged. Significant interactions were followed by post hoc tests for each ZT using independent-samples t tests. A Bonferroni correction was used to adjust for multiple comparisons (in this case, alpha was set at 0.05/24 = 0.002). In DD conditions, we calculated the hourly average activity patterns by calculating the period of each animal’s rhythm and dividing it by 24 to obtain the length of each circadian hour. These activity counts were then averaged during each 24-h interval for 5 days in DD for each animal, to obtain a mean and standard error per hour. Independent-samples t tests were used to compare hourly circadian counts for shams versus SC lesioned grass rats. Again, a Bonferroni correction was used to adjust for multiple comparisons (alpha was set at 0.002, because of 24 comparisons being made).
Total activity counts were analyzed using a 1-way ANOVA in LD presurgery and postsurgery and also in DD. Activity counts were collected over a 5-day period before and after surgery and averaged over a 24-h period. A Tukey test was used as a post hoc test. From this data set, activity counts were separated for the lights-on and lights-off phases, and a percentage of activity occurring during lights-on was calculated by taking the total average activity during the lights-on phase, dividing by the total average activity per 24-h period, and multiplying by 100. Independent-samples t tests were used to analyze differences between presurgery and postsurgery separately for shams and grass rats with complete SC lesions.
Light pulses and dark pulses were analyzed by calculating the activity counts during a 2-h interval on the day of the pulse and comparing it with the same 2-h interval from the day before (with no pulse given on the baseline day). A 3-way ANOVA was used to analyze the pulse data using a 2 × 2 × 3 design for light pulses and dark pulses separately (surgical condition [sham vs. SC lesion] × lighting condition [baseline vs. 2-h pulse] × time of day [ZT14, 18, 22 for light pulses; ZT2, 6, 10 for dark pulses]). Significant interactions were followed by paired-sample t tests for each ZT.
Circadian patterns were also analyzed in DD by calculating the period length and the duration of the active phase (alpha) for each animal. Alpha was calculated based on the onset of activity to the offset of activity. Clocklab (version 2.6.1; Actimetrics, Wilmette, IL) was used along with visual inspection of actograms to calculate onset and offset of activity. The phase angle of entrainment was analyzed in LD by calculating the difference in hours between alpha onset and lights-on. An independent-samples t test was used to analyze differences in period, alpha, and total activity counts between shams and grass rats with complete SC lesions in DD, while in LD, phase angle of entrainment, diurnal activity percentage, and total activity counts were analyzed. These results are reported in Table 1 to compare shams and SC lesioned grass rats on these measures.
Quantitative measures in constant darkness and in a 12:12 LD cycle.
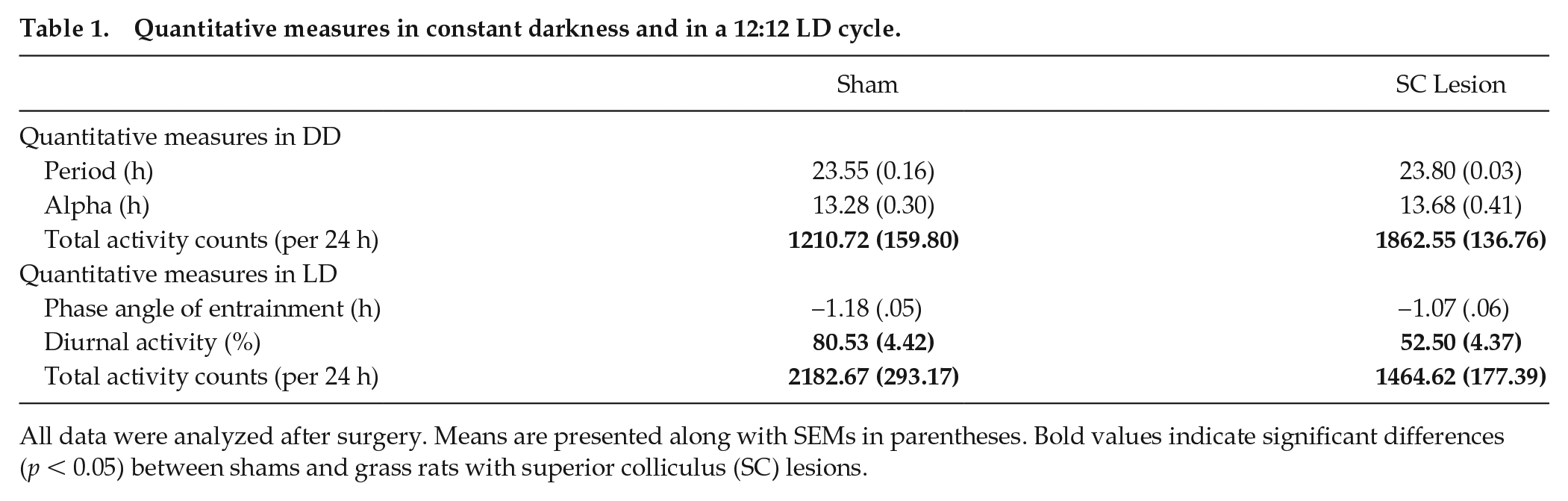
All data were analyzed after surgery. Means are presented along with SEMs in parentheses. Bold values indicate significant differences (p < 0.05) between shams and grass rats with superior colliculus (SC) lesions.
