Abstract
The ventral subparaventricular zone (vSPVZ) receives direct retinal input and influences the daily patterning of activity in rodents, making it a likely candidate for the mediation of acute behavioral responses to light (i.e., masking). We performed chemical lesions aimed at the vSPVZ of diurnal grass rats (Arvicanthis niloticus) using N-methyl-D,L-aspartic acid (NMA), a glutamate agonist. Following NMA lesions, we placed grass rats in various lighting conditions (e.g., 12:12 light-dark, constant dark, constant light); presented a series of light pulses at circadian times (CT) 6, 14, 18, and 22; and placed them in a 7-h ultradian cycle to assess behavioral masking. Extensive bilateral lesions of the vSPVZ disrupted the expression of circadian rhythms of activity and abolished the circadian modulation of masking responses to light, without affecting light-induced masking behavior per se. We also found that in diurnal grass rats, NMA was capable of destroying not only neurons of the vSPVZ but also those of the suprachiasmatic nucleus (SCN), even though excitotoxins have been ineffective at destroying cells within the SCN of nocturnal rodents. The vulnerability of the grass rat’s SCN to NMA toxicity raises the possibility of a difference in density of receptors for glutamate between nocturnal and diurnal species. In cases in which damage extended to the SCN, masking responses to light were present and similar to those displayed by animals with damage restricted to the vSPVZ. Thus, extensive bilateral lesions of the SCN and vSPVZ disrupted the expression of circadian rhythms without affecting acute responses to light in a diurnal species. Our present and previous results suggest that retinorecipient brain areas other than the SCN or vSPVZ, such as the intergeniculate leaflet or olivary pretectal nucleus, may be responsible for the mediation of masking responses to light in the diurnal grass rat.
Light influences the daily pattern of activity in mammals by entraining circadian rhythms and also by acutely affecting activity, a phenomenon known as masking (Redlin, 2001). Entrainment and masking enable organisms to match their behavior to the demands of a cyclic environment while maintaining a system that can respond to immediate changes in the environment (Mrosovsky and Thompson, 2008). In diurnal species, light increases activity, whereas in nocturnal species, light induces sleep (Borbély, 1976). Here, we seek to understand the neural mechanisms underlying masking behavior in response to light, using a diurnal animal model, the Nile grass rat (Arvicanthis niloticus); very little is known about the neural mechanisms of masking in diurnal species. Most of our current knowledge about masking comes from data on nocturnal species.
In nocturnal rodents, both the intergeniculate leaflet (IGL) and olivary pretectal nucleus (OPT) are involved in masking, as lesions of the IGL enhance the inhibitory effects of light on activity in hamsters (Redlin et al., 1999), while lesions of the OPT alter rapid eye movement (REM) sleep regulation by light in rats (Miller et al., 1999). However, lesions of the suprachiasmatic nucleus (SCN) have produced inconclusive results. In hamsters, one group reported that SCN lesions significantly impaired masking (Li et al., 2005), whereas another group saw no effect (Redlin and Mrosovsky, 1999). Li et al. (2005) suggest that this discrepancy may be due to the fact that SCN lesions eliminated retinal projections to the ventral subparaventricular zone (vSPVZ) and other hypothalamic areas.
The vSPVZ is located dorsal to the SCN, receives significant input from the SCN, and projects to many of the same regions as the SCN (Morin, 1994; Schwartz et al., 2011). The vSPVZ plays an important role in the regulation of circadian rhythms of sleep and activity in nocturnal rodents (Lu et al., 2001), and it exhibits increases in Fos expression following light exposure (LeGates et al., 2012).
In diurnal grass rats, the SCN, vSPVZ, OPT, and IGL all receive input from the retina (Smale and Boverhof, 1999; Gaillard et al., 2013; Langel et al., 2015) and contain cells that express an increase in Fos production after exposure to light in grass rats (Shuboni et al., 2015). In addition, we recently provided evidence that the IGL and OPT are involved in the masking response to light in the diurnal grass rat (Gall et al., 2013; Gall et al., 2014). However, it is not yet known if the SCN or vSPVZ contributes to masking in diurnal species. Therefore, we sought to test the hypothesis that the vSPVZ is part of the neural circuitry mediating masking responses to light in a diurnal rodent. Specifically, we delivered the excitotoxin N-methyl-D,L-aspartic acid (NMA) to the vSPVZ expecting that the damage would not extend into the SCN, given the results of previous lesion studies using this method (Peterson and Moore, 1980; Schwarcz and Kohler, 1983; Hastings et al., 1985; Schwartz et al., 2009). The histological analysis, however, revealed several cases in which there was extensive damage to the SCN. Our results suggest that the SCN and the vSPVZ are involved in the circadian regulation of activity but are not necessary for masking behavior to acute pulses of light in grass rats.
Materials and Methods
All experiments were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80-23) and were approved by the Institutional Animal Care and Use Committee of Michigan State University. All efforts were made to minimize the number of animals used.
Subjects
A total of 47 adult female Nile grass rats (A. niloticus) were obtained from a breeding colony at Michigan State University. Animals were run in 2 separate cohorts, following identical protocols, and the data for the two cohorts were combined. Animals were singly housed in Plexiglas cages (34 × 28 × 17 cm) and provided with food (PMI Nutrition Prolab RMH 2000, Brentwood, MO, USA) and water ad libitum. A total of 35 animals received bilateral lesions of the vSPVZ, and 12 animals received sham surgery as controls.
Experimental Procedures
Infrared motion detectors (Visonic, Tel Aviv, Israel) were placed on the top of each cage during the duration of the study to monitor general locomotor activity in 5-min bins using VitalView 4.0 software (Mini-Mitter, Bend, OR, USA) on a computer located in an adjacent room. Activity patterns of grass rats were monitored in 12:12 light-dark (LD) conditions (300 lux of white light during the light phase) for at least 10 days prior to surgery. Animals were randomly assigned to either a vSPVZ-lesioned group or a sham control group and allowed to recover from surgery for at least 2 weeks under a 12:12 LD cycle.
Animals were then tested for their masking response to light using a previously described procedure (Shuboni et al., 2012). Briefly, animals were placed in constant darkness (DD) for 2 days and received a 1-h light pulse (LP) on the second day in DD. They were returned to a 12:12 LD cycle for 2 days, and the procedure was repeated for the other 3 LPs. Each animal received a total of 4 LPs at circadian times (CTs, CT0 = activity onset) 6, 14, 18, and 22. The order of LPs was randomized. Next, animals were exposed to an ultradian light-dark cycle, in which they received 3.5 h of light followed by 3.5 h of darkness repeatedly for 9 days. Finally, animals were placed back in a 12:12 LD cycle until all animals reentrained. Then, circadian rhythms were examined by placing animals in DD for at least 2 weeks, followed by constant light (LL) for at least 2 weeks. Animals were then sacrificed and brains were collected for histological assessment of the lesions.
