Abstract
Plants and animals use circadian and photoperiodic timekeeping mechanisms to respond to daily and seasonal changes in light:dark and appropriately coordinate their development. Although the mechanisms that may connect the circadian and photoperiodic clock are still unclear in many species, researchers have been using Nanda-Hamner protocols for decades to elucidate how seasonal time is measured and determine whether seasonal responses have a circadian basis in a given species. In this brief tutorial we describe how to design and interpret the results of Nanda-Hamner experiments, and provide suggestions on how to use both Nanda-Hamner protocols and modern molecular experiments to better understand the mechanisms of seasonal timekeeping.
Keywords
Photoperiodic responses are ubiquitous in biology and regulate the seasonal timing of numerous traits, including flowering time in plants, overwintering diapause in insects, and breeding cycles for some vertebrates. Photoperiodic timekeeping requires precise measurements of day and/or night length, and several models have been proposed to explain how these measurements are accomplished (reviewed by Vaz Nunes and Saunders, 1999). In the hourglass model, organisms measure either the length of day or night through the accumulation of a hypothetical substance that is synthesized in direct response to either the presence or absence of light (Lees, 1973). As an alternative to the hourglass model, the circadian clock might measure day or night length to generate photoperiodic responses. This hypothesis, first proposed by Bünning (1936), has served as the primary framework for investigating the mechanistic basis of photoperiodism (Saunders, 2021). Within this framework, two qualitative models have been developed: (1) In the external coincidence model, the circadian clock sets a photo-inducible phase or time period, and the day length determines whether the photo-inducible factor is exposed to an appropriate light signal to elicit a response (Pittendrigh, 1966); (2) In the internal coincidence model, light entrains two independent oscillators, which separately track dawn and dusk. Depending on day length, these two oscillators will have different phase relationships with each other, allowing measurement of photoperiod (Pittendrigh, 1972).
The Nanda-Hamner protocol is a classic approach for determining whether photoperiodic responses involve a circadian timekeeping mechanism (Nanda and Hamner, 1958). In these experiments, separate groups of organisms are held at a constant day length (usually at or around the critical day length for triggering a photoperiodic response), and the night length systematically varies (Figure 1a). The total cycle length (T) usually varies from 24 to 72 h in a given Nanda-Hamner experiment. Individuals are kept at these non-standard photoperiods for their entire life cycle or during the photosensitive period when they are actively measuring day/night length and making the decision to initiate a photoperiodic response. After being held at these conditions, the proportion that undergoes short- versus long-day responses at each photoperiod is assessed. If the photoperiodic response is regulated by a circadian clock, short-day responses (or long-day, depending on the phenotype being assessed) should occur when the total cycle length is a multiple of 24 h. For example, for a short-day response that is normally triggered by 12:12 L:D, peaks in short-day phenotypes around 12:36 L:D and 12:60 L:D would be expected, while long-day responses would be expected at 12:24 L:D and 12:48 L:D. In contrast, under an hourglass model, a short-day photoperiodic response should occur regardless of the total cycle length, as the organism would be simply measuring day or night length independent of the total length of the photoperiodic cycle. However, in some cases, interpretation is not straightforward, and a result consistent with an hourglass timer could also be attributed to a damped external coincidence model (see below).