For assessing anxiety-like behaviors in the open-field test, independent-samples t tests were used to examine differences between shams and SC lesions with respect to the amount of time spent in the perimeter versus center (50 × 50 cm) of the open-field test.
The alpha was set at 0.05 for all statistical tests (except when a Bonferroni correction was used; see analyses above). SPSS (version 25) was used for all analyses. Means are presented with standard error of the mean.
Results
Histology
Figure 1 presents the rostral-caudal extent of damage in grass rats with SC lesions. Three groups of grass rats were identified based on histology: (1) those with complete, bilateral damage to the SC (n = 8); (2) shams (n = 6); and (3) those with incomplete lesions to the SC (“misses,” n = 4), with either unilateral damage to the SC (n = 1) or no damage to the SC (n = 3).
In 12:12 LD Conditions, Complete SC Lesions Resulted in Significantly Reduced Activity, Which Was Restored in DD
Figure 2 presents double-plotted actograms of a representative sham (Fig. 2A), grass rats with complete SC lesions (Figs. 2B, C), and a grass rat with no damage to the SC (“miss,” Fig. 2D) in 12:12 LD and DD. Prior to surgery, grass rats were clearly diurnal, with most of their activity during the lights-on phase. After surgery, shams remained diurnal, with no significant change in activity levels, whereas grass rats with complete SC lesions exhibited a significant reduction in activity levels during the lights-on phase. When grass rats with SC lesions were placed in DD, activity levels during the subjective day were restored.
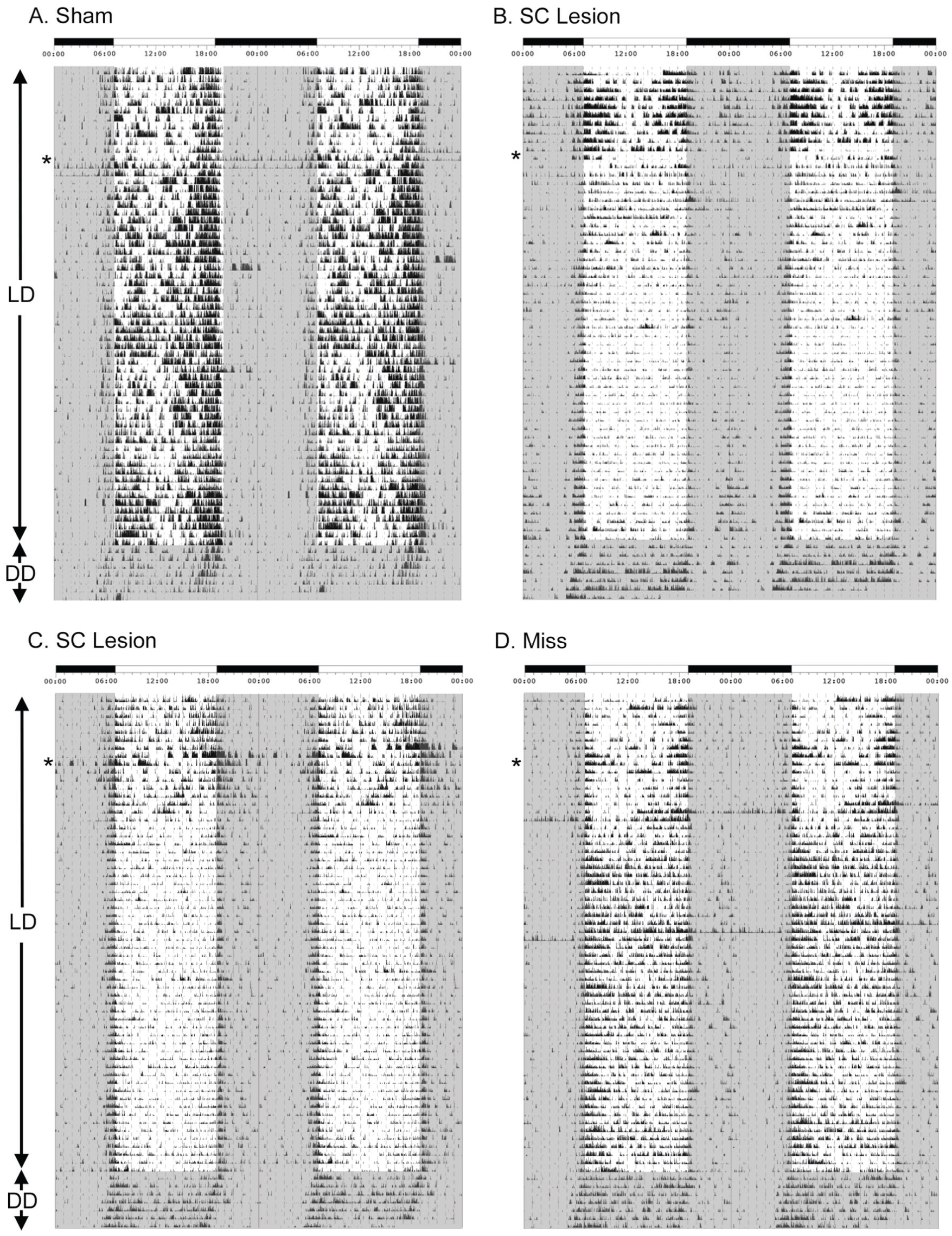
Effects of superior colliculus (SC) lesions on general activity patterns before and after surgery in 12:12 LD followed by complete darkness. Actograms are double plotted and depict general activity patterns before and after surgery (surgery time is noted by the asterisk) in 12:12 LD conditions followed by constant darkness in a representative sham (A), 2 grass rats with complete SC lesions (B and C), and a miss (D), which had lesions that spared the SC completely. A light-dark bar is presented on the top of each figure, indicating darkness (black) and light (white). Times are presented in clock time. Gray-shaded regions on the actograms indicate darkness.