Surgical Procedures
Grass rats were anesthetized with isoflurane (2.5%-5%; Abbott Laboratories, Abbott Park, IL, USA) and secured in a stereotaxic apparatus (Stoelting Co., Chicago, IL, USA) with the tooth bar set to −4.5 mm. Animals were injected subcutaneously with ketoprofen (5 mg/kg body weight). The top of the head was shaved, disinfected with betadine, and injected subcutaneously with lidocaine (0.03 mL). An incision was made in the scalp to expose the surface of the skull. Using a surgical drill, 2 small holes were made at the following coordinates: anterior-posterior (AP): +0.16 mm from bregma; medial-lateral (ML): ±0.05 mm from the midline; dorsal-ventral (DV): –6.0 to −6.2 mm ventral to the dura. Injections were made using a 0.5-µL Hamilton syringe (Hamilton, Reno, NV, USA) filled with either 0.3 M NMA (Sigma, St. Louis, MO, USA) dissolved in 0.2 M saline or a vehicle control, which consisted of 0.2 M saline alone. The needle was lowered into the brain slowly until it reached the target coordinates and allowed to rest for 1 min before and after injection. Then, 180 nL of either NMA or saline was injected bilaterally using a manual stereotaxic microinjector (Stoelting Co.) given over a 5-min period. The incision was closed using autoclips and the animals were injected subcutaneously with 2.0 mL of 0.9% sterile saline. At 24 and 48 h following surgery, all animals received Meloxicam (0.1 mg/kg body weight) orally via a piece of apple.
Histology
At the end of the experiment, animals were euthanized at ZT2 with intraperitoneal injections of sodium pentobarbital (Ovation Pharmaceutical, Deerfield, IL, USA) and perfused transcardially with 0.01 M phosphate-buffered saline (PH), pH 7.2, followed by 4% paraformaldehyde (PFA; Sigma-Aldrich) with 75 mM lysine (Sigma-Aldrich) and 10 mM sodium periodate (Sigma-Aldrich) in 0.1 M phosphate buffer to make a periodate-lysine-paraformaldehyde solution (PLP). The brains were postfixed for 4 h in PLP, then transferred to a 20% sucrose solution (J. T. Baker, Phillipsburg, NJ, USA) and stored at 4 °C for at least 24 h. Brains were sectioned in 30-µm coronal sections using a sliding microtome, separated into 3 series, and stored in cryoprotectant at −20 °C. One series was mounted onto gelatin-coated slides, stained for Nissl using a thionin protocol, and coverslipped with Permount (Fisher Scientific, Fair Lawn, NJ, USA).
The extent of the lesions was determined by 2 experimenters, who independently analyzed the tissue; brain sections were analyzed using a light microscope (Leitz, Wetzlar, Germany) equipped with a drawing tube at 5× magnification. The lesioned area was drawn by both experimenters in one brain section that was rostral to the SCN; 3 sections that included the rostral, middle, and caudal SCN; and a fifth section caudal to the SCN. These drawings were compared between the 2 independent experimenters, and animals were classified as bilateral lesion, unilateral lesion, or miss. The experimenters were in 100% agreement in the classification of groups. To quantify the extent of the SCN and vSPVZ lesions, the number of Nissl-stained cells was counted bilaterally in the same 3 sections by an experimenter blind to condition using the Cell Counter Plugin in ImageJ software (version 1.49; National Institutes of Health, Bethesda, MD, USA). The counting box for the vSPVZ (215-µm × 160-µm rectangle) was placed dorsal to the SCN (Shuboni et al., 2015). The SCN of sham animals was traced and the average size for rostral, middle, and caudal sections was used to delineate the SCN in lesioned animals. We compared the total number of cells that remained in lesioned animals in the vSPVZ and SCN to the average number of cells counted in sham animals in these areas. In addition to the total number of cells, cell size was quantified by randomly selecting 50 cells and measuring the cell size using ImageJ’s Analyze Particle tool.
A total of 12 animals had bilateral lesions of the vSPVZ (Figures 1A and 2A). Of these 12 animals, 6 also sustained significant damage to the SCN (Figures 1B and 2A), with 1 animal having significant damage to the optic tract (this animal was excluded from group analyses). In all animals that sustained SCN damage, the vSPVZ was bilaterally lesioned; therefore, to assess the effects of bilateral vSPVZ lesions, we have analyzed the vSPVZ and vSPVZ/SCN animals as a single “lesion” group. Data from these animals, along with shams, are presented in the analyses below. In the brains of sham animals, needle tracts were observed, but the SCN and vSPVZ were intact (Figure 1C). In 15 animals, NMA did not damage the SCN or vSPVZ on either side of the brain but did damage other brain structures (these cases are referred to as “misses”; Figure 2B), and in 8 animals, damage to the SCN or vSPVZ was unilateral (statistical analyses for misses and unilateral lesions are not presented). The high number of misses is related to the variability of the placement of the needle of the Hamilton syringe, which is inevitable because bregma is extremely variable and is not as reliable for determining coordinates in grass rats as it is in more standard laboratory rodents (e.g., commercially obtained Norway rats, mice or hamsters).
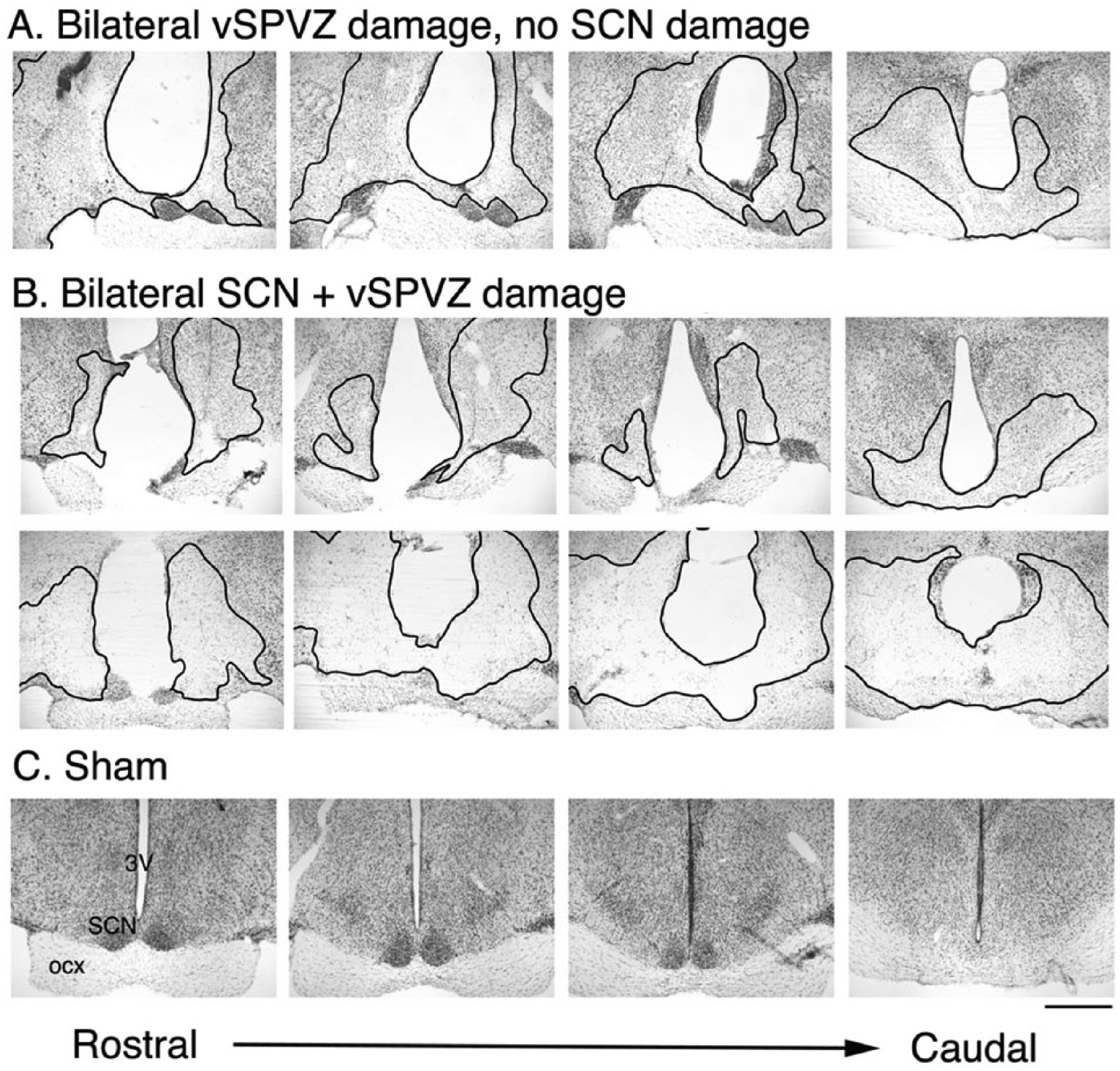
Histology from representative animals with bilateral vSPVZ damage (A), bilateral SCN + vSPVZ damage (B), and sham-operated grass rats (C). Thionin stained coronal sections through the extent of the SCN from rostral (left) to caudal (right). Outlined regions in black indicate the lesioned area. Scale bar represents 1 mm. Abbreviations: ocx = optic chiasm; SCN = suprachiasmatic nucleus; vSPVZ = ventral subparaventricular zone; 3V = third ventricle.
