Abstract
Many temperate insects enter diapause (dormancy) for overwintering in response to short days (long nights). A latitudinal cline in the critical day lengths for the photoperiodic induction of diapause has been reported in various insect species. However, the physiological mechanisms underlying this cline have remained elusive. We approached this issue in the flesh fly Sarcophaga similis, in which the photoperiodic time measurement system meets the “external coincidence model.” In this model, measuring day lengths depends on whether the photoinducible phase (φi), determined by a circadian clock, is exposed to light or not. First, we detected a clear latitudinal cline in the critical day lengths of flies collected from 4 localities at different latitudes. The phase positions of the φi, which can be verified by night interruption photoperiods, also showed a clear latitudinal cline. This result supports the hypothesis that the latitudinal cline in the critical day length is produced by the difference in the phase positions of the φi among different strains. A sexual difference in the critical day length for photoperiodic induction has also been detected in various species. In this study, a sexual difference in the critical day length was observed in the southern strains but there was no sexual difference in the phase positions of the φi. This result indicates that both sexes measure photoperiods in the same manner. Males are less sensitive than females to the light pulse given at the φi, suggesting a quantitative difference in the photoperiodic time measurement and counter systems. This study clearly reveals that distinct mechanisms induce latitudinal and sexual differences in the critical day length for the photoperiodic induction of diapause in a fly.
Keywords
The tilt of the Earth’s axis, from the plane of its orbit around the sun, causes seasons with dramatic environmental contrasts. Some seasons are suitable for development and reproduction, while others are not. Thus, most insects inhabiting the temperate zones enter diapause, a physiological state in which development or reproduction is suppressed or arrested in response to shorter day lengths to overcome adverse conditions. This ability for organisms to respond to the photoperiod is called photoperiodism (Goto and Numata, 2014; Nelson et al., 2010). For example, the flesh fly Sarcophaga similis, the focal species in this study, shows a clear photoperiodic response. They develop without interruption under long-day conditions and enter diapause at the pupal stage in response to short-day conditions (Tanaka et al., 2008), as reported for other sarcophagid species (Denlinger, 1971; Kurahashi and Ohtaki, 1979; Saunders, 1976; Vinogradova, 1976). Even though many insect species measure the “night length” not the “day length” (Saunders, 2013), the term “day length” is widely used in the literature, and thus we also followed this terminology in this paper.
There is little intraspecific variation in the stages at which diapause occurs; however, there is great intraspecific variation in the timing of diapause entry. The timing is determined by the critical day length, which is the length of the light fraction of the light-dark (LD) cycle that separates a strong long-day response from a strong short-day response in the photoperiodic response curve (Fig. 1A). One of the intraspecific variations is critical day length, which varies among geographic strains (Danilevskii 1965; Danks, 1987; Saunders 2002). For example, in an exhaustive survey, 41 geographical strains of Drosophila littoralis were collected from localities ranging from the Black Sea coast (41.6° N) to northern Finland (69.0° N), with geographical variations in critical day length (Lankinen, 1986). The author found that the critical day length ranged from 11.6 to 20.3 h (from south to north), with length highly correlated with latitude; i.e., a clear latitudinal cline. Such a latitudinal cline, with longer critical day lengths in strains from higher latitudes, is observed in several insect species (Bradshaw and Lounibos 1977; Kimura et al., 1993; Lankinen et al., 2013; Lehmann et al., 2015; Paolucci et al., 2013; Shimizu and Kawasaki, 2001; Wang et al., 2012). In addition, the rapid evolution of changes in critical day length for several invasive species has been measured or inferred in the field after colonization (Bean et al., 2012; Gomi et al., 2007; Tanaka and Murata, 2016; Tanaka et al., 2015; Urbanski et al., 2012). The significance of latitudinal cline in critical day length can be explained in terms of ecological aspects. Days are longer at higher latitudes between the spring and autumn equinoxes, but the climate is colder, and summer ends earlier than in areas at lower latitudes. Therefore, insects at higher latitudes likely enter diapause earlier by responding to the longer photoperiod (Saunders, 2002; Tauber et al., 1986).
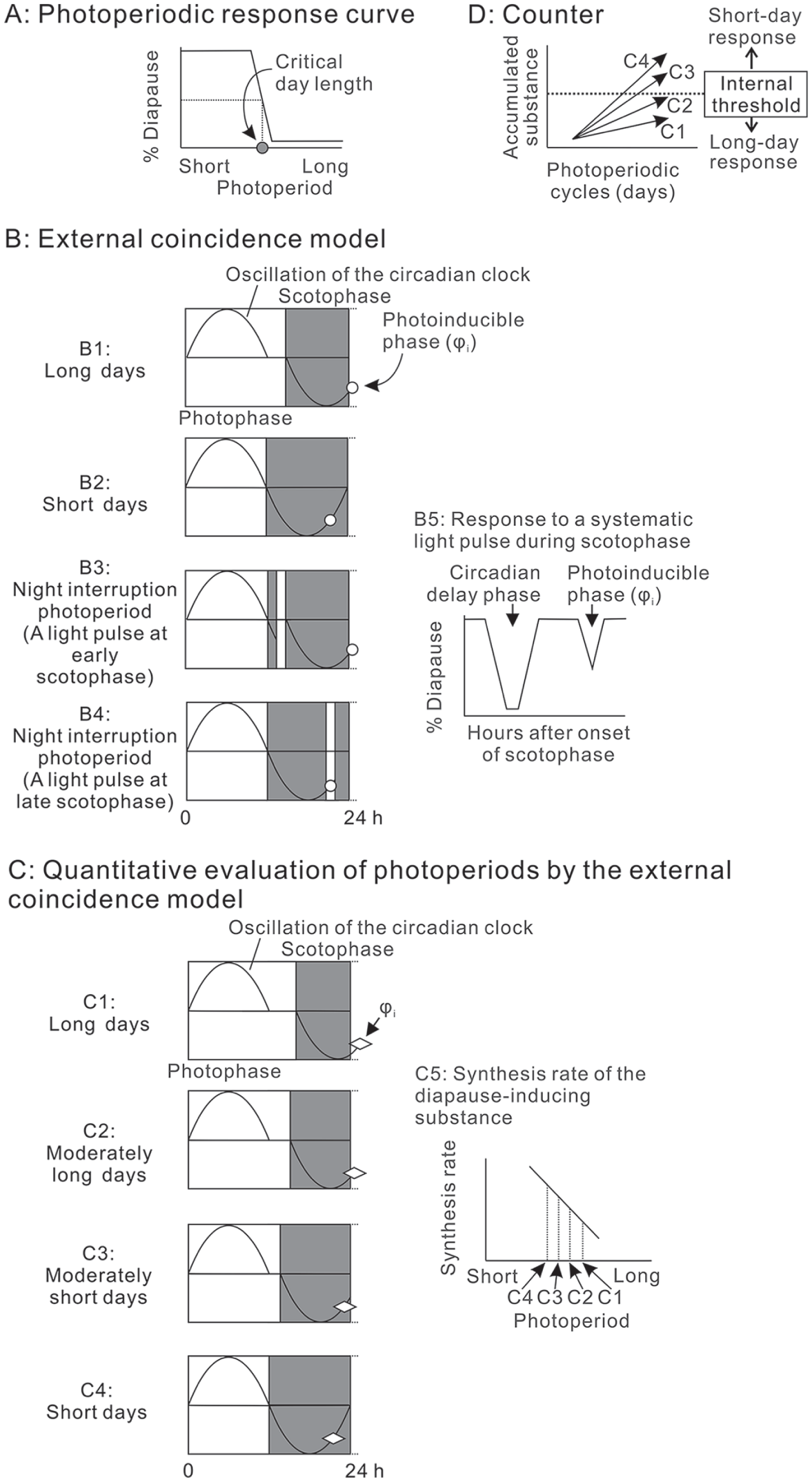
Photoperiodic response curve (A) and conceptual diagrams of the external coincidence model (B and C) and counter system (D), which describes the photoperiodic response of the flesh fly. (A) The flesh fly enters pupal diapause in response to a short-day photoperiod. The critical day length is defined as the length of the light fraction of the LD cycle that separates a strong long-day response from a strong short-day response in the photoperiodic response curve. (B) Photoperiodic time measurement in the external coincidence model is based on an oscillation of the circadian clock, which sets its phase at dawn and dusk, and on the position of the photoinducible phase (φi) in the latter half of the scotophase. During long summer days, the φi is exposed to light and a long-day response is induced (B1). During short autumnal days, the φi falls in the dark phase, eliciting a short-day response (B2). When a light pulse is applied during the early scotophase, the clock delays its phase and thus the φi is pushed out to the main photophase to be exposed to light. This gives rise to a long-day effect (B3). A light pulse during the late scotophase directly illuminates the φi, to induce a long-day response (B4). When the scotophase was systematically scanned with a light pulse, 2 light-sensitive phases averting diapause were observed; the former is the circadian delay phase promoting the phase delay of the clock, and the latter is the φi (B5). (C) Photoperiods are also quantitatively evaluated. In this model, insects quantitatively produce a hypothetical diapause-inducing substance based on the photoperiods, according to how long the φi is exposed to light (C1-C5). (D) The substance accumulates in the counter system. When the level of the accumulated substance exceeds a certain “internal threshold” diapause induction is determined. In contrast, nondiapause development is determined when the accumulation is lower than the threshold.