As shown in Table 1, an independent-samples t test revealed no difference in period (t12 = −1.805, p = 0.096) or alpha (t12 = −0.723, p = 0.484) for shams versus animals with complete SC lesions, suggesting that the circadian clock was not affected by destruction of the SC.
As shown in Figure 3A, for shams, a repeated-measures ANOVA for activity counts revealed no significant main effect of time of surgery (presurgery vs. postsurgery; F1,5 = 0.308, p = 0.603) but did reveal a significant main effect of ZT (F23,115 = 7.670, p < 0.001) and no interaction between the 2 variables (F23,115 = 0.339, p = 0.998). Therefore, for shams, activity counts did not differ between presurgery and postsurgery, but a rhythm in activity counts was detected, as indicated by the significant main effect of ZT. As shown in Figure 3B, for animals with complete SC lesions, a repeated-measures ANOVA for activity counts revealed a significant interaction between time of surgery and ZT (F23,161 = 4.951, p < 0.001). For animals with complete SC lesions, post hoc paired t tests revealed significant differences in activity levels presurgery versus postsurgery at ZTs 23, 0, 10, and 11 (t7’s > −4.785, p’s < 0.002). Importantly, these significant reductions in activity levels in animals with complete SC lesions were found at times near the transitions between lights-on and lights-off.
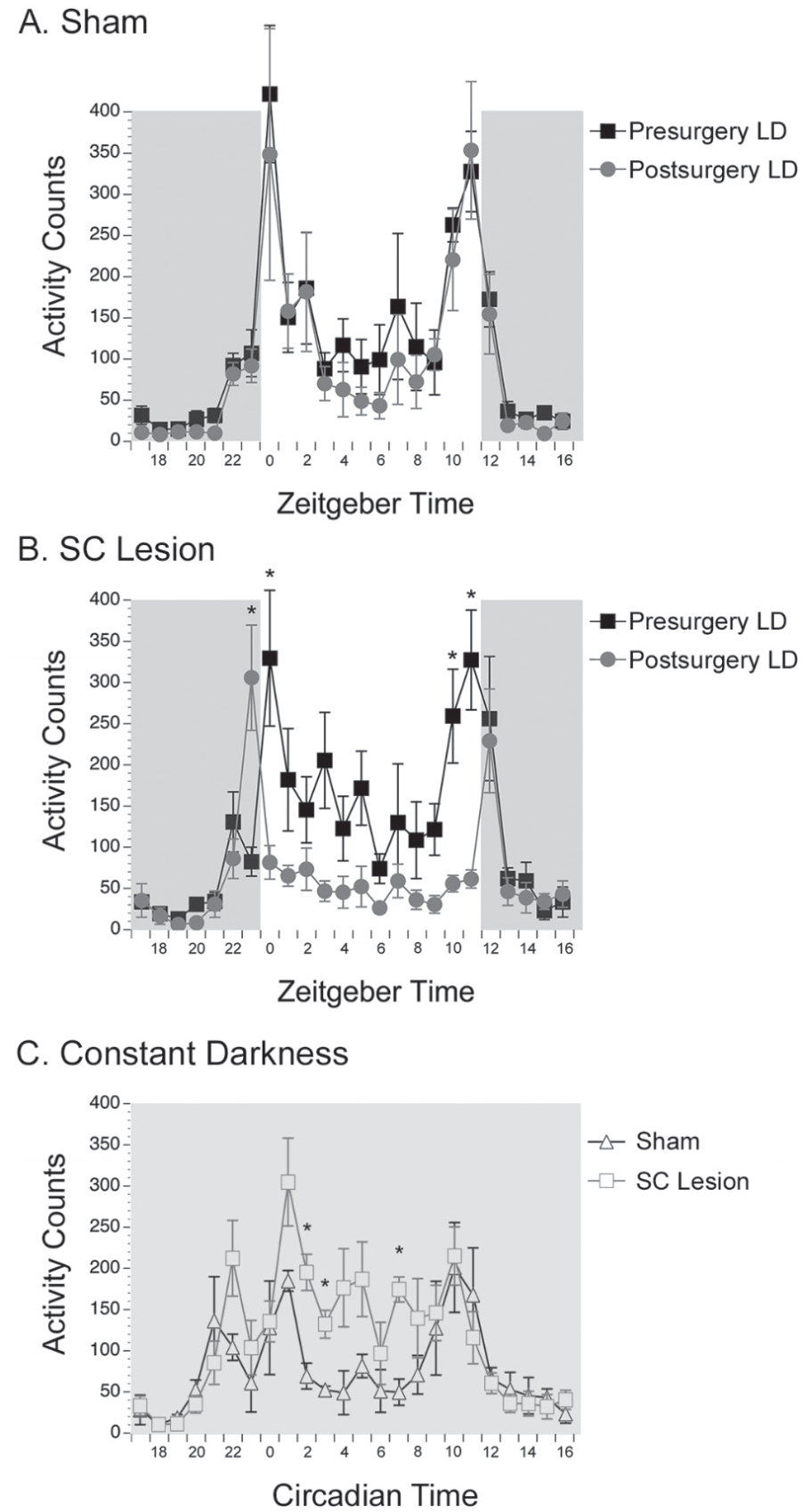
Quantitative analyses of general activity in LD and DD. Average patterns of general activity in 12:12 LD conditions during the day and night in shams (A) and animals with complete superior colliculus (SC) lesions (B). Activity counts presurgery are presented in black squares; postsurgery activity counts are gray circles. Before surgery, all animals were more active during the light phase. After surgery, shams remained more active during the light phase, whereas SC lesioned grass rats become less diurnal because of less activity during lights-on, especially at ZTs 23, 0, 10, and 11. Average patterns of general activity in DD conditions (C) are presented in shams (open triangles) and grass rats with complete SC lesions (open squares). Shams were significantly less active than grass rats with SC lesions at CTs 2, 3, and 7. *Significant difference in activity counts; means are presented with SEMs.