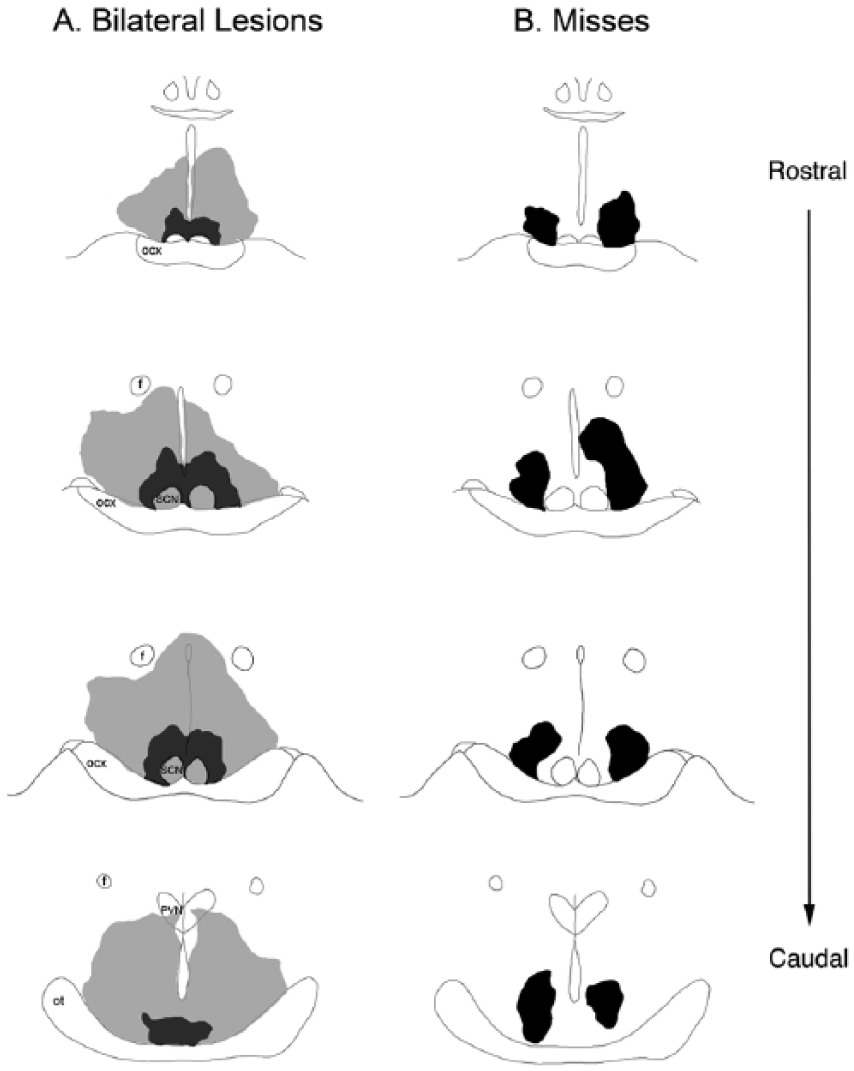
Smallest and largest chemical lesions that destroyed the ventral subparaventricular zone bilaterally (A) and misses that left this area intact (B), seen in 4 coronal sections. Gray shaded areas indicate the largest sized lesion in each group; black shaded areas indicate the smallest sized lesion in each group. Abbreviations: f = fornix; ocx = optic chiasm; ot = optic tract; PVN = paraventricular nucleus; SCN = suprachiasmatic nucleus.
Statistical Analysis
Activity data from VitalView software (Mini-Mitter) were viewed in Microsoft Excel (Microsoft Corp., Redmond, WA, USA) and analyzed statistically with SPSS (version 21; SPSS, Inc., an IBM Company, Chicago, IL, USA). To evaluate the effects of light pulses, activity counts from the same 1-h interval from the pulse day and preceding baseline day in DD were compared. A 3-way analysis of variance (ANOVA) was used to analyze the data with a 2 × 2 × 4 factorial design (surgical condition [sham vs. bilateral lesion] × lighting condition [baseline vs. 1-h pulse] × time of day [CT6, 14, 18, and 22]; surgical condition as the between-group factor and both lighting condition and time of day the within-group factors). This was followed up by analyzing each surgical group separately using 2-way repeated-measures ANOVAs to assess within-subject effects of lighting condition (darkness vs. light pulse) and time-of-day (CT6, 14, 18, and 22). Significant interactions were followed by evaluation of simple main effects of lighting condition using paired-samples t tests; simple main effects of time of day were evaluated using Tukey’s honestly significant difference test.
Masking behavior during the ultradian LD cycle (3.5 h light/3.5 h dark) was assessed by comparing the total amount of activity that occurred during the light period with the total activity that occurred during the dark period. For the ultradian LD cycle, a 2-way mixed ANOVA was used to assess within-subjects effects of lighting condition (light vs. dark) and the between-subjects effects of surgical condition (sham vs. bilateral lesion). Significant interactions were followed by the evaluation of simple main effects of lighting condition and surgical condition using paired-samples t tests and independent-samples t tests, respectively. Masking behavior during DD and LL was assessed by comparing the total amount of activity that occurred in DD with the total activity that occurred in LL and dividing by the total amount of days in each condition. A 2-way mixed ANOVA was used to assess within-subjects effects of lighting condition (DD vs. LL) and between-subjects effects of surgical condition (sham vs. bilateral lesion). Significant interactions were followed by the evaluation of simple main effects of lighting condition and surgical condition using dependent- and independent-samples t tests, respectively.
For the 1 animal that free ran through all lighting conditions, a paired-samples t test was used to assess the effect of lighting condition (light vs. dark). In addition, activity counts from 12-h light periods and 12-h dark periods for this animal were averaged across the 21-day interval before surgery and the 21-day interval that began 2 weeks after surgery. The activity counts after surgery began 2 weeks after surgery, to allow time for the animal to recover from surgery. Daily mean activity counts were calculated during the light phase and compared to the dark phase.