Diapause incidence and the critical day length that triggers diapause can also differ intraspecifically between males and females (Danks, 1987). Males are more likely to enter diapause than are females; i.e., males have a longer critical day length than females (Butler et al., 1985; Denlinger, 1972; Shimizu and Fujisaki, 2002; Wiklund et al., 1992). Despite obvious ecological advantages, diapause itself is a metabolically expensive life history strategy. The costs of diapause are commonly reflected in a lower post-diapause survival and reduced fecundity (Bradshaw et al., 1998; Ellers and van Alphen, 2002; Matsuo, 2006; Sadakiyo and Ishihara 2012a; 2012b). In the flesh fly Sarcophaga crassipalpis, Denlinger (1981) found that the risks associated with overwintering are greater in females than in males. Females that had been in diapause show lower reproductive success, whereas males that had been in diapause do not suffer severe reproductive consequences. This explains the propensity for females to escape from diapause induction in autumn, as delaying the onset of diapause while temperatures are still favorable allows further reproduction before the winter sets in. Thus, for females, the timing of diapause inception becomes a trade-off between high fertility and environmental security. Wiklund et al. (1992) explained the significance of this sexual difference from the viewpoint of evolutionarily stable strategies. In temperate areas where many insects have multiple generations, males should enter diapause at an earlier date than females should, because late-emerging males are penalized in terms of fewer mating opportunities.
We understand the ecological significance of the latitudinal and sexual differences in the critical day length for the photoperiodic induction of diapause. However, little is known about the physiological mechanisms underlying these differences (Hut et al., 2013). Photoperiodism is operated by several physiological modules, including: 1) the photoreceptive system, which extracts light information from environmental conditions; 2) the photoperiodic time measurement system, which measures day or night length; 3) the counter system, which accumulates photoperiodic information; and 4) the endocrine system, which regulates the synthesis and/or secretion of neurohormones and/or hormones to reflect the photoperiodic information into a physiological state (Goto and Numata, 2014). Latitudinal and sexual differences in the critical day length can be derived from some modulations of the photoperiodic time measurement system and/or its downstream cascades. To determine the causal mechanisms regulating intraspecific variation in critical day length, we used a flesh fly species that shows a clear photoperiodic response and has been used for understanding insect photoperiodism.
Sarcophaga similis is widely distributed in Europe and Asia. The photoperiodic time measurement for diapause induction in Sarcophaga species meets the “external coincidence model” (Gnagey and Denlinger, 1984; Goto and Numata, 2009; Lewis and Saunders, 1987; Saunders, 1976; 1978a; 1978b; 1979; Saunders and Lewis, 1987ab). This model hypothesizes the involvement of an endogenous circadian oscillator in photoperiodic time measurement and the existence of a photoinducible phase (φi), which is located in the late phase of the subjective night under the control of a circadian clock (Pittendrigh and Minis, 1964; Fig. 1B). The φi is important for determining the developmental program. Short-day responses are produced when the φi is in darkness, whereas long-day responses are produced when the φi is exposed to light. During summer, the φi is pushed out to the photophase because of the phase delay of the clock and thus falls in the light period, representing a longer day to insects and, accordingly, eliciting a long-day response. Under autumnal short-day conditions, the φi falls in the dark period, which is interpreted as a short day and induces a short-day response. When a light pulse systematically scans the scotophase under short-day conditions, 2 light-sensitive phases can be observed (Goto and Numata, 2009; Gnagey and Denlinger, 1984; Tagaya et al., 2010; Fig. 1B). The phase located during the early scotophase is derived from the phase delay of the circadian clock, whereas the phase located during the late scotophase is the φi (Fig. 1B; Saunders, 2002).
Insects, including flesh fly species, evaluate photoperiods quantitatively. This can be explained by the “quantitative production of a diapause-inducing substance” (Tagaya et al., 2010). In this concept, the φi is not a short period of time but is instead a much longer one. Insects synthesize the hypothetical diapause-inducing substance based on how long the φi is exposed to light. The synthetic rate of the substance is higher during shorter days but lower during longer days (or, the degradation rate of the substance is higher during longer days) (Fig. 1C). The substance accumulates in the counter system. When the level of the accumulated substance exceeds a certain “internal threshold,” diapause induction is determined, whereas nondiapause development is determined when the accumulation is lower than the threshold (Gibbs, 1975; Saunders, 1966; Fig. 1D). Although this concept sufficiently explains the quantitative discrimination of photoperiods, no such product has yet been verified.