As shown in Figure 4, for total activity counts, a repeated-measures ANOVA revealed a significant interaction between lighting condition (LD presurgery vs. LD postsurgery vs. DD) and surgical condition (sham vs. complete SC lesion; F2,24 = 8.065, p = 0.002). For shams, post hoc tests using a Tukey test revealed that activity counts presurgery were not significantly different from those postsurgery (p = 0.487), but activity counts in DD were significantly reduced as compared with presurgery counts (p = 0.048) and postsurgery counts (p = 0.005) in LD. For animals with complete SC lesions, post hoc tests revealed that activity counts postsurgery were significantly reduced compared with presurgery levels in LD (p = 0.040). In addition, activity counts in DD were significantly reduced compared with presurgery levels in LD (p = 0.044) but significantly higher compared with postsurgery levels in LD (p = 0.043). As shown in Table 1, activity counts in DD were significantly higher in female grass rats with SC lesions as compared with shams (t12 = −3.105, p = 0.009), with significant differences in time points found during the subjective day at CTs 2, 3, and 7 (t12’s > 3.952, p’s < 0.002; Fig. 3C). In contrast, activity counts in LD were significantly lower in animals with SC lesions as compared with shams (t12 = 2.211, p = 0.047). Phase angle of entrainment was not affected by SC lesions in LD (t12 = −1.429, p = 0.178).
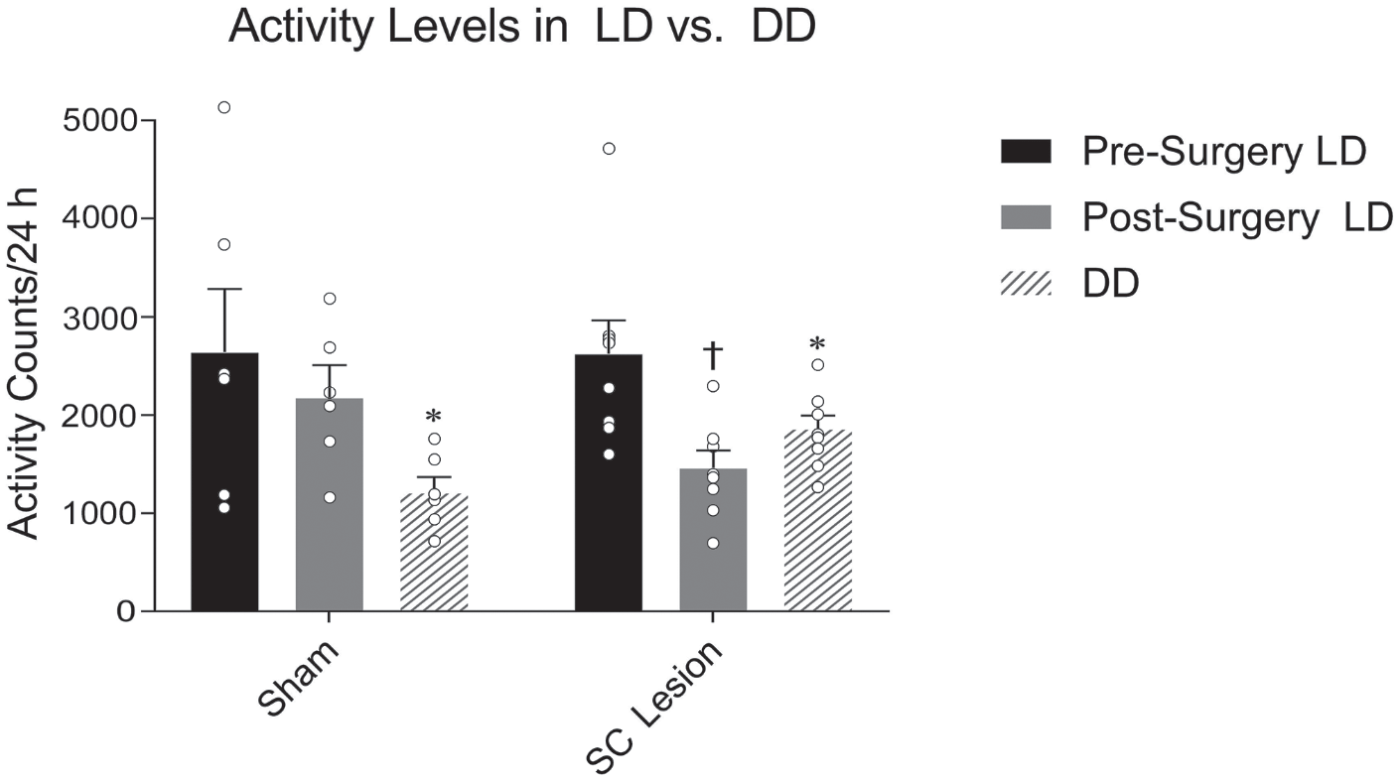
Quantitative analyses of general activity levels in LD versus DD before and after surgery. No significant differences in activity levels were observed for shams before surgery versus after surgery in LD, but significantly reduced activity was observed in DD as compared with LD conditions. For grass rats with complete superior colliculus lesions, significantly reduced activity was observed postsurgery in LD versus presurgery in the same lighting conditions. When these animals were placed in DD, activity levels were reinstated. Black bars represent presurgery LD, gray bars represent postsurgery LD, and hashed bars represent DD. Open circles represent individual data points. *Significantly different from both presurgery LD and postsurgery LD. †Significantly different from both presurgery and DD. Means are presented with SEMs.