To analyze circadian rhythmicity, actograms were first viewed using ClockLab software (Actimetrics, Wilmette, IL, USA). We determined the daily onsets and offsets of activity and the period of the rhythm using ClockLab, but when visual inspection indicated that ClockLab detected these improperly, these measures were manually corrected (Schrader et al., 2009; Gall et al., 2013). We also calculated alpha (i.e., the duration of the active period). An ANOVA was used to assess the between-subjects effects of surgical condition (sham vs. bilateral lesion) on alpha and period.
Finally, to analyze total activity in LD conditions, we examined day-night activity by averaging the total activity during the day and night for 21 days for each animal prior to surgery and comparing it to the average activity during the day and night, also for 21 days, at 14 days after surgery. A 3-way ANOVA was used to analyze the data with a 2 × 2 × 2 factorial design (surgical condition [sham vs. bilateral lesion] × lighting condition [dark vs. light] × surgical time [surgical time]; surgical condition as the between-group factor and both lighting condition and surgical time as the within-group factors).
Significant interactions were followed by the evaluation of simple main effects of lighting condition and surgical condition using paired-samples t tests and independent-samples t tests, respectively. For all comparisons, differences were considered significant when p < 0.05 (2-tailed for all t tests).
Results
Quantification of Lesions
Nissl staining demonstrated the drastic change in cell size and number within the lesioned area compared to shams (Suppl. Figure S1). The size of these cells was significantly smaller in the lesioned tissue within both the vSPVZ (t98 = 18.1, p < 0.05) and SCN (t98 = 16.4, p < 0.05; Suppl. Figure S1). In the vSPVZ, an average of 23.9% of the cells survived in lesioned animals compared to shams (range, 0.0%-53.1%). Importantly, an average of only 3.3% (range, 0.0%-7.9%) of the surviving cells were in the size range of those from shams (Suppl. Figure S1).
In the SCN, an average of 32.5% of the cells survived in lesioned animals compared to shams (range, 3.6%-53.1%). Importantly, an average of only 21.1% (range, 9.8%-30.6%) of the surviving cells were in the size range of those from shams (Suppl. Figure S1). The smaller cells observed following lesions to either the vSPVZ or SCN are likely to be glia rather than surviving neurons.
Masking
Light Pulses
The effects of 1-h light pulses given to shams and those with bilateral lesions of the SCN/vSPVZ were assessed using a 3-way ANOVA, which revealed a significant lighting condition × time interaction (F3,63 = 4.8, p < 0.005; within-subjects effect) but no significant difference in surgical condition (F1,21 = .063, p = 0.805; between-subjects effect). When the SCN-lesioned animals were compared to the vSPVZ-lesioned animals, no significant differences were detected between the groups for masking (F1,10 = 1.6, p = 0.227; between-subjects effect). In addition, neither group was significantly different from shams (F1,21 = 1.1, p = 0.347; between-subjects effect). Therefore, the vSPVZ and vSPVZ/SCN animals are analyzed as a single “lesion” group. This was followed up by analyzing shams and animals with bilateral lesions separately using a within-subjects 2-way ANOVA for lighting condition and time. For animals with bilateral lesions to the SCN/vSPVZ (Figure 3A), there was a significant main effect of lighting condition (F1,10 = 29.0, p < 0.001) but no significant main effect of time (F3,30 = 0.2, p = 0.867) and no interaction between the 2 variables (F3,30 = 0.7, p = 0.570). Therefore, grass rats with bilateral vSPVZ lesions, some of which extended to the SCN, were still capable of masking to light. For shams (Figure 3B), there was a significant interaction between lighting condition and time (F3,33 = 5.9, p < 0.005). Analysis of simple main effects of lighting condition revealed that activity counts were significantly higher following a LP at all time points in shams (t11s > 2.6, ps < 0.05). Analysis of simple main effects of time of day revealed that activity counts were significantly higher at CT14 compared to CT6 and CT18 (ps < 0.05). Therefore, light induced a significant increase in activity (i.e., masking was not affected) in shams and lesioned groups, but the circadian pattern of masking was ablated in the lesioned animals.
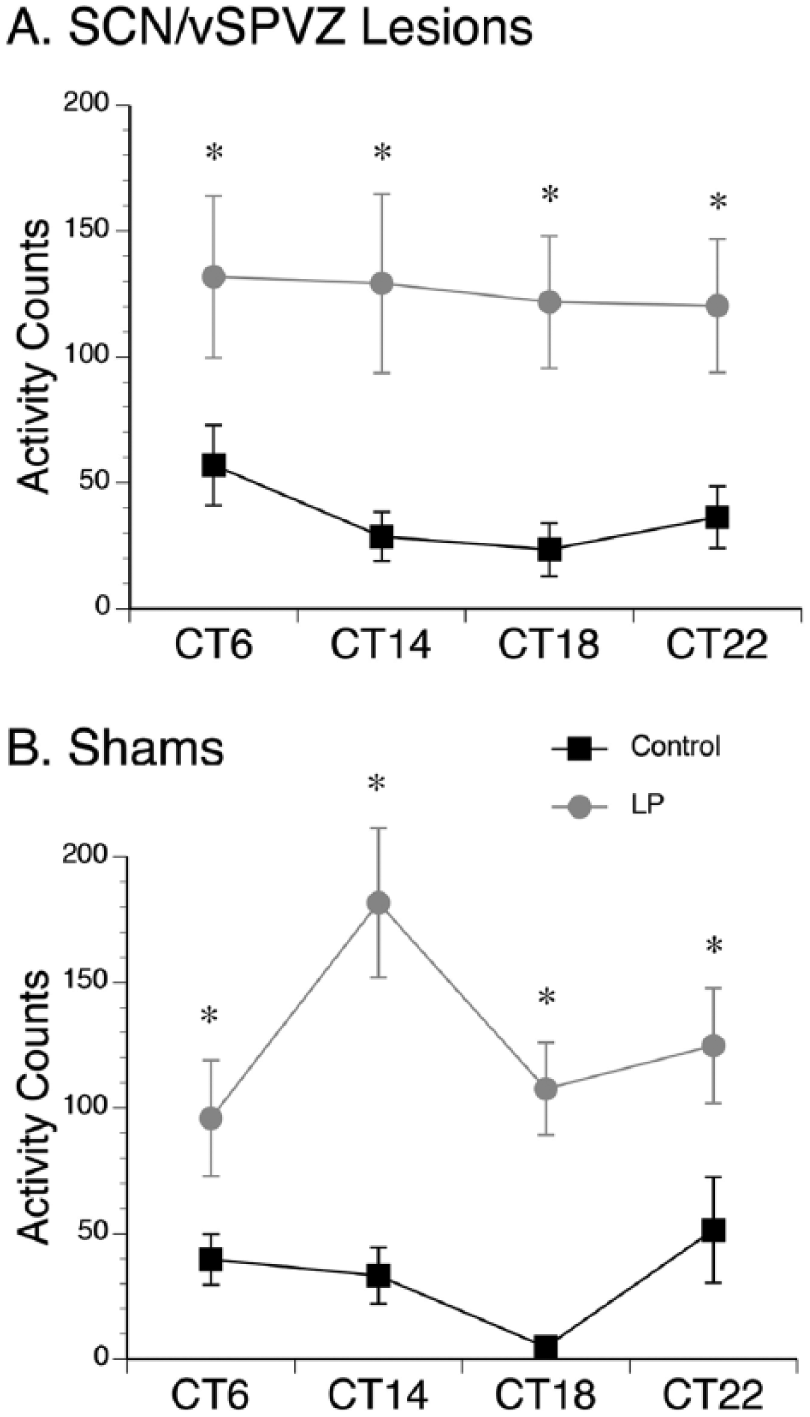
Masking responses to light pulses (LPs) at various time points. (A) Masking response to LPs at circadian time (CT) 6, 14, 18, and 22 in grass rats with lesions to the SCN/ventral subparaventricular zone (vSPVZ). Lesioned animals given LPs (gray lines) exhibited significantly higher activity counts than the same animals when maintained in constant darkness (black lines) on the day prior to the LP at all time points. (B) Sham animals also exhibited significantly higher activity counts during the LP (gray lines) compared to the day prior in constant darkness (black lines). *p < 0.05. Means are presented along with SEM.