Latitudinal and sexual differences in critical day length (Fig. 2A) may be explained by the qualitative differences in photoperiodic time measurements; i.e., the phase-positional difference of the φi (Hypothesis I, Fig. 2B; Saunders 2013). If the φi of a group is located at an earlier point in the scotophase than that of another group, the critical day length of the former will be longer than that of the latter. This hypothesis can be explained by 2 models; the same phase angle of the φi in circadian clocks with different periods (Model A shown in Fig. 2B1-3; Hut and Beersma, 2011) and the different phase angles of the φi in circadian clocks with the same periods (Model B shown in Fig. 2B4-6). In model A, the φi resides in the same position (the same phase angle) in the circadian clocks, but the period of the clock is longer in a group with a short critical day length than in a group with a long critical day length. In model B, the periods of the clocks are identical between the groups, but the φi resides in the different positions (the different phase angle) in the circadian clocks. Such positional differences of the φi can be assessed by the systematic scanning of the scotophase using a short light pulse; i.e., the night interruption photoperiod (Fig. 2B7). Alternatively, the difference in the critical day length might be explained by the quantitative difference in the photoperiodic time measurement and counter systems (Hypothesis II, Fig. 2C; Tagaya et al., 2010). In this hypothesis, there is no positional difference of the φi, but intraspecific variation in the synthetic rate of the diapause-inducing substance is assumed. If a group with a longer critical day length synthesizes the substance at a higher rate than another group with a shorter critical day length, the substance is accumulated at a higher rate in the counter system of the former than in that of the latter. Thus, even under the same photoperiodic conditions, the short-day response is elicited easier in the former than in the latter. The basic concept of Hypothesis II is whether the accumulated amount of the substance exceeds the threshold or not, and thus, the hypothesis includes the same synthetic rates of the substance with different internal thresholds between the groups.
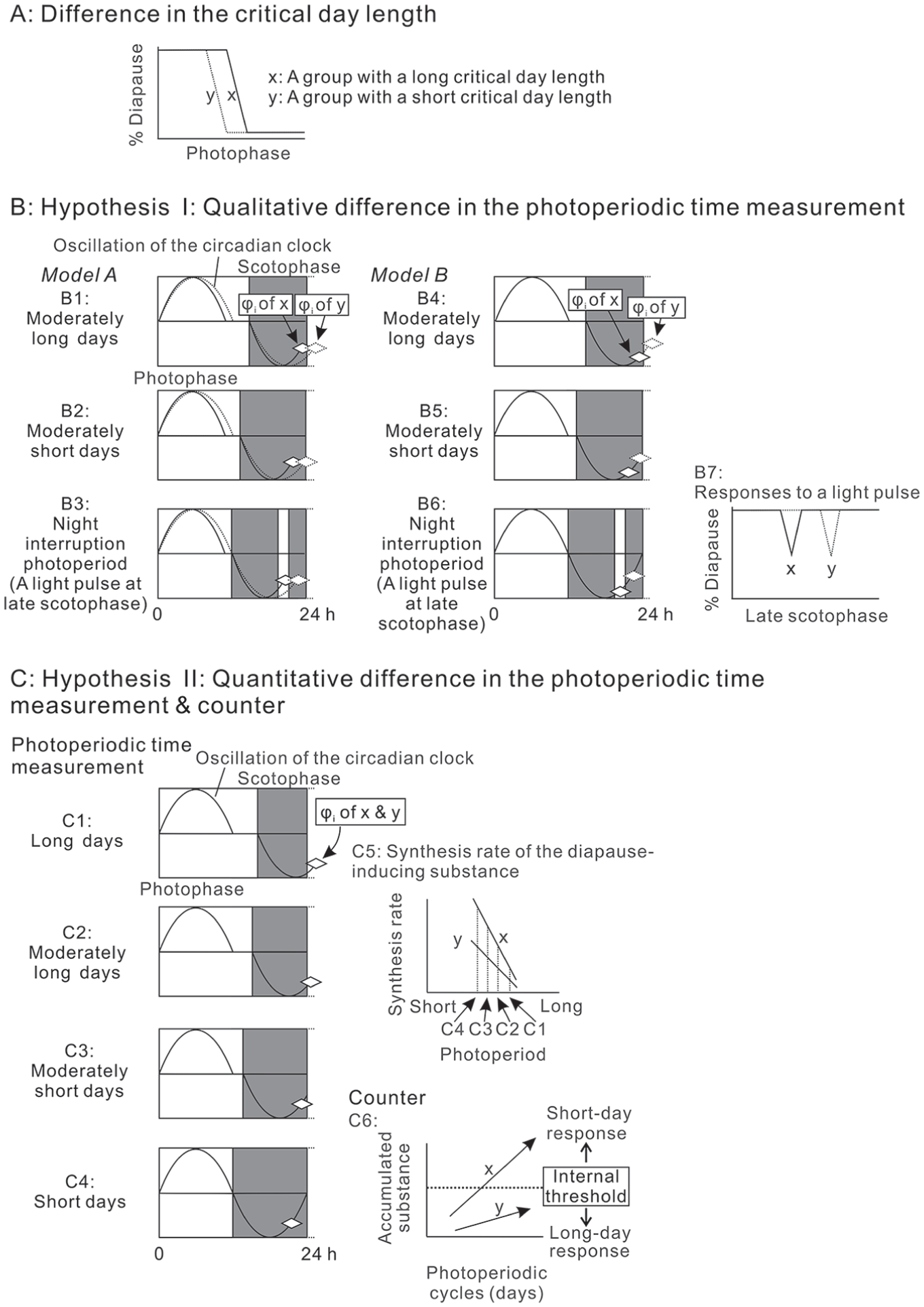
Two groups with different critical day lengths (A) and 2 hypotheses that explain the difference (B and C). (B) Hypothesis I assumes the presence of a qualitative difference in the photoperiodic time measurement between groups; i.e., a difference in the phase positions of the φi. Under this hypothesis, the φi of a group with a longer critical day length (group x) is positioned at an earlier phase of the scotophase than that of a group with a shorter critical day length (group y). This can be achieved by the same phase angle of the φi in the circadian clocks with different periods (Model A; B1-3) or by different phase angles of the φi in the circadian clocks with the same periods (Model B; B4-6). In both of these, the group with a longer critical day length (group x) enters diapause even under a moderately long day length (B1 and B4). During moderately short days, the group with a shorter critical day length (group y) also enters diapause because its φi is in the dark phase (B2 and B5). The systematic interruption of the scotophase by a light pulse will detect this qualitative difference in the phase positions of the φi (B3, B6, and B7). (C) Hypothesis II assumes, not qualitative, but quantitative differences in the photoperiodic time measurement and counter systems. Under this assumption, the positions of the φi are identical between groups (C1-C4), but they respond to the photoperiod differently. A group with a longer critical day length (group x) synthesizes larger amounts of the hypothetical diapause-inducing substance in response to photoperiods compared with another group with a shorter critical day length (group y) (C5). The substance is accumulated daily in the counter system. The substance easily exceeds the internal threshold in the group with a longer critical day length as compared with the group with the shorter critical day length (C6), and thus the former and latter likely enters and avoids diapause, respectively, even under the same photoperiod. The main concept is whether the accumulated amount of the substance exceeds the threshold or not, and thus, this hypothesis includes the same synthetic rates of the substance with different internal thresholds between the groups.