In 12:12 LD Conditions, Complete SC Lesions Resulted in a Significant Reduction in the Percentage of Activity During Lights-on
As shown in Figure 5, for percentage of activity during lights-on, a repeated-measures ANOVA revealed a significant interaction between time of surgery (presurgery vs. postsurgery) and surgical condition (sham vs. lesion; F1,12 = 6.870, p = 0.022). As shown in Table 1, an independent-samples t test revealed a significant reduction in percentage of activity during lights-on for lesioned animals versus shams postsurgery (t12 = 4.420, p = 0.001) but no significant difference presurgery (t12 = 1.363, p = 0.198).
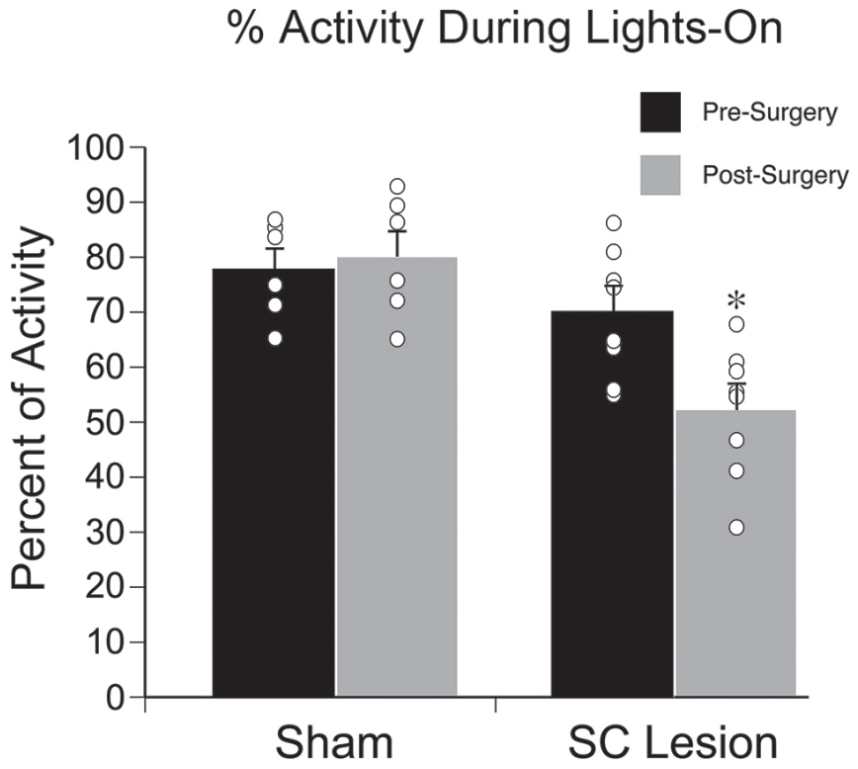
Percentage of general activity during the lights-on phase of a 12:12 LD cycle. No significant difference in percentage activity during lights-on was observed for shams, but grass rats with complete superior colliculus lesions expressed significantly less activity during the lights-on phase after surgery. Black bars represent presurgery, gray bars represent postsurgery. Open circles represent individual data points. *Significantly different from presurgery. Means are presented with SEMs.
Aberrant Responses to Light Pulses Are Observed Only at ZT22 in Grass Rats with Complete SC Lesions
As shown in Figure 6A in shams, a repeated-measures ANOVA revealed a significant interaction between the light pulse (baseline vs. light pulse) and ZT (14, 18, or 22; F2,10 = 7.823, p = 0.009). Paired-samples t tests revealed significantly higher counts during the light pulse versus baseline for all 3 time points (t5’s > −2.579, p’s < 0.049). A significant interaction between these 2 variables was also obtained for animals with complete SC lesions (F2,14 = 13.130, p = 0.001). Paired-samples t tests revealed significantly higher counts during the light pulse versus baseline for ZTs 14 and 18 (t7’s > −2.768, p’s < 0.028) but the opposite direction for ZT22 (t7 = 2.498, p = 0.041).