Ultradian
The effects of a 7-h ultradian light-dark cycle (Figure 4) were assessed using a 2-way mixed ANOVA for lighting condition (within-subjects; light vs. dark) and surgical condition (between-subjects; sham vs. bilateral lesion). The ANOVA revealed a significant main effect of lighting condition (F1,21 = 12.5, p < 0.005) but no main effect of surgical condition (F1,21 = 0.6, p = 0.457) and no interaction between the two variables (F1,21 = 1.0, p = 0.319). Importantly, activity counts were significantly higher during the lights-on condition compared to the lights-off condition, with no significant differences between shams and bilaterally lesioned animals.
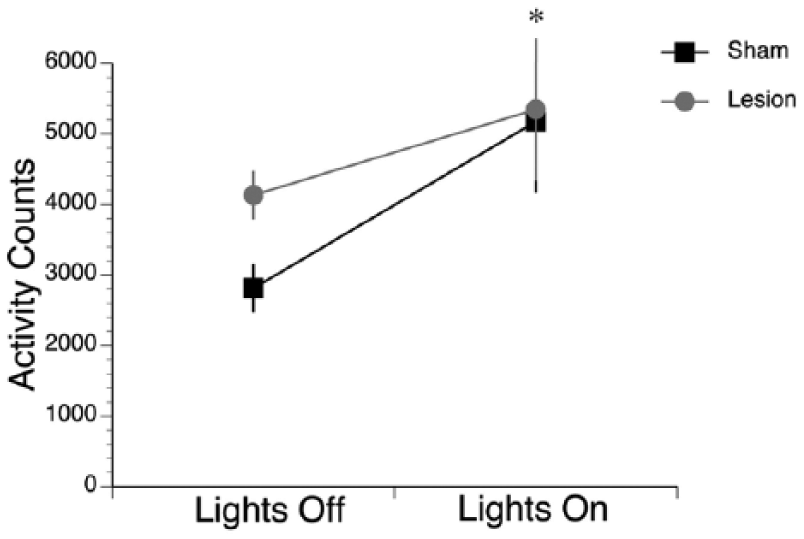
Grass rats maintained on an ultradian cycle exhibited higher amounts of activity during lights-on compared to lights-off. Shams (black lines) and lesioned grass rats (gray lines) both exhibited significantly higher activity counts in lights-on compared to lights-off. No differences were detected between the groups. *p < 0.05. Means are presented along with SEM.
Circadian Rhythms
As shown in Figure 5A, grass rats with bilateral vSPVZ damage and no SCN damage had intervals of arrhythmicity in DD and LL (histology for this animal is in Figure 1A), along with other intervals where a rhythm was detectable, suggesting that in those animals, the circadian clock was still functional. In contrast, as shown in Figure 5B, animals with bilateral damage that included both the SCN and the vSPVZ became completely arrhythmic when placed in DD and LL (histology for one such animal is presented in Figure 1B, second row). This circadian profile after damage restricted to the vSPVZ of grass rats is consistent with previous observations (Schwartz et al., 2009). Shams, when placed in DD or LL (Figure 5C; histology for this animal is in Figure 1C), continued to express circadian rhythms. Importantly, SCN/vSPVZ-lesioned animals that still had a detectable rhythm did not differ from shams in terms of alpha (F1,17 = 0.3, p = 0.577; Figure 6A) or period (F1,17 = 0.4, p = 0.545; Figure 6B) in DD and LL. Therefore, although circadian rhythms become disrupted following lesions to the SCN and/or vSPVZ, when rhythms are detectable, alpha and period in DD and LL did not differ from those of controls.
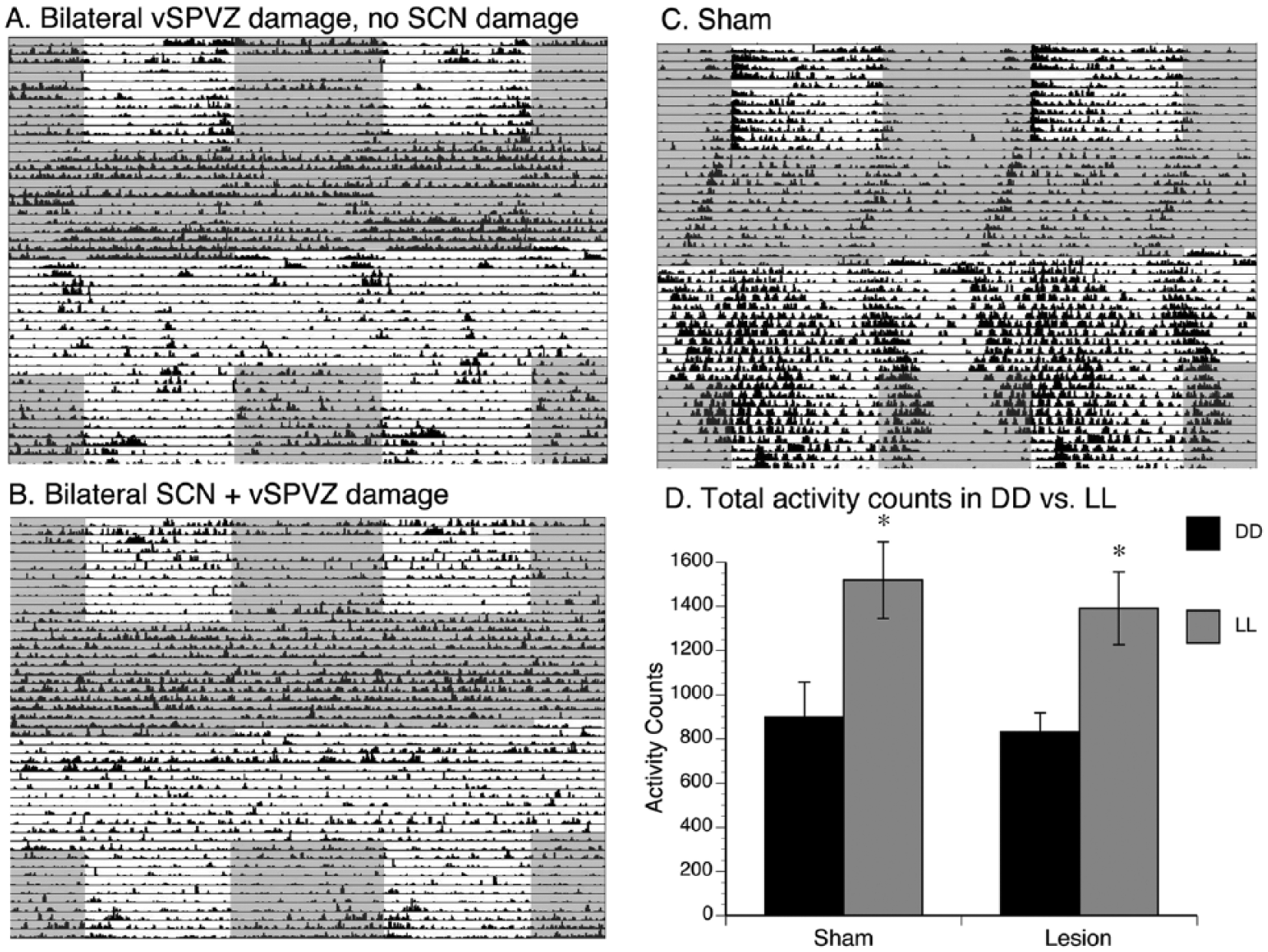
Effects of lesions on behavior in constant conditions. (A) Double-plotted actograms from 1 representative animal with bilateral ventral subparaventricular zone (vSPVZ) damage and no SCN damage (histology for this animal is in Figure 1A), (B) 1 representative animal with bilateral SCN damage and vSPVZ damage (histology for this animal is in Figure 1B, second row), and (C) a sham animal (histology for this animal is in Figure 1C) during periods of LD, DD, and LL, followed by LD. Note the arrhythmicity in SCN-lesioned animals and the periods of arrhythmicity in vSPVZ-lesioned animals, especially in constant conditions. Gray shaded areas indicate darkness, and white indicates light. (D) Total activity counts in DD and LL in shams and animals with bilateral lesions. *p < 0.05. Means are presented along with SEM.