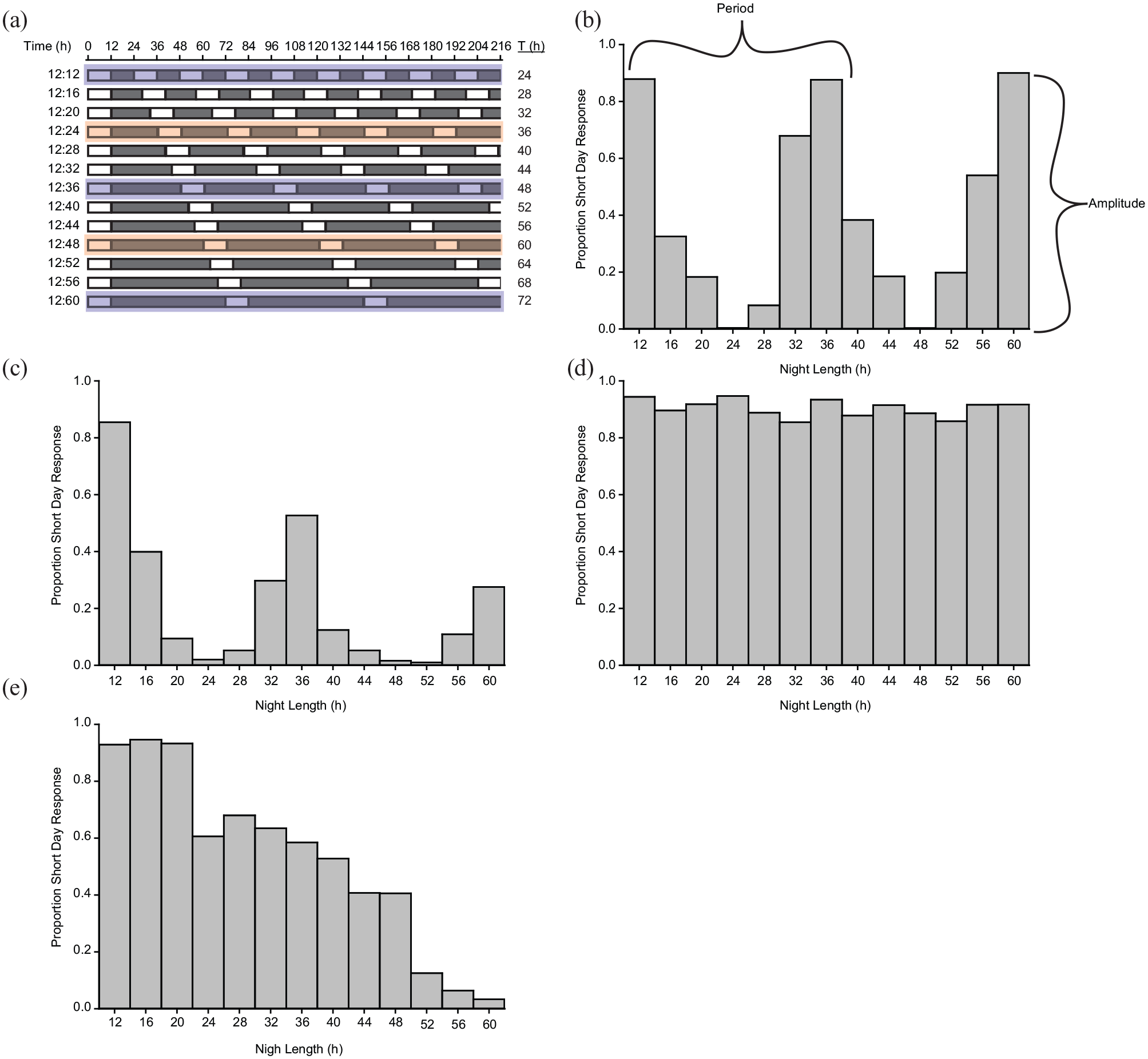
Tutorial on how to set up and interpret a Nanda-Hamner experiment. (a) Set up: organisms are exposed to a short day followed by night lengths that vary, creating photoperiod (T) that ranges from 24 to 72 h. (b-e) Hypothetical results of a Nanda-Hamner experiment. The periodicity of a Nanda-Hamner response is the peak-to-peak interval, while the amplitude is the size of the response. (b) Positive Nanda-Hamner response with consistent amplitude, suggesting an external coincidence model of time measurement. (c) Positive Nanda-Hamner response with decreasing amplitude over long photoperiods, suggesting a damped external coincidence model of time measurement. (d) Negative Nanda-Hamner response, suggesting an hourglass model of photoperiodic time measurement. (e) Negative Nanda-Hamner response with decreasing amplitude, possibly consistent with an hourglass model of photoperiodic time measurement or a highly damped external coincidence model.