Tagaya et al. (2010) reported that a mid-latitude strain of S. similis shows sexual differences in the critical day length for diapause induction; i.e., the critical day length of males is longer than that of females. The phase positions of the φi are identical between the sexes, whereas males are less sensitive to the light pulse given at the φi. These results align with Hypothesis II (Fig. 2C). To expand our knowledge of the physiological mechanisms underlying photoperiodism, we focus on the latitudinal cline of the critical day length for diapause induction in S. similis. In this study, we collected 4 populations from different latitudes in Japan and compared their photoperiodic response curves to clarify their critical day length. A light pulse was emitted during the scotophase to find the phase position of the φi. We further investigated the sexual differences in the phase position of the φi in these 4 geographic strains to assess the similarity between latitudinal and sexual variations in the critical day length for the photoperiodic induction of diapause.
Materials and Methods
Insects
Adult flies of S. similis Meade were collected in Otaru City, Hokkaido Prefecture (OTR; 43.20°N, 141.99°E); Utsunomiya City, Tochigi Prefecture (UTM; 36.57°N, 139.90°E); Osaka City, Osaka Prefecture (OSK; 34.59°N, 135.51°E); and Nagasaki City, Nagasaki Prefecture (NGS; 32.75°N, 129.88°E) between 2014 and 2017 (Fig. 3A). Stock cultures of these strains were maintained under diapause-averting long-day conditions, 16 h of light and 8 h of darkness (LD 16:8 h) at 20°C ± 1°C or 25°C ± 1°C.
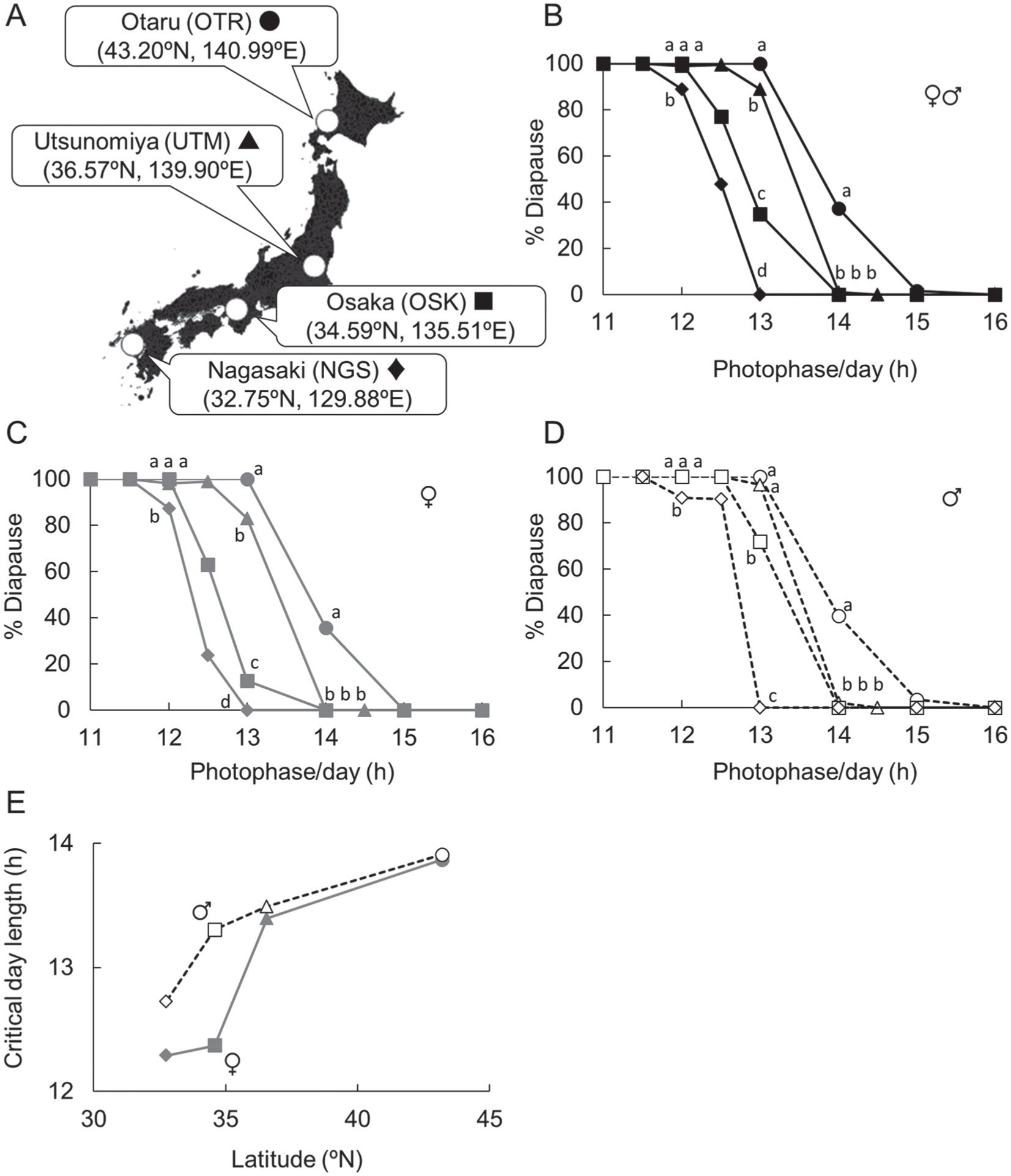
Locations where the strains of Sarcophaga similis originated (A), their photoperiodic responses (B-D), and the relationship between the latitude of where the strain originated and critical day length (E). Photoperiodic response curves obtained from data for both sexes (B; n = 36-182), females only (C; n = 17-86), and males only (D; n = 19-96) of the OTR (circles), UTM (triangles), OSK (squares), and NGS (diamonds) strains at 20 °C are shown. Flies were reared under the photoperiod continuously. Diapause incidences under LD 12:12, 13:11, and 14:10 h conditions were separately compared with Tukey-type multiple comparisons for proportions. The same letters indicate statistical insignificance (P > 0.05).
For the experiments, adult flies were maintained in groups of less than 50 in a plastic container (15 cm in diameter, 9 cm in depth) covered with nylon netting. Water, 2 pieces of block sugar, and a piece of beef or chicken liver were provisioned at 20°C ± 1°C under the experimental photoperiodic conditions. Fifteen days after the first provision of the liver, a new piece of liver was provided as a larviposition site. The next day, larvae on the liver were collected and transferred to a chunk of liver on an aluminum dish. The dish was placed on dry wood chips (30 to 50 mm in depth, pupariation/pupation substrate) in a plastic container. Most larvae ceased feeding 5 days after being transferred, left their food, and moved into the wood chips for pupariation. A few days later, they pupariated and pupated.
White fluorescent lights (FLR15W, FLR20SW/M, FLR30SW, or FL40SW; Panasonic, Kadoma, Osaka, Japan) were used as a white light source at an intensity ranging from 0.3 to 1.3 W·m−2 on the surface of the wood chips in the photophase of complete photoperiods, the main photophase, and the light pulse of night interruption photoperiods. However, larvae were located within their food (liver) during the feeding stage and the wandering larvae were concealed in dry wood chips at a depth of 30 to 50 mm or in wet wood chips to a depth of 10 mm (see below). Therefore, the light intensity that the larvae received was dependent on their position.