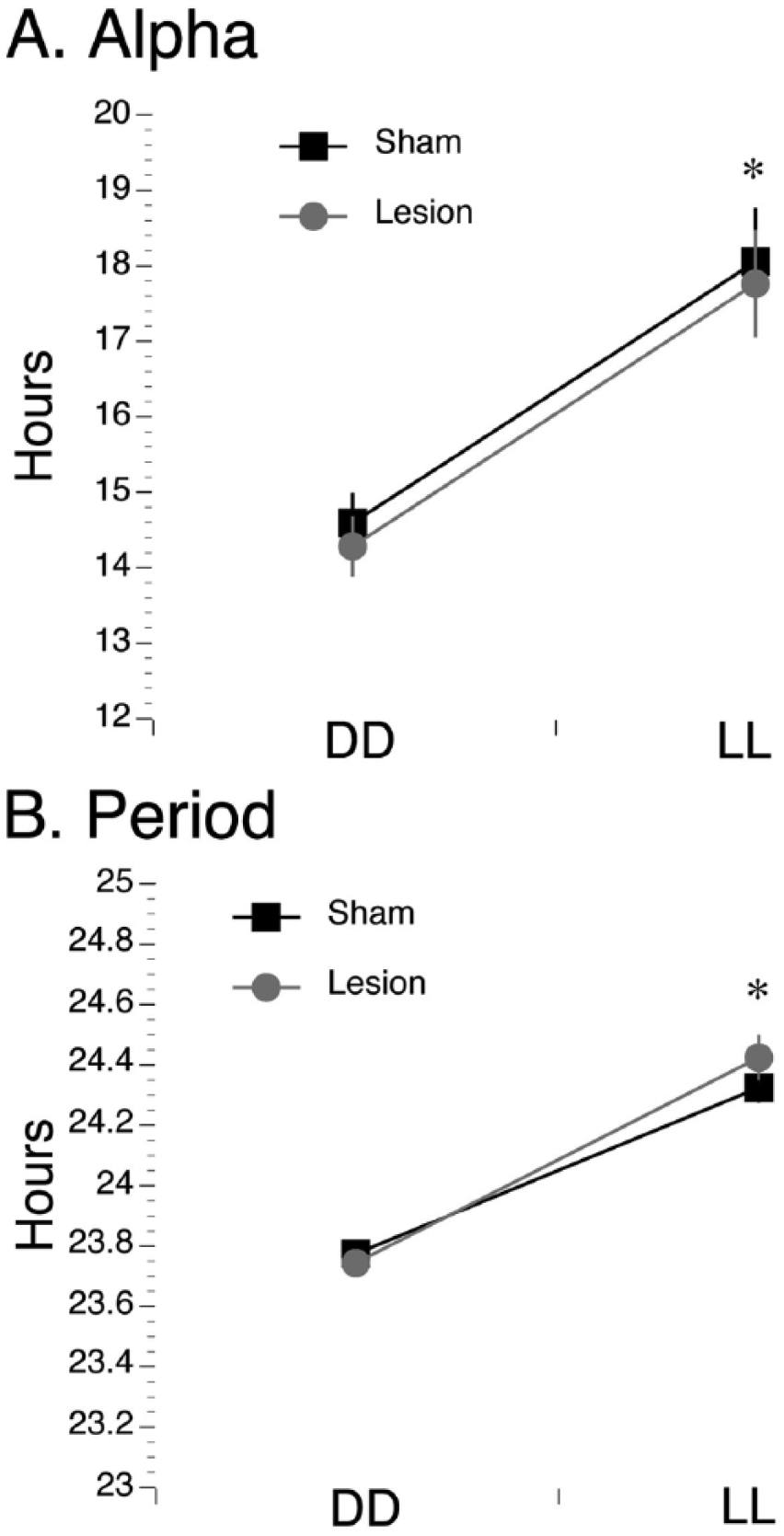
Alpha and period in constant conditions in lesioned and sham-operated grass rats. Alpha (A) and period (B) were significantly higher in LL than in DD for both shams (black lines) and lesioned grass rats (gray lines). No differences were detected between the groups. *p < 0.05. Means are presented along with SEM.
DD vs. LL
The effects of DD and LL on total activity (Figure 5D) were assessed using a 2-way mixed ANOVA for lighting condition (within-subjects; DD vs. LL) and surgical condition (between-subjects; sham vs. bilateral lesion). The ANOVA revealed a significant main effect of lighting condition (F1,21 = 15.1, p < 0.001) but no main effect of surgical condition (F1,21 = 0.4, p = 0.533) and no interaction between the 2 variables (F1,21 = 0.04, p = 0.839). Importantly, activity counts were significantly higher during LL compared to DD, with no significant differences between shams and bilaterally lesioned animals.
Total Activity Levels
For total activity counts (Figure 7), a 3-way ANOVA found a significant main effect of lighting condition (F1,21 = 13.1, p < 0.005; within-subjects) but no significant main effects of surgical time (F1,21 = .646, p = 0.431; within-subjects) or surgical condition (F1,21 = .057, p = 0.814; between-subjects) and no interactions between any variables. Therefore, activity levels were higher in the light than during the dark of an LD cycle, but no difference was found between these activity levels in shams versus bilaterally lesioned animals.