Here, we present a brief tutorial for designing and interpreting Nanda-Hamner experiments. These elegant designs have been used for decades to study photoperiodic processes, but conceptualizing these experiments can be difficult for non-specialists (including the authors of this tutorial!). Thus, our goal is to lay out general considerations for a Nanda-Hamner experiment, and to provide guidance for how results can be interpreted under common photoperiodic timekeeping models (e.g., hourglass timer, internal coincidence, external coincidence, damped oscillators). We will also provide thoughts on how to reconcile these classic protocols with recent molecular studies detailing the molecular regulation of photoperiodic responses.
Designing a Nanda-Hamner Experiment
As discussed above, the general principle of a Nanda-Hamner experiment is to provide a constant day length and vary the night length. Most experiments start with a 24-h photoperiod that induces the trait of interest. Thus, a successful Nanda-Hamner experiment requires prerequisite knowledge of the critical day length for a trait of interest. After establishing the critical day length, the night length is systematically lengthened to generate total cycle lengths longer than 24 h. While the number of photoperiods tested varies from study to study, at minimum, the total cycle length should be extended to at least 48 h so that a cycle length that is a multiple of 24 h can be included. Typically, Nanda-Hamner experiments will include cycle lengths from 24 to 72 h in 4 to 6 h increments (Figure 1a). Including a higher number of intermediate photoperiods increases the resolution to detect positive and negative Nanda-Hamner responses. While a classic Nanda-Hamner protocol typically only varies night length, in some instances, the addition of photoperiods <24 h, or photoperiods with varying day lengths, can help distinguish between hourglass, external coincidence, and internal coincidence models.
There are also some practical considerations to account for when designing a Nanda-Hamner experiment. For many photoperiodic responses, pulses of light during the scotophase are disruptive, so precise light/dark control is essential (although in some cases, periodic pulses of light at night are used in conjunction with Nanda-Hamner protocols; for example, Bünsow experiments; Bunsow, 1960). Also, many photoperiodic responses are influenced by temperature, so it is important to have consistent temperature regimes across light conditions, to avoid artifacts. Finally, some processes (such as growth and reproduction) are reliant on circadian rhythms, independent of photoperiod, such that the non-standard photoperiods used in Nanda-Hamner protocols have the potential to disrupt physiology and cause misleading results. For example, in some insect species, release of molting hormones and/or sex pheromones are controlled by a circadian gate (Haynes and Birch, 1984; Zhukovskaya, 1995; Myers, 2003; Rymer et al., 2007), so disruption of these core processes by non-standard photoperiods could indirectly disrupt photoperiodic responses and lead to incorrect conclusions. However, these potential artifacts have not been widely considered in designing or interpreting the results of Nanda-Hamner experiments.
Interpreting a Nanda-Hamner Experiment
Given the inherent complexity of Nanda-Hamner experiments, a range of outcomes can be obtained. Thus, in some instances, a straightforward interpretation may not be possible. Below, we provide a summary of expected results for a Nanda-Hamner experiment under common models of photoperiodic timekeeping mechanisms.
Circadian model of photoperiodic timekeeping. Under the circadian model of photoperiodic timekeeping, inherent circadian oscillators are co-opted to measure day (or night) length. While the circadian clock is entrained by light, it has a free-running rhythm of ~24 h, so the timing of light and darkness in conjunction with the circadian oscillator can be used as a photoperiodic timekeeping mechanism. As introduced above, two competing models exist for circadian-based photoperiodic timekeepers: the external coincidence model, in which timekeeping is achieved by a single light-sensitive oscillator, and the internal coincidence model, where there are two oscillators whose phase relative to each other depends on the length of day and night. In a Nanda-Hamner experiment, if a photoperiodic response shows approximately 24 h periodicity (i.e., a positive Nanda-Hamner response), this result is consistent with a circadian-based photoperiodic timekeepers (Figure 1b). This result is most often interpreted as being consistent with an external coincidence model, because peaks at 24-h intervals suggest that light must be present in-phase with the free-running circadian oscillator, which is presumed to maintain a ~24 h periodicity regardless of external photoperiod. Under a qualitative version of the external coincidence model, a maximum photoperiodic response is expected at each photoperiod that is a multiple of 24 h, but in practice, a reduced amplitude is often observed as cycle length increases, which is consistent with a damped external coincidence model (Figure 1c). In some cases, slight modifications of Nanda-Hamner protocols that vary day length in addition to night length can be used to test for an internal coincidence mechanism (Saunders, 1974), but these designs are beyond the scope of this tutorial.