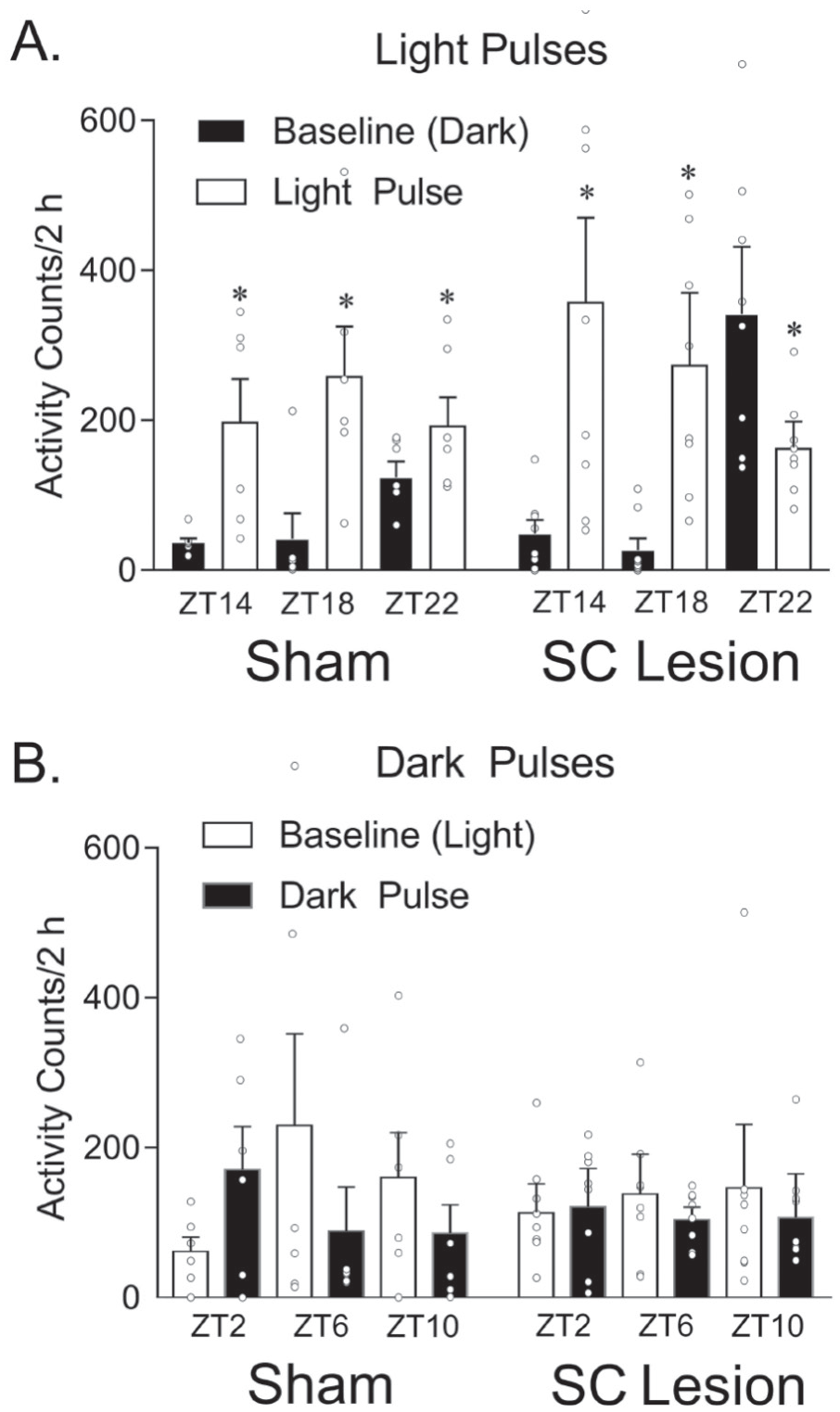
Masking responses to light and dark pulses. (A) Masking response to a light pulse in 12:12 LD conditions during the dark phase at ZT14, 18, and 22. In shams, activity levels were significantly higher at all 3 time points during the light pulse as compared with the day prior in darkness (baseline day). In grass rats with complete superior colliculus (SC) lesions, activity levels were significantly higher at ZT14 and 18 during the light pulse, but this effect was reversed at ZT22. (B) Masking response to a dark pulse in 12:12 LD conditions during the light phase at ZT2, 6, and 10. No significant differences were observed for dark pulses at any time point for either shams or grass rats with complete SC lesions. Open circles represent individual data points. *Significant difference between baseline day (black bars) and 2-h light pulse (white bars). Means are presented with SEMs.
Dark Pulses Were Not Affected by SC Lesions
As shown in Figure 6B for shams, a repeated-measures ANOVA did not reveal a significant main effect of the dark pulse (baseline vs. dark pulse; F1,5 = 0.758, p = 0.424) or ZT (2, 6, or 10; F2,10 = 0.201, p = 0.821), and there was no significant interaction between these 2 variables (F2,10 = 3.020, p = 0.094). For animals with complete SC lesions, we also did not find a significant main effect of the dark pulse (F1,7 = 1.593, p = 0.247) or ZT (F2,14 = 0.023, p = 0.977), and there was no significant interaction between these 2 variables (F2,14 = 0.610, p = 0.557).
Anxiety-Like Behavior Was Not Affected by SC Lesions
As shown in Figure 7, anxiety-like behavior was not affected by destruction of the SC. The amount of time spent in the center of the open-field test was not significantly different for shams versus animals with complete SC lesions (t12 = 0.779, p = 0.451). In addition, the number of entries to the center, total distance in the open-field test and the number of times rearing were not significantly different for shams versus animals with complete SC lesions (t12’s < 1.326, p’s > 0.210).