Photoperiodic Response Curves
Insects were reared from their embryonic stage continuously under experimental photoperiodic conditions to assess their diapause status. We adopted photoperiods yielding 0% to 100% diapause incidences. In this study, we regarded the critical day length as the day length in which half of the individuals in the population entered diapause.
Night Interruption
To determine the phase position of the φi, larvae reared under short-day conditions were exposed to a night interruption photoperiod. Mature larvae (5 days after larviposition), reared under a short-day photoperiod of LD 11.5:12.5 h from their embryonic stage, were placed in wet chips at a depth of 10 mm. This treatment, called water treatment, prolongs the larval photosensitive period and enables us to expose the larvae to photoperiods of a determined time (Goto and Numata, 2009a; 2009b). In this study, the scotophase was systematically interrupted by a 2 h light pulse under LD 10:14 h conditions i.e., LDLD 10:x:2:(12-x) h, for 4 days. In our preliminary experiments, we had maintained flies under LD 10:14 h and exposed them to LDLD 10:x:2:(12-x) h but it did not work, as a light pulse did not avert diapause, possibly because the photoperiod was very short for some strains. Therefore, we maintained flies under LD 11.5:12.5 h and exposed them to LDLD 10:x:2:(12-x) h for night interruption. After water treatment, larvae were transferred onto dry wood chips at a 30-50 mm depth under LD 11.5:12.5 h, to allow them to pupariate and pupate.
The most effective phases for diapause aversion were assessed from the light-pulse response curve. When 2 phases were similarly effective (less than 5% difference), both were regarded as the most effective phase.
Judgment of the Developmental Status and Sex
Twenty or more days after larviposition, the anterior tip of the puparium was removed using fine forceps, to assess the diapause status (diapause or nondiapause) according to the standard described by Fraenkel and Hsiao (1968). After assessment, pupae were pulled from the puparia for sex determination. Sex was judged based on the differences in the sizes of the fourth and fifth tarsomeres on the middle legs: the fifth tarsomere is obviously larger than the fourth in males but not in females (Tagaya et al., 2010).
Results
Latitudinal and Sexual Differences in Critical Day Length
The photoperiodic response curves of 4 geographic strains are shown in Fig. 3B. Flies exhibited a clear photoperiodic response, where short days elicited a high diapause incidence and long days elicited a low diapause incidence. Their critical day lengths were different, with the northern strains entering diapause under longer photoperiods; i.e., a clear latitudinal cline (Fig. 3B). Clear latitudinal clines were also observed when data of females and males were separately analyzed (Fig. 3C-E). It is noteworthy that diapause incidences were significantly higher in males than females under certain photoperiods in southern strains (OSK and NGS) (χ2 test, P < 0.05) but not in northern strains (OTR and UTM) (χ2 test, P > 0.05). Thus, critical day lengths were longer in males than in females in southern strains but were nearly identical in northern strains (Fig. 3E).
Systematic Scanning of the Scotophase by a Light Pulse
The systematic scanning of the scotophase by a 2-h light pulse was conducted on the NGS strain (Fig. 4). The same experiment has been done previously for the mid-latitude strain, with similar but slightly different results (Goto and Numata, 2009b). In both cases, the effects of a light pulse varied markedly among the phases of the scotophase. In the NGS strain, 1 h after scotophase onset, the diapause incidence of the population remained high, but the incidence dropped sharply when a light pulse was presented 2 h after scotophase onset. From 3 to 6 h after scotophase onset, the diapause incidence increased gradually. A light pulse presented 7 to 9 h and 11 h after scotophase onset was ineffective in averting diapause. In contrast, the light pulse 10 h after scotophase onset was effective. The light-sensitive phase 2 to 5 h after scotophase onset is the circadian delay phase, in which the circadian clock delays its phase, and the light-sensitive phase 10 h after scotophase onset is the φi. Diapause incidence in females was consistently lower than that in males, but a qualitative difference was undetectable in these light-sensitive phases; i.e., in both sexes, the light-sensitive phase, in which the circadian clock delays its phase, occurred 2 to 6 h after scotophase onset and the φi occurred 10 h after scotophase onset.
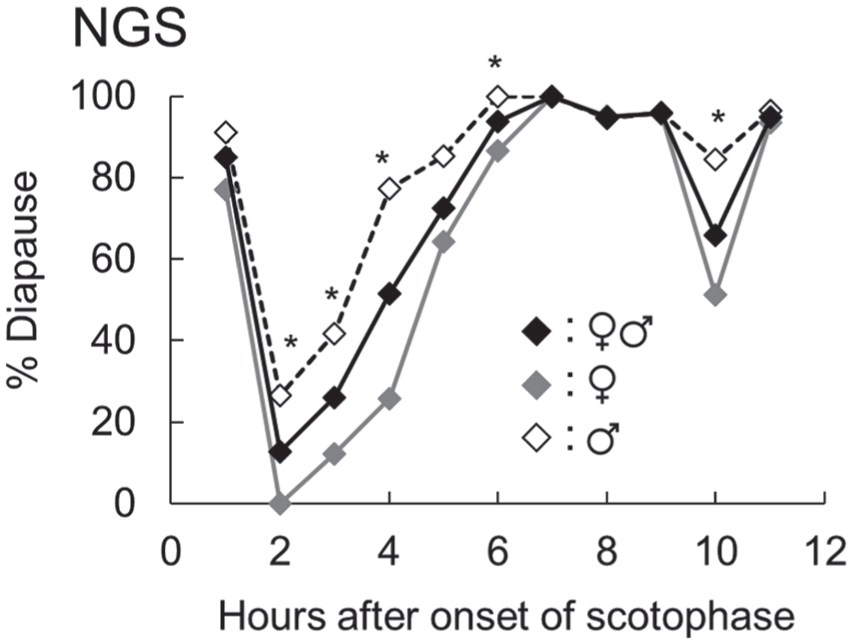
Effect of the systematic interruption of the scotophase by a light pulse on the diapause incidence of the NGS strain of Sarcophaga similis. Diapause-destined larvae were placed on wet wood chips (water treatment) and exposed to night interruption photoperiods. After water treatment, larvae were transferred onto dry wood chips to allow them to pupariate and pupate (n = 23-77). Asterisks indicate significant differences between the sexes (χ2 test, P < 0.05).
To clarify the exact position of the φi in the late scotophase, a 2-h light pulse was systematically applied to the 4 geographic strains at 8.5 to 11 h after scotophase onset, with 15-min intervals. If the phase positions of the φi were different among the geographical strains, the effective phases for diapause aversion by a light pulse are different among them. The most effective phases for diapause aversion among strains varied from 9 to 10.25 h after scotophase onset; i.e., the phases were 9-9.25, 9.50, 9.75-10, and 10.25 h after scotophase onset in the OTR, UTM, OSK, and NGS strains (Fig. 5, left columns), respectively. Therefore, the phase positions of the φi are qualitatively different among strains and a clear negative latitudinal cline of the phase positions of the φi was observed (Fig. 6). The negative latitudinal cline was also clear in the data of the females only and males only (Fig. 5, middle and right columns, respectively; Fig. 6). Although some small differences were detected, the most effective phases for diapause aversion were identical between the sexes in each strain. These results indicate that the phase positions of the φi are identical between the sexes, and both sexes measure photoperiods in the same manner (Fig. 6).