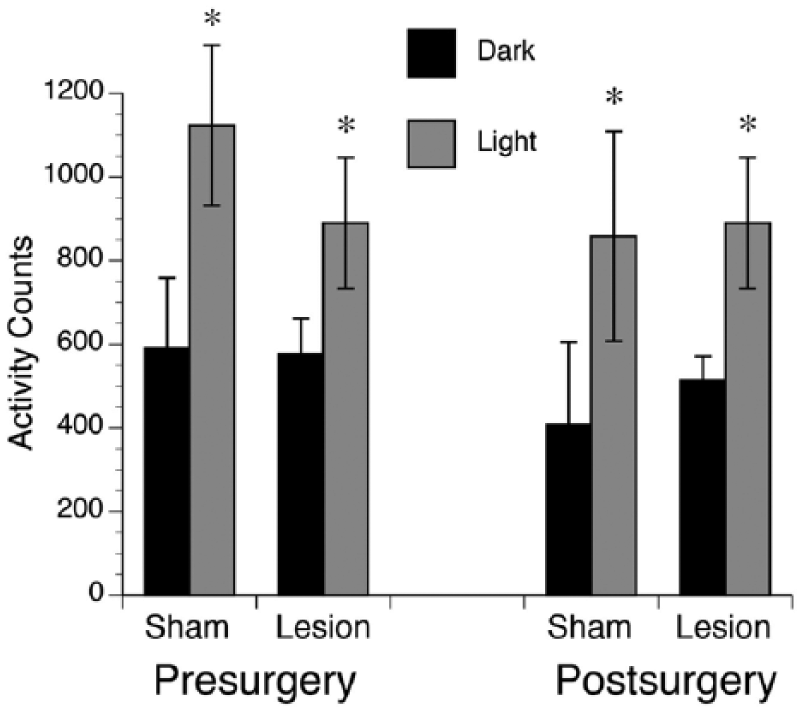
Total activity counts presurgery and postsurgery in shams and animals with bilateral lesions. Both prior to surgery and after surgery, average total activity counts were significantly higher during the light phase (gray bars) than the dark phase (black bars) of a 12:12 LD cycle. No significant differences were found for shams compared to lesioned animals. *p < 0.05. Means are presented along with SEM.
Free-Running Animal
One unique animal with significant damage to the SCN, vSPVZ, and optic chiasm free-ran through all lighting conditions (Figure 8). Even though this animal was not capable of entraining to light, it was still capable of masking to acute pulses of light. A paired t test revealed that activity counts were significantly higher during the light pulse intervals than during the equivalent intervals in the dark (t3 = 3.2, p < 0.05). In addition, average daily activity counts in this animal were higher during the light phase of an LD cycle (397.5 ± 26.4) compared to the dark phase (247.9 ± 25.1), with 61.6% of the activity occurring during lights-on and 38.4% of activity occurring during lights-off, suggesting that the animal was still capable of masking.
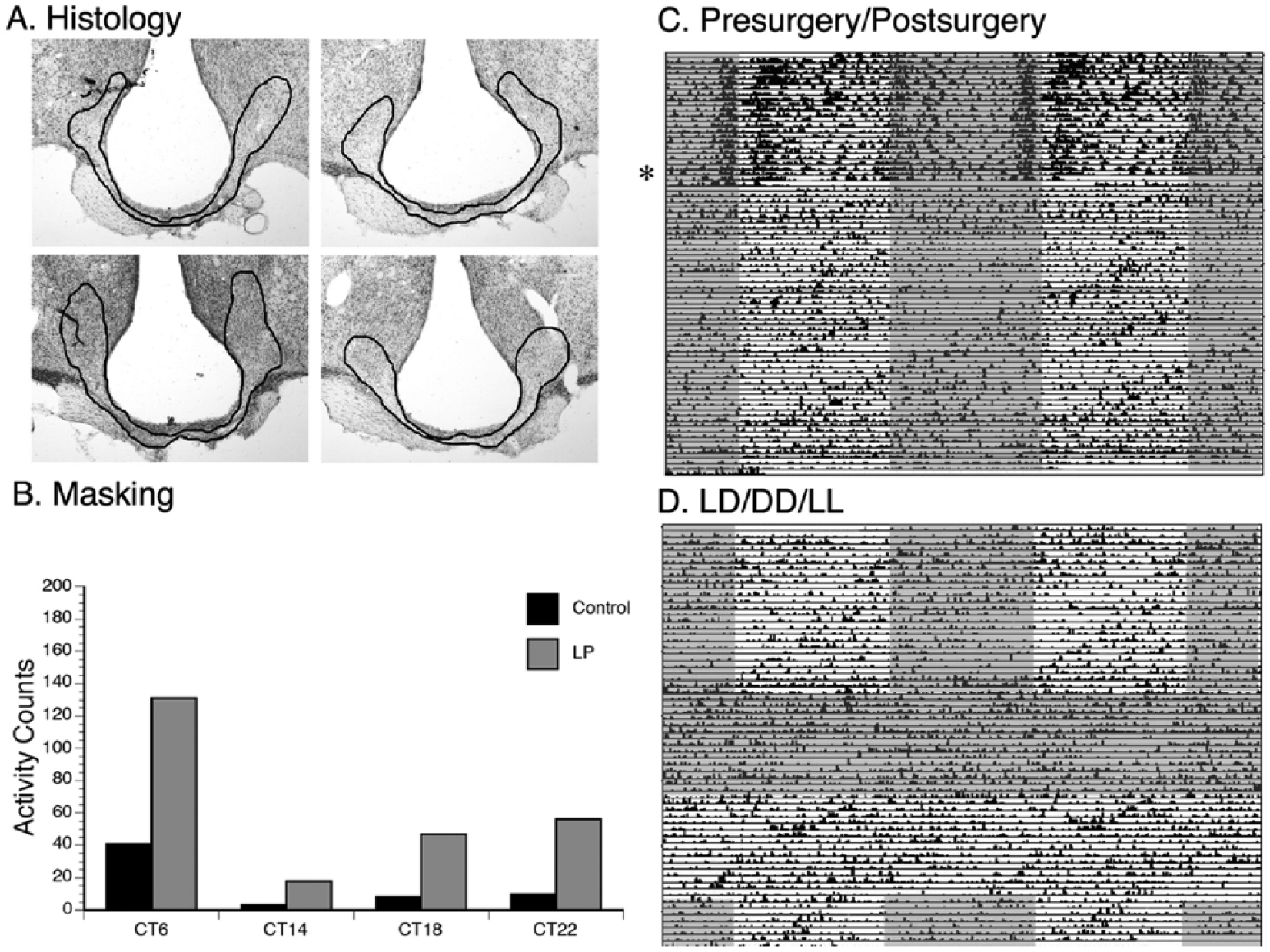
A unique grass rat with damage to the ventral subparaventricular zone (vSPVZ), SCN, and the optic chiasm that free-runs through all lighting conditions. Histology is presented in (A), which shows significant damage to the vSPVZ, SCN, and optic chiasm. Outlined regions in black indicate the lesioned area. (B) Light-induced masking was observed at all CTs. (C) Prior to surgery (denoted by the star), the animal was primarily active during lights-on. After surgery, the animal free-ran through LD conditions and also through DD and LL (D). Gray shaded areas indicate darkness; white areas indicate lights-on.
Discussion
Masking
The major finding of this work was that extensive damage to the vSPVZ, with or without additional damage to the SCN, did not prevent the display of masking responses to light in the diurnal grass rat. Thus, light presented as individual LPs, during the 12:12 LD cycle, under constant conditions or as the recurrent 3.5-h light interval of an ultradian LD cycle increased general activity after the lesions, as was the case for control animals or animals with damage outside the vSPVZ/SCN region. Furthermore, the presence of masking in animals with substantial damage to the SCN also serves to question the importance of that nucleus for mediating responses to light typical of a diurnal species. But the role of the SCN in mediating masking deserves further attention, since none of our cases resulted in complete SCN lesions, and one published report supports the role of the SCN in masking (Li et al., 2005), whereas another does not (Redlin and Mrosovsky, 1999). Importantly, others have shown that masking persists in drinking and wheel running in 12:12 LD cycles following SCN lesions (Fuller et al., 1981; Mistlberger, 1992), providing supporting evidence that the SCN may not be critical for the masking response to light. Clearly, the role for the SCN in masking to light is still under debate (reviewed in Morin, 2013).