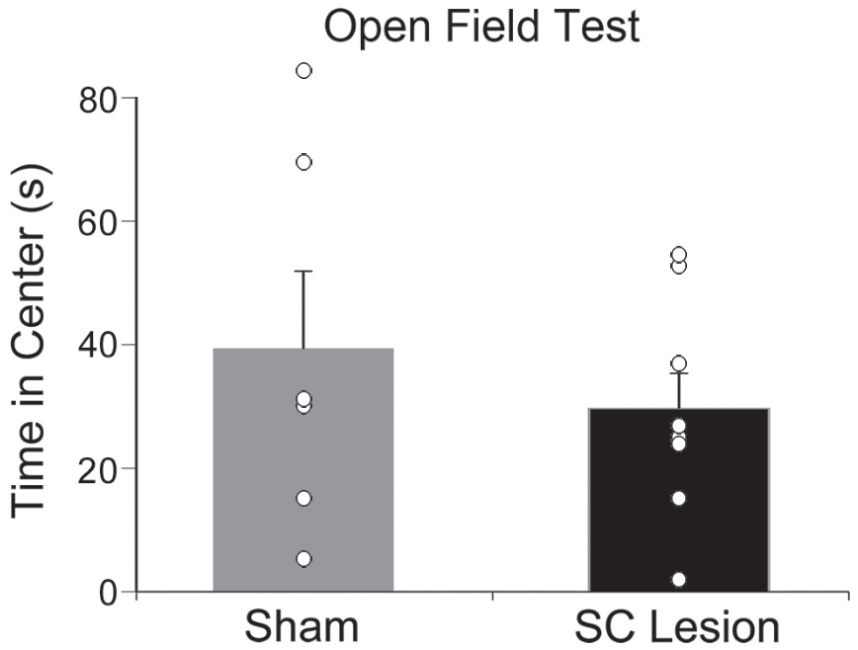
Anxiety-like behavior was unaffected by superior colliculus (SC) lesions. The average time spent in the center zone of the open-field test (seconds) in shams (gray bar) and grass rats with complete SC lesions (black bar). No significant differences were found in the open-field test for shams versus SC lesions. Open circles represent individual data points. Means are presented with SEMs.
Discussion
The SC is a retinorecipient brain structure that is well positioned to be critical for masking responses to light in diurnal species (Arvicanthis niloticus). We hypothesized that complete lesions to this brain area in female grass rats would result in aberrant behavioral responses to light and disrupted circadian rhythms. Importantly, female grass rats with these lesions were significantly less active during the light phase of the 12:12 LD cycle, and this activity was reinstated during the subjective day in DD, suggesting that masking was significantly affected by these lesions. Interestingly, the only time point at which we observed a significant effect of acute pulses of light or darkness was during a 2-h light pulse at ZT22, near the transition from sleep to wakefulness in these diurnal rodents. While diurnality was affected in 12:12 LD conditions, because of the suppression of activity in the light, it was not affected in constant conditions, suggesting that the circadian clock remained intact but masking was driving the reduction in activity levels. Critically, these reductions in activity levels were not directly attributable to anxiety, as no differences emerged in the open-field test. Taken together, our results suggest that the SC is an important brain area for sustaining activity levels during lights-on in diurnal species.
In nocturnal rodents, the SC has been shown to be critical for maintaining sleep when the lights are on (Miller et al., 1998). It has been recently shown that GABAergic neurons in the SC are critical for inducing wakefulness when the lights are off (Zhang et al., 2019). In addition, light input is critical for the SC to develop properly (Prichard et al., 2007). However, until now, it was unclear how light or darkness affects behavior in diurnal species with SC lesions. It is important to understand the differences between nocturnal and diurnal organisms with respect to masking, as it may reveal mechanistic differences between the 2 chronotypes. Our results suggest that dark pulses did not affect behavior in shams or grass rats with SC lesions. These results align with previous studies, showing that grass rats generally do not respond to dark pulses (Shuboni et al., 2012), which is quite different from the way nocturnal mice respond to dark pulses by increasing their activity. In contrast, grass rats were significantly less active during the light phase of a 12:12 LD cycle, and their response to an acute presentation of light at ZT22 was reversed compared with shams. Based on these results, we conclude that female grass rats are significantly less active during the lights-on phase for long periods of time (up to 12 h) following SC lesions, but they are able to sustain activity over shorter periods of time (up to 2 h) at ZT14 and 18 but not at ZT22. Interestingly, when examining the activity profile of SC lesioned female grass rats in 12:12 LD conditions (see Fig. 3B), the significant reductions in activity presurgery to postsurgery are at transition time points (ZT23, 0, 10, and 11). These results suggest that the SC is especially important for modulating activity patterns during these transitions from dark to light, and vice versa, which are important for enabling the organism to efficiently become active when the lights come on and inactive when the lights turn off.