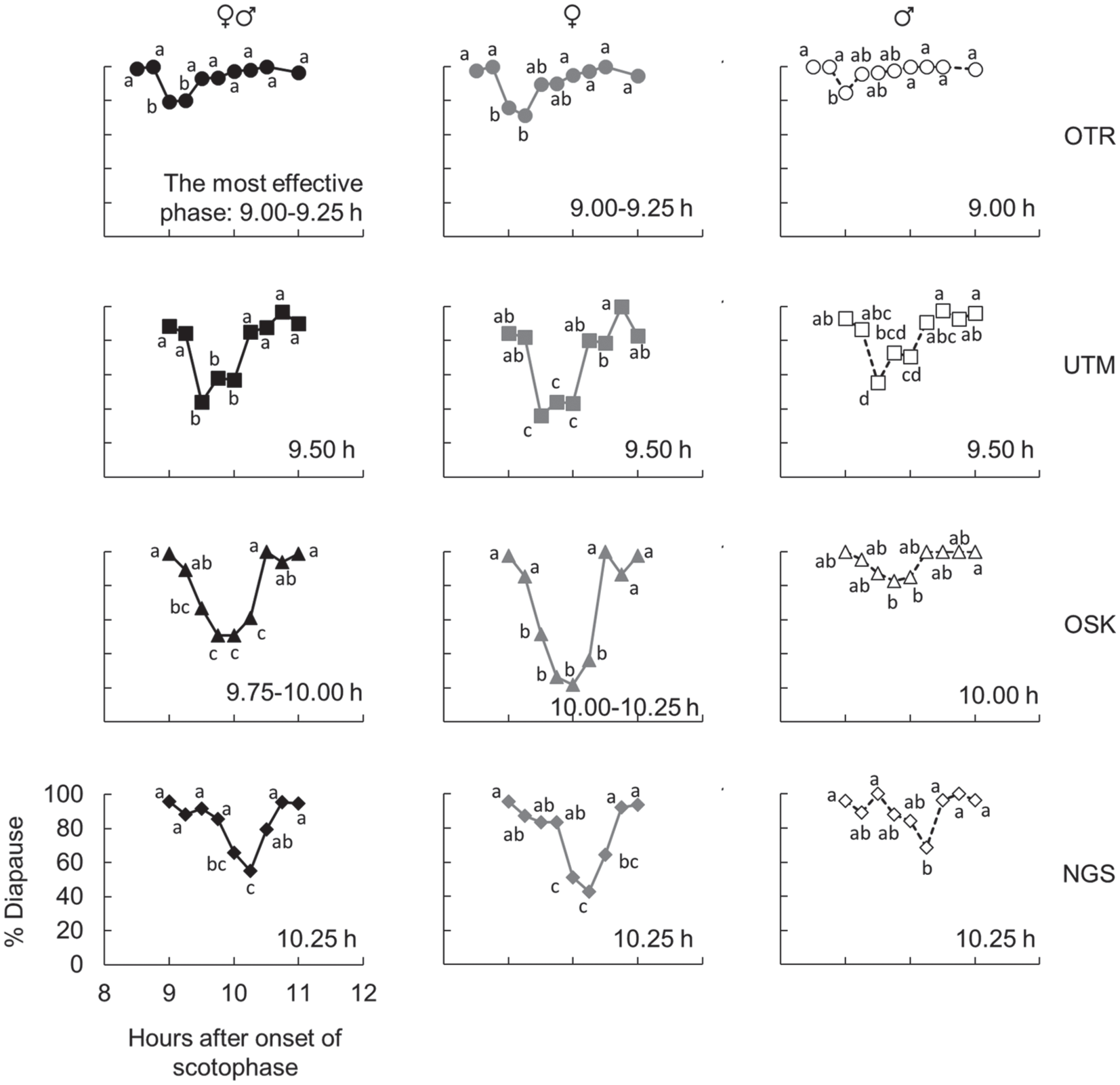
Effects of interrupting the late scotophase with a light pulse on the diapause incidence of the OTR (the top row), UTM (the second), OSK (the third), and NGS (the bottom) strains of Sarcophaga similis. Data from both sexes (left column), those from females only (middle column), and those from males only (right column) are shown (n = 32-137). The same letters indicate statistical insignificance (Tukey-type multiple comparisons for proportions, P > 0.05). The phase that was most affected by the light pulse during the late scotophase is also indicated.
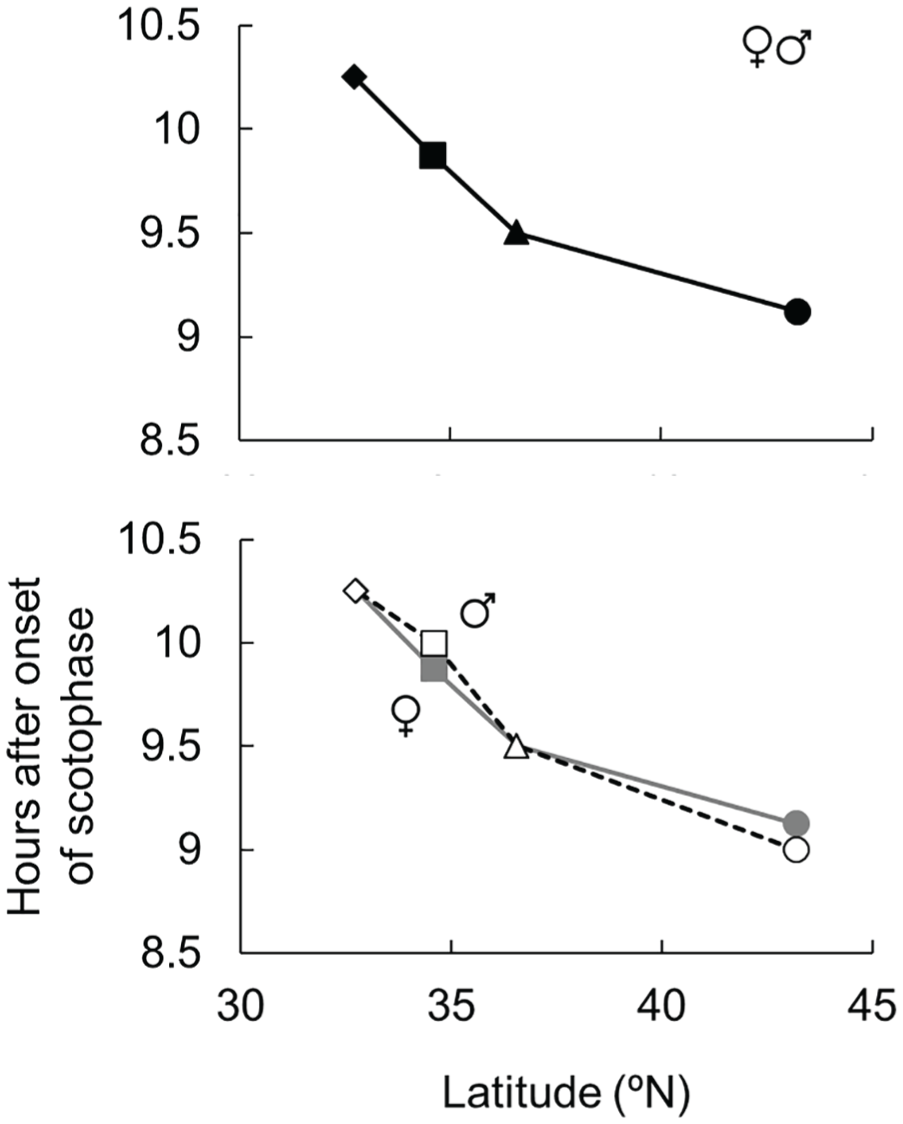
The relationship between the latitudes of where the strain originated and the phase positions of the φi after scotophase onset. The data shown in Figure 6 were arranged to show the latitudinal cline. The mean was plotted when 2 phases were similarly effective (less than 5% difference). The upper panel is derived from the data of both sexes and the lower panel is derived from the data of females (gray marks and gray solid line) and males (open marks and black broken line). The OTR (circles), UTM (triangles), OSK (squares), and NGS (diamonds) strains are shown.