In our study, the only effect of the vSPVZ/SCN lesions on masking responses was to abolish their circadian modulation; there was a significant interaction between time of presentation and the effect of the acute light pulses for both the intact controls and the animals with damage outside the target zone of the lesions, and that interaction was absent in animals with the intended damage. Therefore, in grass rats, the role of the vSPVZ/SCN in masking may be restricted to a modulatory role that results in variations of the magnitude of the responses to light across the subjective day and night (Shuboni et al., 2012).
For nocturnal species, the role of the vSPVZ in masking has not been investigated directly. In a study using lesions of the SCN (Redlin and Mrosovsky, 1999), partial damage to the vSPVZ was described as compatible with normal masking responses by hamsters. Furthermore, several studies documenting the disruptive effects of vSPVZ on circadian rhythms in rats (Cipolla-Neto et al., 1988; Moore and Danchenko, 2002; Abrahamson and Moore, 2006) have reported increased activity at night under an LD cycle, even when activity rhythms are absent or severely disrupted when the animals are monitored under constant conditions, thus suggesting that rats with vSPVZ lesions can mask their activity under an LD cycle. Consistent with the data from nocturnal species are our observations that grass rats with extensive damage to the vSPVZ were more active in the light, when kept in LD. Taken together, the available data for both diurnal and nocturnal rodents indicate that the vSPVZ is not critical for the mediation of masking responses to light. It is important to note that it remains plausible that other retinorecipient hypothalamic structures could be important for masking, especially given that our method of chemically lesioning the SCN and vSPVZ is different from other studies using electrolytic lesions. The differences observed may, therefore, be a methodological difference rather than a species difference.
One unique case among our lesioned animals (Figure 8) had moderate damage to the vSPVZ and almost a complete lesion of the SCN, with some evidence of mechanical damage to the optic chiasm. Behaviorally, this animal retained a clear circadian rhythm, which failed to entrain to the LD cycle. One explanation for this case is that light information was not capable of reaching what was left of the SCN, and therefore, the animal could not entrain in LD. This is similar to what was reported for animals receiving knife cuts that separate the SCN from the optic chiasm (Johnson et al., 1988). Remarkably, this animal was capable of masking, displaying higher amounts of activity following a light pulse compared to darkness. In addition, activity levels were higher during the light phase of an LD cycle compared to the dark phase. These results suggest that although light was not able to reach the SCN directly, it was able to reach other areas of the brain, which allowed the animal to continue to mask (e.g., IGL, OPT). This case provides additional evidence that transduction of light information by the SCN may not be required for the masking response, since entrainment did not occur, but masking did.
Our previous work adds supporting evidence that, rather than the SCN, the IGL and OPT may be involved in masking to light in grass rats. IGL lesions result in a reversal of masking responses to light in grass rats, such that light pulses result in a significant reduction in activity, whereas controls display the opposite behavior (Gall et al., 2013). In addition, IGL lesions resulted in a reversal of Fos responses to light in the OPT, whereas in the SCN, light-induced Fos expression was not affected by IGL lesions (Gall et al., 2014). Therefore, masking may operate via a neural network independent of the SCN (Gall et al., 2014). As noted earlier, in nocturnal species, lesions of the IGL do not have a similar effect (Redlin et al., 1999), suggesting that the role of the IGL on masking may be specific to diurnal species.
Circadian Rhythms
Lesions that ablated the vSPVZ bilaterally produced circadian activity patterns that were significantly disrupted even in animals that received no or minimal damage to the SCN (Figure 5A). These results were similar to those reported earlier for grass rats with partial vSPVZ lesions and no damage to the SCN (Schwartz et al., 2009). Importantly, Schwartz et al. (2009) found that the size of the vSPVZ lesion was inversely correlated with the strength of the postsurgical rhythms and the reentrainment rate of the animals. In rats, vSPVZ lesions also disrupt or eliminate circadian rhythms in activity (Abrahamson and Moore, 2006) and sleep (Lu et al., 2001) but do not affect temperature rhythms (Moore and Danchenko, 2002). This suggests that lesions of the vSPVZ in both rats and grass rats disrupt circadian rhythms but do not completely eliminate them. Importantly, grass rats with lesions in the vSPVZ still had detectable activity rhythms, in which period and alpha were not affected. These data suggest that the vSPVZ is important for the maintenance of circadian rhythms, but when the SCN is at least partially intact, rhythmicity is still detectable, and several parameters of the rhythms are intact. This is in stark contrast to grass rats with IGL lesions, which reverse their masking responses to light, in addition to exhibiting significant changes in alpha (Gall et al., 2013).
Excitotoxicity of the SCN
Six of our 12 animals in which the vSPVZ was ablated also had extensive damage to the SCN (Figures 1B and 2A). Therefore, in the grass rat, NMA was capable of destroying SCN neurons. In previous studies, the SCN has been shown to be resistant to excitotoxins such as NMA and kainic acid (Peterson and Moore, 1980; Schwarcz and Kohler, 1983; Hastings et al., 1985). Previous work from our group showed that NMA destroyed vSPVZ tissue but left the SCN intact in grass rats (Schwartz et al., 2009). The reason for the discrepancy between the 2 studies with grass rats is likely due to differences in methodology; Schwartz et al. (2009) used a lower concentration of NMA than what was used here. Interestingly, in nocturnal rodents, the concentration of NMA used to target the SCN was much less than what we used here (Hastings et al., 1985). Perhaps a higher concentration of NMA would also be effective at lesioning the SCN in nocturnal rodents. Importantly, it is also possible that the discrepancy between our results and those obtained with nocturnal rodents may reflect species differences in the abundance of glutamate receptors in the SCN of diurnal and nocturnal rodents. This hypothesis is supported by evidence that grass rats may be less sensitive to the phase-shifting ability of a glutamatergic agonist presented to the SCN than nocturnal rodents (Novak and Albers, 2002).
Conclusions
The data presented here for grass rats, a diurnal rodent, suggest that 1) the vSPVZ and perhaps the SCN are not necessary for the expression of masking responses to light, and 2) the SCN of grass rats is vulnerable to the actions of NMA, a glutamatergic excitotoxin, challenging the notion that the SCN is resistant to chemical lesions. We initially hypothesized that the vSPVZ and SCN would play a role in masking to light since both brain areas contain cells that express an increase in Fos production after exposure to light in grass rats (Shuboni et al., 2015). However, our data suggest that the neural mechanisms of masking involve interplays between the IGL and OPT (Gall et al., 2013; Gall et al., 2014) that may be independent of the transduction of light information by hypothalamic targets such as the SCN and vSPVZ. Finally, the finding that the SCN of grass rats is vulnerable to excitotoxins opens the possibility of species differences in the density of glutamatergic receptors in the SCN of diurnal and nocturnal rodents.
Footnotes
Acknowledgements
We thank Jennifer Langel, Ken Bennett, Megan Luck, and Thomas Groves for technical assistance. Supported by a National Science Foundation (NSF) grant (IOS-1051919, to L.S., A.A.N., and L.Y.), and A.J.G. was supported by a National Institutes of Health (NIH) Ruth L. Kirschstein National Research Service Award (NRSA) from the National Institute of Neurological Disorders and Stroke (NINDS) (F32 NS083360-01). The authors declare no competing financial interests.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