Importantly, at ZT22, female grass rats with SC lesions had significantly higher baseline activity counts than at any other time point. This increase in baseline activity resulted in the grass rats responding less to the positive effect of light. This result is similar to our findings in grass rats with IGL lesions (i.e., increased activity in baseline activity counts, especially near transition points of dark to light; Gall et al., 2013). This increase in activity at transition points is likely due to the anticipation that the lights are about to turn on. Similarly, as shown in our current findings, female grass rats with SC lesions were significantly more active than shams in constant darkness. The significant increase in activity counts in grass rats with SC lesions suggests that these animals respond differently to darkness than shams. Whereas dark pulses during the light phase had no effect on them, sustained darkness did increase activity levels. Similarly, whereas 2 of the 3 light pulse time points had no effect on grass rats with SC lesions, sustained periods of light during a 12:12 LD cycle significantly reduced activity levels. These findings suggest that sustained periods of light or darkness had the most profound effect on grass rats with SC lesions. Therefore, we conclude that the SC is critical for maintaining normal responses to sustained periods of light or darkness or to behavior near transition points between darkness and light.
Whereas masking responses were aberrant in female grass rats with SC lesions, the circadian system remained intact. Specifically, period and alpha were not significantly different in female grass rats with SC versus shams (see Table 1). Whereas multiple reports have suggested that the SC is involved in acute masking responses, none have suggested that it affects the circadian system (Zhang et al., 2019; Lupi et al., 2008; Miller et al., 1998). Given the SC’s known role in modulating somatosensation, non–image-forming behaviors, auditory responses, and induction of sleep, our data suggest that the SC also plays an important role in masking but not in modulating circadian rhythmicity in a diurnal rodent. Although we did find a reduction in diurnality in 12:12 LD conditions, activity levels were restored in constant darkness, suggesting that the reduction in activity was completely driven by light input. Interestingly, whereas nocturnal rodents with SC lesions become hyperactive (Pope and Dean, 1979; Delay, 1981), diurnal mammals, such as tree shrews (Casagrande et al., 1972), monkeys (Denny-Brown, 1962), and, as shown here, Nile grass rats with SC lesions, become hypoactive. We hypothesize that the SC acts to drive species-typical behavior when lights are present. Therefore, in diurnal species, the SC maintains wakefulness with the lights on, and in nocturnal species, the SC maintains sleep with the lights on. Without a functional SC, behavior becomes hypoactive in diurnal species and hyperactive in nocturnal species.
Importantly, our results cannot be explained by anxiety-like behaviors alone, as there were no significant effects obtained in the open-field test. Specifically, the number of entries to the center, time spent in the center, and overall distance traveled were not significantly different in shams as compared with female grass rats with complete SC lesions. Interestingly, although total activity counts in the home cage were significantly reduced in female grass rats with SC lesions, activity counts did not differ in the open-field test over a 5-min time frame. The differences in locomotor activity, therefore, emerge only over a longer time frame and in a familiar environment. In nocturnal mice, stimulating the SC had no effect on anxiety-like behavior or locomotor activity in an open-field test but rather attenuated threat recognition, leading to approach behavior directed toward a threatening object (Almada et al., 2018). It is widely accepted that the SC, together with the amygdala and pulvinar nucleus of the thalamus, is involved in fear (Almeida et al., 2015; Carr, 2015), which can be distinguished independently from anxiety-like behaviors (Perusini and Fanselow, 2015). Although fear was not specifically assessed in our study, it is clear that anxiety-like behaviors, as measured by the open-field test, were not manifested by lesions of the SC in diurnal female grass rats.
The findings of our study are limited by several methodological procedures. First, our masking pulses of light and darkness lasted for 2 h. Although they were evenly distributed over the day and night equally, it would have been interesting to see how light and dark pulses affected behavior even closer to transitions between light and darkness. The time point from ZT22 to ZT0 was the only one to show an effect, most likely because it was very close to the transition point of dark to light. In retrospect, it would have been interesting to see how a light pulse would affect animals from ZT12-14, at the transition from light to dark. Instead, because of the equal spacing of masking time points, which were chosen because of similar time points in previous studies (Shuboni et al., 2012, 2016), we did not assess the transition from light to dark. It would also be interesting to examine the effects of longer-duration masking pulses, especially given that the effects are seen only under longer-term light stimulation (i.e., in 12:12 LD) and at transition time points. Finally, because electrolytic lesions were used, fibers of passage were also destroyed. It would be optimal to use chemical lesions in a future study so that fibers of passage are not destroyed and so that specific cell types, such as GABAergic neurons, within the SC can be targeted.
Conclusions
Nile grass rats are an excellent model to examine the mechanisms by which light affects behaviors as they are diurnal and have a larger brain region devoted to vision (Blanchong et al., 1999; Gaillard et al., 2013). We have recently shown that the IGL and OPT are critical for the normal masking response to light in these organisms, and here we extend these findings following lesions of the SC. Specifically, female Nile grass rats with complete SC lesions expressed significantly reduced activity in the presence of an 12:12 LD cycle, which was reinstated in constant darkness, exhibited aberrant responses to acute pulses of light at ZT22, but did not exhibit significant effects of dark pulses, anxiety-like behavior, or circadian rhythms in constant conditions. Altogether, our data suggest that the SC is critical for maintaining normal responses to the presentation of light, which is important for maintaining diurnality in these organisms. These results have important implications for the mechanisms underlying masking responses to light in diurnal species.
Footnotes
Acknowledgements
We would like to acknowledge Carolina Marshall for her technical assistance with the project. This research was generously supported by the Howard Hughes Medical Institute through the Undergraduate Science Education Program, a Nyenhuis Grant from Hope College, and the Towsley Research Scholars Award at Hope College. This study was also supported by startup funds from the Social Sciences Division at Hope College. Research data will be made available upon request.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