Discussion
Danilevskii (1965) was the first to demonstrate the correlation between latitude and the critical photoperiod for diapause induction. He showed that the critical photoperiod increases by approximately 1 h with every 5°- to 6°-increase in latitude in the knot grass moth, Acronicta rumicis. Since then, such a latitudinal cline has been observed in several insect species (Bradshaw and Lounibos 1977; Kimura et al., 1993; Lankinen et al., 2013; Lehmann et al., 2015; Paolucci et al., 2013; Shimizu and Kawasaki, 2001; Wang et al., 2012). A latitudinal cline of the critical day length for diapause induction was also obvious in 4 geographic strains of S. similis in this study. The night interruption photoperiods reveal a clear latitudinal cline in the phase positions of the φi. A strain from a higher latitude possesses a φi at an earlier stage in the scotophase than a strain from a lower latitude. These results indicate that the qualitatively different photoperiodic time measurement systems among geographic strains causally induce the latitudinal difference in the critical day lengths; i.e., Hypothesis I (Fig. 2B). Although a similar model has been proposed by Saunders (2013) and Meuti and Denlinger (2013), it has not been empirically tested.
The next question is how the φi is positioned at different phases in the scotophase among strains. At least 2 possibilities exist. One is based on the same phase angle of the φi in the circadian clocks showing different periods (Model A, Fig. 2B1-3) and the other is based on qualitatively different phase angles of the φi with no assumption of circadian system variations (Model B, Fig. 2B4-6). In model A, if the period of the clock of a group with a longer critical day length was shorter than that of another group with a shorter one, the φi of the former is located at an earlier scotophase than that of the latter. Although we still do not fully understand the molecular mechanisms of the circadian clock involved in photoperiodic time measurement, the clock regulating photoperiodism appears to share many of the same genes as the circadian clock that regulates locomotor activity rhythms; i.e., the circadian clock genes including period (per), timeless (tim), cycle (cyc), Clock (Clk), and mammalian-type cryptochrome (cry-m or known as cry2) (Dolezel, 2015; Goto, 2013; Koštal, 2011; Saunders, 2011; 2016). Clock gene mutants show great variations in circadian rhythmicity. For example, a null mutant of per shows a loss of circadian rhythmicity in adult eclosion and locomotor activity rhythms and several mutants in the per alleles shorten or lengthen the period of the clock (Konopka and Benzer, 1971). A recent study revealed that genomic regions containing circadian clock genes (per, cyc, and cry-m) are associated with geographic variations in the photoperiodic diapause induction of a wasp species (Paolucci et al., 2016). A close association between the geographic variations and genomic regions containing per and tim is also reported in a butterfly species (Pruisscher et al., 2018). The conspicuous correlation between the clock gene haplotypes and photoperiodic induction is also reported (Mathias et al. 2007; Pegoraro et al, 2014; Tauber et al. 2007; Yamada and Yamamoto, 2011). Latitudinal clines of several properties of endogenous circadian rhythms, such as a negative latitudinal cline of circadian periods, are observed in Drosophila species (Lankinen, 1986, 1993). These results may support the model A (Hut et al., 2013). On the other hand, considerably low correlations between the period of the Nanda-Hamner rhythm (the period of the circadian clock involved in the photoperiodic response) with latitude and the critical day length were also reported in a mite species and a mosquito species (Bradshaw et al., 2006; Vaz Nunes et al., 1990; Wegis et al., 1997). In addition, there is no genetic association between critical day length and circadian period in the mosquito (Bradshaw et al., 2012). These results may support the idea that phase angles of the φi qualitatively differ among the geographic strains with no assumption of circadian clock variations; i.e., Model B (Fig. 2B4-6). We are unable to determine which models are applicable to the photoperiodic response in S. smilis. It is crucial to investigate the properties of the circadian system involved in photoperiodic induction among these geographic strains, such as the Nanda-Hamner rhythm and the phase-response curve of the circadian clock involved in photoperiodism (Bradshaw et al., 2012; Saunders, 1976).
It is noteworthy that the sexual differences in the critical day length for the photoperiodic induction of diapause was smaller in the northern S. similis strains than in the southern ones. Although a similar trend is also reported in the cotton bollworm Helicoverpa armigera (Shimizu and Fujisaki, 2002), how the sexual difference in the photoperiodic response changes along with latitude has received little attention in other insect species. A small sexual difference at high latitudes might be derived from overwintering risks, as proposed by Denlinger (1981). At higher latitudes, autumn is shorter and winter comes earlier. Thus, reproduction during autumn, by avoiding diapause, would be riskier and the need to enter diapause to survive the winter would be high in both sexes. In this case, the sexual difference in the trade-off between high fertility and environmental security becomes negligible, which promotes the disappearance of the sexual difference in the critical day length. It is of interest to compare the latitudinal sexual difference of the photoperiodic response curves with the latitudinal difference of the risks associated with overwintering.