Hourglass model of photoperiodic timekeeping. The hourglass model indicates that photoperiod is measured by a factor that either accumulates during day or night, and when this factor reaches a certain threshold, it triggers the appropriate photoperiodic response. Under this model, one would expect a “negative” Nanda-Hamner result, meaning that there would be no apparent periodicity in the photoperiodic response (Figure 1d). In the case of short-day responses, which are typically measuring night length, the scotophase is greater than the critical night length for each total photoperiodic cycle so the short-day response is observed in all photoperiods. In some instances, the amplitude of the photoperiodic response decreases as night length increases (Figure 1e), presumably because the organism experiences fewer day/night cycles. In this case, while the results are qualitatively consistent with an hourglass model, they can also be interpreted under a highly damped external coincidence model where extremely long night lengths or continuous darkness reduces the proportion of short-day phenotypes (Vaze and Helfrich-Forster, 2016). Thus, a combination of Nanda-Hamner photoperiods and treatments with continuous darkness would be needed to tease apart whether an organism uses an hourglass timer or a damped external coincidence mechanism to measure day/night length.
Reconciling Nanda-Hamner Protocols With Modern Molecular Approaches
While Nanda-Hamner protocols have provided key insights on the role of the circadian system in photoperiodic time, the specific clock components, hormones, and other factors that trigger photoperiodic responses are unclear. Indeed, Nanda-Hamner and other classic designs have generally not been well-integrated into our understanding of the molecular clock and the hormonal underpinnings of photoperiodism (although see Saunders, 2020). In some cases, there can be discrepancies between Nanda-Hamner experiments and subsequent molecular analyses, so a combination of approaches may be needed to reconcile these inconsistencies. For example, Nanda-Hamner protocols revealed a circadian basis for reproductive diapause in the fruit fly Drosophila melanogaster (Saunders, 1990), but at the same time, flies with a mutation in the circadian clock gene period that render them arrhythmic are still capable of entering diapause (Saunders et al., 1989), suggesting that the molecular machinery underpinning rhythmic Nanda-Hamner responses is distinct from the clock mechanisms that regulate diurnal activity rhythms. Similarly, Bradshaw et al. (2006, 2012) demonstrate that in the pitcher plant mosquito, Wyeomia smithii, photoperiodic responses (e.g., critical photoperiod) and circadian rhythms (e.g., the amplitude and/or periodicity of Nanda-Hamner responses) have distinct genetic architectures, again suggesting distinct mechanisms for circadian and photoperiodic timekeeping mechanisms. However, in other systems, manipulation of clock gene expression via RNAi indicates a direct link between circadian clock genes and photoperiodic time keeping mechanisms (Ikeno et al., 2010; Ikeno et al., 2011; Meuti et al., 2015; Chang and Meuti, 2020) indicating that the relationship between circadian and photoperiodic timekeepers may be species-specific (and/or dependent on methodology). Thus, moving forward, we recommend using a combination of classical photoperiodic manipulations, molecular genetics, and neuroanatomical approaches across distinct systems to tease apart the extent to which circadian and photoperiodic time measurement co-opt similar mechanisms, and how these relationships influence the evolution of photoperiodic responses.
Footnotes
Acknowledgements
We thank Dr. Mary Harrington, editor of Journal of Biological Rhythms, for inviting us to prepare this tutorial. We also thank an anonymous reviewer for pointing out an important reference for the paper. This work is supported by Hatch Project 1010996 from the USDA National Institute of Food and Agriculture and NSF Grants OIA-1826689 and OPP-1850988 to N.M.T. and NSF Grant IOS-1944324 to M.E.M.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