In southern strains (OSK and NGS), sexual differences in both diapause initiation and incidence were obvious, with males having a longer critical length, as reported by Tagaya et al. (2010). The present study reveals that the phase positions of the φi are identical between sexes, irrespective of the difference in the critical day lengths. These results support Hypothesis II; i.e., a sexual difference in the quantitative evaluation of photoperiods and accumulation of the substance (Fig. 2C; Tagaya et al., 2010). This quantitative manner could alter critical day length with no requirement to change the central circadian clockwork (Meuti and Denlinger, 2013). Development is arrested in diapausing pupae, and these individuals do not progress to the next metamorphic stage (adult) until diapause is terminated and the prothoracic gland (PG) releases ecdysteroids (Denlinger et al., 2012). Prothoracicotropic hormone (PTTH), from neurosecretory cells (PTTH cells) in the brain, is responsible for the activation of the PG (Smith and Rybczynski, 2012). In a moth in pupal diapause, PTTH cells are less excitable and neurophysiologically silent; i.e., high voltage thresholds, low input resistance, and few spontaneous action potentials (Tomioka et al., 1995). Richard and Saunders (1987) investigated PTTH activity in the brain, which is assessed by in vitro activation of PG cells to produce and release ecdysteroids in the diapausing flesh fly, Sarcophaga argyrostoma. They revealed that brain extracts from diapausing pupae have as much, if not more, PTTH activity than brain extracts from non-diapausing pupae. Similarly, PTTH activity is higher in larvae of a flesh fly Boettcherisca peregrina destined for pupal diapause than in those destined for direct development (Moribayashi et al., 1992). These results indicate that the PTTH is present in the brains of individuals destined for diapause and even during diapause, but it is simply not secreted. Mizoguchi et al. (2013) clarified in the cabbage army moth, that a low PTTH titer in diapause pupae was promoted by the inhibition of PTTH secretion, not by PTTH production. Together with these results, we predict that PTTH cells of S. similis males will easily be silenced and the release of PTTH from neurosecretory cells will be more easily inhibited, compared with females, even under the same short photoperiod. It is of great interest to investigate the sexual difference in the neurophysiological states of PTTH cells, such as voltage threshold, input resistance, and the number of spontaneous action potentials, under the same photoperiodic conditions.
The latitudinal difference in critical day lengths involve qualitative variations in the photoperiodic time measurement system itself, whereas the sexual difference is possibly based on the quantitative photoperiodic time measurement and counter systems (Fig. 7). Thus, S. similis uses distinct mechanisms to induce variations in the critical day length for seasonal events. However, we still do not know why flies adopt such distinct mechanisms for intraspecific variations in the critical day length for the photoperiodic induction of diapause. The circadian system is crucial for adapting to daily environmental cycles, and thus, governs a range of behaviors and physiological systems, such as locomotor activity, eclosion, sleep, phototaxis, learning, memory, feeding, metabolism, chemosensation, courtship, mating, and immunity (Allada and Chung, 2010). Changing properties of the circadian system would cause an alteration to these rhythms. The mechanism inducing the sexual difference found in the flesh fly can alter the critical day length for the photoperiodic induction of diapause, without changing the circadian properties of many output processes, and thus this might be an easier or less costly option for adjusting critical day length (Meuti and Denlinger, 2013). A latitudinal cline of the circadian clock gene alleles has been reported in various insect species (Paolucci et al., 2016; Pruisscher et al., 2018; Tauber et al., 2007; see also Hut et al., 2013), suggesting insects need to adjust their circadian properties along with latitude. The mechanism inducing the latitudinal cline of critical day length might simply be dependent on the latitudinal cline of the properties of the circadian system.
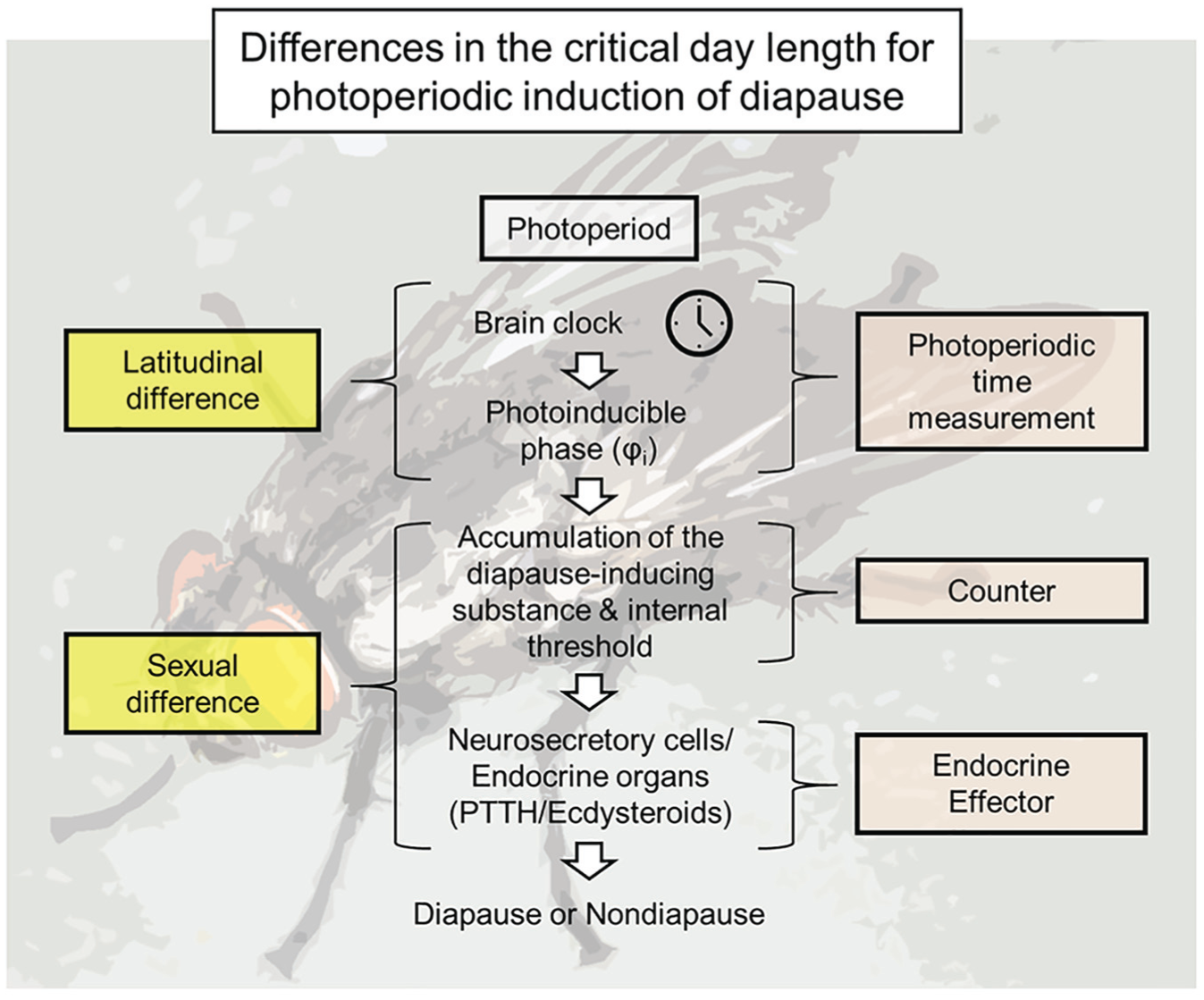
Modules inducing latitudinal and sexual differences in the photoperiodic induction of diapause in Sarcophaga similis. The latitudinal difference in the critical day length involves qualitative variations in the photoperiodic time measurement system itself, whereas sexual difference is based on the quantitative photoperiodic time measurement and counter systems.
Footnotes
Acknowledgements
This study is supported in part by JSPS KAKENHI (Grant-in-Aid for Scientific Research) Grant Number 16K08101 and the Osaka City University (OCU) Strategic Research Grant 2016 and 2017 for Basic Researches to SGG. We thank Editage (![]() ) for English language editing of the manuscript submitted for publication.
) for English language editing of the manuscript submitted for publication.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
